Published online Aug 14, 2014. doi: 10.3748/wjg.v20.i30.10577
Revised: April 6, 2014
Accepted: April 30, 2014
Published online: August 14, 2014
Processing time: 227 Days and 1.9 Hours
AIM: To evaluate the benefit of aerobic exercise on colonic transit time (CTT) for psychiatric inpatients in a closed ward.
METHODS: Sixty consecutive adult inpatients of the Somang Hospital Psychiatry Unit (Eumsung-gun, South Korea), without CTT-related diseases or drug therapies, were recruited for study from March to June of 2012. Upon enrollment, the patients were randomly assigned to partake in a 12-wk instructor-led group aerobic exercise program (exercise group; n = 30) or to maintain their ordinary daily activities (control group; n = 30). The exercise program was structured as 10 min warm-up (stretching), 40 min exercise, and 10 min cool-down (stretching) for three days each week. The exercise sessions consisted of walking only in week one and aerobics from weeks two to 12, with increasing intensity (50% heart rate reserve (HRR) for weeks one to four, 60% HRR for weeks five to eight, and 70% HRR for weeks nine to 12). CTT was measured before (baseline) and after (week 12) the exercise program, in duplicate (on days four and seven), using abdominal radiography and the multiple radio-opaque marker technique. Changes in the exercising patients’ CTT and weight-, cardiovascular- and fitness-related parameters were statistically assessed.
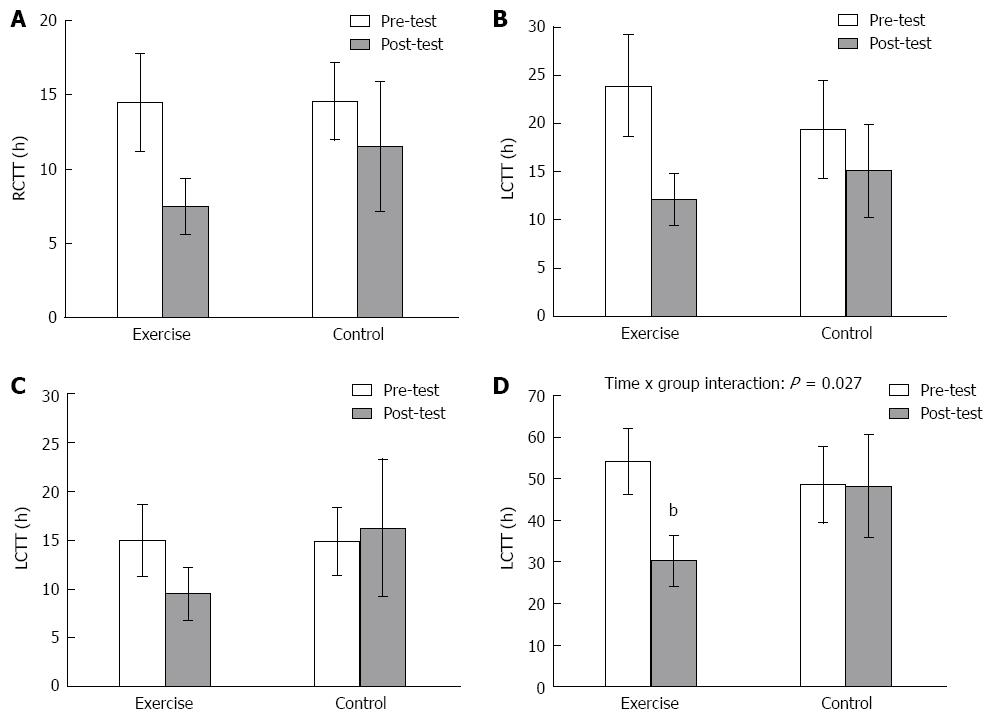
RESULTS: The study dropout rate was 30.0%, with 23 patients in the exercise group and 19 patients in the control group completing the study. At week 12, the exercise group showed decreases in body weight (mean ± SE) baseline: 69.4 ± 2.8 vs study-end: 67.6 ± 2.7; P < 0.635) and body mass index (BMI) (25.2 ± 1.1 vs 24.9 ± 0.8; P < 0.810), but the extent of change was not significantly different from that experienced by the control group (body weight: 68.8 ± 4.0 vs 68.8 ± 3.9; BMI: 24.3 ± 1.1 vs 24.4 ± 1.2). However, the exercise group showed significant improvements in leg muscle strength (baseline: 41.7 ± 4.3 vs study-end: 64.1 ± 5.0; P < 0.001), cardio-respiratory endurance (120.5 ± 4.5 vs 105.4 ± 2.8; P < 0.004), and leg muscle elasticity and power output (21.5 ± 2.6 vs 30.6 ± 2.8; P < 0.001). The exercise group showed an exercise-induced reduction in total CTT (baseline: 54.2 ± 8.0 vs 30.3 ± 6.1), which was significantly different from that experienced by the control group over the 12-wk period (48.6 ± 9.3 vs 48.3 ± 12.3; P = 0.027); however, the exercise-induced decreases in CTT involving the three colonic segments examined (right, left and recto-sigmoid) showed no significant differences from the control group.
CONCLUSION: A 12-wk aerobic exercise program can benefit psychiatric inpatients by increasing intestinal motility, possibly decreasing risk of metabolic- and cardiovascular-related disease.
Core tip: Maintaining a routine of physical activity for inpatients of closed wards in mental health facilities remains a major challenge. Long-term inactivity is a risk factor for decreased gastrointestinal motility, which leads to constipation and can affect drug absorption, as well as weight gain and related metabolic and cardiovascular disorders. In this study, implementation of a 12-wk aerobic exercise program was shown to be beneficial in reducing colonic transit time, decreasing weight and improving blood pressure.
- Citation: Kim YS, Song BK, Oh JS, Woo SS. Aerobic exercise improves gastrointestinal motility in psychiatric inpatients. World J Gastroenterol 2014; 20(30): 10577-10584
- URL: https://www.wjgnet.com/1007-9327/full/v20/i30/10577.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i30.10577
The gastrointestinal (GI) tract plays a key role in gut and systemic health, affecting distant and key organs through its functional relationship with the systemic blood supply. As such, disruption of GI motility can have a broad range of consequences in the GI system itself and beyond. Decreased GI motility reduces the movement of materials, with stagnating masses triggering local chronic inflammatory responses that may affect both the structural and functional integrity of the surrounding tissue. In addition, low GI motility is known to affect the efficiency of absorption of nutrients and therapeutic drugs[1,2].
One of the most common signs of low GI motility is constipation, which may cause discomfort and pain associated with increased gas, abdominal bloating and physical difficulties in passing stools. While the disorder-disease association is still under debate, chronic constipation itself remains a widespread problem. In particular, it has been estimated that up to 28% of the general population in the United States suffers from constipation[3], and it has been demonstrated that the Western lifestyle (low activity, low fiber diet) is related to constipation in populations of East Asian countries as well[4].
Clinical symptoms of constipation include excessive contraction of the anal sphincter, hard feces, anal atresia and a decrease in frequency of defecation or size of stools[5]. An underlying disorder in colonic motility can be determined by measurement of colonic transit time (CTT), by which material passing through the colon is tracked by real-time imaging of ingested radio-opaque markers[4-7]. The technical ease (relatively non-invasive nature), low cost and high rates of reproducibility of this technique have promoted its use as a screening and treatment monitoring method in various patient populations, including those classified as difficult-to-treat (e.g., children and the mentally ill)[5,7-9].
An important cause of low GI motility, and one of the easiest to correct, is physical inactivity. Indeed, some studies have shown that adding a routine of aerobic exercise can help to resolve constipation symptoms[10]. Research to determine the underlying mechanisms of the relationship between exercise and constipation has demonstrated possible roles for increasing peristaltic movement and shortening of the CTT[6,11,12]. Moreover, several epidemiologic studies have shown that decreased physical activity level correlates with increased prevalence of constipation[13].
Certain living conditions foster an inactive (sedentary) lifestyle, such as residence in a closed ward of a hospital. Attaining and sustaining an adequate level of physical activity is a particular challenge facing clinical care teams working with mentally ill patients residing in closed wards. The daily routine of limited physical activity (with planned activities being largely sedentary, such as watching television, or low-impact, such as participating in a sing-along) may increase the risk of further mental illness as well as various chronic physical conditions[14,15], such as low GI motility, and related diseases, such as colorectal cancer[16,17], obesity[18] and diabetes[19]. Indeed, studies have shown that increasing mentally ill patients’ physical activity level may help to prevent cancer among this patient population[20,21].
As a measure of gastric motility, CTT has been shown to be correlated with physical activity level, with more sedentary individuals showing longer CTT than their physically active counterparts[4,22]. Moreover, partaking in the active practices of walking, running, cycling and other aerobic activities has been shown to significantly shorten CTT in healthy individuals[5,23,24]. The previous studies that have investigated the effects of increasing physical activity by participation in an exercise program and changes in CTT have largely involved healthy subjects practicing a normal lifestyle. However, no studies reported in the publicly available literature to date have reported on the effects of aerobic exercise on colonic function in mentally ill patients residing in a closed hospital ward. The current study was designed to evaluate the effects of a long-term (12-wk) aerobic exercise program on CTT to determine the potential benefits to gastric motility, as well as on the weight-, fitness and cardiovascular-related parameters that may affect the overall health of mentally ill patients residing in a closed ward.
Consecutive male inpatients with mental illness admitted for treatment in the closed ward of Somang Hospital Psychiatry Unit (Eumsung-gun, South Korea) over a 4-mo period (March to June of 2012) were recruited to the study. Patients were offered enrollment if they had not participated in any structured exercise program in the past 12 mo and had no physical limitations impeding normal activity. Patients were denied enrollment according to diagnosis of cardiovascular or orthopedic diseases with potential effects on CTT, diagnosis of functional stomach diseases (untreated or under current clinical management), undergoing treatment with anti-constipation drugs and diagnosis with diabetes mellitus or hypertension[4,7,9]. In addition, any patients with indications of irritable bowel syndrome, according to the Rome II criteria[25], or who were taking any drugs to address diarrhea symptoms were excluded from the study.
A total of 60 male patients were enrolled and randomly assigned to participate in a 12-wk instructor-led group aerobic exercise program (exercise group; n = 30) or to maintain their ordinary daily activities (control group; n = 30). During the study period, seven patients from the exercise group were withdrawn due to change in mental illness that inhibited study participation, non-compliance with the exercise program and changes in therapeutic drugs or disease status that impacted the inclusion/exclusion criteria. In the control group, 11 subjects were withdrawn due to refusal to participate in study procedures, cancellation of consent and various personal reasons. The final groups were composed of 23 patients (age: 46.4 ± 2.1 years) in the exercise group and 19 patients (age: 50.8 ± 2.6 years) in the control group. The included patients’ psychiatric diagnoses, according to that determined by the Structured Clinical Interview for Diagnostic and Statistical Manual of Mental Disorders 4th revision (DSM-IV), are listed in Table 1.
The study was designed in accordance with the Declaration of Helsinki (2000; World Medical Association) and carried out with pre-approval from the Institutional Review Board of Seoul National University (IRB No: 1203/001-002). All patients provided written informed consent for study participation and subsequent analysis and publication of all study-related data.
Each of the participants in the exercise group participated in an instructor-led aerobic exercise program that consisted of 60 min sessions, three days a week for 12 wk. Each of the 60-min sessions included 10 min of stretching (warm-up), 40 min of aerobic activity of varying impact (exercise), and 10 min of stretching (cool-down). The aerobic activities started out as walking exercise in the first study week, to help the inactive patients adapt to the exercise routine. Starting in study week two, the aerobic exercise activities were modified to provide increasing impact, using a combination of walking, running and other types of aerobic movements to achieve 50% heart rate reserve (HRR) between weeks one and four, 60% HRR between weeks five and eight, and 70% HRR between weeks nine and 12 (according to the recommendations of the American College of Sports Medicine (ACMS). For each day’s exercise session, the patients wore a RS300X heart rate monitor (Polar Electro, Kempele, Finland) in order for investigators to ensure that the target heart rate was maintained during the aerobic exercise period.
Each of the participants in the control group was instructed to maintain their ordinary daily activities. No restrictions were placed on participation in the normal planned group activities for the ward, such as watching television or partaking in a sing-along.
The InBody 3.0 bioelectrical impedance analysis system (Biospace Co., Seoul, South Korea) was used to measure each patient’s height, weight and body mass index (BMI). The Spirit CK-101 sphygmomanometer (Spirit Medical Co. Ltd., New Tapai City, Taiwan) was used to measure blood pressure with the patient in the prone position after a 5 min rest. In an attempt to rule out any extrinsic factors that might affect blood pressure, the following patient data was collected for consideration during the analysis: body temperature, general degree of physical activity, smoking practice and diet[4,9]. Thigh circumference was measured as the distance around the fullest part of the thigh. Waist circumference was measured as the distance around the abdomen, just above the hip bones.
Each patient’s physical fitness was quantitatively assessed at study enrollment (first day of week one; baseline) and again at the end of the study period (last day of week 12; study-end). Electronic-based measures were made for handgrip strength [Takei Kiki Kogyo (TKK) 5401 grip dynamometer; Takei Scientific Instruments Co. Ltd., Tokyo, Japan], leg strength (TKK 5710 dynamometer), and leg muscle elastic strength and power output (TKK 5406 vertical jump/Sergeant jump measuring instrument). Cardio-respiratory endurance was measured using the YMCA bench step test, which was carried out with patients taking 24 steps per minute for 3 min using a 30.5 cm-high step box and accompanied by a metronome that was set to 96 beats per minute (bpm); the heart rate was measured during the rest period. Reach was measured using a trunk flexion meter (TKK 5404 instrument).
CTT measurement was performed twice at baseline and twice at study-end using abdominal radiography and the multiple radio-opaque marker technique. Each of the patients ingested a single gelatin capsule containing 20 radio-opaque markers at the same time every day for three consecutive days (Kolomark™, MI Tech Co. Ltd., Pyeongtaik, South Korea). Then on the fourth day (study days four and seven for baseline and study-end), a supine abdominal radiography was performed. The mean CTT (h) was calculated by counting the number of radio-opaque markers that remained in the colon (total colon and the three colonic segments: right, left and recto-sigmoid) and multiplying this value by 1.2[4-6,8,23].
Data were expressed as mean ± SE. All statistical analyses were carried out using SPSS for Windows, version 18.0 (SPSS Inc., Chicago, IL, United States). The significance of changes in fitness and segmental CTT following aerobic exercise was assessed by two-way repeated ANOVA. A P value of less than 0.050 was considered statistically significant.
The physical characteristics of the subjects in the exercise and control groups are shown in Table 2. At study-end, the patients in the exercise group showed decreases in the weight parameters of body weight (baseline: 69.4 ± 2.8 vs study end: 67.6 ± 2.7), BMI (25.2 ± 1.1 vs 24.9 ± 0.8), body fat percentage (30.8 ± 2.4 vs 27.9 ± 2.0) and waist circumference (88.3 ± 2.9 vs 85.5 ± 2.4), as well as in the cardiovascular parameters of systolic blood pressure (SBP) (116.5 ± 3.9 vs 113.5 ± 3.9), diastolic blood pressure (DBP) (77.8 ± 2.5 vs 71.9 ± 2.1) and resting heart rate (86.4 ± 3.8 vs 79.8 ± 2.8); however, the patients showed increases in lean body mass (46.8 ± 1.5 vs 48.7 ± 1.4) and thigh circumference (41.4 ± 1.4 vs 49.6 ± 2.9). The control group showed similar trends for changes in each of these parameters, and only the exercise-induced decrease in SBP was significantly different from the decrease seen over the 12-wk period for the control group (P = 0.016).
| Parameter | Exercise group (n = 23) | Control group (n = 19) | P value1 | ||
| Baseline | Study-end | Baseline | Study-end | ||
| Body weight, kg | 69.4 ± 2.8 | 67.6 ± 2.7 | 68.8 ± 4.0 | 68.8 ± 3.9 | 0.706 |
| BMI, kg/m2 | 25.2 ± 1.1 | 24.9 ± 0.8 | 24.3 ± 1.1 | 24.4 ± 1.2 | 0.759 |
| Lean body mass, kg | 46.8 ± 1.5 | 48.7 ± 1.4 | 44.7 ± 1.4 | 46.8 ± 2.0 | 0.909 |
| Body fat, % | 30.8 ± 2.4 | 27.9 ± 2.0 | 33.3 ± 2.8 | 30.8 ± 2.5 | 0.830 |
| SBP, mmHg | 116.5 ± 3.9 | 113.5 ± 3.9 | 126.0 ± 4.7 | 110.8 ± 5.4 | 0.016a |
| DBP, mmHg | 77.8 ± 2.5 | 71.9 ± 2.1 | 87.1 ± 4.2 | 76.9 ± 3.6 | 0.251 |
| Resting heart rate, bpm | 86.4 ± 3.8 | 79.8 ± 2.8 | 87.4 ± 2.8 | 80.5 ± 2.3 | 0.894 |
| Thigh circumference, cm | 41.4 ± 1.4 | 49.6 ± 2.9 | 44.0 ± 1.6 | 46.3 ± 2.3 | 0.074 |
| Waist circumference, cm | 88.3 ± 2.9 | 85.5 ± 2.4 | 91.8 ± 3.5 | 90.5 ± 4.0 | 0.675 |
The changes in variables of physical fitness for the subjects in the exercise and control groups are shown in Table 3. The patients in the exercise group showed significant exercise-induced improvements in leg muscle strength (baseline: 41.7 ± 4.3 vs study-end: 64.1 ± 5.0), cardio-respiratory endurance (120.5 ± 4.5 vs 105.4 ± 2.8) and leg muscle elasticity and power output (21.5 ± 2.6 vs 30.6 ± 2.8). The control group also showed an increasing trend over the 12-wk study period in leg muscle strength (42.5 ± 4.3 vs 43.8 ± 1.5), cardio-respiratory endurance (120.3 ± 4.7 vs 119.6 ± 3.3) and leg muscle elasticity and power output (21.5 ± 3.3 vs 24.3 ± 3.2), but the extent of improvement was significantly better in the experimental group for all three of the parameters (leg muscle strength, P = 0.005; cardio-respiratory endurance, P = 0.048; leg muscle elasticity and power output, P = 0.015). In contrast, the improvements in grip strength and flexibility experienced by the exercise group (grip strength, baseline: 26.3 ± 2.3 vs study-end: 27.7 ± 2.2; flexibility, 6.5 ± 2.3 vs 8.2 ± 2.1) were not significantly different from those experienced by the control group (grip strength, baseline: 29.4 ± 3.0 vs study-end: 29.4 ± 2.2; flexibility, 2.9 ± 2.4 vs 5.4 ± 2.3) (exercise vs control, P = 0.343 and P = 0.679 respectively).
| Parameter | Exercise group (n = 23) | Control group (n = 19) | P value1 | ||
| Baseline | Study-end | Baseline | Study-end | ||
| Grip strength, kg | 26.3 ± 2.3 | 27.7 ± 2.2 | 29.4 ± 3.0 | 29.4 ± 2.2 | 0.343 |
| Leg strength, kg | 41.7 ± 4.3 | 64.1 ± 5.0 | 42.5 ± 4.3 | 43.8 ± 1.5 | 0.005a |
| YMCA bench step test, BPM | 120.5 ± 4.5 | 105.4 ± 2.8 | 120.3 ± 4.7 | 119.6 ± 3.3 | 0.048a |
| Jump, cm | 21.5 ± 2.6 | 30.6 ± 2.8 | 21.5 ± 3.3 | 24.3 ± 3.2 | 0.015a |
| Reach, cm | 6.5 ± 2.3 | 8.2 ± 2.1 | 2.9 ± 2.4 | 5.4 ± 2.3 | 0.679 |
The changes in CTT, in the entire colon [total CTT (TCTT)] and in the three different colonic segments, for the subjects in the exercise and control groups are shown in Figure 1. The exercise group showed reductions in CCTs of each of the colonic segments over the study period (right, baseline: 14.5 ± 3.3 vs study-end: 7.5 ± 1.9; left, 23.9 ± 5.3 vs 12.1 ± 2.7; recto-sigmoid, 15.0 ± 3.7 vs 9.5 ± 2.7) but the differences were not significant from those seen in the control group (right, baseline: 14.6 ± 2.6 vs study-end: 11.5 ± 4.5; left, 19.4 ± 5.1 vs 15.1 ± 4.8; recto-sigmoid, 14.9 ± 3.5 vs 16.2 ± 7.1). Only the reduction in the total CTT over the study period was significantly better for the exercise group (baseline: 54.2 ± 8.0 vs study-end: 30.3 ± 6.1) when compared to the control group (48.6 ± 9.3 vs 48.3 ± 12.3) (P = 0.027).
This study showed that participation in a 12-wk aerobic exercise program benefited inpatients of a psychiatric closed ward by improving CTT. This finding concurs with the results from one of the earliest studies of CTT measurement using abdominal radiography and the radio-opaque marker technique in normal (non-hospitalized) healthy men[26]. However, the mean CTTs reported in subsequent studies of Asian males were relatively shorter[27-29]. The longer CTTs found for our current study cohort may simply reflect the difference of the population studied, namely their mentally ill status and current residence in a closed ward with limited practice of physical activity on a daily basis. We showed that patients who increased their normal physical activity by participating in an aerobic exercise program experienced a corresponding reduction in CTT. Although the current study was not specifically designed to allow for a direct comparison between normal (non-mentally ill, healthy, non-hospitalized) Asian populations and our study cohort, many potential factors may be theorized as contributing to the difference in CTT among these two populations, including activities related to job functions, eating habits and diet, and social and lifestyle patterns, as well as hereditary components.
Several biological and behavioral parameters have been characterized in previous studies as influencing factors of CTT, including age, sex, BMI, dietary fiber intake, water intake and various lifestyle habits[5,29]. However, the precise interplay of these factors and underlying molecular mechanisms leading to changes in CTT remain largely unknown. In addition, studies on the effect of exercise on CTT have provided inconsistent results, with some studies showing no effect of increased physical activity or exercise on CTT[5]. Similarly, when the effect of increasing physical activity level on CTT was evaluated with respect to the various colon segments, in addition to the total colonic structure, the results suggested no benefit of exercise for managing chronic constipation[30]. In the current study, the patients experienced significant improvements in lower extremity muscle strength, endurance and leaping ability as a result of participation in an aerobic exercise program, but the CTT of the three segmental colon structures was not changed; although, the TCTT did appear to decrease significantly in these patients during the same period. The relationship between exercise and CTT remains to be absolutely established, even for the study cohort of mentally ill Asian patients in a closed ward. The study cohort (comprised of mentally ill individuals) is inherently more difficult to control in an experimental study, which complicates analysis of their data and limits the power of a study to make definitive conclusions regarding etiologies and influencing factors of CTT.
The general benefits of exercise are well known and primarily involve strengthening of physical fitness[31]. Exercise has been characterized as a preventive and therapeutic approach for a wide array of metabolic (diabetes, obesity), cardiovascular (hypertension) and bone (osteoporosis) diseases, which are interrelated from a mechanistic standpoint; in addition, there are benefits to GI organs and GI-related disorders, such as colorectal cancer and various gastrointestinal conditions including constipation[31,32]. Certainly, exercise is not an exclusive etiologic factor for these diseases and their prognosis, and a person’s lifestyle and eating habits are important contributing factors for maintaining health by promoting cardiorespiratory endurance, mechanical integrity of muscle and bone, proper intake of nutrients and avoidance of detrimental substances (such as carcinogens). Furthermore, studies have suggested that an inactive (sedentary) lifestyle increases CTT and risk of constipation[33] and have demonstrated that implementing an active (vs sedentary) lifestyle and healthy eating habits can improve symptoms of obstructed defecation[32,34]. The practice of aerobic exercise, in particular, may help to alleviate constipation by stimulating intestinal muscle contraction as a consequence of increased respiration and heart rate.
Several studies have been performed to identify the direct relationship between exercise and CTT. In a group of individuals with a sedentary lifestyle who partook in a one-week aerobic exercise program (one hour per day, three days a week) no significant difference was found in their CTT measurements recorded during the exercise period and the rest period (one week of no exercise)[30]. Another study of a longer (4-wk) aerobic exercise program also showed no benefit, with the participants’ symptoms of constipation being unimproved over the study period[12]. In the current study, a substantially longer exercise period was implemented (12 wk). Again, there was no significant shortening in CTT of the colonic segments in comparison to the control (non-exercise) group; however, the control group experienced shortening of CTT during the study period, which may have been due to their being placed under a well-regulated lifestyle and provided with a balanced diet during their time of residence in the closed psychiatric ward. The exercise group of the current study did show a significant improvement in TCTT when compared to the control group.
A key difference between the previous related studies that failed to observe an exercise-related difference in CTT and the current study is the duration of the exercise program participation (1-4 wk vs 12 wk); thus, it is possible that participation in an aerobic exercise program of sufficient or sustained duration (≥ 12 wk) may be necessary to facilitate a beneficial change in CTT and improvements in gastric health, possibly lowering risks of subsequent development of related disorders, such as colorectal cancer. Since the current study’s subjects were inpatients residing in a closed ward, many variables with potential influence on CTT (i.e., lifestyle and diet) were controlled, which strengthened the likelihood that the observed reduction in TCTT in the exercise group was a result of the exercise program. However, further studies with large sample sizes are needed to understand the dose-response relationship between exercise type and intensity and changes in CTT and in symptoms of constipation.
In summary, increasing psychiatric inpatient physical activity level through a long-term (12-wk) exercise program can benefit the patients’ overall health by helping to reduce CTT and improving their risk for developing subsequent diseases related to low gastrointestinal motility, which can range from colorectal cancer to life-threatening cardiovascular and metabolic disorders.
A large number of studies have demonstrated that aerobic exercise promotes normal colonic function in generally healthy individuals practicing a normal lifestyle. However, no published studies have investigated the effects of aerobic exercise on colonic function in mentally ill patients residing in a closed ward (physically restricted) setting. The current study was designed to investigate the relationship between aerobic exercise and colon transit time (CTT) in this population to help understand the potential benefit of implementing a long-term (12-wk) aerobic exercise program to promote the general gastrointestinal (GI) health of inpatients.
Low motility in the GI tract, including in the colonic segments, can have expansive effects on a patient’s overall health, as the GI system is functionally interrelated with the metabolic system, the cardiovascular system and the immune system. Decreases in GI motility slow the passage of waste, reduce the absorption of nutrients and therapeutic drugs, and promote localized inflammation. Increasing physical activity through aerobic exercise may help to increase CTT and provide wider benefits to overall health by lowering an individual’s risk of developing subsequent systemic disorders with life-threatening potential. Individuals living under conditions with restricted physical activity, such as in a closed psychiatric ward, may benefit greatly from participation in a structured long-term aerobic exercise program.
Implementation of a structured long-term aerobic exercise program can help to improve CTT, as measured by abdominal radiography and the radio-opaque marker method. A 12-wk program of increasing intensity of aerobic exercise was shown to improve the total CTT of psychiatric inpatients and may be a useful approach to improving the overall health of such patients when combined with structured diet programs promoting better water and fiber intake.
Mentally ill patients admitted to closed hospital wards for extended stays frequently experience limited physical activity during their inpatient residence period. When basic physical fitness is weakened, risk of constipation increases. However, as shown in the current study, as a patient’s physical activity level increases (through participation in a structured aerobic exercise program), the total CTT is shortened. Implementation of a structured long-term (≥ 12-wk) aerobic exercise program for mentally ill patients during their inpatient hospitalization may enhance the overall clinical management approach to care for these patients and help to improve their general health prognosis.
Mental illness is a mental or behavioral pattern or anomaly that causes either suffering or an impaired ability to function in ordinary life. Aerobic exercise is physical exercise of relatively low intensity that depends primarily on the aerobic energy-generating process. Measurement of colon transit time (CTT) is useful for evaluating colonic motility, and allows for assessment of the severity of the motility disruption as well as the response to therapy.
The current study by Kim et al is methodologically sound and well written.
P- Reviewer: Sidor MM S- Editor: Qi Y L- Editor: A E- Editor: Ma S
| 1. | Parkman HP. Scintigraphy for evaluation of patients for GI motility disorders--the referring physician’s perspective. Semin Nucl Med. 2012;42:76-78. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 2. | Sarnelli G, D’Alessandro A, Pesce M, Palumbo I, Cuomo R. Genetic contribution to motility disorders of the upper gastrointestinal tract. World J Gastrointest Pathophysiol. 2013;4:65-73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 12] [Cited by in RCA: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 3. | Locke GR, Pemberton JH, Phillips SF. American Gastroenterological Association Medical Position Statement: guidelines on constipation. Gastroenterology. 2000;119:1761-1766. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 146] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 4. | Song BK, Cho KO, Jo Y, Oh JW, Kim YS. Colon transit time according to physical activity level in adults. J Neurogastroenterol Motil. 2012;18:64-69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 58] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 5. | Metcalf AM, Phillips SF, Zinsmeister AR, MacCarty RL, Beart RW, Wolff BG. Simplified assessment of segmental colonic transit. Gastroenterology. 1987;92:40-47. [PubMed] |
| 6. | Xu HM, Han JG, Na Y, Zhao B, Ma HC, Wang ZJ. Colonic transit time in patient with slow-transit constipation: comparison of radiopaque markers and barium suspension method. Eur J Radiol. 2011;79:211-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 7. | Jung HK, Kim DY, Moon IH. Effects of gender and menstrual cycle on colonic transit time in healthy subjects. Korean J Intern Med. 2003;18:181-186. [PubMed] |
| 8. | Prokesch RW, Breitenseher MJ, Kettenbach J, Herbst F, Maier A, Lechner G, Mahieu P. Assessment of chronic constipation: colon transit time versus defecography. Eur J Radiol. 1999;32:197-203. [PubMed] |
| 9. | Jung HK, Kim DY, Moon IH, Hong YS. Colonic transit time in diabetic patients--comparison with healthy subjects and the effect of autonomic neuropathy. Yonsei Med J. 2003;44:265-272. [PubMed] |
| 10. | De Schryver AM, Keulemans YC, Peters HP, Akkermans LM, Smout AJ, De Vries WR, van Berge-Henegouwen GP. Effects of regular physical activity on defecation pattern in middle-aged patients complaining of chronic constipation. Scand J Gastroenterol. 2005;40:422-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 117] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 11. | Ducrotte P, Rodomanska B, Weber J, Guillard JF, Lerebours E, Hecketsweiler P, Galmiche JP, Colin R, Denis P. Colonic transit time of radiopaque markers and rectoanal manometry in patients complaining of constipation. Dis Colon Rectum. 1986;29:630-634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 36] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 12. | Meshkinpour H, Selod S, Movahedi H, Nami N, James N, Wilson A. Effects of regular exercise in management of chronic idiopathic constipation. Dig Dis Sci. 1998;43:2379-2383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 66] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 13. | Everhart JE, Go VL, Johannes RS, Fitzsimmons SC, Roth HP, White LR. A longitudinal survey of self-reported bowel habits in the United States. Dig Dis Sci. 1989;34:1153-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 243] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 14. | Shariati A, Haghighi S, Fayyazi S, Tabesh H, Kalboland MM. The effect of exercise on the severity of the fatigue in colorectal cancer patients who received chemotherapy in Ahwaz. Iran J Nurs Midwifery Res. 2010;15:145-149. [PubMed] |
| 15. | Aoi W, Naito Y, Takagi T, Kokura S, Mizushima K, Takanami Y, Kawai Y, Tanimura Y, Hung LP, Koyama R. Regular exercise reduces colon tumorigenesis associated with suppression of iNOS. Biochem Biophys Res Commun. 2010;399:14-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 16. | Kisely S, Campbell LA, Cox M. The effect of study design on the reporting of mortality due to colorectal cancer in adults with mental illness in Nova Scotia. Can J Psychiatry. 2012;57:389-394. [PubMed] |
| 17. | Kisely S, Crowe E, Lawrence D. Cancer-related mortality in people with mental illness. JAMA Psychiatry. 2013;70:209-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 263] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 18. | Lin HY, Huang CK, Tai CM, Lin HY, Kao YH, Tsai CC, Hsuan CF, Lee SL, Chi SC, Yen YC. Psychiatric disorders of patients seeking obesity treatment. BMC Psychiatry. 2013;13:1. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 117] [Cited by in RCA: 126] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 19. | Stanley SH, Laugharne JD. Obesity, cardiovascular disease and type 2 diabetes in people with a mental illness: a need for primary health care. Aust J Prim Health. 2012;18:258-264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 20. | Alvarez-Jiménez M, Hetrick SE, González-Blanch C, Gleeson JF, McGorry PD. Non-pharmacological management of antipsychotic-induced weight gain: systematic review and meta-analysis of randomised controlled trials. Br J Psychiatry. 2008;193:101-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 204] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 21. | Richardson CR, Avripas SA, Neal DL, Marcus SM. Increasing lifestyle physical activity in patients with depression or other serious mental illness. J Psychiatr Pract. 2005;11:379-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 22. | Cho KO, Jo YJ, Song BK, Oh JW, Kim YS. Colon transit time according to physical activity and characteristics in South Korean adults. World J Gastroenterol. 2013;19:550-555. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 16] [Cited by in RCA: 19] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 23. | Chaussade S, Gosselin A, Hostein J, Leman M, Ponsot P. [Determination of global and segmental colonic transit time in a population of 96 healthy volunteers]. Gastroenterol Clin Biol. 1990;14:95-96. [PubMed] |
| 24. | Hinds JP, Stoney B, Wald A. Does gender or the menstrual cycle affect colonic transit? Am J Gastroenterol. 1989;84:123-126. [PubMed] |
| 25. | Xin HW, Fang XC, Zhu LM, Xu T, Fei GJ, Wang ZF, Chang M, Wang LY, Sun XH, Ke MY. Diagnosis of functional constipation: agreement between Rome III and Rome II criteria and evaluation for the practicality. J Dig Dis. 2014;15:314-320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 26. | Martelli H, Devroede G, Arhan P, Duguay C, Dornic C, Faverdin C. Some parameters of large bowel motility in normal man. Gastroenterology. 1978;75:612-618. [PubMed] |
| 27. | Chaussade S, Roche H, Khyari A, Couturier D, Guerre J. [Measurement of colonic transit time: description and validation of a new method]. Gastroenterol Clin Biol. 1986;10:385-389. [PubMed] |
| 28. | Chan YK, Kwan AC, Yuen H, Yeung YW, Lai KC, Wu J, Wong GS, Leung CM, Cheung WC, Wong CK. Normal colon transit time in healthy Chinese adults in Hong Kong. J Gastroenterol Hepatol. 2004;19:1270-1275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 39] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 29. | Meir R, Beglinger C, Dederding JP, Meyer-Wyss B, Fumagalli M, Rowedder A, Turberg J, Brignoli R. [Age- and sex-specific standard values of colonic transit time in healthy subjects]. Schweiz Med Wochenschr. 1992;122:940-943. [PubMed] |
| 30. | Robertson G, Meshkinpour H, Vandenberg K, James N, Cohen A, Wilson A. Effects of exercise on total and segmental colon transit. J Clin Gastroenterol. 1993;16:300-303. [PubMed] |
| 31. | Irwin ML. Physical activity interventions for cancer survivors. Br J Sports Med. 2009;43:32-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 107] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 32. | Stephenson LE, Bebb DG, Reimer RA, Culos-Reed SN. Physical activity and diet behaviour in colorectal cancer patients receiving chemotherapy: associations with quality of life. BMC Gastroenterol. 2009;9:60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 33. | Sandler RS, Drossman DA. Bowel habits in young adults not seeking health care. Dig Dis Sci. 1987;32:841-845. [PubMed] |
| 34. | Papathanasopoulos A, Camilleri M. Dietary fiber supplements: effects in obesity and metabolic syndrome and relationship to gastrointestinal functions. Gastroenterology. 2010;138:65-72.e1-65-72.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 214] [Article Influence: 14.3] [Reference Citation Analysis (0)] |