BRAIN-GUT AXIS AND HELICOBACTER PYLORI
Upper digestive tract diseases, including both functional dyspepsia and dyspeptic symptoms in the course of gastritis or peptic ulcers, were considered for many years to be psychosomatic[1,2]. However, Helicobacter pylori (H. pylori) infection has been recognized as the main risk factor for many upper digestive tract disorders and complications, including life-threatening bleeding from the digestive tract, gastritis, non-ulcer dyspepsia, mucosa-associated lymphoid tissue (MALT) lymphoma and gastric cancer[3,4], and a risk factor for stress-induced peptic ulcers[2,5-8]. As a result, the role of psychosomatic factors in the pathogenesis of these diseases has since lost favor[1,9]. However, the presence of H. pylori-negative upper digestive tract abnormalities, such as peptic ulcers[10,11], and the fact that not all H. pylori-infected patients develop ulcers, with only 10%-15% of them presenting dyspeptic symptoms[3], suggest a role for other individual factors, including nervous system imbalance, as an indispensable cofactor in gastritis or ulcer disease pathogenesis[12]. These arguments imply that H. pylori infection may induce changes in the function and morphology of the digestive tract both directly through cytotoxin (CagA, VacA) release and inflammatory process activation, and indirectly, via the brain-gut axis[13]. This review presents and discusses the interaction between H. pylori infection and the brain-gut axis and the clinical implications for digestive tract function.
The brain-gut axis is anatomically based in the central (CNS), peripheral (PNS, enteric nervous system, or “little brain”), and autonomic nervous systems (ANS), and modulates gastrointestinal function via the regulation of the gastrointestinal immune system, mucosal inflammation and intestinal microbiota in response to stress, emotions and environmental influences[6,14,15]. This circuitry acts bidirectionally, playing a role both in upper (peptic ulcer, functional dyspepsia) and lower [irritable bowel syndrome (IBS), inflammatory bowel disease (IBD)] digestive tract homeostasis, appetite and weight control[6,15-17], modulation of the gut-associated immune system and in the coordination of the gastrointestinal tract with the overall physical and emotional state of the organism[18]. However, so many interactions and functions work not only via the nervous system, but also through the endocrine, immunological and metabolic pathways (neuroendocrine-immune crosstalk)[6,15-22]. The main endocrine role is played by the hypothalamus-pituitary-adrenal (HPA) axis, along with the highly significant neuroendocrine-immune response modulator corticotropin releasing factor (CRF). CRF regulates inflammation, gut permeability, visceral hypersensitivity, gut motility and HPA axis activity, and is recognized as the major endocrine system response to stress[6,14,21-25].
Intestinal bacteria also play an important and proven role in the above-described relationships (“brain-gut-enteric microbiota axis,” or “microbiome-gut-brain axis”), whose composition and function are related to nervous system activity and, in turn, communicate with the brain-gut axis through endocrine, immunological and neural messages[6,18,20,26-28]. Such bidirectional interactions are suspected on the basis of the following observations: (1) stress and mental disorders have a harmful effect on digestive tract function and intestinal bacterial flora[5,6,29]; (2) patients with digestive tract disorders have a high prevalence of neuropsychological comorbidities, such as depression, anxiety and alexithymia[13]; (3) changes in cognitive function occur after diet-induced gut-dysbiosis[18] or with alimentary tract diseases (e.g., hepatic encephalopathy) which may be altered after antibiotics, probiotics or laxative administration[15,30]; (4) patients with major depression show altered intestinal fermentation profiles[31]; (5) individuals with autism have abnormal microbiota and experience shorter antibiotic effects[32-34]; (6) some patients with digestive tract disorders demonstrate an ANS imbalance[6,14,15,21,22,29,35-39]; and (7) digestive tract stimulation and/or infection can produce abnormalities in brain-imaging techniques that show the activation (evoked potentials) or deactivation of specific brain regions, such as functional magnetic resonance imaging, positron emission tomography, magnetoencephalography and electroencephalography[40].
As a gastric mucosa-colonizing microorganism, H. pylori may contribute to the bidirectional nature of the brain-gut axis as evidenced by: (1) the overlapping of upper and lower digestive tract functional symptoms in H. pylori infected patients[18,41-43] and the effect of H. pylori infection on IBS symptoms[43] (but see Breckan et al[44]) and pancreatic juice secretion[45]; (2) the protection against IBD appearance by H. pylori infection[17,46] resulting from changes induced in brain-gut axis function (neuroendocrine-immune crosstalk)[6,15,21,22,26,47,48]; (3) the association of H. pylori infection with ANS-related extra-digestive diseases, such as atherosclerosis or cardiac arrhythmia[3,35]; (4) the improved physical and psychological health-related quality of life and sexual relationships after digestive tract symptom alleviation and H. pylori eradication[49,50]; (5) the proposed association between H. pylori infection and the development of axonal type Guillain-Barré neuropathy, multiple sclerosis and epilepsy[51-53], and case reports of gastric MALT lymphoma followed by primary CNS lymphoma[54]; and (6) the modulation of ANS balance by H. pylori infection[35]. Furthermore, H. pylori can potentially regulate esophageal motility[55], gastric emptying, gastric accommodation of ingested food, gastric acid secretion, mucosal blood flow, hypersensitivity to chemo- and mechano-stimulants[56], food intake[6,19], digestive tract endocrine and immune functions and the composition of gut microbiota[6,18,21,22], by influencing the release of various neurotransmitters, including acetylocholine (vagal nerve, parasympathetic part), noradrenaline, adrenaline and sympathetic dopamine, as well as the neuropeptides leptin, ghrelin, calcitonin gene-related peptide (CGRP), nitric oxide, neuropeptide Y, substance P (SP), somatostatin (STS) and cholecystokinin (CCK). Moreover, H. pylori infection is associated with the up-regulation of toll-like receptors and cytokine overproduction, especially tumor necrosis factor (TNF)-alpha, interleukin (IL)-1, IL-6, and IL-8[57-59], thereby indirectly influencing the brain-gut axis. These immune-mediators may stimulate mast cells (MC) in the gastric mucosa, as well as the hypothalamus and brain stem (via neuroendocrine-immuno crosstalk)[6,22], thereby activating the sympathetic ANS and pituitary-suprarenal axis, resulting in increased cortisol and adrenalin secretion[6,21,22,60,61].
ROLE OF THE BRAIN-GUT AXIS IN ACUTE H. PYLORI INFECTION
H. pylori infection occurs via the oral route and, like other contagions, depends on the balance between bacteria virulence (urease, AmiE, AmiF, hydrogenase and arginase, BabA, SabA and ureaseA), host immunity and environmental factors[62]. The brain-gut axis may modulate the local immunological resistance related to gastric and gut-associated lymphoid tissue (neuroendocrine-immune crosstalk). However, these interactions are bidirectional and seem to be more complicated with regard to H. pylori infection. For example, nervous system disorders and the lack of MC in the gastric mucosa may predispose an individual to H. pylori infection, while at the same time, acute H. pylori infection may change both CNS and ANS activity[2,22,29,37,63,64]. Autonomic neuropathy facilitates gastric mucosal colonization and hinders H. pylori eradication via the slowing of gastric emptying, changes in gastric mucosal blood flow and immunosuppression of relative hyper-sympatheticotonia[2,37,60,63,64]. In an animal model, Guo et al[63] found that psychological stress increased H. pylori colonization and gastric mucosal injury. Studies by Gulcelik et al[65] and Gentile et al[37], demonstrated a higher prevalence of H. pylori in type 2 diabetics with cardiovascular autonomic neuropathy, but not with other common diabetic complications, such as nephropathy and retinopathy. Diabetic patients also commonly experience dyspeptic symptoms as a result of disturbances in gastric emptying, gastric wall accommodation and visceral hypersensitivity. These abnormalities, particularly efferent and vagal dysfunction, may be secondary to ANS activity[66,67]. Testoni et al[68] also showed a significantly greater prevalence of H. pylori infection in patients without evidence of gastric phase III of the migrating motor complex. Collectively, these data indicate that the coexistence of ANS dysfunction and H. pylori infection is a necessary condition for H. pylori invasion, as well as for chronic gastritis and peptic ulcer formation[6,14,15,21,22,29,35-39] (but see Chang et al[69]).
While it is known that acute intestinal infection during psychological or physical stress may evoke gastrointestinal symptoms and lead to increased mucosal MC infiltration[19], there is little information available concerning the effect of acute H. pylori infection on brain-gut axis function. The main mechanism for post-infectious IBS is infiltration and long-term maintenance of MCs in the digestive tract[6,16,21,22]. MC number and activity are increased in the gastric mucosa of patients with functional dyspepsia, independent of inflammation[70], as well as in subjects with H. pylori-associated gastritis[71-75]. MCs are actively involved in the pathogenesis of H. pylori-infected gastritis acting as important effectors of the brain-gut axis by translating stress signals into the release of a wide range of neurotransmitters and proinflammatory cytokines capable of altering gastric nerve and muscle function[6,70]. Furthermore, MCs may act via gastritis maintenance and reparation of injured tissue. MCs can initiate and promote edema by secreting multiple, degranulated chemotactic factors that induce the infiltration of inflammatory cells[73], as well as promote tissue turnover by stimulating the degradation of pericellular matrices and the growth of cells in their vicinity. In chronic H. pylori infection, these processes continue until the bacteria are eradicated. In this way, both acute and chronic H. pylori infection could contribute to the pathogenesis of functional dyspepsia and gastritis symptoms[9,70], especially in patients who have acquired excessive responsiveness to stress, such as a result of environmental influences during early life, genetic abnormalities, residual inflammation after gastrointestinal infections, psychophysiological abnormalities, abnormal secretion of gastric acid, diet and lifestyle[6,76]. Moreover, as with every acute infection, H. pylori may evoke flu-like symptoms and neuroendocrine alterations. Oral administration of Campylobacter jejuni or Citrobacter rodentium leads to the activation of vagal sensory neurons and brain regions associated with viscerosensory pathways and the central autonomic network in mice[77]. These local stomach and gut infections were associated with increased anxiety-like behavior, reminiscent of the sickness syndrome typically seen following systemic administration of bacteria or bacterial products, such as lipopolysaccharides. Furthermore, H. pylori-infected mice show an upregulation of SP expression, which may increase visceral mechanosensitivity in the spinal cord[56].
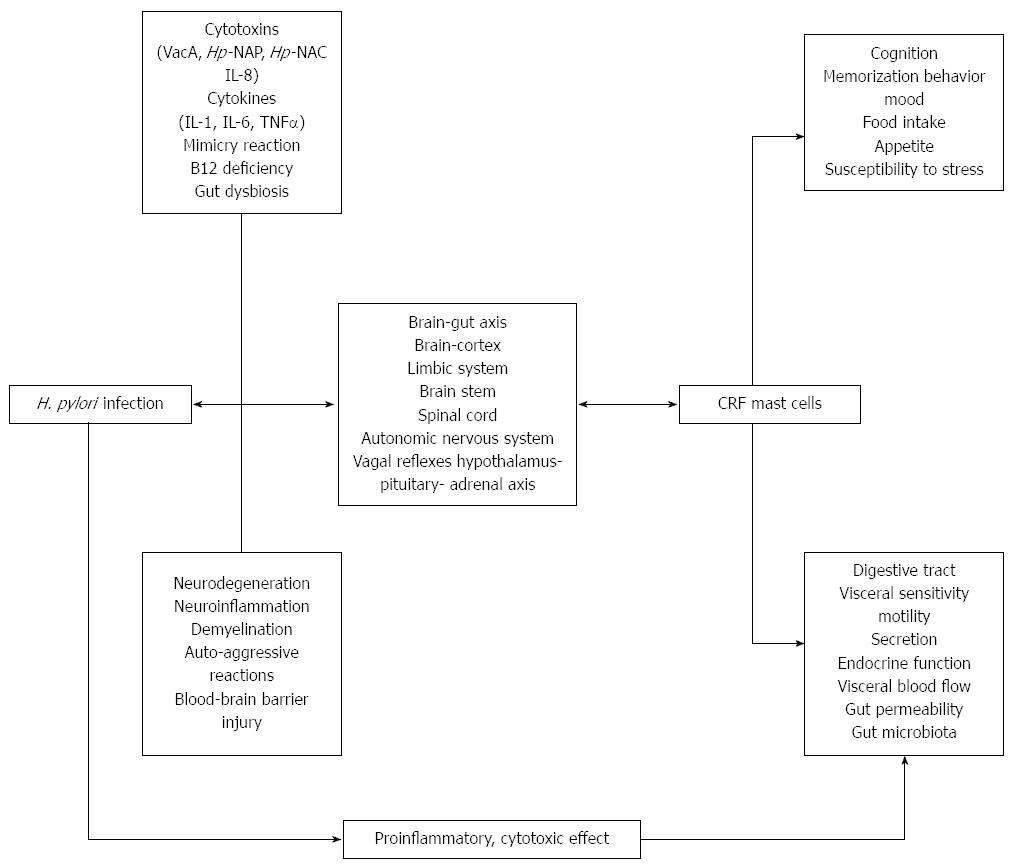
The pathophysiological interactions between H. pylori and the brain-gut axis likely involve (Figure 1): (1) axon injury or stimulation (neuroinflammation) by bacterial cytotoxins (VacA)[60,72,78], neutrophil-attractant chemokine IL-8[79], and/or neutrophil-activating protein (H. pylori-NAP)[75,79]; (2) axon damage or stimulation by autoimmunological reactions due to mimicry of VacA, bacterial aquaporin (AQP), H. pylori-NAP and human antigens[51,53,79,80]; (3) H. pylori-induced production of free radicals[80-82], cytotoxins and cytokines[57-61,78], which may also result in blood-brain barrier disruption; (4) changes in neurotransmitter secretion in gastric mucosa and spinal cord[56,72,75,81,83-85]; (5) neuron injury resulting from gastric mucosa atrophy and a decrease in vitamin B12 absorption[86]; and (6) changes in stomach and intestinal microbiota[6,87-89].
Figure 1 Pathophysiology of Helicobacter pylori infection on the brain-gut axis.
CRF: Corticotropin releasing factor; TNF-α: Tumor necrosis factor-alpha; IL: Interleukin; NAP: Neutrophil-activating protein; H. pylori: Helicobacter pylori; NAC: Neutrophil activating capacity.
ROLE OF THE BRAIN-GUT AXIS IN CHRONIC H. PYLORI INFECTION
Reports concerning chronic H. pylori infection show abnormalities in neuroendocrine activity and in the digestive tract related to H. pylori virulence, and disappearance following eradicative therapy. Li et al[83] showed greater neuronal expressions of c-fos, vasoactive intestinal peptide (VIP) and CGRP in the stomach and spinal cord of H. pylori-infected animals, which they proposed could form a basis for the appearance of functional dyspeptic symptoms in H. pylori-infected patients, as these neuropeptides are involved in the sensitization of afferent neuronal pathways[84]. Similarly, Bercík et al[56] showed a decrease in acetylcholine release by electric field stimulation and a higher density of SP-, CGRP-, and VIP-immunoreactive nerves in the stomach and spinal cord as well as an increase in pro-opiomelanocortin (POMC) secretion[19] in H. pylori-infected mice. Eradication of H. pylori-normalized functional and morphological abnormalities, except for the increased density of gastric SP- and CGRP-immunoreactive nerves, which may be the basis for neuromuscular changes that produce dysmotility and hyperalgesia. This observation may have many clinical consequences, as POMC is a precursor for several important biologically active substances, including N-terminal peptide of pro-opiomelanocortin (or pro-γ-MSH), γ-melanotropin, adrenocorticotropic hormone, α-melanocyte-stimulating hormone, corticotropin-like intermediate peptide, β-lipotropin, γ-lipotropin, β-melanotropin, β-endorphin and met-enkephalin.
Neuronal and non-neuronal levels of SP, VIP, CGRP and neprilysin activity were examined in freshly frozen biopsies taken from patients with chronic gastritis or ulcers in a study by Erin et al[85] showing that neuronal SP, mucosal VIP and neprilysin levels decreased significantly in normal appearing mucosa in patients with gastritis, while levels of mucosal SP increased in areas of gastritis and ulcer. In this study the presence of H. pylori was associated with further decreases in SP levels. Hydrolysis of SP by neprilysin is known to have gastroprotective effects, therefore the decreased levels of VIP, SP and neprilysin were considered by the authors as a predisposition to cellular damage. In contrast, Dömötör et al[90] concluded that capsaicin-sensitive afferent nerves with gastroprotective properties do not participate in the harmful effects of H. pylori as they found that although the expression of the capsaicin receptor, CGRP and SP were significantly higher in the mucosa of patients with chronic gastritis, there were no significant differences between H. pylori-positive and H. pylori-negative cases.
A proportion of H. pylori effects on the brain-gut axis may be secondary to central and peripheral nerve demyelination and blood-brain barrier disruption[79]. The cross-mimicry between VacA and H. pylori-NAP is recognized as the most important immunological reaction[79], which induces autoreactive T cells and initiates or worsens gastric autoimmunity leading to atrophic gastritis and gastric cancer[91,92]. Cross-reactivity of anti-VacA antibodies with ion channels in Schwann cell plasmalemma could potentially induce demyelination, since cross-mimicry between H. pylori VacA and the human Na+/K+-ATPaseA subunit was identified by sequence homology. Cross-mimicry between H. pylori-NAP and/or anti-bacterial AQP antibodies and neural tissues is also possible. Whereas blood-brain barrier disruption may be secondary to the effects of H. pylori-induced inflammatory mediators, it can lead to increased brain vessel permeability for immunomediators, non-specific antibodies, AQP4-specific antibodies and inflammatory cells (e.g., lymphocytes), which can evoke many pathological processes in the CNS, including demyelination, local edema and neuroinflammation. These processes are recognized as important for associations between H. pylori infection and the pathogenesis of demyelination, multiple sclerosis and neuromyelitis optica[51,52,79].
Intermediate effects of chronic H. pylori infection on brain-gut axis function have been clinically observed as: (1) the alteration of feeding patterns[15,19]; (2) cognitive and memory dysfunction[18,19,27,82,93], increased vulnerability to stress[15,19,27] and anxiety- and depressive-like behaviors[19]; (3) alterations in endocrine functions of the stomach, including the production of SP, VIP, CCK, STS, gastrin and ghrelin[81,94]; (4) changes in visceral ANS balance and the action of vagal visceral reflexes[95]; (5) alterations in gastrointestinal motility[21]; (6) increased visceral perception (chemo- and mechano-hypersensitivity)[67,96]; (7) changes in gastrointestinal secretion[21,45]; (8) increased intestinal permeability[23,61]; (9) intestinal microbiota[87-89], with indirect effects on the brain-gut axis[27,28,97-99]; (10) alterations in immunological reactivity, resulting in decreased prevalence of food allergies and inflammatory bowel diseases[46,100]; and (11) the overlapping of gastrointestinal disorders from upper and lower parts of the digestive tract[43]. Moreover, H. pylori eradication has also been shown to normalize some of these alterations[55,95,100-103].
H. PYLORI INTERACTIONS WITH THE BRAIN-GUT AXIS IN THE REGULATION OF FEEDING PATTERNS, APPETITE AND WEIGHT CONTROL
Clinical practice shows appetite improvement and increased body weight following H. pylori eradication[104,105]. Furthermore, corresponding changes in plasma and/or the gastric mucosal levels of hormones regulating appetite and/or energy expenditure were identified, such as ghrelin and leptin, two hormones primarily produced, stored and secreted by the gastric mucosa[106]. An animal model of H. pylori infection also showed abnormal feeding behavior observed as an increase in eating frequency and a decrease in the quantity of food consumed per meal[56]. These changes in feeding behavior were accompanied by elevated plasma ghrelin, postprandial CCK and higher TNF-alpha, along with reduced POMC mRNA in the arcuate nucleus of the brain. However, eradication therapy had no effect on eating behavior in this study, despite the normalization of gastric emptying and sensitivity.
A study of dyspeptic patients by Ulasoglu et al[55] found decreased ghrelin and increased obestatin blood levels after H. pylori eradication, which were greatest in overweight and male patients. However, Francois et al[102] reported a postprandial increase in ghrelin and leptin levels, and an increase in body mass index (BMI) in subjects 7-18 mo after successful H. pylori eradication treatment. Restored tissue levels of ghrelin and an improved appetite were also achieved after gastric ulcer healing and H. pylori eradication in studies by Roper et al[106], Jang et al[107] and Osawa et al[108]. Similar results were also obtained by Yang et al[101], who found lower serum ghrelin levels, reduced body weight and height and lower BMI and body weight gain in H. pylori-infected children, with restored ghrelin levels and increased growth observed one year after H. pylori eradication. Increased plasma and gastric mucosal ghrelin levels after H. pylori eradication were also shown by Deng et al[103] in 50 children with H. pylori-associated functional dyspepsia. However, a study reported by Choe et al[109] failed to show a relationship between H. pylori infection and stomach mucosal ghrelin levels, with no effect of eradication therapy. It should be noted that a greater BMI and corpus predominant gastritis are recognized as independent risk factors for eradication failure[110].
H. PYLORI INTERACTIONS WITH THE BRAIN-GUT AXIS IN BEHAVIOR, COGNITIVE FUNCTION AND MENTAL DISORDERS
The participation of the brain-gut axis in associations between bacterial flora in the bowel and some behavior patterns, and cognitive and mental disorders as well as their improvement following antibiotic, pre- and/or probiotic administration, have been reported previously, both in animals[19,24,25,93,97-99] and in humans[6,14,27,28]. It is also possible that psychiatric disorders in H. pylori-infected subjects result from disease overlap, with comorbidity in 40% of patients with functional dyspepsia, of which depression is the most prevalent (24%)[111].
Bacterial infection of germ-free mice evokes anxiety-like behavior, which is relieved after introducing normal gut microbiota[19,24,25,97-99]. A corresponding increase in brain-derived neurotrophic factor (BDNF) in the hippocampus and amygdala was observed that also diminished after antibiotic therapy in these mice[19]. BDNF is involved in the neurobiology of depression[112], and is also considered a biomarker for gastric hypersensitivity and an overactive bladder[113,114]. Moreover, stress-induced memory dysfunction in mice after bacterial infection was reported by Gareau et al[93].
In a large, cross-sectional study of United States adults by Beydoun et al[115], H. pylori-seropositivity was associated with poor cognition based on declining verbal memory test performance. H. pylori, as with other urease-positive bacteria that produce ammonia, is also recognized as a cause of liver-like encephalopathy[116]. In a study by Roubaud Baudron et al[117] who followed-up individuals aged 65 years or older for 20 years, H. pylori infection was recognized as a significant risk factor for the development of dementia as a result of enhanced neurodegenerative processes, a hypothesis that has been suggested by Deretzi et al[80]. Moreover, a study by Muhsen et al[118] indicated that H. pylori might also negatively affect early cognitive development. In our own study, depressive and alexithymic alcoholics had less intensive H. pylori antral colonization than subjects with lower Beck Depression Inventory and Toronto Alexithymia Scale scores[29]. In a study by Ozdemir et al[119], a significant association was observed between schizophrenia and diminished risk of duodenal and gastric but not gastrojejunal ulcers. The authors concluded that schizophrenia and attendant neurobiological mechanisms (e.g., variability in dopamine pathways) may therefore modify the risk of gastric and duodenal ulcers as both central and peripheral dopamine pathways influence ulcer pathogenesis, and dopamine agonists preclude antagonists from augmenting stress - and chemically - induced gastrointestinal ulcers in preclinical models.
Probiotics reduce the adverse effects, prevent complications and improve the outcome of H. pylori-eradicative therapy[120]. Furthermore, Bifidobacterium bifidum YIT 4007 in fermented milk suppresses H. pylori growth[100]. However, research concerning the effect of probiotic supplementation on brain-gut axis function and mental disorders in subjects with H. pylori is lacking, as is data regarding the adverse effect of eradicative therapy on intestinal microbiota for a sub-form of dysbiosis in the stomach[89]. Myllyluoma et al[88] showed that the triple standard H. pylori-eradicative therapy (clarithromycin, amoxicillin and lansoprazole) significantly changed the quantities of the predominant bacterial groups both in H. pylori-positive and H. pylori-negative patients, with disturbances observed up to nine weeks after treatment completion. In their study, a combination of probiotics containing Lactobacillus rhamnosus GG, L. rhamnosus LC705, Propionibacterium freudenreichii spp., Shermanii JS and Bifidobacterium breve Bb99 only slightly counteracted the effects of anti-H. pylori treatment, seen as significantly fewer alterations in the total numbers of aerobes and lactobacilli/enterococci. However, it was found that probiotics in animal models reduced vagal-dependent activation of GABA receptors in response to physical or psychological stress[6,19]. Moreover, probiotics prevented stress-induced memory deficits in mice, reduced BDNF and c-fos expression in the hippocampus, as well as attenuated corticosterone release in response to stress[27,93,97-99].
H. PYLORI AND ANS ACTIVITY
The ANS is the most important part of the brain-gut axis, containing afferent and efferent pathways regulating saliva and gastric juice secretion, mucosal blood flow, inflammatory process activity, digestive tract motility and visceral pain threshold (visceral sensitivity)[15,21,22,35,56]. Disturbances in ANS balance are observed in patients presenting with H. pylori infection and functional and organic digestive tract disorders[10,14,15,21,22,29,35-39]. Our study found that among patients diagnosed as a result of atypical chest pain, subjects infected with H. pylori had increased ANS activity, an effect that was confirmed by multi-factorial analysis[35]. Moreover, our study showed negative relationships between H. pylori infection and esophageal sensitivity to acid, the number of acid gastroesophageal refluxes, and time spent at a gastric pH < 4, as well as positive relationships between H. pylori and the efficacy of esophageal peristalsis, and the amplitude and duration of esophageal contractions. We therefore concluded that gastric mucosal colonization by H. pylori affects ANS activity and can evoke disturbances in esophageal and gastric function. Vagal nerve predominance, as diagnosed by heart rate variability analyses, was also recognized as a risk factor for duodenal ulcer development[2,38-39,121]. Lugon et al[122] reported blunted sympathetic reactivity and an exacerbated vagal response to feeding in H. pylori-positive subjects. However, Katoh et al[36] have shown that H. pylori eradication in duodenal ulcer patients does not change nocturnal sympatheticotonia or parasympatheticotonia, which may be the cause for persistently increased gastric acid secretion, gastric mucosal vasoconstriction and ulcer recurrence[38,121], though the time course between eradication and ANS evaluation may have been too short to reveal statistically significant differences. Indeed, Stanghellini et al[123] suggest there is long-term recovery of H. pylori-induced neuroplastic changes in the ANS and highlight the importance of an incomplete resolution of gastritis and the presence of a persistent production of inflammatory mediators even after successful H. pylori eradication. However, Thor and Blaut[124] suggest that H. pylori eradication restores the parasympatheticotonic imbalance via a ghrelin-leptin interplay. Shahabi et al[125] further suggest that H. pylori infection of the gastric mucosa may induce a T-helper-1-like immune response and production of proinflammatory cytokines, which can inhibit local, and increase systemic, sympathetic and cholinergic tone. Such ANS changes can inhibit inflammation in the esophagus and stomach.
It is known that the brain-gut axis, including the CNS, PNS and ANS, modulates visceral reflex activity. This activity is responsible for the local regulation of digestive tract function, such as the gastric and intestinal phase of gastric or pancreatic juice secretion, esophageal, gastric and jejunal motility and visceral sensitivity. Furthermore, visceral reflexes regulate the remote effect of gastrointestinal stimulation, including the cardio-esophageal or gastro-cardiac reflexes responsible for coronary artery spasm in response to esophageal acidification or distension. In return, coronary angioplasty can induce esophageal motility abnormalities, a decrease in the pain threshold and esophageal and gastric hyperalgesia, via cardio-esophageal reflex activation[67,95]. As these effects were only observed in 56% of subjects with non-cardiac chest pain[95], reciprocal modulation by the brain-gut axis may have had further influence along with individual reactions to stress[6,21,22]. Thus, more research is needed concerning the effect of H. pylori infection on visceral reflexes.
H. PYLORI AND VISCERAL SENSITIVITY
Visceral hypersensitivity is a symptom of functional digestive tract disorders that may be modulated by H. pylori infection and the brain-gut axis[76]. The brain-gut axis plays an important role in the tonic or phasic up- or downregulation of afferent and efferent nerve sensitivities[18,21,22]. Visceral afferent pathways (enteric, spinal and vagal) transmit signals from the stomach to the CNS. As mentioned above, these afferents may also modulate emotion, pain, satiety and immunity in response to emotional influences or environmental demands. Visceral hypersensitivity may also be affected by hypersympatheticotonia. However, both hypervagotonia and hypersympatheticotonia may change efferent nerve activity due to modulation of vagal visceral reflex activity and the patient’s digestive tract response to visceral or environmental stimuli[67].
H. pylori infection may additionally affect brain-gut axis function and visceral sensitivity by neurotransmitter and hormone release and activation of inflammatory processes[61], as well as induce digestive tract dysmotility and trigger neuroplastic changes in the afferent neural pathways leading to visceral hyperalgesia[123]. A reduction in central antinociceptive control systems, visceral pain modulators both in digestive tract disorders and extra-gastric syndromes, may also play a pathophysiological role in H. pylori infection.
Bercík et al[56] found that an increase in visceral perception (mechanosensitivity) in H. pylori-infected mice was related to an increase in vagal SP expression. Similarly, Mönnikes et al[126] showed that discomfort and pain thresholds on gastric distension were lower in patients with functional dyspepsia that were H. pylori-positive. These patients also showed higher antral mucosal levels of CGRP and SP, which negatively correlated with the levels of discomfort and pain thresholds, demonstrating the role of the brain-gut axis in hypersensitivity and SP and CGRP involvement in the sensitization of afferent neuronal pathways, which result from chronic inflammation[84,126]. Similar inflammation-related changes in sensory-motor function have been observed in subjects with intestinal mucosal inflammation[9,11,123]. The role of H. pylori infection in the symptoms of functional dyspepsia was also confirmed by a Cochrane review, which reported that eradication of H. pylori had a small but statistically significant long-term effect on symptom relief when compared with a placebo, lasting at least 12 mo after one week of eradication therapy, expressed more in patients from Asian rather than Western countries[92,127]. However, Mearin et al[128] found that H. pylori-infected patients with functional dyspepsia presented no distinctive symptoms compared to their H. pylori-negative counterparts. Furthermore, additional placebo-controlled trials and a meta-analysis did not show improvement in dyspeptic symptoms with H. pylori eradication[129-131]. Discrepancies regarding symptomatic response to H. pylori eradication may relate to commonly occurring comorbid psychiatric disorders, which can affect treatment outcome[111]. H. pylori infection is also considered a cause of visceral hypersensitivity in patients with IBS, as individuals reporting typical abdominal discomfort after rectal barostat testing were more frequently H. pylori positive[42,43].
Disturbances in ANS activity, particularly hyperparasympatheticotonia, should also be recognized as a factor responsible for the clinical manifestation of lowered pain thresholds. H. pylori infection may increase sympathetic ANS activity via induction of gastritis and overproduction of systemic inflammatory mediators through a pathogen-burden mechanism[61]. Increased ANS activity is related to symptom thresholds in patients with peptic ulcers[38-39,121], in acid-sensitive and insensitive patients with non-cardiac chest pain[67], in patients with non-specific disorders of esophageal motility[132], as well as in individuals with functional dyspepsia[9,21,42,66,76,94,126,133] and IBS[134]. Such a pathway is also considered as one of the causes of pain in H. pylori-infected patients with fibromyalgia[96].
H. PYLORI AND DIGESTIVE MOTILITY
Digestive tract motor disorders are a source of functional symptoms originating from the upper and lower parts of the digestive tract[76]. Typical gastric motility abnormalities involve impaired fundic accommodation and reduced compliance to meals, impaired initial distribution of a meal, antral hypomotility, altered myoelectric activity and gastric emptying and disturbed gastro- and duodeno-jejunal motor coordination[133]. Delayed gastric emptying has been described in about 30%-80% of patients with functional dyspepsia as a result of antral or fundal hypomotility, uncoordinated antro-pyloro-duodenal activity or inhibitory feedback of the intestine[135]. Symptoms of motility disorders may be evoked through the activation of chemo- (delayed clearance) and mechano-receptors (digestive tract tube contraction or extension due to hypertensive spasm or dilatation proximal to contraction), via activation of viscerosomatic reflexes, and somatic receptors localized in the distended abdominal wall in the course of bloating, especially in individuals with hypersensitivity to stress. Furthermore, symptoms relating to reflux of gastric content into the esophagus, or other causes of delayed gastric emptying and/or its accommodation impairment, may appear in patients with gastroparesis[136].
The effect of H. pylori infection on digestive tract motility may result from its direct local action, or an intermediate effect via the above-described ANS and endocrine effects (i.e., due to alteration in gastrin, STS, CCK and plasma and mucosal ghrelin levels)[137-140]. However, there are no recent studies on the effect of H. pylori infection and its eradication on esophageal and gastric motility. Chiloiro et al[140] showed higher gastrin and lower CCK plasma levels in H. pylori-infected patients with functional dyspepsia, but did not find a difference in gastric emptying. Similarly, Schenk et al[137] showed that severe hypergastrinemia during omeprazole maintenance therapy for gastroesophageal reflux was associated with the duration of therapy and H. pylori infection, but not with abnormalities of gastric emptying or vagal nerve integrity. Currently, there are no reports concerning the effect of H. pylori-induced hypergastrinemia secondary to hypochloremia in the course of H. pylori infection-related gastric corpus mucosa atrophy. However, an in vitro study showed that eradication of H. pylori did not affect muscle contractility and acetylcholine release but augmented antral relaxation after nerve electrical field stimulation[56].
A study by our group found positive relationships between H. pylori infection and the efficacy of esophageal peristalsis and the amplitude and duration of esophageal contractions in patients with atypical chest pain[35]. However, Tsai et al[141] and Zerbib et al[142] did not find any differences in the patterns of esophageal motility and transient lower esophageal sphincter relaxations between H. pylori-positive and H. pylori-negative subjects. Similarly, Tanaka et al[143] did not observe changes in gastric and esophageal function after H. pylori eradication, besides an increase in gastric acidity at night, suggesting that H. pylori does not produce esophageal-motility-related chest pain, although it induced a more active inflammatory process in esophageal mucosa[142].
The presence of H. pylori infection also did not significantly affect gastric emptying rates for solids and liquids, discomfort sensitivity thresholds, or meal-induced gastric relaxation in dyspeptic patients[144]. H. pylori-positive and -negative patients in a study by Mearin et al[128] also suffered from similar severity and frequency of dyspeptic symptoms and global symptom scores, and presented no differences in gastric compliance. However, postprandial antral motility in H. pylori-positive patients was significantly decreased, though without an increase in their perception of gastric distension. A report by Thumshirn et al[145] showed that impaired gastric accommodation is frequent in non-ulcer dyspepsia, but unrelated to H. pylori infection and vagal efferent dysfunction. Additionally, a placebo-controlled double-blind study showed that H. pylori eradication in patients with functional dyspepsia had no impact on gastric emptying as estimated using a standardized scintigraphic double-tracer[146]. Furthermore, a decreased prevalence of normal electrogastrography (EGG) patterns was found in both H. pylori-positive and -negative patients with functional dyspepsia compared to healthy controls, though only subjects with delayed gastric emptying achieved symptom improvement and normalization of the EGG pattern after H. pylori eradication[135]. In contrast, a study by Simrén et al[20] indicated that H. pylori infection was independently associated with the presence of unsuppressed phasic fundic contractility after a meal, which induced a transient increase in gastric wall tension that was observed in 15% of subjects. Lin et al[147] found abnormal gastric myoelectrical activity in approximately 40% of H. pylori-infected patients with functional dyspepsia, which was normalized in some following H. pylori eradication, with similar observations reported by Thor et al[148]. In a double-blind study performed in patients with duodenal ulcers, Konturek et al[139] showed that H. pylori infection was accompanied by enhanced gastric emptying and reduced luminal release of STS, although a CCK antagonist had no effect in H. pylori-infected subjects. The authors therefore concluded that, since gastric motor function and STS secretion were normalized after H. pylori eradication, CCK and STS contribute to the normalization of gastric emptying following H. pylori eradication in patients with duodenal ulcer.
Current data demonstrate contrary outcomes of the effect of H. pylori-infection on digestive tract motor function, and further studies are therefore needed to address these discrepancies as well as the involvement of the brain-gut axis. Indeed, this has been suggested by Manes et al[149] who highlighted the low quality of the studies performed, methodological problems relating to the small numbers of patients, differences in methodological approaches and the well-known difficulties in studying both gastrointestinal motility and functional dyspepsia.
H. PYLORI AND GASTROINTESTINAL SECRETION
The known phrase, “no acid - no ulcer” emphasizes the important role of gastric acid secretion in the pathogenesis of upper digestive tract symptoms and injuries. As H. pylori infection is recognized as the main ulcerogenic factor, it is an important pathomechanism of direct and immediate harmful H. pylori action via the brain-gut axis. The effect of H. pylori infection on gastric acid secretion depends on the location of the gastritis. In localization of the antral inflammatory process, acid hypersecretion was observed, leading to stomach mucosal atrophy and hypochlorhydria in patients with corporal gastritis or pan-gastritis[141]. After H. pylori eradication, intragastric pH in the postprandial period was significantly lower, whereas baseline pH remained unchanged[150]. These changes were expressed particularly in patients with the proinflammatory IL-1B genotype, which is associated with an increased risk of gastric mucosa atrophy, lower gastric acidity and gastric carcinoma[151].
Chronic gastritis also affects a variety of endocrine functions of the stomach, including the production of STS, gastrin and ghrelin[94]. As gastric parietal cells are regulated by hormones and neurotransmitters, such as gastrin, histamine and acetylocholine, analysis of the effect of H. pylori-infection on the brain-gut axis is reasonable. H. pylori infection leads to a decrease in gastric acid secretion due to mucosal atrophy and vagal nerve dysfunction[81], which may result in alteration of both gastric motility and secretion, as well as food intake.
Abnormal vagal nerve activity should be considered as a potential mediator of H. pylori-induced changes in secretion of gastric acid, gastrin and ghrelin. However, there are no recent studies investigating the effect of H. pylori infection and eradication on gastric acidity. Lucini et al[39] examined the association between ANS activity and gastric acid secretion and found that sham-fed patients with duodenal ulcers had greater vagal and lower sympathetic nerve activities using heart rate variability (HRV) analyses. In these patients, the sham feeding to basal acid output ratio value correlated with markers of ANS activity. A study by Nada et al[121] reported high sympatho-vagal tone at night in patients with peptic ulcers, which can result in gastric artery spasms and excess secretion of gastric acid. In the same study, HRV markers of vagal nerve activity and gastric juice secretion increased in adult mongrel dogs following an intravenous injection of insulin.
H. pylori-positive patients with duodenal ulcers showed increased plasma gastrin concentrations in a study by Iijima et al[152], while H. pylori-positive patients with gastric ulcers showed lower concentrations compared to H. pylori-negative counterparts. In both groups, the gastrin plasma levels changed after H. pylori eradication, decreasing in individuals with duodenal ulcers, and increasing in those with gastric ulcers. Ghrelin is also secreted by gastric mucosa cells, and not only regulates food intake, but also exhibits an effect on gastroprotection and increases esophageal and gastric motility and gastric acid secretion[6,14,124,148,153].
CONCLUSION
H. pylori infection, the main pathogenic factor of upper digestive tract organic diseases, displays not only local cytotoxic and proinflammatory effects, but may also exert systemic action via modulation of the brain-gut axis. These relationships are bidirectional and influence the contagion process and the host neuro-endocrine immunological reaction, including alterations in cognitive functions, modification of symptom sensitivity thresholds and digestive tract secretion and motility. The effect on the brain-gut axis by this microorganism may arise from a direct neurotoxic effect, activation of inflammatory processes in nerves and microelement deficiency. However, many of the investigations concerning the interplay between H. pylori and the effects on brain-gut axis activation are dated. Therefore, further studies are necessary to evaluate the clinical importance of these host-bacteria interactions. Results from such studies will improve our understanding of H. pylori infection pathophysiology and suggest new therapeutic approaches.