Published online May 14, 2014. doi: 10.3748/wjg.v20.i18.5177
Revised: January 20, 2014
Accepted: March 6, 2014
Published online: May 14, 2014
Processing time: 229 Days and 17.6 Hours
Obesity-associated diseases account for a large portion of public health challenges. Among obesity-related disorders, a direct and independent relationship has been ascertained for colorectal cancer (CRC). The evidence that adipocyte hypertrophy and excessive adipose tissue accumulation (mainly visceral) can promote pathogenic adipocyte and adipose tissue-related diseases, has led to formulate the concept of “adiposopathy”, defined as adipocyte and adipose tissue dysfunction that contributes to metabolic syndrome. Adipose tissue can, indeed, be regarded as an important and highly active player of the innate immune response, in which cytokine/adipokine secretion is responsible for a paracrine loop between adipocytes and macrophages, thus contributing to the systemic chronic low-grade inflammation associated with visceral obesity, which represents a favorable niche for tumor development. The adipocyte itself participates as a central mediator of this inflammatory response in obese individuals by secreting hormones, growth factors and proinflammatory cytokines, which are of particular relevance for the pathogenesis of CRC. Among adipocyte-secreted hormones, the most relevant to colorectal tumorigenesis are adiponectin, leptin, resistin and ghrelin. All these molecules have been involved in cell growth and proliferation, as well as tumor angiogenesis and it has been demonstrated that their expression changes from normal colonic mucosa to adenoma and adenocarcinoma, suggesting their involvement in multistep colorectal carcinogenesis. These findings have led to the hypothesis that an unfavorable adipokine profile, with a reduction of those with an anti-inflammatory and anti-cancerous activity, might serve as a prognostic factor in CRC patients and that adipokines or their analogues/antagonists might become useful agents in the management or chemoprevention of CRC.
Core tip: Obesity-associated diseases account for a large portion of public health challenges, and adipose tissue can be regarded as an important and highly active player of the innate immune response, in which cytokine/adipokine secretion is responsible for a paracrine loop between adipocytes and macrophages. This interplay contributes to the systemic chronic low-grade inflammation associated with visceral obesity, which represents a favorable niche for tumor development. Available findings suggest that an unbalanced adipokine profile might serve as a prognostic factor in colorectal cancer patients and that adipokines or their analogues/antagonists might become useful agents in the management or chemoprevention of colon cancer.
- Citation: Riondino S, Roselli M, Palmirotta R, Della-Morte D, Ferroni P, Guadagni F. Obesity and colorectal cancer: Role of adipokines in tumor initiation and progression. World J Gastroenterol 2014; 20(18): 5177-5190
- URL: https://www.wjgnet.com/1007-9327/full/v20/i18/5177.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i18.5177
The incidence of obesity, defined as abnormal fat accumulation in adipose tissue that may impair health (http://www.who.int/mediacentre/factsheets/fs311/en, updated March 2013), is increasing worldwide, and obesity-associated diseases account for a large portion of public health challenges. Among obesity-related disorders, a direct and independent relationship has been ascertained for colorectal cancer (CRC)[1].
Previous studies indicated that the carcinogenic effect of excess weight differs among cancer sites and gender[2,3]. Several systematic reviews and meta-analyses summarized the evidence that while the relative risk associated with obesity [defined by a body mass index (BMI) ≥ 30 kg/m2] was higher for colon cancer than for rectum cancer and it was higher in men than in women, abdominal adiposity (as determined by waist circumference or waist-to-hip ratio) had a strong association with colon cancer in both sexes[4-6]. This evidence suggests that, in women, fat localization is a more important risk factor for colon cancer than body weight or BMI. It is worth noting that when abdominal obesity is measured by waist circumference (WC), which is more closely related to metabolic changes compared to general obesity as determined by BMI, weight change is associated with excess colon cancer risk in people with attained high WC at age 50 years, but not in those having low WC[7]. Conversely, no differences can be attained for BMI[7]. Although the association between obesity and colon cancer is strongest, a weak correlation has been observed between waist circumference and fat mass and risk of rectal cancer[8]. However, given all the above, there is one consideration that should be outlined, since it might represent a strong bias in the correct interpretation of the study results, that is the choice of the measurement and, most of all, the use of self-reported measures. Thus, it should be advisable to adjust the data according to morphometric cut offs.
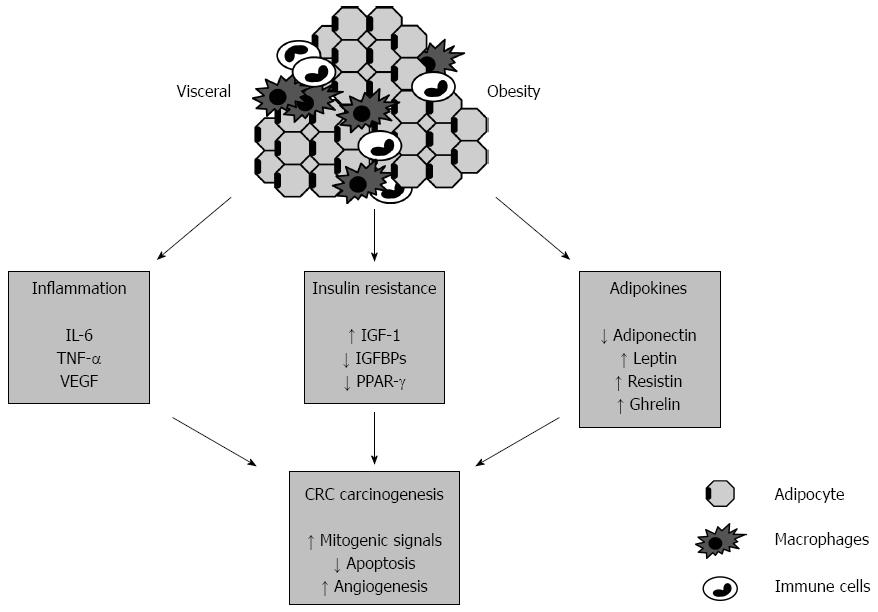
Despite these limitations, several mechanisms linking adiposity to CRC risk have been proposed, among these, obesity-related insulin resistance, hyperinsulinemia, sustained hyperglycemia, oxidative stress[9], adipocytokine production[10] and hyperinsulinemia-related increase of insulin-like growth factor-1 (IGF-1)[11], all responsible for cancer promoting effects, favoring tumor growth, increasing cell migration, and ultimately leading to metastasis (Figure 1). In this respect, it is worth noting that obesity appears to be associated with worse cancer outcomes, both in terms of cancer recurrence and mortality (reviewed in[12]).
In order to relate CRC to obesity, the biochemical behavior of adipose tissue must be considered.
Adipose tissue, which has long been regarded to have only energy storage, thermal regulation and mechanical protection functions, it is now recognized as an endocrine/metabolic organ[13]. Two different tissues differing in structure, function, and lineage must be distinguished: white adipose tissue (WAT) and brown adipose tissue (BAT). Of the two, WAT is known to produce cytokines and chemokines that induce inflammation[14], while BAT is highly metabolic and produces heat through a specialized metabolic pathway mediated by the expression of the tissue-specific uncoupling protein 1[15]. Contrary to what previously believed, it has been recently demonstrated that functional BAT is prevalent in adult females and that BMI is inversely correlated with the amount of BAT, thus suggesting a possible role of BAT in protecting against obesity[15]. On the other hand, the accumulation of WAT in the abdominal cavity represents the visceral adipose tissue (VAT), which exhibits increased insulin resistance, lipolysis, and inflammatory cytokine expression respect to subcutaneous adipose tissue (SAT).
The evidence that adipocyte hypertrophy and excessive adipose tissue accumulation can promote pathogenic adipocyte and adipose tissue effects, has led to formulate the concept of “adiposopathy”, defined as adipocyte and adipose tissue dysfunction that, together with other factors (any two of raised TG levels, reduced HDL cholesterol, raised blood pressure, or impaired glucose metabolism) contributes to the onset of metabolic syndrome[16]. Adiposopathy is also characterized by recruitment of committed mesothelial stem cell progenitors of preadipocytes[17]. Moreover, the increasing knowledge on the role of WAT in immunological responses, vascular diseases, and appetite regulation has evoked adiposopathy to define the association among obesity, inflammation and metabolic/vascular complications as a formal disease[18,19].
Existing evidence suggest that VAT is more predictive than SAT of obesity-associated comorbidity and mortality[20]. This has been related to the unique architecture of VAT that is highly cellular, vascular and innervated and contains cells with inflammatory and immunological functions[21]. Moreover, the close proximity of VAT to the portal vein causes drainage of excess free fatty acids and inflammatory mediators directly to the liver, thus affecting metabolism[22] and creating a condition of low-grade chronic inflammation, a favorable niche for tumor development. Epidemiological and clinical evidences, in fact, support the concept that longstanding inflammation represents a risk factor for the development of CRC arising in individuals with colitis-associated CRC[23].
The possibility of a direct involvement of VAT in inflammatory processes has been suggested by the direct finding that preadipocytes could function as macrophage-like cells[24], that adipocytes exert phagocytosis and microbicidal activities similar to macrophages[25] and that adipose tissue is infiltrated by significant numbers of macrophages[26]. Adipose tissue macrophages that infiltrate into VAT exhibit a phenotypic switch from an anti-inflammatory and adipostatic (M2) to a proinflammatory and pro-adipogenic (M1) phenotype, thus contributing to obesity-related inflammation and insulin resistance[27]. M1 macrophages express a series of proinflammatory cytokines [e.g., tumor necrosis factor-α (TNF-α), Interleukin-1 (IL-1), IL-6, IL-12, and IL-23] and release high levels of superoxide anions, oxygen radicals, and nitrogen radicals[28]. The M1/M2 balance is also actively involved in the microenvironment of tumor growth[28]. Once activated, macrophages are a further source of growth factors and cytokines, which contribute to affect the local microenvironment (Figure 1).
Adipose tissue can thus be regarded as an important and highly active player of the innate immune response, in which cytokine/adipokine secretion is responsible for a paracrine loop between adipocytes and macrophages, thus contributing to the systemic chronic low-grade inflammation associated with visceral obesity[29]. The adipocyte itself participates as a central mediator of this inflammatory response in obese individuals by secreting hormones (e.g., leptin, adiponectin), growth factors [e.g., IGF-1, vascular endothelial growth factor (VEGF)] and proinflammatory cytokines (e.g., TNF-α and IL-6)[14], which are of particular relevance in the pathogenesis of CRC. These altered factors act on a common pathway, through phosphoinositide kinase-3 (PI3K)/serine-threonine-protein kinase (AKT) activation, which, in turn, regulates downstream targets leading to increased cell survival and cell growth[30], ultimately causing hyperplasia, proliferation and carcinogenesis in colonic cells[31].
It is worth mentioning that in the tumor microenvironment, cancer cells interact with normal colonocytes and that in the presence of cancer cells, adipocytes can revert from mature, differentiated adipocytes to pre-adipocytes[32]. Adipocytes, thus represent a major source of energy for the cancer cell. Indeed, among the factors evoked to represent important contributors to tumor progression there are several energy balance-associated factors[33], of which adipokines are directly involved in colon carcinogenesis and tumor development.
Adipokines have a wide variety of functions, acting not only as local paracrine signaling cytokines, but also at distant levels, through secretion in the circulation.
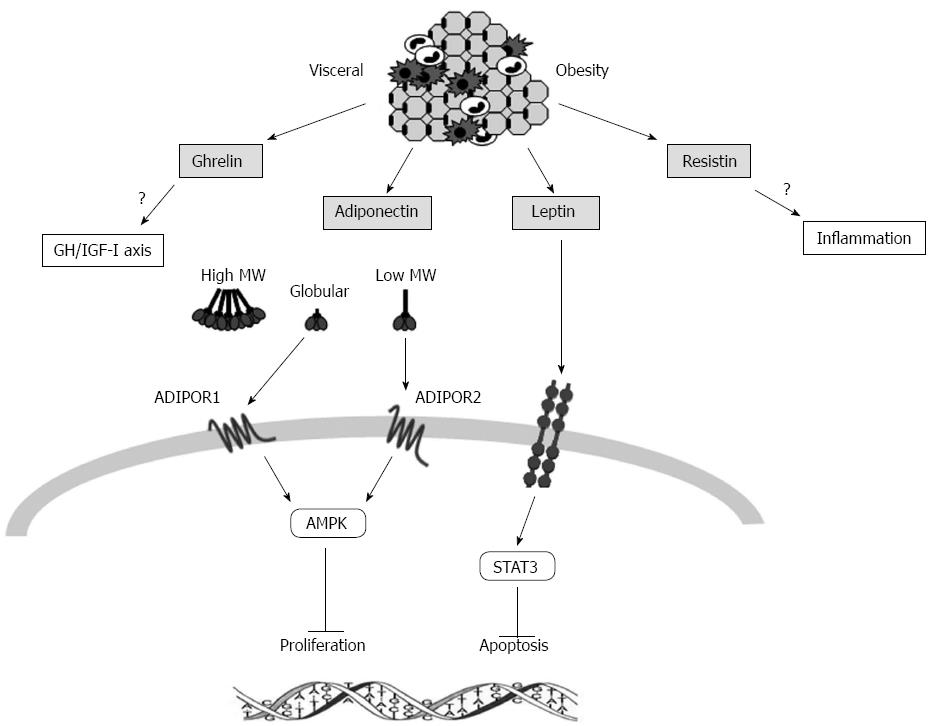
As the adipose tissue expands, the vasculature is insufficient to adequately oxygenate the adipocytes, thus, the resulting hypoxia and oxidative stress contribute to the overproduction of cytokines in obesity. Indeed, hypoxia, probably mediated by hypoxia-inducible factor-1 (HIF-1), initiates an inflammatory/angiogenic response similar to that operating in tumor growth[34]. In addition, adipocyte hypertrophy generates cellular metabolic and structural changes leading to endoplasmic reticular (ER) stress that, through activation of nuclear factor κB (NF-κB), stimulates the production of adipokines[35,36]. Finally, IR and impaired glucose metabolism in obesity may result in oxidative stress further amplyfing this pathogenetic loop[37]. Whatever the mechanism involved, adipokines may exert direct pro-tumorigenic properties in the gastrointestinal tract, although it is not yet known whether they act directly on gastrointestinal mucosal cells in an endocrine manner and/or in a paracrine manner through induction of local tissue inflammation[38]. Among adipocyte-secreted hormones, the most relevant to colorectal tumorigenesis are adiponectin, leptin, resistin and ghrelin (Figure 2).
Adiponectin is a 30-kDa insulin-sensitizing protein hormone with a collagen-like motif, sharing homologies with complement factors and TNF-α[39-42]. It is an adipose tissue-derived protein, exclusively secreted from adipocites[28], capable of stimulating insulin secretion, as well as increasing fatty acid combustion and energy consumption[41]. Evidence exists that adiponectin levels inversely correlate with IR and visceral obesity[42,43] and it has been suggested that low levels of adiponectin may provide a link between obesity, IR, and the risk of CRC[44,45]. In this regard, a protective role of adiponectin for several malignancies, including CRC has been proposed. Since colon epithelium expresses both isoforms of the adiponectin receptor, AdipoR1 and AdipoR2[46], such a protective role might be exerted either directly on cancer cells by affecting signal pathways involved in cell growth and proliferation[47] or indirectly by altering hormone and cytokine levels thus regulating whole-body insulin sensitivity[48].
Leptin, a 167-amino acid peptide in humans, is a product of the Ob gene produced primarily by fat cells, it is involved in the control of food intake and energy expenditure[49,50], and it has proven to be able to regulate cell proliferation in various normal and neoplastic cell types. Indeed, in CRC leptin acts as a potent mitogen[51-53] and antiapoptotic cytokine (Figure 2)[54-57] and promotes the invasiveness of familial adenomatous colonic cells[58]. Accordingly, leptin expression has been reported to be elevated as tumorigenesis progresses[59]. Indeed, it has been shown that leptin expression dramatically increases from normal colonic mucosa to adenoma and adenocarcinoma, suggesting its involvement in multistep colorectal carcinogenesis[60].
Similarly to what reported for other adipokines, leptin is capable of stimulating an angiogenic response in vitro, in a manner that resembles that observed with fibroblast growth factor 2 (FGF-2)[61], thus playing a critical role in the maintenance and regulation of vascular permeability in the adipose tissue[62]. The possible effects of leptin on tumor angiogenesis have not been fully elucidated. Among the raised hypothesis, a paracrine stimulation of leptin-related local new blood vessel formation by leptin receptor-expressing endothelial cells[62,63] and an effect similar to that of IGF-1 gained the highest consents[64].
Resistin is a 12.5-kDa protein of the “resistin-like molecules” (RELMs) family[65], produced by the stromovascular fraction of adipose tissue and peripheral blood monocytes. It has been postulated that a transient upregulation of resistin expression is important for accumulation of intracellular lipid content[66], which has been recently described as a potential factor in obesity-mediated IR, type 2 diabetes and inflammation[67]. Indeed, the involvement of monocytes in resistin production supports the evidence of a role exerted by adipose tissue in the generalized inflammatory process of which monocyte activation is part (Figure 2). Such activity led to suggest that high resistin levels are related to cancer-associated chronic inflammation. To date, it is widely accepted that resistin plays a pivotal role in several inflammatory conditions and in malignancies, such as breast cancer[68] and non-small cell lung cancer[69], however, its role in colorectal carcinogenesis is still not fully elucidated[70].
Ghrelin is a novel orexigenic peptide and represents an endogenous ligand for growth hormone secretagogue receptor type 1a (GHS-R1a)[71]. Ghrelin has a strong stimulatory effect on growth hormone (GH) release, modulates the secretion of other pituitary hormones, participate to glucose homeostasis, stimulates adipogenesis and changes the growth processes of neoplastic tissues[72,73]. Ghrelin exists in the circulation in two molecular forms: acylated (G) and unacylated (UAG). The latter form predominates in circulation and it is active in ghrelin-regulated processes, such as adipogenesis[74] and neoplastic growth influencing proliferation and apoptosis[75,76]. Interestingly, it has been demonstrated that ghrelin may act as either antiapoptotic or pro-apoptotic factor in different cancer cells. Moreover, some cancers possess ghrelin binding sites other than GHS-R1a[75,77]. An alternative hypothesis explaining the role of ghrelin in the development of several neoplasms, including colon cancer, derives by the evidence that ghrelin is a potent regulator of the GH/IGF-I axis, whose inappropriate regulation is known to be positively involved in colon cancer carcinogenesis (Figure 2)[78].
In vitro studies have demonstrated that adiponectin suppresses colon cancer tumor growth and that it negatively correlates with obesity, insulin resistance, immune responses, angiogenesis regulation and intracellular signaling pathways. Adiponectin directly controls cell proliferation, adhesion, invasion and colony formation and regulates metabolic [AMP-activated protein kinase (AMPK)/S6], inflammatory [signal transducer and activator of transcription 3 (STAT3)/VEGF] and cell cycle (p21/p27/p53/cyclins) signaling pathways in both mouse MCA38 and human HT29, HCT116 and LoVo colon cancer cell lines in a LKB1 (a tumor suppressor gene)-dependent way[79]. Moreover, recent experimental evidence suggests that full-length adiponectin is a direct angiogenesis inhibitor that preferentially induces apoptosis in activated endothelial cells in pathological neovascularization[80], whereas globular adiponectin appears to exert opposite effects[81]. Among other cytokines influenced by adiponectin, the reduced TNF-α production in macrophages and its action on endothelial cells, suggests that low levels of this adipokine could potentially lead to carcinogenesis by changing the influence of TNF-α on tumor cell proliferation[47]. Studies have advanced the hypothesis that adiponectin might suppress colon cancer cells through its own receptor mediated AMPK activity (Table 1)[82].
| Adipokine | In vitro evidence | In vivo animal studies | Translational studies | Proposed mechanism(s) |
| Adiponectin | ||||
| Inverse association | Suppression of colon cancer cells by its receptor-mediated AMPK activity[82] | Exogenous adiponectin in ApcMin/+ mice is capable of reducing the growth of intestinal polyps[93]Adiponectin knockout mice are more prone to chronic inflammation-induced colon cancer compared to C57BL/6 wild type mice[94,95] | Inhibition of cell proliferation, adhesion, invasion and colony formation | Reduction of TNF-α levels; regulation of metabolic (AMPK/S6), inflammatory (STAT3/VEGF) and cell cycle (p21/p27/p53/cyclins) signaling pathways[79] |
| Leptin | ||||
| Direct association | Stimulation of the invasive capacity of early neoplastic cells by enhancing cell motility through increased formation of lamellipodial structures[83] | Obese C57BL/6J-Lep (ob) male mice do not display increased tumorigenesis as compared to their lean littermates[97]C57BL/KsJ-db/db obese and diabetic mice have a significant increase in the multiplicity of premalignant lesions[98] | Involvement in CRC carcinogenesis only after the tumor initiation stage[99] Direct correlation with cancer progression[111,112] | Regulation of proliferation and apoptosis through PI3K/AKT/ mTOR signaling pathwayEnhanced expression of VEGF and VEGF-R2, via PI3K, JAK2/STAT3, and ERK1/2 signaling pathways[63] |
| Resistin | ||||
| Direct association | Overexpression in a human colon cancer cell line, LS174T and in the colonic epithelium of many human colon cancer tissues[88] | None reported | Association with the chronic inflammation that leads to cancer pathogenesis[105] Gradual increase with progression in tumor stage[125] | Interference with adipogenesis |
| Ghrelin | ||||
| Conflicting | Induction of apoptosis in human CRC cells[89] Overexpression and enhanced production in malignant intestinal epithelial cells[127] | None reported | Stage-dependent correlation of locally produced autocrine tissue ghrelin with colorectal cancer[127]Gradual decrease with progression in tumor stage[128,129] | Inhibition of the ubiquitin-proteasome system and autophagy activation[90] |
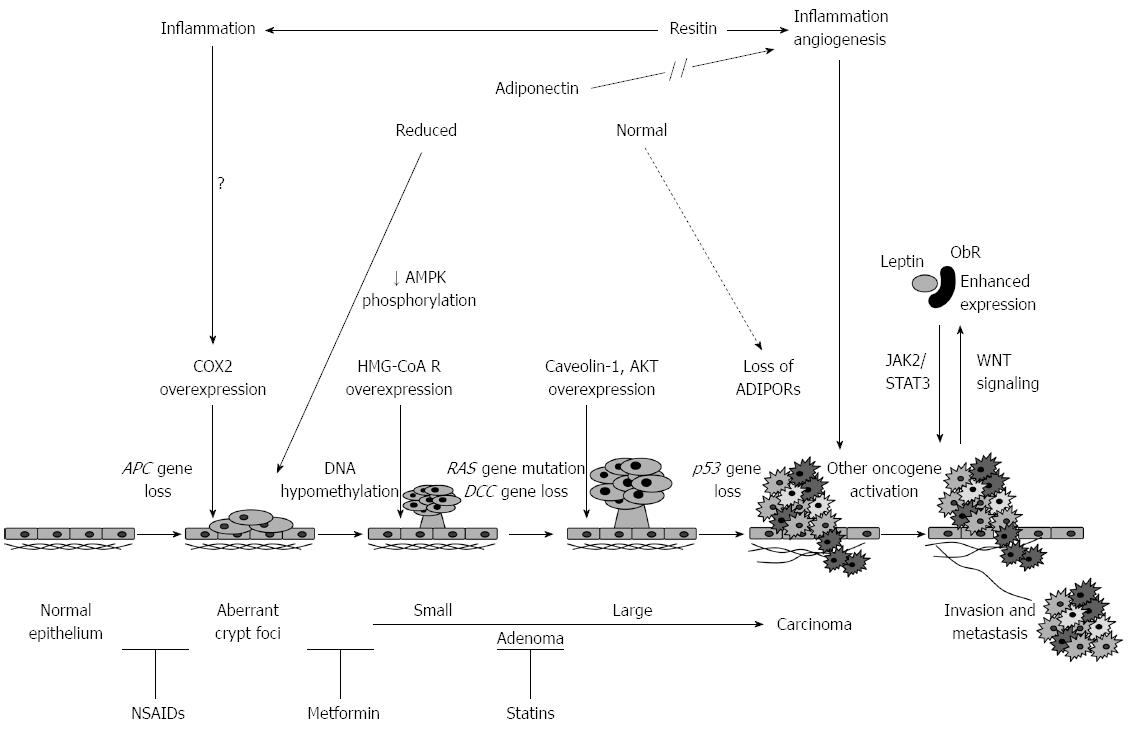
In vitro evidences in support of a role for leptin have been also reported. In particular, leptin has been shown to participate in the malignant behavior of cells stimulating the invasive capacity of early neoplastic cells by enhancing cell motility through increased formation of lamellipodial structures[83] (Table 1). Furthermore, it has been demonstrated that leptin stimulates two oncogenic pathways in colonic adenocarcinoma cell lines, Mitogen-Activated Protein Kinase (MAPK) and NF-κB, with resulting mitosis[64]. Indeed, in the CRC cell line HT-29, leptin (1 nmol/L) treatment increased cell proliferation and p42/44 MAPK phosphorylation, the number of HT-29 cells in S and G2/M phase, cyclin D1 expression in G0/G1 and prevented apoptosis of HT-29 cells, via downstream NF-κB and extracellular signal-related kinase (ERK)1/2 signaling pathways[55]. More recently, by analyzing the association between the regulatory effect of leptin (100 ng/mL) on colon cancer and phosphoinositide kinase-3 (PI3K)/protein kinase B/mammalian target of rapamycin (mTOR) signal transduction, it has been demonstrated that leptin could regulate proliferation and apoptosis of CRC through this signaling pathway[84]. In a human colorectal adenoma-carcinoma sequence it has been observed a role for signal transducer and activator of transcription 3 (STAT3)-mediated leptin signaling through the activation of the long form leptin receptor (ObRL) in colorectal carcinogenesis (Figure 3)[85].
Evidence that leptin stimulates proliferation of colon cells dependent on Apc genotype to induce auto/paracrine signaling cascades of inflammatory mediators and growth factors also arises from experimental models using normal colon epithelial cells, young adult mouse colon cells; Apc+/+ (YAMC), isolated from an immortalized murine cell line model mimicking a genetic mutation in cells linked to stages of carcinogenesis, and Immorto-Min colonic epithelium cells; ApcMin/+ (IMCE), harboring a mutation in Apc, a “gatekeeper” gene linked to human colon cancer. In such a model, the response to leptin at concentrations varying from 0.01 to 50 ng/mL, i.e., a range similar to that detected in mouse serum[86], caused a reduced proliferation and apoptosis in YAMC, and an increased proliferation and inhibition of apoptosis in IMCE[87]. Moreover, beside IMCE proliferation, leptin induced chemokine production, macrophage activation and chemotaxis[88]. These data further highlight leptin-induced cross-talk between preneoplastic epithelial cells and immune cells in the promotional phase of carcinogenesis[88].
If the role of the two former adipokines in colorectal carcinogenesis has been extensively investigated in vitro, only scattered information is available regarding the biochemical mechanisms linking resistin and ghrelin to CRC (Table 1). As regards the family of tissue-specific resistin-like molecules, RELMβ has been found to be over-expressed in a human colon cancer cell line, LS174T and in the colonic epithelium of most human colon cancer tissues analyzed[89]. In a recent study[90] the direct inhibitory effects of both forms of ghrelin (G and UAG) and of the ghrelin receptor type 1a antagonist (D-Lys-GHRP-6; GHS-RA), applied alone or jointly, were analyzed on the growth of colon and prostate cancer cell lines (murine colon cancer MC38 and human prostate cancer DU145), demonstrating an involvement of the ghrelin axis in the growth regulation of colon and prostate cancers. Indeed, the different members of the ghrelin axis, and the ghrelin receptor type 1a antagonist, affected the growth of MC38 colon cancer and DU145 PCa cell lines with diverse potencies, depending on the type of the cancer cell line, the applied substance and the concentration used[90]. More recent evidences indicated that ghrelin (1 μmol/L) induced apoptosis in human colorectal carcinoma cells (HCT116 cells) by inhibiting the ubiquitin-proteasome system and by activating autophagy, two proteolytic pathways strictly related to cell cycle regulation and cell death[91].
Diet-induced obesity mouse models have been employed to verify the epidemiological data linking obesity to colon cancer development. Recent studies aimed at determining whether wild-type diet-induced obese (DIO) mice were at greater risk of colon cancer than their lean male littermates, indicated that DIO mice showed significantly more aberrant crypts and aberrant crypt foci as well as increased proliferation of colonocytes per mouse compared to wild-type control mice, when injected with the procarcinogen azoxymethane[92]. In a similar setting, mice with disruptions in APN developed more intestinal tumors, compared with wild-type mice[93].
At the same time, other studies have used different animal models to gain evidence on the association between adiposopathy and CRC (Table 1). In this context, it has been demonstrated that exogenous administration of adiponectin in a ApcMin/+ mouse model of intestinal carcinogenesis is capable of reducing the growth of intestinal polyps[94] and that adiponectin knockout mice are more prone to chronic inflammation-induced colon cancer compared to wild type mice[95,96].
The idea that adiponectin deficiency contributes to inflammation-induced colon cancer, was further supported by the finding that, when inflammation and colon cancer were induced in both adiponectin knockout and C57BL/6 wild type mice, adiponectin proved to play an important role in CRC prevention by modulating genes involved in chronic inflammation and tumorigenesis[95]. Finally, in adiponectin-deficient mice or wild type mice, fed with either a high-fat (HF) or a low-fat (LF) diet, adiponectin administration suppressed the increase in epithelial cell proliferation[97] and the implanted tumor growth, causing larger central necrotic areas[79]. Moreover, adiponectin treatment suppressed expression of angiogenic factors (CD31, VEGFb and VEGFd) in tumors obtained from all HF mice and from adiponectin-deficient mice either on HF or on LF diet, decreased serum insulin levels in HF mice and increased serum IL-12 levels in all adiponectin-deficient mice[79].
In analogy to what has been shown for adiponectin, the role exerted by leptin in the increased risk of colon cancer associated with obesity has been highlighted by demonstrating that obese C57BL/6J-Lepob male mice, deficient in leptin, did not show signs of increased tumorigenesis, as compared to their lean littermates, suggesting that leptin might serve as a key hormone mediating the increased risk of colonic tumorigenesis associated with obesity[98]. These data further support previous findings that in C57BL/KsJ-db/db obese and diabetic mice with hyperleptinemia and hyperinsulinemia due to disruption of the leptin receptor, a significant increase in the multiplicity of premalignant lesions was found, as compared to their lean littermates[99]. Of interest, in another animal model of syngeneic mice, leptin was also shown to enhance the expression of VEGF and VEGF-R2, via PI3K, Janus kinases 2 (JAK2)/STAT3, and ERK1/2 signaling pathways, thus suggesting its possible role in stimulating tumor neoangiogenesis[63]. Finally, in experimental studies on leptin-deficient and leptin receptor (ObR)-deficient models of obesity, a significant decrease of tumor cell proliferation was observed in leptin-deficient tumors, concomitantly with a higher ObR expression levels in the colon tumors as compared to normal epithelium (Figure 3)[100]. In the colorectal adenoma (CRA) tissues, a significant increase in the phosphorylation level of the JAK/STAT signaling pathway molecules, and transcriptional regulation of STAT3-downstream target molecules were observed, and in colon tumors the proliferative and survival effects of leptin were found to be mediated by ObRL/STAT3 signaling[100]. Accordingly, tumor growth was dramatically inhibited in leptin-deficient and leptin-receptor-deficient mice despite their severe obesity[100]. Interestingly, it seems that leptin stimulates the proliferation of tumor cells in which Wnt signaling pathway is activated (Figure 3)[100].
As regards resistin, it has been demonstrated that in primary preadipocytes derived from Zucker obese rats, adipokine expression differed significantly in visceral adipose tissue as compared to lean animals[101]. Indeed, while resistin showed a characteristic expression profile during differentiation and maturation of 3T3-L1 cells and primary preadipocytes, its expression was lower in the visceral adipose tissue of Zucker obese rats[101] suggesting that this adipokine strongly influences adipogenesis of 3T3-L1 cells in the early maturation period[66].
Several clinical and epidemiological studies have demonstrated that altered circulating levels of adipokines are inversely correlated with the risk of CRC (Table 1)[99-117]. Results from meta-analyses suggest that changes in the levels of adipocytokines may indicate the initiation and progression of CRC and adenoma but conflicting results have been reported in other studies[107-109]. Decreased concentrations of plasma adiponectin have been associated with the development of colon adenomas in Japanese patients[118], the association being particularly significant with the number/size of tumors and histological progression from tubular to tubulovillous/villous adenomas[118]. A large prospective study demonstrated that men with low plasma adiponectin levels had a higher risk of CRC than men with higher levels, the association being independent of BMI, waist circumference, WHR and physical activity[102]. Furthermore, several case-control studies[104-106] have confirmed the occurrence of lower adiponectin levels in patients with CRC than in healthy controls and it has been recently suggested that adiponectin might represent a prognostic parameter in risk prediction for CRC recurrence[104,119]. In support of the hypothesis that adiponectin can inhibit the growth of CRC, immunostaining studies demonstrated an inverse correlation between the T stage of CRC and the grade of adiponectin receptors, AdipoR1 and AdipoR2, expression[120]. Indeed, abundant expression of adiponectin receptors in CRC tissue may facilitate the anticarcinogenic effect of adiponectin; conversely, low expression levels of AdipoRs may promote progression of CRC by counteracting the protective effects of adiponectin (Figure 3)[120].
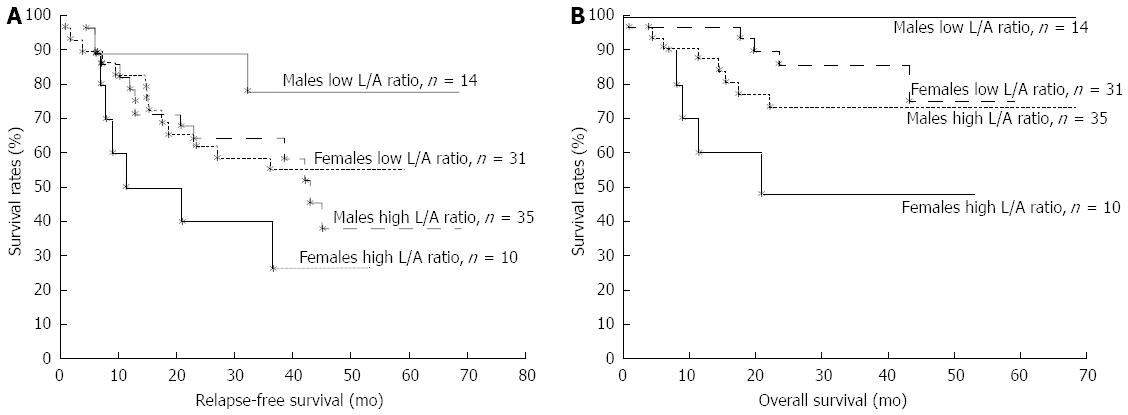
Recent studies exploring high-molecular weight (HMW) adiponectin, which is involved in insulin sensitivity regulation and non-HMW adiponectin fractions in relation to CRC risk, showed that, when stratified by cancer site, non-HMW adiponectin was inversely associated with both colon and rectal cancer, suggesting an important role of the relative proportion of non-HMW adiponectin in CRC pathogenesis[121]. Meanwhile, evidence from a large prospective study evaluating the association of plasma adiponectin and soluble leptin receptor (sObR) with CRC risk demonstrated that plasma adiponectin was significantly associated with reduced risk of CRC among men, but not among women and that sObR was significantly associated with increased risk of rectal cancer but not colon cancer[122]. Recently, the hypothesis that an unfavorable adipokine profile [as indicated by a high leptin/adiponectin (L/A) ratio] might serve as a prognostic factor in CRC patients has been proposed[109,119]. In particular, the evidence that a high L/A ratio had a negative prognostic value with respect to both disease-free and overall survival in CRC patients suggested that combined measurement of both adipokines may represent an adjunctive tool in risk prediction for CRC recurrence[119]. Of interest, the negative prognostic value of L/A ratio was independent of gender (Figure 4)[119], contrarily to what previously demonstrated for adiponectin alone[122].
Leptin is also directly correlated with the risk of CRC[112,113] and a more aggressive tumor phenotype, as demonstrated by the association with stage[114,119], microvascular invasion[115] or decreased relapse free survival[119], although low or undetectable leptin concentrations were observed in other studies[116,117,123]. It appears that leptin is not involved in the early-stage CRC carcinogenesis but, rather, only after the tumor initiation stage[100]. As regards the sex-specific differences observed in relating leptin to CRC risk, there is no agreement, since some Authors reported an association in men but not in women[111,112], while others found an association also in women[113].
Resistin is another adipokine that has been recently proposed as an independent predictor of CRC[106,112,124], independently of different anthropometric measures of adiposity, such as WC[124-127]. Indeed, as no association between resistin and IR has been detected, the hypothesis that resistin may play a role in CRC independently of obesity, but due to its close association with the chronic inflammation that leads to cancer pathogenesis has been advanced[106]. In this regard, although resistin levels positively correlated with inflammatory markers, association with tumor stage is debated. In fact, while some studies reported no differences between early and late stages CRC[125], Nakajima et al[126] showed that resistin levels gradually increased with progression in tumor stage. Similarly, no study, so far, has reported any association between resistin and age, sex, BMI or lifestyle parameters, rather than C-reactive protein levels[127]. Notably, enhanced expression of tissue-specific resistin-like molecules RELMβ has been positively correlated with the differentiation degree of colon adenocarcinoma and mucinous carcinoma, while loss of expression was observed in undifferentiated carcinoma[89]. Based on these findings, it has also been suggested that, independently from BMI, resistin may be a good biomarker of CRC, while adiponectin may be superior in CRA[126].
As regards the role of ghrelin axis in CRC development, studies on CRC progression showed that malignant intestinal epithelial cells differentially overexpress ghrelin receptors and produce more ghrelin as compared to normal human colonocytes, thus causing their enhanced proliferative and invasive behavior[128]. Contrasting results were obtained when comparing locally produced autocrine ghrelin with its sistemically available form. In support of the role of ghrelin in promoting malignancy in CRC patients, it has been demonstrated that while locally produced autocrine ghrelin correlated with CRC in a stage-dependent manner, systemically available endocrine ghrelin levels did not exhibit significant correlation with any tumor stage or grade[128]. These data were in contrast with an earlier study in which ghrelin serum levels showed a decreasing pattern from early to advanced stage of disease in CRC patients[129,130].
Many attempts at developing prediction models to identify those patients at higher risk to develop colon cancer (reviewed in[131]), in order to provide clear indication for prevention, diagnosis and treatment have been performed. The vast majority of the epidemiological evidence clearly indicates mutual factors linking obesity and colon cancer[132]. Among the proposed risk factors, increased consumption of high-energy diets and reduced consumption of fruit, vegetables and fibre, a sedentary lifestyle and increased age, have proven to influence adipose tissue functionality and to provide a common soil for a strong link between obesity-driven chronic inflammation and CRC[133]. Reversing obesity-associated inflammation and adiposopathy by lifestyle interventions, including weight loss, physical activity and dietary modifications, might have a clinically relevant role in reducing CRC risk or progression[134-136] and survival outcomes of patients with recurrent colon cancer[137]. The mechanistic and pathophysiology studies on adipokines highlight the important role of these adipocyte-secreted hormones in colon cancer. Literature data agree in indicating that adiponectin or analogues might become useful agents in the management or chemoprevention of CRC[79]. In conclusion, dissecting the mechanisms underlying adipokine involvement in obesity-driven CRC will be of utmost importance in risk reduction and design of tailored therapies to prevent chemoresistance and recurrence.
P- Reviewers: Coleman HG, Crea F, Leitman IM, Liu X S- Editor: Gou SX L- Editor: A E- Editor: Ma S
| 1. | Moghaddam AA, Woodward M, Huxley R. Obesity and risk of colorectal cancer: a meta-analysis of 31 studies with 70,000 events. Cancer Epidemiol Biomarkers Prev. 2007;16:2533-2547. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 431] [Cited by in RCA: 450] [Article Influence: 26.5] [Reference Citation Analysis (0)] |
| 2. | Larsson SC, Wolk A. Obesity and colon and rectal cancer risk: a meta-analysis of prospective studies. Am J Clin Nutr. 2007;86:556-565. [PubMed] |
| 3. | Lukanova A, Björ O, Kaaks R, Lenner P, Lindahl B, Hallmans G, Stattin P. Body mass index and cancer: results from the Northern Sweden Health and Disease Cohort. Int J Cancer. 2006;118:458-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 122] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 4. | MacInnis RJ, English DR, Haydon AM, Hopper JL, Gertig DM, Giles GG. Body size and composition and risk of rectal cancer (Australia). Cancer Causes Control. 2006;17:1291-1297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 5. | Pischon T, Lahmann PH, Boeing H, Friedenreich C, Norat T, Tjønneland A, Halkjaer J, Overvad K, Clavel-Chapelon F, Boutron-Ruault MC. Body size and risk of colon and rectal cancer in the European Prospective Investigation Into Cancer and Nutrition (EPIC). J Natl Cancer Inst. 2006;98:920-931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 384] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 6. | Renehan AG, Tyson M, Egger M, Heller RF, Zwahlen M. Body-mass index and incidence of cancer: a systematic review and meta-analysis of prospective observational studies. Lancet. 2008;371:569-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3505] [Cited by in RCA: 3681] [Article Influence: 216.5] [Reference Citation Analysis (1)] |
| 7. | Renehan AG, Soerjomataram I, Tyson M, Egger M, Zwahlen M, Coebergh JW, Buchan I. Incident cancer burden attributable to excess body mass index in 30 European countries. Int J Cancer. 2010;126:692-702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 196] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 8. | Aleksandrova K, Pischon T, Buijsse B, May AM, Peeters PH, Bueno-de-Mesquita HB, Jenab M, Fedirko V, Dahm CC, Siersema PD. Adult weight change and risk of colorectal cancer in the European Prospective Investigation into Cancer and Nutrition. Eur J Cancer. 2013;49:3526-3536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 53] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 9. | Fernández-Sánchez A, Madrigal-Santillán E, Bautista M, Esquivel-Soto J, Morales-González A, Esquivel-Chirino C, Durante-Montiel I, Sánchez-Rivera G, Valadez-Vega C, Morales-González JA. Inflammation, oxidative stress, and obesity. Int J Mol Sci. 2011;12:3117-3132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1127] [Cited by in RCA: 1012] [Article Influence: 72.3] [Reference Citation Analysis (0)] |
| 10. | Orgel E, Mittelman SD. The links between insulin resistance, diabetes, and cancer. Curr Diab Rep. 2013;13:213-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 125] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 11. | Renehan AG, Zwahlen M, Minder C, O’Dwyer ST, Shalet SM, Egger M. Insulin-like growth factor (IGF)-I, IGF binding protein-3, and cancer risk: systematic review and meta-regression analysis. Lancet. 2004;363:1346-1353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1246] [Cited by in RCA: 1236] [Article Influence: 58.9] [Reference Citation Analysis (0)] |
| 12. | Bardou M, Barkun AN, Martel M. Republished: obesity and colorectal cancer. Postgrad Med J. 2013;89:519-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 13. | Wozniak SE, Gee LL, Wachtel MS, Frezza EE. Adipose tissue: the new endocrine organ? A review article. Dig Dis Sci. 2009;54:1847-1856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 359] [Cited by in RCA: 354] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 14. | Fontana L, Eagon JC, Trujillo ME, Scherer PE, Klein S. Visceral fat adipokine secretion is associated with systemic inflammation in obese humans. Diabetes. 2007;56:1010-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1059] [Cited by in RCA: 991] [Article Influence: 55.1] [Reference Citation Analysis (0)] |
| 15. | Cypess AM, Lehman S, Williams G, Tal I, Rodman D, Goldfine AB, Kuo FC, Palmer EL, Tseng YH, Doria A. Identification and importance of brown adipose tissue in adult humans. N Engl J Med. 2009;360:1509-1517. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3598] [Cited by in RCA: 3367] [Article Influence: 210.4] [Reference Citation Analysis (0)] |
| 16. | Bays HE, Toth PP, Kris-Etherton PM, Abate N, Aronne LJ, Brown WV, Gonzalez-Campoy JM, Jones SR, Kumar R, La Forge R. Obesity, adiposity, and dyslipidemia: a consensus statement from the National Lipid Association. J Clin Lipidol. 2013;7:304-383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 327] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 18. | Bays HE, González-Campoy JM, Henry RR, Bergman DA, Kitabchi AE, Schorr AB, Rodbard HW. Is adiposopathy (sick fat) an endocrine disease? Int J Clin Pract. 2008;62:1474-1483. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 64] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 19. | Bays H. Adiposopathy: role of adipocyte factors in a new paradigm. Expert Rev Cardiovasc Ther. 2005;3:187-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 20. | Harvey AE, Lashinger LM, Hursting SD. The growing challenge of obesity and cancer: an inflammatory issue. Ann N Y Acad Sci. 2011;1229:45-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 205] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 21. | Kursawe R, Eszlinger M, Narayan D, Liu T, Bazuine M, Cali AM, D’Adamo E, Shaw M, Pierpont B, Shulman GI. Cellularity and adipogenic profile of the abdominal subcutaneous adipose tissue from obese adolescents: association with insulin resistance and hepatic steatosis. Diabetes. 2010;59:2288-2296. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 105] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 22. | O’Rourke RW. Inflammation in obesity-related diseases. Surgery. 2009;145:255-259. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 101] [Cited by in RCA: 100] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 23. | Rizzo A, Pallone F, Monteleone G, Fantini MC. Intestinal inflammation and colorectal cancer: a double-edged sword? World J Gastroenterol. 2011;17:3092-3100. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 56] [Reference Citation Analysis (0)] |
| 24. | Cousin B, Munoz O, Andre M, Fontanilles AM, Dani C, Cousin JL, Laharrague P, Casteilla L, Pénicaud L. A role for preadipocytes as macrophage-like cells. FASEB J. 1999;13:305-312. [PubMed] |
| 25. | Dalmas E, Clément K, Guerre-Millo M. Defining macrophage phenotype and function in adipose tissue. Trends Immunol. 2011;32:307-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 177] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 26. | Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW. Obesity is associated with macrophage accumulation in adipose tissue. J Clin Invest. 2003;112:1796-1808. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 3626] [Article Influence: 172.7] [Reference Citation Analysis (0)] |
| 27. | Lumeng CN, Bodzin JL, Saltiel AR. Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J Clin Invest. 2007;117:175-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3164] [Cited by in RCA: 3579] [Article Influence: 198.8] [Reference Citation Analysis (0)] |
| 28. | Hao NB, Lü MH, Fan YH, Cao YL, Zhang ZR, Yang SM. Macrophages in tumor microenvironments and the progression of tumors. Clin Dev Immunol. 2012;2012:948098. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 499] [Cited by in RCA: 688] [Article Influence: 52.9] [Reference Citation Analysis (0)] |
| 29. | Suganami T, Nishida J, Ogawa Y. A paracrine loop between adipocytes and macrophages aggravates inflammatory changes: role of free fatty acids and tumor necrosis factor alpha. Arterioscler Thromb Vasc Biol. 2005;25:2062-2068. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 779] [Cited by in RCA: 791] [Article Influence: 39.6] [Reference Citation Analysis (0)] |
| 30. | Huang XF, Chen JZ. Obesity, the PI3K/Akt signal pathway and colon cancer. Obes Rev. 2009;10:610-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 159] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 31. | Yehuda-Shnaidman E, Schwartz B. Mechanisms linking obesity, inflammation and altered metabolism to colon carcinogenesis. Obes Rev. 2012;13:1083-1095. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 91] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 32. | Dirat B, Bochet L, Escourrou G, Valet P, Muller C. Unraveling the obesity and breast cancer links: a role for cancer-associated adipocytes? Endocr Dev. 2010;19:45-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 82] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 33. | Ramos-Nino ME. The role of chronic inflammation in obesity-associated cancers. ISRN Oncol. 2013;2013:697521. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 34. | Braun S, Bitton-Worms K, LeRoith D. The link between the metabolic syndrome and cancer. Int J Biol Sci. 2011;7:1003-1015. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 179] [Cited by in RCA: 198] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 35. | Zick Y. Role of Ser/Thr kinases in the uncoupling of insulin signaling. Int J Obes Relat Metab Disord. 2003;27 Suppl 3:S56-S60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 60] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 36. | Shoelson SE, Lee J, Yuan M. Inflammation and the IKK beta/I kappa B/NF-kappa B axis in obesity- and diet-induced insulin resistance. Int J Obes Relat Metab Disord. 2003;27 Suppl 3:S49-S52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 326] [Cited by in RCA: 344] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 37. | Brownlee M. Biochemistry and molecular cell biology of diabetic complications. Nature. 2001;414:813-820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6145] [Cited by in RCA: 6205] [Article Influence: 258.5] [Reference Citation Analysis (0)] |
| 38. | Kant P, Hull MA. Excess body weight and obesity--the link with gastrointestinal and hepatobiliary cancer. Nat Rev Gastroenterol Hepatol. 2011;8:224-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 87] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 39. | Scherer PE, Williams S, Fogliano M, Baldini G, Lodish HF. A novel serum protein similar to C1q, produced exclusively in adipocytes. J Biol Chem. 1995;270:26746-26749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2259] [Cited by in RCA: 2303] [Article Influence: 76.8] [Reference Citation Analysis (0)] |
| 40. | Hu E, Liang P, Spiegelman BM. AdipoQ is a novel adipose-specific gene dysregulated in obesity. J Biol Chem. 1996;271:10697-10703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1581] [Cited by in RCA: 1572] [Article Influence: 54.2] [Reference Citation Analysis (0)] |
| 41. | Kadowaki T, Yamauchi T, Kubota N, Hara K, Ueki K, Tobe K. Adiponectin and adiponectin receptors in insulin resistance, diabetes, and the metabolic syndrome. J Clin Invest. 2006;116:1784-1792. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1945] [Cited by in RCA: 2062] [Article Influence: 108.5] [Reference Citation Analysis (0)] |
| 42. | Nakashima R, Kamei N, Yamane K, Nakanishi S, Nakashima A, Kohno N. Decreased total and high molecular weight adiponectin are independent risk factors for the development of type 2 diabetes in Japanese-Americans. J Clin Endocrinol Metab. 2006;91:3873-3877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 130] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 43. | Hanley AJ, Bowden D, Wagenknecht LE, Balasubramanyam A, Langfeld C, Saad MF, Rotter JI, Guo X, Chen YD, Bryer-Ash M. Associations of adiponectin with body fat distribution and insulin sensitivity in nondiabetic Hispanics and African-Americans. J Clin Endocrinol Metab. 2007;92:2665-2671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 100] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 44. | Scheid MP, Sweeney G. The role of adiponectin signaling in metabolic syndrome and cancer. Rev Endocr Metab Disord. 2013;Sep 10; Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 45. | Xu Y, He B, Pan Y, Gu L, Nie Z, Chen L, Li R, Gao T, Wang S. The roles of ADIPOQ genetic variations in cancer risk: evidence from published studies. Mol Biol Rep. 2013;40:1135-1144. [PubMed] |
| 46. | Drew JE, Farquharson AJ, Padidar S, Duthie GG, Mercer JG, Arthur JR, Morrice PC, Barrera LN. Insulin, leptin, and adiponectin receptors in colon: regulation relative to differing body adiposity independent of diet and in response to dimethylhydrazine. Am J Physiol Gastrointest Liver Physiol. 2007;293:G682-G691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 47. | Ouchi N, Kihara S, Arita Y, Nishida M, Matsuyama A, Okamoto Y, Ishigami M, Kuriyama H, Kishida K, Nishizawa H. Adipocyte-derived plasma protein, adiponectin, suppresses lipid accumulation and class A scavenger receptor expression in human monocyte-derived macrophages. Circulation. 2001;103:1057-1063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1551] [Cited by in RCA: 1540] [Article Influence: 59.2] [Reference Citation Analysis (0)] |
| 48. | Gialamas SP, Petridou ET, Tseleni-Balafouta S, Spyridopoulos TN, Matsoukis IL, Kondi-Pafiti A, Zografos G, Mantzoros CS. Serum adiponectin levels and tissue expression of adiponectin receptors are associated with risk, stage, and grade of colorectal cancer. Metabolism. 2011;60:1530-1538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 62] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 49. | Koerner A, Kratzsch J, Kiess W. Adipocytokines: leptin--the classical, resistin--the controversical, adiponectin--the promising, and more to come. Best Pract Res Clin Endocrinol Metab. 2005;19:525-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 308] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 50. | Zhang Y, Proenca R, Maffei M, Barone M, Leopold L, Friedman JM. Positional cloning of the mouse obese gene and its human homologue. Nature. 1994;372:425-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9119] [Cited by in RCA: 8855] [Article Influence: 285.6] [Reference Citation Analysis (0)] |
| 51. | Somasundar P, Riggs D, Jackson B, Vona-Davis L, McFadden DW. Leptin stimulates esophageal adenocarcinoma growth by nonapoptotic mechanisms. Am J Surg. 2003;186:575-578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 38] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 52. | Pai R, Lin C, Tran T, Tarnawski A. Leptin activates STAT and ERK2 pathways and induces gastric cancer cell proliferation. Biochem Biophys Res Commun. 2005;331:984-992. [PubMed] |
| 53. | Aparicio T, Kotelevets L, Tsocas A, Laigneau JP, Sobhani I, Chastre E, Lehy T. Leptin stimulates the proliferation of human colon cancer cells in vitro but does not promote the growth of colon cancer xenografts in nude mice or intestinal tumorigenesis in Apc(Min/+) mice. Gut. 2005;54:1136-1145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 79] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 54. | Hardwick JC, Van Den Brink GR, Offerhaus GJ, Van Deventer SJ, Peppelenbosch MP. Leptin is a growth factor for colonic epithelial cells. Gastroenterology. 2001;121:79-90. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 256] [Cited by in RCA: 258] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 55. | Rouet-Benzineb P, Aparicio T, Guilmeau S, Pouzet C, Descatoire V, Buyse M, Bado A. Leptin counteracts sodium butyrate-induced apoptosis in human colon cancer HT-29 cells via NF-kappaB signaling. J Biol Chem. 2004;279:16495-16502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 105] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 56. | Hoda MR, Keely SJ, Bertelsen LS, Junger WG, Dharmasena D, Barrett KE. Leptin acts as a mitogenic and antiapoptotic factor for colonic cancer cells. Br J Surg. 2007;94:346-354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 87] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 57. | Amemori S, Ootani A, Aoki S, Fujise T, Shimoda R, Kakimoto T, Shiraishi R, Sakata Y, Tsunada S, Iwakiri R. Adipocytes and preadipocytes promote the proliferation of colon cancer cells in vitro. Am J Physiol Gastrointest Liver Physiol. 2007;292:G923-G929. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 93] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 58. | Attoub S, Noe V, Pirola L, Bruyneel E, Chastre E, Mareel M, Wymann MP, Gespach C. Leptin promotes invasiveness of kidney and colonic epithelial cells via phosphoinositide 3-kinase-, rho-, and rac-dependent signaling pathways. FASEB J. 2000;14:2329-2338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 183] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 59. | Koda M, Sulkowska M, Kanczuga-Koda L, Surmacz E, Sulkowski S. Overexpression of the obesity hormone leptin in human colorectal cancer. J Clin Pathol. 2007;60:902-906. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 82] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 60. | Paik SS, Jang SM, Jang KS, Lee KH, Choi D, Jang SJ. Leptin expression correlates with favorable clinicopathologic phenotype and better prognosis in colorectal adenocarcinoma. Ann Surg Oncol. 2009;16:297-303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 56] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 61. | Ribatti D, Nico B, Belloni AS, Vacca A, Roncali L, Nussdorfer GG. Angiogenic activity of leptin in the chick embryo chorioallantoic membrane is in part mediated by endogenous fibroblast growth factor-2. Int J Mol Med. 2001;8:265-268. [PubMed] |
| 62. | Vona-Davis L, Rose DP. Angiogenesis, adipokines and breast cancer. Cytokine Growth Factor Rev. 2009;20:193-201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 119] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 63. | Gonzalez RR, Cherfils S, Escobar M, Yoo JH, Carino C, Styer AK, Sullivan BT, Sakamoto H, Olawaiye A, Serikawa T. Leptin signaling promotes the growth of mammary tumors and increases the expression of vascular endothelial growth factor (VEGF) and its receptor type two (VEGF-R2). J Biol Chem. 2006;281:26320-26328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 185] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 64. | Sikalidis AK, Varamini B. Roles of hormones and signaling molecules in describing the relationship between obesity and colon cancer. Pathol Oncol Res. 2011;17:785-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 65. | Steppan CM, Bailey ST, Bhat S, Brown EJ, Banerjee RR, Wright CM, Patel HR, Ahima RS, Lazar MA. The hormone resistin links obesity to diabetes. Nature. 2001;409:307-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3205] [Cited by in RCA: 3212] [Article Influence: 133.8] [Reference Citation Analysis (1)] |
| 66. | Ikeda Y, Tsuchiya H, Hama S, Kajimoto K, Kogure K. Resistin affects lipid metabolism during adipocyte maturation of 3T3-L1 cells. FEBS J. 2013;280:5884-5895. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 67. | Mojiminiyi OA, Abdella NA. Associations of resistin with inflammation and insulin resistance in patients with type 2 diabetes mellitus. Scand J Clin Lab Invest. 2007;67:215-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 68. | Filková M, Haluzík M, Gay S, Senolt L. The role of resistin as a regulator of inflammation: Implications for various human pathologies. Clin Immunol. 2009;133:157-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 318] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 69. | Karapanagiotou EM, Tsochatzis EA, Dilana KD, Tourkantonis I, Gratsias I, Syrigos KN. The significance of leptin, adiponectin, and resistin serum levels in non-small cell lung cancer (NSCLC). Lung Cancer. 2008;61:391-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 61] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 70. | Tiaka EK, Manolakis AC, Kapsoritakis AN, Potamianos SP. The implication of adiponectin and resistin in gastrointestinal diseases. Cytokine Growth Factor Rev. 2011;22:109-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 71. | Kojima M, Hosoda H, Date Y, Nakazato M, Matsuo H, Kangawa K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature. 1999;402:656-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5961] [Cited by in RCA: 5889] [Article Influence: 226.5] [Reference Citation Analysis (0)] |
| 72. | Baragli A, Lanfranco F, Allasia S, Granata R, Ghigo E. Neuroendocrine and metabolic activities of ghrelin gene products. Peptides. 2011;32:2323-2332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 73. | Jeffery PL, Herington AC, Chopin LK. The potential autocrine/paracrine roles of ghrelin and its receptor in hormone-dependent cancer. Cytokine Growth Factor Rev. 2003;14:113-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 132] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 74. | Thompson NM, Gill DA, Davies R, Loveridge N, Houston PA, Robinson IC, Wells T. Ghrelin and des-octanoyl ghrelin promote adipogenesis directly in vivo by a mechanism independent of the type 1a growth hormone secretagogue receptor. Endocrinology. 2004;145:234-242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 299] [Cited by in RCA: 286] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 75. | Cassoni P, Ghé C, Marrocco T, Tarabra E, Allia E, Catapano F, Deghenghi R, Ghigo E, Papotti M, Muccioli G. Expression of ghrelin and biological activity of specific receptors for ghrelin and des-acyl ghrelin in human prostate neoplasms and related cell lines. Eur J Endocrinol. 2004;150:173-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 158] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 76. | Joint statement of the role of the registered nurse in the care of the patient with cardiovascular diseases in coronary care or intensive care units; approved by the Maryland Nurses Association. Md Nurs News. 1967;36:43-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 397] [Cited by in RCA: 397] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 77. | Barzon L, Pacenti M, Masi G, Stefani AL, Fincati K, Palù G. Loss of growth hormone secretagogue receptor 1a and overexpression of type 1b receptor transcripts in human adrenocortical tumors. Oncology. 2005;68:414-421. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 78. | Bustin SA, Jenkins PJ. The growth hormone-insulin-like growth factor-I axis and colorectal cancer. Trends Mol Med. 2001;7:447-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 61] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 79. | Moon HS, Liu X, Nagel JM, Chamberland JP, Diakopoulos KN, Brinkoetter MT, Hatziapostolou M, Wu Y, Robson SC, Iliopoulos D. Salutary effects of adiponectin on colon cancer: in vivo and in vitro studies in mice. Gut. 2013;62:561-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 77] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 80. | Bråkenhielm E, Veitonmäki N, Cao R, Kihara S, Matsuzawa Y, Zhivotovsky B, Funahashi T, Cao Y. Adiponectin-induced antiangiogenesis and antitumor activity involve caspase-mediated endothelial cell apoptosis. Proc Natl Acad Sci USA. 2004;101:2476-2481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 511] [Cited by in RCA: 533] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 81. | Adya R, Tan BK, Chen J, Randeva HS. Protective actions of globular and full-length adiponectin on human endothelial cells: novel insights into adiponectin-induced angiogenesis. J Vasc Res. 2012;49:534-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 52] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 82. | Kim AY, Lee YS, Kim KH, Lee JH, Lee HK, Jang SH, Kim SE, Lee GY, Lee JW, Jung SA. Adiponectin represses colon cancer cell proliferation via AdipoR1- and -R2-mediated AMPK activation. Mol Endocrinol. 2010;24:1441-1452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 172] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 83. | Jaffe T, Schwartz B. Leptin promotes motility and invasiveness in human colon cancer cells by activating multiple signal-transduction pathways. Int J Cancer. 2008;123:2543-2556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 100] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 84. | Wang D, Chen J, Chen H, Duan Z, Xu Q, Wei M, Wang L, Zhong M. Leptin regulates proliferation and apoptosis of colorectal carcinoma through PI3K/Akt/mTOR signalling pathway. J Biosci. 2012;37:91-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 93] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 85. | Uchiyama T, Takahashi H, Endo H, Sugiyama M, Sakai E, Hosono K, Nagashima Y, Inayama Y, Wada K, Hippo Y. Role of the long form leptin receptor and of the STAT3 signaling pathway in colorectal cancer progression. Int J Oncol. 2011;39:935-940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 86. | Sarraf P, Frederich RC, Turner EM, Ma G, Jaskowiak NT, Rivet DJ, Flier JS, Lowell BB, Fraker DL, Alexander HR. Multiple cytokines and acute inflammation raise mouse leptin levels: potential role in inflammatory anorexia. J Exp Med. 1997;185:171-175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 568] [Cited by in RCA: 557] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 87. | Fenton JI, Hord NG. Stage matters: choosing relevant model systems to address hypotheses in diet and cancer chemoprevention research. Carcinogenesis. 2006;27:893-902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 88. | Fenton JI, Hursting SD, Perkins SN, Hord NG. Leptin induces an Apc genotype-associated colon epithelial cell chemokine production pattern associated with macrophage chemotaxis and activation. Carcinogenesis. 2007;28:455-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 89. | Zheng LD, Tong QS, Weng MX, He J, Lv Q, Pu JR, Jiang GS, Cai JB, Liu Y, Hou XH. Enhanced expression of resistin-like molecule beta in human colon cancer and its clinical significance. Dig Dis Sci. 2009;54:274-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 33] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 90. | Lawnicka H, Mełeń-Mucha G, Motylewska E, Mucha S, Stępień H. Modulation of ghrelin axis influences the growth of colonic and prostatic cancer cells in vitro. Pharmacol Rep. 2012;64:951-959. [PubMed] |
| 91. | Bonfili L, Cuccioloni M, Cecarini V, Mozzicafreddo M, Palermo FA, Cocci P, Angeletti M, Eleuteri AM. Ghrelin induces apoptosis in colon adenocarcinoma cells via proteasome inhibition and autophagy induction. Apoptosis. 2013;18:1188-1200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 92. | Sikalidis AK, Fitch MD, Fleming SE. Diet induced obesity increases the risk of colonic tumorigenesis in mice. Pathol Oncol Res. 2013;19:657-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 93. | Mutoh M, Teraoka N, Takasu S, Takahashi M, Onuma K, Yamamoto M, Kubota N, Iseki T, Kadowaki T, Sugimura T. Loss of adiponectin promotes intestinal carcinogenesis in Min and wild-type mice. Gastroenterology. 2011;140:2000-208, 2000-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 57] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 94. | Otani K, Kitayama J, Yasuda K, Nio Y, Iwabu M, Okudaira S, Aoki J, Yamauchi T, Kadowaki T, Nagawa H. Adiponectin suppresses tumorigenesis in Apc(Min)(/+) mice. Cancer Lett. 2010;288:177-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 95. | Saxena A, Chumanevich A, Fletcher E, Larsen B, Lattwein K, Kaur K, Fayad R. Adiponectin deficiency: role in chronic inflammation induced colon cancer. Biochim Biophys Acta. 2012;1822:527-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 66] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 96. | Boddicker RL, Whitley E, Birt DF, Spurlock ME. Early lesion formation in colorectal carcinogenesis is associated with adiponectin status whereas neoplastic lesions are associated with diet and sex in C57BL/6J mice. Nutr Cancer. 2011;63:1297-1306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 97. | Fujisawa T, Endo H, Tomimoto A, Sugiyama M, Takahashi H, Saito S, Inamori M, Nakajima N, Watanabe M, Kubota N. Adiponectin suppresses colorectal carcinogenesis under the high-fat diet condition. Gut. 2008;57:1531-1538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 125] [Cited by in RCA: 144] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 98. | Sikalidis AK, Fitch MD, Fleming SE. Risk of colonic cancer is not higher in the obese Lep(ob) mouse model compared to lean littermates. Pathol Oncol Res. 2013;19:867-874. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 99. | Hirose Y, Hata K, Kuno T, Yoshida K, Sakata K, Yamada Y, Tanaka T, Reddy BS, Mori H. Enhancement of development of azoxymethane-induced colonic premalignant lesions in C57BL/KsJ-db/db mice. Carcinogenesis. 2004;25:821-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 68] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 100. | Endo H, Hosono K, Uchiyama T, Sakai E, Sugiyama M, Takahashi H, Nakajima N, Wada K, Takeda K, Nakagama H. Leptin acts as a growth factor for colorectal tumours at stages subsequent to tumour initiation in murine colon carcinogenesis. Gut. 2011;60:1363-1371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 116] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 101. | Ikeda Y, Hama S, Kajimoto K, Okuno T, Tsuchiya H, Kogure K. Quantitative comparison of adipocytokine gene expression during adipocyte maturation in non-obese and obese rats. Biol Pharm Bull. 2011;34:865-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 102. | An W, Bai Y, Deng SX, Gao J, Ben QW, Cai QC, Zhang HG, Li ZS. Adiponectin levels in patients with colorectal cancer and adenoma: a meta-analysis. Eur J Cancer Prev. 2012;21:126-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 68] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 103. | Wei EK, Giovannucci E, Fuchs CS, Willett WC, Mantzoros CS. Low plasma adiponectin levels and risk of colorectal cancer in men: a prospective study. J Natl Cancer Inst. 2005;97:1688-1694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 343] [Cited by in RCA: 362] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 104. | Otake S, Takeda H, Suzuki Y, Fukui T, Watanabe S, Ishihama K, Saito T, Togashi H, Nakamura T, Matsuzawa Y. Association of visceral fat accumulation and plasma adiponectin with colorectal adenoma: evidence for participation of insulin resistance. Clin Cancer Res. 2005;11:3642-3646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 162] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 105. | Ferroni P, Palmirotta R, Spila A, Martini F, Raparelli V, Fossile E, Mariotti S, Del Monte G, Buonomo O, Roselli M. Prognostic significance of adiponectin levels in non-metastatic colorectal cancer. Anticancer Res. 2007;27:483-489. [PubMed] |
| 106. | Gonullu G, Kahraman H, Bedir A, Bektas A, Yücel I. Association between adiponectin, resistin, insulin resistance, and colorectal tumors. Int J Colorectal Dis. 2010;25:205-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 107. | Lukanova A, Söderberg S, Kaaks R, Jellum E, Stattin P. Serum adiponectin is not associated with risk of colorectal cancer. Cancer Epidemiol Biomarkers Prev. 2006;15:401-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 73] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 108. | Fukumoto J, Otake T, Tajima O, Tabata S, Abe H, Mizoue T, Ohnaka K, Kono S. Adiponectin and colorectal adenomas: Self Defense Forces Health Study. Cancer Sci. 2008;99:781-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 109. | Stocks T, Lukanova A, Johansson M, Rinaldi S, Palmqvist R, Hallmans G, Kaaks R, Stattin P. Components of the metabolic syndrome and colorectal cancer risk; a prospective study. Int J Obes (Lond). 2008;32:304-314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 122] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 110. | Wolf I, Sadetzki S, Kanety H, Kundel Y, Pariente C, Epstein N, Oberman B, Catane R, Kaufman B, Shimon I. Adiponectin, ghrelin, and leptin in cancer cachexia in breast and colon cancer patients. Cancer. 2006;106:966-973. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 124] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 111. | Stattin P, Palmqvist R, Söderberg S, Biessy C, Ardnor B, Hallmans G, Kaaks R, Olsson T. Plasma leptin and colorectal cancer risk: a prospective study in Northern Sweden. Oncol Rep. 2003;10:2015-2021. [PubMed] |
| 112. | Stattin P, Lukanova A, Biessy C, Söderberg S, Palmqvist R, Kaaks R, Olsson T, Jellum E. Obesity and colon cancer: does leptin provide a link? Int J Cancer. 2004;109:149-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 218] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 113. | Tamakoshi K, Toyoshima H, Wakai K, Kojima M, Suzuki K, Watanabe Y, Hayakawa N, Yatsuya H, Kondo T, Tokudome S. Leptin is associated with an increased female colorectal cancer risk: a nested case-control study in Japan. Oncology. 2005;68:454-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 71] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 114. | Tutino V, Notarnicola M, Guerra V, Lorusso D, Caruso MG. Increased soluble leptin receptor levels are associated with advanced tumor stage in colorectal cancer patients. Anticancer Res. 2011;31:3381-3383. [PubMed] |
| 115. | Healy LA, Howard JM, Ryan AM, Beddy P, Mehigan B, Stephens R, Reynolds JV. Metabolic syndrome and leptin are associated with adverse pathological features in male colorectal cancer patients. Colorectal Dis. 2012;14:157-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 116. | Wallace AM, Sattar N, Mcmillan DC. The co-ordinated cytokine/hormone response to acute injury incorporates leptin. Cytokine. 2000;12:1042-1045. [PubMed] |
| 117. | Bolukbas FF, Kilic H, Bolukbas C, Gumus M, Horoz M, Turhal NS, Kavakli B. Serum leptin concentration and advanced gastrointestinal cancers: a case controlled study. BMC Cancer. 2004;4:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 54] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 118. | Erarslan E, Turkay C, Koktener A, Koca C, Uz B, Bavbek N. Association of visceral fat accumulation and adiponectin levels with colorectal neoplasia. Dig Dis Sci. 2009;54:862-868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 65] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 119. | Guadagni F, Roselli M, Martini F, Spila A, Riondino S, D’Alessandro R, Del Monte G, Formica V, Laudisi A, Portarena I. Prognostic significance of serum adipokine levels in colorectal cancer patients. Anticancer Res. 2009;29:3321-3327. [PubMed] |
| 120. | Byeon JS, Jeong JY, Kim MJ, Lee SM, Nam WH, Myung SJ, Kim JG, Yang SK, Kim JH, Suh DJ. Adiponectin and adiponectin receptor in relation to colorectal cancer progression. Int J Cancer. 2010;127:2758-2767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 121. | Aleksandrova K, Boeing H, Jenab M, Bueno-de-Mesquita HB, Jansen E, van Duijnhoven FJ, Fedirko V, Rinaldi S, Romieu I, Riboli E. Total and high-molecular weight adiponectin and risk of colorectal cancer: the European Prospective Investigation into Cancer and Nutrition Study. Carcinogenesis. 2012;33:1211-1218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 65] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 122. | Song M, Zhang X, Wu K, Ogino S, Fuchs CS, Giovannucci EL, Chan AT. Plasma adiponectin and soluble leptin receptor and risk of colorectal cancer: a prospective study. Cancer Prev Res (Phila). 2013;6:875-885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 123. | Sălăgeanu A, Tucureanu C, Lerescu L, Caraş I, Pitica R, Gangurà G, Costea R, Neagu S. Serum levels of adipokines resistin and leptin in patients with colon cancer. J Med Life. 2010;3:416-420. [PubMed] |
| 124. | Steppan CM, Lazar MA. The current biology of resistin. J Intern Med. 2004;255:439-447. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 260] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 125. | Kumor A, Daniel P, Pietruczuk M, Małecka-Panas E. Serum leptin, adiponectin, and resistin concentration in colorectal adenoma and carcinoma (CC) patients. Int J Colorectal Dis. 2009;24:275-281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 100] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 126. | Nakajima TE, Yamada Y, Hamano T, Furuta K, Matsuda T, Fujita S, Kato K, Hamaguchi T, Shimada Y. Adipocytokines as new promising markers of colorectal tumors: adiponectin for colorectal adenoma, and resistin and visfatin for colorectal cancer. Cancer Sci. 2010;101:1286-1291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 163] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 127. | Danese E, Montagnana M, Minicozzi AM, Bonafini S, Ruzzenente O, Gelati M, De Manzoni G, Lippi G, Guidi GC. The role of resistin in colorectal cancer. Clin Chim Acta. 2012;413:760-764. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 128. | Waseem T, Javaid-Ur-Rehman F, Azam M, Qureshi MA. Role of ghrelin axis in colorectal cancer: a novel association. Peptides. 2008;29:1369-1376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 64] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 129. | D’Onghia V, Leoncini R, Carli R, Santoro A, Giglioni S, Sorbellini F, Marzocca G, Bernini A, Campagna S, Marinello E. Circulating gastrin and ghrelin levels in patients with colorectal cancer: correlation with tumour stage, Helicobacter pylori infection and BMI. Biomed Pharmacother. 2007;61:137-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 50] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 130. | Kemik O, Sumer A, Kemik AS, Hasirci I, Purisa S, Dulger AC, Demiriz B, Tuzun S. The relationship among acute-phase response proteins, cytokines and hormones in cachectic patients with colon cancer. World J Surg Oncol. 2010;8:85. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 131. | Win AK, Macinnis RJ, Hopper JL, Jenkins MA. Risk prediction models for colorectal cancer: a review. Cancer Epidemiol Biomarkers Prev. 2012;21:398-410. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 132. | Johnson IT, Lund EK. Review article: nutrition, obesity and colorectal cancer. Aliment Pharmacol Ther. 2007;26:161-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 88] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 133. | Drew JE. Molecular mechanisms linking adipokines to obesity-related colon cancer: focus on leptin. Proc Nutr Soc. 2012;71:175-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 134. | Irwin ML. Physical activity interventions for cancer survivors. Br J Sports Med. 2009;43:32-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 107] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 135. | Jeon JY, Meyerhardt JA. Energy in and energy out: what matters for survivors of colorectal cancer? J Clin Oncol. 2012;30:7-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 136. | Vazzana N, Riondino S, Toto V, Guadagni F, Roselli M, Davi G, Ferroni P. Obesity-driven inflammation and colorectal cancer. Curr Med Chem. 2012;19:5837-5853. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 137. | Jeon J, Sato K, Niedzwiecki D, Ye X, Saltz LB, Mayer RJ, Mowat RB, Whittom R, Hantel A, Benson A. Impact of physical activity after cancer diagnosis on survival in patients with recurrent colon cancer: Findings from CALGB 89803/Alliance. Clin Colorectal Cancer. 2013;12:233-238. [PubMed] |