Published online Nov 7, 2013. doi: 10.3748/wjg.v19.i41.7205
Revised: July 21, 2013
Accepted: August 12, 2013
Published online: November 7, 2013
Processing time: 189 Days and 7.1 Hours
Intramural duodenal hematoma is a rare cause of a proximal gastrointestinal tract obstruction. Presentation of intramural duodenal hematoma most often occurs following blunt abdominal trauma in children, but spontaneous non-traumatic cases have been linked to anticoagulant therapy, pancreatitis, malignancy, vasculitis and endoscopy. We report an unusual case of spontaneous intramural duodenal hematoma presenting as an intestinal obstruction associated with acute pancreatitis in a patient with established von Willebrand disease, type 2B. The patient presented with abrupt onset of abdominal pain, nausea, and vomiting. Computed tomography imaging identified an intramural duodenal mass consistent with blood measuring 4.7 cm × 8.7 cm in the second portion of the duodenum abutting on the head of the pancreas. Serum lipase was 3828 units/L. Patient was managed conservatively with bowel rest, continuous nasogastric decompression, total parenteral nutrition, recombinant factor VIII (humateP) and transfusion. Symptoms resolved over the course of the hospitalization. This case highlights an important complication of an inherited coagulopathy.
Core tip: Spontaneous non-traumatic duodenal hematoma has been linked to coagulopathy, but this case includes a rare form of coagulopathy in von Willebrand type 2B. Early identification with computed tomography imaging and treatment of the coagulopathy aided the usage of conservative therapy which allowed the patient to avoid surgical intervention consistent with prior successful cases of duodenal hematomas.
- Citation: Eichele DD, Ross M, Tang P, Hutchins GF, Mailliard M. Spontaneous intramural duodenal hematoma in type 2B von Willebrand disease. World J Gastroenterol 2013; 19(41): 7205-7208
- URL: https://www.wjgnet.com/1007-9327/full/v19/i41/7205.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i41.7205
Intramural duodenal hematoma is a rare occurrence of proximal gastrointestinal tract obstruction. Presentation of intramural duodenal hematoma most often occurs following blunt abdominal trauma in children, but non-traumatic cases have been linked to anticoagulant therapy, pancreatitis, bleeding disorders, malignancy, vasculitis and upper endoscopy[1-4]. We report a unique case of spontaneous intramural duodenal hematoma presenting as an intestinal obstruction associated with acute pancreatitis infection in a patient with established von Willebrand disease, type 2B.
A 24-year-old male was transferred to our tertiary center with a 10 h history of abrupt onset abdominal pain, nausea, and vomiting. History was notable for type 2B von Willebrand disease initially diagnosed at 12 years of age following recurrent hemorrhagic episodes (epistaxis, gingival bleeding and hemarthrosis) and diagnosis in his dizygotic sibling for a spontaneous bleeding episode. Both dizygotic siblings were genetically tested for mutations and found to have the G3946A genotype. Physical examination on admission revealed heart rate 66 beat/min, blood pressure 115/53 mmHg, temperature 36.7 °C and respiration rate 16. Patient was in moderate amount of pain. There were minor facial petechiae. Oropharynx was pink and moist and no evidence of gingival bleeding or epistaxis. There was no conjunctival injection. Neck was supple without adenopathy or thyromegaly. Heart was regular rate and rhythm without murmur. Respirations were unlabored and auscultation was clear bilaterally. Abdominal exam demonstrated hypoactive bowel sounds, epigastric tenderness, without rebound tenderness or guarding. Testicular exam revealed bilateral ecchymoses without edema or tenderness. The lower extremities noted multiple small ecchymosis in various stages of healing. There was normal range of motion in upper and lower extremities, no focal joint pain or effusions noted. Hemoglobin was 15.0 g/dL, white blood cell count 18.8 cells/mL, segmented neutrophils 85%, bands 6%, platelets were immeasurable due to clumping. Sodium 140 mmol/L, potassium 4.0 mmol/L, chloride 107 mmol/L, bicarbonate 26 mmol/L, BUN 21 mg/dL, creatinine 0.9 mg/dL, total protein 7.1 gm/dL, albumin 4.3 gm/dL, aspartate aminotransferase 33 U/L, alanine aminotransferase 34 U/L, alkaline phosphatase 46 U/L, bilirubin 2.4 mg/dL, Lipase 3828 units/L.
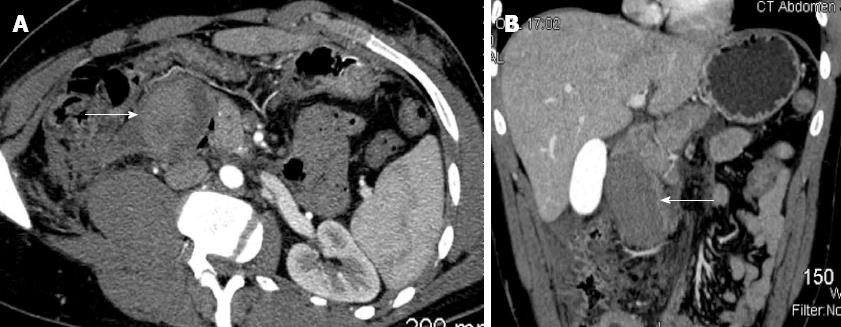
Computed tomography (CT) of the abdomen without contrast revealed a hyperdense mass in the second and third portions of the duodenum measuring 4.7 cm× 8.7 cm with associated retroperitoneal hemorrhage, extending from the right anterior pararenal space to the right pelvic sidewall. The duodenal mass resembled a hematoma and was abutting the pancreatic head to the left. Liver, spleen, adrenal glands and genitourinary organs were normal. The findings led to a diagnosis of intramural duodenal hematoma complicated by acute pancreatitis (Figure 1).
The patient was transfused one unit of single donor platelets and administered human antihemophilic factor/von Willebrand factor complex (Humate P®) 80 Units/kg prior to transfer from outside facility. A nasogastric (NG) tube was placed, and intravenous bolus of three liters of normal saline was given. Surgery consultation was obtained. He received factor replacement therapy (Humate P® 40 units/kg every 12 h) for the next 72 h, following an initial loading bolus.
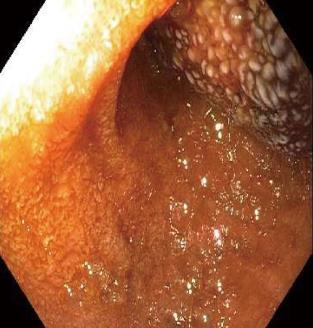
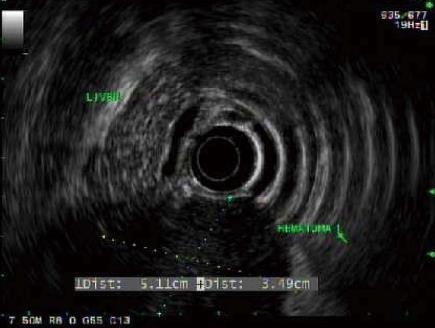
Esophagogastroduodenoscopy and endoscopic ultrasound were delayed until day eleven of the hospitalization due to potential risk for hemorrhage from the hematoma during endoscopy. Endoscopy showed a near complete obstruction in the second portion of the duodenum with large amount of gastric fluid (Figures 2 and 3). The patient was discharged to home on day 15 of hospitalization after patient’s pain receded, repeat imaging noted unchanged duodenal and retroperitoneal hematoma and resolution of gastric outlet obstruction. The patient was readmitted to the hospital the following day with worsening abdominal pain with development of nausea and vomiting. On readmission a repeat abdominal CT was performed due to concern of enlargement and extravasation of the hematoma. Imaging revealed a minute increase in size of the duodenal hematoma that measured 5.3 cm × 5.8 cm (previously 5.3 cm × 5.2 cm) in the AP and transverse dimensions and persistent retroperitoneal hematoma. After control of the patient’s symptoms and his ability to tolerate a modified and limited oral diet the patient was finally discharged home on day 23 of the second hospitalization. Following discharge the patient had not experienced return of abdominal pain, nausea and vomiting. Patient was able to tolerate a full diet and total parenteral nutrition (TPN) was discontinued shortly after discharge. Abdominal CT performed at day 68 from presentation revealed decreased size of the duodenal hematoma with measurements of 2.1 cm × 2.6 cm (initially 4.7 cm × 8.7 cm).
McLachlan described the first published report of duodenal hematoma in 1838 as a false aneurismal tumor[5]. Decades later, the first case of a non-traumatic intramural hematoma was reported by Sutherland in a child with Henoch-Schonlein purpura that presented as intussusception.
The most common clinical scenario for presentation of intramural duodenal hematoma is seen in blunt abdominal trauma in children[3,4], but non-traumatic cases have been linked to anticoagulant therapy, pancreatitis, bleeding disorders, malignancy, vasculitis and upper endoscopy[1,3,4,6]. In adults the most common risk factor for spontaneous intramural small-bowel hematoma reported is supratherapeutic anticoagulation[7]. Other notable risk factors include bleeding disorders (hemophilia, idiopathic thrombocytopenic purpura), malignancies and its therapies (leukemia, lymphoma, myeloma, pancreatic cancer, and chemotherapy), vasculitis and pancreatitis[7,8].
Common signs and symptoms on presentation typically include abdominal pain that ranges from vague abdominal complaints to intestinal tract obstruction with vomiting and tenderness mimicking an acute abdomen[7]. Abbas noted that most of the patients presented after having symptoms for several days[7]. Other infrequent signs and symptoms include jaundice and right upper quadrant tenderness due to bile duct obstruction secondary to compression by the hematoma[8]. In this case, the patient’s elevated lipase identified pancreatitis induced by the hematoma as opposed to the case of Bellens et al[9] in which pancreatitis was the inciting event.
The location of the hemorrhage in spontaneous non-traumatic intraluminal hematoma is submucosal and originates from a small vessel[7]. In our case, based upon the imaging findings, the bleeding originated from the gastroduodenal artery. Extravasation of submucosal hematomas can occur into intraluminal, intra-mesenteric, and retroperitoneal sites[7]. Whereas traumatic small-bowel hematomas commonly affect the duodenum, most spontaneous small-bowel hematomas involve the jejunum[7,10]. Additionally, Abbas noted that no spontaneous intramural hematoma was less than 8 cm in length, consistent with our findings of an 8.7 cm length[7].
In recent years, there has been an increase in detection of non-traumatic intramural small bowel hematomas in patients that are anti-coagulated. This increase has also coincided with more sensitive imaging modalities that have led to earlier detection. In general, CT is the imaging choice for diagnosis of spontaneous intramural small bowel hematoma, but as a rapid screening modality, ultrasound can be used effectively during the initial evaluation, as it is inexpensive, rapid, noninvasive and readily available in most instances[5,7]. Characteristic features have been described include circumferential bowel wall thickening, intramural hyperdensity with Hounsfield units characteristic for blood (30-80 H), luminal narrowing and intestinal obstruction[7]. Reassessment of the status of the hematoma is generally recommended with CT in 2 wk following diagnosis and conservative management[8].
In prior case reports, the initial treatment regimen for IDH favored medical management; if feasible in a stable patient, as spontaneous local absorption of the hematoma occurs in most circumstances[5-8]. Conservative management constituted bowel rest, continuous NG decompression, TPN, blood transfusion and correction of coagulopathy. Minimally invasive drainage through image-guided modalities is generally not recommended in consideration to the technical difficulty required to access the precarious location of the pathology as well as the risk for bowel perforation[8]. Complete resolution of the hematoma and healing of the intestine usually occurs within 2 mo after diagnosis in cases of non-extensive hematomas[5,7]. Surgical intervention is only indicated if there is significant intraluminal hemorrhage, bowel perforation or identifiable risk of ischemia[4,5,7]. Surgical evacuation of the hematoma through laparotomy or laparoscopy tends to be the best surgical treatment in these situations, specifically in instances in which the abdominal pain or obstruction did not resolve and objective findings of bowel infarction were present[11-13]. Limited case reports have detailed successful outcomes with percutaneous aspiration and endoscopic dilation[4,14,15]. Finally, bypass surgery is reserved for patients with severe duodenal perforation and multiple comorbidities precluding resectional surgery[8].
Our case highlights a rare but important complication of coagulopathy. Acute abdominal pain with signs and symptoms of obstruction in patients with coagulopathy should raise suspicion for spontaneous intramural small-bowel hematoma. Prompt diagnosis with CT imaging is a crucial contribution to patient treatment, because conservative medical management can be achieved in most patients. Whereas, in cases of failed conservative medical management surgical intervention is performed.
P- Reviewer: Michiels JJ S- Editor: Zhai HH L- Editor: A E- Editor: Ma S
| 1. | Hoenisch K, Prommegger R, Schwaighofer H, Freund M, Schocke M, Vogel W, Kaser A. Intramural duodenal hematoma after upper gastrointestinal endoscopy. Wien Med Wochenschr. 2011;161:441-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 2. | Moore SW, Erlandson ME. Intramural Hematoma of the Duodenum. Ann Surg. 1963;157:798-807. [PubMed] |
| 3. | Weil BR, Howard TJ, Zyromski NJ. Spontaneous duodenal hematoma: a rare cause of upper gastrointestinal tract obstruction. Arch Surg. 2008;143:794-796. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 4. | Hou SW, Chen CC, Chen KC, Ko SY, Wong CS, Chong CF. Sonographic diagnosis of spontaneous intramural small bowel hematoma in a case of warfarin overdose. J Clin Ultrasound. 2008;36:374-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 5. | McLachlan J. Fatal false aneurismal tumor occupying nearly the whole of the duodenum. Lancet. 1838;2:203-205. |
| 6. | Touloukian RJ. Protocol for the nonoperative treatment of obstructing intramural duodenal hematoma during childhood. Am J Surg. 1983;145:330-334. [PubMed] |
| 7. | Abbas MA, Collins JM, Olden KW. Spontaneous intramural small-bowel hematoma: imaging findings and outcome. AJR Am J Roentgenol. 2002;179:1389-1394. [PubMed] |
| 8. | Ma JK, Ng KK, Poon RT, Fan ST. Pancreatic-induced intramural duodenal haematoma. Asian J Surg. 2008;31:83-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Bellens L, Van Hee R, Vanderstighelen Y, Vanderputte S. Intramural duodenal hematoma of pancreatic origin. Hepatogastroenterology. 1999;46:930-932. [PubMed] |
| 10. | Jones WR, Hardin WJ, Davis JT, Hardy JD. Intramural hematoma of the duodenum: a review of the literature and case report. Ann Surg. 1971;173:534-544. [PubMed] |
| 11. | Beal JM, Haid S, Sherrick J, Scarff J, Rambach W, Method H. Small bowel obstruction secondary to hematoma of the mesentery. IMJ IllMed J. 1966;130:422-426. |
| 12. | Xeropotamos NS, Tsakayannis DE, Kappas AM. Intramural post-traumatic haematomas of the duodenum: are there any late sequelae of operative intervention? Injury. 1997;28:349-352. [PubMed] |
| 13. | Simi S, Anoop TM, George KC. Spontaneous intramural duodenal hematoma--a rare cause of upper gastrointestinal obstruction. Am J Emerg Med. 2010;28:642.e1-642.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 14. | Aizawa K, Tokuyama H, Yonezawa T, Doi M, Matsuzono Y, Matumoto M, Uragami K, Nishioka S, Yataka I. A case of traumatic intramural hematoma of the duodenum effectively treated with ultrasonically guided aspiration drainage and endoscopic balloon catheter dilation. Gastroenterol Jpn. 1991;26:218-223. [PubMed] |
| 15. | Gullotto C, Paulson EK. CT-guided percutaneous drainage of a duodenal hematoma. AJR Am J Roentgenol. 2005;184:231-233. [PubMed] |