Published online Sep 14, 2013. doi: 10.3748/wjg.v19.i34.5598
Revised: July 13, 2013
Accepted: July 17, 2013
Published online: September 14, 2013
Processing time: 121 Days and 11.7 Hours
Esophageal cancer (EsC) is one of the least studied and deadliest cancers worldwide because of its extremely aggressive nature and poor survival rate. It ranks sixth among all cancers in mortality. In retrospective studies of EsC, smoking, hot tea drinking, red meat consumption, poor oral health, low intake of fresh fruit and vegetables, and low socioeconomic status have been associated with a higher risk of esophageal squamous cell carcinoma. Barrett’s esophagus is clearly recognized as a risk factor for EsC, and dysplasia remains the only factor useful for identifying patients at increased risk, for the development of esophageal adenocarcinoma in clinical practice. Here, we investigated the epidemiologic patterns and causes of EsC. Using population based cancer data from the Surveillance, Epidemiology and End Results Program of the United States; we generated the most up-to-date stage distribution and 5-year relative survival by stage at diagnosis for 1998-2009. Special note should be given to the fact that esophageal cancer, mainly adenocarcinoma, is one of the very few cancers that is contributing to increasing death rates (20%) among males in the United States. To further explore the mechanism of development of EsC will hopefully decrease the incidence of EsC and improve outcomes.
Core tip: Here, we investigated the epidemiologic patterns and causes of esophageal cancer. Using population based cancer data from the Surveillance, Epidemiology and End Results Program of the United States; we generated the most up-to-date stage distribution and 5-year relative survival by stage at diagnosis for 1998-2009.
- Citation: Zhang Y. Epidemiology of esophageal cancer. World J Gastroenterol 2013; 19(34): 5598-5606
- URL: https://www.wjgnet.com/1007-9327/full/v19/i34/5598.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i34.5598
Esophageal cancer (EsC) including squamous cell carcinoma (SCC) and adenocarcinoma is considered as a serious malignancy with respect to prognosis and a fatal outcome in the great majority of cases[1,2]. Esophageal carcinoma affects more than 450000 people worldwide and the incidence is rapidly increasing[3]. Currently, EsC is the eighth most common incident cancer in the world because of its extremely aggressive nature and poor survival rate[4,5].
EsC exhibits an epidemiologic pattern distinct from all other cancers[6,7]. The incidence of esophageal adenocarcinoma has increased sharply over the past few decades, both by period and birth cohort. Etiological studies are required to explain the rapid increase of this lethal cancer[8]. Understanding the epidemiology of EsC will be the key to elucidating the causes and risk factors for esophageal cancer and thus the cornerstone of developing any prevention strategies.
Cancer of the esophagus typically occurs in one of two forms, SCCs arising from the stratified squamous epithelial lining of the organ, and adenocarcinomas affecting columnar glandular cells that replace the squamous epithelium[9]. Sarcomas and small cell carcinomas generally represent less than 1%-2% of all esophageal cancers[10,11]. On rare occasions, other carcinomas, melanomas, leiomyosarcomas, carcinoids, and lymphomas may develop in the esophagus as well[5].
SCC is the predominant histologic type of esophageal cancer worldwide[12]. The incidence of squamous cell cancer of the esophagus increases with age as well and peaks in the seventh decade of life. The incidence of squamous cell esophageal cancer is three times higher in blacks than in whites, whereas adenocarcinomas are more common in white men.
The natural histories of SCCs and adenocarcinomas of esophagus appear to differ substantially. For squamous cell cancers, transition models have described squamous epithelium undergoing inflammatory changes that progress to dysplasia and in situ malignant change[13,14].
Most adenocarcinomas, however, tend to arise in the distal esophagus from columnar-lined metaplastic epithelium, commonly known as Barrett’s esophagus[15,16], which replaces the squamous epithelium during the healing reflux esophagitis and may progress to dysplasia. Gastroesophageal reflux disease (GERD), or just reflux[17-19] can damage the lining of esophagus which causes Barrett’s esophagus[17], characterized by abnormal “tongues” of salmon-colored mucosa extending proximally from the gastroesophageal junction into the normal pale esophageal mucosa, develops in approximately 5 to 8 percent of patients with gastroesophageal reflux disease.
Cancers that start at the area where the esophagus joins the stomach (the GE junction), which includes about the first 2 inches of the stomach (called the cardia), tend to behave like esophagus cancers (and are treated like them, as well), so they are grouped with esophagus cancers. Approximately three quarters of all adenocarcinomas are found in the distal esophagus, whereas SCCs are more evenly distributed between the middle and lower third. The cervical esophagus is an uncommon site of disease. Nowadays the terminology used for the definition of adenocarcinomas at the GE junction is “cardiac carcinoma”, which can be easily misunderstood. This definition of adenocarcinomas of the GE junction does not allow correct comparison of diagnosis (endoscopic, radiological and pathologic), epidemiology and surgical therapy in national and international aspects, because different tumor can develop in the same area, and all called cardia tumors[20]. Siewert and Stein recommended a classification to solve this problem[21]. The classification of the tumors is morphological/topographical[21,22]. Type I is adenocarcinoma of the distal part of the esophagus. Type II is adenocarcinoma of the real cardia and type III is subcardial gastric adenocarcinoma. The importance of this classification is it enables unified pre-operative assessment and it can also help to decide the type of the surgical intervention[20,23-27].
Cancers arising from the esophagus, including the GE junction, are relatively uncommon in the United States[28,29]. The rate of cancer of the distal esophagus is about equal to that of the more proximal two-thirds[30]. SCC is the predominant histologic type of esophageal cancer worldwide. The incidence of SCC increases with age as well and peaks in the seventh decade of life, which is three times higher in blacks than in whites, whereas adenocarcinomas are more common in white men.
The most important precancerous disease is Barrett’s esophagus[31-34]. Patients with Barrett’s esophagus have a 50 to 100 times increase in their risk of developing cancer compared to the general population. People with Barrett’s esophagus are much more likely to develop cancer of the esophagus. These people require close medical follow-up in order to find cancer early. Still, although they have a higher risk, most people with Barrett’s esophagus do not go on to develop cancer of the esophagus. In their population-based cohort study, Hvid-Jensen et al[35] reported an annual risk of esophageal adenocarcinoma of 0.12% among patients with Barrett’s esophagus.
For different types of esophageal cancer, the risk increases with age, with a mean age at diagnosis of 67 years. Esophageal cancer age-adjusted incidence of blacks was about twice that of whites (8.63/100000 vs 4.39/100000, P < 0.05)[36]. Squamous cell carcinoma was more commonly diagnosed in blacks and white females, whereas adenocarcinoma was more common among white males.
Although the disease is relatively uncommon in the United States, it is a major global health threat[37]. Esophageal cancer is four times more common and slightly more lethal in men than in women. According to the National Cancer Institute (Cancer.gov) in 2012, it is estimated that 17460 persons (13950 men and 3510 women) will be diagnosed with and 15070 persons will die of cancer of the esophagus in 2012.
Esophageal cancer occurs at a rate 20 to 30 times higher in China than in the United States. An esophageal “cancer belt,” primarily squamous cell cancers, extends from northeast China to the Middle East[38-40]. Evidence of an association between environment and diet and esophageal cancer comes from the profound differences in incidence observed in various parts of the world. The majority of the factors so far implicated in cancer of the esophagus appear to act directly on the esophagus rather than systemically. Nutritional deficiencies can develop by chronic alcohol use as well as by poverty and lack of an adequate food supply, but diet does not explain the whole picture. External carcinogens are necessary to affect the end result. The association between nutrition and esophagitis may suggest methods of primary prevention of esophageal cancer and provide a chance of lowering the incidence of this deadly disease[31].
From 1996-2009, the annual percentage change was increased by 0.5% in all races and 0.4% in white. However, the increase of incidence is because of the increase incidence in men. Actually, the incidence in woman dropped by 0.4% (Surveillance, Epidemiology and End Results, SEER).
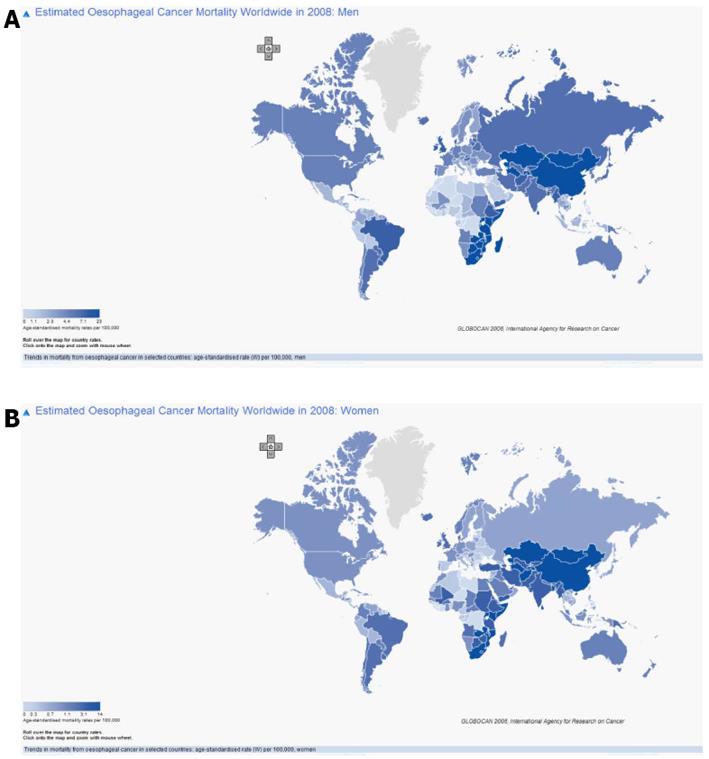
Figure 1 shows the age-adjusted esophageal cancer mortality. It is in line with the incidence rate in the world but there is no difference between men and women. Age-adjusted mortality for blacks, although showing a declining trend, was nearly twice that of whites (7.79 vs 3.96, P < 0.05). Squamous cell carcinoma was more commonly diagnosed in blacks and white females, whereas adenocarcinoma was more common among white males (P < 0.001)[41]. The reasons are economic status, diet, and poor eating habits, etc.
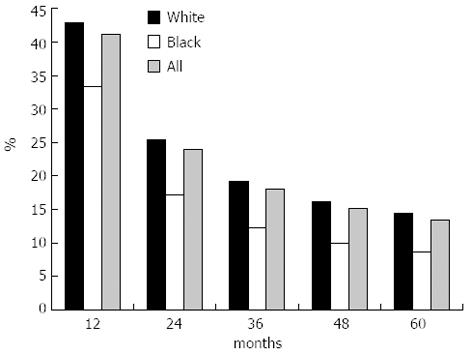
Survival varied widely according to cancer site. The differences in survival related to histology were also expected[42]. Although survival was poor for all groups, it was significantly poorer in blacks than in whites (Figure 2). The overall 5-year relative survival for 2002-2008 from 18 SEER geographic areas was 16.9%. Five-year relative survival by race and sex was: 18.1% for white men; 17.0% for white women; 10.4% for black men; 12.6% for black women.
The overall relative 5-year survival rates over time increase gradually in white and black, man and women. For example, the rate was below 2% in 1995 to over 10% in 2008 in black men (SEER).
Although the overall outlook for patients diagnosed with esophageal cancer has improved in the past 30 years, most patients still present with advanced disease, and their survival remains poor[43]. One-third to one-half of patients treated with either chemoradiation therapy or chemoradiation therapy plus surgery are alive at 2 years, without recurrence of esophageal cancer.
The reason is because esophageal cancer is diagnosed at rather late stage. Overall, more than 30 percent of patients have metastatic disease at the time of presentation (32.15% in white and 31.83% in black). None was found that has in situ cancer, due to the fact that it can be difficult to diagnose esophageal cancer early. Among patients who are undergoing primary surgery, 22 percent have localized disease, 30 percent have regional cancer (Table 1).
| Stage at diagnosis | Stage distribution | 5-year relative survival |
| Localized (confined to primary site) | 22% | 37.80% |
| Regional (spread to regional lymphnodes) | 30% | 19.80% |
| Distant (cancer has metastasized) | 35% | 3.40% |
| Unknown (unstaged) | 13% | 10.50% |
The patterns of esophageal cancer are dramatically changing in the United States. However, the mechanisms of esophageal tumorigenesis are not fully understood[5]. Three decades ago the large majority of these cancers were SCCs, but the incidence of esophageal adenocarcinoma has been steadily increasing[44]. Tobacco and alcohol consumption are the primary causes of SCCs of the esophagus[45]. One of the strongest emerging risk factors, however, is obesity. Increases in the prevalence of obesity and the incidence of esophageal adenocarcinoma are parallel, and several epidemiologic studies have shown upwards of threefold excess risks among overweight individuals. Further research into the causes of these usually fatal cancers may help identify other potential determinants and provide needed information to help stem their increase.
Cigarettes, red meat, alcohol and hookah smoking[4], nass use (a chewing tobacco product), opium consumption, hot tea drinking, poor oral health, low intake of fresh fruit and vegetables, and low socioeconomic status have been associated with a higher risk of esophageal SCC (Table 2). Barrett’s esophagus is clearly recognized as a risk factor for EsC, and dysplasia remains the only factor useful for identifying patients at increased risk, for the development of esophageal adenocarcinoma in clinical practice.
| Risk factor | Squmous-cell carcinoma | Adenocarcinoma | |
| First or second hand smoke | +++ | ++ | |
| Alcohol consumption | +++ | - | |
| Consumption of red meat | + | + | |
| Barrett’s esophagus | - | ++++ | |
| Reflux symptoms | - | +++ | |
| Being overweight | - | ++ | |
| Poverty | ++ | - | |
| Caustic injury to the esophagus | ++++ | - | |
| History of head and neck cancer | ++++ | - | |
| History with radiotherapy | +++ | +++ | |
| Frequent consumption of extremely hot drinks | + | - | |
| Polymorphism | Cyclin D1 (CCND1) G870A polymorphism | - | + |
| p53 | + | - | |
| polymorphism | |||
| TERT A279T polymorphism | + | + | |
Moderate to heavy smokers face an increased risk of both SCC and adenocarcinoma of the esophagus. Research suggests that when a smoker ingests tobacco condensates, it causes tobacco carcinogens, particularly nitrosamines, to come in contact with the esophageal mucosa. There is a direct correlation between the number of cigarettes a smoker smokes per day; the length of time the smoker spends smoking, and the risk of esophageal cancer[2].
The incidence of SCC of the esophagus has been found to dramatically increase in the presence of any factor that causes chronic irritation and inflammation, such as excessive alcohol intake, especially in combination with smoking[46,47]. This does not hold true for adenocarcinoma. This may account for more than 90 percent of all cases of SCC of the esophagus in developed countries[48].
Chronic esophageal irritation also occurs when food is retained and decomposed by bacteria, releasing various chemical irritants. Frequent consumption of hot beverages also appears to increase the incidence of SCC[49].
Esophageal squamous cell carcinoma (ESCC) is clearly linked to a low socioeconomic status. The increasing prevalence of obesity in the Western world is thought to add to the rising incidence of esophageal adenocarcinoma. More specifically, it has been postulated that obesity increases intraabdominal pressure and gastroesophageal reflux by a specific mechanism, although some studies provided contradictory results. On the other hand, adipose tissue itself influences tumor development[50-54]. Adipocytes and inflammatory cells secrete adipokines and cytokines which are known to promote tumor development. The abundant availability of lipids from adipocytes in the tumor microenvironment, supports tumor progression and uncontrolled growth. Given that adipocytes are a major source of adipokines and energy for the cancer cell, understanding the mechanisms of metabolic symbiosis between cancer cells and adipocytes, should reveal new therapeutic possibilities.
The genetic and molecular changes underlying the development of EsC remain poorly understood. Genetic analysis of these cancers reveals frequent chromosomal losses (4q, 5q, 9p, and 18q), chromosomal gains (8q, 17q, and 20q), and occasional gene amplifications (7, 8, and 17q)[5].
In the past decade, efforts have been made to use candidate gene approaches to identify genetic susceptibility factors for ESCC. The genome-wide association studies (GWAS) has emerged as a powerful and successful tool to identify common disease alleles by using high-throughput genotyping technology to interrogate a large number of tagging single nucleotide polymorphisms (SNPs) that serve as surrogates for untested common SNPs across the genome. So far, GWAS of esophageal cancers including ESCC in individuals of European and Japanese ancestry, have shown that variants in ADH genes and/or ALDH2 are associated with risk of ESCC[55-58]. More recently, Wu et al further reported that nine new ESCC susceptibility loci, of which seven, at chromosomes 4q23, 16q12.1, 17q21, 22q12, 3q27, 17p13 and 18p11, had a significant marginal effect (P = 1.78 × 10-39 to P = 2.49 × 10-11) and two of which, at 2q22 and 13q33, had a significant association only in the gene-alcohol drinking interaction [gene-environment interaction P (PG × E) = 4.39 × 10-11 and PG × E = 4.80 × 10-8, respectively]. Variants at the 4q23 locus, which includes the ADH cluster, each had a significant interaction with alcohol drinking in their association with ESCC risk (PG × E = 2.54 × 10-7 to 3.23 × 10-2). They confirmed the known association of the ALDH2 locus on 12q24 to ESCC, and a joint analysis showed that drinkers with both of the ADH1B and ALDH2 risk alleles had a fourfold increased risk for ESCC compared to drinkers without these risk alleles. Their results underscore the direct genetic contribution to ESCC risk, as well as the genetic contribution to ESCC through interaction with alcohol consumption[55].
There are also some studies on polymorphism on other locations for esophageal adenocarcinoma with smaller samples. Cyclin D1 (CCND1) G870A polymorphism has been known to be a risk factor in multiple cancers[59-63]. However, investigations concerning the association of CCND1 G870A polymorphism with esophageal cancer risk have generated conflicting results[64-69]. The overall data suggest that CCND1 G870A variations might have an association with increased esophageal cancer susceptibility. The earliest findings, published in 2005, reported that CCND1 G870A was a risk factor for esophageal adenocarcinoma[67]. A study conducted by Liu’s group, drew the exact opposite conclusion: CCND1 G870A was not associated with susceptibility to esophageal adenocarcinoma[70]. Liu’s group explained the discrepancy by noting that all previous studies were based on small samplings.
Since the definition of G870A is the same for both groups, the significant difference lies in the methods that they used. Casson’s group did polymerase chain reaction (PCR) followed by enzyme digestion, and visualized the result by running the products in a 15% acrylamide gel, and is referred to as “PCR-restriction fragment length polymorphism (RFLP),” which was widely used ten years ago. Liu’s group genotyped by the 5′-nuclease assay (TaqMan), using the ABI Prism 7900HT Sequence Detection System (Applied Biosystems, Foster City, CA, United States). This is currently considered the gold standard in genotyping. Casson’s group included patients with GERD. Since GERD is rather common in the general population, they selected strictly asymptomatic individuals for their control groups. Liu’s group chose instead to use healthy visitors as their control group. And those healthy controls might have had some undiagnosed diseases related to GERD, such as Barrett’s esophagus.
One source of bias between the two groups may lie in the different controls that were used. This may explain why the rate of G/G is different between the two groups. The second reason may be due to the detection method used. Usually, sequencing is viewed as the gold standard, but it is not always correct[71]. To detect polymorphism, the PCR-RFLP that Casson’s group used, might have been a better choice because PCR-RFLP tests detect the correct genotype. Direct sequencing of the PCR products, obtained with one of the primers located adjacent to a mutated nucleotide, may cause unequal amplification of alleles in heterozygous samples. This effect is even stronger when mismatched primers are used. Therefore, there is a potential pitfall in DNA sequencing, indicating that sequencing may not always be the gold standard. The third reason may be due to the inherent differences between the two groups. As we know, the minor allele frequency (maf) of a SNP is different among different populations. Since it is ethnicity related, more information is needed to know the demographic information of the patient and the control group.
Although Li argues that others may be drawing different conclusions than his group, due to smaller samplings, it cannot be ruled out that other factors are involved, such as how the different control groups were recruited. Zhuo et al[64] reported that homozygous AA alleles might elevate esophageal cancer risk among Asians, but not Caucasians. This might partially explain why the two groups drew different conclusions.
CCND1 G870A polymorphism might be a low-penetrant risk factor for esophageal carcinoma, particularly among Asians. More information is needed to study large samples in relationship to pertinent demographic data.
The keys to prevention of esophageal cancer vary by cell type. For SCC, reduction or elimination of tobacco and alcohol consumption provide the best means to reduce the incidence of this cancer. However, no one particular risk factor is responsible for the rising incidence of esophageal adenocarcinoma. Several preventive strategies are under investigation using such agents as nonsteroidal anti-inflammatory drugs, selenium, alpha-difluoromethylornithine, and retinoids[72]. Vegetable intake, and fruit intake is considered to be a preventive role. Carotene, vitamin C, and vitamin E are protective, most likely in combination with each other and other micronutrients. The role of vitamin A is not clear because of conflicting findings in the studies reviewed[73]. When intake of raw vegetables and cooked vegetables was analyzed separately, raw vegetables were found to be more protective. Because fruits are relatively expensive in most places, increased consumption may reflect higher socioeconomic status.
Since obesity is closely related to the incidence of the esophageal cancer, it would be interesting to follow up those patients with precancerous lesion to monitor their weight.
In patients with high-grade dysplasia, the options for preventive approaches include surveillance, endoscopic therapies, and surgical resection, but the optimum approach is debated[3]. In an analysis of more than 15 studies, the mean incidence of occult adenocarcinoma in patients with a preoperative diagnosis of high-grade dysplasia treated with esophagectomy was 41%. This high incidence provides a rationale for use of esophagectomy, but there is concern about the risk of morbidity. Use of endoscopic treatments for high-grade dysplasia has been supported in two randomised trials. In one trial of photodynamic therapy plus proton-pump inhibitors compared with proton-pump inhibitors alone, progression to cancer was significantly decreased in the photodynamic-therapy group (13% vs 28%). In the other, which assessed endoscopic radiofrequency ablation in patients with Barrett’s esophagus and high-grade dysplasia, radio frequency ablation was more effective in eradication of high-grade dysplasia than a proton-pump inhibitor alone, and the progression to cancer was lower (4% vs 22%) during short-term follow-up[74-77].
Although several potential preventive measures exist, none has been proven to decrease the risk of esophageal carcinoma in prospective well-designed trials[3]. The relatively low incidence of esophageal cancer, the absence of early symptoms, and the rarity of a hereditary form of the disease make population-based screening untenable except in certain high-risk areas of the world[5].
Patients who are found to have Barrett’s esophagus, however, may be candidates for regular endoscopic surveillance, since the incidence of low-grade dysplasia, high-grade dysplasia, and cancer is approximately 4 percent, 1 percent, and 0.5 percent per year, respectively, among such patients[5]. Whether endoscopic screening programs to detect Barrett’s esophagus in patients with chronic reflux disease symptoms are useful has been debated. Critics point out the high number of people in the general population who have reflux symptoms and the fact that at least 40% of patients with Barrett’s esophagus do not have reflux symptoms, and question the cost-effectiveness of screening. Proponents of screening for Barrett’s esophagus point to the clear associations between reflux, Barrett’s esophagus, and esophageal adenocarcinoma, and suggest that the rising incidence of esophageal adenocarcinoma justifies screening. No definitive data are available on whether endoscopic screening for Barrett’s esophagus is associated with a reduction in cancer-related mortality and, therefore, screening is not routinely recommended.
However, some experts have recommended that endoscopy be performed every three to five years in patients who have Barrett’s esophagus in the absence of epithelial dysplasia and more frequently if they are found to have low-grade dysplasia. Diagnostic endoscopy for early detection can be conducted in 2 steps: at first detection of an abnormal area through changes in relief, in color or in the course of superficial capillaries; then characterization of the morphology of the lesion. Then treatment decision offers 3 options according to histologic prediction: abstention, endoscopic resection, surgery. The rigorous quality control of endoscopy will reduce the miss rate of lesions and the occurrence of interval cancer[78].
The precise causes of EsC have not been identified. Despite uncertainties in our understanding of the causes of mechanistic pathways of esophageal cancer, there is sufficient evidence to take effective steps to prevent the majority of SCC in western countries, while more information is needed to curb the epidemic increase in adenocarcinoma[7,79].
P- Reviewers Fakheri H, Manfredi S, Milone M, Osawa S S- Editor Gou SX L- Editor A E- Editor Zhang DN
| 1. | Retraction: The candidate tumor suppressor gene ECRG4 inhibits cancer cells migration and invasion in esophageal carcinoma. J Exp Clin Cancer Res. 2011;30:19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 2. | Esophageal cancer: epidemiology, pathogenesis and prevention. Nat Clin Pract Gastroenterol Hepatol. 2008;5:517-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 147] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 3. | Pennathur A, Gibson MK, Jobe BA, Luketich JD. Oesophageal carcinoma. Lancet. 2013;381:400-412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1956] [Cited by in RCA: 1961] [Article Influence: 163.4] [Reference Citation Analysis (5)] |
| 4. | Mao WM, Zheng WH, Ling ZQ. Epidemiologic risk factors for esophageal cancer development. Asian Pac J Cancer Prev. 2011;12:2461-2466. [PubMed] |
| 5. | Enzinger PC, Mayer RJ. Esophageal cancer. N Engl J Med. 2003;349:2241-2252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2115] [Cited by in RCA: 2219] [Article Influence: 100.9] [Reference Citation Analysis (0)] |
| 6. | Engel LS, Chow WH, Vaughan TL, Gammon MD, Risch HA, Stanford JL, Schoenberg JB, Mayne ST, Dubrow R, Rotterdam H. Population attributable risks of esophageal and gastric cancers. J Natl Cancer Inst. 2003;95:1404-1413. [PubMed] |
| 7. | Blot WJ, McLaughlin JK. The changing epidemiology of esophageal cancer. Semin Oncol. 1999;26:2-8. [PubMed] |
| 8. | Lepage C, Rachet B, Jooste V, Faivre J, Coleman MP. Continuing rapid increase in esophageal adenocarcinoma in England and Wales. Am J Gastroenterol. 2008;103:2694-2699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 194] [Article Influence: 11.4] [Reference Citation Analysis (1)] |
| 9. | Blot WJ, Devesa SS, Fraumeni JF. Continuing climb in rates of esophageal adenocarcinoma: an update. JAMA. 1993;270:1320. [PubMed] |
| 10. | Young JL, Percy CL, Asire AJ, Berg JW, Cusano MM, Gloeckler LA, Horm JW, Lourie WI, Pollack ES, Shambaugh EM. Cancer incidence and mortality in the United States, 1973-77. Natl Cancer Inst Monogr. 1981;1-187. [PubMed] |
| 11. | Kwatra KS, Prabhakar BR, Jain S, Grewal JS. Sarcomatoid carcinoma (carcinosarcoma) of the esophagus with extensive areas of osseous differentiation: a case report. Indian J Pathol Microbiol. 2003;46:49-51. [PubMed] |
| 12. | Cook MB. Non-acid reflux: the missing link between gastric atrophy and esophageal squamous cell carcinoma? Am J Gastroenterol. 2011;106:1930-1932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 13. | Dawsey SM, Lewin KJ, Wang GQ, Liu FS, Nieberg RK, Yu Y, Li JY, Blot WJ, Li B, Taylor PR. Squamous esophageal histology and subsequent risk of squamous cell carcinoma of the esophagus. A prospective follow-up study from Linxian, China. Cancer. 1994;74:1686-1692. [PubMed] |
| 14. | Dawsey SM, Lewin KJ, Liu FS, Wang GQ, Shen Q. Esophageal morphology from Linxian, China. Squamous histologic findings in 754 patients. Cancer. 1994;73:2027-2037. [PubMed] |
| 15. | Huang Q, Fang DC, Yu CG, Zhang J, Chen MH. Barrett’s esophagus-related diseases remain uncommon in China. J Dig Dis. 2011;12:420-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 16. | Kountourakis P, Papademetriou K, Ardavanis A, Papamichael D. Barrett’s esophagus: treatment or observation of a major precursor factor of esophageal cancer? J BUON. 2011;16:425-430. [PubMed] |
| 17. | Conteduca V, Sansonno D, Ingravallo G, Marangi S, Russi S, Lauletta G, Dammacco F. Barrett’s esophagus and esophageal cancer: an overview. Int J Oncol. 2012;41:414-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 18. | Hongo M, Nagasaki Y, Shoji T. Epidemiology of esophageal cancer: Orient to Occident. Effects of chronology, geography and ethnicity. J Gastroenterol Hepatol. 2009;24:729-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 271] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 19. | Shaheen N, Ransohoff DF. Gastroesophageal reflux, barrett esophagus, and esophageal cancer: scientific review. JAMA. 2002;287:1972-1981. [PubMed] |
| 20. | Pedrazzani C, Bernini M, Giacopuzzi S, Pugliese R, Catalano F, Festini M, Rodella L, de Manzoni G. Evaluation of Siewert classification in gastro-esophageal junction adenocarcinoma: What is the role of endoscopic ultrasonography? J Surg Oncol. 2005;91:226-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 21. | Suh YS, Han DS, Kong SH, Lee HJ, Kim YT, Kim WH, Lee KU, Yang HK. Should adenocarcinoma of the esophagogastric junction be classified as esophageal cancer? A comparative analysis according to the seventh AJCC TNM classification. Ann Surg. 2012;255:908-915. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 87] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 22. | Schumacher G, Schmidt SC, Schlechtweg N, Roesch T, Sacchi M, von Dossow V, Chopra SS, Pratschke J, Zhukova J, Stieler J. Surgical results of patients after esophageal resection or extended gastrectomy for cancer of the esophagogastric junction. Dis Esophagus. 2009;22:422-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 23. | Hasegawa S, Yoshikawa T, Aoyama T, Hayashi T, Yamada T, Tsuchida K, Cho H, Oshima T, Yukawa N, Rino Y. Esophagus or stomach? The seventh TNM classification for Siewert type II/III junctional adenocarcinoma. Ann Surg Oncol. 2013;20:773-779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 24. | Hosokawa Y, Kinoshita T, Konishi M, Takahashi S, Gotohda N, Kato Y, Daiko H, Nishimura M, Katsumata K, Sugiyama Y. Clinicopathological features and prognostic factors of adenocarcinoma of the esophagogastric junction according to Siewert classification: experiences at a single institution in Japan. Ann Surg Oncol. 2012;19:677-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 90] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 25. | Fang WL, Wu CW, Chen JH, Lo SS, Hsieh MC, Shen KH, Hsu WH, Li AF, Lui WY. Esophagogastric junction adenocarcinoma according to Siewert classification in Taiwan. Ann Surg Oncol. 2009;16:3237-3244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 49] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 26. | Yoon HY, Kim HI, Kim CB. [Clinicopathologic characteristics of adenocarcinoma in cardia according to Siewert classification]. Korean J Gastroenterol. 2008;52:293-297. [PubMed] |
| 27. | Szántó I, Vörös A, Gonda G, Nagy P, Altorjay A, Banai J, Gamal EM, Cserepes E. [Siewert-Stein classification of adenocarcinoma of the esophagogastric junction]. Magy Seb. 2001;54:144-149. [PubMed] |
| 28. | Ku GY, Ilson DH. Adjuvant therapy in esophagogastric adenocarcinoma: controversies and consensus. Gastrointest Cancer Res. 2012;5:85-92. [PubMed] |
| 29. | Fein R, Kelsen DP, Geller N, Bains M, McCormack P, Brennan MF. Adenocarcinoma of the esophagus and gastroesophageal junction. Prognostic factors and results of therapy. Cancer. 1985;56:2512-2518. [PubMed] |
| 30. | Patel AN, Preskitt JT, Kuhn JA, Hebeler RF, Wood RE, Urschel HC. Surgical management of esophageal carcinoma. Proc (Bayl Univ Med Cent). 2003;16:280-284. [PubMed] |
| 31. | McCollum AD, Wu B, Clark JW, Kulke MH, Enzinger PC, Ryan DP, Earle CC, Michelini A, Fuchs CS. The combination of capecitabine and thalidomide in previously treated, refractory metastatic colorectal cancer. Am J Clin Oncol. 2006;29:40-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 32. | Jiao X, Krasna MJ. Clinical significance of micrometastasis in lung and esophageal cancer: a new paradigm in thoracic oncology. Ann Thorac Surg. 2002;74:278-284. [PubMed] |
| 33. | Erasmus JJ, Munden RF. The role of integrated computed tomography positron-emission tomography in esophageal cancer: staging and assessment of therapeutic response. Semin Radiat Oncol. 2007;17:29-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 34. | Lordick F, Ebert M, Stein HJ. Current treatment approach to locally advanced esophageal cancer: is resection mandatory? Future Oncol. 2006;2:717-721. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 35. | Hvid-Jensen F, Pedersen L, Drewes AM, Sørensen HT, Funch-Jensen P. Incidence of adenocarcinoma among patients with Barrett’s esophagus. N Engl J Med. 2011;365:1375-1383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 985] [Cited by in RCA: 980] [Article Influence: 70.0] [Reference Citation Analysis (1)] |
| 36. | Cummings LC, Cooper GS. Descriptive epidemiology of esophageal carcinoma in the Ohio Cancer Registry. Cancer Detect Prev. 2008;32:87-92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 37. | Ilson DH. Esophageal cancer chemotherapy: recent advances. Gastrointest Cancer Res. 2008;2:85-92. [PubMed] |
| 38. | Rasool S, A Ganai B, Syed Sameer A, Masood A. Esophageal cancer: associated factors with special reference to the Kashmir Valley. Tumori. 2012;98:191-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 19] [Reference Citation Analysis (0)] |
| 39. | Khuroo MS, Zargar SA, Mahajan R, Banday MA. High incidence of oesophageal and gastric cancer in Kashmir in a population with special personal and dietary habits. Gut. 1992;33:11-15. [PubMed] |
| 40. | Li JY. Epidemiology of esophageal cancer in China. Natl Cancer Inst Monogr. 1982;62:113-120. [PubMed] |
| 41. | Baquet CR, Commiskey P, Mack K, Meltzer S, Mishra SI. Esophageal cancer epidemiology in blacks and whites: racial and gender disparities in incidence, mortality, survival rates and histology. J Natl Med Assoc. 2005;97:1471-1478. [PubMed] |
| 42. | Faivre J, Trama A, De Angelis R, Elferink M, Siesling S, Audisio R, Bosset JF, Cervantes A, Lepage C. Incidence, prevalence and survival of patients with rare epithelial digestive cancers diagnosed in Europe in 1995-2002. Eur J Cancer. 2012;48:1417-1424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 43. | Cesas A, Bagajevas A. [Combined treatment of esophageal cancer: a review]. Medicina (Kaunas). 2004;40 Suppl 1:161-165. [PubMed] |
| 44. | Lepage C, Drouillard A, Jouve JL, Faivre J. Epidemiology and risk factors for oesophageal adenocarcinoma. Dig Liver Dis. 2013;45:625-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 46] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 45. | Blot WJ. Invited commentary: more evidence of increased risks of cancer among alcohol drinkers. Am J Epidemiol. 1999;150:1138-140; discussion 1141. [PubMed] |
| 46. | D’Onofrio V, Bovero E, Iaquinto G. Characterization of acid and alkaline reflux in patients with Barrett’s esophagus. G.O.S.P.E. Operative Group for the study of Esophageal Precancer. Dis Esophagus. 1997;10:16-22; discussion 22-3. [PubMed] |
| 47. | Guanrei Y, Songliang Q. Endoscopic surveys in high-risk and low-risk populations for esophageal cancer in China with special reference to precursors of esophageal cancer. Endoscopy. 1987;19:91-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 34] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 48. | Oze I, Matsuo K, Wakai K, Nagata C, Mizoue T, Tanaka K, Tsuji I, Sasazuki S, Inoue M, Tsugane S. Alcohol drinking and esophageal cancer risk: an evaluation based on a systematic review of epidemiologic evidence among the Japanese population. Jpn J Clin Oncol. 2011;41:677-692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 49. | Lin J, Zeng R, Cao W, Luo R, Chen J, Lin Y. Hot beverage and food intake and esophageal cancer in southern China. Asian Pac J Cancer Prev. 2011;12:2189-2192. [PubMed] |
| 50. | Lagergren J, Lagergren P. Recent developments in esophageal adenocarcinoma. CA Cancer J Clin. 2013;Epub ahead of print. [PubMed] [DOI] [Full Text] |
| 51. | Nieman KM, Romero IL, Van Houten B, Lengyel E. Adipose tissue and adipocytes support tumorigenesis and metastasis. Biochim Biophys Acta. 2013;Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 568] [Article Influence: 47.3] [Reference Citation Analysis (0)] |
| 52. | Duggan C, Onstad L, Hardikar S, Blount PL, Reid BJ, Vaughan TL. Association Between Markers of Obesity and Progression From Barrett’s Esophagus to Esophageal Adenocarcinoma. Clin Gastroenterol Hepatol. 2013;11:934-943. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 97] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 53. | Carr JS, Zafar SF, Saba N, Khuri FR, El-Rayes BF. Risk factors for rising incidence of esophageal and gastric cardia adenocarcinoma. J Gastrointest Cancer. 2013;44:143-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 54. | Löfdahl HE, Lu Y, Lagergren P, Lagergren J. Risk factors for esophageal adenocarcinoma after antireflux surgery. Ann Surg. 2013;257:579-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 55. | Wu C, Kraft P, Zhai K, Chang J, Wang Z, Li Y, Hu Z, He Z, Jia W, Abnet CC. Genome-wide association analyses of esophageal squamous cell carcinoma in Chinese identify multiple susceptibility loci and gene-environment interactions. Nat Genet. 2012;44:1090-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 217] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 56. | Wu C, Hu Z, He Z, Jia W, Wang F, Zhou Y, Liu Z, Zhan Q, Liu Y, Yu D. Genome-wide association study identifies three new susceptibility loci for esophageal squamous-cell carcinoma in Chinese populations. Nat Genet. 2011;43:679-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 243] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 57. | Wang LD, Zhou FY, Li XM, Sun LD, Song X, Jin Y, Li JM, Kong GQ, Qi H, Cui J. Genome-wide association study of esophageal squamous cell carcinoma in Chinese subjects identifies susceptibility loci at PLCE1 and C20orf54. Nat Genet. 2010;42:759-763. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 338] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 58. | Abnet CC, Freedman ND, Hu N, Wang Z, Yu K, Shu XO, Yuan JM, Zheng W, Dawsey SM, Dong LM. A shared susceptibility locus in PLCE1 at 10q23 for gastric adenocarcinoma and esophageal squamous cell carcinoma. Nat Genet. 2010;42:764-767. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 405] [Cited by in RCA: 421] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 59. | Sameer AS, Parray FQ, Dar MA, Nissar S, Banday MZ, Rasool S, Gulzar GM, Chowdri NA, Siddiqi MA. Cyclin D1 G870A polymorphism and risk of colorectal cancer: a case control study. Mol Med Rep. 2013;7:811-815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 60. | Chen B, Cao L, Yang P, Zhou Y, Wu XT. Cyclin D1 (CCND1) G870A gene polymorphism is an ethnicity-dependent risk factor for digestive tract cancers: a meta-analysis comprising 20,271 subjects. Cancer Epidemiol. 2012;36:106-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 61. | Hong Y, Eu KW, Seow-Choen F, Fook-Chong S, Cheah PY. GG genotype of cyclin D1 G870A polymorphism is associated with increased risk and advanced colorectal cancer in patients in Singapore. Eur J Cancer. 2005;41:1037-1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 62. | Ceschi M, Sun CL, Van Den Berg D, Koh WP, Yu MC, Probst-Hensch N. The effect of cyclin D1 (CCND1) G870A-polymorphism on breast cancer risk is modified by oxidative stress among Chinese women in Singapore. Carcinogenesis. 2005;26:1457-1464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 39] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 63. | Grieu F, Malaney S, Ward R, Joseph D, Iacopetta B. Lack of association between CCND1 G870A polymorphism and the risk of breast and colorectal cancers. Anticancer Res. 2003;23:4257-4259. [PubMed] |
| 64. | Zhuo W, Zhang L, Wang Y, Zhu B, Chen Z. Cyclin D1 G870A polymorphism is a risk factor for esophageal cancer among Asians. Cancer Invest. 2012;30:630-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 65. | Hussain S, M Y, Thakur N, Salam I, Singh N, Mir MM, Bhat MA, Siddiqi MA, Das BC, Bharadwaj M. Association of cyclin D1 gene polymorphisms with risk of esophageal squamous cell carcinoma in Kashmir Valley: a high risk area. Mol Carcinog. 2011;50:487-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 66. | Jain M, Kumar S, Lal P, Tiwari A, Ghoshal UC, Mittal B. Role of BCL2 (ala43thr), CCND1 (G870A) and FAS (A-670G) polymorphisms in modulating the risk of developing esophageal cancer. Cancer Detect Prev. 2007;31:225-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 67. | Casson AG, Zheng Z, Evans SC, Geldenhuys L, van Zanten SV, Veugelers PJ, Porter GA, Guernsey DL. Cyclin D1 polymorphism (G870A) and risk for esophageal adenocarcinoma. Cancer. 2005;104:730-739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 68. | Zhang J, Li Y, Wang R, Wen D, Sarbia M, Kuang G, Wu M, Wei L, He M, Zhang L. Association of cyclin D1 (G870A) polymorphism with susceptibility to esophageal and gastric cardiac carcinoma in a northern Chinese population. Int J Cancer. 2003;105:281-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 56] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 69. | Yu C, Lu W, Tan W, Xing D, Liang G, Miao X, Lin D. Lack of association between CCND1 G870A polymorphism and risk of esophageal squamous cell carcinoma. Cancer Epidemiol Biomarkers Prev. 2003;12:176. [PubMed] |
| 70. | Liu G, Cescon DW, Zhai R, Zhou W, Kulke MH, Ma C, Xu W, Su L, Asomaning K, Heist RS. p53 Arg72Pro, MDM2 T309G and CCND1 G870A polymorphisms are not associated with susceptibility to esophageal adenocarcinoma. Dis Esophagus. 2010;23:36-39. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 71. | van der Heiden IP, van der Werf M, Lindemans J, van Schaik RH. Sequencing: not always the “gold standard”. Clin Chem. 2004;50:248-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 72. | Heath EI, Limburg PJ, Hawk ET, Forastiere AA. Adenocarcinoma of the esophagus: risk factors and prevention. Oncology (Williston Park). 2000;14:507-514; discussion 518-520, 522-523. [PubMed] |
| 73. | Chainani-Wu N. Diet and oral, pharyngeal, and esophageal cancer. Nutr Cancer. 2002;44:104-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 48] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 74. | Inadomi JM, Somsouk M, Madanick RD, Thomas JP, Shaheen NJ. A cost-utility analysis of ablative therapy for Barrett’s esophagus. Gastroenterology. 2009;136:2101-2114.e1-6. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 151] [Cited by in RCA: 143] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 75. | Beaumont H, Gondrie JJ, McMahon BP, Pouw RE, Gregersen H, Bergman JJ, Boeckxstaens GE. Stepwise radiofrequency ablation of Barrett’s esophagus preserves esophageal inner diameter, compliance, and motility. Endoscopy. 2009;41:2-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 76. | Pouw RE, Gondrie JJ, Curvers WL, Sondermeijer CM, Ten Kate FJ, Bergman JJ. Successful balloon-based radiofrequency ablation of a widespread early squamous cell carcinoma and high-grade dysplasia of the esophagus: a case report. Gastrointest Endosc. 2008;68:537-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 77. | Pouw RE, Gondrie JJ, Sondermeijer CM, ten Kate FJ, van Gulik TM, Krishnadath KK, Fockens P, Weusten BL, Bergman JJ. Eradication of Barrett esophagus with early neoplasia by radiofrequency ablation, with or without endoscopic resection. J Gastrointest Surg. 2008;12:1627-1636; discussion 1627-1636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 82] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 78. | Lambert R. Endoscopy in screening for digestive cancer. World J Gastrointest Endosc. 2012;4:518-525. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 18] [Cited by in RCA: 27] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 79. | Blot WJ, Fraumeni JF. Time-related factors in cancer epidemiology. J Chronic Dis. 1987;40 Suppl 2:1S-8S. [PubMed] |
| 80. | Xing EP, Yang GY, Wang LD, Shi ST, Yang CS. Loss of heterozygosity of the Rb gene correlates with pRb protein expression and associates with p53 alteration in human esophageal cancer. Clin Cancer Res. 1999;5:1231-1240. [PubMed] |
| 81. | von Brevern MC, Hollstein MC, Cawley HM, De Benedetti VM, Bennett WP, Liang L, He AG, Zhu SM, Tursz T, Janin N. Circulating anti-p53 antibodies in esophageal cancer patients are found predominantly in individuals with p53 core domain mutations in their tumors. Cancer Res. 1996;56:4917-4921. [PubMed] |
| 82. | Casson AG, Mukhopadhyay T, Cleary KR, Ro JY, Levin B, Roth JA. p53 gene mutations in Barrett’s epithelium and esophageal cancer. Cancer Res. 1991;51:4495-4499. [PubMed] |
| 83. | Cross AJ, Freedman ND, Ren J, Ward MH, Hollenbeck AR, Schatzkin A, Sinha R, Abnet CC. Meat consumption and risk of esophageal and gastric cancer in a large prospective study. Am J Gastroenterol. 2011;106:432-442. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 148] [Cited by in RCA: 133] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 84. | Cross AJ, Leitzmann MF, Gail MH, Hollenbeck AR, Schatzkin A, Sinha R. A prospective study of red and processed meat intake in relation to cancer risk. PLoS Med. 2007;4:e325. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 322] [Cited by in RCA: 306] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 85. | Ward MH, Cross AJ, Abnet CC, Sinha R, Markin RS, Weisenburger DD. Heme iron from meat and risk of adenocarcinoma of the esophagus and stomach. Eur J Cancer Prev. 2012;21:134-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 54] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 86. | Ward MH, Sinha R, Heineman EF, Rothman N, Markin R, Weisenburger DD, Correa P, Zahm SH. Risk of adenocarcinoma of the stomach and esophagus with meat cooking method and doneness preference. Int J Cancer. 1997;71:14-19. [PubMed] |
| 87. | Kawaguchi H, Ohno S, Araki K, Miyazaki M, Saeki H, Watanabe M, Tanaka S, Sugimachi K. p53 polymorphism in human papillomavirus-associated esophageal cancer. Cancer Res. 2000;60:2753-2755. [PubMed] |