Published online Jul 14, 2013. doi: 10.3748/wjg.v19.i26.4137
Revised: May 22, 2013
Accepted: June 8, 2013
Published online: July 14, 2013
Processing time: 135 Days and 16.8 Hours
AIM: To investigate the effects of rhein on intestinal epithelial tight junction proteins in rats with IgA nephropathy (IgAN).
METHODS: Twenty-eight female Sprague-Dawley rats were randomly divided into four groups (7 per group): Control, IgAN, Rhein-treated, and Rhein-prevented. Bovine serum albumin, lipopolysaccharide and CCl4 were used to establish the rat model of IgA nephropathy. The Rhein-treated group was given rhein from week 7 until the rats were sacrificed. The Rhein-prevented group was given rhein from week 1. Animals were sacrificed at the end of week 10. We observed the changes in the intestinal epithelial tight junctions using transmission electron microscopy, and expression of intestinal epithelial tight junction proteins zona occludens protein (ZO)-1 and occludin by immunofluorescence using laser confocal microscopy. Changes in mRNA and protein expression of ZO-1 and occludin were measured by reverse transcriptase polymerase chain reaction and Western blotting. The ratio of urinary lactulose/mannitol was measured by high performance liquid chromatography (HPLC) for assessing the intestinal permeability.
RESULTS: In the control group, the tight junctions lied between epithelial cells on the top of the outer side of the cell membrane, and appeared in dense dotted crystal structures, the neighboring cells were binded tightly with no significant gap, and the tight junction protein ZO-1 and occludin were evenly distributed in the intestinal epithelial cells at the top of the junction. Compared with the control group, in the IgAN group, the structure of the tight junction became obscured and the dotted crystal structures had disappeared; the fluorescence of ZO-1 and occludin was uneven and weaker (5.37 ± 1.27 vs 10.03 ± 1.96, P < 0.01; 4.23 ± 0.85 vs 12.35 ± 4.17, P < 0.01); the mRNA expression of ZO-1 and occludin decreased (0.42 ± 0.19 vs 0.92 ± 0.24, P < 0.01; 0.40 ± 0.15 vs 0.97 ± 0.25, P < 0.01); protein expression of ZO-1 and occludin was decreased (0.85 ± 0.12 vs 1.98 ± 0.43, P < 0.01; 0.72 ± 0.15 vs 1.38 ± 0.31, P < 0.01); and the ratio of urinary lactulose/mannitol increased (3.55 ± 0.68 vs 2.72 ± 0.21, P < 0.01). In the Rhein-prevented and Rhein-treated groups, compared with the IgAN group, the intestinal epithelial tight junctions were repaired; fluorescence of ZO-1 and occludin was stronger (11.16 ± 3.52 and 8.81 ± 2.30 vs 5.37 ± 1.27, P < 0.01; 10.97 ± 3.40 and 9.46 ± 2.40 vs 4.23 ± 0.85, P < 0.01); mRNA of ZO-1 and occludin increased (0.81 ± 0.17 and 0.64 ± 0.16 vs 0.42 ± 0.19, P < 0.01; 0.82 ± 0.22 and 0.76 ± 0.31 vs 0.40 ± 0.15, P < 0.01); protein expression of ZO-1 and occludin was increased (2.07 ± 0.41 and 1.57 ± 0.23 vs 0.85 ± 0.12, P < 0.01; 1.34 ± 0.21 and 1.15 ± 0.17 vs 0.72 ± 0.15, P < 0.01); and the ratio of urinary lactulose/mannitol decreased (2.83 ± 0.43 and 2.87 ± 0.18 vs 3.55 ± 0.68, P < 0.01).
CONCLUSION: Rhein can enhance the expression of ZO-1 and occludin, repair damaged tight junctions, and protect the intestinal barrier.
Core tip: It has been reported that the incidence and aggravation of IgA nephropathy (IgAN) are often accompanied with intestinal mucosal damage. We speculate that various factors cause the destruction of the intestinal mucosal barrier, food proteins activate the mucosal immune system, and a large amount of secretory IgA is deposited in kidney and causes IgAN. Rhubarb has a protective effect on the intestine. Rhein is isolated from rhubarb and we speculate that it also has a protective effect, although this has not been reported to date. We used various biochemical approaches to confirm this.
- Citation: Peng SN, Zeng HH, Fu AX, Chen XW, Zhu QX. Effects of rhein on intestinal epithelial tight junction in IgA nephropathy. World J Gastroenterol 2013; 19(26): 4137-4145
- URL: https://www.wjgnet.com/1007-9327/full/v19/i26/4137.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i26.4137
IgA nephropathy (IgAN) is the most common primary glomerular disease worldwide[1]. Although the etiology and pathogenesis of IgAN are still not clear, and it lacks effective treatment, the incidence and aggravation of IgAN are often accompanied with intestinal mucosal damage[2]. We speculate that various virulence factors cause destruction of the intestinal mucosal barrier, the permeability of the intestinal mucosa increases, food proteins activate the intestinal mucosal immune system, and a large amount of secretory IgA is produced and deposited in the kidney, which causes renal damage and IgAN. We suggest that the protection of the intestinal mucosal barrier can decrease the permeability of intestinal mucosa and prevent or reduce the occurrence of IgAN.
It has been reported that rhubarb has a protective effect on the intestinal mucosal barrier[3]. Rhein (1.8-dihydroxy-3-carboxy-anthraquinone, CAS number: 478-43-3) is an anthraquinone monomer isolated from rhubarb, and we speculate that it may also have a protective effect on the intestinal mucosal barrier and delay or prevent the course of IgAN. The function of the intestinal mucosal barrier mainly depends on the integrity of the tight junction proteins in the intestinal epithelial cells. A decrease in tight junction proteins increases intestinal permeability and leads to dysfunction of the intestinal mucosal barrier[4]. As far as we are aware, a protective effect of rhein on the intestinal epithelial tight junction proteins in rats with IgAN has not yet been reported. Therefore, we used various biochemical approaches to determine how rhein regulates the expression of intestinal epithelial tight junction proteins in IgAN.
Rhein (> 95% purity) was extracted and identified by Chengdu Mansite Pharmaceutical Co. Ltd. (batch number: MUST-11032801; China). Antibodies against occludin were purchased from Abcam (Cambridge, United Kingdom). Antibodies against zonula occludens protein (ZO)-1 were purchased from Invitrogen (Carlsbad, CA). Bovine serum albumin (BSA) was purchased from Roche (Mannheim, Germany). Lipopolysaccharide (LPS), lactulose and mannitol were purchased from Sigma (St Louis, MO). Carbon tetrachloride and castor oil were purchased from Shanghai Reagents (China). Antibodies against β-actin, horseradish-peroxidase-conjugated secondary antibodies and fluorescein isothiocyanate (FITC)-conjugated secondary antibodies were purchased from Beijing Zhongshan (China). Trizol and reverse transcriptase polymerase chain reaction (RT-PCR) kit were purchased from Transgen (Beijing, China).
Twenty-eight female Sprague-Dawley rats weighing 180-220 g were obtained from the Animal Center of Nanchang University. They were housed in the animal facilities of the Nanchang University, with free access to food and water. Animals were treated humanely by use of protocols that were approved by the Institutional Animal Use and Care Committee of Nanchang University. Rats were divided randomly into the control group, IgAN group, Rhein-prevented group, and Rhein-treated group (n = 7 each). The IgAN experimental animal model was established by treatment with BSA, LPS and CCl4[5], and specific implementation was as follows: BSA (400 mg/kg, oral every other day) for 6 wk plus LPS (0.05 mg, intravenous injection at wk 6 and 8) and CCl4 (0.1 mL dissolved in 0.5 mL castor oil, subcutaneous injection weekly for 9 wk). The Rhein-treated group was given rhein (100 mg/kg per day)[6] from week 7 until sacrifice. The Rhein-prevented group was given rhein (100 mg/kg per day) from week 1. The control and IgAN groups were given the same volume of normal saline. All the rats were sacrificed at week 10.
Seven rats per group were analyzed by transmission electron microscopy (TEM) (H-600). Pieces of ileum, 2 mm × 2 mm, were fixed in 2.5% glutaraldehyde overnight at 4°C. The fixed tissues were then post-fixed in 1% osmium tetroxide for 2 h and then rinsed and stored in 0.1 mol/L sodium cacodylate buffer containing 6% sucrose for 12 h. The pieces of ileum were dehydrated through a graded acetone series and embedded in epoxy resin. Semi-thin sections (1.5 μm) were cut and stained with toluidine blue. Ultra-thin sections were stained with 4% uranyl acetate solution in 50% ethanol and lead citrate and then the intestinal epithelial tight junctions were examined by TEM.
Seven rats per group were analyzed by immunofluorescence. Pieces of ileum, 5 mm × 5 mm, were frozen in liquid nitrogen and 10-μm frozen sections were cut. The frozen sections were fixed with cold acetone for 10 min at 4 °C. After extensive washing three times (5 min per wash) with cold PBS, the frozen sections were blocked with 10% normal sheep serum in PBS and then incubated with the antibodies against occludin (1:200, Abcam) and ZO-1 (1:100, Invitrogen) at 4 °C overnight, followed by staining with FITC-conjugated secondary antibodies. Stained frozen sections were examined with a laser confocal microscope equipped with a digital camera, identifying occludin and ZO-1 by light green color (excitation light wave length of 490 nm). Stained frozen sections were analyzed by a morphological analysis system to determine semi-quantitatively the expression of occludin and ZO-1. Five visual fields were randomly observed under high magnification, with two sections selected from each specimen. The integrated optical density of the positive material in each visual field and its area were measured by morphological analysis system; the ratio of which showed the relative content of occludin and ZO-1.
Five rats per group were analyzed by RT-PCR. RT-PCR was used for mRNA detection and semi-quantitative assessment. Total RNA was extracted from the small intestine using Trizol reagent (Transgen), measured and verified with a UV spectrophotometer. cDNA was synthesized using an One-Step RT-PCR kit (Transgen) from 1 μg total RNA. Primers were designed by the Primer Premier 5.0 software (Premier Biosoft International, Palo Alto, CA) according to mRNA sequences (by GenBank) of occludin, ZO-1 and β-actin (as control). The sequences of primers were as follows: forward primer of β-actin gene was 5’-TCAGGTCATCACTATCGGCAAT-3’ and its reverse primer was 5’-AAAGAAAGGGTGT AAAACGCA-3’. The forward primer of the occludin gene was 5’-TGCGTGGCTTCCACACTT GCT-3’ and its reverse primer was 5’-TTTGCCGCTCTGGGGTCTGT-3’. The forward primer of the ZO-1 gene was 5’-TGCCCGGCCATTTGAACGCA-3’ and its reverse primer was 5’-TCAGG CGGCTGTGTGGAACT-3’. The PCR products were separated by electrophoresis on a 2% agarose gel stained with ethidium bromide to confirm that products of the expected size were detected. The electrophoretic bands were analyzed using a gel image analysis system. The results were normalized to the respective β-actin expression. RT-PCR experiments were repeated twice.
Five rats per group were analyzed by Western blotting. For Western blotting, the small intestines were frozen in liquid nitrogen until further use. Protein extraction was carried out using the RIPA lysate (Solarbio, Beijing, China). Protein (20-50 μg per lane) was separated by SDS-PAGE. Occludin was separated on 10% gel and ZO-1 on 8% gel. Proteins were transblotted to polyvinylidene difluoride membranes (Solarbio) in standard Tris-glycine transfer buffer, pH 8.3, containing 0.5% SDS. After transfer, membranes were blocked for 1 h at room temperature in TBST (10 mmol/L Tris-HCl, pH 8.0, 150 mmol/L NaCl, 0.2% Tween-20) containing 5% non-fat milk powder, and incubated overnight at 4 °C with either anti-occludin (Abcam) or anti-ZO-1 (Invitrogen) diluted 1:200 in TBST containing 1% non-fat milk powder. Membranes were then washed in TBST for 30 min, incubated with horseradish-peroxidase-conjugated goat anti-rabbit IgG, diluted 1:5000 (Beijing Zhongshan, China) in TBST, washed in TBST for 30 min, and resolved by chemiluminescence (Thermo, Waltham, MA). All membranes were stripped and reprobed with anti-β-actin antibodies (Beijing Zhongshan) as loading controls. Intensities of immunoreactive bands were quantified by densitometry, and normalized to the respective β-actin content. Western blotting experiments were repeated twice.
Intestinal permeability was determined using two non-metabolized sugars. Three grams lactulose and 1.5 g mannitol were dissolved in 60 mL distilled water. After a fasting period of 12 h, all animals received 2 mL lactulose/mannitol solution by orogastric tube. One hour after feeding, the 6-h urine was collected using metabolic cages before sacrifice. The ratio of urine concentrations of lactulose and mannitol was measured to assess the intestinal permeability.
All measurement data were expressed as mean ± SE. Statistical analysis was performed using SPSS 17.0 software. Comparison between groups was made using one-way analysis of variance followed by Student-Newman-Keuls test. P < 0.05 was considered to be statistically significant.
ZO-1 and occludin are important components in tight junctions, so we performed a morphological analysis of the junctions (Figure 1). Tight junctions are belt-shaped and expand around the apex of epithelial cells. TEM indicated that the cell membrane was intact, and distinct junction complexes were observed in the Control Group. However, in the IgAN Group, the structure of the tight junctions became obscured and the dotted crystal structures disappeared. The microvilli were sparse with irregular length and arrangement. The situations in rhein-treated group and rhein-prevented group were improved compared with IgAN group. The intestinal epithelial tight junctions were repaired with respect to structural integration, with close intercellular connection and high electron density.
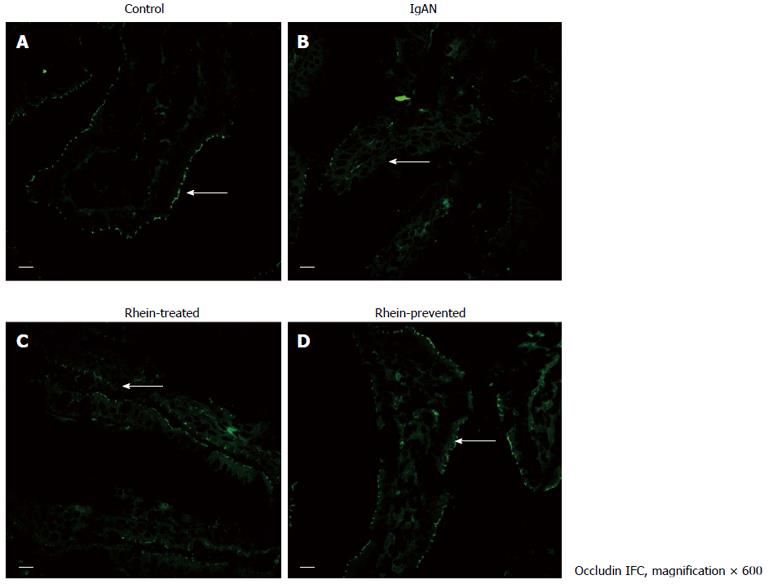
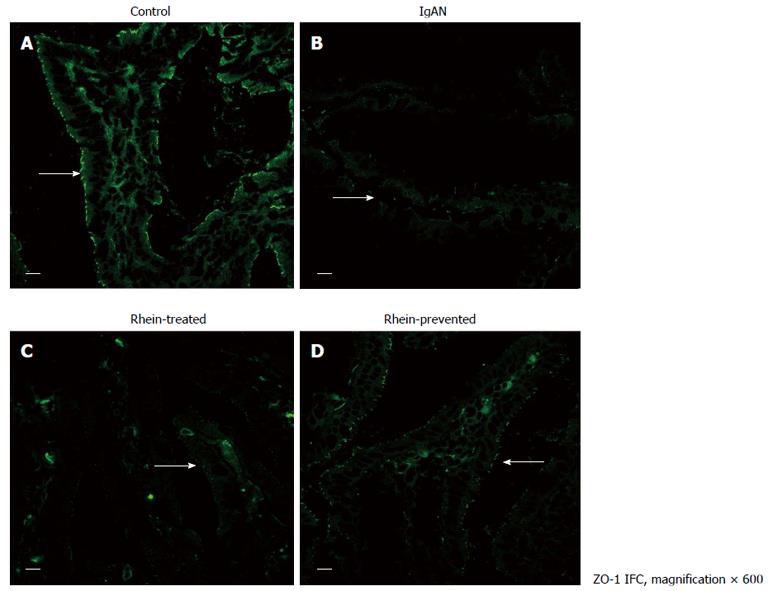
Indirect immunofluorescence staining for occludin (Figure 2; intestinal epithelium by occludin immunofluorescence staining) and ZO-1 (Figure 3; intestinal epithelium by ZO-1 immunofluorescence staining) was performed. In the control group, occludin and ZO-1 staining was found at the apical part of the lateral membranes of the polar epithelial cells and distributed continuously, similar to an intestinal mechanical barrier. In the IgAN Group, the green signals were intermittent and markedly weaker than those in the Control group (P < 0.01), and the integrity of the barrier was damaged. The condition of the Rhein-treated and Rhein-prevented groups was ameliorated compared with the IgAN group (P < 0.01). The green signals of ZO-1 in the Rhein-prevented group were stronger than in the Rhein-treated group (P < 0.01). In contrast, no change in the green signals of occludin was observed between these two groups (Table 1).
| Control group | IgAN group | Rhein-treated group | Rhein-prevented group | |
| In immunofluorescence (× 10-2) | ||||
| Occludin | 12.35 ± 4.17 | 4.23 ± 0.85b | 9.46 ± 2.40bd | 10.97 ± 3.40d |
| ZO-1 | 10.03 ± 1.96 | 5.37 ± 1.27b | 8.81 ± 2.30d | 11.16 ± 3.52df |
| In reverse transcriptase polymerase chain reaction | ||||
| Occludin | 0.97 ± 0.25 | 0.40 ± 0.15b | 0.76 ± 0.31ad | 0.82 ± 0.22d |
| ZO-1 | 0.92 ± 0.24 | 0.42 ± 0.19b | 0.64 ± 0.16bd | 0.81 ± 0.17de |
Occludin, ZO-1 and β-actin RNAs were 170, 150 and 432 bp long, respectively. RT-PCR semi-quantitative analyses showed that the levels of occludin and ZO-1 expression in the IgAN group were significantly lower than in the control group (P < 0.01). In the Rhein-treated and Rhein-prevented group, the levels were markedly higher than in the IgAN Group (P < 0.01). The level of ZO-1 expression in the Rhein-prevented group was higher than in the Rhein-treated group (P < 0.05). In contrast, no change in occludin expression was observed between these groups (Figure 4 and Table 1).
Western blotting analysis showed that occludin and ZO-1 protein expression decreased significantly in the IgAN group compared with the control group (P < 0.01). In the Rhein-treated and Rhein-prevented groups, occludin and ZO-1 protein expression was higher than in the IgAN group (P < 0.01). ZO-1 protein expression in the Rhein-prevented group was higher than in the Rhein-treated group (P < 0.01). However, no change in occludin protein expression was observed between the groups (Figure 5 and Table 2). These findings are consistent with the immunofluorescence results.
The intestinal permeability was assessed by differential uptake of lactulose and mannitol in all four groups. Measurement of mannitol and lactulose by HPLC showed that the ratio of urinary lactulose/mannitol increased in the IgAN group compared with the control group (P < 0.01), indicating an increase of intestinal permeability. In the Rhein-treated and Rhein-prevented groups, the ratio of urinary lactulose/mannitol decreased compared with the IgAN group (P < 0.05), indicating decreased intestinal permeability. The decrease in intestinal permeability in the Rhein-prevented group was more obvious than that in the Rhein-treated group (P < 0.05) (Table 3).
IgAN is defined as the predominant deposition of IgA in the glomerular mesangium[7]. The etiology of IgAN has not been completely clarified, but one hypothesis involves the stimulation of antigen by an intestinal route causing an increase in IgA production in the intestinal mucosa, such as ulcerative colitis or Crohn’s disease[8,9]. In recent years, an IgAN animal model induced by oral immunization has supported this hypothesis[10].
It has been reported that the permeability of the intestinal mucosa of IgAN patients is significantly higher than normal[11]. This is consistent with the experimental results, but its mechanism is not yet clear. Intestinal mucosal permeability is closely related to the integrity of the intestinal barrier. Mucosal barrier plays an important role in protecting the body from food antigens, microorganisms and their harmful metabolites[12]. The mucosal barrier includes mechanical, immune, chemical and biological barriers, among which, the mechanical barrier is essential for maintaining the integrity of the intestinal barrier. It is mainly composed of the intestinal epithelial cells and cellular junctions among them. The function of the intestinal barrier is affected by the morphology and number of epithelial cells and cellular junctions[13]. The cellular junctions include tight junctions, intermediate junctions, desmosomes, and gap junctions, and tight junctions are closely related to the mechanical barrier[14]. Tight junctions, or ZO, expand around the apex of epithelial cells and form a semipermeable barrier in the paracellular pathway in most vertebrate epithelia[15]. Disruption of the tight junctions can cause increased permeability and leakiness[16,17]. Three groups of macromolecules are considered as integral components of the tight junctions: occludins, claudins and junction adhesion molecules[18]. ZO-1 is the major tight junction protein that binds to the intracellular domain of occludins[19]. The interaction between occludin and ZO-1 plays a crucial role in maintaining the structure of tight junctions and epithelial barrier function[20,21]. Therefore, detection of occludin and ZO-1 reflects the condition of the tight junctions and intestinal mucosa barrier.
Our results showed that expression of intestinal epithelial tight junction proteins occludin and ZO-1 was significantly reduced in the IgAN Group. Therefore, we hypothesized that increased intestinal permeability in that group might be related to the decrease in expression of intestinal epithelial tight junction proteins. This decrease may be due to excess secretion of inflammatory cytokines, such as tumor necrosis factor (TNF)-α, interferon (IFN)-γ, NO, and oxygen free radicals, which is caused by intestinal antigen stimulation in the modeling process[22,23]. It has been reported that TNF-α affects the interaction of occludin and ZO-1 with components of the actin cytoskeleton[24]. Moreover, a series of recent reports has indicated that TNF-α disrupts tight junction assembly and decreases expression of ZO-1[25,26]. The synergy between TNF-α and IFN-γ can downregulate occludin expression of the occludin promoter[27].
Increased expression of tight junction proteins improves intestinal mucosal barrier function[28,29]. It has been reported that rhubarb can promote intestinal mucosal barrier function recovery and alleviate intestinal bacterial translocation in animal models of burns[30]. Therefore, we speculated that rhein, as the main pharmacological component of rhubarb, would also have a protective effect on the intestinal mucosal barrier and delay or prevent progression of IgAN. Western blotting showed that occludin and ZO-1 protein expression in the Rhein-treated and Rhein-prevented Groups was higher than that in the IgAN Group, which was consistent with the immunofluorescence and RT-PCR results. The protective effect of rhein on tight junction proteins may be associated with the following aspects. First, rhein lowers the activity of macrophages and inhibits the secretion of TNF-α and other inflammatory cytokines that damage the structure and function of tight junctions[31]. Second, rhein has a positive effect on the peristaltic reflex of the small intestine[32], speeds up the excretion of intestinal bacteria and LPS, and reduces LPS-induced secretion of inflammatory cytokines. Third, reactive oxygen species destroy tight junction proteins by affecting the signal transduction pathway[33]. Rhein can remove reactive oxygen species and alleviate oxidative damage[34]. Lastly, dysfunction of the intestinal microcirculation results in structural damage of tight junctions[35]. Rhein inhibits intestinal microvascular endothelial cell secretion of NO, endothelin-1 and other vasoconstrictor substances, which improves the intestinal microcirculation[36].
In summary, rhein reduces intestinal permeability by protecting intestinal epithelial tight junction proteins ZO-1 and occludin, which alleviates the damage to the intestinal mucosa in IgAN. In this regard, rhein may be a potential therapeutic agent for protecting the intestinal mucosa in IgAN.
IgA nephropathy (IgAN) is the most common primary glomerular disease worldwide. The etiology and pathogenesis of IgAN are still not clear, and it lacks effective treatment. The incidence and aggravation of IgAN are often accompanied with damage to the intestinal mucosa. Rhein is an anthraquinone monomer isolated from rhubarb. It has been reported that rhubarb has a protective effect on the intestinal mucosal barrier in burns and pancreatitis, but a protective effect on the intestinal mucosal barrier in IgAN has not yet been reported.
The intestinal mucosal barrier is an important area in research related to the etiology and pathogenesis of IgAN. The research hotspot is how to protect the intestinal mucosal barrier to prevent the occurrence of IgAN.
It has been reported previously that rhubarb has a protective effect on the intestinal mucosal barrier in burns and pancreatitis. Rhein is one of the anthraquinone monomers isolated from rhubarb. The protective effect of rhein on the intestinal mucosal barrier in IgAN has not yet been reported. The authors observed the protective effect of rhein on the intestinal mucosa in a rat model of IgAN; further demonstrated the pathogenesis of intestinal mucosal barrier injury in IgAN; and showed that intestinal protection and repair play an important role in the prevention and treatment of IgAN.
The results suggest that rhein is a potential therapeutic material that could be used in protecting intestinal mucosa barrier and preventing and treating IgAN.
Rhein (1.8-dihydroxy-3-carboxy-anthraquinone, CAS number: 478-43-3) is a substance in the anthraquinone group obtained from rhubarb. Originally the rhubarb plant was used as a laxative, and it was believed that rhein along with other anthraquinone glycosides imparted this activity.
This was a good descriptive study in which the authors analyzed the preventive effect of rhein on the intestinal mucosa barrier in rats with IgAN. The results are interesting and suggest that rhein is a potential therapeutic substance that could be used in protecting the intestinal mucosa and preventing IgAN.
P- Reviewer Rampoldi L S- Editor Zhai HH L- Editor A E- Editor Zhang DN
| 1. | Lai KN. Pathogenesis of IgA nephropathy. Nat Rev Nephrol. 2012;8:275-283. [PubMed] |
| 2. | Suzuki H, Kiryluk K, Novak J, Moldoveanu Z, Herr AB, Renfrow MB, Wyatt RJ, Scolari F, Mestecky J, Gharavi AG. The pathophysiology of IgA nephropathy. J Am Soc Nephrol. 2011;22:1795-1803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 429] [Cited by in RCA: 596] [Article Influence: 42.6] [Reference Citation Analysis (0)] |
| 3. | Mao XB, Wang SQ, Mao Y. [Effects of rhubarb on the intestinal banier function of patients with acute myocardial infarction-heart]. Zhongguo Zhong Xi Yi Jie He Zazhi. 2012;32:1046-1050. [PubMed] |
| 4. | Yang DH, Ye ZY, Xie YJ, He XJ, Xu WJ, Zhou WM. Effect of salvianolate on intestinal epithelium tight junction protein zonula occludens protein 1 in cirrhotic rats. World J Gastroenterol. 2012;18:7040-7047. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 6] [Cited by in RCA: 9] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 5. | Xing L, Bai L, Yu CY, Xie RJ. [Effect of telmisartan on tubulointerstitial injury and expression of PPARγ in rat renal tissue of IgA nephropathy model]. Zhonghua Yi Xue Zazhi. 2010;90:2860-2863. [PubMed] |
| 6. | Guo MZ, Li XS, Xu HR, Mei ZC, Shen W, Ye XF. Rhein inhibits liver fibrosis induced by carbon tetrachloride in rats. Acta Pharmacol Sin. 2002;23:739-744. [PubMed] |
| 7. | Coppo R, Cattran D, Roberts Ian SD, Troyanov S, Camilla R, Cook T, Feehally J. The new Oxford Clinico-Pathological Classification of IgA nephropathy. Prilozi. 2010;31:241-248. [PubMed] |
| 8. | Ku E, Ananthapanyasut W, Campese VM. IgA nephropathy in a patient with ulcerative colitis, Graves’ disease and positive myeloperoxidase ANCA. Clin Nephrol. 2012;77:146-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Choi JY, Yu CH, Jung HY, Jung MK, Kim YJ, Cho JH, Kim CD, Kim YL, Park SH. A case of rapidly progressive IgA nephropathy in a patient with exacerbation of Crohn’s disease. BMC Nephrol. 2012;13:84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 10. | Emancipator SN. Prospects and perspectives on IgA nephropathy from animal models. Contrib Nephrol. 2011;169:126-152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 11. | Kovács T, Kun L, Schmelczer M, Wagner L, Davin JC, Nagy J. Do intestinal hyperpermeability and the related food antigens play a role in the progression of IgA nephropathy I. Study of intestinal permeability. Am J Nephrol. 1996;16:500-505. [PubMed] |
| 12. | Camilleri M, Madsen K, Spiller R, Greenwood-Van Meerveld B, Verne GN. Intestinal barrier function in health and gastrointestinal disease. Neurogastroenterol Motil. 2012;24:503-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 459] [Cited by in RCA: 639] [Article Influence: 49.2] [Reference Citation Analysis (0)] |
| 13. | Ma Y, Semba S, Khan RI, Bochimoto H, Watanabe T, Fujiya M, Kohgo Y, Liu Y, Taniguchi T. Focal adhesion kinase regulates intestinal epithelial barrier function via redistribution of tight junction. Biochim Biophys Acta. 2013;1832:151-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 14. | Vaziri ND, Yuan J, Norris K. Role of urea in intestinal barrier dysfunction and disruption of epithelial tight junction in chronic kidney disease. Am J Nephrol. 2013;37:1-6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 278] [Cited by in RCA: 280] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 15. | Nasu Y, Ido A, Tanoue S, Hashimoto S, Sasaki F, Kanmura S, Setoyama H, Numata M, Funakawa K, Moriuchi A. Hepatocyte growth factor stimulates the migration of gastric epithelial cells by altering the subcellular localization of the tight junction protein ZO-1. J Gastroenterol. 2013;48:193-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 16. | Hu CH, Xiao K, Luan ZS, Song J. Early weaning increases intestinal permeability, alters expression of cytokine and tight junction proteins, and activates mitogen-activated protein kinases in pigs. J Anim Sci. 2013;91:1094-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 296] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 17. | Scudamore CL, Jepson MA, Hirst BH, Miller HR. The rat mucosal mast cell chymase, RMCP-II, alters epithelial cell monolayer permeability in association with altered distribution of the tight junction proteins ZO-1 and occludin. Eur J Cell Biol. 1998;75:321-330. [PubMed] |
| 18. | Ulluwishewa D, Anderson RC, McNabb WC, Moughan PJ, Wells JM, Roy NC. Regulation of tight junction permeability by intestinal bacteria and dietary components. J Nutr. 2011;141:769-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 754] [Cited by in RCA: 855] [Article Influence: 61.1] [Reference Citation Analysis (1)] |
| 19. | Van Itallie CM, Fanning AS, Bridges A, Anderson JM. ZO-1 stabilizes the tight junction solute barrier through coupling to the perijunctional cytoskeleton. Mol Biol Cell. 2009;20:3930-3940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 354] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 20. | Noth R, Lange-Grumfeld J, Stüber E, Kruse ML, Ellrichmann M, Häsler R, Hampe J, Bewig B, Rosenstiel P, Schreiber S. Increased intestinal permeability and tight junction disruption by altered expression and localization of occludin in a murine graft versus host disease model. BMC Gastroenterol. 2011;11:109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 67] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 21. | Tash BR, Bewley MC, Russo M, Keil JM, Griffin KA, Sundstrom JM, Antonetti DA, Tian F, Flanagan JM. The occludin and ZO-1 complex, defined by small angle X-ray scattering and NMR, has implications for modulating tight junction permeability. Proc Natl Acad Sci USA. 2012;109:10855-10860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 50] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 22. | Bandyopadhaya A, Sarkar M, Chaudhuri K. Transcriptional upregulation of inflammatory cytokines in human intestinal epithelial cells following Vibrio cholerae infection. FEBS J. 2007;274:4631-4642. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 23. | Treede I, Braun A, Jeliaskova P, Giese T, Füllekrug J, Griffiths G, Stremmel W, Ehehalt R. TNF-alpha-induced up-regulation of pro-inflammatory cytokines is reduced by phosphatidylcholine in intestinal epithelial cells. BMC Gastroenterol. 2009;9:53. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 67] [Cited by in RCA: 84] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 24. | Fischer A, Gluth M, Pape UF, Wiedenmann B, Theuring F, Baumgart DC. Adalimumab prevents barrier dysfunction and antagonizes distinct effects of TNF-α on tight junction proteins and signaling pathways in intestinal epithelial cells. Am J Physiol Gastrointest Liver Physiol. 2013;304:G970-G979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 95] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 25. | Poritz LS, Garver KI, Tilberg AF, Koltun WA. Tumor necrosis factor alpha disrupts tight junction assembly. J Surg Res. 2004;116:14-18. [PubMed] |
| 26. | Song HL, Lv S, Liu P. The roles of tumor necrosis factor-alpha in colon tight junction protein expression and intestinal mucosa structure in a mouse model of acute liver failure. BMC Gastroenterol. 2009;9:70. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 45] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 27. | Mankertz J, Tavalali S, Schmitz H, Mankertz A, Riecken EO, Fromm M, Schulzke JD. Expression from the human occludin promoter is affected by tumor necrosis factor alpha and interferon gamma. J Cell Sci. 2000;113:2085-2090. [PubMed] |
| 28. | Suzuki T, Hara H. Quercetin enhances intestinal barrier function through the assembly of zonula [corrected] occludens-2, occludin, and claudin-1 and the expression of claudin-4 in Caco-2 cells. J Nutr. 2009;139:965-974. [PubMed] |
| 29. | Hu C, Song J, Li Y, Luan Z, Zhu K. Diosmectite-zinc oxide composite improves intestinal barrier function, modulates expression of pro-inflammatory cytokines and tight junction protein in early weaned pigs. Br J Nutr. 2013;11:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 74] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 30. | Meng YB, Lei J, Hao ZM, Cao RL. [Influence of rhubarb on gastrointestinal motility and intestinal mucosal barrier in patients with severe burn]. Zhonghua Shao Shang Zazhi. 2011;27:337-340. [PubMed] |
| 31. | Cong XD, Ding MJ, Dai DZ, Wu Y, Zhang Y, Dai Y. ER stress, p66shc, and p-Akt/Akt mediate adjuvant-induced inflammation, which is blunted by argirein, a supermolecule and rhein in rats. Inflammation. 2012;35:1031-1040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 45] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 32. | Nijs G, de Witte P, Geboes K, Meulemans A, Schuurkes J, Lemli J. In vitro demonstration of a positive effect of rhein anthrone on peristaltic reflex of guinea pig ileum. Pharmacology. 1993;47 Suppl 1:40-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 33. | Schreibelt G, Kooij G, Reijerkerk A, van Doorn R, Gringhuis SI, van der Pol S, Weksler BB, Romero IA, Couraud PO, Piontek J. Reactive oxygen species alter brain endothelial tight junction dynamics via RhoA, PI3 kinase, and PKB signaling. FASEB J. 2007;21:3666-3676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 281] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 34. | Zhong XF, Huang GD, Luo T, Deng ZY, Hu JN. Protective effect of rhein against oxidative stress-related endothelial cell injury. Mol Med Rep. 2012;5:1261-1266. [PubMed] |
| 35. | Nakajima Y, Baudry N, Duranteau J, Vicaut E. Microcirculation in intestinal villi: a comparison between hemorrhagic and endotoxin shock. Am J Respir Crit Care Med. 2001;164:1526-1530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 88] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 36. | Pelletier JP, Mineau F, Fernandes JC, Duval N, Martel-Pelletier J. Diacerhein and rhein reduce the interleukin 1beta stimulated inducible nitric oxide synthesis level and activity while stimulating cyclooxygenase-2 synthesis in human osteoarthritic chondrocytes. J Rheumatol. 1998;25:2417-2424. [PubMed] |