Published online Jun 28, 2013. doi: 10.3748/wjg.v19.i24.3824
Revised: February 5, 2013
Accepted: March 23, 2013
Published online: June 28, 2013
Processing time: 267 Days and 6.4 Hours
AIM: To investigate the characteristics of mucosal lesions and their relation to laboratory data and long-term follow up in breast-fed infants with allergic colitis.
METHODS: In this study 31 breast-fed infants were prospectively evaluated (mean age, 17.4 wk) whose rectal bleeding had not ceased after a maternal elimination diet for cow’s milk. Thirty-four age-matched and breast-fed infants (mean age, 16.9 wk) with no rectal bleeding were enrolled for laboratory testing as controls. Laboratory findings, colonoscopic and histological characteristics were prospectively evaluated in infants with rectal bleeding. Long-term follow-up with different nutritional regimes (L-amino-acid based formula or breastfeeding) was also included.
RESULTS: Iron deficiency, peripheral eosinophilia and thrombocytosis were significantly higher in patients with allergic colitis in comparison to controls (8.4 ± 3.2 μmol/L vs 13.7 ± 4.7 μmol/L, P < 0.001; 0.67 ± 0.49 G/L vs 0.33 ± 0.17 G/L, P < 0.001; 474 ± 123 G/L vs 376 ± 89 G/L, P < 0.001, respectively). At colonoscopy, lymphonodular hyperplasia or aphthous ulceration were present in 83% of patients. Twenty-two patients were given L-amino acid-based formula and 8 continued the previous feeding. Time to cessation of rectal bleeding was shorter in the special formula feeding group (mean, 1.4 wk; range, 0.5-3 wk) when compared with the breast-feeding group (mean, 5.3 wk; range, 2-9 wk). Nevertheless, none of the patients exhibited rectal bleeding at the 3-mo visit irrespective of the type of feeding. Peripheral eosinophilia and cessation of rectal bleeding after administration of elemental formula correlated with a higher density of mucosal eosinophils.
CONCLUSION: Infant hematochezia, after cow’s milk allergy exclusion, is generally a benign and probably self-limiting disorder despite marked mucosal abnormality. Formula feeding results in shorter time to cessation of rectal bleeding; however, breast-feeding should not be discouraged in long-lasting hematochezia.
Core tip: Rectal bleeding is a common problem in otherwise healthy breast-fed infants; our primary aim was to find characteristic lesions at colonoscopy and determine the cessation of rectal bleeding when administering different nutritional regimes (L-amino-acid based formula or breast-feeding). Our secondary aim was to find correlations between laboratory data, severity of mucosal lesions and cessation of rectal bleeding in allergic colitis infants with no cow’s milk allergy.
- Citation: Molnár K, Pintér P, Győrffy H, Cseh &, Müller KE, Arató A, Veres G. Characteristics of allergic colitis in breast-fed infants in the absence of cow’s milk allergy. World J Gastroenterol 2013; 19(24): 3824-3830
- URL: https://www.wjgnet.com/1007-9327/full/v19/i24/3824.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i24.3824
Rectal bleeding is a common problem in otherwise healthy breast-fed infants[1]. The differential diagnosis of this condition includes anal fissures, infectious colitis, congenital bleeding disorders, inflammatory bowel disease (IBD) and, most frequently, allergic colitis (AC)[2]. Currently there is no reliable diagnostic test available for AC, and the diagnosis is often made presumptively in healthy infants with rectal bleeding who show no anal fissures or infectious colitis[3]. AC is believed to be a hypersensitive gastrointestinal disorder to allergens present in breast milk, and formula is regarded as a form of food allergy in infancy[4]. As a resulting first-line therapeutic intervention, cow’s milk (CM) is generally eliminated from the maternal diet[5].
Recent guidelines state that the elimination diet for the mother should be continued for a minimum of at least 2 wk, and up to 4 wk in cases of AC[6]. According to the guidelines, if an infant has no cessation of rectal bleeding after 4 wk of maternal CM-free diet then the patient has no CM allergy. It is of interest that the majority of infants with rectal bleeding, despite the general belief, have no CM allergy as an underlying cause of hematochezia. This phenomenon was supported by recent studies showing that even in breast-fed infants with rectal bleeding, CM allergy is more uncommon than previously believed[7]. Only 18% of 40 infants with rectal bleeding had proven CM allergy, none had positive specific immunoglobulin E (IgE) to cow’s milk, egg or wheat on admission, and only 5% had a positive skin-prick test to cow’s milk[8]. Nevertheless, an atopy patch test was useful to identify sensitization for cow’s milk (50%), soy (28%), egg (21%), rice (14%) and wheat (7%) in 14 AC infants with multiple food allergy[9].
Lymphonodular hyperplasia (LNH), patchy granularity and aphthous lesions were found at colonoscopy in patients with AC; however, most patients showed no abnormal mucosal lesions[5,7,8]. These studies included patients with short-term rectal bleeding which may not have shown the development of characteristic lesions at colonoscopy. Therefore, in our study, there was a minimum 4-wk timeline between onset of rectal bleeding and the procedure of colonoscopy. We hypothesized that 4 wk would be enough to find a typical mucosal abnormality in otherwise healthy breast-fed infants with rectal bleeding not caused by CM allergy.
Taken together, our primary aim was to find characteristic lesions at colonoscopy and determine the cessation of rectal bleeding when administering different nutritional regimes (L-amino-acid based formula or breast-feeding). Our secondary aim was to find correlations between laboratory data, severity of mucosal lesions and cessation of rectal bleeding in AC infants with no CM allergy.
It should be noted that there is no previous, prospective study in AC patients with no CM allergy.
During a 4-year period (January 2006-February 2010) at the First Department of Pediatrics, Semmelweis University, breast-fed infants were invited to join the present study. Inclusion criteria were age less than 6 mo, exclusive breast-feeding, normal stool cultures (Salmonella, Shigella, E. coli, Yersinia, Campylobacter), and no cessation of rectal bleeding after introduction of a maternal elimination diet for CM for a minimum of 4 wk. All subjects were evaluated by history and physical examination for signs of fissure, infection and allergic diseases. Rectal bleeding as bloody spots or streaks with or without mucus was confirmed macroscopically before the colonoscopy. A complete blood count with differential, C-reactive protein, prothrombin time, activated partial thromboplastin time, serum albumin, serum IgE, and specific IgE to common food antigens including CM, egg, wheat, rye, soy, fish, nuts, peanuts, sesame, almond, tomato, banana, celery, carrots, apple, peach, lemon, and orange were obtained. Thirty-four age-matched and breast-fed infants (mean age, 16.9 wk; range, 4-25 wk; girls, 15) with no rectal bleeding (apnea, 10; gastro-esophageal reflux, 12; minor trauma, 7; minor surgical intervention, 5) were enrolled as controls for the laboratory testing. Characteristics of patients and controls are summarized in Table 1.
| Characteristic | AC (n = 30) | Controls (n = 34) |
| Gender (F:M) | 19:11 | 19:15 |
| Age at presentation (wk, mean) | 17.2 | 16.9 |
| Family history of atopy | 17 (57) | 13 (38) |
| Atopic dermatitis | 8 (27) | 7 (21) |
| Duration of hematochezia (wk, mean) | 8.6 | - |
| Increased level of IgE (> 5 U/mL) | 3 | Not done |
| Positivity to specific IgE to food antigens | 0 | Not done |
The Institutional Ethical Committee approved the study; written parental informed consent was obtained.
To characterize allergic colitis and exclude other rare forms of rectal bleeding in infancy such as angiodysplasia, hemangioma, polyp, IBD or blue rubber bleb syndrome, colonoscopy with multiple biopsies was performed using a flexible pediatric gastroscope with narrow band imaging technique included (Olympus GIF-Q180, Olympus Hungary KFT). Ambulatory colonoscopy was done by the same gastroenterologist (Veres G) with the assistance of a trained endoscopy nurse. The procedure was done under general anesthesia. The colon was gently cleansed by a single water enema (5 mL/kg) before the procedure.
Using standard biopsy forceps, 4 biopsies were taken from the sigmoid colon (about 10 cm from the anal verge) targeted to areas with gross endoscopic findings. Biopsy samples were fixed in 4% buffered formalin for 24 h then embedded into paraffin. Hematoxylin eosin (HE) stain was performed on 3-4 μm thin slides. The biopsy was examined for routine histology by a pathologist (Győrffy H). Eosinophils were counted in 10 high-power fields (HPF) and the average number of cells was recorded. Although there are no standard accepted criteria for the diagnosis of AC, several studies have demonstrated eosinophilic infiltration (≥ 6/HPF) in the lamina propria of the left colon or rectum[10-12]. Based on previous studies[11,13], patients with AC were subdivided according to the number of eosinophils on biopsy specimens. AC1 consisted of patients with eosinophils between 6 and 19/HPF, and AC2 with eosinophils ≥ 20/HPF. Using these criteria, AC2 patients fulfill the diagnostic criteria for eosinophilic colitis.
All subjects were followed up for 6 mo to assess outcome, including visits after one week following colonoscopy, after 3 mo, and after 6 mo. Elemental L-amino acid formula (Neocate; SHS Int., Liverpool, United Kingdom) was offered to the parents to treat their children. Assessment of rectal bleeding during the follow-up period was qualitative and assessed by parental self-report. In addition, if macroscopic bleeding eased, parents were asked to provide feces to exclude occult bleeding (HSV10; Diagnosticum Zrt, Budapest, Hungary). Because macroscopic bleeding was the main inclusion criteria, the cessation of macroscopic bleeding was considered to be the endpoint.
Up to 1 year of age, patients received AA formula or continued breast-feeding. At the age of 1 year, CM was introduced to both groups with no macroscopic bleeding in the follow-up. After 4 wk of the challenge, parents were asked to provide feces to exclude occult bleeding (HSV10; Diagnosticum Zrt, Budapest, Hungary). In addition to CM, parents were asked to avoid eggs, wheat, soya, nuts and fish (“six food elimination diet”) up to one year of age. After CM introduction, other foods were introduced gradually (wheat, soya, eggs, fish and nuts).
At the age of 6 mo, introduction of supplemental feeding (except the 6 foods listed above) was recommended irrespective of whether the patient was on the formula or the breast-feeding arm. Subjects with worsening of symptoms or having a severe form of colitis underwent repeat colonoscopy with biopsy. Six month time-point was chosen for this procedure to allow enough time for healing in the latter group.
Our data followed normal distribution, therefore parametric statistical tests were implicated and data were expressed as means with standard deviation. For comparison of datasets, unpaired t test and analysis of variance (ANOVA) with Bonferroni’s post hoc tests were used. For analyses of contributing effects, factorial ANOVA, logistic regression and Pearson’s correlation were used. The level of significance was 5% (P < 0.05) and Bonferroni’s adjustment for multiple comparisons was introduced where needed. Statistical analysis was performed with Statistica 8 (Statsoft, Tulsa, OK, United States).
Thirty-one healthy, breast-fed infants (mean age, 17.4 wk; range, 5-26 wk; 12 girls) were enrolled. The mean (range) duration of bloody stools before colonoscopy was 8.7 (4-16) wk (Table 1). All mothers strictly followed a CM-free diet with the help of a trained dietician. In addition to CM-free diet, 7 mothers had an elimination diet for egg and 4 had an exclusion diet for egg, fish, wheat, nuts and soy. In all patients, rectal bleeding was the main symptom that prompted the request for gastroenterological evaluation. In addition to bloody stools, watery (42%) or mucous stools (68%) were common. None of the patients had fissures at rectal examination. Stool frequency averaged 4.2 (range, 1-7) bowel movements per day. Three patients had a classic history of infantile colic. One patient had hemangioma on the thorax with no involvement of the liver. One patient had transient hypothyroidism. The others had no previous hospitalization or chronic disease. Growth was reported as normal in all patients whose weight and height charts were reviewed for an objective assessment of their growth pattern.
During follow-up, one subject had failure to thrive and was subsequently diagnosed with infantile Crohn’s disease. She was 24 wk old at study entry with a history of rectal bleeding for 12 wk. Her data were excluded from the study (final study group consisted of 30 infants).
Eight patients (27%) had atopic dermatitis which is comparable with the percentage of control subjects (21%). Seventeen patients (57%) had a positive family history of atopy among first-degree relatives which was higher than in controls (38%). Only 3 patients had an elevated level of serum IgE (> 5 kU/L). Specific IgE to the most common food antigens was determined in 25 of 31 patients (81%) with negative results (Table 1).
The laboratory abnormalities are presented in Table 2. The mean iron level was significantly decreased in patients (8.43 ± 3.2 μmol/L) in comparison to controls (13.7 ± 4.7 μmol/L, P < 0.001). We found a markedly increased thrombocyte count in patients (474 ± 123 G/L) when compared with controls (376 ± 89 G/L, P < 0.001). Similarly, patients exhibited increased peripheral eosinophilia (6.7 ± 4.9 G/L) when compared with controls (3.3 ± 1.7 G/L, P < 0.001). On the other hand, 60% of patients had thrombocytosis, 50% showed low iron level, and 40% demonstrated eosinophilia (Table 3). There was no difference concerning hemoglobin level between patients and controls.
| Characteristic | On admission | 6-mo visit | ND |
| Thrombocytosis (> 450 × 109/L) | 16 (60) | 5 | 2 |
| Iron deficiency (< 9 μmol/L) | 15 (50) | 4 | 2 |
| Eosinophilia (> 5% of leukocytes) | 12 (40) | 5 | 1 |
Patients were subdivided into two groups (AC1 and AC2: eosinophilic colitis) according to the number of eosinophils determined by histology on biopsies (see above). Iron level, hemoglobin and thrombocytes were comparable in AC1 and AC2. However, peripheral eosinophils were significantly elevated in AC2 (9.1 ± 4.7 G/L) when compared with AC1 (5.1 ± 4.5 G/L, P < 0.01).
Only one patient had marked anemia which required blood transfusion. This patient was the only one who had low albumin level (< 35 g/L). Her other characteristics (including macroscopic and microscopic mucosal abnormalities) were not different from those of the other subjects. Laboratory data in patients who were subsequently diagnosed with Crohn’s disease showed iron deficiency (4 mol/L) without anemia (113 g/L), and thrombocytosis (524 × 109/L).
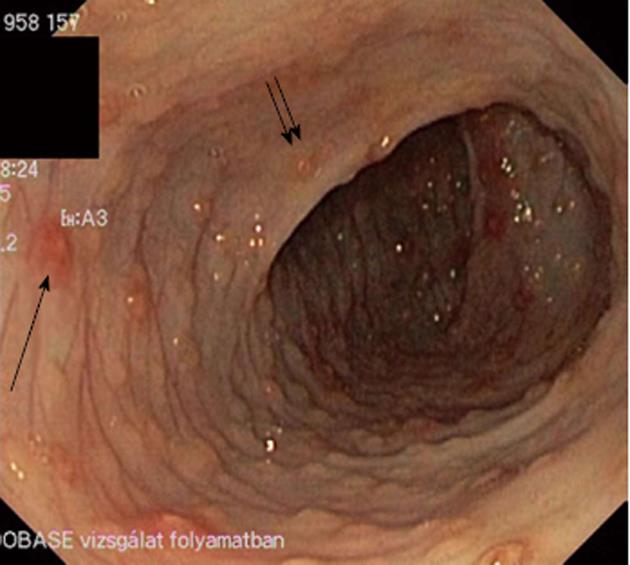
At colonoscopy, the cecum was reached in 23 (74%) patients. In all other subjects the colonic mucosa was visualized from the anus to the splenic flexure, two of them to the hepatic flexure. Three patients had normal visual colonoscopy findings. Twenty-seven (90%) showed abnormal mucosal lesions such as LNH, aphthous ulceration, and marked erythema (2 patients). LNH or aphthous ulceration was present in 25/30 patients (83%) (Figure 1). LNH was found in 73% of patients (22/30) and aphthous ulceration in 47% of the patients (14/30). Subdividing the patients with regard to the number of eosinophils on biopsies, LNH was seen in 15 patients with AC1 (79%) and in 7 patients with AC2 (64%). Aphthous ulceration was visualized in 8 patients with AC1 (42%) and in 6 patients with AC2 (55%). More patients with AC2 (45%) depicted both phenomenon, which were present in 32% of subjects with AC1. However, the discrepancies failed to reach significance (Table 4).
| Characteristic | AC1 (n = 19) | AC2 (n = 11) |
| LNH | 15 (79) | 7 (64) |
| Aphthous ulceration | 8 (42) | 6 (55) |
| LNH and aphthous ulceration | 6 (32) | 5 (45) |
| Marked erythema | 1 (5) | 1 (9) |
| No. of eosinophils/HPF (mean) | 12.2 | 29.5 |
LNH and aphthous ulceration without any specific signs for IBD were present in the patient with Crohn’s disease at her first colonoscopy. She was the only patient with no cessation of rectal bleeding at the 3-mo visit. Deep ulceration in the transverse colon and non-caseating granulomas on histology established the diagnosis of Crohn’s disease at her second colonoscopy (after 6-mo visit). Abnormal findings at colonoscopy are depicted in Table 4. There were no adverse events recorded under general anesthesia and colonoscopy.
Light microscopy revealed normal mucosal architecture in all patients studied, including the first biopsy of the patient with Crohn’s disease which showed normal mucosal architecture with an elevated number of eosinophils (29/HPF). All patients had more than 6 eosinophils/HPF on histology (Table 4). All three patients with normal macroscopic findings at colonoscopy showed an elevated eosinophil count on biopsies (6, 20 and 39 cells/HPF, respectively). Nineteen patients had an eosinophil count between 6 and 19/HPF (AC1) and 11 patients had more than 20/HPF (AC2). Mean number of eosinophils was 12.2/HPF in AC1 and 29.5/HPF in AC2 (eosinophilic colitis).
Based on marked mucosal abnormalities and the long-lasting rectal bleeding, we decided to treat patients with special formula. At the first visit one week after colonoscopy, elemental L-amino acid formula was offered to the parents to treat their children. Parents refused the formula feeding in 8 cases (5 in AC1 and 3 in AC2) and continued the previous feeding type. Twenty-two patients received the special formula (14 in AC1 and 8 in AC2). There were no significant differences concerning baseline characteristics between the 2 groups.
The duration of bleeding was significantly shorter in the special formula feeding group (mean, 1.4 wk; range, 0.5-3 wk) when compared with the breast-feeding group (mean, 5.3 wk; range, 2-9 wk). Nevertheless, none of the patients exhibited gastrointestinal complaints or visible rectal bleeding at the 3-mo visit irrespective of the type of feeding (Table 5). Three patients with serum iron level below 5 μmol/L were treated with iron-containing syrup (Maltofer; dosage, 2 drops/kg) for 6 wk. At 6-mo visit, control blood count and iron level were analyzed in patients who had abnormal levels on admission.
| Characteristic | Amino-acid based formula feeding | Breast-feeding |
| No. of patients | 22 | 8 |
| Age at presentation (wk) | 16.0 (5-26) | 20.6 (16-25)a |
| Rectal bleeding at baseline (wk) | 7.9 (4-14) | 10.5 (6-16) |
| Cessation of rectal bleeding (wk) | 1.4 (0.5-3) | |
| Rectal bleeding at 3-mo visit (n) | 0 | 0 |
Due to the cessation of bleeding, only four parents agreed for a control colonoscopy to be performed. At baseline all of them had LNH and aphthous ulceration. One patient was in AC1 (13 eosinophils/HPF) and 3 patients were in AC2 (eosinophils > 30/HPF). At control colonoscopy none of the patients had aphthous ulceration; only scattered LNH without bleeding spots were visible. The number of eosinophils normalized in AC1 patients (5/HPF) and markedly decreased in AC2 patients (8-17/HPF).
Correlation analysis revealed that the time to stop the bleeding was negatively correlated with the administration of elemental formula (P < 0.001, r = -0.762). In addition, the number of eosinophils on biopsy specimens at baseline correlated negatively with elemental formula concerning cessation of rectal bleeding (P = 0.025, r = -0.667). There was no correlation between abnormal serum laboratory data (thrombocytes, eosinophils, iron) and the ending of rectal bleeding. In patients with AC1 and AC2, we found similar correlations between elemental diet and the time to stop the bleeding.
The elimination of CM from the diet of the lactating mother is a commonly used and recommended practice[10-12] for infants with rectal bleeding. However, recent studies have suggested that there are significant proportions of infants with rectal bleeding with no CM allergy[8,9,13]. Based on these results we focused on AC patients with no cessation of rectal bleeding after a minimum of 4 wk of maternal CM-free diet. Four weeks is a long enough time-span to exclude rare forms of surgical diseases, to cure an undetected viral gastrointestinal disorder or for elimination of CM protein from patients[6,8,14].
In our study, frequency of atopic dermatitis, measurement of serum IgE, and specific IgE to different foods were not significant factors in the diagnostic procedure, as has been suggested by previous studies[14,15]. On the other hand, a recent Italian study suggested a delayed-type immunogenic mechanism in this process, hence the atopy patch test was useful in the diagnostic procedure in 14 AC infants with multiple gastrointestinal food allergy[9].
In our study, iron deficiency, peripheral eosinophilia and thrombocytosis were significantly higher in AC patients in comparison to controls. Surprisingly, no previous study has analyzed iron levels in patients with AC. Peripheral eosinophilia and thrombocytosis were reported previously[7,8,15]. To determine the risk factors for the clinical outcome, none of the 3 abnormal laboratory findings correlated with the cessation of rectal bleeding.
Theoretically, colonoscopy would have detected other rare causes of rectal bleeding such as polyps, hemangioma, blue rubber bleb syndrome and angiodysplasia. However, all but 3 patients showed mucosal abnormalities such as LNH, aphthous ulceration and marked focal erythema. As a bleeding source, LNH with circumscribed erythema or/and central pit-like bleeding spots (Figure 1) or aphthous ulceration were present in 25 patients with AC (83%). This is in contrast to previous studies, which reported a much lower percentage of characteristic abnormalities at colonoscopy. In a prospective evaluation of 34 infants with rectal bleeding, only 10 (29%) showed LNH including three of them with normal histology[5]. Xanthakos et al[7] followed 22 infants younger than 6 mo of age with hematochezia. Only 12 patients showed gross endoscopic abnormalities at colonoscopy including diffuse erythema, friability, LNH and aphthous ulceration. In addition, a recent study followed 40 infants with visible rectal bleeding where 41% of patients had normal mucosa, 33% had aphthous ulceration, and 51% depicted focal erythema with no report of LNH[8]. One of the explanations for the high rate of LNH and aphthous ulceration found in our study is the long-lasting rectal bleeding (minimum 4 wk). LNH has been described as a sign of food allergy in children with CM allergy[16-18]. In our patients with no CM allergy, LNH was also a characteristic abnormality at colonoscopy. We do not think that LNH is a specific sign of food allergy or AC, just an indicator of a focal inflammation beneath the epithelium. However, LNH with circumscribed erythema or/and central pit-like bleeding spots and aphthous ulceration may have a role in the diagnostic procedure of AC.
The number of eosinophils on biopsies in patients with AC is a question of debate[19]. Previous studies[20,21] have suggested, as a diagnostic criteria for AC, eosinophilic infiltration in the lamina propria of ≥ 6/HPF, whereas Machida et al[5] suggested for this entity of ≥ 20/HPF. All patients in the present study had ≥ 6 eosinophils/HPF in the lamina propria. To find a possible difference of laboratory, colonoscopy and clinical outcome, patients were subdivided to moderate (AC1) or high (AC2) mucosal density of eosinophils. It should be noted that AC2 patients fulfill the present criteria for eosinophilic colitis (≥ 20 eosinophils/HPF)[22]. Peripheral eosinophilia and cessation of rectal bleeding after administration of elemental formula correlated with the higher number of eosinophils on biopsies. In conclusion, these data suggest that both groups represent similar entities, and a cut-off > 20 eosinophils/HPF for eosinophilic colitis may be artificial.
Due to the marked mucosal abnormality and the long-lasting rectal bleeding, elemental formula was given to the patients. It was refused by 8 cases who continued their previous feeding. Elemental diet shortened the time to cessation of hematochezia; nevertheless, it should be emphasized that none of the patients exhibited gastrointestinal complaints or rectal bleeding at the 3-mo visit irrespective of the type of feeding. This result suggests that the underlying cause could be food-related other than CM, hence AA formula shortened the resolutions of symptoms. Probably, our study population may have multiple food allergy; at least, we cannot exclude this because the nursing mothers conducted only limited types of food elimination diet (beside CM).
The beneficial effect of amino acid-based formula was reported previously[23], suggesting an allergy-related mechanism even in AC patients with no CM allergy.
On the other hand, we cannot exclude the possibility that our patients had no food allergy at all. This phenomenon is similar to infantile eczema[24] or eosinophilic esophagitis in infancy, where considerable proportions of patients have no food allergy but an allergic-immunologic component does exist[25]. Exclusive amino acid formula-based dietary trials resulted in more than 90% remission in children with eosinophilic esophagitis. Similarly to current guidelines in AC, empiric elimination diets of avoidance of foods commonly known to cause hypersensitivity reactions resulted in 50%-74% disease remission in eosinophilic esophagitis[26]. Taken together, previous studies and the beneficial effect of AA formula in the present study suggest that our study population probably has multiple food allergy. One of the most interesting points of data in our study is that patients were able to tolerate a non-restricted diet after a period of elimination diet (AA-based formula) or after a more prolonged persistence of symptoms on breast-feeding. Acquired tolerance may explain this finding, which is supported by our previous study where depleted FOXP3 regulatory T cells normalized after feeding of AA formula[27].
Nevertheless, the only patient who had not ceased the rectal bleeding was subsequently diagnosed with infantile Crohn’s disease and she was excluded from the study. As reported previously[7,28,29], at baseline, none of her laboratory, colonoscopy or histology parameters were different from the patients with AC.
In conclusion, rectal bleeding in infancy, even after exclusion of CM, is generally benign and is probably a self-limiting disorder, despite the marked mucosal abnormality. Iron deficiency, peripheral eosinophilia and thrombocytosis are characteristic findings in these patients. At colonoscopy, LNH and aphthous ulceration may be characteristic signs. Administration of amino acid formula shortened the timeline of the rectal bleeding in patients with AC; however, breast-feeding should not be discouraged in this population with long-lasting hematochezia.
Allergic inflammation of the large intestine is believed to be a hypersensitive gastrointestinal disorder to allergens present in breast milk, and formula is regarded as a form of food allergy in infancy. As a resulting first line therapeutic intervention, cow’s milk is generally eliminated from the maternal diet; however, significant numbers of infants with rectal bleeding, despite the general belief, have no cow’s milk allergy as an underlying cause of hematochezia. Currently there is no reliable diagnostic test available for allergic colitis, and the diagnosis is often made presumptively in healthy breast-fed infants with rectal bleeding who show no anal abnormalities or infectious colitis.
A research group in Hungary investigated the characteristics of endoscopic findings in colonic mucosal lesions and their relation to laboratory data in breast-fed infants with allergic colitis. Long-term follow-up with different nutritional regimes (L-amino acid-based formula or breast-feeding) was also included. According to the literature there is no previous, prospective study in allergic colitis patients with no cow’s milk allergy.
In this study, 31 breast-fed infants were prospectively evaluated whose rectal bleeding had not ceased after a maternal elimination diet for cow’s milk allergy. As controls, 34 age-matched and breast-fed infants without rectal bleeding were enrolled for the laboratory testing. In the laboratory findings, iron deficiency and elevation of peripheral eosinophilic cell and platelet counts were significantly higher in patients with allergic colitis in comparison to controls. At colonoscopy, increments of lymphoid tissue appearing as a lump (called lymphonodular hyperplasia) or aphthous ulceration were present in 83% of patients. Twenty-two patients were given L-amino acid-based formula and 8 continued the previous feeding. Time to cessation of rectal bleeding was shorter in the special formula feeding group when compared with the breast-feeding group. Nevertheless, none of the patients exhibited rectal bleeding at the 3-mo visit irrespective of the type of feeding. The elevated peripheral eosinophilic cell count and cessation of rectal bleeding after administration of elemental formula correlated with the higher density of mucosal eosinophilic cells in the colon.
Administration of amino acid formula shortened the timeline of rectal bleeding in patients with allergic colitis; however, breast-feeding should not be discouraged in this population with long-lasting rectal bleeding. These data suggest that the significance of real food allergy in breast-fed infants with allergic colitis should be revised in the future. This phenomenon is similar to infantile eczema or eosinophilic esophagitis in infancy where considerable proportions of patients had no food allergy but an allergic-immunologic component does exist. It may indicate that the “food allergy” theory is currently much less applicable, and probably a significant number of nursing mothers continue an unnecessary restriction diet worldwide.
Allergic colitis is explained as an allergic condition of the gastrointestinal tract in breast-fed infants that results from an immune response to the allergens present in breast milk.
This is a very interesting article from a teaching institution in Hungary. It is well written and conclusions are valid.
P- Reviewer Camilleri-Brennan J S- Editor Wen LL L- Editor Logan S E- Editor Li JY
| 1. | Eigenmann PA. Mechanisms of food allergy. Pediatr Allergy Immunol. 2009;20:5-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 2. | Fox VL. Gastrointestinal bleeding in infancy and childhood. Gastroenterol Clin North Am. 2000;29:37-66, v. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 56] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 3. | Troncone R, Discepolo V. Colon in food allergy. J Pediatr Gastroenterol Nutr. 2009;48 Suppl 2:S89-S91. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 4. | Moon A, Kleinman RE. Allergic gastroenteropathy in children. Ann Allergy Asthma Immunol. 1995;74:5-12; quiz 12-16. [PubMed] |
| 5. | Machida HM, Catto Smith AG, Gall DG, Trevenen C, Scott RB. Allergic colitis in infancy: clinical and pathologic aspects. J Pediatr Gastroenterol Nutr. 1994;19:22-26. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 101] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 6. | Vandenplas Y, Koletzko S, Isolauri E, Hill D, Oranje AP, Brueton M, Staiano A, Dupont C. Guidelines for the diagnosis and management of cow’s milk protein allergy in infants. Arch Dis Child. 2007;92:902-908. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 239] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 7. | Xanthakos SA, Schwimmer JB, Melin-Aldana H, Rothenberg ME, Witte DP, Cohen MB. Prevalence and outcome of allergic colitis in healthy infants with rectal bleeding: a prospective cohort study. J Pediatr Gastroenterol Nutr. 2005;41:16-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 99] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 8. | Arvola T, Ruuska T, Keränen J, Hyöty H, Salminen S, Isolauri E. Rectal bleeding in infancy: clinical, allergological, and microbiological examination. Pediatrics. 2006;117:e760-e768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 147] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 9. | Lucarelli S, Di Nardo G, Lastrucci G, D’Alfonso Y, Marcheggiano A, Federici T, Frediani S, Frediani T, Cucchiara S. Allergic proctocolitis refractory to maternal hypoallergenic diet in exclusively breast-fed infants: a clinical observation. BMC Gastroenterol. 2011;11:82. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 39] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 10. | Sampson HA, Anderson JA. Summary and recommendations: Classification of gastrointestinal manifestations due to immunologic reactions to foods in infants and young children. J Pediatr Gastroenterol Nutr. 2000;30 Suppl:S87-S94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 129] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 11. | Winter HS, Antonioli DA, Fukagawa N, Marcial M, Goldman H. Allergy-related proctocolitis in infants: diagnostic usefulness of rectal biopsy. Mod Pathol. 1990;3:5-10. [PubMed] |
| 12. | Hill SM, Milla PJ. Colitis caused by food allergy in infants. Arch Dis Child. 1990;65:132-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 54] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 13. | Hwang JB, Park MH, Kang YN, Kim SP, Suh SI, Kam S. Advanced criteria for clinicopathological diagnosis of food protein-induced proctocolitis. J Korean Med Sci. 2007;22:213-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 14. | Lake AM. Food-induced eosinophilic proctocolitis. J Pediatr Gastroenterol Nutr. 2000;30 Suppl:S58-S60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 141] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 15. | Lake AM, Whitington PF, Hamilton SR. Dietary protein-induced colitis in breast-fed infants. J Pediatr. 1982;101:906-910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 141] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 16. | Anveden-Hertzberg L, Finkel Y, Sandstedt B, Karpe B. Proctocolitis in exclusively breast-fed infants. Eur J Pediatr. 1996;155:464-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 43] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 17. | Ravelli A, Villanacci V, Chiappa S, Bolognini S, Manenti S, Fuoti M. Dietary protein-induced proctocolitis in childhood. Am J Gastroenterol. 2008;103:2605-2612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 35] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 18. | Kokkonen J, Karttunen TJ, Niinimäki A. Lymphonodular hyperplasia as a sign of food allergy in children. J Pediatr Gastroenterol Nutr. 1999;29:57-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 38] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 19. | Yan BM, Shaffer EA. Primary eosinophilic disorders of the gastrointestinal tract. Gut. 2009;58:721-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 119] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 20. | Odze RD, Bines J, Leichtner AM, Goldman H, Antonioli DA. Allergic proctocolitis in infants: a prospective clinicopathologic biopsy study. Hum Pathol. 1993;24:668-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 103] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 21. | Odze RD, Wershil BK, Leichtner AM, Antonioli DA. Allergic colitis in infants. J Pediatr. 1995;126:163-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 88] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 22. | Bischoff SC. Food allergy and eosinophilic gastroenteritis and colitis. Curr Opin Allergy Clin Immunol. 2010;10:238-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 23. | Alfadda AA, Storr MA, Shaffer EA. Eosinophilic colitis: epidemiology, clinical features, and current management. Therap Adv Gastroenterol. 2011;4:301-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 85] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 24. | Kvenshagen B, Jacobsen M, Halvorsen R. Atopic dermatitis in premature and term children. Arch Dis Child. 2009;94:202-205. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 34] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 25. | Hogan SP, Rothenberg ME. Eosinophil Function in Eosinophil-associated Gastrointestinal Disorders. Curr Allergy Asthma Rep. 2006;6:65-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 42] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 26. | Chehade M, Aceves SS. Food allergy and eosinophilic esophagitis. Curr Opin Allergy Clin Immunol. 2010;10:231-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 52] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 27. | Cseh A, Molnár K, Pintér P, Szalay B, Szebeni B, Treszl A, Arató A, Vásárhelyi B, Veres G. Regulatory T cells and T helper subsets in breast-fed infants with hematochezia caused by allergic colitis. J Pediatr Gastroenterol Nutr. 2010;51:675-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 28. | Ojuawo A, St Louis D, Lindley KJ, Milla PJ. Non-infective colitis in infancy: evidence in favour of minor immunodeficiency in its pathogenesis. Arch Dis Child. 1997;76:345-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 29. | Cannioto Z, Berti I, Martelossi S, Bruno I, Giurici N, Crovella S, Ventura A. IBD and IBD mimicking enterocolitis in children younger than 2 years of age. Eur J Pediatr. 2009;168:149-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 80] [Article Influence: 5.0] [Reference Citation Analysis (0)] |