Published online Jun 21, 2013. doi: 10.3748/wjg.v19.i23.3707
Revised: March 27, 2013
Accepted: April 27, 2013
Published online: June 21, 2013
Processing time: 135 Days and 20.2 Hours
Bowel endometriosis affects between 3.8% and 37% of women with endometriosis. The evaluation of symptoms and clinical examination are inadequate for an accurate diagnosis of intestinal endometriosis. We describe the case of a 41-year-old woman who presented to our hospital because of six months of recurrent abdominal pain, vomiting and diarrhea, without previous history of bowel disease. Physical examination revealed a palpable 3 cm × 5 cm mass in the right lower quadrant abdomen. Laboratory tests showed slightly elevated levels of CA19-9 and CA125. Small bowel computer tomography scanning revealed an ileocecal mass with bowel wall thickening and luminal narrowing. Small bowel endoscopy identified a deep longitudinal ulcer and mucosal edema in the distal ileum. All these findings supported the diagnosis of Crohn’s disease. The patient underwent a laparotomy, which identified a 5 cm × 5 cm ileocecal mass with severe mucosal edema and luminal stricture in the distal ileum. Histopathological examination confirmed a diagnosis of ileocecal endometriosis without other areas involved. After one-year follow-up, there was no recurrence of the symptoms.
Core tip: We describe the case of a 41-year old woman who had recurrent abdominal pain with vomiting and diarrhea on presentation to our hospital. The results of computer tomography scanning and small bowel endoscopy were strongly suspicious for Crohn’s disease. However, surgery and histopathological examination confirmed a diagnosis of ileocecal endometriosis without other areas involved.
- Citation: Tong YL, Chen Y, Zhu SY. Ileocecal endometriosis and a diagnosis dilemma: A case report and literature review. World J Gastroenterol 2013; 19(23): 3707-3710
- URL: https://www.wjgnet.com/1007-9327/full/v19/i23/3707.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i23.3707
Intestinal endometriosis affects 12%-15% of menstruating women, and is generally an asymptomatic condition[1]. Ileal involvement is very rare and the patients generally present with an asymptomatic or painful mass[2]. Symptoms of bowel endometriosis are numerous, ranging from being asymptomatic to a constellation of symptoms like painful bowel movements, cramps, constipation, diarrhea, vomiting, rectal pain, infertility, abdominal mass, increased urinary frequency and cyclical hematochezia[3]. Classically, the symptoms become worse during menses, but this is not always the case. This myriad of symptoms makes the condition difficult to diagnose. Small bowel endometriosis tends to affect bowel serosa and only 10% of intestinal cases have mucosal involvement[1,4,5]. It can be difficult to discern between ileal Crohn’s disease (CD) and endometriosis.
In this report, we describe a further case of ileocecal mass with longitudinal ulcer which was suspected as being CD. Surgery and histopathological examination confirmed a diagnosis of ileocecal endometriosis with no other areas involved. This report serves as a reminder of this rare condition as well as highlighting the diagnostic difficulties it can pose.
A 41-year-old lady with no significant past medical history was presented to our hospital because of six months of recurrent abdominal pain, vomiting and diarrhea. The patient had no past history of tuberculosis or other infectious diseases. She also denied radiation exposure, a poisonous chemical contact history and genetic history. Her family had no history of bowel disease. Physical examination revealed a palpable 3 cm × 1.5 cm mass in the right lower quadrant abdomen.
Laboratory tests showed slightly elevated levels of C-reactive protein, CA19-9 and CA125. No other abnormalities were found in tests such as erythrocyte sedimentation rate, immune index or tuberculosis series check. Routine stool test was normal with occult blood negative.
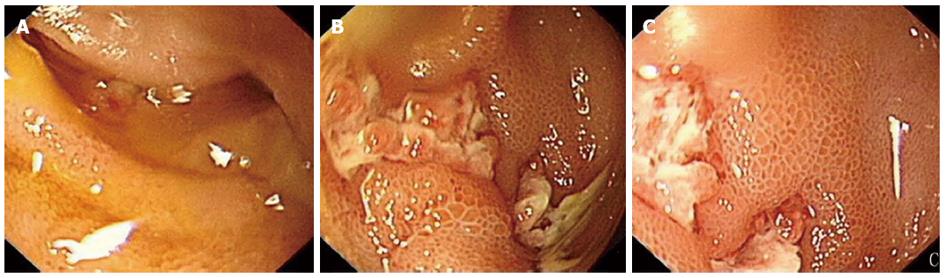
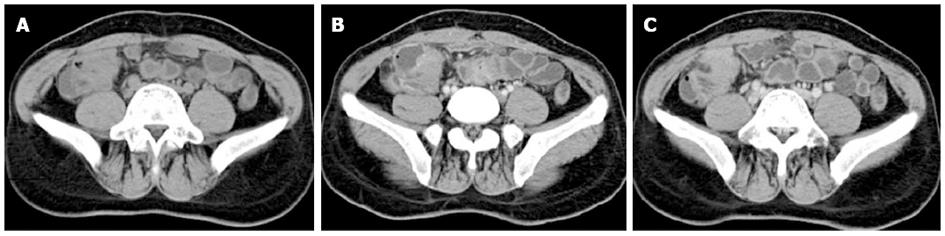
Computer tomography (CT) scanning revealed an ileocecal mass with multiple mesenteric lymph nodes enlarged. A colonoscopy she underwent three months previously showed introverted mucosa surrounding the appendix hole without colon abnormalities. Small bowel endoscopy identified a deep longitudinal ulcer in the distal ileum, mucosal edema and luminal stricture which the endoscope couldn’t go through (Figure 1). All these findings supported the diagnosis of CD. To evaluate other intestinal lesions, we undertook small bowel CT scanning, which revealed an ileocecal mass with bowel wall thickening and luminal narrowing, without other intestinal areas being involved (Figure 2).
Due to the bowel obstruction, the patients underwent a laparotomy, which revealed an ileocecal mass, 5 cm × 5 cm in size, with severe mucosal edema and luminal stricture in the distal ileum. No other organs were invaded. The frozen-section diagnosis was endometriosis. En bloc resection was taken and the histopathological examination confirmed ileocecal endometriosis. No subsequent medical treatment was undertaken. The patient recovered well after the surgery, and her quality of life has been significantly improved. After one-year follow-up, there was no recurrence of the symptoms.
Endometriosis is defined as the presence of ectopic endometrial tissue in extrauterine sites. It affects 10%-15% of women of reproductive age and usually becomes apparent in the reproductive years when the lesions are stimulated by ovarian hormones[6]. Intestinal endometriosis occurs in 12%-15% of cases, and the incidence of the involvement of different intestinal sites varies greatly in the literature, with the rectosigmoid colon, small bowel, appendix and cecum affected in 50%-90%, 2%-16%, 3%-18% and 2%-5% of cases, respectively[4]. As in our case, ileocecal involvement is rare with an incidence of 4.1% in intestinal cases[7].
The etiology of endometriosis is still elusive. The most widely accepted theory is that “retrograde menstruation’’ causes the implantation and growth of endometriosis on the serosal surface of extra-uterine organs or that this occurs secondary to metaplasia in the pelvic peritoneum[2,8-10].
Symptoms of bowel endometriosis can be associated with the patient’s menstrual cycle in 18%-40% of cases but may become permanent when the lesions progress[2,11,12]. Under cyclical hormonal influences, serosal implants may proliferate and infiltrate the bowel wall, and lead to inflammation, fibrosis, and metaplasia or hyperplasia of intestinal smooth muscles that can involve the serosa, submucosa and (uncommonly) mucosa[13]. This then leads to introverted mucosa surrounding the appendix hole, luminal stricture, longitudinal ulcer, and ileocecal mass as we believe happened in our case.
Symptoms range from an asymptomatic state to a constellation of symptoms like painful bowel movements, cramps, constipation, diarrhea, vomiting, rectal pain, infertility, abdominal mass, increased urinary frequency and cyclical hematochezia[3]. Those symptoms can mimic a wide spectrum of diseases, including irritable bowel syndrome, infectious diseases, ischemic enteritis/colitis, inflammatory bowel disease and neoplasm, so it is difficult to establish a preoperative diagnosis of bowel endometriosis[2,14,15].
Laboratory tests such as CA125 detection are not sensitive enough for diagnosis[9]. Transvaginal sonography should be used as the first-line diagnostic technique; this has shown a sensitivity and specificity of 43.7% and 50%, respectively[16,17]. Saline contrast sonovaginography was more accurate in diagnosing the condition than was transvaginal ultrasonography, with a sensitivity and specificity of 90.6% and 85.7%, respectively[16-18]. Contrast CT with enteroclysis protocols can be useful in diagnosis as this may demonstrate focal or constricting bowel lesions[4,9]. Magnetic resonance imaging (MRI) is currently the best imaging modality for enteric endometriosis with a sensitivity of 77%-93%[1,9]. Endoscopy may provide no valuable results because of the intact mucosa, but it is still recommended in all patients with suspected endometriosis to rule out mucosal involvement and malignant lesions with the help of biopsies, if needed. In our patient, symptoms relapsed irregularly and were not related with menses. The imaging results showed an ileocecal mass and longitudinal ulcer with luminal stricture under endoscopy, which strongly suggested CD. Due to the bowel obstruction, surgery was recommended.
Histopathological confirmation required presence of both glandular and stromal tissue. In our patient, the pathologist’s findings showed that the annular lesion of endometriosis and mucosa was not involved.
The treatment of uncomplicated intestinal endometriosis depends on the patient’s age and intention to conceive. Medical treatment with hormonal therapy such as the oral contraceptive pill, danazol or gonadotropin antagonists can be attempted for intestinal disease when there is no obstruction[1,2,19]. Bowel resection is indicated if there are symptoms of obstruction or bleeding, and if malignancy cannot be excluded. Post-operative hormonal therapy does not demonstrate benefits, according to a recent meta-analysis[20].
In summary, bowel endometriosis should be borne in mind when a woman of reproductive age presents with episodic gastrointestinal symptoms. A careful history may elicit symptoms related to the patient’s menses. Small bowel CT and MRI is indicated, and endoscopy is still recommended in all patients to rule out mucosal involvement and malignant lesions. In our case, the final diagnosis could only be given by the pathologist’s report. Multidisciplinary care should be encouraged to ensure correct evaluation and improve the management of these patients.
P- Reviewers Cosmi E, Petraglia F S- Editor Zhai HH L- Editor Logan S E- Editor Zhang DN
| 1. | Bianchi A, Pulido L, Espín F, Hidalgo LA, Heredia A, Fantova MJ, Muns R, Suñol J. [Intestinal endometriosis. Current status]. Cir Esp. 2007;81:170-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 2. | Scarmato VJ, Levine MS, Herlinger H, Wickstrom M, Furth EE, Tureck RW. Ileal endometriosis: radiographic findings in five cases. Radiology. 2000;214:509-512. [PubMed] |
| 3. | Kazadi Buanga J, Alcazar JL, Laparte MC, Lopez Garcia G. [Catamenial rectal bleeding and sigmoid endometriosis]. J Gynecol Obstet Biol Reprod (Paris). 1992;21:773-774. [PubMed] |
| 4. | Teke Z, Aytekin FO, Atalay AO, Demirkan NC. Crohn’s disease complicated by multiple stenoses and internal fistulas clinically mimicking small bowel endometriosis. World J Gastroenterol. 2008;14:146-151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 16] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 5. | Kavallaris A, Köhler C, Kühne-Heid R, Schneider A. Histopathological extent of rectal invasion by rectovaginal endometriosis. Hum Reprod. 2003;18:1323-1327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 99] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 6. | Podgaec S, Abrao MS, Dias JA, Rizzo LV, de Oliveira RM, Baracat EC. Endometriosis: an inflammatory disease with a Th2 immune response component. Hum Reprod. 2007;22:1373-1379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 155] [Cited by in RCA: 180] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 7. | Chapron C, Chopin N, Borghese B, Foulot H, Dousset B, Vacher-Lavenu MC, Vieira M, Hasan W, Bricou A. Deeply infiltrating endometriosis: pathogenetic implications of the anatomical distribution. Hum Reprod. 2006;21:1839-1845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 294] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 8. | Szucs RA, Turner MA. Gastrointestinal tract involvement by gynecologic diseases. Radiographics. 1996;16:1251-170; quiz 1251-170;. [PubMed] |
| 9. | De Ceglie A, Bilardi C, Blanchi S, Picasso M, Di Muzio M, Trimarchi A, Conio M. Acute small bowel obstruction caused by endometriosis: a case report and review of the literature. World J Gastroenterol. 2008;14:3430-3434. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 69] [Cited by in RCA: 55] [Article Influence: 3.2] [Reference Citation Analysis (1)] |
| 10. | Siristatidis CS. What have the ‘omics done for endometriosis? Med Sci Monit. 2009;15:RA116-RA123. [PubMed] |
| 11. | Popoutchi P, dos Reis Lemos CR, Silva JC, Nogueira AA, Feres O, Ribeiro da Rocha JJ. Postmenopausal intestinal obstructive endometriosis: case report and review of the literature. Sao Paulo Med J. 2008;126:190-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 12. | Denève E, Maillet O, Blanc P, Fabre JM, Nocca D. [Ileocecal intussusception secondary to a cecal endometriosis]. J Gynecol Obstet Biol Reprod (Paris). 2008;37:796-798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 13. | Itoga T, Matsumoto T, Takeuchi H, Yamasaki S, Sasahara N, Hoshi T, Kinoshita K. Fibrosis and smooth muscle metaplasia in rectovaginal endometriosis. Pathol Int. 2003;53:371-375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 82] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 14. | Yantiss RK, Clement PB, Young RH. Endometriosis of the intestinal tract: a study of 44 cases of a disease that may cause diverse challenges in clinical and pathologic evaluation. Am J Surg Pathol. 2001;25:445-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 155] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 15. | Dimoulios P, Koutroubakis IE, Tzardi M, Antoniou P, Matalliotakis IM, Kouroumalis EA. A case of sigmoid endometriosis difficult to differentiate from colon cancer. BMC Gastroenterol. 2003;3:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 45] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Dessole S, Farina M, Rubattu G, Cosmi E, Ambrosini G, Nardelli GB. Sonovaginography is a new technique for assessing rectovaginal endometriosis. Fertil Steril. 2003;79:1023-1027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 98] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 17. | Saccardi C, Cosmi E, Borghero A, Tregnaghi A, Dessole S, Litta P. Comparison between transvaginal sonography, saline contrast sonovaginography and magnetic resonance imaging in the diagnosis of posterior deep infiltrating endometriosis. Ultrasound Obstet Gynecol. 2012;40:464-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 61] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 18. | Cosmi E, Saccardi C, Litta P. The sonographic diagnosis of deep endometriosis. J Ultrasound Med. 2009;28:410-411. [PubMed] |
| 19. | Lin YH, Kuo LJ, Chuang AY, Cheng TI, Hung CF. Extrapelvic endometriosis complicated with colonic obstruction. J Chin Med Assoc. 2006;69:47-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 20. | Yap C, Furness S, Farquhar C. Pre and post operative medical therapy for endometriosis surgery. Cochrane Database Syst Rev. 2004;CD003678. [PubMed] |