Published online Feb 21, 2012. doi: 10.3748/wjg.v18.i7.601
Revised: July 8, 2011
Accepted: July 15, 2011
Published online: February 21, 2012
Epithelial-to-mesenchymal and mesenchymal-to-epithelial transitions are well established biological events which have an important role in not just normal tissue and organ development, but in the pathogenesis of diseases. Increasing evidence has established their presence in the human colon during colorectal carcinogenesis and cancer invasion, chronic inflammation-related fibrosis and in the course of mucosal healing. A large body of evidence supports the role for transforming growth factor-β and its downstream Smad signaling, the phosphatidylinositol 3’-kinase/Akt/mTOR axis, the Ras-mitogen-activated protein kinase/Snail/Slug and FOXC2 pathway, and Hedgehog signaling and microRNAs in the development of colorectal cancers via epithelial-to-mesenchymal transition. C-met and Frizzled-7, among others, seem to be the principle effectors of mesenchymal-to-epithelial transition, hence have a role not just in mucosal regeneration but in the progression of colonic wall fibrosis. Here we discuss a role for these pathways in the initiation and development of the transition events. A better understanding of their induction and regulation may lead to the identification of pathways and factors that could be potent therapeutic targets. The inhibition of epithelial-to-mesenchymal transition using mTOR kinase inhibitors targeting the ATP binding pocket and which inhibit both mTORC1 and mTORC2, RNA aptamers or peptide mimetics, such as a Wnt5A-mimetic, may all be useful in both cancer treatment and delaying fibrosis, while the induction of mesenchymal-to-epithelial transition in induced pluripotent stem cells may enhance epithelial healing in the case of severe mucosal damage. The preliminary results of the current studies are promising, but more clinical investigations are needed to develop new and safe therapeutic strategies for diseases of the colon.
- Citation: Sipos F, Galamb O. Epithelial-to-mesenchymal and mesenchymal-to-epithelial transitions in the colon. World J Gastroenterol 2012; 18(7): 601-608
- URL: https://www.wjgnet.com/1007-9327/full/v18/i7/601.htm
- DOI: https://dx.doi.org/10.3748/wjg.v18.i7.601
Human colonic diseases are some of the most common diseases worldwide, and their incidence is increasing[1,2]. Although the diversity of the etiologic and pathophysiologic factors of colonic diseases is very wide, there exist two biological processes whose presence is indispensable in the progression or healing phase of these conditions.
One of these processes is the epithelial-to-mesenchymal transition (EMT), which has a significant role in the development of the human body. EMT is also involved in the initial step, acquisition of migratory and invasive capability of colorectal cancer (CRC), and even has an important role in tissue fibrosis[3].
The other process is a reverse phenomenon, namely the mesenchymal-to-epithelial transition (MET). MET is also essential for normal tissue and organ development, but it is also involved in colorectal carcinogenesis, and it seems to have an important role in colonic mucosal regeneration[4,5].
The induction and regulation of these complex, reversible biological programs are not fully understood. Therapeutically, the influence of EMT and MET is recognized, but, unfortunately, the exact description of these biological phenomena is still missing. As the understanding of the steps of EMT and MET is of great clinical importance, and, at the same time, data about their complex induction and regulation, as well as their role in the pathogenesis of colonic diseases are scarce in the scientific literature, we aimed to summarize the current knowledge in this review.
EMT is a physiological mechanism which is present during development, including mesoderm formation and neural tube formation, and is also encountered in several pathological situations, such as renal interstitial fibrosis, endometrial adhesion, and cancer metastasis[3].
Cells that undergo EMT exhibit dramatic shape changes during which they can lose many of their epithelial characteristics, such as the loss of apico-basal polarity and cell adhesion, the repression of E-cadherin, occludin, tight junction protein 1, or cytokeratin expression, and increased cell mobility[6]. At the same time, elevated expression of tyrosine kinases or their activation, upregulation of N-cadherin, vimentin, fibronectin, zinc-finger domain proteins (SNAI1/SAIL, SNAI2/SLUG, ZEB2/SIP1), and matrix metalloproteinases, as well as basic helix-loop-helix domain protein Twist1 expression are often linked to a mesenchymal-like phenotype[7,8].
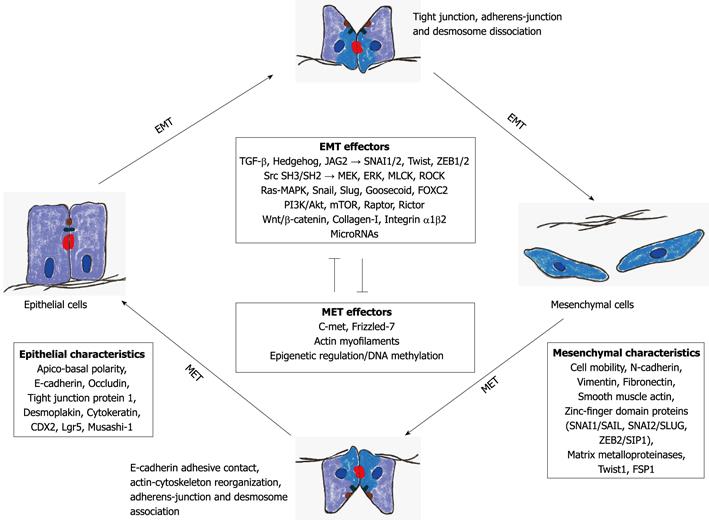
Several oncogenic pathways (i.e., peptide growth factors, Src, Ras, integrin, Wnt/β-catenin, Notch) may induce EMT (Figure 1).
Transforming growth factor-β (TGF-β) is a potent inducer of EMT. TGF-β directly activates the expression of transcription factors such as SNAI1/2, Twist and ZEB1/2. These factors are the key regulators of the EMT program[9].
The Src SH3 and SH2 domains cooperate with extracellular signal-regulated kinase (ERK), MEK (ERK kinase), myosin light chain kinase (MLCK), and Rho-dependent protein kinase (ROCK) signaling to accumulate phosphomyosin at the colon cancer cell periphery and promote a mesenchymal-like phenotype[6]. It was recently shown that actomyosin contractility is a key determinant of EMT[6].
Ras-mitogen-activated protein kinase has been shown to activate two related transcription factors, namely Snail and Slug, both of which are transcriptional repressors of E-cadherin, and their expression induces EMT[10].
Recently, activation of the phosphatidylinositol 3’-kinase (PI3K)/Akt axis has emerged as a central feature of EMT[11]. Activation of PI3K/Akt signaling is associated with growth and progression of CRC. It was reported that the mTOR kinase, a downstream effector of PI3K/Akt signaling, regulates tumorigenesis in CRC[12,13]. Increased expression of mTOR, Raptor, and Rictor mRNA was noted with advanced stages of CRC, suggesting that mTOR signaling may be associated with CRC progression and metastasis[14]. Gulhati et al[14] showed that the inhibition of mTORC1 and mTORC2 attenuated migration and invasion of colon cancer cells concomitant with altered cytoskeletal rearrangement and decreased activation of RhoA and Rac1. The inhibition of mTORC1 and mTORC2 induces changes reminiscent of mesenchymal-to-epithelial transition, and it was also shown that the establishment of metastasis in vivo was completely abolished upon targeted inhibition of mTORC1 and mTORC2. Based on these results, one may propose that mTORC1 and mTORC2 regulate motility of colon cancer cells via RhoA and Rac1 signaling[11]. Twist, another transcription factor, has been shown to possibly induce EMT too, and is also implicated in the regulation of metastasis[7,8].
Expression of FOXC2, an important element of embryonic development is supposed to induce EMT and regulate metastasis. In addition, the expression of FOXC2 is induced when epithelial cells undergo EMT by Snail, Twist, Goosecoid, and TGF-β1[15-17].
The majority of human colon cancers carry mutations that lead to the activation of Wnt signaling, a pathway that also has a pivotal role in intestinal stem cell biology[18]. Despite the underlying genetic background, cells within individual tumors display differential Wnt signaling, suggesting further regulation by the microenvironment. A local loss of basement membrane at the invasive edge has been suggested to expose cancer cells to a different microenvironment, which promotes Wnt signaling (nuclear β-catenin expression), EMT-like changes and loss of differentiation[19]. Type 1 collagen is a known component of the microenvironment at the host–tumor interface in CRC[20], and is more highly expressed in tumors displaying infiltrative growth compared with those with expansive growth[21]. Type 1 collagen also reduces CDX2 expression, an early marker of epithelial commitment, in human CRC cell lines in vitro[20], and enhances tumorigenicity in human CRC cells in xenografts[22]. Integrin α1β2 has a central role in type 1 collagen-induced EMT[23].
Hedgehog signaling cascade cross-talks with Wnt, epithelial growth factor/fibroblast growth factor, and TGFβ/Activin/Nodal/bone morphogenic protein signaling cascades, which are implicated in EMT through E-cadherin repression[8,24,25]. Although the Hedgehog signaling cascade induces the SNAI1 upregulation, there is no evidence for its direct SNAI1 transcriptional activation. On the other hand, Hedgehog signals induce JAG2 upregulation, and TGF-β1 secretion to promote motility and invasiveness of cancer cells[26]. JAG2 signaling induces transition of the Notch receptor to the Notch intracellular domain (NICD). NICD is then associated with the CSL transcription factor in the nucleus to induce SNAI1 upregulation[27]. TGF-β1 activates the TGF-β receptor for nuclear factor-κB-mediated transcriptional upregulation of ZEB1 and ZEB2 (zinc-finger transcription factors)[28], and also for the SMAD-Sp1-mediated transcriptional upregulation of mesenchymal markers, such as vimentin. Together these facts indicate that the Hedgehog signals indirectly induce EMT through the upregulation of multiple EMT regulators via Notch and TGF-β signaling cascades[29].
The 20-22 bp nucleotide noncoding RNAs, the microRNAs (miRNAs), regulate gene expression at post-transcriptional levels. Earlier profiling experiments have identified cohorts of miRNAs whose levels undergo significant changes upon TGF-β induced EMT, suggesting possible involvements of miRNAs in this process[30]. The miR 200 family has been linked to inhibition of EMT (promotion of the epithelial phenotype) through inhibition of ZEB1/2, known transcriptional repressors of the human E-cadherin gene[31].
In LIM 1863 colon carcinoma cells, the upregulation of miR-21 and miR-31 had been reported during EMT[32]. Overexpression as well as inhibition experiments support the contributions of both miR-21 and miR-31 not only in the TGF-β-induced morphological changes, but also in cell motility and invasion. It was also shown that T lymphoma and metastasis gene 1 (TIAM1) is a direct target of both miR-21 and miR-31, and that the suppression of TIAM1 is important for the pro-migration and invasion activities of miR-21 and miR-31. Based on these results, miR-21 and miR-31 were indentified as positive regulators of the EMT in colon carcinoma cells[32].
Interestingly, it was recently shown[33] that nicotine enhanced the expression level of fibronectin, an important EMT-related maker, in a dose-dependent manner. Furthermore, an α7-nicotinic acetylcholine receptor antagonist and siRNA reversed the nicotine-enhanced fibronectin expression in both SW480 and DLD-1 cells[33].
The switch between epithelial and mesenchymal phenotypes occurs during the advanced stages of cancer development. As a general rule, epithelial cancer cells that have undergone EMT are thought of as being more migratory, which may contribute to the invasive or metastatic phenotype.
In adult organisms, it has been proposed that restrictive mechanisms repress EMT and MET[34]. During tumor development, these mechanisms appear to fail, allowing EMT as described in metastasis generation[35]. Colonic stroma tissue, including subepithelial lymphoid aggregates surrounding the cancer cells, plays an important role in both EMT regulation and tumor behavior. Mesker et al[36] analyzed the expression of markers involved in pathways related to stroma production and EMT (β-catenin, TGF-β-R2, Smad4) in high-risk CRC patients, and found that patients with high stroma and Smad4 loss are at high risk. The anti-EMT effect of Smad4 was also proven in colon carcinoma cells[37].
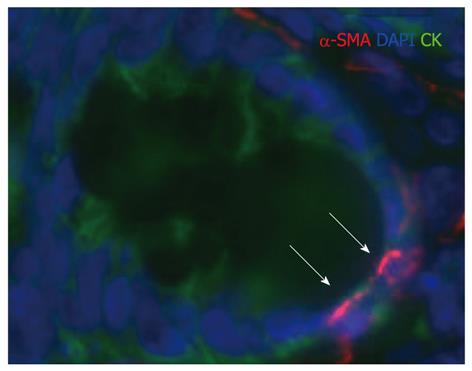
Besides colon cancer cell migration and invasion, EMT is also involved in organ fibrosis. The epithelium has been proposed to be a significant source of matrix-producing fibroblasts and of myofibroblasts[38]. Tissue accumulation of myofibroblasts shows strong correlation with the severity and progression of colonic fibrosis[39]. TGF-β1 has been long known as the chief inducer not just of EMT, but fibrosis, and myofibroblast generation. Accordingly, receptor Smads (Smad2 and particularly Smad3), the direct targets of the activated TGF-β receptor have been implicated as critical mediators in fibrogenesis and EMT[40,41] (Figure 2).
Inhibition of EMT would be an ideal choice for the treatment of CRC, as well as colonic fibrosis developed on the basis of chronic inflammation. Despite the incontrovertible rationale for treating diseases belonging to PI3K/Akt/mTOR signaling with rapamycin, clinical results have been disappointing. One proposed mechanism of resistance to rapamycin arises from its inability to inhibit mTORC2[14,42]. The findings of Gulhati et al[11] support a role for elevated mTORC1 and mTORC2 activity in regulating EMT and metastasis of CRC. Taken together with previous results, showing that both mTORC1 and mTORC2 contribute to CRC tumorigenesis[14], it may be hypothesized that the inherent redundancy in functions of both complexes may allow mTORC2 to compensate for loss of mTORC1 activity upon rapamycin treatment, thereby leading to rapamycin resistance. Based on these data, Gulhati et al[11] provides the rationale for including mTOR kinase inhibitors targeting the ATP binding pocket, which inhibit both mTORC1 and mTORC2 more completely, as part of the therapeutic regimen for treating CRC patients.
miRNAs targeted to mRNAs, encoding stem cell signaling components or EMT regulators, are also potent drug targets. miRNAs inducing proliferative, anti-apoptotic, pro-angiogenic, or pro-metastatic effects on tumor cells could be downregulated for cancer therapy, while those with proapoptotic, anti-angiogenic, or anti-metastatic effects could be applied for synthetic miRNA[43,44]. An RNA aptamer is a short RNA oligonucleotide with a stable 3D structure[44]. RNA aptamers binding to the extracellular region of Patched1 could be utilized for drug delivery to cancer cells with Hedgehog signaling activation. RNA aptamers binding to the cytoplasmic region of Smoothened, and those binding to Fused or GLI1 could be utilized as Hedgehog signaling inhibitors. Peptide mimetics, resembling Wnt and fibroblast growth factor family members, have been developed[45,46]. Because Wnt5A is involved in the non-canonical signaling cascade for the induction of EMT partly through SNAI1 upregulation[47-50], a Wnt5A mimetic is able to suppress invasion and metastasis of cancer cells. On the other hand, great care should be taken before clinical application of these technologies, as the miRNA and siRNA off-target effects are serious problems.
Mesenchymal-to-epithelial transition is a reversible biological process that involves the transition from motile, multipolar or spindle-shaped mesenchymal cells to polarized epithelial cells. MET, just like EMT, also takes place during normal development in processes such as somitogenesis, kidney development, cardiogenesis, hepatogenesis and celomic cavity formation[4,51,52], moreover MET occurs in cancer metastasis, induced pluripotent stem cell reprogramming and mucosal healing[5].
While the mechanism in which MET occurs during each organ morphogenesis is similar, in that epithelium-associated genes are upregulated and mesenchymal ones are downregulated, each process has a unique signaling pathway to induce MET and related changes in gene expression.
Though our knowledge about the mechanism and regulation of EMT in the colon is increasing nowadays, the reverse phenomenon, MET, is still not a well understood mechanism (Figure 1).
MET during carcinogenesis has been shown to be induced by the c-met proto-oncogene[53-55]. C-met, also known as hepatocyte growth factor receptor, is a receptor tyrosine kinase for hepatocyte growth factor and its increased expression leads to epithelial differentiation[56,57]. In addition to epithelial specification by C-met, 5-azacytidine, a DNA methyltransferase inhibitor with broad spectrum epigenetic effects, has been used to induce MET in vitro[58]. Recent research on the transcription factor Snail has been linked to aberrant DNA methylation of the epithelial specific E-cadherin promoter in association with EMT, and stable RNA interference of Snail expression in carcinoma cell lines induced a complete MET[59-61].
Frizzled-7 (FZD7) is necessary for MET in the LIM1863-Mph CRC model. The loss of FZD7 in cancer cells results in the persistence of a mesenchymal state (increased SNAI2/decreased E-cadherin)[62]. Moreover, FZD7 is also required for migration of the LIM1863-Mph monolayer cells. This suggests that FZD7 induced either migratory or epithelialization events depending on the context.
The role of DNA methylation in MET induction and induced pluripotent stem (iPS) cell reprogramming had been also highlighted[63].
In the inflamed colon, signs of MET can be detected in the subepithelial lymphoid follicles[64-66]. These data suggest that migrating stem cells undergoing MET may be sensitive to the chemokine/cytokine milieu of the inflammatory environment.
Regarding the colon, the need for MET is high when severe mucosal damage is present. In inflammation, MET can also be altered because mesenchymal stem cells are mobilized to the site of injury and consequently subjected to the inflammatory response[67]. Bone marrow-derived stem cells could differentiate into mature-appearing epithelial cells in response to tissue damage[68]. It was recently published that versican, a large chondroitin sulfate proteoglycan, mediates MET[69]. The results of Hirose et al[70] indicate that versican can bind specific chemokines through its chondroitin sulfate chains and by doing so, it tends to downregulate the chemokine function. This raises the possibility that versican is a potent regenerative factor in the colonic mucosa.
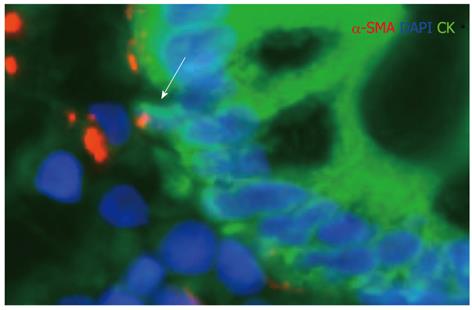
In the inflamed colon, the presence of CDX2- and cytokeratin-positive subepithelial cells in the marginal zone of subepithelial lymphoid follicles also suggests that MET may have a role in colonic mucosal regeneration[64]. Presumably the mesenchymal cells committed to an epithelial fate are sensitive for the regeneration-associated paracrine cytokine and chemokine environment in the inflammatory stroma (Figure 3).
Induced pluripotent stem cells can be derived from somatic cells by the induction of a small number of genes, like POU5F1, MYC, KLF4 and SOX2[71-74]. Originating from an individual’s own tissue, iPS cells offer considerable therapeutic promise, avoiding both immunologic and ethical barriers to their use. Upon induction, mouse fibroblasts must undergo MET to successfully begin the initiation phase of reprogramming. Epithelial-associated genes (i.e., E-cadherin, Claudin-3, -4, -7, -11, occludin, epithelial cell adhesion molecule, Crumbs homolog 3) were all upregulated before the turning on of Nanog, a key transcription factor in maintaining pluripotency. Additionally, mesenchymal-associated genes like Snail, Slug, Zeb-1/2, and N-cadherin were downregulated[75].
Epigenetic changes, DNA methylation, seems to be also involved in MET regulation, which may also have therapeutic significance[63].
According to our current knowledge, both EMT and MET are highly significant biological events, not just in physiological, but in pathological circumstances. A better understanding of their induction and regulation may lead to the identification of pathways and factors that can be potent therapeutic targets.
The inhibition of EMT seems to be a useful technique to avoid CRC cell invasion and metastasis generation, and chronic inflammation-related colonic wall fibrosis may be also delayed, or even reversed.
The induction of MET is also of major clinical importance. In the case of severe mucosal damage, the time for complete epithelial regeneration may be reduced by enhancing the phenotype change of mesenchymal stem cells to epithelial cells.
The preliminary results of ongoing studies are promising, but more investigations are needed to develop new therapeutic strategies that can be safely used in the near future.
Peer reviewers: Stefano Pontone, Dr., Department of Surgical Sciences, “Sapienza” University of Rome, Viale Regina Margherita 324, Rome, Italy; Wai Lun Law, FACS, Clinical Professor, Chief, Division of Colorectal Surgery, Department of Surgery, Queen Mary Hospital, The University of Hong Kong, Pokfulam Road, Hong Kong, China
S- Editor Lv S L- Editor Cant MR E- Editor Zhang DN
| 1. | Center MM, Jemal A, Ward E. International trends in colorectal cancer incidence rates. Cancer Epidemiol Biomarkers Prev. 2009;18:1688-1694. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 532] [Cited by in RCA: 566] [Article Influence: 35.4] [Reference Citation Analysis (0)] |
| 2. | Thukkani N, Williams JL, Sonnenberg A. Epidemiologic characteristics of patients with inflammatory bowel disease undergoing colonoscopy. Inflamm Bowel Dis. 2011;17:1333-1337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 3. | Thiery JP. Epithelial-mesenchymal transitions in development and pathologies. Curr Opin Cell Biol. 2003;15:740-746. [PubMed] |
| 4. | Rubio D, Garcia S, De la Cueva T, Paz MF, Lloyd AC, Bernad A, Garcia-Castro J. Human mesenchymal stem cell transformation is associated with a mesenchymal-epithelial transition. Exp Cell Res. 2008;314:691-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 73] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 5. | Sipos F, Muzes G. Isolated lymphoid follicles in colon: switch points between inflammation and colorectal cancer? World J Gastroenterol. 2011;17:1666-1673. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 37] [Cited by in RCA: 37] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 6. | Avizienyte E, Brunton VG, Fincham VJ, Frame MC. The SRC-induced mesenchymal state in late-stage colon cancer cells. Cells Tissues Organs. 2005;179:73-80. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 7. | Thiery JP. Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer. 2002;2:442-454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4877] [Cited by in RCA: 5116] [Article Influence: 222.4] [Reference Citation Analysis (0)] |
| 8. | Lee JM, Dedhar S, Kalluri R, Thompson EW. The epithelial-mesenchymal transition: new insights in signaling, development, and disease. J Cell Biol. 2006;172:973-981. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1482] [Cited by in RCA: 1597] [Article Influence: 84.1] [Reference Citation Analysis (0)] |
| 9. | Moustakas A, Heldin CH. Signaling networks guiding epithelial-mesenchymal transitions during embryogenesis and cancer progression. Cancer Sci. 2007;98:1512-1520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 10. | Waerner T, Alacakaptan M, Tamir I, Oberauer R, Gal A, Brabletz T, Schreiber M, Jechlinger M, Beug H. ILEI: a cytokine essential for EMT, tumor formation, and late events in metastasis in epithelial cells. Cancer Cell. 2006;10:227-239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 152] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 11. | Gulhati P, Bowen KA, Liu J, Stevens PD, Rychahou PG, Chen M, Lee EY, Weiss HL, O'Connor KL, Gao T. mTORC1 and mTORC2 regulate EMT, motility, and metastasis of colorectal cancer via RhoA and Rac1 signaling pathways. Cancer Res. 2011;71:3246-3256. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 443] [Cited by in RCA: 463] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 12. | Rychahou PG, Jackson LN, Silva SR, Rajaraman S, Evers BM. Targeted molecular therapy of the PI3K pathway: therapeutic significance of PI3K subunit targeting in colorectal carcinoma. Ann Surg. 2006;243:833-842; discussion 843-844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 95] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 13. | Rychahou PG, Kang J, Gulhati P, Doan HQ, Chen LA, Xiao SY, Chung DH, Evers BM. Akt2 overexpression plays a critical role in the establishment of colorectal cancer metastasis. Proc Natl Acad Sci USA. 2008;105:20315-20320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 147] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 14. | Gulhati P, Cai Q, Li J, Liu J, Rychahou PG, Qiu S, Lee EY, Silva SR, Bowen KA, Gao T. Targeted inhibition of mammalian target of rapamycin signaling inhibits tumorigenesis of colorectal cancer. Clin Cancer Res. 2009;15:7207-7216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 139] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 15. | Sano H, Leboeuf JP, Novitskiy SV, Seo S, Zaja-Milatovic S, Dikov MM, Kume T. The Foxc2 transcription factor regulates tumor angiogenesis. Biochem Biophys Res Commun. 2010;392:201-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 51] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 16. | Hader C, Marlier A, Cantley L. Mesenchymal-epithelial transition in epithelial response to injury: the role of Foxc2. Oncogene. 2010;29:1031-1040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 17. | Hugo HJ, Kokkinos MI, Blick T, Ackland ML, Thompson EW, Newgreen DF. Defining the E-cadherin repressor interactome in epithelial-mesenchymal transition: the PMC42 model as a case study. Cells Tissues Organs. 2011;193:23-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 59] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 18. | Reya T, Clevers H. Wnt signalling in stem cells and cancer. Nature. 2005;434:843-850. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2659] [Cited by in RCA: 2765] [Article Influence: 138.3] [Reference Citation Analysis (0)] |
| 19. | Spaderna S, Schmalhofer O, Hlubek F, Berx G, Eger A, Merkel S, Jung A, Kirchner T, Brabletz T. A transient, EMT-linked loss of basement membranes indicates metastasis and poor survival in colorectal cancer. Gastroenterology. 2006;131:830-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 384] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 20. | Brabletz T, Spaderna S, Kolb J, Hlubek F, Faller G, Bruns CJ, Jung A, Nentwich J, Duluc I, Domon-Dell C. Down-regulation of the homeodomain factor Cdx2 in colorectal cancer by collagen type I: an active role for the tumor environment in malignant tumor progression. Cancer Res. 2004;64:6973-6977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 110] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 21. | Oku Y, Shimoji T, Takifuji K, Hotta T, Yokoyama S, Matsuda K, Higashiguchi T, Tominaga T, Nasu T, Tamura K. Identification of the molecular mechanisms for dedifferentiation at the invasion front of colorectal cancer by a gene expression analysis. Clin Cancer Res. 2008;14:7215-7222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 22. | Del Buono R, Pignatelli M, Hall PA. Control of differentiation in a rectal adenocarcinoma cell line: the role of diffusable and cell-associated factors. J Pathol. 1991;164:59-66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 23. | Kirkland SC. Type I collagen inhibits differentiation and promotes a stem cell-like phenotype in human colorectal carcinoma cells. Br J Cancer. 2009;101:320-326. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 110] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 24. | van den Brink GR, Bleuming SA, Hardwick JC, Schepman BL, Offerhaus GJ, Keller JJ, Nielsen C, Gaffield W, van Deventer SJ, Roberts DJ. Indian Hedgehog is an antagonist of Wnt signaling in colonic epithelial cell differentiation. Nat Genet. 2004;36:277-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 273] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 25. | Bailey JM, Singh PK, Hollingsworth MA. Cancer metastasis facilitated by developmental pathways: Sonic hedgehog, Notch, and bone morphogenic proteins. J Cell Biochem. 2007;102:829-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 160] [Cited by in RCA: 156] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 26. | Yoo YA, Kang MH, Kim JS, Oh SC. Sonic hedgehog signaling promotes motility and invasiveness of gastric cancer cells through TGF-beta-mediated activation of the ALK5-Smad 3 pathway. Carcinogenesis. 2008;29:480-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 129] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 27. | Sahlgren C, Gustafsson MV, Jin S, Poellinger L, Lendahl U. Notch signaling mediates hypoxia-induced tumor cell migration and invasion. Proc Natl Acad Sci USA. 2008;105:6392-6397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 568] [Cited by in RCA: 651] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 28. | Chua HL, Bhat-Nakshatri P, Clare SE, Morimiya A, Badve S, Nakshatri H. NF-kappaB represses E-cadherin expression and enhances epithelial to mesenchymal transition of mammary epithelial cells: potential involvement of ZEB-1 and ZEB-2. Oncogene. 2007;26:711-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 439] [Cited by in RCA: 505] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 29. | Katoh Y, Katoh M. Hedgehog signaling, epithelial-to-mesenchymal transition and miRNA (review). Int J Mol Med. 2008;22:271-275. [PubMed] |
| 30. | Zavadil J, Narasimhan M, Blumenberg M, Schneider RJ. Transforming growth factor-beta and microRNA: mRNA regulatory networks in epithelial plasticity. Cells Tissues Organs. 2007;185:157-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 118] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 31. | Loboda A, Nebozhyn MV, Watters JW, Buser CA, Shaw PM, Huang PS, Van't Veer L, Tollenaar RA, Jackson DB, Agrawal D. EMT is the dominant program in human colon cancer. BMC Med Genomics. 2011;4:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 228] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 32. | Cottonham CL, Kaneko S, Xu L. miR-21 and miR-31 converge on TIAM1 to regulate migration and invasion of colon carcinoma cells. J Biol Chem. 2010;285:35293-35302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 224] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 33. | Wei PL, Kuo LJ, Huang MT, Ting WC, Ho YS, Wang W, An J, Chang YJ. Nicotine enhances colon cancer cell migration by induction of fibronectin. Ann Surg Oncol. 2011;18:1782-1790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 76] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 34. | Prindull G, Zipori D. Environmental guidance of normal and tumor cell plasticity: epithelial mesenchymal transitions as a paradigm. Blood. 2004;103:2892-2899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 94] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 35. | Kang Y, Massagué J. Epithelial-mesenchymal transitions: twist in development and metastasis. Cell. 2004;118:277-279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1074] [Cited by in RCA: 1182] [Article Influence: 56.3] [Reference Citation Analysis (0)] |
| 36. | Mesker WE, Liefers GJ, Junggeburt JM, van Pelt GW, Alberici P, Kuppen PJ, Miranda NF, van Leeuwen KA, Morreau H, Szuhai K. Presence of a high amount of stroma and downregulation of SMAD4 predict for worse survival for stage I-II colon cancer patients. Cell Oncol. 2009;31:169-178. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 73] [Reference Citation Analysis (0)] |
| 37. | Pohl M, Radacz Y, Pawlik N, Schoeneck A, Baldus SE, Munding J, Schmiegel W, Schwarte-Waldhoff I, Reinacher-Schick A. SMAD4 mediates mesenchymal-epithelial reversion in SW480 colon carcinoma cells. Anticancer Res. 2010;30:2603-2613. [PubMed] |
| 38. | Willis BC, Borok Z. TGF-beta-induced EMT: mechanisms and implications for fibrotic lung disease. Am J Physiol Lung Cell Mol Physiol. 2007;293:L525-L534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 695] [Cited by in RCA: 817] [Article Influence: 45.4] [Reference Citation Analysis (0)] |
| 39. | Ma Y, Guan Q, Bai A, Weiss CR, Hillman CL, Ma A, Zhou G, Qing G, Peng Z. Targeting TGF-beta1 by employing a vaccine ameliorates fibrosis in a mouse model of chronic colitis. Inflamm Bowel Dis. 2010;16:1040-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 43] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 40. | Di Sabatino A, Jackson CL, Pickard KM, Buckley M, Rovedatti L, Leakey NA, Picariello L, Cazzola P, Monteleone G, Tonelli F. Transforming growth factor beta signalling and matrix metalloproteinases in the mucosa overlying Crohn's disease strictures. Gut. 2009;58:777-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 176] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 41. | Masszi A, Kapus A. Smaddening complexity: the role of Smad3 in epithelial-myofibroblast transition. Cells Tissues Organs. 2011;193:41-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 53] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 42. | Sarbassov DD, Ali SM, Sengupta S, Sheen JH, Hsu PP, Bagley AF, Markhard AL, Sabatini DM. Prolonged rapamycin treatment inhibits mTORC2 assembly and Akt/PKB. Mol Cell. 2006;22:159-168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1994] [Cited by in RCA: 2109] [Article Influence: 111.0] [Reference Citation Analysis (0)] |
| 43. | Marquez RT, McCaffrey AP. Advances in microRNAs: implications for gene therapists. Hum Gene Ther. 2008;19:27-38. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 44. | Katoh M. RNA technology targeted to the WNT signaling pathway. Cancer Biol Ther. 2008;7:275-277. [PubMed] |
| 45. | Säfholm A, Leandersson K, Dejmek J, Nielsen CK, Villoutreix BO, Andersson T. A formylated hexapeptide ligand mimics the ability of Wnt-5a to impair migration of human breast epithelial cells. J Biol Chem. 2006;281:2740-2749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 93] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 46. | Li S, Christensen C, Kiselyov VV, Køhler LB, Bock E, Berezin V. Fibroblast growth factor-derived peptides: functional agonists of the fibroblast growth factor receptor. J Neurochem. 2008;104:667-682. [PubMed] |
| 47. | Katoh M, Katoh M. Comparative genomics on ROR1 and ROR2 orthologs. Oncol Rep. 2005;14:1381-1384. [PubMed] |
| 48. | Fukuda T, Chen L, Endo T, Tang L, Lu D, Castro JE, Widhopf GF, Rassenti LZ, Cantwell MJ, Prussak CE. Antisera induced by infusions of autologous Ad-CD154-leukemia B cells identify ROR1 as an oncofetal antigen and receptor for Wnt5a. Proc Natl Acad Sci USA. 2008;105:3047-3052. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 266] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 49. | Katoh M, Katoh M. STAT3-induced WNT5A signaling loop in embryonic stem cells, adult normal tissues, chronic persistent inflammation, rheumatoid arthritis and cancer (Review). Int J Mol Med. 2007;19:273-278. [PubMed] |
| 50. | Dissanayake SK, Wade M, Johnson CE, O'Connell MP, Leotlela PD, French AD, Shah KV, Hewitt KJ, Rosenthal DT, Indig FE. The Wnt5A/protein kinase C pathway mediates motility in melanoma cells via the inhibition of metastasis suppressors and initiation of an epithelial to mesenchymal transition. J Biol Chem. 2007;282:17259-17271. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 293] [Cited by in RCA: 283] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 51. | Nakajima Y, Yamagishi T, Hokari S, Nakamura H. Mechanisms involved in valvuloseptal endocardial cushion formation in early cardiogenesis: roles of transforming growth factor (TGF)-beta and bone morphogenetic protein (BMP). Anat Rec. 2000;258:119-127. [PubMed] |
| 52. | Li B, Zheng YW, Sano Y, Taniguchi H. Evidence for mesenchymal-epithelial transition associated with mouse hepatic stem cell differentiation. PLoS One. 2011;6:e17092. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 53. | Ouyang P. An in vitro model to study mesenchymal-epithelial transformation. Biochem Biophys Res Commun. 1998;246:771-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 54. | Naka T, Iwamoto Y, Shinohara N, Ushijima M, Chuman H, Tsuneyoshi M. Expression of c-met proto-oncogene product (c-MET) in benign and malignant bone tumors. Mod Pathol. 1997;10:832-838. [PubMed] |
| 55. | Scotlandi K, Baldini N, Oliviero M, Di Renzo MF, Martano M, Serra M, Manara MC, Comoglio PM, Ferracini R. Expression of Met/hepatocyte growth factor receptor gene and malignant behavior of musculoskeletal tumors. Am J Pathol. 1996;149:1209-1219. [PubMed] |
| 56. | Jeffers M, Rong S, Woude GF. Hepatocyte growth factor/scatter factor-Met signaling in tumorigenicity and invasion/metastasis. J Mol Med (. Berl). 1996;74:505-513. [PubMed] |
| 57. | Tsarfaty I, Rong S, Resau JH, Rulong S, da Silva PP, Vande Woude GF. The Met proto-oncogene mesenchymal to epithelial cell conversion. Science. 1994;263:98-101. [PubMed] |
| 58. | Darmon M, Nicolas JF, Lamblin D. 5-Azacytidine is able to induce the conversion of teratocarcinoma-derived mesenchymal cells into epithelia cells. EMBO J. 1984;3:961-967. [PubMed] |
| 59. | Jordà M, Vinyals A, Marazuela A, Cubillo E, Olmeda D, Valero E, Cano A, Fabra A. Id-1 is induced in MDCK epithelial cells by activated Erk/MAPK pathway in response to expression of the Snail and E47 transcription factors. Exp Cell Res. 2007;313:2389-2403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 60. | Bathula CS, Garrett SH, Zhou XD, Sens MA, Sens DA, Somji S. Cadmium, vectorial active transport, and MT-3-dependent regulation of cadherin expression in human proximal tubular cells. Toxicol Sci. 2008;102:310-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 61. | Fitzgerald MP, Gourronc F, Teoh ML, Provenzano MJ, Case AJ, Martin JA, Domann FE. Human Chondrosarcoma Cells Acquire an Epithelial-Like Gene Expression Pattern via an Epigenetic Switch: Evidence for Mesenchymal-Epithelial Transition during Sarcomagenesis. Sarcoma. 2011;2011:598218. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 62. | Vincan E, Darcy PK, Farrelly CA, Faux MC, Brabletz T, Ramsay RG. Frizzled-7 dictates three-dimensional organization of colorectal cancer cell carcinoids. Oncogene. 2007;26:2340-2352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 60] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 63. | Doi A, Park IH, Wen B, Murakami P, Aryee MJ, Irizarry R, Herb B, Ladd-Acosta C, Rho J, Loewer S. Differential methylation of tissue- and cancer-specific CpG island shores distinguishes human induced pluripotent stem cells, embryonic stem cells and fibroblasts. Nat Genet. 2009;41:1350-1353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 912] [Cited by in RCA: 874] [Article Influence: 54.6] [Reference Citation Analysis (0)] |
| 64. | Sipos F, Muzes G, Valcz G, Galamb O, Tóth K, Leiszter K, Krenács T, Tulassay Z, Molnár B. Regeneration associated growth factor receptor and epithelial marker expression in lymphoid aggregates of ulcerative colitis. Scand J Gastroenterol. 2010;45:440-448. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 65. | Sipos F, Galamb O, Herszényi L, Molnár B, Solymosi N, Zágoni T, Berczi L, Tulassay Z. Elevated insulin-like growth factor 1 receptor, hepatocyte growth factor receptor and telomerase protein expression in mild ulcerative colitis. Scand J Gastroenterol. 2008;43:289-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 66. | Sipos F, Molnár B, Zágoni T, Berczi L, Tulassay Z. Growth in epithelial cell proliferation and apoptosis correlates specifically to the inflammation activity of inflammatory bowel diseases: ulcerative colitis shows specific p53- and EGFR expression alterations. Dis Colon Rectum. 2005;48:775-786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 41] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 67. | Spees JL, Olson SD, Ylostalo J, Lynch PJ, Smith J, Perry A, Peister A, Wang MY, Prockop DJ. Differentiation, cell fusion, and nuclear fusion during ex vivo repair of epithelium by human adult stem cells from bone marrow stroma. Proc Natl Acad Sci USA. 2003;100:2397-2402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 403] [Cited by in RCA: 389] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 68. | Van Arnam JS, Herzog E, Grove J, Bruscia E, Ziegler E, Swenson S, Krause DS. Engraftment of bone marrow-derived epithelial cells. Stem Cell Rev. 2005;1:21-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 69. | Sheng W, Wang G, La Pierre DP, Wen J, Deng Z, Wong CK, Lee DY, Yang BB. Versican mediates mesenchymal-epithelial transition. Mol Biol Cell. 2006;17:2009-2020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 74] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 70. | Hirose J, Kawashima H, Yoshie O, Tashiro K, Miyasaka M. Versican interacts with chemokines and modulates cellular responses. J Biol Chem. 2001;276:5228-5234. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 164] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 71. | Takahashi K, Yamanaka S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17989] [Cited by in RCA: 18186] [Article Influence: 957.2] [Reference Citation Analysis (0)] |
| 72. | Takahashi K, Tanabe K, Ohnuki M, Narita M, Ichisaka T, Tomoda K, Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14327] [Cited by in RCA: 14312] [Article Influence: 841.9] [Reference Citation Analysis (0)] |
| 73. | Park IH, Zhao R, West JA, Yabuuchi A, Huo H, Ince TA, Lerou PH, Lensch MW, Daley GQ. Reprogramming of human somatic cells to pluripotency with defined factors. Nature. 2008;451:141-146. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2182] [Cited by in RCA: 2125] [Article Influence: 118.1] [Reference Citation Analysis (0)] |
| 74. | Yu J, Vodyanik MA, Smuga-Otto K, Antosiewicz-Bourget J, Frane JL, Tian S, Nie J, Jonsdottir GA, Ruotti V, Stewart R. Induced pluripotent stem cell lines derived from human somatic cells. Science. 2007;318:1917-1920. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7589] [Cited by in RCA: 7243] [Article Influence: 402.4] [Reference Citation Analysis (0)] |
| 75. | Samavarchi-Tehrani P, Golipour A, David L, Sung HK, Beyer TA, Datti A, Woltjen K, Nagy A, Wrana JL. Functional genomics reveals a BMP-driven mesenchymal-to-epithelial transition in the initiation of somatic cell reprogramming. Cell Stem Cell. 2010;7:64-77. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 792] [Cited by in RCA: 801] [Article Influence: 53.4] [Reference Citation Analysis (0)] |