Published online Dec 14, 2012. doi: 10.3748/wjg.v18.i46.6836
Revised: March 20, 2012
Accepted: May 13, 2012
Published online: December 14, 2012
AIM: Evaluation of the wide range of normal findings in asymptomatic women undergoing dynamic magnetic resonance (MR) defecography.
METHODS: MR defecography of 10 healthy female volunteers (median age: 31 years) without previous pregnancies or history of surgery were evaluated. The rectum was filled with 180 mL gadolinium ultrasound gel mixture. MR defecography was performed in the supine position. The pelvic floor was visualized with a dynamic T2-weighted sagittal plane where all relevant pelvic floor organs were acquired during defecation. The volunteers were instructed to relax and then to perform straining maneuvers to empty the rectum. The pubococcygeal line (PCGL) was used as the line of reference. The movement of pelvic floor organs was measured as the vertical distance to this reference line. Data were recorded in the resting position as well as during the defecation process with maximal straining. Examinations were performed and evaluated by two experienced abdominal radiologists without knowledge of patient history.
RESULTS: Average position of the anorectal junction was located at -5.3 mm at rest and -29.9 mm during straining. The anorectal angle widened significantly from 93° at rest to 109° during defecation. A rectocele was diagnosed in eight out of 10 volunteers showing an average diameter of 25.9 mm. The bladder base was located at a position of +23 mm at rest and descended to -8.1 mm during defecation in relation to the PCGL. The bladder base moved below the PCGL in six out of 10 volunteers, which was formally defined as a cystocele. The uterocervical junction was located at an average level of +43.1 mm at rest and at +7.9 mm during straining. The uterocervical junction of three volunteers fell below the PCGL; described formally as uterocervical prolapse.
CONCLUSION: Based on the range of standard values in asymptomatic volunteers, MR defecography values for pathological changes have to be re-evaluated.
- Citation: Schreyer AG, Paetzel C, Fürst A, Dendl LM, Hutzel E, Müller-Wille R, Wiggermann P, Schleder S, Stroszczynski C, Hoffstetter P. Dynamic magnetic resonance defecography in 10 asymptomatic volunteers. World J Gastroenterol 2012; 18(46): 6836-6842
- URL: https://www.wjgnet.com/1007-9327/full/v18/i46/6836.htm
- DOI: https://dx.doi.org/10.3748/wjg.v18.i46.6836
As a result of the complex anatomy and synergism of pelvic organs and their muscular structures, there is a wide variety of static and functional disorders. Outlet obstruction, anism, dyskinesia of the puborectal muscle, intussusception, prolapse of the anus and rectum, vaginal prolapse, rectocele, cystocele, and enterocele represent common diagnoses in proctology, urology and gynecology. Besides constipation, fecal or urinary incontinence is the most common symptom, with serious problems for the patients that have a major negative impact on quality of life. Women are affected at a significantly higher rate with a ratio of 9:1[1]. Patients usually attend gynecologists, urologists and proctologists. Therefore, a comprehensive interdisciplinary approach for diagnosis and therapy is the most promising strategy.
A systematic medical history as well as a thorough proctological examination (inspection, palpation, rectoscopy, and proctoscopy) along with manometry and endosonography is the basic diagnostic approach for complex pelvic floor disorders. Furthermore, radiological imaging with evacuation proctography can give important additional information[2].
With the development of magnetic resonance imaging (MRI) technology, dynamic MRI of the pelvic floor has become an important alternative for the diagnosis of complex combined pelvic floor disorders. Since its first introduction by Yang et al[3] and Kruyt et al[4] in 1991, MRI has increasingly replaced evacuation proctography, which was first described in 1952 by Walldén[5], for evaluation of outlet obstruction.
For the evaluation of extraluminal pelvic disorders such as enterocele or utero-vaginal prolapse, MRI of the pelvic floor is favorable in several aspects compared to clinical examination and conventional evacuation proctography, which does not depict extraluminal structures[3,4,6-10]. Additionally, MRI examination can be repeated because of the total lack of ionizing radiation. This may increase the chances of detecting pathological findings in some patients[9]. The horizontal position of the patient during MR defecography may be a disadvantage, because it could influence the pelvic floor physiology as well as the dynamic defecation process. Therefore, some authors consider that videoproctography is still superior to MRI for assessment of an enterocele or rectocele[11,12].
So far, patient preparation, examination technique, as well as reference lines for the evaluation of MRI are still not standardized and findings differ widely in the current literature. Most of the previously performed studies did not examine the defecation process itself. Normal findings and values were defined in small control groups and therefore were only applicable within the particular study setting.
The aim of the present study was to show the wide range of normal findings in asymptomatic female volunteers and therefore the necessity of obtaining common standards not only in terms of patient preparation, but also in the evaluation of numeric values for the definition of pathological and normal dynamic MR examinations of the pelvic floor.
Ten healthy female volunteers (median age: 31 years, range: 22-40 years) without previous pregnancy or history of any gynecological, urological or proctological surgery were evaluated by dynamic pelvic MRI. The volunteers had no symptoms of incontinence, constipation or other stool evacuation problems. None of the volunteers had any contraindications for MRI. Informed consent was obtained from all volunteers. We applied a uniform admission questionnaire for standardized documentation of patient history. The study was approved by the local ethics committee.
All healthy volunteers were asked to empty their bladder 3 h before the examination to achieve a medium filling of the urinary bladder during MRI. The rectum was filled with 180 mL ultrasound gel mixture (1% Gd-DTPA-GEL-Mixture). MRI (1.5 Tesla MRI, Magnetom Symphony; Siemens, Erlangen, Germany) was performed in the supine position with hips and knees bent at 45°. The pelvic floor was visualized in three planes (transversal, coronal, sagittal, T1 and T2) to find the appropriate sagittal plane in which all relevant pelvic floor organs were acquired during defecation over 55 s at a frequency of one shot per 1.1 s [True FISP (True Fast Imaging with Steady State Precession), TR: 5.8 ms, TE: 2.8 ms). Slice thickness was 7 mm (field of view: 270 mm × 270 mm, matrix 256 × 256). During the examination, healthy volunteers were instructed via headphones. They first were asked to relax and then to perform straining maneuvers to empty the rectum as completely as possible. The sequences were acquired digitally and analyzed cinematographically.
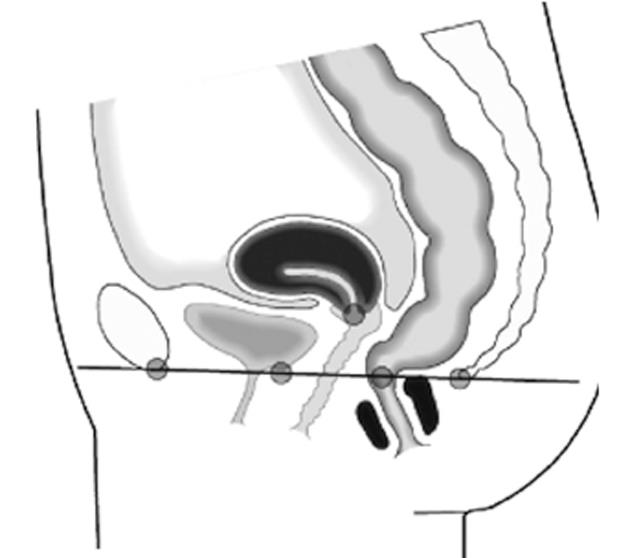
The pubococcygeal line (PCGL) was defined as the line of reference (Figure 1). Movement of pelvic floor organs was measured as the vertical distance with reference to this line. The location of structures above the PCGL were marked with positive values (+), structures located below this line were marked negative (-).
Data were recorded in the resting position as well as during the defecation process with maximal straining. All data represents mean values with corresponding SD.
The anorectal junction was defined as the intersection point of the central axis of the anal canal and a line along the posterior rectal wall. The anorectal angle was measured between these two lines[3,7,9].
According to the current literature, based on different evaluation techniques, a rectocele is defined as a bulge of the anterior rectal wall of > 20-30 mm[11,13]. For our data analysis we applied the method described by Delemarre, who defined the distance from the anorectal junction to the tip of the protrusion of the anterior rectal wall as the correct measurement[11].
MRI revealed data about the position of the bladder base, the uterocervical junction and the vagina. Additionally the location of small bowel in relation to the PCGL was assessed.
A cystocele and a uterovaginal prolapse were diagnosed if the bladder base or the uterocervical junction fell below the PCGL during defecation[9,10]. Widening of the rectovaginal space or a descent of mesenteric parts, small bowel or sigmoid colon beyond the PCGL during defecation was defined as an enterocele[8,10].
All examinations were performed and evaluated in consensus by two experienced board-certified abdominal radiologists without any knowledge of the volunteers history or age.
All 10 volunteers were able to hold the rectal enema and perform defecation within the MR scanner according to the examination protocol. The average position of the anorectal junction was located at -5.3 mm (± 9.9 mm) at rest and descended to -29.9 mm (± 10.3 mm) during maximal straining. The average anorectal angle widened significantly from 93° (± 4.8°) at rest to 108.7° (± 14.7°) at maximal straining during defecation. A rectocele based on the current definition of an anterior bulging of the anterior rectum wall of at least 20 mm was diagnosed in eight of 10 volunteers, showing an average size of 26 mm (± 6 mm).
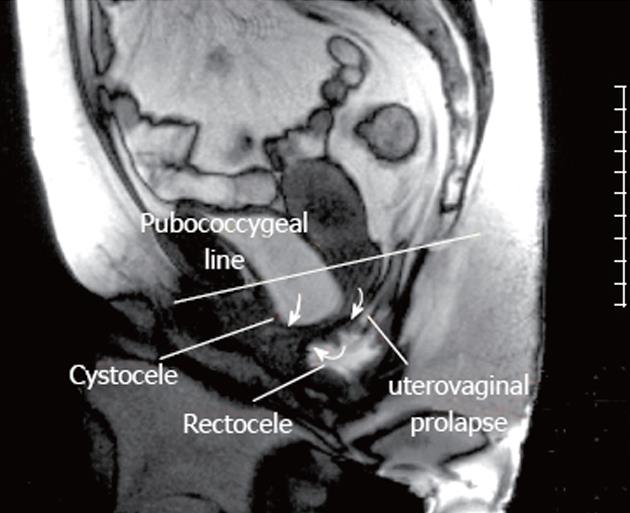
The bladder base was located at an average position of +23 mm (± 4.6 mm) at rest and descended to -8.1 mm (± 11.1 mm) during defecation. The bladder base moved below the PCGL in six of 10 healthy volunteers during maximal straining, which is defined as a cystocele (Figure 2).
The uterocervical junction or vaginal vault was located at an average level of +43.1 mm (± 7.8 mm) at rest and at +7.9 mm (± 16.5 mm) during maximal straining. The uterocervical junction of three volunteers descended below the PCGL at maximal straining during defecation, described as uterocervical prolapse according to the common definition in the literature (Figure 2). No enterocele or other additional pathological findings were detected in any volunteers. Table 1 shows an overview of all results and findings of the dynamic MRI examinations.
| Anatomic structure | Rest | Straining | Relative movement |
| Annorectal junction (mm) | -5.3 (± 9.9) | -29.9 (± 10.3) | -24.6 |
| Anorectal angle (°) | 93. 0 (± 4.8) | 108.7 (± 14. 7) | 15.7 |
| Bladder base (mm) | 23.0 (± 4.6) | -8.1 (± 11.1) | -14.9 |
| Uterovaginal junction (mm) | 43.1 (± 7.8) | 7.9 (± 16.5) | -35.2 |
Dynamic MRI of the pelvic floor, also known as MR defecography, is increasingly used for the diagnosis of complex pelvic floor disorders and replaces conventional videoproctography. Different medical specialties such as radiology, urology, gynecology and surgery are involved in the interpretation and evaluation of findings and therefore need an accepted definition of normal values and clinical practical reference lines for image assessment.
Back in 1991, Kruyt et al[4] were the first authors using MRI to study functional aspects of the anorectal region. In their study, the anatomy of the anorectum and the anorectal angle was examined in 10 healthy volunteers placed in the prone position within the MRI scanner. The symphysiosacral line reaching from the most superior part of the pubic symphysis to the lower part of the sacrum, was taken as the reference line to evaluate the mobility of the pelvic organs. Dynamics were measured during contraction and relaxation of the pelvic floor and during straining. In this study, the defecation process itself was not visualized because no contrast medium or enema was applied to the rectum.
Also in 1991, Yang et al[3] introduced dynamic MRI as a new method for the diagnosis of the descending perineum in women. They compared 26 patients with 16 asymptomatic women in the supine position. Values were documented with various degrees of straining maneuvers. Again, defecation itself was not because the rectum was not filled with contrast medium. The PCGL was taken as the reference line to evaluate the degree of descent of the pelvic structures.
Another study comparing clinical examination, videoproctography and dynamic MRI in the diagnosis of anterior rectoceles was published by Delemarre et al[11]. In this study patients were examined in the prone position without any rectal enema, which again made evaluation of the defecation process itself impossible. The pubosacral line reaching from the most inferior part of the pubic symphysis to the lower part of the sacrum was chosen as the reference line for MRI. Measurements were performed at rest and during straining for both imaging techniques.
The pubosacral line was also used by Goodrich et al[6]
who examined five female patients with descending perineum syndrome pre- and postoperatively, as well as 10 asymptomatic female volunteers undergoing MRI. In contrast to the previous study, patients were placed in the supine position. The authors did not state whether the rectum, vagina or bladder had been contrasted.
Healy et al[8,14] have analyzed various aspects of pelvic floor disorders in patients and healthy volunteers comparing videoproctography with dynamic MRI with patients placed in the supine position. The defecation maneuver was not acquired. In contrast to other studies[9,10,12], instead of contrast medium, a tampon was placed in the rectum. Measurements were performed during maximal straining, while the lower PCGL was the reference landmark.
Tacke[15] has tested a new method of dynamic MRI with radial real-time imaging and a reduced image area for defecography. Patients in the supine position were asked to void a condom filled with a gadolinium-based contrast gel. The authors discussed this form of rectal filling critically as it may mask intussusception or latent incontinence.
Vanbeckevoort et al[12] compared colpocystoproctography (videoproctography with opacification of vagina and bladder) and dynamic MRI in supine position. For MRI, the rectum was filled with 100 mL of ultrasound gel, which was not meant to be voided. Measurements were taken during maximal straining using the PCGL as a reference line.
Lienemann et al[10] compared colpocystoproctography and MRI for the diagnosis of enteroceles. In MRI, patients and healthy volunteers were placed in a supine position and the rectum was filled with 200 mL of ultrasound gel, which was to be defecated during imaging. The study does not reveal data on frequency and completeness of the defecation process. Reference line was the PCGL.
In a technically equal approach, Lienemann et al[9] examined 44 female patients and five asymptomatic volunteers for descent of the pelvic floor. Patients and volunteers were asked not to void the gel. Additionally, there was no discussion of the findings of the healthy volunteers in this study.
Hilfiker et al[16] and Schoenberger et al[17] presented an “open system” MRI where patients were examined in an upright position analogous to videoproctography. In the study of Schoenberger et al[17], the findings of 15 patients examined with videoproctography and this new form of open configured MRI were compared. Five healthy volunteers were included for the definition of normal values, which are not mentioned in the paper.
This overview illustrates the problems arising with the introduction of a new examination technique. So far, dynamic MRI is widely accepted as a promising technique for the diagnosis of the pelvic floor[3,4,6,9,10], particularly for functional aspects of pelvic floor disorders because pelvic organs and muscles can be visualized and evaluated without invasive opacification and without any exposure to ionizing radiation. Still, without standardization of patient preparation, examination technique and evaluation of the data according to standardized reference lines and landmarks, it may not yet replace a well-established technique like videoproctography or colpocystoproctography. We believe that a comparison of both procedures has to take place under standardized conditions. In this context, the documentation of the defecation process is most important for subsequent comparability of both techniques. Vanbeckevoort et al[12] have compared the results of 35 patients examined with colpocystoproctography with and without defecation with the findings of dynamic MRI without defecation. For colpocystoproctography the urinary bladder and the small bowel were filled with a contrast medium. In their analysis, colpocystoproctography including defecation was by far superior to the same technique without defecation. Based on the observation that the pelvic floor reached its maximum downward movement only during defecation, and supported by the fact that patients are placed in a horizontal position in MRI, the authors concluded that colpocystoproctography including defecation may also be superior to MRI without defecation. Our results also revealed that the formation of a rectocele and a cystocele as well as the maximal descent of the anorectal junction and of the uterocervical junction was only completely visible at the end of the defecation process but not during straining alone.
In our study, the rectum was filled with 180 mL ultrasound gel with a gadolinium-based contrast medium. Ikenberry et al[18] have shown that varying viscosity of the contrast medium does not significantly influence the findings in videoproctography. Due to the natural differences in signal intensity of the urinary bladder, vagina, small bowel, and peritoneum, the procedure can be kept on a low level of invasivity.
The lower PCGL was used to visualize relative movements of the pelvic organs during defecation. The anorectal junction and anorectal angle were determined by the central axis of the anal canal and a line along the posterior wall of the rectum[8-10,14]. According to Delemarre et al[11], rectoceles were measured from the anorectal junction to the tip of protrusion of the anterior rectum wall. Table 2 gives an overview of different techniques and normal values as described in other studies.
| Yang et al[3] | Kruyt et al[4] | Vanbeckevoort et al [12] | Lienemann et al [9] | Healy et al [14] | Paetzel et al l[19] | |
| Defecation | No | No | No | No | No | Yes |
| Rectal contrast | No | No | 100 mL μg | 200 mL μg | Plastic tube | 180 mL μg |
| Bladder contrast | No | No | No | 60 mL sodium chloride | No | No |
| Vaginal contrast | No | No | No | 50 mL ug | Plastic tube | No |
| Position | Horizontal | Prone | Horizontal | Horizontal | Horizontal | Horizontal |
| Reference line | Pubococcygeal | Symphysiosacral | Pubosacral | Pubococcygeal | Pubococcygeal | Pubococcygeal |
| Anorectal junction | -25 mm | -30 mm to -40 mm | -25 mm | No information | -20 mm | -5.3 mm |
| Bladder base | -10 mm | No information | ± 0 | ± 0 | -10 mm | +23 mm |
| Uterocervical junction | +10 mm | No information | No information | +0 | ± 0 | +43 mm |
| Rectocele (evaluation) | No information | No information | Yoshioka | Yoshioka | Yoshioka | Delemarre |
| Rectocele (size) | No information | no information | < 30 mm | < 30 mm | no information | 26 mm |
| Anorectal angle | No information | < 130° (rest) | No information | No information | No information | 93 ° (rest) |
The position of the anorectal junction related to the PCGL at rest and during defecation is one parameter in the evaluation of a descent of the perineum. In the present study the anorectal junction moved 30 mm on average below the PCGL during defecation. In a previously published study by our group we evaluated symptomatic patients with pelvic floor disorders. In this study the anorectal junction moved an average 49 mm below the PCGL, while in patients suffering from a rectal incontinence, an average of 51 mm was measured[19].
Besides the descent of the anorectal junction, changes in the anorectal angle are commonly used as an indicator for the functional status of the pelvic floor[20]. A narrowing of the anorectal angle may indicate a disorder of the puborectal muscle[21]. This may lead to constipation with subsequent straining leading to rectal intussusception, rectocele and mucosal prolapse with a solitary ulcer of the rectum[21,22]. If the anorectal angle is already widened at rest, this may be a sign of weakness of the pelvic floor and is commonly observed along with incontinence and rectal prolapse[23-25].
Standard values in the literature vary enormously. For videoproctography, Hardcastle et al[26] described normal values between 60° and 105° at rest whereas the findings of other groups[27-29] relying on control groups with up to 150 volunteers show values between 90° and 104° at rest and 103° to 137° during defecation. Comparing the data with our study we determined an average angle of 93° at rest and 108.7° during straining. Accordingly, the use of changes in the anorectal angle as a diagnostic parameter is difficult not only because data vary significantly but also because findings of patients and asymptomatic volunteers tend to overlap[4,7,8,15,29].
A patient with an anterior rectocele often presents with symptoms of incomplete defecation and are observed along with a descent of the pelvic floor[13]. However, a rectocele can also frequently be found in asymptomatic patients. Therefore, some authors assume that symptoms depend on the diameter of the rectocele[9,29,30]. Due to different approaches in the attempt to measure the expansion of a rectocele, there are still no well-defined normal values available. Delemarre et al[11] used videoproctography and MRI to examine 38 patients in the prone position without rectal filling and without defecation. He concluded that videoproctography was superior to MRI for diagnosis of rectoceles. He postulated that a symptomatic rectocele > 20 mm needs to be operated. Lienemann et al[9] and Yoshioka et al[13] have defined a rectocele as a protrusion of the anterior rectal wall of > 30 mm. Yoshioka et al[13] have found that, in comparison to clinical examination, MRI was superior to colpocystoproctography in detecting rectoceles. Patients were examined in the supine position with rectal filling during maximal straining but without defecation. In contrast, Healy et al[8] rated videoproctography as superior to MRI. They have defined expansion of > 20 mm as pathological. In addition, they found that a rectocele < 13 mm measured by videoproctography was totally missed by MRI. MRI examination again was performed without defecation.
According to our observations the horizontal position does not seem to be a disadvantage because a rectocele was found in eight out of 10 healthy volunteers, with an average size of 26 mm (Table 1). This was in accordance with data from the literature[9,14] where the incidence of a rectocele in asymptomatic volunteers was about 80%, although these are described as “small” rectoceles.
Incidence and degree of a cystocele and an uterocervical prolapse is usually connected with the number of vaginal deliveries, preceding hysterectomy, and chronic constipation with increased straining maneuvers[12]. Besides clinical examination, imaging techniques make quantification of findings possible[9].
Yang et al[3], Lienemann et al[9], Vanbeckevoort et al[12] and Healy et al[7,14] have used normal values for the descent of the bladder base and uterocervical junction during straining in relation to the PCGL. These values were raised partly in healthy volunteers and partly determined at random.
Due to the natural differences in signal intensity of the urinary bladder and the vagina there was no necessity to apply additional contrast media[16]. In our study, six of 10 asymptomatic healthy female volunteers without a history of previous delivery or surgery showed the finding of a cystocele, and in three of 10 a vaginal prolapse was diagnosed.
In the studies of Lienemann et al[31] and Sprenger et al[32], 20 and 39 healthy females were examined with dynamic MRI of the pelvic floor, including defecation. A cystocele or vaginal prolapse was detected in none of them.
An enterocele is defined as herniation of the peritoneum into the rectovaginal space[33], which may contain small bowel loops or sigmoid colon. They are often accompanied by severe defecation disorders and a sensation of pressure as well as downward movement of the pelvic floor[34]. The prevalence of an enterocele in women is between 18% and 37%[35,36]. They often occur after hysterectomy[33]. No enterocele was detected in any volunteer. Lienemann et al[10] examined 55 patients and 11 asymptomatic volunteers with colpocystoproctography and dynamic MRI, without application of contrast medium into the peritoneum or small bowel. MRI had a clear advantage because the peritoneum and contents of the enterocele were easily identified. He concluded that MRI may replace colpocystoproctography in the diagnosis of an enterocele.
It proved to be useful to acquire the complete defecation process after a rectal enema to detect potential pathological changes. A rectocele can easily be identified by dynamic MRI. The pelvic organs of healthy volunteers show a relatively high mobility, therefore, presently suggested normal values for the position of pelvic organs in relation to the PCGL have to be redefined. It is necessary to evaluate normal values under standardized investigation conditions and with large groups of healthy volunteers.
Pelvic floor disorders are commonly seen in proctology, urology and gynecology, and the patients are mostly women. Due to the complexity of anatomy and physiology of the pelvic floor we have come across a wide variety of disorders.
Imaging plays an important role in the diagnostic workup of complex combined pelvic floor disorders. Functional examinations like videoproctography provide valuable information but it has been increasingly replaced by dynamic magnetic resonance imaging (MRI) that can visualize intra- and extraluminal structures. Further strengths of MRI are excellent soft tissue contrast and the lack of ionizing radiation.
There have been several studies about the diagnostic value of dynamic MRI of the pelvic floor; most of them with symptomatic patients. This investigation is believed to be the first to document the range of normal findings in MR defecography of asymptomatic female volunteers.
The wide range of normal findings in this study emphasizes the necessity of obtaining common standards for the evaluation of MR defecography.
MR defecography is a dynamic pelvic MRI examination. After optional application of a rectal enema, defecation is visualized by a series of fast MRI sequences.
This is a good study but the authors should make some revision.
Peer reviewers: Edward J Ciaccio, PhD, Research Scientist, Department of Medicine, HP 804, Columbia University, 180 Fort Washington Avenue, New York, NY 10032, United States; Nageshwar D Reddy, Professor, Asian Institute of Gastroenterology, 6-3-652, Somajiguda, Hyderabad 500082, India
S- Editor Cheng JX L- Editor Kerr C E- Editor Xiong L
| 1. | Winkler R. Proktologie- Ein Leitfaden für die Praxis. Stuttgart-New York: Georg Thieme Verlag 1997; . |
| 2. | Herold A, Müller-Lobeck H, Jost WH, Duschka L, Leder D. [Diagnostic evaluation of the rectum and pelvic floor in chronic constipation]. Zentralbl Chir. 1999;124:784-795. [PubMed] |
| 3. | Yang A, Mostwin JL, Rosenshein NB, Zerhouni EA. Pelvic floor descent in women: dynamic evaluation with fast MR imaging and cinematic display. Radiology. 1991;179:25-33. [PubMed] |
| 4. | Kruyt RH, Delemarre JB, Doornbos J, Vogel HJ. Normal anorectum: dynamic MR imaging anatomy. Radiology. 1991;179:159-163. [PubMed] |
| 5. | Wallden L. Defecation block in cases of deep rectogenital pouch. Acta Chir Scand. 1952;103:236-238. [PubMed] |
| 6. | Goodrich MA, Webb MJ, King BF, Bampton AE, Campeau NG, Riederer SJ. Magnetic resonance imaging of pelvic floor relaxation: dynamic analysis and evaluation of patients before and after surgical repair. Obstet Gynecol. 1993;82:883-891. [PubMed] |
| 7. | Healy JC, Halligan S, Reznek RH, Watson S, Bartram CI, Kamm MA, Phillips RK, Armstrong P. Magnetic resonance imaging of the pelvic floor in patients with obstructed defaecation. Br J Surg. 1997;84:1555-1558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 8. | Healy JC, Halligan S, Reznek RH, Watson S, Bartram CI, Phillips R, Armstrong P. Dynamic MR imaging compared with evacuation proctography when evaluating anorectal configuration and pelvic floor movement. AJR Am J Roentgenol. 1997;169:775-779. [PubMed] |
| 9. | Lienemann A, Anthuber C, Baron A, Kohz P, Reiser M. Dynamic MR colpocystorectography assessing pelvic-floor descent. Eur Radiol. 1997;7:1309-1317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 163] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 10. | Lienemann A, Anthuber C, Baron A, Reiser M. Diagnosing enteroceles using dynamic magnetic resonance imaging. Dis Colon Rectum. 2000;43:205-12; discussion 212-3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 80] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 11. | Delemarre JB, Kruyt RH, Doornbos J, Buyze-Westerweel M, Trimbos JB, Hermans J, Gooszen HG. Anterior rectocele: assessment with radiographic defecography, dynamic magnetic resonance imaging, and physical examination. Dis Colon Rectum. 1994;37:249-259. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 53] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 12. | Vanbeckevoort D, Van Hoe L, Oyen R, Ponette E, De Ridder D, Deprest J. Pelvic floor descent in females: comparative study of colpocystodefecography and dynamic fast MR imaging. J Magn Reson Imaging. 1999;9:373-377. |
| 13. | Yoshioka K, Matsui Y, Yamada O, Sakaguchi M, Takada H, Hioki K, Yamamoto M, Kitada M, Sawaragi I. Physiologic and anatomic assessment of patients with rectocele. Dis Colon Rectum. 1991;34:704-708. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 70] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 14. | Healy JC, Halligan S, Reznek RH, Watson S, Phillips RK, Armstrong P. Patterns of prolapse in women with symptoms of pelvic floor weakness: assessment with MR imaging. Radiology. 1997;203:77-81. [PubMed] |
| 15. | Tacke J, Nolte-Ernsting C, Glowinski A, Schäffter T, Adam G, Günther RW. [MR defecography at 1.5 Tesla with radial real-time imaging and reduced image field]. Rofo. 1999;171:187-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 16. | Hilfiker PR, Debatin JF, Schwizer W, Schoenenberger AW, Fried M, Marincek B. MR defecography: depiction of anorectal anatomy and pathology. J Comput Assist Tomogr. 1998;22:749-755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | Schoenenberger AW, Debatin JF, Guldenschuh I, Hany TF, Steiner P, Krestin GP. Dynamic MR defecography with a superconducting, open-configuration MR system. Radiology. 1998;206:641-646. [PubMed] |
| 18. | Ikenberry S, Lappas JC, Hana MP, Rex DK. Defecography in healthy subjects: comparison of three contrast media. Radiology. 1996;201:233-238. [PubMed] |
| 19. | Paetzel C, Strotzer M, Fürst A, Rentsch M, Lenhart M, Feuerbach S. [Dynamic MR defecography for diagnosis of combined functional disorders of the pelvic floor in proctology]. Rofo. 2001;173:410-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 10] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 20. | Yang XM, Partanen K, Farin P, Soimakallio S. Defecography. Acta Radiol. 1995;36:460-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (1)] |
| 21. | Johansson C, Ihre T, Holmström B, Nordström E, Dolk A, Brodén G. A combined electromyographic and cineradiologic investigation in patients with defecation disorders. Dis Colon Rectum. 1990;33:1009-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 22. | Goei R, Baeten C, Janevski B, van Engelshoven J. The solitary rectal ulcer syndrome: diagnosis with defecography. AJR Am J Roentgenol. 1987;149:933-936. [PubMed] |
| 23. | Goei R. Anorectal function in patients with defecation disorders and asymptomatic subjects: evaluation with defecography. Radiology. 1990;174:121-123. [PubMed] |
| 24. | Hardcastle JD. The descending perineum syndrome. Practitioner. 1969;203:612-619. [PubMed] |
| 25. | Porter NH. A physiological study of the pelvic floor in rectal prolapse. Ann R Coll Surg Engl. 1962;31:379-404. [PubMed] |
| 26. | Hardcastle JD, Parks AG. A study of anal incontinence and some principles of surgical treatment. Proc R Soc Med. 1970;63 Suppl:116-118. [PubMed] |
| 27. | Mahieu P, Pringot J, Bodart P. Defecography: I. Description of a new procedure and results in normal patients. Gastrointest Radiol. 1984;9:247-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 301] [Cited by in RCA: 218] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 28. | Piloni V, Fioravanti P, Spazzafumo L, Rossi B. Measurement of the anorectal angle by defecography for the diagnosis of fecal incontinence. Int J Colorectal Dis. 1999;14:131-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 29. | Shorvon PJ, McHugh S, Diamant NE, Somers S, Stevenson GW. Defecography in normal volunteers: results and implications. Gut. 1989;30:1737-1749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 575] [Cited by in RCA: 450] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 30. | Kelvin FM, Maglinte DD, Benson JT. Evacuation proctography (defecography): an aid to the investigation of pelvic floor disorders. Obstet Gynecol. 1994;83:307-314. [PubMed] |
| 31. | Lienemann A, Sprenger D, Janssen U, Anthuber C, Reiser M. [Functional MRI of the pelvic floor. The methods and reference values]. Radiologe. 2000;40:458-464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 32. | Sprenger D, Lienemann A, Anthuber C, Reiser M. [Functional MRI of the pelvic floor: its normal anatomy and pathological findings]. Radiologe. 2000;40:451-457. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 33. | Holley RL. Enterocele: a review. Obstet Gynecol Surv. 1994;49:284-293. [PubMed] |
| 34. | Shull BL. Clinical evaluation of women with pelvic support defects. Clin Obstet Gynecol. 1993;36:939-951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 56] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 35. | Maglinte DD, Kelvin FM, Hale DS, Benson JT. Dynamic cystoproctography: a unifying diagnostic approach to pelvic floor and anorectal dysfunction. AJR Am J Roentgenol. 1997;169:759-767. [PubMed] |
| 36. | Mellgren A, Johansson C, Dolk A, Anzén B, Bremmer S, Nilsson BY, Holmström B. Enterocele demonstrated by defaecography is associated with other pelvic floor disorders. Int J Colorectal Dis. 1994;9:121-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 68] [Article Influence: 2.2] [Reference Citation Analysis (0)] |