Published online Sep 28, 2012. doi: 10.3748/wjg.v18.i36.4985
Revised: April 12, 2012
Accepted: April 20, 2012
Published online: September 28, 2012
Cholestasis results in a buildup of bile acids in serum and in hepatocytes. Early studies into the mechanisms of cholestatic liver injury strongly implicated bile acid-induced apoptosis as the major cause of hepatocellular injury. Recent work has focused both on the role of bile acids in cell signaling as well as the role of sterile inflammation in the pathophysiology. Advances in modern analytical methodology have allowed for more accurate measuring of bile acid concentrations in serum, liver, and bile to very low levels of detection. Interestingly, toxic bile acid levels are seemingly far lower than previously hypothesized. The initial hypothesis has been based largely upon the exposure of μmol/L concentrations of toxic bile acids and bile salts to primary hepatocytes in cell culture, the possibility that in vivo bile acid concentrations may be far lower than the observed in vitro toxicity has far reaching implications in the mechanism of injury. This review will focus on both how different bile acids and different bile acid concentrations can affect hepatocytes during cholestasis, and additionally provide insight into how these data support recent hypotheses that cholestatic liver injury may not occur through direct bile acid-induced apoptosis, but may involve largely inflammatory cell-mediated liver cell necrosis.
- Citation: Woolbright BL, Jaeschke H. Novel insight into mechanisms of cholestatic liver injury. World J Gastroenterol 2012; 18(36): 4985-4993
- URL: https://www.wjgnet.com/1007-9327/full/v18/i36/4985.htm
- DOI: https://dx.doi.org/10.3748/wjg.v18.i36.4985
A reduction in bile flow (cholestasis) can result from gall stones, impingement from a local tumor, intrahepatic cholestasis of pregnancy, genetic deficiency in bile export proteins or in autoimmune disorders amongst other etiologies[1,2]. Cholestasis results in a dramatic increase in liver and serum bile acid levels that eventually lead to acute liver toxicity, proliferation of bile ducts, and fibrosis progressing to cirrhosis[3]. Neonatal disorders such as progressive familial intrahepatic cholestasis and biliary atresia are particularly difficult to treat effectively[4]. While surgical means can prevent disease progression in the case of gall stones or biliary atresia, there are a limited number of treatment options pharmacologically for diseases such as primary sclerosing cholangitis or primary biliary cirrhosis[5]. Ursodiol, a pharmaceutical form of ursodeoxycholic acid, is the current mainline defense against cholestasis, although its efficacy is limited in some circumstances[5,6].
While many of the etiologies of disease initiation are well described, the molecular mechanisms behind the early liver injury associated with cholestasis are extensively studied, but not well understood. A major hypothesis was based on the assumption that the buildup of bile acids in hepatocytes and blood results in hepatocellular apoptosis dependent upon bile acid concentrations[6,7]. While these studies on the direct toxicity of bile acids have been supported by many in vitro studies from multiple groups, recent methodology measuring the concentrations of individual bile acids after the onset of cholestasis in vivo has cast some doubt that bile acid levels may actually reach the levels necessary for cell toxicity[8-10]. A growing volume of work has instead focused on how sterile inflammation and innate immunity may result in the initial injury[11-15]. This review will discuss current and developing paradigms in the pathophysiology of early cholestatic liver injury, and provide insight into how recent advances in methodology may affect these theories.
High concentrations of bile acids given to cultured hepatocytes in vitro[16] and bile acid-induced cholestasis[17], or bile duct ligation in vivo[18], have long been established as models of toxicity to hepatocytes. After the discovery that ursodeoxycholic acid could be used pharmacologically as a choleretic agent with protective effects against cholestasis[19], a renewed interest in the subject emerged. While previous papers had focused on the detergent properties of amphipathic bile acids[20,21] recent research resulted in a series of papers illustrating a detailed mechanism of how toxic bile acids or the respective bile salt, could induce apoptosis in primary human and rat hepatocytes[22-27]. Much of this work has spearheaded the current revival of direct bile acid toxicity as a model of cholestasis, or liver injury, in vitro and in vivo.
The most commonly studied bile acid for its toxicity to hepatocytes is glycochenodeoxycholic acid (GCDCA), or its bile salt glycochenodeoxycholate, due to it being a major constituent of human serum and bile after cholestasis[28] and its relatively higher level of toxicity when compared to other bile acids[29]. Galle et al[22] first established GCDCA as toxic to primary human hepatocytes and in the same paper also established ursodeoxycholate (UDCA) as protective against the GCDCA-induced injury. Due to the limited availability of primary human hepatocytes, much of the subsequent research into mechanism of injury occurred in rodent, especially rat hepatocytes. GCDCA-induced hepatocellular death was first established as apoptotic using electron microscopy[23]. Incubation of primary rat hepatocytes with 50 μmol/L GCDCA induced DNA fragmentation, chromatin margination and condensation, cellular shrinkage and blebbing of the membrane with intact organelles enclosed, all of these classical hallmark characteristics of apoptosis[23]. While it has been extensively shown that DNA fragmentation is common to both necrosis and apoptosis[30-33], the morphological changes present after GCDCA exposure provided strong evidence of bile acid-induced apoptosis of primary hepatocytes. In addition, caspase activation is a feature of GCDCA-induced apoptosis[27,34-36] and caspase inhibitors were shown to protect[27,34,36].
Investigation into intracellular mechanisms of bile acid toxicity showed cellular apoptosis was associated with mitochondrial instability and mitochondrial membrane permeability transition pore (MPTP) formation that was preventable by either UDCA, ketohexoses such as fructose, or cyclosporine A and trifluoroperazine[36-39]. As mitochondria are essential for ATP production and GCDCA depleted ATP levels[29], the idea that UDCA, or other protective interventions, were sustaining ATP levels was tested; however, protection was not due to reduced ATP depletion, but by blockage of the MPTP[37]. This suggested that UDCA may prevent injury during cholestasis not only by enhancing bile flow, but by preventing initiation of the MPTP[37], a facet which has been confirmed in other models[40-42]. Mitochondrial reactive oxygen species (ROS) production and MPTP initiation was later also shown in human hepatocytes exposed to toxic bile acids such as GCDCA indicating the mitochondrial dysfunctions occur in more than just rodent species[43]. Adult rats fed a diet supplemented with deoxycholic acid (DCA) for 10 d also showed significant mitochondrial swelling, peroxynitrite formation and ROS production[44]. These data suggest that exposure of cultured hepatocytes to high levels of hydrophobic bile acids causes formation of ROS, reactive nitrogen species, and initiation of the MPTP, which is directly involved in the injury. In immortalized cell lines transfected with the basolateral transporter NTCP, bile acids like GCDCA could directly stimulate both Fas-dependent apoptosis via activation of Fas receptor[26] and Fas-independent apoptosis via previously established mitochondrial damaging mechanisms[41,42]. These experiments indicate that uptake of bile acids can cause both intrinsic and extrinsic apoptosis, and that cell death could be ameliorated by protecting against MPTP formation. These observations were critical in establishing the role of intracellular signaling and mitochondrial stress in the pathophysiology of bile acid-induced toxicity, as previous hypotheses revolved largely around the idea of inherent bile acid toxicity.
Although GCDCA and its bile salt have been extensively studied, other toxic bile acids have also effects on intracellular signaling mechanisms. Taurolithocholic acid (TLCA), taurochenodeoxycholic acid, and DCA increase intracellular ROS species in hepatocytes[45,46] and toxicity from these bile acids is responsive to antioxidants such as N-acetylcysteine and α-tocopherol[45]. DCA has also been shown to induce apoptosis and mitochondrial instability in adult rats and primary hepatocytes at high concentrations[44,47,48]. Interestingly the taurine conjugate of UDCA, TUDCA, is protective against bile acid-induced apoptosis through activation of mitogen-activated protein kinase survival pathway signaling including p38 and extracellular signal-regulated kinase[35]. While GCDCA may be one of the most toxic and perhaps the most interesting bile acid scientifically and mechanistically, the total bile acid milieu probably contributes to the overall effect more than a single bile acid, with anti-apoptotic bile acids such as TUDCA and UDCA counteracting some of the effects of pro-apoptotic bile acids such as GCDCA.
Bile acids are also known to target intracellular nuclear receptors, especially the liver X receptor (LXR) family such as farnesoid X receptor (FXR), pregnane X receptor (PXR), constitutive androstane receptor (CAR) and more[49]. The first LXRs identified as bile acid receptors were FXR[50,51], LXR[52], and later PXR[53]. FXR has been established as a key regulator in bile acid synthesis and transport[54]. Agonism of FXR with the synthetic drug GW4064 is hepatoprotective against both intra- and extrahepatic cholestasis[55] and agonism of PXR is protective against certain bile acid-induced cholestatic syndromes[56]. Nevertheless, the FXR knockout mouse were also protected against bile duct ligation-induced injury[57,58], which was found to be via increases in phase I detoxifying reactions and renal excretion of bile acids[59]. This suggests that both agonism and antagonism may be beneficial, depending on the type of cholestasis and the individual response to alterations in master regulators such as FXR. There is evidence for reduced bile acid load by either a reduction in synthesis in the case of agonism or enhanced excretion in antagonism of FXR. In cases of agonism a reduced bile acid load also correlated with a reduction in the hepatic inflammatory response in both steatotic and non-steatotic models[55,60] and loss of FXR has also protected against inflammatory injury in other organs[61]. These data indicate that increasing bile acid levels may have a strong signaling effect through FXR, PXR and more in hepatocytes that results in decreased bile acid synthesis, increased bile acid excretion and recruitment of inflammatory cells to the area. Early growth response factor-1 (Egr-1) has been clearly established as a master regulator in the sterile inflammatory response that hepatocytes express after exposure to high levels of bile acids[62]. Future research investigating interplay between FXR and Egr-1 in this signaling axis may be fruitful.
While it seems undeniable that bile acids have the capacity to elicit multiple different signaling cascades ranging from mitochondrial dysfunction and cell death to bile acid synthesis and glucose homeostasis[49,63], there remains some debate over the models used to dissect these actions and their physiological relevancy. In particular, there appears to be a discrepancy between the concentration of bile acids used in vitro to cause toxicity, and the levels of bile acids occurring during cholestasis both in rodents and in man[8,9].
Table 1 illustrates the increases in serum bile acids found in sex and age matched patients after multiple forms of cholestasis in man[9]. Full details on the patients are provided by Trottier et al[9] and Lai et al[64]. In brief, bile acid concentrations were measured in human serum from healthy volunteers and from patients with extrahepatic cholestasis[9]. Seventeen patients (8 men and 9 women) were recruited to the study. Clinical diagnoses of the patients included 8 common bile duct stones, 5 pancreatic tumors, 1 bile duct tumor, 1 benign common bile duct stenosis and 1 chronic pancreatitis. Age and sex matched (20 male, 20 female) non-cholestatic volunteers were used as controls[9], selected from the Genetics of Lipid Lowering Drugs and Diet Network study[64].
| BA | Healthy volunteers (nmol/L) | Cholestatic patients (nmol/L) |
| CDCA | 257 ± 56 | 84 ± 60 |
| TCDCA | 120 ± 22 | 16 578 ± 4370 |
| GCDCA | 776 ± 112 | 28 535 ± 5734 |
| CA | 182 ± 83 | 173 ± 137 |
| TCA | 180 ± 47 | 54 487 ± 13 419 |
| GCA | 233 ± 56 | 56 222 ± 15 714 |
| UDCA | 138 ± 25 | 20 ± 11 |
| TUDCA | 5 ± 1 | 75 ± 22 |
| LCA | 13 ± 2 | 2 ± 1 |
| TLCA | 23 ± 4 | 12 ± 2 |
| GLCA | 16 ± 4 | 11 ± 2 |
| LCA-S | 7 ± 1 | 4 ± 2 |
| DCA | 387 ± 66 | 21 ± 16 |
| TDCA | 45 ± 12 | 213 ± 71 |
| GDCA | 246 ± 43 | 418 ± 91 |
Previous work suggests serum bile acid levels are highly similar to total liver levels when comparing bile acid concentrations in vivo[8]. The bile acids that show the greatest increase in concentration in the serum are not toxic bile acids, but largely non-toxic bile acids such as TCA and GCA in man[9] or TCA, α- and β-muricholic acid (MCA) and TMCA in mice[8]. Levels of the primary toxic bile acid in humans, GCDCA, rise substantially, to almost 30 μmol/L under severe cholestatic conditions as measured by serum levels in man[9]. However, studies with primary human hepatocytes have shown little to no increase in toxicity until levels greater than 100 μmol/L for 24 h with significant toxicity beginning around 500 μmol/L[22,65]. The only paper to demonstrate toxicity at a level close to 30 μmol/L reported that GCDCA showed morphological characteristics in primary human hepatocytes that resembled apoptosis and a 60% increase in DNA fragmentation at 50 μmol/L[66]. However, a significant increase in serum aspartate aminotransferase activities over control levels was only observed at 100 μmol/L GCDCA[66]. These data suggest that direct bile acid toxicity may not be the main mechanism of cholestatic liver injury in humans and seem to be supported by findings in mouse models of obstructive cholestasis[8,15]. Table 2 shows plasma bile acid concentrations in sham-operated controls or after 6 h of bile duct ligation (BDL)[8]. This is a time point corresponding with either maximal or near maximal increases in bile acid concentrations. Data are expressed as mean ± SE of n = 6 per group. For toxic bile acids such as GCDCA or LCA, the levels required to initiate toxicity in murine hepatocyte cultures are up to 3000-fold higher than the corresponding bile acid levels in vivo[8]. In the case of GCDCA, the most commonly used bile acid for in vitro toxicity, in vivo levels never exceed about 24 nmol/L throughout the first 3 d of injury[8]. This compares to 50 μmol/L GCDCA that is required to cause toxicity in cultured rodent hepatocytes in vitro[23,26]. In addition, during cholestasis in vivo, hepatocytes are exposed to a mixture of bile acids, some of which have actually an anti-apoptotic effect by causing the upregulation of inhibitor of apoptosis proteins, which may serve to counterbalance the apoptotic effect of specific bile acids[67,68].
| BA | Sham control (μmol/L) | BDL 6 h (μmol/L) |
| TMCA | 0.229 ± 0.029 | 1.003 ± 133 |
| TCA | 0.122 ± 0.027 | 1.072 ± 137 |
| TCDCA | 0.029 ± 0.003 | 7 ± 0.557 |
| GCA | 0.007 ± 0.0002 | 1 ± 0.118 |
| GCDCA | 0.003 ± 0.003 | 0 ± 0 |
| αMCA | 0.697 ± 0.273 | 87 ± 41 |
| βMCA | 0.175 ± 0.23 | 253 ± 100 |
| CA | 0.219 ± 0.059 | 17 ± 10 |
| CDCA | 0.0001 ± 0.0001 | 0.144 ± 0.085 |
| TUDCA | 0.031 ± 0.002 | 16 ± 14 |
| TDCA | 0.016 ± 0.004 | 0.330 ± 0.107 |
| TLCA | 0.007 ± 0.002 | 0.027 ± 0.002 |
| UDCA | 0.131 ± 0.002 | 0.492 ± 0.215 |
| DCA | 0.042 ± 0.018 | 0.109 ± 0.032 |
| LCA | 0.054 ± 0.013 | 0.033 ± 0.022 |
While it is known that cultured hepatocytes lose expression of bile acid transporters such as NTCP and the OATPs over time in culture, cells freshly taken from the liver express nearly the same amount of transporter as whole liver tissue[69], and nearly the same level at 3 h after plating[70], when many of these experiments begin[23,71,72]. Even given a 30% decrease in mRNA levels of transporters such as NTCP 3 h after plating, cells maintain over 50% of their NTCP function up to 24 h after plating as measured by kinetic parameters[70]. Thus, it stands to reason that these hepatocytes are concentrating bile acids in vitro at levels within a 2 fold difference of the in vivo physiology, yet it takes multiple orders of magnitude, up to a 3000 fold difference in the case of GCDCA, to account for the difference between bile acid levels used on rodent hepatocytes in vitro and levels observed in murine samples in vivo. Still, even given that serum or liver levels may underestimate the true concentrations of bile acids hepatocytes are exposed to after BDL when bile leaks back into the parenchyma[73,74], the principle bile acid composition in bile is similar to serum, i.e. the non-toxic bile acids predominate, thus it is unlikely that cells are exposed to micromolar concentrations of the toxic bile acids in vivo[75].
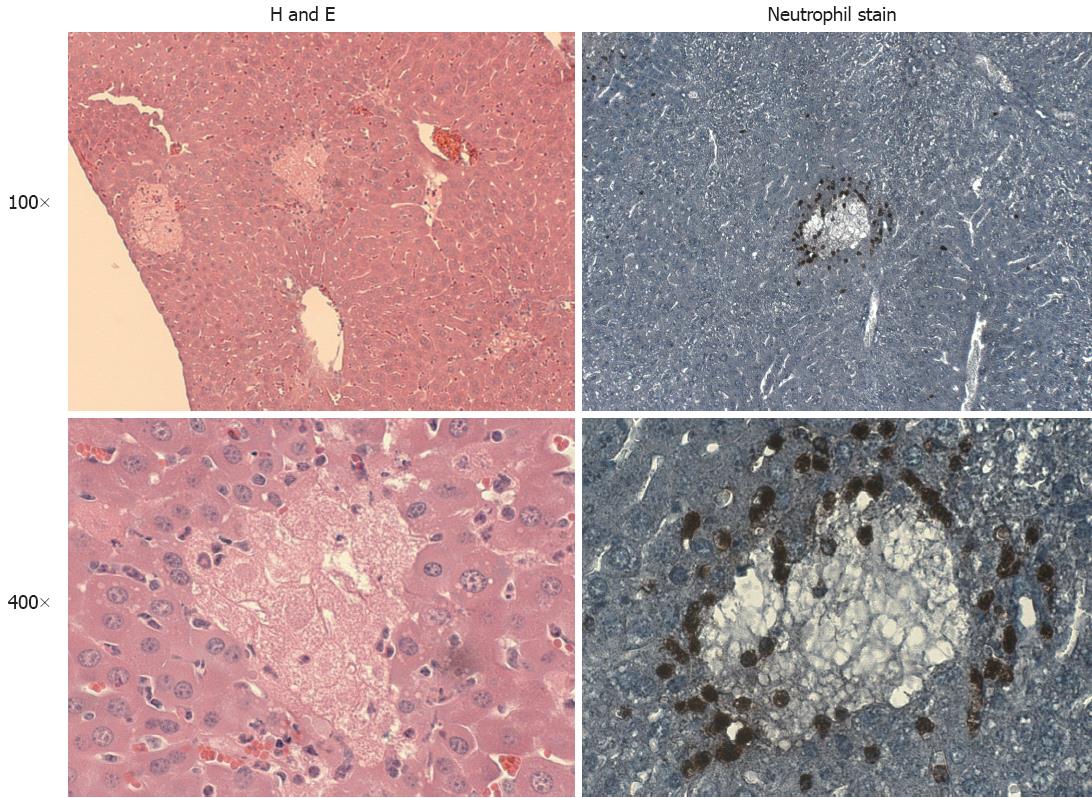
If bile acids are not directly responsible for cholestatic liver injury in vivo, then what is the mechanism of injury, and what role do bile acids play in the pathophysiology and progression of the injury? Accumulation of neutrophils, largely around sites of cell injury, within 24 h after bile duct ligation has been reported numerous times[11,12,76,77]. In addition, an increase in macrophage accumulation is observed approximately one week post ligation[77,78]. Figure 1 shows typical histology and neutrophil extravasation after bile duct ligation in mice. H&E staining shows multiple areas of hepatic focal necrosis throughout the liver. Neutrophils are present around the site of injury as early as six hours post BDL, and peak around 48-72 h after BDL, which is also the peak of liver injury[8,11,77].
Neutrophil accumulation has been directly associated with the injury, as deficiency of adhesion molecules such as ICAM-1 or CD18 reduced the number of neutrophils that extravasate into liver parenchyma, and drastically reduced liver injury[11,12]. A neutrophil is a particularly toxic leukocyte due to its capacity to produce superoxide via nicotinamide adenine dinucleotide phosphate (NADPH) oxidase and the potent oxidant hypochlorous acid via myeloperoxidase[79]. Neutrophil accumulation after bile duct ligation has been associated with an increase in myeloperoxidase activity[80] and intracellular chlorotyrosine adduct formation in hepatocytes[11,12,81]. This indicates that neutrophil-derived hypochlorous acid can cause an intracellular oxidant stress in hepatocytes, which is responsible for cell injury[82,83]. Inhibition of NADPH oxidase reduces neutrophil-mediated oxidant stress and protects against neutrophil cytotoxicity[84,85]. In addition, hepatic NADPH oxidases located in hepatic stellate cells (Nox1) and Kupffer cells (Nox2) contribute to BDL-induced fibrosis[86,87] but their contribution to the initial liver injury (bile infarcts) has not been determined.
Together these data strongly implicate neutrophils in the pathogenesis of early cholestatic liver injury. In support of this hypothesis is recent data that exposure of cultured hepatocytes to both toxic and non-toxic bile acids at levels that recapitulate the in vivo concentrations result in dramatic increases in chemotactic factors such as MIP-2 and mKC, and adhesion molecules such as ICAM-1, all factors that can be involved in hepatic neutrophil recruitment[15]. The inflammatory mediator induction is dependent on the activation of the transcription factor Egr-1 in bile duct-ligated mice and in bile acid-exposed hepatocytes[15,62]. Egr-1 expression is also increased in human patients with cholestatic liver disease and the hepatic Egr-1 levels correlate with interleukin-8 and ICAM-1 induction[15]. Therefore, we initially hypothesized that after BDL, bile leaks back into the parenchyma after rupture of bile ducts and hepatocytes are locally exposed to high concentrations of pro-inflammatory bile acids such as TCA, β-MC and TMC that initiate an inflammatory response[8,13,15]. Additionally these hepatocytes are exposed to low concentrations of cytotoxic bile acids such as DCA, TLCA, and GCDCA that may initiate a low level of mitochondrial dysfunction that stresses the hepatocytes, but is insufficient to directly kill the cells[8,29,37,38,41-43]. This bile acid-induced stress triggers the expression of ICAM-1, MIP-2, and mKC in hepatocytes[8,15]. The subsequent release of these CXC chemokines can activate neutrophils in circulation[88] and provide a chemotactic gradient for the extravasation of neutrophils, which then attack the stressed hepatocytes and cause cell death through reactive oxygen formation[11,12,81,82]. Given a dramatic enough increase in toxic bile acid concentrations by feeding e.g. a lithocholic acid-containing diet[89], it is possible to directly cause cell death via bile acid toxicity but independent of neutrophils (Woolbright and Jaeschke unpublished observation). However the in vivo bile acid concentrations in models such as BDL are insufficient to meet this threshold, and thus the neutrophil-mediated cell death is dominant.
A central but controversial issue in the debate on the mechanisms of cell death in cholestatic liver disease is whether the injury occurs through apoptosis or necrosis[90]. This is a key point as the therapeutic options to prevent injury will progress in two very different directions depending on the answer. Necrosis is characterized by cellular swelling, membrane blebbing, DNA fragmentation and release of cellular components, whereas apoptosis is characterized by cellular shrinking, caspase activation, DNA fragmentation, chromatin condensation with initially no release of cellular contents[32]. These processes lead to dramatically different outcomes after cell death in regards to progression of disease with necrosis leading to increased inflammation and further aggravation by inflammatory cells[81,90,91].
Although there is no question that high concentrations of certain hydrophobic bile acids can cause apoptotic cell death in cultured rodent hepatocytes[22-27,34-42,67,68] as discussed earlier, the pathophysiological relevance of these observations for in vivo models, e.g., BDL, is questioned due to the fact that the pro-apoptotic bile acids do not achieve sufficiently high levels in vivo to directly cause cell death[8,9]. Thus, the evaluation of the mechanism of cell injury has to be based on the in vivo evidence. Early studies concluded that BDL-induced liver injury was caused by apoptosis mainly due to the positive terminal deoxynucleotidyl transferase dUTP nick-end labeling (TUNEL) assay[92-94] which is clearly not specific for apoptotic cell death[30-32]. In addition, immunofluorescence was used to detect cells with active caspases 3/7[95] but this was not confirmed by others[96]. Additional arguments for apoptosis were the reduced BDL-induced liver injury in Fas receptor-deficient lpr mice[94] and after anti-sense treatment to reduce Bid expression[97]. However, lpr mice are protected due to the reduced inflammatory response[98] and genetically Bid-deficient mice did not show reduced liver injury[96]. In contrast, numerous studies did not find any morphological evidence of apoptosis[11,12,68,98-101] and no evidence of caspase activation (enzyme activity and/or active fragments) or immunohistochemical staining for cytokeratin 18 cleavage products or active caspase-3/7[96,98,99]. In addition, potent caspase inhibitors, which effectively eliminated caspase activation and cell death in Fas- or TNF-mediated apoptosis models[98,102], did not protect against BDL-induced liver injury[98]. The one study reporting reduced injury after a caspase inhibitor in the absence of relevant caspase activation[95] may be due to off-target effects on other proteases[103]. Likewise, the anti-sense knock-down of Bid gene expression may have caused compensatory protective gene activation, which may have attenuated BDL-induced liver injury[95]. Clearly, Bid-deficient mice were not protected against BDL-induced liver injury[96]. The cathepsin B knockout mouse also showed protection against apoptotic injury[92], although apoptosis was assessed entirely based on the TUNEL assay. While the reduction in injury was attributed to lack of cathepsin B, and thus a lack of apoptotic processing proteins, the paper also showed a significant reduction in the neutrophilic inflammatory response[92]. Thus, the reduced injury in cathepsin B-deficient mice may have been due to reduced inflammation rather than apoptotic cell death. Taken together, the preponderance of experimental evidence supports the conclusion that liver injury after BDL involves mainly necrotic cell death.
In conclusion, when using morphological criteria of apoptosis and caspase activation in combination with positive controls of apoptotic cell death in vivo, the overwhelming experimental evidence supports the conclusion that there is no relevant apoptotic cell death but almost exclusively necrosis after BDL. The main paradigm shift comes from the recognition that the well-known hepatotoxic bile acids never reach concentrations in the liver after BDL that would directly cause cell death. In contrast, the most common bile acids that reach close to mmol/L levels trigger inflammatory mediator formation, which initiates an inflammatory response and cell death caused by neutrophils through oxidant stress. Initial data from human patients with cholestatic liver disease appear to confirm several aspects of the mouse model. Thus, the focus of future research should be on evaluating the cell stress mechanisms in hepatocytes considering pathophysiologically relevant bile acid mixtures and on further characterizing the initiation of the inflammatory response after BDL. Understanding these early injury mechanisms may not only reveal therapeutic approaches to reduce liver injury but also the subsequent fibrosis.
Peer reviewers: Dr. Richard A Rippe, Department of Medicine, The University of North Carolina at Chapel Hill, Chapel Hill, NC 27599-7038, United States; Bruno Stieger, Professor, Department of Medicine, Division of Clinical Pharmacology and Toxicology, University Hospital, 8091 Zurich, Switzerland
S- Editor Lv S L- Editor A E- Editor Li JY
| 1. | Kim WR, Ludwig J, Lindor KD. Variant forms of cholestatic diseases involving small bile ducts in adults. Am J Gastroenterol. 2000;95:1130-1138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 2. | Poupon R, Chazouillères O, Poupon RE. Chronic cholestatic diseases. J Hepatol. 2000;32:129-140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 122] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 3. | Kisseleva T, Brenner DA. Anti-fibrogenic strategies and the regression of fibrosis. Best Pract Res Clin Gastroenterol. 2011;25:305-317. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 143] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 4. | Bessho K, Bezerra JA. Biliary atresia: will blocking inflammation tame the disease? Annu Rev Med. 2011;62:171-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 67] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 5. | Zein CO, Lindor KD. Latest and emerging therapies for primary biliary cirrhosis and primary sclerosing cholangitis. Curr Gastroenterol Rep. 2010;12:13-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 6. | Maillette de Buy Wenniger L, Beuers U. Bile salts and cholestasis. Dig Liver Dis. 2010;42:409-418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 7. | Perez MJ, Briz O. Bile-acid-induced cell injury and protection. World J Gastroenterol. 2009;15:1677-1689. [PubMed] |
| 8. | Zhang Y, Hong JY, Rockwell CE, Copple BL, Jaeschke H, Klaassen CD. Effect of bile duct ligation on bile acid composition in mouse serum and liver. Liver Int. 2012;32:58-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 156] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 9. | Trottier J, Białek A, Caron P, Straka RJ, Milkiewicz P, Barbier O. Profiling circulating and urinary bile acids in patients with biliary obstruction before and after biliary stenting. PLoS One. 2011;6:e22094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 83] [Cited by in RCA: 86] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 10. | Burkard I, von Eckardstein A, Rentsch KM. Differentiated quantification of human bile acids in serum by high-performance liquid chromatography-tandem mass spectrometry. J Chromatogr B Analyt Technol Biomed Life Sci. 2005;826:147-159. [PubMed] |
| 11. | Gujral JS, Farhood A, Bajt ML, Jaeschke H. Neutrophils aggravate acute liver injury during obstructive cholestasis in bile duct-ligated mice. Hepatology. 2003;38:355-363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 253] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 12. | Gujral JS, Liu J, Farhood A, Hinson JA, Jaeschke H. Functional importance of ICAM-1 in the mechanism of neutrophil-induced liver injury in bile duct-ligated mice. Am J Physiol Gastrointest Liver Physiol. 2004;286:G499-G507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 124] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 13. | Copple BL, Jaeschke H, Klaassen CD. Oxidative stress and the pathogenesis of cholestasis. Semin Liver Dis. 2010;30:195-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 160] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 14. | Dold S, Laschke MW, Lavasani S, Menger MD, Thorlacius H. Cholestatic liver damage is mediated by lymphocyte function antigen-1-dependent recruitment of leukocytes. Surgery. 2008;144:385-393. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Allen K, Jaeschke H, Copple BL. Bile acids induce inflammatory genes in hepatocytes: a novel mechanism of inflammation during obstructive cholestasis. Am J Pathol. 2011;178:175-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 401] [Article Influence: 26.7] [Reference Citation Analysis (0)] |
| 16. | Schölmerich J, Becher MS, Schmidt K, Schubert R, Kremer B, Feldhaus S, Gerok W. Influence of hydroxylation and conjugation of bile salts on their membrane-damaging properties--studies on isolated hepatocytes and lipid membrane vesicles. Hepatology. 1984;4:661-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 217] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 17. | King JE, Schoenfield LJ. Cholestasis induced by sodium taurolithocholate in isolated hamster liver. J Clin Invest. 1971;50:2305-2312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 42] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 18. | Schaffner F, Bacchin PG, Hutterer F, Scharnbeck HH, Sarkozi LL, Denk H, Popper H. Mechanism of cholestasis. 4. Structural and biochemical changes in the liver and serum in rats after bile duct ligation. Gastroenterology. 1971;60:888-897. [PubMed] |
| 19. | Poupon RE, Balkau B, Eschwège E, Poupon R. A multicenter, controlled trial of ursodiol for the treatment of primary biliary cirrhosis. UDCA-PBC Study Group. N Engl J Med. 1991;324:1548-1554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 570] [Cited by in RCA: 549] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 20. | Denk H, Schenkman JB, Bacchin PG, Hutterer F, Schaffner F, Popper H. Mechanism of cholestasis. 3. Interaction of synthetic detergents with the microsomal cytochrome P-450 dependentbiotransformation system in vitro. A comparison between the effects of detergents, the effects of bile acids, and the findings in bile duct ligated rats. Exp Mol Pathol. 1971;14:263-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 21. | Billington D, Evans CE, Godfrey PP, Coleman R. Effects of bile salts on the plasma membranes of isolated rat hepatocytes. Biochem J. 1980;188:321-327. [PubMed] |
| 22. | Galle PR, Theilmann L, Raedsch R, Otto G, Stiehl A. Ursodeoxycholate reduces hepatotoxicity of bile salts in primary human hepatocytes. Hepatology. 1990;12:486-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 182] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 23. | Patel T, Bronk SF, Gores GJ. Increases of intracellular magnesium promote glycodeoxycholate-induced apoptosis in rat hepatocytes. J Clin Invest. 1994;94:2183-2192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 193] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 24. | Kristiansen S, Jones J, Handberg A, Dohm GL, Richter EA. Eccentric contractions decrease glucose transporter transcription rate, mRNA, and protein in skeletal muscle. Am J Physiol. 1997;272:C1734-C1738. [PubMed] |
| 25. | Benz C, Angermüller S, Töx U, Klöters-Plachky P, Riedel HD, Sauer P, Stremmel W, Stiehl A. Effect of tauroursodeoxycholic acid on bile-acid-induced apoptosis and cytolysis in rat hepatocytes. J Hepatol. 1998;28:99-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 98] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 26. | Faubion WA, Guicciardi ME, Miyoshi H, Bronk SF, Roberts PJ, Svingen PA, Kaufmann SH, Gores GJ. Toxic bile salts induce rodent hepatocyte apoptosis via direct activation of Fas. J Clin Invest. 1999;103:137-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 420] [Cited by in RCA: 411] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 27. | Higuchi H, Bronk SF, Takikawa Y, Werneburg N, Takimoto R, El-Deiry W, Gores GJ. The bile acid glycochenodeoxycholate induces trail-receptor 2/DR5 expression and apoptosis. J Biol Chem. 2001;276:38610-38618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 138] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 28. | Greim H, Trülzsch D, Czygan P, Rudick J, Hutterer F, Schaffner F, Popper H. Mechanism of cholestasis. 6. Bile acids in human livers with or without biliary obstruction. Gastroenterology. 1972;63:846-850. [PubMed] |
| 29. | Spivey JR, Bronk SF, Gores GJ. Glycochenodeoxycholate-induced lethal hepatocellular injury in rat hepatocytes. Role of ATP depletion and cytosolic free calcium. J Clin Invest. 1993;92:17-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 190] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 30. | Grasl-Kraupp B, Ruttkay-Nedecky B, Koudelka H, Bukowska K, Bursch W, Schulte-Hermann R. In situ detection of fragmented DNA (TUNEL assay) fails to discriminate among apoptosis, necrosis, and autolytic cell death: a cautionary note. Hepatology. 1995;21:1465-1468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 196] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 31. | Jaeschke H, Williams CD, Farhood A. No evidence for caspase-dependent apoptosis in acetaminophen hepatotoxicity. Hepatology. 2011;53:718-719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 32. | Jaeschke H, Lemasters JJ. Apoptosis versus oncotic necrosis in hepatic ischemia/reperfusion injury. Gastroenterology. 2003;125:1246-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 441] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 33. | Cover C, Mansouri A, Knight TR, Bajt ML, Lemasters JJ, Pessayre D, Jaeschke H. Peroxynitrite-induced mitochondrial and endonuclease-mediated nuclear DNA damage in acetaminophen hepatotoxicity. J Pharmacol Exp Ther. 2005;315:879-887. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 279] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 34. | Yerushalmi B, Dahl R, Devereaux MW, Gumpricht E, Sokol RJ. Bile acid-induced rat hepatocyte apoptosis is inhibited by antioxidants and blockers of the mitochondrial permeability transition. Hepatology. 2001;33:616-626. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 250] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 35. | Schoemaker MH, Conde de la Rosa L, Buist-Homan M, Vrenken TE, Havinga R, Poelstra K, Haisma HJ, Jansen PL, Moshage H. Tauroursodeoxycholic acid protects rat hepatocytes from bile acid-induced apoptosis via activation of survival pathways. Hepatology. 2004;39:1563-1573. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 178] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 36. | Rust C, Wild N, Bernt C, Vennegeerts T, Wimmer R, Beuers U. Bile acid-induced apoptosis in hepatocytes is caspase-6-dependent. J Biol Chem. 2009;284:2908-2916. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 70] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 37. | Botla R, Spivey JR, Aguilar H, Bronk SF, Gores GJ. Ursodeoxycholate (UDCA) inhibits the mitochondrial membrane permeability transition induced by glycochenodeoxycholate: a mechanism of UDCA cytoprotection. J Pharmacol Exp Ther. 1995;272:930-938. [PubMed] |
| 38. | Zeid IM, Bronk SF, Fesmier PJ, Gores GJ. Cytoprotection by fructose and other ketohexoses during bile salt-induced apoptosis of hepatocytes. Hepatology. 1997;25:81-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 39. | Gores GJ, Miyoshi H, Botla R, Aguilar HI, Bronk SF. Induction of the mitochondrial permeability transition as a mechanism of liver injury during cholestasis: a potential role for mitochondrial proteases. Biochim Biophys Acta. 1998;1366:167-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 84] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 40. | Azzaroli F, Mehal W, Soroka CJ, Wang L, Lee J, Crispe IN, Boyer JL. Ursodeoxycholic acid diminishes Fas-ligand-induced apoptosis in mouse hepatocytes. Hepatology. 2002;36:49-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 63] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 41. | Rodrigues CM, Fan G, Ma X, Kren BT, Steer CJ. A novel role for ursodeoxycholic acid in inhibiting apoptosis by modulating mitochondrial membrane perturbation. J Clin Invest. 1998;101:2790-2799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 383] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 42. | Rodrigues CM, Ma X, Linehan-Stieers C, Fan G, Kren BT, Steer CJ. Ursodeoxycholic acid prevents cytochrome c release in apoptosis by inhibiting mitochondrial membrane depolarization and channel formation. Cell Death Differ. 1999;6:842-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 196] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 43. | Sokol RJ, Dahl R, Devereaux MW, Yerushalmi B, Kobak GE, Gumpricht E. Human hepatic mitochondria generate reactive oxygen species and undergo the permeability transition in response to hydrophobic bile acids. J Pediatr Gastroenterol Nutr. 2005;41:235-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 79] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 44. | Rodrigues CM, Fan G, Wong PY, Kren BT, Steer CJ. Ursodeoxycholic acid may inhibit deoxycholic acid-induced apoptosis by modulating mitochondrial transmembrane potential and reactive oxygen species production. Mol Med. 1998;4:165-178. [PubMed] |
| 45. | Sokol RJ, Devereaux M, Khandwala R, O'Brien K. Evidence for involvement of oxygen free radicals in bile acid toxicity to isolated rat hepatocytes. Hepatology. 1993;17:869-881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 46. | Sokol RJ, Winklhofer-Roob BM, Devereaux MW, McKim JM. Generation of hydroperoxides in isolated rat hepatocytes and hepatic mitochondria exposed to hydrophobic bile acids. Gastroenterology. 1995;109:1249-1256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 206] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 47. | Qiao L, Studer E, Leach K, McKinstry R, Gupta S, Decker R, Kukreja R, Valerie K, Nagarkatti P, El Deiry W. Deoxycholic acid (DCA) causes ligand-independent activation of epidermal growth factor receptor (EGFR) and FAS receptor in primary hepatocytes: inhibition of EGFR/mitogen-activated protein kinase-signaling module enhances DCA-induced apoptosis. Mol Biol Cell. 2001;12:2629-2645. [PubMed] |
| 48. | Zucchini-Pascal N, de Sousa G, Pizzol J, Rahmani R. Pregnane X receptor activation protects rat hepatocytes against deoxycholic acid-induced apoptosis. Liver Int. 2010;30:284-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 49. | Trauner M, Baghdasaryan A, Claudel T, Fickert P, Halilbasic E, Moustafa T, Zollner G. Targeting nuclear bile acid receptors for liver disease. Dig Dis. 2011;29:98-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 50. | Makishima M, Okamoto AY, Repa JJ, Tu H, Learned RM, Luk A, Hull MV, Lustig KD, Mangelsdorf DJ, Shan B. Identification of a nuclear receptor for bile acids. Science. 1999;284:1362-1365. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2012] [Cited by in RCA: 2093] [Article Influence: 80.5] [Reference Citation Analysis (0)] |
| 51. | Parks DJ, Blanchard SG, Bledsoe RK, Chandra G, Consler TG, Kliewer SA, Stimmel JB, Willson TM, Zavacki AM, Moore DD. Bile acids: natural ligands for an orphan nuclear receptor. Science. 1999;284:1365-1368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1717] [Cited by in RCA: 1754] [Article Influence: 67.5] [Reference Citation Analysis (0)] |
| 52. | Song C, Hiipakka RA, Liao S. Selective activation of liver X receptor alpha by 6alpha-hydroxy bile acids and analogs. Steroids. 2000;65:423-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 90] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 53. | Staudinger JL, Goodwin B, Jones SA, Hawkins-Brown D, MacKenzie KI, LaTour A, Liu Y, Klaassen CD, Brown KK, Reinhard J. The nuclear receptor PXR is a lithocholic acid sensor that protects against liver toxicity. Proc Natl Acad Sci USA. 2001;98:3369-3374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1020] [Cited by in RCA: 1043] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 54. | Kalaany NY, Mangelsdorf DJ. LXRS and FXR: the yin and yang of cholesterol and fat metabolism. Annu Rev Physiol. 2006;68:159-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 449] [Cited by in RCA: 470] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 55. | Liu Y, Binz J, Numerick MJ, Dennis S, Luo G, Desai B, MacKenzie KI, Mansfield TA, Kliewer SA, Goodwin B. Hepatoprotection by the farnesoid X receptor agonist GW4064 in rat models of intra- and extrahepatic cholestasis. J Clin Invest. 2003;112:1678-1687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 56. | Teng S, Piquette-Miller M. Hepatoprotective role of PXR activation and MRP3 in cholic acid-induced cholestasis. Br J Pharmacol. 2007;151:367-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 110] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 57. | Stedman C, Liddle C, Coulter S, Sonoda J, Alvarez JG, Evans RM, Downes M. Benefit of farnesoid X receptor inhibition in obstructive cholestasis. Proc Natl Acad Sci USA. 2006;103:11323-11328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 128] [Cited by in RCA: 138] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 58. | Fickert P, Fuchsbichler A, Moustafa T, Wagner M, Zollner G, Halilbasic E, Stöger U, Arrese M, Pizarro M, Solís N. Farnesoid X receptor critically determines the fibrotic response in mice but is expressed to a low extent in human hepatic stellate cells and periductal myofibroblasts. Am J Pathol. 2009;175:2392-2405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 144] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 59. | Marschall HU, Wagner M, Bodin K, Zollner G, Fickert P, Gumhold J, Silbert D, Fuchsbichler A, Sjövall J, Trauner M. Fxr(-/-) mice adapt to biliary obstruction by enhanced phase I detoxification and renal elimination of bile acids. J Lipid Res. 2006;47:582-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 87] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 60. | Zhang S, Wang J, Liu Q, Harnish DC. Farnesoid X receptor agonist WAY-362450 attenuates liver inflammation and fibrosis in murine model of non-alcoholic steatohepatitis. J Hepatol. 2009;51:380-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 221] [Cited by in RCA: 245] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 61. | Lian F, Xing X, Yuan G, Schäfer C, Rauser S, Walch A, Röcken C, Ebeling M, Wright MB, Schmid RM. Farnesoid X receptor protects human and murine gastric epithelial cells against inflammation-induced damage. Biochem J. 2011;438:315-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 62. | Allen K, Kim ND, Moon JO, Copple BL. Upregulation of early growth response factor-1 by bile acids requires mitogen-activated protein kinase signaling. Toxicol Appl Pharmacol. 2010;243:63-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 63. | Hylemon PB, Zhou H, Pandak WM, Ren S, Gil G, Dent P. Bile acids as regulatory molecules. J Lipid Res. 2009;50:1509-1520. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 527] [Cited by in RCA: 530] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 64. | Lai CQ, Arnett DK, Corella D, Straka RJ, Tsai MY, Peacock JM, Adiconis X, Parnell LD, Hixson JE, Province MA. Fenofibrate effect on triglyceride and postprandial response of apolipoprotein A5 variants: the GOLDN study. Arterioscler Thromb Vasc Biol. 2007;27:1417-1425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 103] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 65. | González R, Cruz A, Ferrín G, López-Cillero P, Fernández-Rodríguez R, Briceño J, Gómez MA, Rufián S, Mata Mde L, Martínez-Ruiz A. Nitric oxide mimics transcriptional and post-translational regulation during α-tocopherol cytoprotection against glycochenodeoxycholate-induced cell death in hepatocytes. J Hepatol. 2011;55:133-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 66. | Benz C, Angermüller S, Otto G, Sauer P, Stremmel W, Stiehl A. Effect of tauroursodeoxycholic acid on bile acid-induced apoptosis in primary human hepatocytes. Eur J Clin Invest. 2000;30:203-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 66] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 67. | Schoemaker MH, Gommans WM, Conde de la Rosa L, Homan M, Klok P, Trautwein C, van Goor H, Poelstra K, Haisma HJ, Jansen PL. Resistance of rat hepatocytes against bile acid-induced apoptosis in cholestatic liver injury is due to nuclear factor-kappa B activation. J Hepatol. 2003;39:153-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 102] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 68. | Schoemaker MH, Ros JE, Homan M, Trautwein C, Liston P, Poelstra K, van Goor H, Jansen PL, Moshage H. Cytokine regulation of pro- and anti-apoptotic genes in rat hepatocytes: NF-kappaB-regulated inhibitor of apoptosis protein 2 (cIAP2) prevents apoptosis. J Hepatol. 2002;36:742-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 125] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 69. | Tchaparian EH, Houghton JS, Uyeda C, Grillo MP, Jin L. Effect of culture time on the basal expression levels of drug transporters in sandwich-cultured primary rat hepatocytes. Drug Metab Dispos. 2011;39:2387-2394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 70. | Liang D, Hagenbuch B, Stieger B, Meier PJ. Parallel decrease of Na(+)-taurocholate cotransport and its encoding mRNA in primary cultures of rat hepatocytes. Hepatology. 1993;18:1162-1166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 71. | Kwo P, Patel T, Bronk SF, Gores GJ. Nuclear serine protease activity contributes to bile acid-induced apoptosis in hepatocytes. Am J Physiol. 1995;268:G613-G621. [PubMed] |
| 72. | Pusl T, Vennegeerts T, Wimmer R, Denk GU, Beuers U, Rust C. Tauroursodeoxycholic acid reduces bile acid-induced apoptosis by modulation of AP-1. Biochem Biophys Res Commun. 2008;367:208-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 73. | Fickert P, Zollner G, Fuchsbichler A, Stumptner C, Weiglein AH, Lammert F, Marschall HU, Tsybrovskyy O, Zatloukal K, Denk H. Ursodeoxycholic acid aggravates bile infarcts in bile duct-ligated and Mdr2 knockout mice via disruption of cholangioles. Gastroenterology. 2002;123:1238-1251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 240] [Cited by in RCA: 238] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 74. | Wagner M, Fickert P, Zollner G, Fuchsbichler A, Silbert D, Tsybrovskyy O, Zatloukal K, Guo GL, Schuetz JD, Gonzalez FJ. Role of farnesoid X receptor in determining hepatic ABC transporter expression and liver injury in bile duct-ligated mice. Gastroenterology. 2003;125:825-838. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 213] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 75. | Alnouti Y, Csanaky IL, Klaassen CD. Quantitative-profiling of bile acids and their conjugates in mouse liver, bile, plasma, and urine using LC-MS/MS. J Chromatogr B Analyt Technol Biomed Life Sci. 2008;873:209-217. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 223] [Cited by in RCA: 211] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 76. | Saito JM, Maher JJ. Bile duct ligation in rats induces biliary expression of cytokine-induced neutrophil chemoattractant. Gastroenterology. 2000;118:1157-1168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 94] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 77. | Georgiev P, Jochum W, Heinrich S, Jang JH, Nocito A, Dahm F, Clavien PA. Characterization of time-related changes after experimental bile duct ligation. Br J Surg. 2008;95:646-656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 183] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 78. | Ito Y, Bethea NW, Baker GL, McCuskey MK, Urbaschek R, McCuskey RS. Hepatic microcirculatory dysfunction during cholestatic liver injury in rats. Microcirculation. 2003;10:421-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 79. | Bilzer M, Lauterburg BH. Effects of hypochlorous acid and chloramines on vascular resistance, cell integrity, and biliary glutathione disulfide in the perfused rat liver: modulation by glutathione. J Hepatol. 1991;13:84-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 39] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 80. | Demirbilek S, Akin M, Gürünlüoğlu K, Aydin NE, Emre MH, Taş E, Aksoy RT, Ay S. The NF-kappaB inhibitors attenuate hepatic injury in bile duct ligated rats. Pediatr Surg Int. 2006;22:655-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 81. | Gujral JS, Hinson JA, Jaeschke H. Chlorotyrosine protein adducts are reliable biomarkers of neutrophil-induced cytotoxicity in vivo. Comp Hepatol. 2004;3 Suppl 1:S48. [PubMed] |
| 82. | Jaeschke H. Mechanisms of Liver Injury. II. Mechanisms of neutrophil-induced liver cell injury during hepatic ischemia-reperfusion and other acute inflammatory conditions. Am J Physiol Gastrointest Liver Physiol. 2006;290:G1083-G1088. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 329] [Cited by in RCA: 350] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 83. | Jaeschke H, Hasegawa T. Role of neutrophils in acute inflammatory liver injury. Liver Int. 2006;26:912-919. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 189] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 84. | Jaeschke H, Ho YS, Fisher MA, Lawson JA, Farhood A. Glutathione peroxidase-deficient mice are more susceptible to neutrophil-mediated hepatic parenchymal cell injury during endotoxemia: importance of an intracellular oxidant stress. Hepatology. 1999;29:443-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 154] [Cited by in RCA: 147] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 85. | Gujral JS, Hinson JA, Farhood A, Jaeschke H. NADPH oxidase-derived oxidant stress is critical for neutrophil cytotoxicity during endotoxemia. Am J Physiol Gastrointest Liver Physiol. 2004;287:G243-G252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 82] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 86. | Paik YH, Iwaisako K, Seki E, Inokuchi S, Schnabl B, Osterreicher CH, Kisseleva T, Brenner DA. The nicotinamide adenine dinucleotide phosphate oxidase (NOX) homologues NOX1 and NOX2/gp91(phox) mediate hepatic fibrosis in mice. Hepatology. 2011;53:1730-1741. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 174] [Cited by in RCA: 177] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 87. | Cui W, Matsuno K, Iwata K, Ibi M, Matsumoto M, Zhang J, Zhu K, Katsuyama M, Torok NJ, Yabe-Nishimura C. NOX1/nicotinamide adenine dinucleotide phosphate, reduced form (NADPH) oxidase promotes proliferation of stellate cells and aggravates liver fibrosis induced by bile duct ligation. Hepatology. 2011;54:949-958. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 97] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 88. | Bajt ML, Farhood A, Jaeschke H. Effects of CXC chemokines on neutrophil activation and sequestration in hepatic vasculature. Am J Physiol Gastrointest Liver Physiol. 2001;281:G1188-G1195. [PubMed] |
| 89. | Fickert P, Fuchsbichler A, Marschall HU, Wagner M, Zollner G, Krause R, Zatloukal K, Jaeschke H, Denk H, Trauner M. Lithocholic acid feeding induces segmental bile duct obstruction and destructive cholangitis in mice. Am J Pathol. 2006;168:410-422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 287] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 90. | Jaeschke H, Gujral JS, Bajt ML. Apoptosis and necrosis in liver disease. Liver Int. 2004;24:85-89. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 115] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 91. | Jaeschke H. Molecular mechanisms of hepatic ischemia-reperfusion injury and preconditioning. Am J Physiol Gastrointest Liver Physiol. 2003;284:G15-G26. [PubMed] |
| 92. | Canbay A, Guicciardi ME, Higuchi H, Feldstein A, Bronk SF, Rydzewski R, Taniai M, Gores GJ. Cathepsin B inactivation attenuates hepatic injury and fibrosis during cholestasis. J Clin Invest. 2003;112:152-159. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 160] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 93. | Miyoshi H, Rust C, Guicciardi ME, Gores GJ. NF-kappaB is activated in cholestasis and functions to reduce liver injury. Am J Pathol. 2001;158:967-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 62] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 94. | Miyoshi H, Rust C, Roberts PJ, Burgart LJ, Gores GJ. Hepatocyte apoptosis after bile duct ligation in the mouse involves Fas. Gastroenterology. 1999;117:669-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 237] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 95. | Canbay A, Feldstein A, Baskin-Bey E, Bronk SF, Gores GJ. The caspase inhibitor IDN-6556 attenuates hepatic injury and fibrosis in the bile duct ligated mouse. J Pharmacol Exp Ther. 2004;308:1191-1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 166] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 96. | Nalapareddy P, Schüngel S, Hong JY, Manns MP, Jaeschke H, Vogel A. The BH3-only protein bid does not mediate death-receptor-induced liver injury in obstructive cholestasis. Am J Pathol. 2009;175:1077-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 97. | Higuchi H, Miyoshi H, Bronk SF, Zhang H, Dean N, Gores GJ. Bid antisense attenuates bile acid-induced apoptosis and cholestatic liver injury. J Pharmacol Exp Ther. 2001;299:866-873. [PubMed] |
| 98. | Gujral JS, Liu J, Farhood A, Jaeschke H. Reduced oncotic necrosis in Fas receptor-deficient C57BL/6J-lpr mice after bile duct ligation. Hepatology. 2004;40:998-1007. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 35] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 99. | Fickert P, Trauner M, Fuchsbichler A, Zollner G, Wagner M, Marschall HU, Zatloukal K, Denk H. Oncosis represents the main type of cell death in mouse models of cholestasis. J Hepatol. 2005;42:378-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 69] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 100. | Kim ND, Moon JO, Slitt AL, Copple BL. Early growth response factor-1 is critical for cholestatic liver injury. Toxicol Sci. 2006;90:586-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 89] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 101. | Wang H, Zhang Y, Heuckeroth RO. Tissue-type plasminogen activator deficiency exacerbates cholestatic liver injury in mice. Hepatology. 2007;45:1527-1537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 102. | Jaeschke H, Fisher MA, Lawson JA, Simmons CA, Farhood A, Jones DA. Activation of caspase 3 (CPP32)-like proteases is essential for TNF-alpha-induced hepatic parenchymal cell apoptosis and neutrophil-mediated necrosis in a murine endotoxin shock model. J Immunol. 1998;160:3480-3486. [PubMed] |
| 103. | Schotte P, Declercq W, Van Huffel S, Vandenabeele P, Beyaert R. Non-specific effects of methyl ketone peptide inhibitors of caspases. FEBS Lett. 1999;442:117-121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 235] [Cited by in RCA: 244] [Article Influence: 9.4] [Reference Citation Analysis (0)] |