Published online Aug 21, 2012. doi: 10.3748/wjg.v18.i31.4199
Revised: June 26, 2012
Accepted: June 28, 2012
Published online: August 21, 2012
AIM: To investigate the potential mechanism of Arg-Gly-Asp (RGD) peptide-labeled liposome loading oxymatrine (OM) therapy in CCl4-induced hepatic fibrosis in rats.
METHODS: We constructed a rat model of CCl4-induced hepatic fibrosis and treated the rats with different formulations of OM. To evaluate the antifibrotic effect of OM, we detected levels of alkaline phosphatase, hepatic histopathology (hematoxylin and eosin stain and Masson staining) and fibrosis-related gene expression of matrix metallopeptidase (MMP)-2, tissue inhibitor of metalloproteinase (TIMP)-1 as well as type I procollagen via quantitative real-time polymerase chain reaction. To detect cell viability and apoptosis of hepatic stellate cells (HSCs), we performed 3-(4,5)-dimethylthiahiazo(-z-y1)-3,5-diphenytetrazoliumromide assay and flow cytometry. To reinforce the combination of oxymatrine with HSCs, we constructed fluorescein-isothiocyanate-conjugated Arg-Gly-Asp peptide-labeled liposomes loading OM, and its targeting of HSCs was examined by fluorescent microscopy.
RESULTS: OM attenuated CCl4-induced hepatic fibrosis, as defined by reducing serum alkaline phosphatase (344.47 ± 27.52 U/L vs 550.69 ± 43.78 U/L, P < 0.05), attenuating liver injury and improving collagen deposits (2.36% ± 0.09% vs 7.70% ± 0.60%, P < 0.05) and downregulating fibrosis-related gene expression, that is, MMP-2, TIMP-1 and type I procollagen (P < 0.05). OM inhibited cell viability and induced apoptosis of HSCs in vitro. RGD promoted OM targeting of HSCs and enhanced the therapeutic effect of OM in terms of serum alkaline phosphatase (272.51 ± 19.55 U/L vs 344.47 ± 27.52 U/L, P < 0.05), liver injury, collagen deposits (0.26% ± 0.09% vs 2.36% ± 0.09%, P < 0.05) and downregulating fibrosis-related gene expression, that is, MMP-2, TIMP-1 and type I procollagen (P < 0.05). Moreover, in vitro assay demonstrated that RGD enhanced the effect of OM on HSC viability and apoptosis.
CONCLUSION: OM attenuated hepatic fibrosis by inhibiting viability and inducing apoptosis of HSCs. The RGD-labeled formulation enhanced the targeting efficiency for HSCs and the therapeutic effect.
-
Citation: Chai NL, Fu Q, Shi H, Cai CH, Wan J, Xu SP, Wu BY. Oxymatrine liposome attenuates hepatic fibrosis
via targeting hepatic stellate cells. World J Gastroenterol 2012; 18(31): 4199-4206 - URL: https://www.wjgnet.com/1007-9327/full/v18/i31/4199.htm
- DOI: https://dx.doi.org/10.3748/wjg.v18.i31.4199
Hepatic fibrosis is characterized by excessive deposition of extracellular matrix (ECM) components in the interstitial space of the liver[1,2]. The fibrogenesis is triggered by a variety of events that lead to chronic injury, including viral infection, drug or alcoholic toxicity, autoimmune disorders and metabolic diseases. As a consequence of liver damage, nonparenchymal cells are activated by a concert of mediators released from injured hepatocytes. A population of nonparenchymal cells, the hepatic stellate cells (HSCs), have been identified and recognized for their contributing role in the fibrotic process after transformation towards myofibroblasts[3]. Thus, HSCs represent an attractive target for the development of antifibrotic strategies incorporating a selective targeting approach for hepatic fibrosis[4]. Recently, several therapeutic strategies have been developed by means of targeting hepatic fibrosis, including inhibition of collagen synthesis[5], interruption of matrix deposition[6], stimulation of matrix degradation, modulation of HSC activation[7], or induction of HSC death[8]. Despite advance in understanding hepatic fibrogenesis, therapeutic repertoire for hepatic fibrosis treatment is still limited.
Oxymatrine (OM), an alkaloid extracted from the medicinal plant Sophora alopecuraides L, has received increasing attention for its multiple pharmacological functions. OM has been demonstrated to exert an inhibitory effect on the replication of hepatitis B[9] and C[10] viruses in vitro. Preclinical and clinical studies have shown that OM effectively inhibited infection with hepatitis B virus[11]. In addition to antiviral effects, OM has been reported to have a beneficial effect on progression of CCl4-induced hepatic fibrosis in rats. Recent studies have demonstrated that OM induces apoptosis in a variety of cells; mainly malignant cells[12]. Apoptosis-inducing activity of OM makes it an attractive antifibrotic agent. However, there is limited evidence for the efficacy of OM in hepatic fibrosis and the underlying mechanism.
In the present study we aimed to investigate whether Arg-Gly-Asp (RGD)-mediated targeting delivery of OM exerted antifibrogenic action with improved efficiency of fibrogenic liver[13]. In vitro experiments showed that uptake of OM in HSCs was enhanced and the apoptotic process was induced. In CCl4-induced rats, delivery of OM to HSCs with this formulation strategy improved the efficacy of this medication in the treatment of hepatic fibrosis.
The lipid phase consisting of a mixture of lecithin and cholesterol in a ratio of 2:1 was dissolved in CHCl3-MeOH (1:1) followed by evaporation and addition of pleic acid and polysorbate. Lipids were mixed with the aqueous solution containing OM and polyvinylpyrrolidone in phosphate-buffered saline (PBS; pH 7.4). The mixture was sonicated for 5 min at 50% amplifying strength resulting in a water-in-oil emulsion. After removal of the organic solvent with a rotary evaporator under vacuum, the dispersion of liposomes was formed.
RGD peptide was synthesized by the Chinese Peptide Company (Hangzhou, China). RGD peptide coupling was performed as described previously[14]. In brief, 4 nmol cyclo-Arg-Gly-Asp (cRGD) peptide per mmol total lipid was added after deacetylation of the peptide in 0.5 mol hydroxylamine solution, and incubated for 1 h at room temperature. Unloaded liposomes and unbound RGD were separated by CL-4B column (Amersham, Piscataway, NJ, United States).
HSCs were isolated by collagenase perfusion through the portal vein in Sprague-Dawley rats, followed by Nycodenz gradient centrifugation. Cells were incubated in Dulbecco’s Modified Eagle’s Medium (DMEM) supplemented with 10% fetal bovine serum (v/v) and 100 U/mL penicillin and streptomycin, and maintained at 37 °C in a humidified incubator (90% humidity) containing 50 mL/L CO2. HSCs seeded at a density of 104 cells/cm2 attained confluence in 6 d and formed a monolayer of closely apposed polygonal cells. The morphology and growth of HSCs were confirmed and evaluated by microscopy.
Cell viability was determined through 3-(4,5)-dimethylthiahiazo(-z-y1)-3,5-diphenytetrazoliumromide (MTT) assay. The MTT assay depends on the extent to which viable cells convert MTT bromide to an insoluble colored formazan product that can be determined spectrophotometrically. After treatment, cells were harvested and washed in PBS, and 200 mL DMEM without phenol red, containing 5 mg/mL MTT, was added to each cell. Three hours later, the medium was aspirated, and the converted dye was solubilized with isopropanol (0.1 mol/L HCl in isopropanol). The resulting absorbance from each cell was measured at a wavelength of 570 nm with background subtraction at 630 nm.
HSCs were treated with different concentration of OM-RGD liposomes for 48 h at a cell density of 2 × 105 cells/mL, and then stained with annexin V-fluorescein isothiocyanate (FITC) and propidium iodide (PI) (Sigma, St Louis, MO, United States). Annexin V-FITC-positive and PI-negative cells were considered to be apoptotic cells.
HSCs were cultured with different formulation of OM at 37 °C for 24 h and then harvested by trypsinization and centrifugation for 10 min at 3500 rpm at room temperature. Cells were fixed in 4% (v/v) glutaraldehyde for 4 h at 4 °C. The specimens were washed with sodium cacodylate buffer (pH 7.4) followed by post-fixation in 1% osmium tetroxide at 4 °C. Cells were then washed with cacodylate buffer (pH 7.4) and dehydrated with a graded series of acetone. The cells were embedded with 100% resin in a beam capsule. A sample block was sectioned using an ultramicrotome. The sections were placed into a grid and stained with uranyl acetate for 10 min followed by 50% filtered acetone, and finally stained with lead. The stained samples were then viewed under a transmission electron microscope (Phillips, Eindhoven, The Netherlands).
Total RNA was extracted from cells using the TRIzol reagent. The amount of each RNA sample was determined by Qubit fluorometer. Reverse transcription was performed in a 20-μL reaction system with 200 ng total RNA using high capacity cDNA reverse transcription kits. Relative quantification of designated genes including MMP-2, TIMP and type I procollagen were assessed by real-time PCR through the ABI 7900HT system. A housekeeping gene glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal control. Primer sequences used are shown as follows: type I procollagen, 5’-CCTGGCAGAACGGAGATGAT-3’, 5’-ACCA CAGCACCATCGTTACC-3’; MMP-2, 5’-CTATTCTGT CAGCACTT TGG-3’, 5’-CAGACTTTGGTTCTCCA ACTT-3’; TIMP-1, 5’-ACA GCTTT CTGCA ACTCG-3’, 5’-CTATAGGTCTTT ACGAAGGCC-3’; GADPH, 5’-AC CCCCC AATGTATCCGTTGT-3’, 5’-TA CTCCTTGGAGGCCATGTA-3’. The relative abundance of target mRNA was determined with the comparative cycle threshold method.
Male Sprague-Dawley rats came from the Experimental Animal Center of Beijing Medical University (Beijing, China) and were caged in an environment with regulated temperature (21 ± 1.6 °C), humidity (45% ± 10%), and an alternating 12-h light and dark cycle. The animals had free access to water and diet throughout the study.
For the chronic liver injury model, animals were injected intraperitoneally with 50% CCl4 (CCl4:vegetable oil = 1:1) at a dose of 0.15 mL/100 g body weight and were also divided into four groups: Group A (administered with OM-loaded liposome); Group B (administered with OM-RGD liposome); Group C (hepatic fibrosis); and Group D (blank vector). Injections were given three times weekly for 8 wk. After treatment, the rats received an intravenous injection according to their subgrouping. Groups A and B were given OM liposomes and OM-RGD liposomes, respectively. The rats in Group D were administered with 0.5 mL blank liposomes. The rats were sacrificed 8 wk after injection, and the livers and blood samples were collected for further assessments.
For HSC targeting, CCl4-treated animals were injected with FITC-labeled OM liposomes and OM-RGD liposomes via the tail vein. Hepatocytes and HSCs were isolated from each individual rat 24 h post-injection, as described previously[15].
Animals were sacrificed 8 wk after treatment. Liver samples from each group were harvested, fixed with 10% formaldehyde, embedded in olefin, and stained with hematoxylin and eosin (HE) as well as Masson collagen staining.
The data are expressed as mean ± SD. Student’s t-test or Dunnett’s t-test was used to compare the differences between treated and control groups, and differences were considered significant at P < 0.05.
We evaluated the therapeutic effect of OM-RGD liposomes on CCl4-induced liver injury in rats. As shown in Table 1, elevated levels of alkaline phosphatase were reduced by OM. Histopathological analysis revealed that CCl4-induced hepatic fibrosis was ameliorated by OM (Figure 1A). Excessive collagen deposited in response to CCl4-induced liver injury was ameliorated by OM (Table 1, Figure 1B). Moreover, the RGD-labeled liposomal formulation exerted a more aggressive therapeutic effect in hepatic fibrosis than did OM in terms of alkaline phosphatase (Table 1), histopathology (Figure 1A), and collagen deposits (Table 1, Figure 1B).
| Group | n | ALP (U/L) |
| Normal | 10 | 73.91 ± 5.97 |
| CCl4-induced hepatic fibrosis | 10 | 550.69 ± 43.80e |
| OM-RGD liposomes | 10 | 272.51 ± 19.55ac |
| OM liposomes | 10 | 344.47 ± 27.52c |
| RGD liposomes | 10 | 562.78 ± 40.22 |
| Collagen area (%) | ||
| CCl4-induced hepatic fibrosis | 5 | 7.70 ± 0.60 |
| RGD liposomes | 5 | 8.32 ± 0.42 |
| OM liposomes | 5 | 2.36 ± 0.09c |
| OM-RGD liposomes | 5 | 0.26 ± 0.09e |
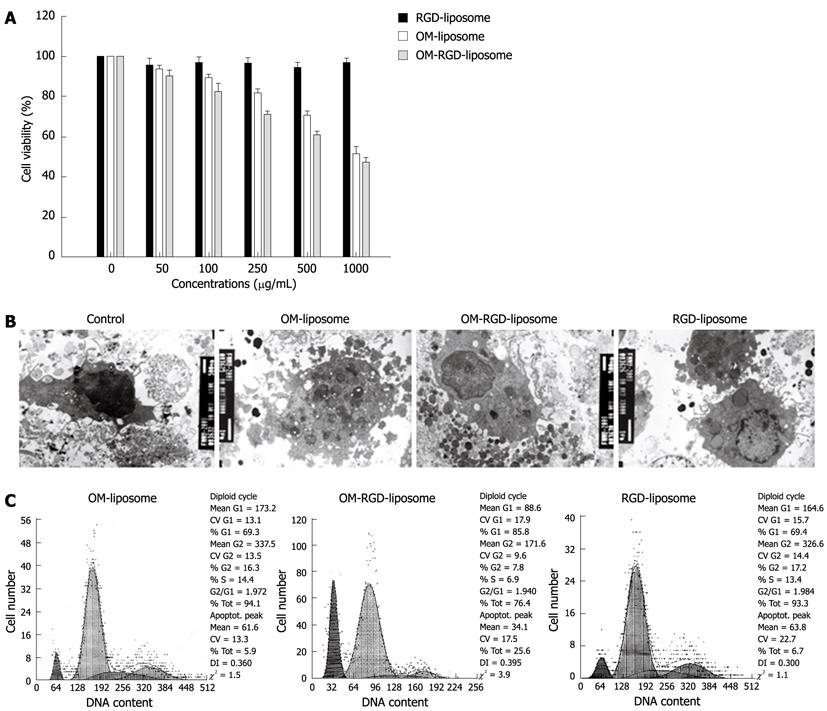
The inhibitory effect of OM-RGD liposomes on the viability of primary HSCs was determined by MTT assay. As shown in Figure 2A, OM-RGD liposomes significantly inhibited the viability of HSCs, whereas OM liposomes exhibited low cytotoxicity in HSCs. The ultrastructure of HSCs treated with different formulation of OM was examined by transmission electron microscopy (TEM). TEM revealed that treatment with OM-RGD liposomes resulted in typical morphological sign of apoptosis, including cell shrinkage, increased cellular granularity, and formation of apoptotic bodies (Figure 2B). The apoptotic effect of different formulations of OM was determined by using flow cytometry. Cell cycle analysis revealed that incubation with OM-RGD liposomes resulted in a significant increase in sub-G1 phase accumulation that was recognized as a biomarker of apoptosis (Figure 2C). Moreover, the RGD-labeled liposomal formulation had a more aggressive effect on HSCs than that of OM in terms of cell viability (Figure 2) and apoptosis (Figures 3 and 4)
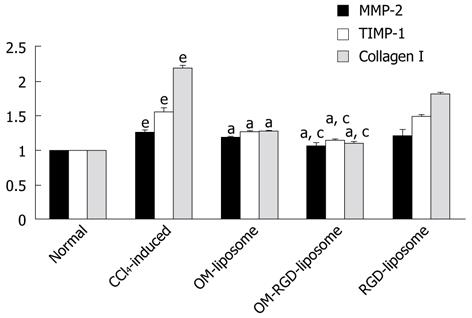
We also examined the change in mRNA expression of fibrosis-related genes upon treatment with OM in different liposomal formulations. As shown in Figure 3, mRNA expression of MMP-2, TIMP-1 and type I procollagen was considerably elevated upon CCl4 induction (vs normal, P < 0.05). Treatment with OM resulted in significant decreases in mRNA expression of these designated fibrosis-related genes and fibrosis-related gene expression (vs CCl4-induced group, P < 0.05). Moreover, RGD-labeled liposomal formulation had a more aggressive downregulation of fibrosis-related gene expression than that of OM (vs CCl4-induced hepatic fibrosis group, P < 0.05; as compared to OM liposomes, P < 0.05).
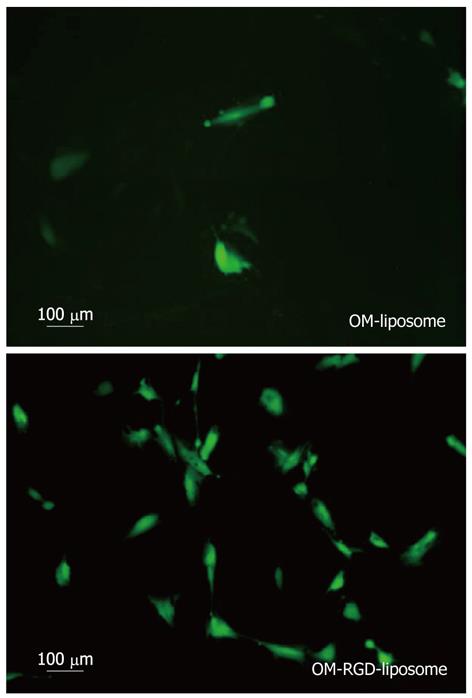
To evaluate the specificity of binding to HSCs in fibrotic liver, OM-RGD liposomes were conjugated with FITC and injected intravenously to rats with CCl4-induced hepatic fibrosis. HSCs were isolated and examined by fluorescence microscopy. As shown in Figure 4, a significantly high number of FITC-positive HSCs was found in the OM-RGD liposome group compared with the OM liposome group.
Increased understanding of the pathogenesis of hepatic fibrosis has led to drug discovery for its treatment. Preclinical and clinical studies have reported that hepatic fibrosis is dynamic and possibly reversible[16,17]. During recent decades, antifibrotic strategies have predominantly focused on eradication of causative factors, for example, clearance of virus[18]. Since the pathogenesis of fibrosis was clarified recently[19], researches have focused on agents that could prevent or reverse fibrosis. OM has been reported for its pharmacological potential to treat liver disorders, particularly inhibiting viral infection[10,11,20,21]. It has been demonstrated to exert antifibrotic action[22]. In our study, we confirmed that OM could attenuate CCl4-induced hepatic fibrosis in a rat model, as defined by a significant decrease in the serum level of alkaline phosphatase and improvement of histopathological change.
OM was recently referred to as an antifibrosis agent in clinical and preclinical studies. However, its mechanisms of action were still puzzling. In preclinical studies, it was proved that OM showed prophylactic and therapeutic effects in D-galactosamine-induced rat hepatic fibrosis, partly by protecting hepatocytes and suppressing fibrosis accumulation through ating against lipid peroxidation[23]. Another study also demonstrated that OM was effective in reducing the production and deposition of collagen in the liver tissue of experimental rats in ways that relate to modulating the fibrogenic signal transduction via the p38 mitogen-activated protein kinase signaling pathway[24]. Moreover, OM could promote the expression of Smad 7 and inhibit the expression of Smad 3 and cAMP-response element binding protein-binding protein in CCl4-induced hepatic fibrosis in rats, and modulate the fibrogenic signal transduction of the transforming growth factor (TGF)-β-Smad pathway[25]. Clinical studies have proved that the effect of OM against hepatic fibrosis is mediated by lowering the levels of TGF-β1 and tumor necrosis factor-α and increasing the level of interleukin-10 in chronic hepatitis B patients[26]. OM could also target directly fibrogenic effector cells, which has received much attention[27]. Various cells are involved in the fibrogenic process. HSCs, the most important fibrogenic cells, are activated during injury and contribute to excessive synthesis of ECM, resulting in deposition of scar or fibrous tissue[28-31]. OM has been demonstrated to prevent pig-serum-induced liver fibrosis in rats by inhibiting the activation of HSCs and synthesis of collagen[32]. However, how OM inhibits HSC activation was not determined in that study[32].
We explored the effect of OM on HSCs in vitro. OM inhibited viability and induced apoptosis of HSCs. This might be the underlying mechanism involved in OM therapy of hepatic fibrosis. Furthermore, we also detected fibrosis-related gene expression after OM administration. MMP-2, produced by HSCs, plays an important role in liver fibrogenesis[33]. TIMPs, especially TIMP-1 and -2 expression and activity, were significantly increased at 8 wk in a rat porcine-serum-induced hepatic fibrosis model[34]. Furthermore, inhibition of cell viability and type I procollagen expression in rat HSCs could improve recovery from CCl4-induced liver fibrogenesis in rats[35]. As shown in our study, mRNA expression of MMP-2, TIMP-1 and type I procollagen was considerably elevated upon CCl4 induction. Treatment with OM resulted in significant decreases in mRNA expression of these designated fibrosis-related genes. All the data indicated that OM could attenuate hepatic fibrosis via its effect on HSCs, such as inhibiting cell viability, inducing apoptosis and downregulation of fibrosis-related gene expression.
Since OM therapy was dependent on its interaction with HSCs, binding to HSCs became a key factor for its function. Due to a relatively small population of HSCs in the liver and lack of specific membrane receptors, HSC-specific targeting therapy has remained unavailable. Several studies have attempted to use different formulation approaches for targeting HSCs. Beljaars et al[36] have reported that human serum albumin (HSA) modified with mannose 6-phosphate (M6P) accumulated in HSCs by binding to the M6P-insulin-like growth factor II receptors found on activated HSCs. Modification of HSA with a cyclic peptide that recognizes the collagen type VI receptor has been demonstrated to enhance effectiveness and tissue specificity of antifibrogenic drugs[37]. Moreover, the affinity of a cyclic peptide, cRGD, for collagen type VI receptor on HSCs was confirmed in both in vitro and in vivo experiments[13]. In our study, in order to facilitate OM binding to HSCs, we conjugated liposomes targeted to HSCs in rats with CCl4-induced hepatic fibrosis. Fluorescence microscopy showed more FITC-positive HSCs in the OM-RGD-liposome group compared with the OM-liposome group. We compared the difference in therapeutic effect of the alternative formulations of OM on liver fibrosis. We demonstrated better results in the OM-RGD-liposome group, as demonstrated by significant decreases in serum alkaline phosphatase and improvement of histopathological changes, compared with the OM-liposome group. Moreover, OM-RGD liposomes showed a more aggressive effect on viability, apoptosis and fibrosis-related gene expression of HSCs, compared with the OM liposomes. The results showed that specific binding of this liposomal formulation to HSCs enhanced the liver protective effect of OM.
In conclusion, we conjugated OM with RGD liposomes and confirmed that this formulation could enhance OM binding to HSCs and the therapeutic effect on hepatic fibrosis induced by CCl4. We also demonstrated that the therapeutic effects of OM on hepatic fibrosis were partly dependent on inhibiting cell viability, inducing apoptosis, and downregulating fibrosis-related gene expression of HSCs, thus highlighting OM-RGD liposomes as an attractive novel therapy in liver fibrosis.
Oxymatrine (OM) has been reported to have a beneficial effect on progression of CCl4-induced hepatic fibrosis in rats, however, its mechanism of action is still uncertain. Hepatic stellate cells (HSCs) have been identified as an important factor in the hepatic fibrotic process. Drugs that could induce HSC apoptosis or death might be the potential strategy for treatment of hepatic fibrosis. Recent studies have demonstrated that OM induces apoptosis in a variety of cells; mainly malignant cells. Thus, the authors performed an assay to demonstrate whether OM could attenuate hepatic fibrosis via inducing HSC apoptosis.
OM was demonstrated to attenuate hepatic fibrosis but its mechanisms of action were still uncertain. Moreover, targeting of HSCs might facilitate the therapeutic effect of OM. The research hotspot is to clarify the mechanism of action of OM in attenuating hepatic fibrosis and how to enhance OM binding to HSCs.
Apoptosis-inducing activity of OM makes it an attractive antifibrotic agent. However, there is limited evidence for the efficacy of OM in hepatic fibrosis and the underlying mechanism of action. The authors demonstrated for the first time that OM could attenuate hepatic fibrosis via inhibiting viability and inducing apoptosis of HSCs. Moreover, Arg-Gly-Asp (RGD) could promote OM targeting to HSCs and enhance its effect on hepatic fibrosis.
This study aimed to analyze the effects of RGD-peptide-labeled liposomes on CCl4-induced hepatic fibrosis in rats. This is an interesting approach that clearly improves the efficacy of treatment. The research combined in vitro and in vitro studies, and data are clear and well presented.
Peer reviewer: Javier Gonzalez-Gallego, Professor, Institute of Biomedicine, University of Leon, 24071 Leon, Spain
S- Editor Gou SX L- Editor Kerr C E- Editor Zhang DN
| 1. | Friedman SL. Molecular regulation of hepatic fibrosis, an integrated cellular response to tissue injury. J Biol Chem. 2000;275:2247-2250. [PubMed] |
| 2. | Mormone E, George J, Nieto N. Molecular pathogenesis of hepatic fibrosis and current therapeutic approaches. Chem Biol Interact. 2011;193:225-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 198] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 3. | Kastanis GJ, Hernandez-Nazara Z, Nieto N, Rincón-Sanchez AR, Popratiloff A, Dominguez-Rosales JA, Lechuga CG, Rojkind M. The role of dystroglycan in PDGF-BB-dependent migration of activated hepatic stellate cells/myofibroblasts. Am J Physiol Gastrointest Liver Physiol. 2011;301:G464-G474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 4. | Kim MR, Kim HS, Lee MS, Lee MJ, Jang JJ. Cell cycle protein profile of the hepatic stellate cells(HSCs)in dimethylnitrosamine-induced rat hepatic fibrosis. Exp Mol Med. 2005;37:335-342. [PubMed] |
| 5. | Zhang Y, Liu P, Gao X, Qian W, Xu K. rAAV2-TGF-β(3) decreases collagen synthesis and deposition in the liver of experimental hepatic fibrosis rat. Dig Dis Sci. 2010;55:2821-2830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 25] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Baroni GS, D'Ambrosio L, Curto P, Casini A, Mancini R, Jezequel AM, Benedetti A. Interferon gamma decreases hepatic stellate cell activation and extracellular matrix deposition in rat liver fibrosis. Hepatology. 1996;23:1189-1199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 204] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 7. | Rosenberg P, Sjöström M, Söderberg C, Kinnman N, Stål P, Hultcrantz R. Attenuated liver fibrosis after bile duct ligation and defective hepatic stellate cell activation in neural cell adhesion molecule knockout mice. Liver Int. 2011;31:630-641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 8. | Li J, Liu P, Zhang R, Cao L, Qian H, Liao J, Xu W, Wu M, Yin Z. Icaritin induces cell death in activated hepatic stellate cells through mitochondrial activated apoptosis and ameliorates the development of liver fibrosis in rats. J Ethnopharmacol. 2011;137:714-723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 45] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 9. | Wang YP, Zhao W, Xue R, Zhou ZX, Liu F, Han YX, Ren G, Peng ZG, Cen S, Chen HS. Oxymatrine inhibits hepatitis B infection with an advantage of overcoming drug-resistance. Antiviral Res. 2011;89:227-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 78] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 10. | Wu XN, Wang GJ. Experimental studies of oxymatrine and its mechanisms of action in hepatitis B and C viral infections. Chin J Dig Dis. 2004;5:12-16. [PubMed] |
| 11. | Chen XS, Wang GJ, Cai X, Yu HY, Hu YP. Inhibition of hepatitis B virus by oxymatrine in vivo. World J Gastroenterol. 2001;7:49-52. [PubMed] |
| 12. | Ling Q, Xu X, Wei X, Wang W, Zhou B, Wang B, Zheng S. Oxymatrine induces human pancreatic cancer PANC-1 cells apoptosis via regulating expression of Bcl-2 and IAP families, and releasing of cytochrome c. J Exp Clin Cancer Res. 2011;30:66. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 72] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 13. | Du SL, Pan H, Lu WY, Wang J, Wu J, Wang JY. Cyclic Arg-Gly-Asp peptide-labeled liposomes for targeting drug therapy of hepatic fibrosis in rats. J Pharmacol Exp Ther. 2007;322:560-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 86] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 14. | Schiffelers RM, Koning GA, ten Hagen TL, Fens MH, Schraa AJ, Janssen AP, Kok RJ, Molema G, Storm G. Anti-tumor efficacy of tumor vasculature-targeted liposomal doxorubicin. J Control Release. 2003;91:115-122. [PubMed] |
| 15. | Li Q, Yan Z, Li F, Lu W, Wang J, Guo C. The improving effects on hepatic fibrosis of interferon-γ liposomes targeted to hepatic stellate cells. Nanotechnology. 2012;23:265101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 16. | Hoefs JC, Shiffman ML, Goodman ZD, Kleiner DE, Dienstag JL, Stoddard AM. Rate of progression of hepatic fibrosis in patients with chronic hepatitis C: results from the HALT-C Trial. Gastroenterology. 2011;141:900-908.e1-2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Hemmann S, Graf J, Roderfeld M, Roeb E. Expression of MMPs and TIMPs in liver fibrosis - a systematic review with special emphasis on anti-fibrotic strategies. J Hepatol. 2007;46:955-975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 349] [Cited by in RCA: 392] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 18. | Ricchi P, Lanza AG, Ammirabile M, Costantini S, Cinque P, Spasiano A, Di Matola T, Di Costanzo GG, Pagano L, Prossomariti L. Hepatitis C virus distribution and clearance following interferon-monotherapy among thalassaemia major and intermedia patients. Br J Haematol. 2011;155:524-527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 19. | Fallowfield J, Hayes P. Pathogenesis and treatment of hepatic fibrosis: is cirrhosis reversible? Clin Med. 2011;11:179-183. [PubMed] |
| 20. | Lin M, Yang LY, Li WY, Peng YP, Zheng JK. Inhibition of the replication of hepatitis B virus in vitro by oxymatrine. J Int Med Res. 2009;37:1411-1419. [PubMed] |
| 21. | Lu LG, Zeng MD, Mao YM, Li JQ, Wan MB, Li CZ, Chen CW, Fu QC, Wang JY, She WM. Oxymatrine therapy for chronic hepatitis B: a randomized double-blind and placebo-controlled multi-center trial. World J Gastroenterol. 2003;9:2480-2483. [PubMed] |
| 22. | Shi GF, Li Q, Weng XH, Wu XH. [Effects of Oxymatrine on the expression of tissue inhibitor of metalloproteinase-1 and alpha-smooth muscle actin in the livers of rats with hepatic fibrosis]. Zhonghua Gan Zang Bing Zazhi. 2004;12:56. [PubMed] |
| 23. | Yang W, Zeng M, Fan Z, Mao Y, Song Y, Jia Y, Lu L, Chen CW, Peng YS, Zhu HY. [Prophylactic and therapeutic effect of oxymatrine on D-galactosamine-induced rat liver fibrosis]. Zhonghua Gan Zang Bing Zazhi. 2002;10:193-196. [PubMed] |
| 24. | Deng ZY, Li J, Jin Y, Chen XL, Lü XW. Effect of oxymatrine on the p38 mitogen-activated protein kinases signalling pathway in rats with CCl4 induced hepatic fibrosis. Chin Med J (. Engl). 2009;122:1449-1454. [PubMed] |
| 25. | Wu XL, Zeng WZ, Jiang MD, Qin JP, Xu H. Effect of Oxymatrine on the TGFbeta-Smad signaling pathway in rats with CCl4-induced hepatic fibrosis. World J Gastroenterol. 2008;14:2100-2105. [PubMed] |
| 26. | Shen BS, Song XW. [Effects of oxymatrine on serum cytokines and hepatic fibrotic indexes in patients with chronic hepatitis B]. Zhongguo Zhong Xi Yi Jie He Zazhi. 2008;28:17-19. [PubMed] |
| 27. | Xiang X, Wang G, Cai X, Li Y. Effect of oxymatrine on murine fulminant hepatitis and hepatocyte apoptosis. Chin Med J (. Engl). 2002;115:593-596. [PubMed] |
| 28. | Moreira RK. Hepatic stellate cells and liver fibrosis. Arch Pathol Lab Med. 2007;131:1728-1734. [PubMed] |
| 29. | Reeves HL, Friedman SL. Activation of hepatic stellate cells--a key issue in liver fibrosis. Front Biosci. 2002;7:d808-d826. [PubMed] |
| 30. | Kisseleva T, Brenner DA. Hepatic stellate cells and the reversal of fibrosis. J Gastroenterol Hepatol. 2006;21 Suppl 3:S84-S87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 208] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 31. | Gäbele E, Mühlbauer M, Dorn C, Weiss TS, Froh M, Schnabl B, Wiest R, Schölmerich J, Obermeier F, Hellerbrand C. Role of TLR9 in hepatic stellate cells and experimental liver fibrosis. Biochem Biophys Res Commun. 2008;376:271-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 111] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 32. | Shi GF, Li Q. Effects of oxymatrine on experimental hepatic fibrosis and its mechanism in vivo. World J Gastroenterol. 2005;11:268-271. [PubMed] |
| 33. | Li J, Fan R, Zhao S, Liu L, Guo S, Wu N, Zhang W, Chen P. Reactive Oxygen Species Released from Hypoxic Hepatocytes Regulates MMP-2 Expression in Hepatic Stellate Cells. Int J Mol Sci. 2011;12:2434-2447. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 41] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 34. | Hasegawa-Baba Y, Doi K. Changes in TIMP-1 and -2 expression in the early stage of porcine serum-induced liver fibrosis in rats. Exp Toxicol Pathol. 2011;63:357-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 35. | Chen MH, Wang QF, Chen LG, Shee JJ, Chen JC, Chen KY, Chen SH, Su JG, Liu YW. The inhibitory effect of Gynostemma pentaphyllum on MCP-1 and type I procollagen expression in rat hepatic stellate cells. J Ethnopharmacol. 2009;126:42-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 36. | Beljaars L, Molema G, Weert B, Bonnema H, Olinga P, Groothuis GM, Meijer DK, Poelstra K. Albumin modified with mannose 6-phosphate: A potential carrier for selective delivery of antifibrotic drugs to rat and human hepatic stellate cells. Hepatology. 1999;29:1486-1493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 109] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 37. | Beljaars L, Molema G, Schuppan D, Geerts A, De Bleser PJ, Weert B, Meijer DK, Poelstra K. Successful targeting to rat hepatic stellate cells using albumin modified with cyclic peptides that recognize the collagen type VI receptor. J Biol Chem. 2000;275:12743-12751. [PubMed] |