Published online May 21, 2012. doi: 10.3748/wjg.v18.i19.2364
Revised: March 18, 2011
Accepted: April 12, 2012
Published online: May 21, 2012
AIM: To determine the efficacy of external beam radiotherapy (EBRT), with or without intraluminal brachytherapy (ILBT), in patients with non-resected locally advanced hilar cholangiocarcinoma.
METHODS: We analyzed 64 patients with locally advanced hilar cholangiocarcinoma, including 25 who underwent resection (17 curative and 8 non-curative), 28 treated with radiotherapy, and 11 who received best supportive care (BSC). The radiotherapy group received EBRT (50 Gy, 30 fractions), with 11 receiving an additional 24 Gy (4 fractions) ILBT by iridium-192 with remote after loading. ILBT was performed using percutaneous transhepatic biliary drainage (PTBD) route. Uncovered metallic stents (UMS) were inserted into non-resected patients with obstructive jaundice, with the exception of four patients who received percutaneous transhepatic biliary drainage only. UMS were placed endoscopically or percutaneously, depending on the initial drainage procedure. The primary endpoints were patient death or stent occlusion. Survival time of patients in the radiotherapy group was compared with that of patients in the resection and BSC groups. Stent patency was compared in the radiotherapy and BSC groups.
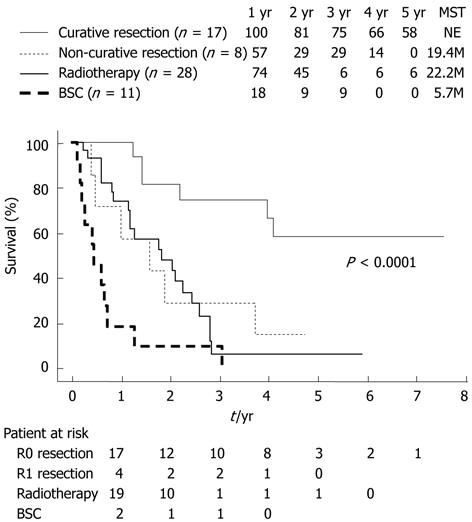
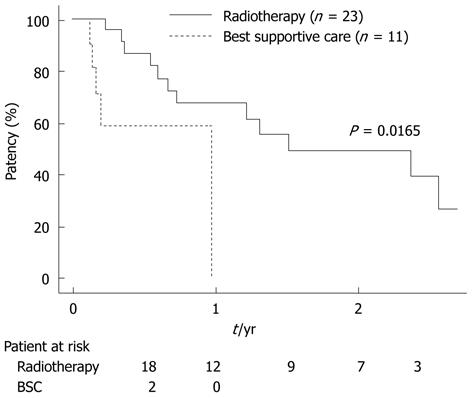
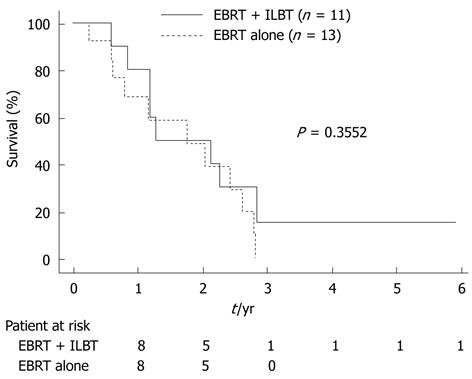
RESULTS: No statistically significant differences in patient characteristics were found among the resection, radiotherapy, and BSC groups. Three patients in the radiotherapy group and one in the BSC group did not receive UMS insertion but received PTBD alone; cholangitis occurred after endoscopic stenting, and patients were treated with PTBD. A total of 16 patients were administered additional systemic chemotherapy (5-fluorouracil-based regimen in 9, S-1 in 6, and gemcitabine in 1). Overall survival varied significantly among groups, with median survival times of 48.7 mo in the surgery group, 22.1 mo in the radiotherapy group, and 5.7 mo in the BSC group. Patients who underwent curative resection survived significantly longer than those who were not candidates for surgery (P = 0.0076). Cumulative survival in the radiotherapy group was significantly longer than in the BSC group (P = 0.0031), but did not differ significantly from those in the non-resection group. Furthermore, the median survival time of patients in the radiotherapy group who were considered for possible resection (excluding the seven patients who were not candidates for surgery due to comorbid disease or age) was 25.9 mo. Stent patency was evaluated only in the 24 patients who received a metallic stent. Stent patency was significantly longer in the radiotherapy than in the BSC group (P = 0.0165). Biliary drainage was not eliminated in any patient. To determine the efficacy of ILBT, we compared survival time and stent patency in the EBRT alone and EBRT plus ILBT groups. However, we found no significant difference in survival time between groups or for stent patencies. Hemorrhagic gastroduodenal ulcers were observed in 5 patients (17.9%), three in the EBRT plus ILBT group and two in the EBRT alone group. Ulcers occurred 5 mo, 7 mo, 8 mo, 16 mo, and 29 mo following radiotherapy. All patients required hospitalization, but blood transfusions were unnecessary. All 5 patients recovered following the administration of anti-ulcer medication.
CONCLUSION: Radiotherapy improved patient prognosis and the patency of uncovered metallic stents in patients with locally advanced hilar cholangiocarcinoma, but ILBT provided no additional benefits.
- Citation: Isayama H, Tsujino T, Nakai Y, Sasaki T, Nakagawa K, Yamashita H, Aoki T, Koike K. Clinical benefit of radiation therapy and metallic stenting for unresectable hilar cholangiocarcinoma. World J Gastroenterol 2012; 18(19): 2364-2370
- URL: https://www.wjgnet.com/1007-9327/full/v18/i19/2364.htm
- DOI: https://dx.doi.org/10.3748/wjg.v18.i19.2364
Hilar cholangiocarcinoma is a relatively rare, slow-growing, late-metastasizing tumor associated with poor patient prognosis[1-7]. The only known effective treatment is surgery, but only a small proportion of patients are suitable candidates. The operation is technically demanding and may be too invasive for high-risk elderly patients.
External beam radiotherapy (EBRT), with or without intraluminal brachytherapy (ILBT), is widely used to treat patients with hilar cholangiocarcinoma. A randomized trial comparing stenting alone with stenting plus radiotherapy showed that both procedures significantly prolonged patient survival and stent patency[8]. However, the relatively long survival time of untreated patients (median, 298 d) suggests that some of these patients may have been resectable. Thus, the effects of radiotherapy alone, and the benefits of ILBT, are not known.
Biliary stenting is a widely-accepted palliative procedure used to treat patients with unresectable hilar cholangiocarcinoma and obstructive jaundice. Patient prognosis, however, is poor, even with the absence of metastasis. A randomized trial showed that uncovered metallic stents remained patent longer than plastic stents in patients with hilar cholangiocarcinoma[9]. The primary cause of uncovered metallic stent occlusion is tumor ingrowth into the mesh[10]. Covered metallic stents prevent tumor ingrowth in patients with distal biliary obstruction and have a longer patency than uncovered metallic stents[11-16]. Although covered metallic stents are not feasible for treating hilar cancer patients because of the complex anatomy of the hilar duct, radiotherapy may be used to prevent tumor ingrowth after placement of an uncovered metallic stent[17].
The aims of this study were therefore twofold: to test the effects of radiotherapy, with or without ILBT, on the survival of hilar cholangiocarcinoma patients who did not undergo surgical resection; and to evaluate whether radiotherapy prolongs stent patency and thus improves patient quality of life.
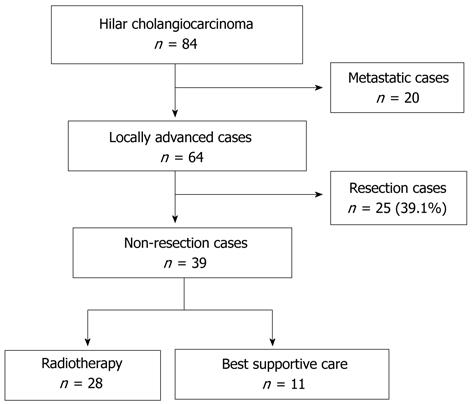
Between 1986 and 2008, 84 patients with hilar cholangiocarcinoma were admitted to the Department of Gastroenterology of Tokyo University Hospital. Of these 84 patients, 20 had metastatic and 64 had locally advanced disease (Figure 1 and Table 1). Of those with locally advanced tumors, 34 fulfilled our resectability criteria, and 25 underwent surgical resection. The remaining 9 patients were not candidates for surgery due to comorbid disease or advanced age. Bile duct cancer was diagnosed by pathological examination, clinical course, or imaging results. Cholangiocarcinoma staging was based on computed tomography, magnetic resonance cholangiopancreatography, or direct cholangiogram using endoscopic retrograde cholangiopancreatography or percutaneous transhepatic biliary drainage (PTBD). In addition, Bismuth’s classification was applied[6].
| Patient group | ||||
| Radiotherapy | BSC | Resection | Pvalue | |
| Cases | 28 | 11 | 25 | |
| Gender (M/F) | 14/14 | 4/7 | 20/5 | |
| Mean age (yr) | 70.1 ± 9.7 (52-86) | 74.0 ± 9.0 (61-90) | 67.0 ± 9.7 (55-78) | |
| Reason for non-resection | > 0.9999 | |||
| Tumor factor | 21 | 9 | - | |
| Patient factor | 7 | 2 | - | |
| Performance status | ||||
| 0 | 12 | 3 | 17 | |
| 1 | 15 | 6 | 8 | |
| 2 | 1 | 2 | 0 | |
| TNM stage | 0.4953 | |||
| 1a | 0 | 0 | 7 | |
| 1b | 2 | 2 | 1 | |
| 2a | 1 | 0 | 3 | |
| 2b | 1 | 1 | 8 | |
| 3 | 24 | 8 | 6 | |
| Bismuth classification | 0.2332 | |||
| 1 | 3 | 0 | 2 | |
| 2 | 6 | 3 | 2 | |
| 3 | 7 | 6 | 10 | |
| 4 | 12 | 2 | 11 | |
The resectability of each tumor was determined after consultation with the hepato-biliary surgeon. Surgical contraindications included: invasion of the celiac and super mesenteric arteries, biliary invasion of the third branch of the future remnant liver, and invasion of the both hepatic artery and portal vein of the future remnant liver.
All patients with locally advanced tumors underwent endoscopic or percutaneous biliary drainage (Table 2). Prophylactic antibiotics were administered intravenously prior to the endoscopic procedure and for 3 d afterwards. An Amsterdam-type plastic stent (7 or 8.5 Fr; Flexima stent, Microvasive, Boston Scientific, Natick, MA, United States) or a nasobiliary drainage tube (Cook Medical Company) was inserted into patients. PTBD using 8 Fr balloon catheters was performed under ultrasonographic and fluoroscopic guidance. Additional stents were inserted or PTBD was performed if jaundice did not improve.
| Patient group | |||
| Radiotherapy | BSC | Pvalue | |
| Cases | 28 | 11 | |
| Biliary metallic stenting | 24 | 10 | |
| Drainage area | 0.0630 | ||
| Bilateral | 14 | 2 | |
| Unilateral | 10 | 8 | |
| Insertion route | 0.0339 | ||
| Endoscopic | 4 | 6 | |
| Percutaneous | 20 | 4 | |
| PTBD only | 3 | 1 | > 0.9999 |
| No need for drainage | 1 | 0 | > 0.9999 |
| Anti-cancer therapy | |||
| Radiotherapy | 28 | - | |
| Extra corporeal (50 Gy) | 28 | - | |
| Intra bile duct (24 Gy) | 11 | - | |
| Additional chemotherapy | 16 | - | |
| Gemcitabine | 1 | - | |
| S-1 | 6 | - | |
| 5FU-based regimen | 9 | - | |
Radiotherapy was recommended for patients whose jaundice did not improve and who were not candidates for surgery. A total of 28 patients agreed to undergo EBRT (54 Gy, 30 fractions); of these, 11 also underwent percutaneous ILBT (24 Gy, 4 fractions) using a high-dose iridium-192 remote after loading system (Table 2). A metallic stent was inserted into the 11 patients who refused EBRT after their jaundice improved; these patients constituted the best supportive care (BSC) group.
After radiotherapy, a metallic stent was inserted endoscopically or percutaneously, depending on the initial drainage procedure. Some patients received bilateral metallic stents because multiple insertion routes were necessary for internal brachytherapy and unilateral stents produced insufficient drainage.
Patient symptoms were assessed and blood tests were performed at monthly intervals in the outpatient clinic. Computed tomography scans were taken every 6 mo (every 2 mo during chemotherapy), with additional scans taken when patients were symptomatic or showed an increase in hepatic-biliary enzymes and bilirubin. Patients received chemotherapy every 2 mo.
The primary endpoints were patient death or stent occlusion through November 2008. Stents were considered patent if the jaundice did not worsen, with or without cholangitis. Patient survival time was measured from the date of diagnosis to the date of death, and stent patency was measured from placement to occlusion or to patient death if the stent remained patent. Survival time of patients in the radiotherapy group was compared with that of patients in the resection and BSC groups. Stent patency was compared in the radiotherapy and BSC groups.
Cumulative patient survival and stent patency were analyzed using the Kaplan-Meier method and compared using the log rank test. Patients whose stents were not obstructed were excluded from stent patency analysis. The Mann-Whitney U-test was used to compare quantitative variables, and Fisher’s exact test was used to analyze qualitative variables. All analyses were performed using StatView 5.0 software (SAS Institute Inc., Cary, NC, United States).
Overall survival varied significantly among groups, with median survival times of 48.7 mo in the surgery group, 22.1 mo in the radiotherapy group, and 5.7 mo in the BSC group (Figure 2). Patients who underwent curative resection survived significantly longer than those who were not candidates for surgery (P = 0.0076). Cumulative survival in the radiotherapy group was significantly longer than in the BSC group (P = 0.0031), but did not differ significantly from those in the non-resection group (Figure 2). Furthermore, the median survival time of patients in the radiotherapy group who were considered for possible resection (excluding the seven patients who were not candidates for surgery due to comorbid disease or age) was 25.9 mo.
Stent patency was evaluated only in the 24 patients who received a metallic stent (Table 3). Stent patency was significantly longer in the radiotherapy than in the BSC group (P = 0.0165; Figure 3). Biliary drainage was not eliminated in any patient.
To determine the efficacy of ILBT, we compared survival time and stent patency in the EBRT alone and EBRT plus ILBT groups. However, we found no significant difference in survival time between groups (Figure 4) or for stent patencies.
Hemorrhagic gastroduodenal ulcers were observed in 5 patients (17.9%), three in the EBRT plus ILBT group and two in the EBRT alone group. Ulcers occurred 5 mo, 7 mo, 8 mo, 16 mo, and 29 mo following radiotherapy. All patients required hospitalization, but blood transfusions were unnecessary. All 5 patients recovered following the administration of anti-ulcer medication.
Surgical resection is the only known cure for hilar cholangiocarcinoma. The surgery is highly invasive and recommended for only a small percentage of patients; thus, alternative treatments are necessary. Our results suggest that radiotherapy prolongs survival in patients who cannot undergo resection. Survival was significantly longer in patients who received radiotherapy than in those who received BSC, but was similar to that of patients who underwent non-curative resection.
Radiotherapy has been shown to improve survival times of patients with locally advanced hilar cholangiocarcinoma who cannot undergo curative resection. For example, a randomized trial reported that median survival times were 12.9 mo in patients who received both EBRT and ILBT and 9.3 mo in patients did not receive radiotherapy[8]. A second study found that median survival in patients who received chemoradiotherapy (EBRT plus 5-fluorouracil) was 22 mo, but that study did not include an untreated control group[18]. A retrospective analysis found that the median survival time in patients with extrahepatic cholangiocarcinoma who received EBRT plus ILBT was 9 mo, compared with 5 mo in patients treated with EBRT alone[19]. This wide range in survival times may reflect differences in patient backgrounds. Most reports did not describe the percentage of patients with resectable tumors, the reasons that they were not resected, or the resection criteria; thus, it is difficult to generalize from these results. In contrast, we have stated the inclusion criteria for non-surgical treatment. Moreover, in our radiotherapy group, the median survival of non-resected patients considered for possible resection was 25.9 mo, longer than previously reported survival times.
We observed no significant differences between the radiotherapy and BSC groups in patient background, including the reasons for not undergoing resection and performance score. We expected that selection bias may have resulted in longer median survival of the radiotherapy than of the BSC group, but we found no significant difference in background between these two groups. Our results therefore provide additional evidence suggesting that radiotherapy increases survival time in patients with locally advanced hilar cholangiocarcinoma.
Although surgery is the only known cure for cholangiocarcinoma, we found that median survival of the radiotherapy group did not differ significantly from that of the non-curative resection group. This finding supports the survival benefits of radiotherapy for patients with locally advanced hilar cholangiocarcinoma. Thus, radiotherapy is a treatment option for patients in poor condition or those with highly invasive tumors.
Biliary stenting is a widely accepted palliative treatment for non-resected patients with biliary obstruction, with uncovered metallic stents maintaining patency longer than plastic stents[9]. The primary cause of obstruction in patients who receive an uncovered metallic stent is tumor ingrowth into the stent mesh. In a previous randomized trial, we prevented distal biliary obstruction by inhibiting tumor ingrowth and found that a covered metallic stent was patent longer than an uncovered stent[11]. However, covered metallic stents cannot be used in patients with hilar obstruction, and it is necessary to develop strategies to prevent tumor ingrowth of hilar lesions. Stents remained patent significantly longer in the radiotherapy group than in the BSC group (stenting alone); suggesting that local tumor control using radiotherapy prolongs stent patency. Previous studies of the efficacy of metallic stents in the treatment of hilar cholangiocarcinoma did not use radiotherapy, and studies investigating the survival benefits of radiotherapy have not reported results of metallic stenting. Thus, no evidence was available to determine the efficacy of radiotherapy in improving stent patency.
Multiple laser-cut-type Nitinol stents were found to have a mean patency of 150 d[20], with Wallstents having a median patency of 169 d[21]. A comparison of the patency of metallic stents inserted endoscopically and percutaneously showed that the median stent patency of these two groups was 9.8 mo and 11.0 mo, respectively[22]. In comparison, we found that median and mean stent patency in our radiotherapy group were 557 d and 604 d, respectively. The length of time the stents remained patent suggests that radiotherapy had a significant effect on patency.
We found that the addition of ILBT to EBRT did not improve patient survival or stent patency. Stent patency is a surrogate indicator of local anti-tumor effectiveness, because the main cause of stent occlusion is tumor ingrowth through the stent mesh. ILBT requires an additional treatment period and multiple PTBD insertions. Since it did not improve patient survival or stent patency, we do not recommend this additional treatment. A retrospective study of 31 patients found that, although additional ILBT improved the 2-year survival rate, it had no effect on cumulative survival time[20]. No randomized studies to date have compared the effect on survival of radiotherapy with and without ILBT. Thus, the efficacy of additional ILBT remains unclear.
Photodynamic therapy is a promising local anti-tumor therapy for patients with non-resectable hilar cholangiocarcinoma, with median survival times of patients who did and not receive photodynamic therapy of 493 d and 98 d, respectively, a difference that was statistically significant[23]. Although photodynamic therapy can be used to treat local tumors extending along the bile duct, it cannot be used to treat large tumors that have invaded other organs, and it cannot be used to treat loco-regional lymph node metastasis because it is performed in the bile duct. The combination of photodynamic therapy with EBRT may be a more feasible treatment option.
The major limitation of this study was its retrospective design. Moreover, despite all included patients having unresectable, locally advanced tumors, the characteristics of the radiation and BSC groups differed. Since there were no selection criteria for the BSC group, the differences in outcomes between this group and the radiation group may be due not only to the effects of radiation, but to patient condition as well.
In conclusion, radiotherapy may improve the prognosis of patients with non-resected, locally advanced hilar cholangiocarcinoma and may increase the patency of uncovered metallic stents. ILBT provides no benefit for these patients.
Hilar cholangiocarcinoma is a relatively rare, slow-growing and late-metastasizing tumor associated with poor patient prognosis. Surgery is the only known curative treatment, but it may be technically difficult and too invasive for high-risk elderly patients. The treatment of patients with unresectable tumors has not been established.
Biliary stenting and radiotherapy, including external beam radiotherapy (EBRT) with or without intraluminal brachytherapy (ILBT), are widely used to treat patients with locally advanced hilar cholangiocarcinoma. These treatments, however, are not well established, although a recent randomized study showed good results for photodynamic therapy.
Several recent studies have shown the efficacy of radiation therapy in patients with locally advanced hilar cholangiocarcinoma. In many of those patients, however, the tumors may have been resectable. The authors clarified the reasons for non-resection of the patients and compared outcomes with resected patients. The authors found that radiation therapy effectively prolonged patient survival and stent patency. They also showed that additional ILBT did not affect survival or stent patency.
EBRT is indicated for patients with locally advanced hilar cholangiocarcinoma, with similar survival times as patients who underwent non-curative resection. Patients who undergo uncovered metallic stent placement should be considered for EBRT to prolong stent patency and survival.
EBRT is a conventional radiation method, and ILBT is performed with iridium wire through a PTBD catheter using an after loading system. A metallic stent is a braided metallic wire or tubular nitinol cut with a laser. Metallic stents have self-expandability and are mounted onto a thin delivery system (6-9Fr). This type of stent, however, tends to be occluded by tumor ingrowth into the stent mesh.
As stated by the authors, the limitation of the study is that it is a retrospective study and that the groups are not the results of a randomization but the consequence of different situations such not being suitable for surgery. In any case, the paper is a well presented study on a very difficult subject and is worthy of publication.
Peer reviewer: Antonio Basoli, Professor, General Surgery “Paride Stefanini”, Università di Roma-Sapienza, Viale del Policlinico 155, Roma 00161, Italy
S- Editor Cheng JX L- Editor Rutherford A E- Editor Zhang DN
| 1. | Klatskin G. Adenocarcinoma of the hepatic duct at its bifurcation within the porta hepatis. An unusual tumor with distinctive clinical and pathological features. Am J Med. 1965;38:241-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 577] [Cited by in RCA: 499] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 2. | Bismuth H, Nakache R, Diamond T. Management strategies in resection for hilar cholangiocarcinoma. Ann Surg. 1992;215:31-38. [PubMed] |
| 3. | Inouye AA, Whelan TJ. Carcinoma of the extrahepatic bile ducts: a ten year experience in Hawaii. Am J Surg. 1978;136:90-95. [PubMed] |
| 4. | Blumgart LH, Hadjis NS, Benjamin IS, Beazley R. Surgical approaches to cholangiocarcinoma at confluence of hepatic ducts. Lancet. 1984;1:66-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 177] [Cited by in RCA: 152] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 5. | Langer JC, Langer B, Taylor BR, Zeldin R, Cummings B. Carcinoma of the extrahepatic bile ducts: results of an aggressive surgical approach. Surgery. 1985;98:752-759. [PubMed] |
| 6. | Bismuth H, Castaing D, Traynor O. Resection or palliation: priority of surgery in the treatment of hilar cancer. World J Surg. 1988;12:39-47. [PubMed] |
| 7. | Seyama Y, Kubota K, Sano K, Noie T, Takayama T, Kosuge T, Makuuchi M. Long-term outcome of extended hemihepatectomy for hilar bile duct cancer with no mortality and high survival rate. Ann Surg. 2003;238:73-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 8. | Válek V, Kysela P, Kala Z, Kiss I, Tomásek J, Petera J. Brachytherapy and percutaneous stenting in the treatment of cholangiocarcinoma: a prospective randomised study. Eur J Radiol. 2007;62:175-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 42] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Wagner HJ, Knyrim K, Vakil N, Klose KJ. Plastic endoprostheses versus metal stents in the palliative treatment of malignant hilar biliary obstruction. A prospective and randomized trial. Endoscopy. 1993;25:213-218. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 234] [Cited by in RCA: 234] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 10. | De Palma GD, Pezzullo A, Rega M, Persico M, Patrone F, Mastantuono L, Persico G. Unilateral placement of metallic stents for malignant hilar obstruction: a prospective study. Gastrointest Endosc. 2003;58:50-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 130] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 11. | Isayama H, Komatsu Y, Tsujino T, Sasahira N, Hirano K, Toda N, Nakai Y, Yamamoto N, Tada M, Yoshida H. A prospective randomised study of "covered" versus "uncovered" diamond stents for the management of distal malignant biliary obstruction. Gut. 2004;53:729-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 460] [Cited by in RCA: 458] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 12. | Isayama H, Komatsu Y, Tsujino T, Yoshida H, Tada M, Shiratori Y, Kawabe T, Omata M. Polyurethane-covered metal stent for management of distal malignant biliary obstruction. Gastrointest Endosc. 2002;55:366-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 123] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 13. | Nakai Y, Isayama H, Komatsu Y, Tsujino T, Toda N, Sasahira N, Yamamoto N, Hirano K, Tada M, Yoshida H. Efficacy and safety of the covered Wallstent in patients with distal malignant biliary obstruction. Gastrointest Endosc. 2005;62:742-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 111] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 14. | Isayama H, Kawabe T, Nakai Y, Ito Y, Togawa O, Kogure H, Yashima Y, Yagioka H, Matsubara S, Sasaki T. Management of distal malignant biliary obstruction with the ComVi stent, a new covered metallic stent. Surg Endosc. 2010;24:131-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 52] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 15. | Kubota Y, Mukai H, Nakaizumi A, Tanaka K, Okabe Y, Sakagami T, Kitano M, Mitsufuji S, Shirasaka D, Kikuchi E. Covered Wallstent for palliation of malignant common bile duct stricture: prospective multicenter evaluation. Digest Endosc. 2005;17:218-223. [DOI] [Full Text] |
| 16. | Kahaleh M, Tokar J, Conaway MR, Brock A, Le T, Adams RB, Yeaton P. Efficacy and complications of covered Wallstents in malignant distal biliary obstruction. Gastrointest Endosc. 2005;61:528-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 128] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 17. | Bowling TE, Galbraith SM, Hatfield AR, Solano J, Spittle MF. A retrospective comparison of endoscopic stenting alone with stenting and radiotherapy in non-resectable cholangiocarcinoma. Gut. 1996;39:852-855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 82] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 18. | Deodato F, Clemente G, Mattiucci GC, Macchia G, Costamagna G, Giuliante F, Smaniotto D, Luzi S, Valentini V, Mutignani M. Chemoradiation and brachytherapy in biliary tract carcinoma: long-term results. Int J Radiat Oncol Biol Phys. 2006;64:483-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 41] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 19. | Shin HS, Seong J, Kim WC, Lee HS, Moon SR, Lee IJ, Lee KK, Park KR, Suh CO, Kim GE. Combination of external beam irradiation and high-dose-rate intraluminal brachytherapy for inoperable carcinoma of the extrahepatic bile ducts. Int J Radiat Oncol Biol Phys. 2003;57:105-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 57] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 20. | Kawamoto H, Tsutsumi K, Harada R, Fujii M, Kato H, Hirao K, Kurihara N, Nakanishi T, Mizuno O, Ishida E. Endoscopic deployment of multiple JOSTENT SelfX is effective and safe in treatment of malignant hilar biliary strictures. Clin Gastroenterol Hepatol. 2008;6:401-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 38] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 21. | Cheng JL, Bruno MJ, Bergman JJ, Rauws EA, Tytgat GN, Huibregtse K. Endoscopic palliation of patients with biliary obstruction caused by nonresectable hilar cholangiocarcinoma: efficacy of self-expandable metallic Wallstents. Gastrointest Endosc. 2002;56:33-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 157] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 22. | Paik WH, Park YS, Hwang JH, Lee SH, Yoon CJ, Kang SG, Lee JK, Ryu JK, Kim YT, Yoon YB. Palliative treatment with self-expandable metallic stents in patients with advanced type III or IV hilar cholangiocarcinoma: a percutaneous versus endoscopic approach. Gastrointest Endosc. 2009;69:55-62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 188] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 23. | Ortner ME, Caca K, Berr F, Liebetruth J, Mansmann U, Huster D, Voderholzer W, Schachschal G, Mössner J, Lochs H. Successful photodynamic therapy for nonresectable cholangiocarcinoma: a randomized prospective study. Gastroenterology. 2003;125:1355-1363. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 383] [Article Influence: 17.4] [Reference Citation Analysis (0)] |