Published online Nov 21, 2011. doi: 10.3748/wjg.v17.i43.4831
Revised: April 16, 2011
Accepted: April 23, 2011
Published online: November 21, 2011
A large cell neuroendocrine carcinoma (LCNEC) of the stomach is very rare. A 76-year-old Japanese man was admitted to our hospital because of epigastralgia and nausea. Endoscopy revealed 2 large tumors in the stomach. He did not have multiple endocrine neoplasia type I or Zollinger-Ellison syndrome. Imaging modalities, including computed tomography and magnetic resonance imaging, revealed no other tumors. Gastrectomy, cholecystectomy, and lymph node dissection were performed. The resected stomach had 2 tumors: one was an antral ulcerated type 3 tumor measuring 5 cm x 5 cm, and the other was a polypoid type 1 tumor measuring 6 cm x 6 cm x 3 cm in the fundus. Microscopically, the antral ulcerated tumor was a well differentiated adenocarcinoma with deep invasion. The fundus polypoid tumor was a LCNEC, being composed of malignant large cells arranged in trabecular and nested patterns. The tumor cells were large and the nuclei were vesicular. Nucleoli were frequently present, and there were many mitotic figures, apoptotic bodies, and necrotic areas. Much lymphovascular permeation was seen. Seven out of 29 dissected lymph nodes showed metastatic foci; 6 were from the LCNEC and 1 from the adenocarcinoma. Many intravascular tumor emboli of LCNEC were seen in the peritoneum around the lymph nodes. Mucins were present in the adenocarcinoma but not in the LCNEC. Immunohistochemically, the LCNEC tumor cells were positive for pancytokeratins, synaptophysin (50% positive), chromogranin A (10% positive), Ki-67 (90% labeled), and platelet-derived growth factor-α (80% positive). They were negative for KIT, p53, CD56, and neuron-specific enolase. The non-cancerous stomach showed a normal number of endocrine cells. The patient is now treated with adjuvant chemotherapy.
- Citation: Terada T, Maruo H. Simultaneous large cell neuroendocrine carcinoma and adenocarcinoma of the stomach. World J Gastroenterol 2011; 17(43): 4831-4834
- URL: https://www.wjgnet.com/1007-9327/full/v17/i43/4831.htm
- DOI: https://dx.doi.org/10.3748/wjg.v17.i43.4831
Large cell neuroendocrine carcinoma (LCNEC) of the stomach is very rare, and has been reported mainly in Japan[1-4]. In the World Health Organization (WHO) Blue Book[5], gastric neuroendocrine tumors were classified into well differentiated endocrine neoplasms (carcinoid) and poorly differentiated endocrine neoplasms (small cell carcinoma). In the latter section of the Blue Book, LCNEC of the stomach was defined as follows: LCNEC is a malignant neoplasm composed of large cells having organoid, nesting, trabecular, rosette-like and palisading patterns that suggest endocrine differentiation, and in which the last can be confirmed by immunohistochemistry and electron microscopy[5]. In contrast to small cell carcinoma, LCNEC has abundant cytoplasm, more vesicular nuclei, and prominent nucleoli[5]. Although single LCNEC[1-4] and composite LCNEC and adenocarcinoma[6] of the stomach have been reported, there has been no English literature of simultaneous and distinct LCNEC and adenocarcinoma of the stomach, to the best of the author’s knowledge.
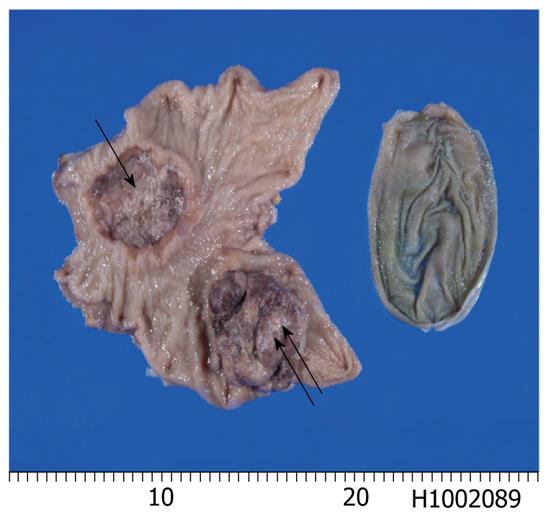
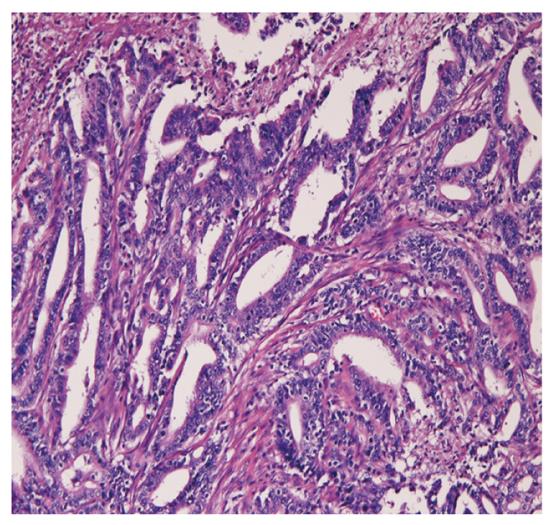
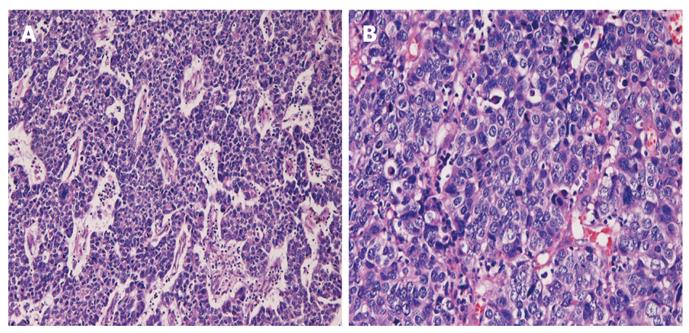
A 76-year-old Japanese man presented with epigastralgia and nausea, and attended our hospital. Upper gastric endoscopy revealed 2 large tumors in the stomach, and a biopsy from one tumor showed well differentiated tubular adenocarcinoma. He did not have multiple endocrine neoplasia type I or Zollinger-Ellison syndrome. Preoperative imaging techniques, including computed tomography and magnetic resonance imaging, revealed no other tumors. Gastrectomy, cholecystectomy, and lymph node dissection were performed. The resected stomach showed 2 large tumors (Figure 1). One was an antral ulcerated type 3 tumor measuring 5 cm × 5 cm, and the other was a polypoid type 1 tumor measuring 6 cm × 6 cm × 3 cm located in the fundus (Figure 1). Microscopically, the antral ulcerated tumor was a well differentiated adenocarcinoma invading the subserosa (Figure 2). The fundus polypoid tumor was composed of malignant large cells arranged in trabecular and nested patterns (Figure 3A). No tubular formations were recognized. The tumor cells were large and the nuclei were vesicular (Figure 3B). Nucleoli were frequently identified, and there were many mitotic figures, apoptotic bodies, and small necrotic areas (Figure 3B). The tumor was located in the mucosa and submucosa, and invasion into proper muscle and subserosa was not noted. Much lymphovascular invasion was seen. Seven out of 29 dissected lymph nodes showed metastatic foci; 6 were from the LCNEC and 1 was from the adenocarcinoma. Many intravascular tumor emboli of the LCNEC were seen in the peritoneum around the lymph nodes. The non-cancerous stomach showed chronic atrophic gastritis.
The adenocarcinoma was positive for mucins, while the LCNEC was negative for mucins, as revealed by mucin stains including d-periodic acid-Schiff, Alcian blue, and mucicarmine.
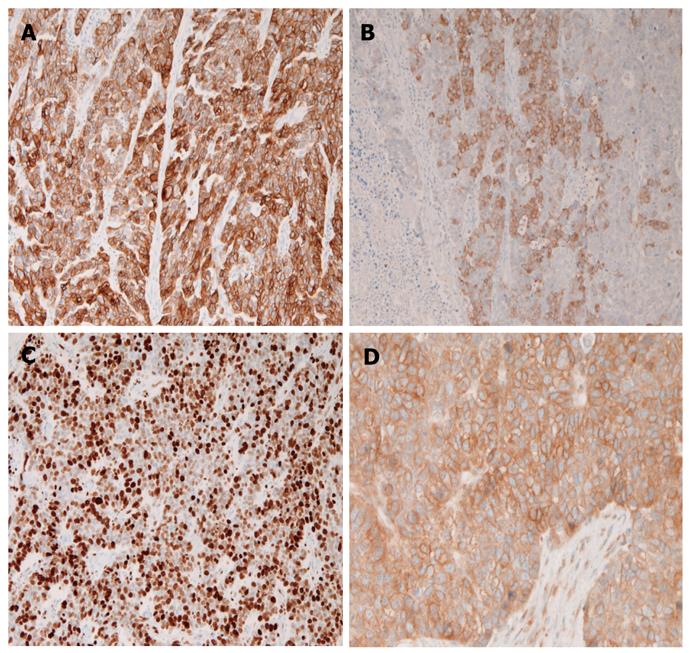
An immunohistochemical study was performed with the Dako Envision method (Dako Corp., Glostrup, Denmark) as previously described[7,8]. The tumor cells of the polypoid tumor were positive for pancytokeratins (AE1/3 and CAM 5.2) (Figure 4A), synaptophysin (50% positive) (Figure 4B), chromogranin A (10% positive), Ki-67 (90% labeled) (Figure 4C), and platelet-derived growth factor-α (PDGFRA) (80% positive) (Figure 4D). The cells were negative for KIT, p53, CD56, and neuron-specific enolase. The non-cancerous stomach showed a normal number of endocrine cells.
The gallbladder showed no significant changes. No peritoneal dissemination was recognized. The patient is now treated with adjuvant chemotherapy.
LCNEC of the stomach is not a well recognized entity. In the WHO Blue Book[5], neuroendocrine tumors of the stomach are classified into well differentiated tumors (carcinoids) and poorly differentiated neuroendocrine carcinoma (small cell carcinoma), and LCNEC is not included as a clinicopathological entity. However, as in the present case, a non-carcinoid and non-small neuroendocrine carcinoma may be present[1-6]. Therefore, LCNEC of the stomach should be listed as a neuroendocrine carcinoma in the WHO Blue Book.
The presentation of the current case is different from a carcinoid tumor because it showed significant atypia, mitosis, apoptotic cells, and necrosis, all of which are not seen in carcinoids. The present case is apparently different from small cell carcinoma because of the atypical features of the tumor cells.
The present case is not an adenocarcinoma with endocrine differentiation. Endocrine cells of the latter are a very minor component, accounting for less than 20%[2]. In the present case, the neuroendocrine component accounted for 50% of LCNEC cells, thus confirming that the current case is not an adenocarcinoma with neuroendocrine differentiation but a LCNEC.
The frequency of gastric LCNEC is unclear. Jiang et al[2]
examined 2835 cases of gastric carcinoma and found 42 cases (1.5%) of LCNEC and 44 cases (1.6%) of adenocarcinoma with endocrine differentiation. The frequency is relatively high. In general, pathologists do not perform an immunohistochemical study of surgical specimens of gastric cancers. It is recommended that pathologists examine at least the immunohistochemistry of neuroendocrine markers in poorly differentiated carcinoma showing neuroendocrinoid histology.
The biological behavior of gastric LCNEC is significantly more aggressive than that of gastric adenocarcinoma[2]. Adenocarcinoma with an endocrine component is more aggressive than ordinary adenocarcinoma[2]. The authors think that the prognosis of the present case is poor because the adenocarcinoma is advanced and lymph node metastasis was found in 6 of 29 lymph nodes. In addition, there were many intravascular tumor emboli of LCNEC in the perilymphatic peritoneum.
The present tumor is not a composite or combined tumor, but a separate tumor. There is one report of combined or collision LCNEC and adenocarcinoma[6]. In the present case, the LCNEC and adenocarcinoma were remote from each other, thus were simultaneous. Such a simultaneous LCNEC and adenocarcinoma of the stomach has not been reported in the English literature, to the best of the author’s knowledge. It is very interesting that the present LCNEC expressed PDGFRA, but not KIT. Tumors expressing PDGFRA have been not examined. The PDGFRA may be related to neuroendocrine features of a LCNEC, as in gastrointestinal stromal tumors. Thus, a stomach LCNEC can be included in a group of tumors expressing PDGFRA.
Peer reviewer: Jian-Zhong Zhang, Professor, Department of Pathology and Laboratory Medicine, Beijing 306 Hospital, 9 North Anxiang Road, PO Box 9720, Beijing 100101, China
S- Editor Tian L L- Editor Cant MR E- Editor Zhang DN
| 1. | Boo YJ, Park SS, Kim JH, Mok YJ, Kim SJ, Kim CS. Gastric neuroendocrine carcinoma: clinicopathologic review and immunohistochemical study of E-cadherin and Ki-67 as prognostic markers. J Surg Oncol. 2007;95:110-117. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 2. | Jiang SX, Mikami T, Umezawa A, Saegusa M, Kameya T, Okayasu I. Gastric large cell neuroendocrine carcinomas: a distinct clinicopathologic entity. Am J Surg Pathol. 2006;30:945-953. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 80] [Cited by in F6Publishing: 86] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 3. | Matsui K, Jin XM, Kitagawa M, Miwa A. Clinicopathologic features of neuroendocrine carcinomas of the stomach: appraisal of small cell and large cell variants. Arch Pathol Lab Med. 1998;122:1010-1017. [PubMed] [Cited in This Article: ] |
| 4. | Iso Y, Tagaya N, Nemoto T, Kita J, Sawada T, Kubota K. Incarceration of a large cell neuroendocrine carcinoma arising from the proximal stomach with an organoaxial gastric volvulus through an esophageal hiatal hernia: report of a case. Surg Today. 2009;39:148-152. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 5. | Capella C, Solcia E, Sobin LH, Arnold R. Endocrine tumours of the stomach. Hamilton SR and Aaltonen LA, editors. World Health Organization Classification of Tumours. Pathology and genetics of tumors of the digestive system. Lyon: IARC Press 2000; 53-57. [Cited in This Article: ] |
| 6. | Jang KY, Moon WS, Lee H, Kim CY, Park HS. Gastric collision tumor of large cell neuroendocrine carcinoma and adenocarcinoma--a case report. Pathol Res Pract. 2010;206:387-390. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 15] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 7. | Terada T, Kawaguchi M, Furukawa K, Sekido Y, Osamura Y. Minute mixed ductal-endocrine carcinoma of the pancreas with predominant intraductal growth. Pathol Int. 2002;52:740-746. [PubMed] [Cited in This Article: ] |
| 8. | Terada T, Kawaguchi M. Primary clear cell adenocarcinoma of the peritoneum. Tohoku J Exp Med. 2005;206:271-275. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 107] [Cited by in F6Publishing: 119] [Article Influence: 6.3] [Reference Citation Analysis (0)] |