INTRODUCTION
The rapid increase in the prevalence of obesity is a major global health problem. Its associated complications are burdened by an increased risk of death by 20%-40% in overweight individuals and by 2-3-fold in obese individuals compared to normal controls, even though the strength of the association between body mass index and heart failure events declines with age[1]. Obesity is a known risk factor for non-alcoholic fatty liver disease (NAFLD)[2], hypertension, stroke, gallbladder disease, osteoarthritis, obstructive sleep apnea, and other breathing problems as well and some forms of cancer (breast, colorectal, endometrial and kidney). Type 2 diabetes (T2D) and obesity, now collectively referred to as “diabesity”, are interrelated, in that obesity is known to exacerbate the pathology of T2D and greater than 60% of diabetics are obese. Low grade chronic inflammation, strictly linked to overweight/obesity, causes insulin resistance (IR) that interacts with other complex mechanisms such as hypercholesterolemia, smoking, hypertension, hyperglycemia, type A behavioral patterns, hemostatic factors, hereditary differences in such diverse aspects as lipoprotein structure and that of their associated receptors, homocysteine processing/metabolism, and high levels of lipoprotein(a) to increase the risk of coronary heart disease (CHD). CHD is present not only in nondiabetic and normotensive obese adult subjects[3], but also in obese children[4]. IR is a condition in which normal amounts of insulin are inadequate to produce a normal insulin response from fat, muscle and liver cells. Indeed, lipolysis, which is normally inhibited by insulin, is overstimulated in insulin-resistant states leading to an increase in free fatty acid (FFA) flux[5]. IR in muscle reduces glucose uptake whereas IR in liver reduces glucose storage, both effects serving to elevate blood glucose. High plasma levels of insulin and glucose due to IR often leads to metabolic syndrome (MS) and T2D. Plasma FFAs from white adipose tissue lipolysis have been shown to be the major contributor to triglyceride accumulation[6] observed in NAFLD, further expression of MS, which ranges from simple fatty liver (FL) through the more severe form, non alcoholic steatohepatitis (NASH) to cryptogenic cirrhosis. Sharing the same mechanisms of NASH, FL is no longer considered completely benign, accordingly to recent data[7]. Activation of the endoplasmic reticulum (ER) by stress has been reported in most models of hepatic steatosis in rodents and its contribution to hepatic fat deposition has been recently documented, with lipogenesis being the main metabolic pathway affected. ER stress-related activation, observed in adipose tissue of obese humans[8], could have metabolic consequences and participate in fat deposition in the liver. Activation of ER could directly induce an insulin-resistant state in adipocytes. Indeed, it has been shown that activation of the ER stress sensor kinase/endonuclease inositol-requiring protein 1 (IRE1), a component of the unfolded protein response (UPR) could stimulate c-Jun amino-terminal kinase [JNKs or stress-activated protein kinases (SAPKs)][9], which, by phosphorylating serine residues of insulin substrate receptor 1, is a key player in the development of IR[10]. The IRE1/box binding protein 1, a branch of the ER stress signaling pathway, has been recently shown to regulate and be regulated by innate immune signaling pathways in both the presence and absence of ER stress[11]. Disruption of ER homeostasis has been observed in liver and adipose tissue of humans with NAFLD and/or obesity. Importantly, the signaling pathways activated by disruption of ER homeostasis, the UPR, has been linked to inflammation and apoptosis, lipid biosynthesis, insulin effects, all of which are involved in the initiation/evolution of NAFLD. The ER is a crucial organelle for cellular homeostasis, in which the synthesis and the post-translational modifications of membrane and secreted proteins take place, as well as the synthesis of lipids and cholesterol for membranes formation. However, the ER quality control system can be compromised under a variety of conditions such as accumulation of unfolded protein, alteration of calcium homeostasis or disruption of the redox state. The UPR activates JNKs[9,12]. Up-to-date results provide evidence that heat shock proteins protect cells from ER stress-induced apoptosis. Obesity and associated disorders constitute a serious problem, for example NAFLD can lead to hepatocarcinoma, and the contribution of adipose tissue to metabolic homeostasis has become a focus of interest. Adipose tissue secretes FFAs and hormones, known as adipokines, and thus seems to play a major role in the development of NAFLD. Apoptotic cell death is a prominent feature in NASH. Indeed, toxic FFAs can activate the intrinsic apoptosis pathway in hepatocytes via c-JNK. JNK activates the proapoptotic protein Bim, resulting in Bax activation[13] and enhanced apoptosis, termed “lipoapoptosis”.
JUN AMINO-TERMINAL KINASES
Jun N-terminal kinases (JNKs), also named SAPKs, are one of 3 members of the mitogen-activated protein kinase (MAPK) superfamily, which also includes the extracellular signal-regulated kinases (ERKs) or classical MAPKs and the p38 MAPK. JNKs bind and phosphorylate c-Jun on Ser63 and Ser73 within its transcriptional activation domain. MAPK kinases (MKK) are responsive to stress stimuli, mainly inflammatory signals, but also to a lesser extent, to ultraviolet irradiation, heat and osmotic shock, and are involved in apoptosis and T cell differentiation. This latter immunological aspect should not be overlooked.
JNKs consist of 10 isoforms derived from 3 genes: JNK1 (4 isoforms), JNK2 (4 isoforms), and JNK3 (2 isoforms). JNK1 and JNK2 are found in all cells of every tissue. JNK3 is found mainly in the brain, but is also found in the heart and the testes. JNK1 is involved in apoptosis, neurodegeneration, cell differentiation and proliferation, inflammatory conditions and cytokine production mediated by activation protein-1 (AP-1) such as regulated upon activation, normal T-cell expressed, and secreted cytokine, interleukin-8 and granulocyte-macrophage colony-stimulating factor. Recently, JNK1 has been found to regulate Jun protein turnover by phosphorylation and activation of the ubiquitin ligase Itch (polyubiquitination marks proteins for degradation by the proteasome). JNKs can associate with scaffold proteins, JNK-interacting proteins as well as their upstream Jun N-terminal kinase kinase 1 and Jun N-terminal kinase kinase 2 following their activation. JNK, by phosphorylation, modifies the activity of numerous proteins that reside at the mitochondria or act in the nucleus. Thus, JNK activity regulates several important cellular functions. One way this activation may occur is through disruption of the conformation of sensitive protein phosphatase enzymes; specific phosphatases normally inhibit the activity of JNK itself and the activity of proteins linked to JNK activation. The JNK proteins lead to varied and seemingly contradictory cellular responses. In particular, JNKs have been reported to have a role in the induction of apoptosis, but have also been implicated in enhancing cell survival and proliferation. The opposing roles of JNKs have been attributed to the observation that JNKs activate different substrates based on a specific stimulus, cell type or temporal aspects[14].
JNK signaling pathway
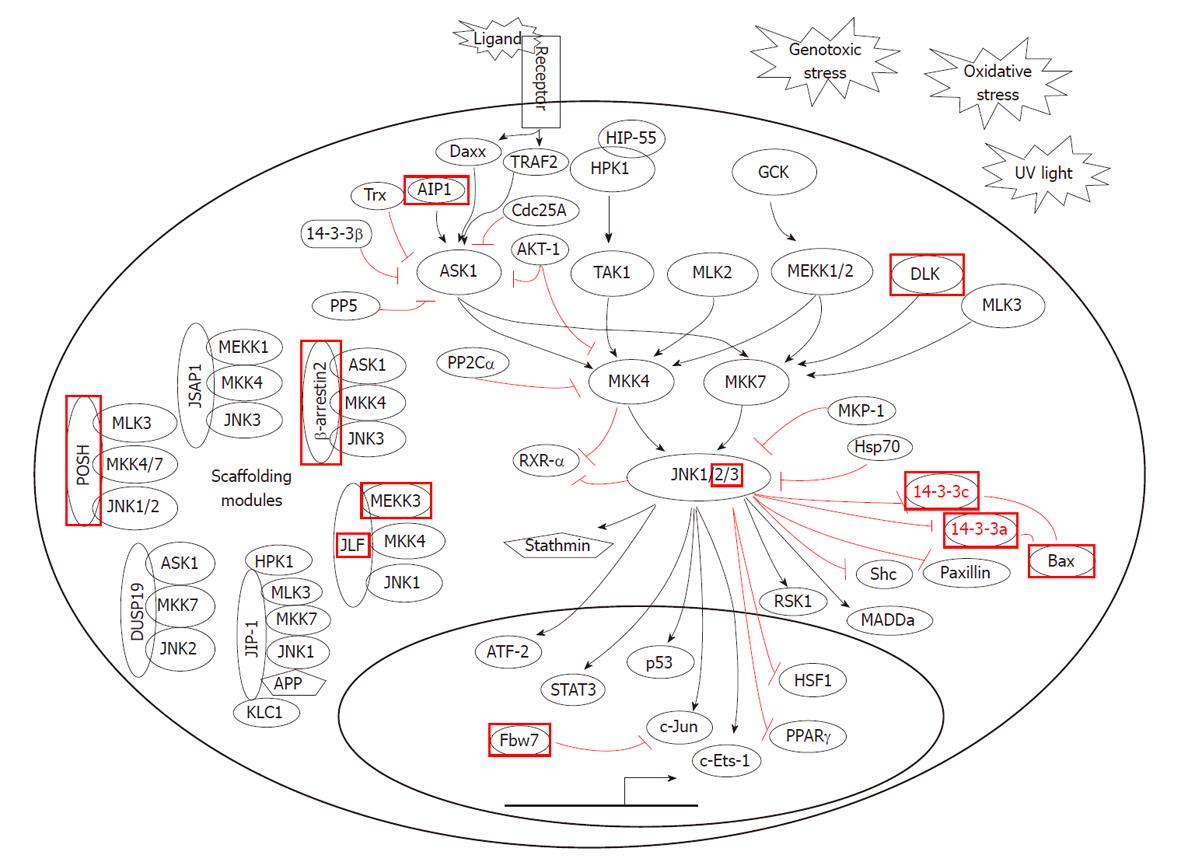
JNKs are activated by MAPK kinases such as MKK-4, MKK-6 and MKK-7 (Figure 1). These kinases are in turn activated by the MAP3 kinases, such as apoptosis signal-regulating kinase 1 (ASK1) also known as mitogen-activated protein kinase kinase kinase 5 (MAP3K5), mixed lineage kinases (MLKs) (MLK1, MLK2 and MLK3), MAP/ERK (extracellular signal-regulated kinase) kinase kinase 1 (MEKK1), MEKK4 and transforming growth factor (TGF)-β-activated kinase 1 (TAK1). JNK MAP3 kinase pathways are activated by MAP4 kinases that link to a variety of cell receptors which sense stress and inflammation, including death receptors (Fas), inflammatory cytokine receptors of tumor necrosis factor alpha (TNF-α) and TGF-β, G-protein-coupled receptors (GPCRs) and antigen receptors. Signals are communicated to JNK pathway MAP4 kinases by tyrosine kinase receptor associated adapter and effector molecules and/or by G-protein mediated signaling. GPCRs signal the JNK pathway through trimeric G-proteins to monomeric p21RhoGTPases, Ras-related C3 botulinum toxin substrate 1 (Rac1) and cell division control protein 42 homolog (CDC42). ASK1 links to stress receptors such as TNF receptor (TNFR) and Fas, and is activated by reactive oxygen species (ROS)-mediated dissociation of thioredoxin, binding to TNFR associated factor 2 (TRAF2) or death domain-associated protein (DAXX) and oligomerization. ASK1 activates the JNK MAP2Ks, MKK4 (SEK1) and MKK6 (MKK3/MAPKK6). The JNK MAP3 kinases: TAK1, MEKK1 and MLK3 are activated by the tyrosine kinase (TK) receptor-activated MAP4 kinase, hematopoietic progenitor kinase 1 (HPK-1). HPK-1 associates with TK receptors through adaptor proteins, such as CT10-regulated kinase (Crk), Crk-like (CrkL) and growth factor receptor-bound 2 (Grb2). Activation of HPK1 requires multiple phosphorylation events including autophosphorylation and protein kinase phosphorylation by protein kinase D1 (PKD1). The JNK MAP3 kinases, MEKK1, MEKK4 and MLK3 are also activated by the p21Rho-GTPases, Ras-related C3 botulinum toxin substrate 1 also known as Rac1 and Cdc42 through the MAP4K, P21-activated kinase- 1 (PAK-1). This links the JNK pathways to a wide variety of GPCR, integrin and receptor pathways. In addition to participating in the stress response, the MAPKs c-Jun N-terminal Kinases JNK1 and JNK2 regulate the proliferation of normal and neoplastic cells. JNKs contribute to these processes largely by phosphorylating c-Jun and thus contributing to the activation of the AP-1 complex. Furthermore, JNKs control entry into mitosis. It has been observed that JNK activity and phosphorylation of c-Jun become elevated during the G/M transition of the cell cycle in immortalized fibroblasts and ovarian granulosa cells. Pharmacological inhibition of JNK causes a profound cell cycle arrest at the G/M transition in both cell types. This effect is specific as it occurs with 2 distinct small molecular compounds. Inactivation of JNK prior to mitosis prevents expression of aurora B and phosphorylation of histone-H3 at Ser 10. Silencing of JNK1 and 2 causes a similar effect, whereas overexpression of JNK1 and 2 causes the opposite effect. Inhibition of JNK delays activation of Cdc2 and prevents downregulation of cyclin B1, whereas Ras controls the activation of MAPKs. Authors have recently observed that in certain cells, the small guanosine triphosphate (GTP)-binding proteins Rac1 and Cdc42 but not Rho regulate the activity of JNKs[15]. Furthermore, because Rac1 and Cdc42 but not Rho, bind and activate a P21-activated kinase 1 (Pak1), it has been suggested that Pak1 is the most upstream component of the pathway linking these GTPases to JNK. However, in mammalian cells, Rho1p, a Rho homologue, and RhoA directly interact with a number of proteins, including kinases related to protein kinase C. Exploring the ability of Ras and Rho-related GTP-binding proteins to activate MAPK or JNK in a variety of cell lines, it was found that in the human kidney epithelial cell line, 293T, Cdc42 and all Rho proteins, RhoA, RhoB, and RhoC, but not Rac or Ras can induce activation of JNK. Furthermore, other researchers provided evidence that signaling from Rho proteins to JNK in 293T cells does not involve Pak1[16]. c-JNK activity is abnormally elevated in obesity. Furthermore, an absence of JNK1 results in decreased adiposity, significantly improved insulin sensitivity and enhanced insulin receptor signaling capacity[17]. As previously mentioned, elevated FFAs and hepatocyte lipoapoptosis are the main features of NAFLD. However, the mechanism by which FFAs mediate lipoapoptosis is unclear. Recently, data have indicated that saturated FFAs induce JNK-dependent hepatocyte lipoapoptosis by activating the pro-apoptotic Bcl-2 family Bim and Bax-mediated apoptosis, which triggers the mitochondrial apoptotic pathway[18]. Additional support for involvement of JNK1 overactivation in conditions associated with IR and MS has been provided[19]. A positive correlation was found between the expression intensity of JNK1 and IR[20], and JNK1 contributes to the development of liver fibrosis by inducing chronic inflammation as ascertained in a mouse NASH model[21]. Methionine-choline-deficient feeding causes NASH coincident with the activation of c-JNK and caspase-12 in a murine model[22].
Figure 1 Jun N-terminal kinase signaling pathway.
ERK: Extracellular signal-regulated kinase; MAPK: Mitogen-activated protein kinase; MKK: MAPK kinase; MEKK: MAPK/ERK kinase kinase; MLK: Mixed lineage kinase; JNK: Jun N-terminal kinase; ASK: Apoptosis signal-regulating kinase; HSP: Heat shock protein; RSK: p90 ribosomal S6 kinase; HPK: Hematopoietic progenitor kinase; PPAR: Peroxisome proliferator-activated receptor; DLK: Dual leucine zipper-bearing kinase; RXR: Retinoid X receptor; ATF: Activating transcription factor; STAT: Signal transducers and activators of transcription; MADD: Mitogen-activated kinase activating death domain protein; TRAF: Tumor necrosis factor receptor-associated factor; MKP: MAPK phosphatase; TAK: Transforming growth factor-activated kinase; KLC: Kinesin light chain; DAXX: Death domain-associated protein.
Further data indicated that the increased oxidative stress and its associated JNK activation, as well as an imbalance in pro- and anti-apoptotic proteins in the Bcl-2 family all contribute to marked hepatocyte apoptosis in a rat NASH model[23]. Examining fat biopsy samples from obese insulin-resistant nondiabetic individuals, UPR activation in subcutaneous adipose tissue was demonstrated, with JNK being a link between obesity, IR, and inflammation[8]. ER stress activates the proteolytic cleavage of the lipogenic transcription factor sterol regulatory element binding protein-1c leading to the induction of lipogenic enzyme expression. A role for X box-binding protein 1, an ER stress-activated transcription factor, has also recently emerged. ER stress, by inhibiting apoB100 secretion, has associated with impaired very low density lipoprotein (VLDL) secretion. In rodents, treatment with molecular or chemical chaperones that reduce ER stress markers have demonstrated effectiveness in the treatment of hepatic steatosis[24].
INSULIN RESISTANCE
SAPK/JNKs are activated by inflammatory cytokines, and JNK signaling is involved in IR and β-cell secretory function and survival. An up-to-date study suggested that FFAs stimulate functional autophagy of β cells, possibly through the RNA-dependent protein kinase (PKR)-JNK1 pathway independent of the ER or oxidative stress[25].
Post-transcriptional modifications altering activity of insulin signaling molecules are the most proposed mechanism for inhibition of the insulin pathway. Various kinases including stress activated protein kinase, c-JNK, and protein kinase C (PKC) can phosphorylate insulin receptor substrate (IRS) 1-2 at specific serine and threonine residues, leading to inhibition of insulin signaling[26]. A high fat diet is an established cause of systemic and adipose tissue IR. Activated JNK is a major contributor to FFA-induced cellular IR, and TNF-α is an autocrine/paracrine downstream effector of activated JNK that can also mediate IR[27]. TNF-α is over-expressed in adipose tissue of obese rodents and humans, and its concentration is reduced after weight loss. TNF-α inhibits insulin signaling in the liver by mechanisms which include the activation of serine kinases such as JNK-1 and induction of suppressor of cytokine signaling proteins[28]. An important consequence of IR in adipose tissue is to reduce the anti-lipolytic effect of insulin that in turn leads to elevated plasma FFA in obese and diabetic patients. Released FFAs from adipose tissue is taken up by liver and muscle cells. In the liver, FFAs induce gluconeogenesis and VLDL overproduction. Increased FFAs, especially metabolites such as acyl-CoAs, ceramides, and diacylglycerol, have been shown to inhibit insulin signaling by activating protein kinases such as PKC, JNK, and the inhibitor of nuclear factor-κB (IKK-β)[29].
Chronic high glucose concentrations and leptin induce IL-1β secretion from pancreatic islets, an event that is possibly promotes β-cell dysfunction and death. A recent study provided evidence that chronically elevated concentrations of leptin and glucose induced β-cell apoptosis through activation of the JNK pathway in human islets and in insulinoma (INS 832/13) cells. JNK inhibition by the dominant inhibitor JNK-binding domain of IB1/JIP-1 (JNKi) reduced JNK activity and apoptosis induced by leptin and glucose. Exposure of human islets to leptin and high glucose concentrations led to a decrease of glucose-induced insulin secretion, which was partly restored by JNKi. An interplay between the JNK cascade and the caspase 1/IL-1β-converting enzyme in human islets has been found. The caspase 1 gene, which contains a potential activating protein-1 binding site, is upregulated in pancreatic sections and in isolated islets from T2D patients. Similarly, cultured human islets exposed to high glucose- and leptin-induced caspase 1 and JNK inhibition prevents this upregulation. Therefore, JNK inhibition may protect β-cells from the deleterious effects of high glucose and leptin in diabetes[30].
Obesity is closely associated with IR and is established as a leading risk factor for T2D, yet the molecular mechanisms of this association are poorly understood. JNKs can interfere with insulin action in cultured cells and are activated by inflammatory cytokines and FFAs, molecules that have been implicated in the development of T2D. As previously highlighted, it has been shown that JNK activity is abnormally elevated in obesity. Furthermore, an absence of JNK1 results in decreased adiposity, significantly improved insulin sensitivity and enhanced insulin receptor signaling capacity in 2 different models of mouse obesity. Thus, JNK is a crucial mediator of obesity and IR and a potential therapeutic target[31]. Chronic oxidative stress results in decreased responsiveness to insulin, eventually leading to T2D and CHD. Activation of the JNK signaling pathway can mediate many of the effects of stress on IR through inhibitory phosphorylation of IRS-1. In contrast, exercise, which acutely increases oxidative stress in muscle, improves insulin sensitivity and glucose tolerance in patients with T2D. Authors used a cellular model of insulin-resistant muscle to induce either chronic or acute oxidative stress and investigate their contrasting effects on insulin and JNK signaling. Chronic oxidative stress resulted in increased levels of phosphorylated (activated) JNK in the cytoplasm, whereas acute oxidative stress led to redistribution of JNK-specific phosphatase MKP7 from the nucleus into the cytoplasm, a reduction in cytoplasmic phospho-JNK, and concurrent accumulation of phospho-JNK in the nucleus. Acute oxidative stress restored normal insulin sensitivity and glucose uptake in insulin-resistant muscle cells, and this effect was dependent on MKP7[32].
Finally, it is likely that JNK activity modulates pancreatic islet function and/or survival in numerous ways. First, there is convincing evidence for the involvement of JNK in islet cell inflammation and death mediated by cytokines[33]. Second, JNK activation may generate a state of β-cell dysfunction and defective insulin production, thereby contributing to the development of overt diabetes[34]. Third, administration of SP600125, a synthetic inhibitor of JNK, results in improved glucose-stimulated insulin production in isolated islets in the db/db model of obesity and diabetes[35]. Hence, there is a strong possibility that JNK may integrate defects in insulin secretion with peripheral IR in T2D through its actions in pancreatic β-cells as well as peripheral sites of insulin action. If this is the case, it is also likely that JNK may be important in the pathogenesis of type 1 diabetes, and recent studies have provided evidence to support a role for the JNK-2 isoform in this disease[36].
LIPOLYTIC MACHINERY, ADIPOCYTES AND NAFLD
Several lines of evidence implicate an inadequate response to lipid storage/catabolism of cellular fat stores as being important in NAFLD. Subcutaneous adipose tissue (AT) is composed mostly of small, differentiated adipocytes that absorb circulating FFAs due to their insulin-sensitivity. They form triglycerides (lipogenesis) and store them in cellular lipid droplets (LD) or lipid bodies (surrounded by a monolayer of lipase-regulating proteins) until FFAs are needed during fasting. They also secrete adiponectin, which by opposing hepatic lipogenesis and stimulating long chain fatty acid β-oxidation, protects the liver from harmful effects of lipid accumulation, such as IR[37]. In MS, failure of subcutaneous AT to store energy leads to swollen adipocytes that are stressed and de-differentiated. They continually release FFAs from triglycerides (lipolysis). Lipolysis is the biochemical pathway responsible for the catabolism of triacylglycerol (TAG) stored in cellular LD. The hydrolytic cleavage of TAG generates FFAs, which are subsequently used as energy substrates, essential precursors for lipid and membrane synthesis, or mediators in cell signaling processes. Consistent with its central importance in lipid and energy homeostasis, lipolysis mostly occurs in white and brown adipose tissue. Over the last few years, important enzymes and regulatory protein factors involved in lipolysis have been identified. These include an essential TAG hydrolase named adipose triglyceride lipase (ATGL) [a patatin-like phospholipase domain-containing protein A2 (PNPLA2)], the ATGL activator comparative gene identification-58 (an α/β hydrolase containing protein 5), and the ATGL inhibitor G0/G1 switch gene 2. ATGL catalyzes the first step in adipocyte and muscle triglyceride hydrolysis. Together with the established hormone-sensitive lipase (lipase E) and monoglyceride lipase, these proteins constitute the basic “lipolytic machinery”. Additionally, a large number of hormonal signaling pathways and lipid droplet-associated protein factors regulate substrate access and the activity of the “lipolysome”[38]. Activation of β-(AR) in adipocytes triggers acute changes in metabolism that can alter patterns of gene expression. A recent work examined the mechanisms by which activation of hormone sensitive lipase induces expression of inflammatory cytokines in adipocytes in vivo and model adipocytes in vitro. β3-adrenergic receptor (AR) activation in mice triggered expression of inflammatory genes CCL2, IL-6, and PAI-1, as well as ER stress markers GRP78 and CHOP[39]. Recent findings suggest that genetic variants in PNPLA3 predispose towards hepatic steatosis and, in the context of other environmental stressors, progression to irreversible liver failure. PNPLA3 is predominantly expressed in human liver and adipose tissue, possesses both lipolytic and lipogenic activity in vitro, and is localized on the surface of lipid droplets in hepatocytes. The 148M mutant protein has reduced lipolytic activity, with attendant increased cellular triglycerides[40], only recently confirmed[41]. Studies in animal models of NAFLD demonstrate that inhibition of acyl-coenzyme A:diacylglycerol acyltransferase (DGAT)-1, the enzymes that catalyze the final step in triglyceride synthesis, results in improvement in hepatic steatosis and insulin sensitivity. Researchers recently confirmed that hepatic-specific inhibition of DGAT-1 with antisense oligonucleotides improved hepatic steatosis in obese, diabetic mice but, unexpectedly, exacerbated injury and fibrosis in that model of progressive NAFLD. When hepatocyte triglyceride synthesis was inhibited, FFA accumulated in the liver, leading to induction of fatty acid oxidizing systems that increased hepatic oxidative stress and liver damage. These findings suggest that the ability to synthesize triglycerides may, in fact, be protective in obesity[42]. This is a key point. MEK1/2 inhibition significantly increased both cellular and microsomal triglycerides mass, and mRNA levels for DGAT-1 and DGAT-2. In contrast to ERK, modulation of the phosphatidylinositol 3-kinases pathway or inhibition of the p38 MAP kinase, had no effect on lipoprotein density profile[43]. The biogenesis of LD induced by serum depends on group IVA phospholipase A(2) [cPLA(2)α/GIVA PLA(2)], a regulatory enzyme that releases arachidonic acid for production of prostaglandins and leukotrienes. Recent data suggest that cPLA(2)α regulates the transport of tight junction and adherens junction proteins through Golgi cell-cell contacts in confluent endothelial cells. Expression of specific activators of different MAP kinases show that phosphorylation of cPLA(2)α at Ser-505 is due to JNK. This was confirmed by pharmacological inhibition and expression of a dominant-negative form of the upstream activator MEKK1. LD biogenesis was accompanied by increased synthesis of ceramide 1-phosphate. Over-expression of its synthesizing enzyme ceramide kinase increased phosphorylation of cPLA(2)α at Ser-505 and the formation of LD, and its downregulation blocked the phosphorylation of cPLA(2)α and LD biogenesis. These results demonstrate that LD biogenesis induced by serum is regulated by JNK and ceramide kinase[44].
The effect of glucose and palmitate on the phosphorylation of proteins is associated with cell growth and survival. Fresh results suggest that short-term changes in MAPK and AKT signaling pathways, and c-fos and c-JNK expressions induced by glucose are abolished by palmitate through phosphatidylinositol 3-kinase inhibition via ceramide synthesis[45].
Activation of β-AR in mouse adipocytes triggered expression of inflammatory genes CCL2, IL-6, and PAI-1, as well as ER stress markers GRP78 and CHOP. Pharmacological inhibition of hormone sensible lipase (HSL) blocked induction of inflammatory genes, but not ER stress markers. Promoting intracellular accumulation of FFAs in 3T3-L1 adipocytes increased the expression of inflammatory cytokines, whereas inhibiting ceramide synthesis partly blocked PAI-1 expression, but not IL-6. Induction of inflammatory markers in vivo and in vitro was preceded by phosphorylation of p38 and JNK, and inhibition of HSL prevented activation of these kinases. Together, these results demonstrate that FFAs liberated by HSL activate p38 and JNK, and p38 mediates pro-inflammatory cytokine expression in adipose tissue[39].
As previously emphasized, accumulation of lipid metabolites within non-adipose tissues can induce chronic inflammation by promoting macrophage infiltration and activation. Oxidized and glycated lipoproteins, FFAs, free cholesterol, triacylglycerols, diacylglycerols and mainly ceramides have long been known to induce cellular dysfunction through their pro-inflammatory and pro-apoptotic properties. Emerging evidence suggests that macrophage activation by lipid metabolites and further modulation by lipid signaling represents a common pathogenic mechanism underlying lipotoxicity in atherosclerosis, obesity-associated IR and inflammatory diseases related to MS such as NAFLD and chronic kidney disease.
The sphingolipid ceramide is an important second signaling molecule that regulates diverse signaling pathways involving apoptosis, cell senescence, the cell cycle and differentiation. For the most part, effects of ceramide are antagonistic to growth and survival. Interestingly, ceramide and the pro-growth agonist, diacylglycerol (DAG) appear to be regulated simultaneously but in opposite directions in the sphingomyelin cycle. While ceramide stimulates signal transduction pathways that are associated with cell death or at least are inhibitory to cell growth (SAPK), DAG activates the classical and novel isoforms of the PKC family. These PKC isoforms are associated with cell growth and cell survival. Furthermore, DAG activation of PKC stimulates other signal transduction pathways that support cell proliferation, e.g., MAPK pathways. Thus, ceramide and DAG generation may serve to monitor cellular homeostasis by inducing pro-death or pro-growth pathways, respectively. The production of ceramide is emerging as a fixture in programmed cell death. Ceramide levels are elevated in response to diverse stress challenges including treatment with pro-death ligands such as TNF-α, chemotherapeutic drug treatment or irradiation. Consistent with this notion, ceramide itself is a potent apoptogenic agent. Ceramide activates c-JNK and thus affects its transcription pathways. Ceramide activates protein phosphatases such as protein phosphatase 1 and PP2A. Ceramide activation of protein phosphatases has been shown to promote inactivation of a number of pro-growth cellular regulators including the kinases PKCα and Akt, Bcl-2 and the retinoblastoma protein. A new role has recently emerged for ceramide in the regulation of protein synthesis. Ceramide-induced activation of PKR, a protein kinase important in anti-viral host defense mechanisms and recently implicated in cellular stress pathways, results in the inhibition of protein synthesis as a prelude to cell death[46].
ENDOPLASMIC RETICULUM, CHAPERONES AND LONGEVITY
Heat shock proteins (HSPs) have proven to be effective tools for extending invertebrate lifespan, and in C. elegans daf-2 mutants, longevity resulting from loss of insulin/insulin-like signals is at least partly dependent upon elevated HSP expression. In mice, inhibition of the orthologous growth hormone/insulin-like growth factor I(GH/IGF-I) pathway has similar pro-longevity effects. A recent study, however, suggested that loss of GH/IGF-Isignaling in long-lived mice did not broadly elevate HSP expression, but in fact decreased HSP expression in many tissue types, such as liver and kidney. The contribution of chaperones to the longevity of long-lived mice with altered GH/IGF-I signals may therefore differ from that described in C. elegans daf-2 mutants. This result, in combination with other recent findings, underscores the possibility that systemic overexpression of chaperones will have dissimilar effects on longevity in vertebrate and invertebrate systems[47].
MS AND CARDIOVASCULAR DISEASE RISK
MS is a constellation of common metabolic disorders that is strictly linked to CHD. It is now commonly accepted that low-grade chronic inflammation associated with obesity induces IR in the liver. Low-grade chronic inflammation is characterized by the production of abnormal cytokines and adipokines such as IL-6, TNF-α, IL-1, leptin and resistin. These factors inhibit insulin signaling in hepatocytes by activating SOCS proteins, several kinases such as JNK, IKK-β and PKC and protein tyrosine phosphatases such as PTP1B and PTEN that in turn impair insulin signaling at the insulin receptor and IRS level. Hepatic IR in turn causes impaired suppression of glucose production by insulin in hepatocytes leading to hyperglycemia. An important and early complication of hepatic IR is the induction of hepatic VLDL production, via changes in the rate of apoB synthesis and degradation and de novo lipogenesis, or increased FFA flux from adipose tissue into the liver. IR also stimulates the production of C-reactive protein (CRP) and plasminogen activator inhibitor-1 (PAI-1), both markers of an inflammatory state. All these subsequent metabolic abnormalities related to hepatic IR have been shown to directly or indirectly promote atherosclerosis. Hyperglycemia induces a series of alterations including endothelial dysfunction, cellular proliferation, changes in extracellular matrix conformation and impairment of low density lipoprotein (LDL) receptor-mediated uptake, decreasing the in vivo clearance of LDL. Small dense LDLs associated with high circulating VLDL levels have higher affinity for the intimal proteoglycans leading, to the penetration of more LDL particles into the arterial wall. CRP can also accelerate atherosclerosis by increasing the expression of PAI-1 and adhesion molecules in endothelial cells, inhibiting nitric oxide formation and increasing LDL uptake into macrophages.
Recently it has been shown that a small molecule pan-JNK inhibitor, dosed orally and compared to rimonabant and rosiglitazone, significantly impacted parameters such as adiposity, glucose levels, and insulin sensitization without any effect on liver enzymes, thus establishing the role of JNK as a useful target for metabolic syndrome linked to the pre-diabetic state[48]. A JNK1 specific antisense oligonucleotide was studied in ob/ob and diet-induced obese mouse models. Profound improvement in insulin sensitivity, glucose levels, plasma cholesterol level, and adiposity without a negative impact on liver function was observed. Decreased body weight and lowered adiposity were attributed to increased food combustion/metabolic rate and decreased lipogenesis[49].
Describing the common mechanisms by which lipid derivatives, through modulation of macrophage function, promote plaque instability in the arterial wall, impair insulin responsiveness and contribute to inflammatory liver and discussing the molecular mechanism of lipid activation of pro-inflammatory pathways [JNK, nuclear factor (NF)κB], the key roles played by the proliferator-activated receptor and liver X receptor α, nuclear receptors-lipid sensors that link lipid metabolism and inflammation, should be emphasized[50]. Atherosclerosis begins as local inflammation of artery walls at sites of disturbed blood flow. JNK is thought to be among the major regulators of flow-dependent inflammatory gene expression in endothelial cells in atherosclerosis. Researchers have shown that JNK activation by both onset of laminar flow and long-term oscillatory flow is matrix-specific, with enhanced activation on fibronectin compared to basement membrane protein or collagen. Flow-induced JNK activation on fibronectin requires new integrin ligation and requires both MKK4 and p21-activated kinase. In vivo, JNK activation at sites of early atherogenesis correlates with the deposition of fibronectin. Inhibiting p21-activated kinase reduces JNK activation in atheroprone regions of the vasculature in vivo. These results identify JNK as a matrix-specific, flow-activated inflammatory event. These data elucidate a network of matrix-specific pathways that determine inflammatory events in response to fluid shear[51].
Visceral AT is known to confer a significantly higher risk of T2D and CHD. Epicardial AT has been shown to be related to cardiovascular disease and myocardial function. Epicardial AT expresses an inflammatory profile of proteins. Authors studied key mediators of the NFκB and c-JNK pathways in paired epicardial and gluteofemoral (thigh) AT from CHD and investigated circulating endotoxin levels in CHD and control subjects. Serums and AT biopsies (epicardial and thigh) were obtained from CHD and non-CHD patients. Inflammation was assessed in tissue and serum samples through western blot, real-time PCR, ELISAs, and activity studies. Western blotting showed epicardial AT had significantly higher NFκB, inhibitory-κB kinase (IKK)-γ, IKK-β, and JNK-1 and -2 compared with thigh AT. Epicardial mRNA data showed strong correlations between CD-68 (again the impaired immunity function) and toll-like receptor-2, toll-like receptor-4, and TNF-α. Circulating endotoxin was elevated in patients with CHD compared with matched controls. Epicardial AT from patients with CHD shows increased NFκB, IKK-β, and JNK expression compared with both CHD thigh AT and non-CHD epicardial AT, suggesting a depot-specific as well as a disease-linked response to inflammation[52].
METHODS TO DETECT JNK
The detection of protein kinases is possible in biological liquids such as blood serum or cell lysate. Sandwich ELISAs for detecting phosphoproteins have commonly been used to quantify kinase function and can be performed in 2 configurations. In the first configuration, polyclonal antibodies directed against the structural part of the protein and away from the phosphorylation site (panprotein) are coated onto the bottom of a microwell plate. A cell lysate containing the phosphorylated target protein is added to the well, allowed to bind and the excess lysate is removed by washing. A monoclonal antibody of either mouse or rabbit origin, specific for the phosphorylated form of the protein, is added followed by an enzyme-labeled secondary antibody specific for the monoclonal antibody species. A chromagen is added and the color is quantified spectrophotometrically. In the second configuration, the capture antibody is directed against the phosphor-antibody and the detection antibody is an antibody directed against the panprotein. The latter configuration is sometimes preferred as the amount of phosphor-protein present may be small compared to the total amount of the panprotein.
In this situation, the large amount of non-phosphorylated panprotein can outcompete the phosphorylated protein for binding to the microwells. This decreases the overall sensitivity of the assay for the phosphor-protein. Using a phosphor-specific capture antibody enriches for the desired target and significantly increases the sensitivity of the assay. Phospho-ELISAs can be used to assess kinase activity in cell lysates or, alternatively, to screen drug candidates targeting a purified kinase. Quantitative measurement of protein phosphorylation has become essential for the development of kinase-inhibiting drugs aimed at therapy of various metabolic diseases. Since kinases are a major source of drug targeting, complex but reliable assay technologies that quantify phosphorylation will continue to be in demand. Biochemical assays that rely on antibodies for assay function are limited by the availability of phosphor-specific antibodies with high affinity and specificity. While many phosphospecific antibodies exist, most are unsuitable for use in quantitative assays due to poor sensitivity or nonspecificity. Alternative methods such as mobility shift, IMAP (a variation of fluorescence polarization that employs nanoparticles bearing immobilized trivalent metal co-ordination complexes that bind specifically to phosphate groups), IQ (using as signal a peptide comprised of an aminoacid sequence recognized by the desired kinase that is synthesized with a fluorophore end-label) and light-speed assays (the signal is generated from a polystyrene microsphere that is coated with a modified fluorescent polyelectrolyte) do not rely on antibodies and allow assessment of targets for which no suitable antibodies exist. As a result, these formats will find wider use in the future[53].
CONCLUSION
As repeatedly emphasized, inflammation is the common mechanism underlying obesity, MS, NAFLD[54], longevity, CHD and perhaps some cancers. The chicken-and-egg dilemma of IR being cause or effect of inflammation is unsolved at present. Further studies are warranted of anti-inflammatory drugs such as aspirin, anti IL-6 receptors, immune-modulators (calcineurin inhibitors)[55], substances enhancing the expression of HSPs (which protect cells from ER stress-induced apoptosis), and anti-JNKs in well-designed trials to try to minimize the high impact of these illnesses, and the different expressions of the diseases, on the whole population[56].
Peer reviewer: Ching Chung Lin, MD, MMS, Division of Gastroenterology, Department of Internal Medicine, Mackay Memorial Hospital, Taipei 111, Taiwan, China
S- Editor Sun H L- Editor Cant MR E- Editor Xiong L