Published online Oct 7, 2010. doi: 10.3748/wjg.v16.i37.4627
Revised: June 21, 2010
Accepted: June 28, 2010
Published online: October 7, 2010
The phosphoinositide 3-kinase (PI3K)/phosphatase and tensin homolog (PTEN)/Akt axis is a key signal transduction node that regulates crucial cellular functions, including insulin and other growth factors signaling, lipid and glucose metabolism, as well as cell survival and apoptosis. In this pathway, PTEN acts as a phosphoinositide phosphatase, which terminates PI3K-propagated signaling by dephosphorylating PtdIns(3,4)P2 and PtdIns(3,4,5)P3. However, the role of PTEN does not appear to be restricted only to PI3K signaling antagonism, and new functions have been recently discovered for this protein. In addition to the well-established role of PTEN as a tumor suppressor, increasing evidence now suggests that a dysregulated PTEN expression and/or activity is also linked to the development of several hepatic pathologies. Dysregulated PTEN expression/activity is observed with obesity, insulin resistance, diabetes, hepatitis B virus/hepatitis C virus infections, and abusive alcohol consumption, whereas mutations/deletions have also been associated with the occurrence of hepatocellular carcinoma. Thus, it appears that alterations of PTEN expression and activity in hepatocytes are common and recurrent molecular events associated with liver disorders of various etiologies. These recent findings suggest that PTEN might represent a potential common therapeutic target for a number of liver pathologies.
- Citation: Peyrou M, Bourgoin L, Foti M. PTEN in liver diseases and cancer. World J Gastroenterol 2010; 16(37): 4627-4633
- URL: https://www.wjgnet.com/1007-9327/full/v16/i37/4627.htm
- DOI: https://dx.doi.org/10.3748/wjg.v16.i37.4627
Obesity, metabolic syndrome, hepatitis virus infections, and abusive alcohol consumption are major etiological factors contributing together, or independently, to the development of severe liver diseases[1-3]. Interestingly, the hepatic pathologies induced by these various factors are associated with common metabolic disorders, i.e. insulin resistance and dysregulated lipid metabolism, and encompass similar histological abnormalities, ranging from hepatic steatosis to steatohepatitis, fibrosis, and cirrhosis[4]. Hepatocellular adenoma (HCA) or hepatocellular carcinoma (HCC) might then occur as a likely end stage of these diseases[5].
Deregulations of numerous signaling pathways leading to insulin resistance, steatosis, inflammation, fibrosis, aberrant cell proliferation, and resistance to cell death have been reported. Among these, abnormal signaling through the phosphoinositide 3-kinase (PI3K)/phosphatase and tensin homolog (PTEN)/Akt pathway critically contributes to the development of non-alcoholic fatty liver disease (NAFLD), non-alcoholic steatohepatitis (NASH), alcoholic liver disease (ALD), viral hepatitis [hepatitis B virus (HBV) and hepatitis C virus (HCV)], and HCC[6-10]. Of particular interest in this signaling pathway is the role of PTEN, an important tumor suppressor having a protein and phosphoinositide phosphatase activity. Indeed, PTEN switches off signaling through the PI3K-Akt axis and by doing that, controls growth factor signaling, thereby acting as a potent tumor suppressor. In the context of hepatic metabolic disorders and cancer, increasing evidence now supports a crucial role of PTEN in the development of these diseases.
The PTEN protein was first identified as a potent tumor suppressor that was frequently mutated or deleted in several human cancers, including HCC[11-16]. The best-characterized function of PTEN is its phosphoinositide phosphatase activity, where it dephosphorylates the PtdIns(3,4)P2 and PtdIns(3,4,5)P3 second messengers on the 3’-position of the inositol ring. PTEN thus antagonizes PI3K activation and acts as a potent regulator of growth factor signaling, in particular insulin/insulin-like growth factor (IGF)-1 signaling, in peripheral tissues such as the liver[8,17]. In vitro studies also suggested that PTEN might have a protein phosphatase activity; however additional studies are required to confirm this enzymatic activity[18-20].
Compared to other classical tumor suppressor, PTEN represents a particular case, as loss of heterozygosity, or partial inhibition of its expression/activity, is sufficient to promote carcinogenesis, in addition to affecting critical cellular functions, such as glucose and lipid metabolism. Consistent with these observations, PTEN expression and activity appears to be regulated by numerous and complex mechanisms. Among these mechanisms, epigenetic silencing by hypermethylation of its promoter[11,21] or histone deacetylase activity[22] strongly affect PTEN expression. Several transcription factors, including Egr1[23], p53[24], peroxisome proliferator-activated receptor γ[25], Spry2[26], Atf2[27], and Myc[28], have been shown to directly bind the PTEN promoter and to upregulate PTEN transcription. On the other hand, transcription factors such as nuclear factor (NF)-κB[29,30], p300/CBP[29], Hes-1[28], Cbf-1[31,32], and c-Jun[33] have been shown to negatively regulate PTEN transcription. Adding to the complexity of PTEN expression regulation, recent evidence also indicated that the PTEN mRNA undergoes post-transcriptional repression/degradation by specific microRNAs (miRNAs). Several miRNAs, including miR-21, miR-19a, miR-17-92, miR-214, miR-216a, and miR-217, have been shown to specifically modulate PTEN mRNA expression[34-39]. Finally, additional processes, whereby the PTEN protein level and activity are modulated, occur post-translationally. These include modifications of the protein, such as phosphorylation, acetylation, ubiquitination, and the REDOX state, which affect PTEN stability, degradation and enzymatic activity[21,40,41]. Similarly, PTEN sequestration in specific subcellular compartments or membranes through interactions with distinct proteins, i.e. FAK[42], MAGI proteins[43], MAST proteins[44], p53[45], NHERF1/2[46], and PICT1[47] likely represent additional mechanisms controlling PTEN stability and/or activity.
Functional defects of key intracellular signaling proteins, in particular in the mammalian target of rapamycin (mTOR)/PI3K/PTEN/Akt pathways, are clearly associated with insulin unresponsiveness of peripheral tissues[48,49]. Indeed, the imbalance between propagation and termination of signaling through mTOR/PI3K/PTEN/Akt represents basal molecular dysfunctions triggering the development of insulin resistance and type II diabetes. Given the enzymatic activity of PTEN, dysregulation of its activity/expression are potentially important mechanisms contributing to insulin resistance in tissues such as the liver[8].
Several in vivo studies, where PTEN expression was genetically altered in various organs of mice, support a crucial role for PTEN expression/activity in insulin sensitivity. PTEN haploinsufficiency and PTEN muscle-specific deletion were shown to improve skeletal muscle insulin sensitivity and to protect mice from insulin resistance and diabetes caused by high fat feeding, respectively[50,51]. Deletion of PTEN in the adipose tissue prevented the development of streptozotocin-induced diabetes[52]. Treatment of db/db mice with PTEN antisense oligonucleotides normalized plasma glucose levels in these animals[53].
Interestingly, liver-specific PTEN knockout mice also have an improved systemic insulin sensitivity and glucose tolerance[54,55]. However, whether this is related to increased insulin sensitivity specifically in the liver, or to a complex in vivo systemic crosstalk between a PTEN-deficient liver and other peripheral tissues, remains unclear. In support of this latter hypothesis, PTEN deletion in the liver is accompanied by decreased circulating levels of leptin and body fat content[55]. In addition, we observed that although constitutive Akt activity is increased in cultured hepatoma cells having downregulated PTEN, insulin signaling upstream of Akt, i.e. insulin receptor/IRS1 expression and phosphorylation, is impaired, as it has been previously described in cancer cells[30,56]. Consistent with these findings, we observed a lack of insulin responsiveness in terms of gene expression in PTEN deficient hepatocytes[30]. These data raise the hypothesis that PTEN downregulation in hepatocytes might, paradoxically, cause insulin resistance despite an increased activation of specific insulin effectors, such as Akt. Further studies are needed to clarify whether PTEN downregulation in hepatocytes is a causal factor for insulin resistance in this organ.
Although liver-specific PTEN knockout mice have an overall improved systemic insulin sensitivity, they develop an important steatosis in the liver, suggesting that PTEN is required for an appropriate control of the hepatic lipid metabolism[54,55]. Consistent with these studies, hepatic PTEN expression is downregulated in obese and insulin resistant rat animal models and in humans having steatosis[30]. Further studies indicate that high levels of circulating free fatty acids, but not glucose or insulin, downregulate PTEN expression in hepatocytes[30]. PTEN downregulation by free fatty acids is triggered by an increase in miR-21, a miRNA targeting PTEN mRNA for degradation, through mTOR/NF-κB-dependent mechanisms[30,36]. In addition to an excess of circulating free fatty acids, a deregulated production of inflammatory cytokines by immune cells and/or of adipokines by the adipose tissue, as observed with NAFLD[57], can also independently, or synergistically with fatty acids, alter PTEN expression. Indeed, inflammatory cytokines, such as transforming growth factor β[39,58-60], tumor necrosis factor (TNF)α[29,61], interleukin (IL)-6[62], IL-1[63], or adipokines such as leptin, resistin and adiponectin[64,65], have been reported to either up- or downregulate PTEN expression or activity in various cells. Although most of these cytokines/adipokines are clearly involved in liver insulin sensitivity and steatosis/fibrosis/inflammation[57,66], it remains to be firmly established whether these factors modulate PTEN expression/activity in the liver and whether there is a causal relationship between potential PTEN alterations induced by these cytokines/adipokines and their beneficial/detrimental effects on the liver physiology.
PTEN loss of function in the liver leads to a progressive and step-wise development of steatohepatitis and fibrosis[54,55,67]. Consistent with studies using liver-specific PTEN knockout mice, decreased PTEN expression was also observed in the liver of rodents with hepatic fibrosis induced either by biliary stenosis or a choline-deficient diet[68,69]. Finally, PTEN depletion in hepatoma cells induces the expression of genes promoting inflammation, epithelial-to-mesenchymal transition, and fibrosis[70]. Taken together, these studies suggest that pathological dysregulation of PTEN expression/activity causing steatosis might also promote progression of this disorder towards different clinical stages of increasing severity. The molecular mechanisms by which PTEN deficiency triggers steatosis, inflammation, and fibrosis development in hepatocytes are still poorly defined. However, evidence indicates that de novo fatty acids synthesis is enhanced[54,55] in liver-specific PTEN knockout mice, whereas in cultured cells, accumulation of neutral lipids seems to rely mainly on increased fatty acids uptake and esterification[30]. These discrepant data might originate either from the different methodologies used to investigate fatty acid metabolism or, more likely, from the different extents of PTEN repression, i.e. complete deletion in knockout mice vs 40%-80% downregulation induced by fatty acids or silencing RNAs in cultured cells. Partial PTEN downregulation or total deletion can indeed mediate very different effects, as it was elegantly demonstrated in studies examining the role of PTEN in prostate tumor progression[41,71].
The first evidence supporting a critical role for PTEN in liver cancer came from genetic studies in mice, where heterozygous deletion of PTEN was shown to induce atypical adenomatous liver hyperplasia[72]. Additional studies then demonstrated that PTEN deficiency in the liver induces hepatomegaly, HCA, and HCC with ageing[54,55]. Weak expression or mutation/deletion of PTEN, as well as upregulation of miRNAs specifically targeting PTEN for degradation, are also frequently observed in human HCC[11,13,16,34,73-76]. However, the tumor suppressor activity of PTEN seems to principally involve its antagonistic effects on the anti-apoptotic, proliferative, and hypertrophic activity of PI3K[77]; recent studies demonstrated that PTEN also plays an essential role in the nucleus to maintain chromosomal stability and for DNA repair[78]. In addition, there is evidence indicating that PTEN can modulate cancer cell invasiveness by stabilizing E-cadherin/ β-catenin adherens junctional complexes[79,80]. Finally, we demonstrated that fatty acids-mediated PTEN downregulation in hepatocytes promotes cell proliferation, migration, and invasiveness, in addition to modulating a set of genes involved in cell cycle regulation and HCC[70]. As inflammation, EMT and genomic alterations are typical features of HCC[81,82], impaired PTEN expression or activity can thus represent an important step in progression of NAFLD towards HCC. Further studies are however still needed to confirm the relevance of PTEN as a prognostic marker for the risk of HCA/HCC development.
Infections by HBV and HCV are major contributors to the high incidence of HCC, particularly in South-East Asia and Africa[83,84]. Similarly to NAFLD and NASH, HCV infection is strongly associated with liver insulin resistance and causes steatosis and fibrosis[85,86]. However, whether HBV infection causes similar liver disorders remains unclear[87].
Only a few studies have examined the involvement of PTEN in HBV/HCV-associated hepatocyte dysfunction. The HBV-X protein (HBx) was shown to trigger uncontrolled Akt activation by downregulating PTEN expression in Chang liver cells, thereby enhancing the invasive potential of these cells[88,89]. In accordance with these data, PTEN overexpression in Chang cells reversed pro-survival signaling and inhibited apoptosis induced by HBx[90]. In addition, PTEN was also shown to prevent HBx-mediated induction of IGF-II expression in hepatoma cell lines[91]. IGF-II plays an essential role in HCC development; therefore, these data suggest that PTEN downregulation might make an important contribution to cell proliferation induced by HBx.
Direct evidence implicating a PTEN loss or gain of function in HCV-mediated liver diseases is scarce and only few studies investigate this issue. PTEN inactivation by post-translational phosphorylation was suggested to contribute to transactivation of SREBPs, which are major regulators of the lipid metabolism, in HCV-infected Huh-7 cells[92]. Correlative immunohistochemical analyses of human HCV-positive cirrhotic HCC also indicated that PTEN is downregulated in these tumors and that its expression was inversely correlated with expression of inducible nitric oxide synthase and cyclooxygenase II. Interestingly, high PTEN expression was evaluated as a positive independent prognostic factor for the survival of HCV-positive cirrhotic HCC patients[93].
Further molecular, clinical, and epidemiological studies are now warranted to understand the detailed mechanisms by which HBV/HCV infections alter PTEN expression or activity in the liver, as well as the pathological outcomes of PTEN dysfunctions in HBV/HCV infections.
ALD also encompass a spectrum of histological and functional liver disorders, ranging from a relatively benign steatosis to alcoholic hepatitis, cirrhosis, and cancer[2]. Given the outcomes of alcohol abuse in the liver, and the effects of PTEN deletion or downregulation for the liver physiology, it could be expected that ethanol induces alterations of PTEN expression/activity in the liver. Surprisingly, increased apoptosis and decreased insulin signaling in hepatocytes of Long-Evans rats chronically fed with ethanol (serum ethanol levels is about 50 mmol/L) was associated with increased levels of PTEN mRNA and protein, thus suggesting that ethanol upregulates PTEN expression in the liver[94]. Increased hepatic PTEN expression was also observed in rats exposed to alcohol in utero[95]. Consistent with these studies, chronic exposure of hepatoma HepG2E47 cells to ethanol increased PTEN expression and subsequently increased the sensitivity of cells to TNFα-induced cytotoxicity and apoptosis[96]. In contrast, acute ethanol exposure did not affect PTEN expression in Huh-7 hepatoma cells. However, in these cells, ethanol increased the physical association between PTEN and the PI3K regulatory subunit p85a, which functionally resulted in a decreased Akt and downstream effectors activity[97].
Taken together, these studies indicated that, in contrast to the PTEN downregulation occurring with NAFLD, PTEN expression/activity is upregulated with ALD. This opposite regulation of a critical signaling effector strongly suggests that the mechanisms of insulin resistance and steatosis development in the context of NAFLD and ALD are distinct. In addition, PTEN might represent a differential diagnostic marker to distinguish between liver disorders with these different etiologies.
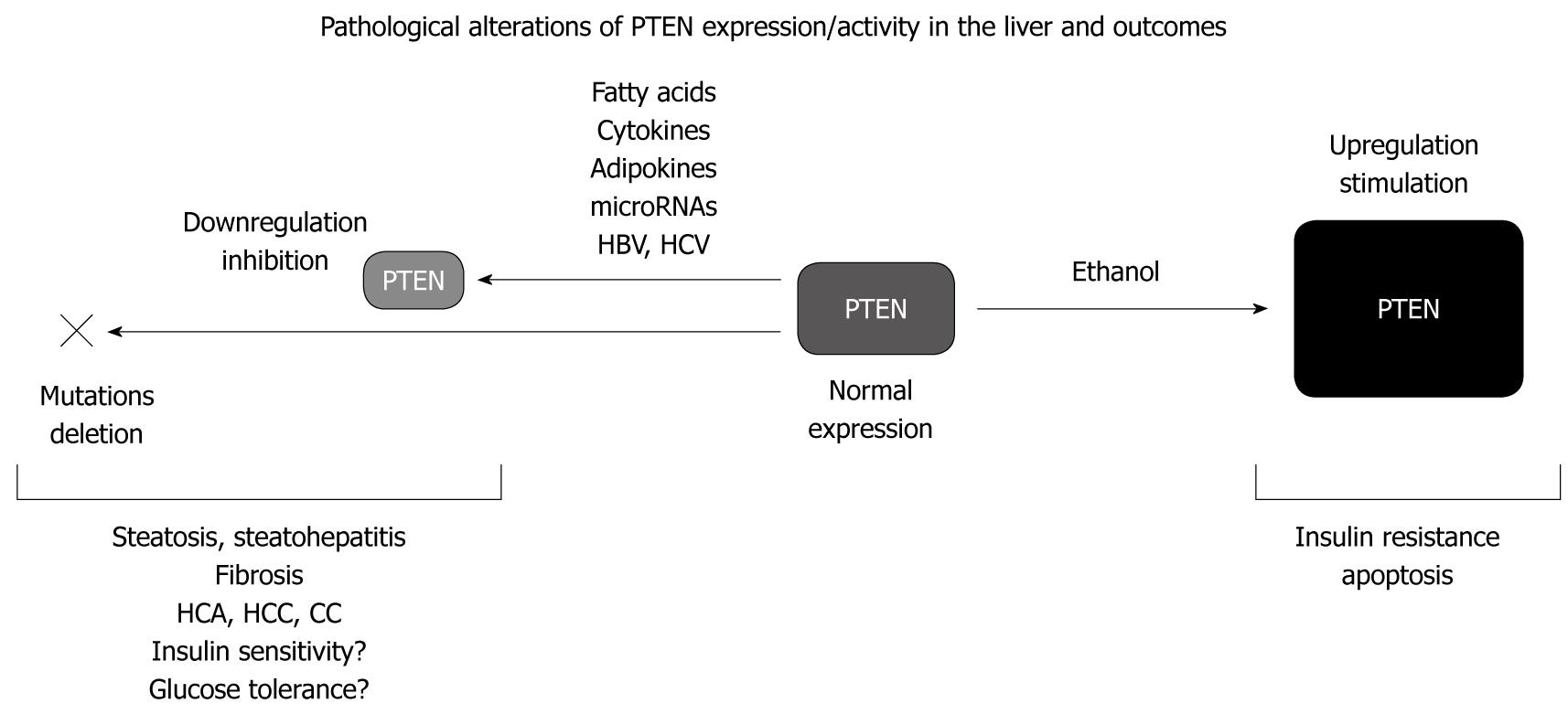
Accumulating evidence indicates that PTEN is a major dysregulated cellular factor contributing to the development of a broad spectrum of hepatic disorders, i.e. insulin resistance, steatosis, steatohepatitis, fibrosis, cirrhosis, and cancer (Figure 1). Indeed, hepatic PTEN expression/activity is altered in liver diseases associated with obesity, metabolic syndrome, viral infection, and alcohol consumption. Thus, it appears that dysregulation of PTEN expression/activity in hepatocytes represents an important and recurrent molecular mechanism contributing to the development of liver disorders with distinct etiologies.
Although hepatic steatosis is currently regarded as a benign disease, progression to inflammation, fibrosis, and cirrhosis can lead to liver failure and development of HCC. There are multiple molecular factors involved in the progression of hepatic steatosis towards more severe stages and among those, dysregulation of PTEN expression/activity, more than PTEN mutations or deletions, could be a critical step in the occurrence and development of these diseases. In addition, PTEN alterations induced by high levels of free fatty acids or inflammatory cytokines, provide an interesting link between insulin resistance and steatosis, which might also explain, at least in part, the high risk factor for HCA/HCC associated with diabetes and obesity[98,99]. Given the tumor suppressor activity of PTEN, the role of steatosis and steatohepatitis as preneoplastic states in the hepatocyte malignant transformation should also be re-evaluated. Additional studies are now required to carefully evaluate PTEN as a differential prognostic marker in liver pathologies with distinct etiologies and to assess the pertinence of future therapeutic interventions to restore physiological PTEN expression in the liver to prevent, or to alleviate, hepatic metabolic disorders and HCC.
Peer reviewers: Thomas Kietzmann, Professor, Department of Biochemistry, University of Oulu, Oulu 90014, Finland; Dr. Juan Carlos Laguna Egea, Catedràtic de Farmacologia/Pharmacology Professor, Unitat de Farmacologia/Pharmacology laboratory, Facultat de Farmàcia/School of Pharmacy, Universitat de Barcelona/University of Barcelona, Avda Diagonal 643, Barcelona 08028, Spain; Dr. Joao Batista Teixeira Rocha, Department of Toxicological Biochemistry, Universidade Federal De Santa Maria, Santa Maria 97105-900, Brazil
S- Editor Wang JL L- Editor Stewart GJ E- Editor Zheng XM
| 1. | Williams R. Global challenges in liver disease. Hepatology. 2006;44:521-526. |
| 2. | Yip WW, Burt AD. Alcoholic liver disease. Semin Diagn Pathol. 2006;23:149-160. |
| 3. | Marchesini G, Babini M. Nonalcoholic fatty liver disease and the metabolic syndrome. Minerva Cardioangiol. 2006;54:229-239. |
| 4. | Contos MJ, Choudhury J, Mills AS, Sanyal AJ. The histologic spectrum of nonalcoholic fatty liver disease. Clin Liver Dis. 2004;8:481-500, vii. |
| 5. | Farazi PA, DePinho RA. Hepatocellular carcinoma pathogenesis: from genes to environment. Nat Rev Cancer. 2006;6:674-687. |
| 6. | Avila MA, Berasain C, Sangro B, Prieto J. New therapies for hepatocellular carcinoma. Oncogene. 2006;25:3866-3884. |
| 7. | Thorgeirsson SS, Grisham JW. Molecular pathogenesis of human hepatocellular carcinoma. Nat Genet. 2002;31:339-346. |
| 8. | Vinciguerra M, Foti M. PTEN and SHIP2 phosphoinositide phosphatases as negative regulators of insulin signalling. Arch Physiol Biochem. 2006;112:89-104. |
| 9. | Vinciguerra M, Foti M. PTEN at the crossroad of metabolic diseases and cancer in the liver. Ann Hepatol. 2008;7:192-199. |
| 10. | He L, Hou X, Kanel G, Zeng N, Galicia V, Wang Y, Yang J, Wu H, Birnbaum MJ, Stiles BL. The critical role of AKT2 in hepatic steatosis induced by PTEN loss. Am J Pathol. 2010;176:2302-2308. |
| 11. | Wang L, Wang WL, Zhang Y, Guo SP, Zhang J, Li QL. Epigenetic and genetic alterations of PTEN in hepatocellular carcinoma. Hepatol Res. 2007;37:389-396. |
| 12. | Chow LM, Baker SJ. PTEN function in normal and neoplastic growth. Cancer Lett. 2006;241:184-196. |
| 13. | Hu TH, Huang CC, Lin PR, Chang HW, Ger LP, Lin YW, Changchien CS, Lee CM, Tai MH. Expression and prognostic role of tumor suppressor gene PTEN/MMAC1/TEP1 in hepatocellular carcinoma. Cancer. 2003;97:1929-1940. |
| 14. | Dong-Dong L, Xi-Ran Z, Xiang-Rong C. Expression and significance of new tumor suppressor gene PTEN in primary liver cancer. J Cell Mol Med. 2003;7:67-71. |
| 15. | Fujiwara Y, Hoon DS, Yamada T, Umeshita K, Gotoh M, Sakon M, Nishisho I, Monden M. PTEN / MMAC1 mutation and frequent loss of heterozygosity identified in chromosome 10q in a subset of hepatocellular carcinomas. Jpn J Cancer Res. 2000;91:287-292. |
| 16. | Yao YJ, Ping XL, Zhang H, Chen FF, Lee PK, Ahsan H, Chen CJ, Lee PH, Peacocke M, Santella RM. PTEN/MMAC1 mutations in hepatocellular carcinomas. Oncogene. 1999;18:3181-3185. |
| 17. | Downes CP, Perera N, Ross S, Leslie NR. Substrate specificity and acute regulation of the tumour suppressor phosphatase, PTEN. Biochem Soc Symp. 2007;69-80. |
| 18. | Weng LP, Brown JL, Eng C. PTEN coordinates G(1) arrest by down-regulating cyclin D1 via its protein phosphatase activity and up-regulating p27 via its lipid phosphatase activity in a breast cancer model. Hum Mol Genet. 2001;10:599-604. |
| 19. | Cai XM, Tao BB, Wang LY, Liang YL, Jin JW, Yang Y, Hu YL, Zha XL. Protein phosphatase activity of PTEN inhibited the invasion of glioma cells with epidermal growth factor receptor mutation type III expression. Int J Cancer. 2005;117:905-912. |
| 20. | Davidson L, Maccario H, Perera NM, Yang X, Spinelli L, Tibarewal P, Glancy B, Gray A, Weijer CJ, Downes CP. Suppression of cellular proliferation and invasion by the concerted lipid and protein phosphatase activities of PTEN. Oncogene. 2010;29:687-697. |
| 21. | Tamguney T, Stokoe D. New insights into PTEN. J Cell Sci. 2007;120:4071-4079. |
| 22. | Pan L, Lu J, Wang X, Han L, Zhang Y, Han S, Huang B. Histone deacetylase inhibitor trichostatin a potentiates doxorubicin-induced apoptosis by up-regulating PTEN expression. Cancer. 2007;109:1676-1688. |
| 23. | Virolle T, Adamson ED, Baron V, Birle D, Mercola D, Mustelin T, de Belle I. The Egr-1 transcription factor directly activates PTEN during irradiation-induced signalling. Nat Cell Biol. 2001;3:1124-1128. |
| 24. | Stambolic V, MacPherson D, Sas D, Lin Y, Snow B, Jang Y, Benchimol S, Mak TW. Regulation of PTEN transcription by p53. Mol Cell. 2001;8:317-325. |
| 25. | Zhang W, Wu N, Li Z, Wang L, Jin J, Zha XL. PPARgamma activator rosiglitazone inhibits cell migration via upregulation of PTEN in human hepatocarcinoma cell line BEL-7404. Cancer Biol Ther. 2006;5:1008-1014. |
| 26. | Edwin F, Singh R, Endersby R, Baker SJ, Patel TB. The tumor suppressor PTEN is necessary for human Sprouty 2-mediated inhibition of cell proliferation. J Biol Chem. 2006;281:4816-4822. |
| 27. | Shen YH, Zhang L, Gan Y, Wang X, Wang J, LeMaire SA, Coselli JS, Wang XL. Up-regulation of PTEN (phosphatase and tensin homolog deleted on chromosome ten) mediates p38 MAPK stress signal-induced inhibition of insulin signaling. A cross-talk between stress signaling and insulin signaling in resistin-treated human endothelial cells. J Biol Chem. 2006;281:7727-7736. |
| 28. | Palomero T, Sulis ML, Cortina M, Real PJ, Barnes K, Ciofani M, Caparros E, Buteau J, Brown K, Perkins SL. Mutational loss of PTEN induces resistance to NOTCH1 inhibition in T-cell leukemia. Nat Med. 2007;13:1203-1210. |
| 29. | Vasudevan KM, Gurumurthy S, Rangnekar VM. Suppression of PTEN expression by NF-kappa B prevents apoptosis. Mol Cell Biol. 2004;24:1007-1021. |
| 30. | Vinciguerra M, Veyrat-Durebex C, Moukil MA, Rubbia-Brandt L, Rohner-Jeanrenaud F, Foti M. PTEN down-regulation by unsaturated fatty acids triggers hepatic steatosis via an NF-kappaBp65/mTOR-dependent mechanism. Gastroenterology. 2008;134:268-280. |
| 31. | Chappell WH, Green TD, Spengeman JD, McCubrey JA, Akula SM, Bertrand FE. Increased protein expression of the PTEN tumor suppressor in the presence of constitutively active Notch-1. Cell Cycle. 2005;4:1389-1395. |
| 32. | Whelan JT, Forbes SL, Bertrand FE. CBF-1 (RBP-J kappa) binds to the PTEN promoter and regulates PTEN gene expression. Cell Cycle. 2007;6:80-84. |
| 33. | Hettinger K, Vikhanskaya F, Poh MK, Lee MK, de Belle I, Zhang JT, Reddy SA, Sabapathy K. c-Jun promotes cellular survival by suppression of PTEN. Cell Death Differ. 2007;14:218-229. |
| 34. | Meng F, Henson R, Wehbe-Janek H, Ghoshal K, Jacob ST, Patel T. MicroRNA-21 regulates expression of the PTEN tumor suppressor gene in human hepatocellular cancer. Gastroenterology. 2007;133:647-658. |
| 35. | Pezzolesi MG, Platzer P, Waite KA, Eng C. Differential expression of PTEN-targeting microRNAs miR-19a and miR-21 in Cowden syndrome. Am J Hum Genet. 2008;82:1141-1149. |
| 36. | Vinciguerra M, Sgroi A, Veyrat-Durebex C, Rubbia-Brandt L, Buhler LH, Foti M. Unsaturated fatty acids inhibit the expression of tumor suppressor phosphatase and tensin homolog (PTEN) via microRNA-21 up-regulation in hepatocytes. Hepatology. 2009;49:1176-1184. |
| 37. | Xiao C, Srinivasan L, Calado DP, Patterson HC, Zhang B, Wang J, Henderson JM, Kutok JL, Rajewsky K. Lymphoproliferative disease and autoimmunity in mice with increased miR-17-92 expression in lymphocytes. Nat Immunol. 2008;9:405-414. |
| 38. | Yang H, Kong W, He L, Zhao JJ, O'Donnell JD, Wang J, Wenham RM, Coppola D, Kruk PA, Nicosia SV. MicroRNA expression profiling in human ovarian cancer: miR-214 induces cell survival and cisplatin resistance by targeting PTEN. Cancer Res. 2008;68:425-433. |
| 39. | Kato M, Putta S, Wang M, Yuan H, Lanting L, Nair I, Gunn A, Nakagawa Y, Shimano H, Todorov I. TGF-beta activates Akt kinase through a microRNA-dependent amplifying circuit targeting PTEN. Nat Cell Biol. 2009;11:881-889. |
| 42. | Tamura M, Gu J, Matsumoto K, Aota S, Parsons R, Yamada KM. Inhibition of cell migration, spreading, and focal adhesions by tumor suppressor PTEN. Science. 1998;280:1614-1617. |
| 43. | Wu X, Hepner K, Castelino-Prabhu S, Do D, Kaye MB, Yuan XJ, Wood J, Ross C, Sawyers CL, Whang YE. Evidence for regulation of the PTEN tumor suppressor by a membrane-localized multi-PDZ domain containing scaffold protein MAGI-2. Proc Natl Acad Sci USA. 2000;97:4233-4238. |
| 44. | Valiente M, Andrés-Pons A, Gomar B, Torres J, Gil A, Tapparel C, Antonarakis SE, Pulido R. Binding of PTEN to specific PDZ domains contributes to PTEN protein stability and phosphorylation by microtubule-associated serine/threonine kinases. J Biol Chem. 2005;280:28936-28943. |
| 45. | Tang Y, Eng C. p53 down-regulates phosphatase and tensin homologue deleted on chromosome 10 protein stability partially through caspase-mediated degradation in cells with proteasome dysfunction. Cancer Res. 2006;66:6139-6148. |
| 46. | Takahashi Y, Morales FC, Kreimann EL, Georgescu MM. PTEN tumor suppressor associates with NHERF proteins to attenuate PDGF receptor signaling. EMBO J. 2006;25:910-920. |
| 47. | Okahara F, Ikawa H, Kanaho Y, Maehama T. Regulation of PTEN phosphorylation and stability by a tumor suppressor candidate protein. J Biol Chem. 2004;279:45300-45303. |
| 48. | Sesti G, Federici M, Hribal ML, Lauro D, Sbraccia P, Lauro R. Defects of the insulin receptor substrate (IRS) system in human metabolic disorders. FASEB J. 2001;15:2099-2111. |
| 49. | Shepherd PR. Mechanisms regulating phosphoinositide 3-kinase signalling in insulin-sensitive tissues. Acta Physiol Scand. 2005;183:3-12. |
| 50. | Wijesekara N, Konrad D, Eweida M, Jefferies C, Liadis N, Giacca A, Crackower M, Suzuki A, Mak TW, Kahn CR. Muscle-specific Pten deletion protects against insulin resistance and diabetes. Mol Cell Biol. 2005;25:1135-1145. |
| 51. | Wong JT, Kim PT, Peacock JW, Yau TY, Mui AL, Chung SW, Sossi V, Doudet D, Green D, Ruth TJ. Pten (phosphatase and tensin homologue gene) haploinsufficiency promotes insulin hypersensitivity. Diabetologia. 2007;50:395-403. |
| 52. | Kurlawalla-Martinez C, Stiles B, Wang Y, Devaskar SU, Kahn BB, Wu H. Insulin hypersensitivity and resistance to streptozotocin-induced diabetes in mice lacking PTEN in adipose tissue. Mol Cell Biol. 2005;25:2498-2510. |
| 53. | Butler M, McKay RA, Popoff IJ, Gaarde WA, Witchell D, Murray SF, Dean NM, Bhanot S, Monia BP. Specific inhibition of PTEN expression reverses hyperglycemia in diabetic mice. Diabetes. 2002;51:1028-1034. |
| 54. | Horie Y, Suzuki A, Kataoka E, Sasaki T, Hamada K, Sasaki J, Mizuno K, Hasegawa G, Kishimoto H, Iizuka M. Hepatocyte-specific Pten deficiency results in steatohepatitis and hepatocellular carcinomas. J Clin Invest. 2004;113:1774-1783. |
| 55. | Stiles B, Wang Y, Stahl A, Bassilian S, Lee WP, Kim YJ, Sherwin R, Devaskar S, Lesche R, Magnuson MA. Liver-specific deletion of negative regulator Pten results in fatty liver and insulin hypersensitivity [corrected]. Proc Natl Acad Sci USA. 2004;101:2082-2087. |
| 56. | Lackey J, Barnett J, Davidson L, Batty IH, Leslie NR, Downes CP. Loss of PTEN selectively desensitizes upstream IGF1 and insulin signaling. Oncogene. 2007;26:7132-7142. |
| 57. | Tilg H, Moschen AR. Insulin resistance, inflammation, and non-alcoholic fatty liver disease. Trends Endocrinol Metab. 2008;19:371-379. |
| 58. | Zhang L, Yu Q, He J, Zha X. Study of the PTEN gene expression and FAK phosphorylation in human hepatocarcinoma tissues and cell lines. Mol Cell Biochem. 2004;262:25-33. |
| 59. | Mahimainathan L, Das F, Venkatesan B, Choudhury GG. Mesangial cell hypertrophy by high glucose is mediated by downregulation of the tumor suppressor PTEN. Diabetes. 2006;55:2115-2125. |
| 60. | Yang Y, Zhou F, Fang Z, Wang L, Li Z, Sun L, Wang C, Yao W, Cai X, Jin J. Post-transcriptional and post-translational regulation of PTEN by transforming growth factor-beta1. J Cell Biochem. 2009;106:1102-1112. |
| 61. | Kim S, Domon-Dell C, Kang J, Chung DH, Freund JN, Evers BM. Down-regulation of the tumor suppressor PTEN by the tumor necrosis factor-alpha/nuclear factor-kappaB (NF-kappaB)-inducing kinase/NF-kappaB pathway is linked to a default IkappaB-alpha autoregulatory loop. J Biol Chem. 2004;279:4285-4291. |
| 62. | Löffler D, Brocke-Heidrich K, Pfeifer G, Stocsits C, Hackermüller J, Kretzschmar AK, Burger R, Gramatzki M, Blumert C, Bauer K. Interleukin-6 dependent survival of multiple myeloma cells involves the Stat3-mediated induction of microRNA-21 through a highly conserved enhancer. Blood. 2007;110:1330-1333. |
| 63. | Barbieri SS, Ruggiero L, Tremoli E, Weksler BB. Suppressing PTEN activity by tobacco smoke plus interleukin-1beta modulates dissociation of VE-cadherin/beta-catenin complexes in endothelium. Arterioscler Thromb Vasc Biol. 2008;28:732-738. |
| 64. | Ning K, Miller LC, Laidlaw HA, Burgess LA, Perera NM, Downes CP, Leslie NR, Ashford ML. A novel leptin signalling pathway via PTEN inhibition in hypothalamic cell lines and pancreatic beta-cells. EMBO J. 2006;25:2377-2387. |
| 65. | Lam JB, Chow KH, Xu A, Lam KS, Liu J, Wong NS, Moon RT, Shepherd PR, Cooper GJ, Wang Y. Adiponectin haploinsufficiency promotes mammary tumor development in MMTV-PyVT mice by modulation of phosphatase and tensin homolog activities. PLoS One. 2009;4:e4968. |
| 66. | Budhu A, Wang XW. The role of cytokines in hepatocellular carcinoma. J Leukoc Biol. 2006;80:1197-1213. |
| 67. | Suzuki A, de la Pompa JL, Stambolic V, Elia AJ, Sasaki T, del Barco Barrantes I, Ho A, Wakeham A, Itie A, Khoo W. High cancer susceptibility and embryonic lethality associated with mutation of the PTEN tumor suppressor gene in mice. Curr Biol. 1998;8:1169-1178. |
| 68. | Hao LS, Zhang XL, An JY, Karlin J, Tian XP, Dun ZN, Xie SR, Chen S. PTEN expression is down-regulated in liver tissues of rats with hepatic fibrosis induced by biliary stenosis. APMIS. 2009;117:681-691. |
| 69. | Wang B, Majumder S, Nuovo G, Kutay H, Volinia S, Patel T, Schmittgen TD, Croce C, Ghoshal K, Jacob ST. Role of microRNA-155 at early stages of hepatocarcinogenesis induced by choline-deficient and amino acid-defined diet in C57BL/6 mice. Hepatology. 2009;50:1152-1161. |
| 70. | Vinciguerra M, Carrozzino F, Peyrou M, Carlone S, Montesano R, Benelli R, Foti M. Unsaturated fatty acids promote hepatoma proliferation and progression through downregulation of the tumor suppressor PTEN. J Hepatol. 2009;50:1132-1141. |
| 71. | Trotman LC, Niki M, Dotan ZA, Koutcher JA, Di Cristofano A, Xiao A, Khoo AS, Roy-Burman P, Greenberg NM, Van Dyke T. Pten dose dictates cancer progression in the prostate. PLoS Biol. 2003;1:E59. |
| 72. | Podsypanina K, Ellenson LH, Nemes A, Gu J, Tamura M, Yamada KM, Cordon-Cardo C, Catoretti G, Fisher PE, Parsons R. Mutation of Pten/Mmac1 in mice causes neoplasia in multiple organ systems. Proc Natl Acad Sci USA. 1999;96:1563-1568. |
| 73. | Wan XW, Jiang M, Cao HF, He YQ, Liu SQ, Qiu XH, Wu MC, Wang HY. The alteration of PTEN tumor suppressor expression and its association with the histopathological features of human primary hepatocellular carcinoma. J Cancer Res Clin Oncol. 2003;129:100-106. |
| 74. | Cotler SJ, Hay N, Xie H, Chen ML, Xu PZ, Layden TJ, Guzman G. Immunohistochemical expression of components of the Akt-mTORC1 pathway is associated with hepatocellular carcinoma in patients with chronic liver disease. Dig Dis Sci. 2008;53:844-849. |
| 75. | Wu SK, Wang BJ, Yang Y, Feng XH, Zhao XP, Yang DL. Expression of PTEN, PPM1A and P-Smad2 in hepatocellular carcinomas and adjacent liver tissues. World J Gastroenterol. 2007;13:4554-4559. |
| 76. | Hu TH, Wang CC, Huang CC, Chen CL, Hung CH, Chen CH, Wang JH, Lu SN, Lee CM, Changchien CS. Down-regulation of tumor suppressor gene PTEN, overexpression of p53, plus high proliferating cell nuclear antigen index predict poor patient outcome of hepatocellular carcinoma after resection. Oncol Rep. 2007;18:1417-1426. |
| 77. | Yuan TL, Cantley LC. PI3K pathway alterations in cancer: variations on a theme. Oncogene. 2008;27:5497-5510. |
| 78. | Shen WH, Balajee AS, Wang J, Wu H, Eng C, Pandolfi PP, Yin Y. Essential role for nuclear PTEN in maintaining chromosomal integrity. Cell. 2007;128:157-170. |
| 79. | Kotelevets L, van Hengel J, Bruyneel E, Mareel M, van Roy F, Chastre E. The lipid phosphatase activity of PTEN is critical for stabilizing intercellular junctions and reverting invasiveness. J Cell Biol. 2001;155:1129-1135. |
| 80. | Kotelevets L, van Hengel J, Bruyneel E, Mareel M, van Roy F, Chastre E. Implication of the MAGI-1b/PTEN signalosome in stabilization of adherens junctions and suppression of invasiveness. FASEB J. 2005;19:115-117. |
| 82. | Lee TK, Poon RT, Yuen AP, Ling MT, Kwok WK, Wang XH, Wong YC, Guan XY, Man K, Chau KL. Twist overexpression correlates with hepatocellular carcinoma metastasis through induction of epithelial-mesenchymal transition. Clin Cancer Res. 2006;12:5369-5376. |
| 83. | Michielsen PP, Francque SM, van Dongen JL. Viral hepatitis and hepatocellular carcinoma. World J Surg Oncol. 2005;3:27. |
| 84. | Tan A, Yeh SH, Liu CJ, Cheung C, Chen PJ. Viral hepatocarcinogenesis: from infection to cancer. Liver Int. 2008;28:175-188. |
| 85. | Negro F, Alaei M. Hepatitis C virus and type 2 diabetes. World J Gastroenterol. 2009;15:1537-1547. |
| 86. | Negro F, Sanyal AJ. Hepatitis C virus, steatosis and lipid abnormalities: clinical and pathogenic data. Liver Int. 2009;29 Suppl 2:26-37. |
| 87. | Wang CC, Hsu CS, Liu CJ, Kao JH, Chen DS. Association of chronic hepatitis B virus infection with insulin resistance and hepatic steatosis. J Gastroenterol Hepatol. 2008;23:779-782. |
| 88. | Chung TW, Lee YC, Ko JH, Kim CH. Hepatitis B Virus X protein modulates the expression of PTEN by inhibiting the function of p53, a transcriptional activator in liver cells. Cancer Res. 2003;63:3453-3458. |
| 89. | Chung TW, Lee YC, Kim CH. Hepatitis B viral HBx induces matrix metalloproteinase-9 gene expression through activation of ERK and PI-3K/AKT pathways: involvement of invasive potential. FASEB J. 2004;18:1123-1125. |
| 90. | Kang-Park S, Im JH, Lee JH, Lee YI. PTEN modulates hepatitis B virus-X protein induced survival signaling in Chang liver cells. Virus Res. 2006;122:53-60. |
| 91. | Kang-Park S, Lee YI, Lee YI. PTEN modulates insulin-like growth factor II (IGF-II)-mediated signaling; the protein phosphatase activity of PTEN downregulates IGF-II expression in hepatoma cells. FEBS Lett. 2003;545:203-208. |
| 92. | Waris G, Felmlee DJ, Negro F, Siddiqui A. Hepatitis C virus induces proteolytic cleavage of sterol regulatory element binding proteins and stimulates their phosphorylation via oxidative stress. J Virol. 2007;81:8122-8130. |
| 93. | Rahman MA, Kyriazanos ID, Ono T, Yamanoi A, Kohno H, Tsuchiya M, Nagasue N. Impact of PTEN expression on the outcome of hepatitis C virus-positive cirrhotic hepatocellular carcinoma patients: possible relationship with COX II and inducible nitric oxide synthase. Int J Cancer. 2002;100:152-157. |
| 94. | Yeon JE, Califano S, Xu J, Wands JR, De La Monte SM. Potential role of PTEN phosphatase in ethanol-impaired survival signaling in the liver. Hepatology. 2003;38:703-714. |
| 95. | Yao XH, Nyomba BL. Hepatic insulin resistance induced by prenatal alcohol exposure is associated with reduced PTEN and TRB3 acetylation in adult rat offspring. Am J Physiol Regul Integr Comp Physiol. 2008;294:R1797-R1806. |
| 96. | Shulga N, Hoek JB, Pastorino JG. Elevated PTEN levels account for the increased sensitivity of ethanol-exposed cells to tumor necrosis factor-induced cytotoxicity. J Biol Chem. 2005;280:9416-9424. |
| 97. | He J, de la Monte S, Wands JR. Acute ethanol exposure inhibits insulin signaling in the liver. Hepatology. 2007;46:1791-1800. |
| 98. | Caldwell SH, Crespo DM, Kang HS, Al-Osaimi AM. Obesity and hepatocellular carcinoma. Gastroenterology. 2004;127:S97-S103. |
| 99. | El-Serag HB, Hampel H, Javadi F. The association between diabetes and hepatocellular carcinoma: a systematic review of epidemiologic evidence. Clin Gastroenterol Hepatol. 2006;4:369-380. |