Published online Sep 14, 2010. doi: 10.3748/wjg.v16.i34.4264
Revised: March 3, 2010
Accepted: March 10, 2010
Published online: September 14, 2010
The pathogenesis of inflammatory bowel diseases (IBDs) seems to involve a primary defect in one or more of the elements responsible for the maintenance of intestinal homeostasis and oral tolerance. The most important element is represented by the intestinal barrier, a complex system formed mostly by intestinal epithelial cells (IECs). IECs have an active role in producing mucus and regulating its composition; they provide a physical barrier capable of controlling antigen traffic through the intestinal mucosa. At the same time, they are able to play the role of non-professional antigen presenting cells, by processing and presenting antigens directly to the cells of the intestinal immune system. On the other hand, immune cells regulate epithelial growth and differentiation, producing a continuous bi-directional cross-talk within the barrier. Several alterations of the barrier function have been identified in IBD, starting from mucus features up to its components, from epithelial junctions up to the Toll-like receptors, and altered immune responses. It remains to be understood whether these defects are primary causes of epithelial damage or secondary effects. We review the possible role of the epithelial barrier and particularly describe the role of IECs in the pathogenesis of IBD.
- Citation: Roda G, Sartini A, Zambon E, Calafiore A, Marocchi M, Caponi A, Belluzzi A, Roda E. Intestinal epithelial cells in inflammatory bowel diseases. World J Gastroenterol 2010; 16(34): 4264-4271
- URL: https://www.wjgnet.com/1007-9327/full/v16/i34/4264.htm
- DOI: https://dx.doi.org/10.3748/wjg.v16.i34.4264
Inflammatory bowel diseases (IBDs) - mostly represented by Crohn’s disease (CD) and ulcerative colitis (UC) - are a group of inflammatory disorders of the gastrointestinal tract characterized by an abnormal immune response to antigens of the intestinal content that leads to a persistent inflammatory state[1].
Intestinal homeostasis in healthy subjects is ensured by a complex system called “intestinal barrier”, a dynamic structure that separates intestinal contents from the host tissues, regulates nutrient absorption and allows interactions between the resident bacterial flora and the mucosal immune system.
The intestinal barrier is composed of a thick mucus layer containing antimicrobial products, a monolayer of intestinal epithelial cells (IECs) and an underlying set of cells (mesenchymal cells, dendritic cells, lymphocytes and macrophages)[2].
IECs are exactly at the centre of this system because of their anatomical and functional position: on the luminal side they secrete and regulate the composition of the mucus layer, while on the basolateral side they interact and cross-talk with the underlying cells.
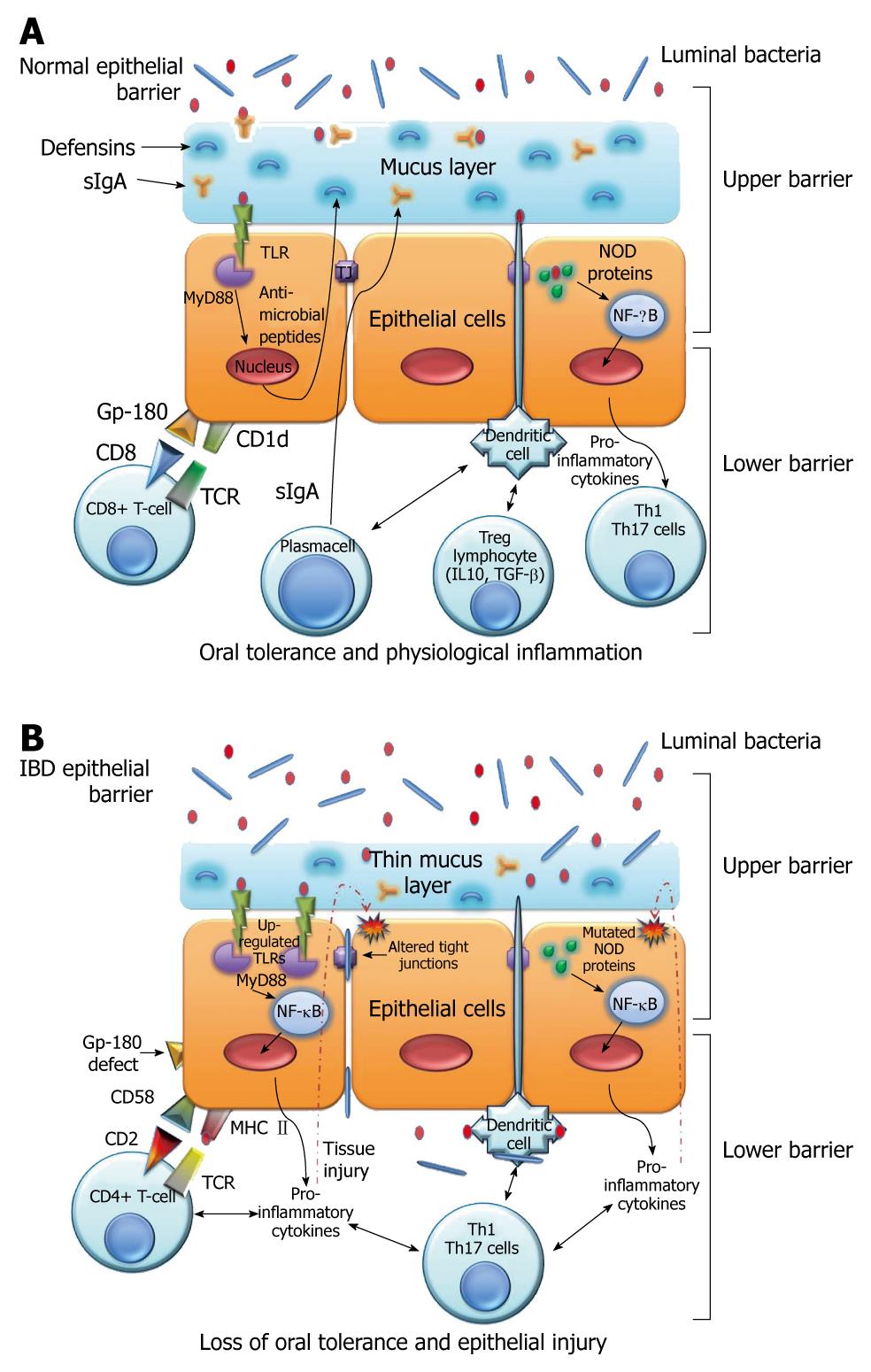
By putting the IECs at the centre of the barrier system we can divide it into an “upper barrier” and a “lower barrier”. The former constitutes a physical barrier, which prevents bacterial adhesion and paracellular diffusion to the underlying host tissues, and a functional barrier, which is able to discriminate commensal bacteria from pathogens; the latter operates by regulating antigen traffic through an intensive cross-talking with immune cells of the lamina propria (Figure 1A).
Given the importance of the epithelium in intestinal immune regulation mechanisms, it is clear how defects at one of these levels could be the primary pathogenetic mechanism that causes the loss of oral tolerance and therefore the establishment of an inflammatory response against luminal antigens, as happens in IBDs (Figure 1B).
Our purpose is to review the state of the art in understanding the key role played by IECs within the epithelial barrier system, in both healthy subjects and IBD patients.
The “upper barrier” is the intestinal epithelial single layer of columnar cells consisting of four IEC types: the absorbent enterocytes, the goblet cells, the Paneth cells and the enteroendocrine cells[3]. Upper barrier features are similar in small and large bowel. The main difference is constituted by the presence of elevations and projections (circular folds, villi and microvilli) in duodenum, jejunum and ileum that allows the increase of the absorption surface. This is not observed in the colon, which instead shows a flat surface.
Amongst the mucous membrane protrusions termed villi, there are inflections called crypts of Lieberkühn, which are distinct glandular invaginations.
Enterocytes are the most representative type of cells, which present finger-like projections, known as microvilli. These arise on the luminal side of the cells, constituting the so-called buffy coat, and are completely coated by glycocalyx and joined by apical junctional complexes, which prevent the entry of pathogens by keeping the cells tight.
In addition, the enterocyte monolayer is interrupted by the presence of goblet cells, secreting mucus, and by the presence of enteroendocrine cells that produce peptide hormones. These hormones are involved in cellular trophism, tissue repair, angiogenesis, enterocyte differentiation and polarization along the crypt-villus axis.
The epithelial cells derive from multipotent stem cells located at the base of the crypts. When these cells reach their maturity, they migrate toward the top of the villus, where aged cells are expelled in the intestinal lumen.
In the depth of the crypts there are also the Paneth cells that have regulatory functions since they produce antimicrobial peptides - called defensins - which constitute an inducible system against pathogens.
Hence, it is clear that the epithelial layer represents an anatomical and functional barrier, an upper interface, which is needed to maintain the whole intestinal homeostasis.
Evidence demonstrates that impairment of the upper barrier plays a key role in the pathogenesis of IBD. In fact, alterations of the mucus layer, disabled epithelial junctions, increased permeability and a defective production of antimicrobial peptides have been observed in CD as well in UC[2].
The upper barrier presents a mechanical external protection, which consists of a thick mucus layer (about 150 μm) synthesized by goblet cells and full of several molecular peptides. The latter allow mucus to act as a chemical boundary, preventing pathogen invasion.
The most important role of mucus in protecting the mucosa is carried out by its viscosity, due to the concomitant presence of glycosylated mucins and trefoil factors (TFFs) which modulate the defensive properties of the mucus layer, most likely through the check of mucin polymerization and by trapping microbes, which would otherwise be conveyed by the peristaltic process[4]. TFFs are a family of small, yet abundant, secreted proteins which maintain the epithelial continuity and restitution[5].
Furthermore, Mashimo et al[6] demonstrated that mice lacking intestinal trefoil factors (ITF) had impaired mucosal healing and died after oral administration of dextran sulfate sodium (DSS).
Taken together, these findings highlight the central role for TFFs in the maintenance and repair of the intestinal mucosa.
As mentioned previously, mucus is composed of a large and complex variety of molecules. The most representative element is mucin, a glycoprotein encoded by the MUC2 gene and synthesized by goblet cells. Another important component is constituted by trefoil factors (mainly TFF3), which are small protease-resistant peptides, secreted together with the mucin by the same kind of cells[7,8]. A recent finding demonstrates that TLR2 activation induces synthesis of TFF3 which protects the inflamed mucosa during acute intestinal injury, such as IBD[9].
The other constitutive elements of the mucus are secretory immunoglobulins, especially sIgA, produced by B lymphocytes; antimicrobial peptides such as defensins and lectins, secreted by Paneth cells; antimicrobial protease inhibitors, synthesized by epithelial cells; and enterocyte hydrophobic phospholipids[2,10].
In IBD, a substantial reduction of trefoil factors, which leads to the production of less viscous mucus, has been found. This fact proves that mucus viscosity and not its thickness, as thought previously, is the most important factor in the protection of the epithelium[11]. For instance, in CD we observe goblet cell hypertrophy with an increase of mucus production and a weakness of antimicrobial activity of defensins and peptides[12].
UC is instead characterised by a reduction of MUC2 expression, by a thinning of the mucus layer and by a decreased goblet cell number. Despite this, UC is often clinically represented by a mucus diarrhea, because of a worsening of mucus quality, probably due to an accumulation of non-glycosylated mucin[13].
These alterations could lead to a lessened capability of the mucus layer to limit antigenic traffic and bacterial translocation in the lamina propria, but it is still to be clarified if they are primary defects or secondary effects of the inflammatory state[14].
The absorbent IECs regulate intestinal permeability through the epithelial junctions, by limiting access of microbes to host tissues and mediating the antigenic traffic from the lumen to the lamina propria, where antigens are processed, presented and eliminated. The junctions consist of desmosomes, adherent junctions (AJs) and tight junctions (TJs), necessary for maintenance of intercellular adhesion and to regulate paracellular transport[3].
The adhesive junctional complexes are characterized by transmembrane proteins that interact with adjacent cells and with intracellular adaptor proteins, which are linked to the cytoskeleton. All together they form a connecting network[15].
The most representative structure of AJs is formed by cadherin-catenin interactions, which not only connect the junctional complex to the cellular cytoskeletal network but also help to maintain cell polarity by regulating epithelial migration and proliferation[16,17].
A dysfunction of AJ proteins has been described and consists of a down-regulation of E-cadherin, which weakens intercellular adhesion. This could be responsible for promoting intestinal inflammation, such as in IBD[18]. However, the most important impact on IBD pathogenesis seems to be the impairment of the tight junctions[19].
The tight junctions, located at the apical end of the intercellular space, consist of a complex structure composed of different proteins, such as hyperphosphorylated occludins, proteins of the zonula occludens and proteins of the claudin family[20]. Far from being static structures, tight junctions are highly regulated by cytokines, which play a central role in modulating intestinal barrier function.
Recent studies have showed that proinflammatory cytokines, such as interferon (IFN)-γ and tumor necrosis factor (TNF)-α, induce a downregulation of the constituent proteins of the tight junctions, mainly zonula occludens-1. This phenomenon can produce an actin-cytoskeletal disarrangement. Probably these proinflammatory cytokines induce internalization of the apical-junctional complex due to an increase in macropinocytosis of the tight junction proteins[21-23].
Other recent studies have demonstrated that IFN-γ and TNF-α can cause a reorganization of numerous tight junction proteins such as zonulin-1, JAM-A, occludin, claudin-1 and claudin-4, increasing intestinal permeability[24].
A downregulation of occludin, claudin-5 and claudin-8 has been found in CD; an upregulation of claudin-2 has been observed in UC. The expression of this pore-forming protein is due to the stimulation of IECs by interleukin (IL)-13. This cytokine induces an increase of barrier permeability and promotes the epithelial apoptotic events in UC[25-27].
However, the extent of defects in the barrier is more significant in UC than in CD, because of an early presence of apoptotic foci that degenerate into erosion and ulcer-type lesions, which has already occurred in the mild stage of disease[28,29].
Overall, the increase of proinflammatory cytokines leads to an impairment of tight junctions and consequently to a loss of barrier function.
Transforming growth factor (TGF)-β, prototype of anti-inflammatory cytokines and produced by Th3 regulatory cells, can preserve the integrity of the tight junctions by acting directly on IECs, probably through a cAMP-dependent mechanism[30]. Hence, regulatory T cells are involved not only in suppressing inflammatory responses but also in preserving the integrity of the tight junctions.
Many studies have shown that TGF-β also plays a role in epithelial restitution, which occurs after an injury, and its secretion is promoted by the wounded epithelium itself[31].
The intestinal mucosa produces antimicrobial peptides, called defensins, which contribute to maintaining host immunity and protect from pathological flora. The antimicrobial activity of defensins is expressed by the formation of micropores in the bacterial membranes that cause the lost of pathogen integrity.
In IBD there is a deficiency in defensin expression; however it is not clear whether this alteration contributes to the pathogenesis or is a secondary phenomenon[32].
The gastrointestinal mucosa produces ten types of defensins that help protect the epithelium from microbes. They play an important role, especially in protecting epithelial stem cells, thanks to their location in small bowel at the base of the crypts of Lieberkühn[33].
Defensins can be differentiated into two groups: α-defensins and β-defensins. Their type-expression is modulated along the different intestinal sections. For instance, α-defensins (HD) - especially HD5 and HD6 - are synthesized by Paneth cells positioned in the crypts, while β-defensins (HBD) are produced by colonic epithelial cells[34,35].
These peptides are produced as pro-peptides, a precursor form that needs an enzymatic digestion by trypsin to turn them into their active form. This process is necessary to allow a conformational folding of these proteins, helpful to accomplish their function.
Evidence demonstrates that in CD there is a defective expression of HD5 and HD6 with a release of non-functional peptide that forms an unfolded structure, probably due to a defective enzymatic digestion[36]. A decreased concentration of defensins is responsible for the presence of a less efficient mucus membrane as a biochemical barrier for pathogenic bacteria.
The cause of deficient defensin production has not been clearly determined but the NOD2 signalling pathway could be involved in this process. In fact, NOD2 receptors are highly expressed in Paneth cells and an association between NOD2 gene mutation and a reduced expression of HD5 mRNA in Paneth cells of CD patients has been found by Wehkamp et al[37]. However, it should be pointed out that the primary cause of the α-defensin deficiency is due to an epithelial cell loss[38].
As stated above, HBD are produced by colonic epithelial cells. The type HBD1 is constitutively expressed in all subjects and its concentration does not change, in spite of the presence of inflammatory cytokines or bacteria. In contrast, HBD3 and HBD4 are minimally represented in normal intestinal mucosa and their expression is maintained in CD, and increased in UC. The most important difference in IBD patients with respect to controls is provided by HBD2 production, which is significantly increased in these patients, especially in active UC. This fact is probably due to an increase of pro-inflammatory cytokines[39].
Conversely, in CD, it seems that genetic factors induce a lower expression of inducible β-defensins by means of a suppression of nuclear factor (NF)-κB, which is caused by a direct mechanism or a NOD2 mutation[40].
The innate immune system is able to recognize a limited set of conserved bacteria and viral motifs known as pathogen-associated molecular patterns (PAMPs), through pattern recognition receptors (PRRs), including above all the Toll-like receptors (TLRs) and the nucleotide-binding oligomerization domain (NOD) families.
TLRs are a family of receptors which recognize specific PAMPs and activate signal transduction through the NF-κB pathway. As a consequence, a pro-inflammatory cascade initiates to induce cytokine and chemokine genes[41]. Activation of the TLR pathway occurs through an adapter molecule, myeloid differentiation factor 88 (MyD88)[42,43].
IECs express several members of the TLR family: TLR2, which recognizes peptidoglycan (PGN), a component of the bacterial cell walls of Gram+ bacteria; TLR3, a receptor for viral double-stranded RNA; TLR4, which recognizes lipopolysaccharide (LPS), the major component of the outer membrane of Gram- bacteria; and finally, TLR5, which binds bacterial flagellin. TLR1, TLR3 and TLR4 are all located on the apical surface of IECs; alternatively, TLR5 is restricted to the basolateral surface of IECs and it is only activated when bacteria invade the epithelium[42].
These findings suggest that PRRs are positioned in order to trigger a response in the event of bacterial penetration of the epithelium. Therefore, pathogens, which unlike commensals are able to penetrate the barrier, are recognized by basolateral TLRs[44]. Moreover, TLRs bind saturated fatty acids in acetylated form, which are essential for the agonistic activity.
Wolowczuk et al[45] report that saturated fatty acids are able to induce the activation of TLR2 and TLR4, whereas unsaturated fatty acids - such as PUFAs - inhibit the TLR-mediated signaling pathway and gene expression by suppressing NF-κB activation and inflammation. These data suggest the protective role of unsaturated fatty acids such as omega-3 and the regulation of immune responses by fatty acid types.
Recognition by TLRs protects against pathogens and is carefully regulated to shut down a proinflammatory response to commensal organisms[41].
An interesting observation is that various TLRs are also expressed in cells of the adaptive immune system, such as B and T cells and dendritic cells (DCs), inducing differentiation and cytokine production, connecting innate to adaptive immunity. We can consequently consider the TLRs as a link from upper to lower barrier function.
For all these reasons it is clear that TLRs may have a dual role. Under normal conditions they maintain oral tolerance and eliminate pathogens, while in IBD they can also amplify inappropriate immune responses.
Several polymorphisms of TLRs have been associated with IBD, such as those of TLR1, TLR2, TLR4, TLR5, TLR6 and TLR9. However, the functional effects of these variants are not well defined[43].
Szebeni et al[46] have demonstrated that in IECs of IBD patients, there is an abnormal production of certain TLR subtypes with a significant upregulation of TLR2 and TLR4 expression in the inflamed mucosa. These alterations could compromise the capability to distinguish commensals from pathogens, or amplify inappropriate immune responses. It is still to be clarified whether this upregulation is one of the causes of IBD or just one of the consequences or even a concomitant factor.
NOD2 is a gene encoding for a cytoplasmatic protein (also known as CARD15), which recognizes bacterial muramyl dipeptide (MDP). This is the major component of peptidoglycan (PGN) and it is present in both Gram+ and Gram- bacteria. The binding of MDP to NOD2 results in the activation of the NF-κB pathway and IL-12 production[47].
In recent years researchers have identified a large number of NOD2 polymorphisms (SNPs) and the most common are associated with susceptibility to CD[48].
It seems that NOD2 variations lead to an impaired intracellular microorganism recognition and a consequent perpetual nuclear translocation of NF-κB, which results in an inadequate phlogosis state.
Using transgenic mice, Watanabe et al[49] have demonstrated that mice overexpressing NOD2 exhibit greatly decreased IL-12 responses to systemic administration of PGN but not to LPS, indicating that defects in NOD2 signaling lead to excessive TLR2-dependent inflammatory responses. Indeed, under normal conditions, PGN from commensal bacteria leads to innate immune responses, which are subsequently made weak by NOD2 modulation and other regulatory responses[49]. In the case of a NOD2 signaling defect, a TLR2-dependent inflammatory response cannot be controlled, leading to mucosal injuries[50].
Strober’s group demonstrated that mice with cells with increased NOD2 function have decreased responses to TLR stimulation, resulting in protection against DSS-induced colitis. They showed that prestimulation of cells with NOD2 ligand renders them unresponsive to TLR stimulation, because of an inhibitor of TLR-induced inflammatory pathways (IRF4)[51,52].
Greater understanding of the relationship between NOD2 variations and the pathomechanisms of IBD is required, but recent studies indicate that these mutations could participate, together with other barrier dysfunctions described previously, in the progression to CD[48].
TLRs and NOD proteins are also key sensors of bacterial-epithelial interactions. The intestinal microbiota contribute to protecting the host against invasions by pathogenic bacteria, competing for nutrients and stimulating immune responses, and play a crucial role in correct epithelial cell development. In return, commensal bacteria derive benefits from this association with the host since they can inhabit a protected environment from which they receive nutrients[53].
Within the bacterial-epithelial interface there is a continuous cross-talk which is enabled by PRRs; components of the mammalian innate immune system continuously sample the composition of commensal communities. However, only pathogens can activate the innate immune system since they can survive within host tissues.
A recent study showed that the immune status of the host can influence the composition of the commensal community. For instance, in the intestinal epithelium of Drosophila melanogaster, the dysregulation of NF-κB-dependent expression of antimicrobial peptides results in the outgrowth of a pathogenic commensal community[54].
In turn, scientific evidence also shows that commensal bacteria modulate IEC function by inhibiting the NF-κB pathway, through blocking the ubiquitylation and degradation of IκB or by hijacking the peroxisome-proliferation-activated receptor-γ (PPARγ) pathway[44].
Several pathologic features of IBD suggest that they derive in part from dysregulated control of bacterial interactions with the mucosal surface. For example, as reported by Duerkop et al[55] demonstrated that IBD patients exhibit increased numbers of mucosal surface-associated bacteria. This evidence suggests a failure of the mechanisms to prevent the microbiota from direct contact with the surface epithelium.
In 2006, Mizoguchi found that chitinase 3-like1 (CHI3L1), a molecule characterized by a strong binding affinity to chitin (a polymer of N-acetylglucosamine richly found in microorganisms), is specifically up-regulated under intestinal inflammatory conditions; it plays a pathogenic role in acute colitis by enhancing bacterial adhesion and invasion into colonic epithelial cells[56].
Many studies have investigated the presence of specific potentially pathogenic microorganisms in the mucosa of IBD patients; Darfeuille-Michaud et al[57] have demonstrated the association between ileal CD and adherent-invasive Escherichia coli (AIEC), not only as secondary invaders but also as possibly responsible for the initiation of the inflammatory process.
Moreover, it was demonstrated that CD-associated AIEC strains adhere to the brush border of primary ileal enterocytes isolated from CD patients but not from controls; AIEC adhesion is dependent on type 1 pili expression on the bacterial surface and on carcinoembryonic antigen (CEA)-related cell adhesion molecule 6 (CEACAM6) expression on the apical surface of ileal epithelial cells in CD patients. CEACAM6 is up-regulated in intestinal epithelial cells of CD patients and acts as a receptor for AIEC adhesion. Finally, this study suggests that AIEC can promote its own colonization in CD patients, since it is able to increase CEACAM6 expression in infected epithelial cells[58].
Recent advances regarding an active role of intestinal epithelial cells within the mucosal immune system have revealed that they act as non-professional antigen presenting cells (APCs), activating subsets of T-cells with regulatory function. On the other hand, lamina propria lymphocytes are able to influence epithelial cell growth and differentiation along the crypt/villus axis by mediating intercellular interactions and by secreting cytokines and other factors[41]. In IBD patients, the cross-talking between IECs and mucosal lymphocytes is changed by altered production of these factors. This complicated dialogue contributes to promoting mucosal inflammation.
IECs are able to directly process and present antigens to lymphocytes by a highly polarized system with apical antigen sorting, processing and exclusively basolateral presentation.
IECs do not express conventional costimulatory molecules such as CD80 and CD86, but express new B7 family members, such as B7h [inducible costimulatory ligand (ICOS-L)] and B7-H1 [programmed death ligand (PD-1 L)], as well as several non-classical MHC class Ib molecules such as MICA/B, HLA-E, and CD1d[42,54].
By using in vitro IEC: peripheral blood T-cells (PBT) co-cultures, Allez et al[59,60] have demonstrated that intestinal epithelial cells preferentially activate CD8+ regulatory T-cells, in particular a CD28- and CD101/CD103+ subset, characterized by the biased usage of the T cell receptor (TCR) Vβ5.1 chain. These data have been confirmed by the same group in vivo by using CD8+ T-cells isolated from the lamina propria (LP) of healthy subjects.
Hence, under normal conditions, luminal antigens presented by IECs cause a suppression rather than an increase of the immune response.
For the activation of the restricted regulatory T-cell subset, the role is crucial of a unique complex formed by gp-180 (a CEA family member glycoprotein) and by CD1d, which bind CD8 and the TCR on the T-cell surface, respectively. Indeed, blocking gp-180 with a monoclonal antibody (B9) suppresses the proliferation of regulatory T-cells[61-63]. This helps to explain the oral tolerance and controlled inflammation phenomena.
Results from both in vitro IEC:PBT co-cultures and from in vivo within the lamina propria of IBD patients demonstrated a reduced amount of CD8+ regulatory T cells, that might be linked to glycoprotein gp-180. Indeed, the frequency of Vβ5.1 cells among the LP CD8+ T cells is significantly decreased in IBD patients with respect to healthy subjects[64].
In IBD IECs, especially in CD, the same group observed a defective expression of gp-180, and moreover, IECs from IBD patients preferentially stimulate CD4+ T-cell proliferation and secretion of IFN-γ, through MHC class II[65,66].
In a subsequent study, by using freshly isolated IECs and lamina propria lymphocytes (LPLs), as well as T84 cell lines, we have correlated SOX9 to CEACAM5 gene expression: SOX9 is able to downregulate CEACAM5. The former is a transcription factor involved in the differentiation of several tissues such as chondrocytes, male gonads, neural crest and spinal cord glial cells, while the latter is a member of the CEA family. We speculate that LPLs, the main source of cytokines within the gastrointestinal mucosa-associated lymphoid tissue (GALT), influence the nuclear translocation of SOX9 in IECs and consequently the downregulation of the CEA family member gp-180, together with a lack of activation and/or expansion of regulatory cells[67,68].
As mentioned previously, the described cross-talk between epithelium and LPLs has also a role from the standpoint of IECs, in particular affecting their proliferation and differentiation along the crypt/villus axis in the colon. Indeed, starting from the concept that IECs can promote regulatory T-cell responses in the mucosa, Dahan et al[41] have demonstrated that lympho-epithelial interactions occur and play a role in IBD. By using freshly isolated LPLs derived from healthy subjects and CD patients, they suggest that cross-talk leads to an enhanced IEC differentiation, a pattern restricted to CD, which seems to involve the transcription factor CDX2 and PI3K/p38 MAPK pathways.
Moreover, T84 cells co-cultured with CD LPLs display a greater increase of differentiation and CDX2 mRNA levels with respect to normal LPLs.
These data were confirmed in vivo, through immunostaining both in human colonic mucosa and in RAG1-/- mice lacking lymphocytes; studies in which an altered IEC differentiation was observed[41,69,70].
Our purpose was to review the state of the art in understanding the key role of intestinal epithelial cells in maintaining gut homeostasis and the possible role in the pathogenesis of IBD.
Intestinal epithelial cells, because they act as a functional barrier and as non-professional antigen presenting cells, represent important elements in the development and maintenance of immune oral tolerance.
In IBD, we observe a global defect in the mucosal immune system: barrier function, innate and adaptive responses.
Two main strands of research on these defects exist: one is focused on the impairment of the epithelial barrier, the other on defects of epithelial-lymphocyte cross-talk. In both these lines of investigation, IECs occupy a prominent place within the complex and dynamic system of the intestinal barrier. Despite much progress in this area of research, it remains to be clarified whether defects involving IECs are fundamental or a consequence of abnormal signals coming from the lamina propria.
A better understanding of these regulatory mechanisms, which allow us to see intestinal epithelial cells at the interface between an “upper” and “lower” barrier, could help to identify new therapeutic targets.
Peer reviewer: Emiko Mizoguchi, MD, PhD, Department of Medicine, Gastrointestinal Unit, GRJ 702, Massachusetts General Hospital, Boston, MA 02114, United States
S- Editor Tian L L- Editor Logan S E- Editor Ma WH
| 1. | Scaldaferri F, Fiocchi C. Inflammatory bowel disease: progress and current concepts of etiopathogenesis. J Dig Dis. 2007;8:171-178. |
| 2. | McGuckin MA, Eri R, Simms LA, Florin TH, Radford-Smith G. Intestinal barrier dysfunction in inflammatory bowel diseases. Inflamm Bowel Dis. 2009;15:100-113. |
| 3. | Laukoetter MG, Nava P, Nusrat A. Role of the intestinal barrier in inflammatory bowel disease. World J Gastroenterol. 2008;14:401-407. |
| 4. | Atuma C, Strugala V, Allen A, Holm L. The adherent gastrointestinal mucus gel layer: thickness and physical state in vivo. Am J Physiol Gastrointest Liver Physiol. 2001;280:G922-G929. |
| 5. | Taupin D, Podolsky DK. Trefoil factors: initiators of mucosal healing. Nat Rev Mol Cell Biol. 2003;4:721-732. |
| 6. | Mashimo H, Wu DC, Podolsky DK, Fishman MC. Impaired defense of intestinal mucosa in mice lacking intestinal trefoil factor. Science. 1996;274:262-265. |
| 7. | Thim L. Trefoil peptides: from structure to function. Cell Mol Life Sci. 1997;53:888-903. |
| 8. | Madsen J, Nielsen O, Tornøe I, Thim L, Holmskov U. Tissue localization of human trefoil factors 1, 2, and 3. J Histochem Cytochem. 2007;55:505-513. |
| 9. | Podolsky DK, Gerken G, Eyking A, Cario E. Colitis-associated variant of TLR2 causes impaired mucosal repair because of TFF3 deficiency. Gastroenterology. 2009;137:209-220. |
| 10. | Mayer L. Mucosal immunity. Pediatrics. 2003;111:1595-1600. |
| 11. | Swidsinski A, Sydora BC, Doerffel Y, Loening-Baucke V, Vaneechoutte M, Lupicki M, Scholze J, Lochs H, Dieleman LA. Viscosity gradient within the mucus layer determines the mucosal barrier function and the spatial organization of the intestinal microbiota. Inflamm Bowel Dis. 2007;13:963-970. |
| 12. | Trabucchi E, Mukenge S, Baratti C, Colombo R, Fregoni F, Montorsi W. Differential diagnosis of Crohn’s disease of the colon from ulcerative colitis: ultrastructure study with the scanning electron microscope. Int J Tissue React. 1986;8:79-84. |
| 13. | Tytgat KM, van der Wal JW, Einerhand AW, Büller HA, Dekker J. Quantitative analysis of MUC2 synthesis in ulcerative colitis. Biochem Biophys Res Commun. 1996;224:397-405. |
| 14. | Welcker K, Martin A, Kölle P, Siebeck M, Gross M. Increased intestinal permeability in patients with inflammatory bowel disease. Eur J Med Res. 2004;9:456-460. |
| 15. | Groschwitz KR, Hogan SP. Intestinal barrier function: molecular regulation and disease pathogenesis. J Allergy Clin Immunol. 2009;124:3-20; quiz 21-22. |
| 16. | Ebnet K. Organization of multiprotein complexes at cell-cell junctions. Histochem Cell Biol. 2008;130:1-20. |
| 17. | Hartsock A, Nelson WJ. Adherens and tight junctions: structure, function and connections to the actin cytoskeleton. Biochim Biophys Acta. 2008;1778:660-669. |
| 18. | Hermiston ML, Gordon JI. In vivo analysis of cadherin function in the mouse intestinal epithelium: essential roles in adhesion, maintenance of differentiation, and regulation of programmed cell death. J Cell Biol. 1995;129:489-506. |
| 19. | Clayburgh DR, Shen L, Turner JR. A porous defense: the leaky epithelial barrier in intestinal disease. Lab Invest. 2004;84:282-291. |
| 20. | Bruewer M, Luegering A, Kucharzik T, Parkos CA, Madara JL, Hopkins AM, Nusrat A. Proinflammatory cytokines disrupt epithelial barrier function by apoptosis-independent mechanisms. J Immunol. 2003;171:6164-6172. |
| 21. | MacDonald TT, Hutchings P, Choy MY, Murch S, Cooke A. Tumour necrosis factor-alpha and interferon-gamma production measured at the single cell level in normal and inflamed human intestine. Clin Exp Immunol. 1990;81:301-305. |
| 22. | Ye D, Ma I, Ma TY. Molecular mechanism of tumor necrosis factor-alpha modulation of intestinal epithelial tight junction barrier. Am J Physiol Gastrointest Liver Physiol. 2006;290:G496-G504. |
| 23. | Mankertz J, Tavalali S, Schmitz H, Mankertz A, Riecken EO, Fromm M, Schulzke JD. Expression from the human occludin promoter is affected by tumor necrosis factor alpha and interferon gamma. J Cell Sci. 2000;113:2085-2090. |
| 24. | Shao L, Serrano D, Mayer L. The role of epithelial cells in immune regulation in the gut. Semin Immunol. 2001;13:163-176. |
| 25. | Zeissig S, Bürgel N, Günzel D, Richter J, Mankertz J, Wahnschaffe U, Kroesen AJ, Zeitz M, Fromm M, Schulzke JD. Changes in expression and distribution of claudin 2, 5 and 8 lead to discontinuous tight junctions and barrier dysfunction in active Crohn's disease. Gut. 2007;56:61-72. |
| 26. | Heller F, Florian P, Bojarski C, Richter J, Christ M, Hillenbrand B, Mankertz J, Gitter AH, Bürgel N, Fromm M. Interleukin-13 is the key effector Th2 cytokine in ulcerative colitis that affects epithelial tight junctions, apoptosis, and cell restitution. Gastroenterology. 2005;129:550-564. |
| 27. | Turner JR. Intestinal mucosal barrier function in health and disease. Nat Rev Immunol. 2009;9:799-809. |
| 28. | Schulzke JD, Ploeger S, Amasheh M, Fromm A, Zeissig S, Troeger H, Richter J, Bojarski C, Schumann M, Fromm M. Epithelial tight junctions in intestinal inflammation. Ann N Y Acad Sci. 2009;1165:294-300. |
| 29. | Gitter AH, Bendfeldt K, Schulzke JD, Fromm M. Leaks in the epithelial barrier caused by spontaneous and TNF-alpha-induced single-cell apoptosis. FASEB J. 2000;14:1749-1753. |
| 30. | Planchon S, Fiocchi C, Takafuji V, Roche JK. Transforming growth factor-beta1 preserves epithelial barrier function: identification of receptors, biochemical intermediates, and cytokine antagonists. J Cell Physiol. 1999;181:55-66. |
| 31. | Göke M, Kanai M, Lynch-Devaney K, Podolsky DK. Rapid mitogen-activated protein kinase activation by transforming growth factor alpha in wounded rat intestinal epithelial cells. Gastroenterology. 1998;114:697-705. |
| 32. | Wehkamp J, Schmid M, Stange EF. Defensins and other antimicrobial peptides in inflammatory bowel disease. Curr Opin Gastroenterol. 2007;23:370-378. |
| 33. | Ouellette AJ, Bevins CL. Paneth cell defensins and innate immunity of the small bowel. Inflamm Bowel Dis. 2001;7:43-50. |
| 34. | Ramasundara M, Leach ST, Lemberg DA, Day AS. Defensins and inflammation: the role of defensins in inflammatory bowel disease. J Gastroenterol Hepatol. 2009;24:202-208. |
| 35. | Cunliffe RN. Alpha-defensins in the gastrointestinal tract. Mol Immunol. 2003;40:463-467. |
| 36. | Tanabe H, Ayabe T, Maemoto A, Ishikawa C, Inaba Y, Sato R, Moriichi K, Okamoto K, Watari J, Kono T. Denatured human alpha-defensin attenuates the bactericidal activity and the stability against enzymatic digestion. Biochem Biophys Res Commun. 2007;358:349-355. |
| 37. | Wehkamp J, Harder J, Weichenthal M, Schwab M, Schäffeler E, Schlee M, Herrlinger KR, Stallmach A, Noack F, Fritz P. NOD2 (CARD15) mutations in Crohn‘s disease are associated with diminished mucosal alpha-defensin expression. Gut. 2004;53:1658-1664. |
| 38. | Lala S, Ogura Y, Osborne C, Hor SY, Bromfield A, Davies S, Ogunbiyi O, Nuñez G, Keshav S. Crohn’s disease and the NOD2 gene: a role for paneth cells. Gastroenterology. 2003;125:47-57. |
| 39. | Wehkamp J, Harder J, Weichenthal M, Mueller O, Herrlinger KR, Fellermann K, Schroeder JM, Stange EF. Inducible and constitutive beta-defensins are differentially expressed in Crohn’s disease and ulcerative colitis. Inflamm Bowel Dis. 2003;9:215-223. |
| 40. | Voss E, Wehkamp J, Wehkamp K, Stange EF, Schröder JM, Harder J. NOD2/CARD15 mediates induction of the antimicrobial peptide human beta-defensin-2. J Biol Chem. 2006;281:2005-2011. |
| 41. | Dahan S, Roth-Walter F, Arnaboldi P, Agarwal S, Mayer L. Epithelia: lymphocyte interactions in the gut. Immunol Rev. 2007;215:243-253. |
| 42. | Shao L, Kamalu O, Mayer L. Non-classical MHC class I molecules on intestinal epithelial cells: mediators of mucosal crosstalk. Immunol Rev. 2005;206:160-176. |
| 43. | Himmel ME, Hardenberg G, Piccirillo CA, Steiner TS, Levings MK. The role of T-regulatory cells and Toll-like receptors in the pathogenesis of human inflammatory bowel disease. Immunology. 2008;125:145-153. |
| 44. | Artis D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat Rev Immunol. 2008;8:411-420. |
| 45. | Wolowczuk I, Verwaerde C, Viltart O, Delanoye A, Delacre M, Pot B, Grangette C. Feeding our immune system: impact on metabolism. Clin Dev Immunol. 2008;2008:639803. |
| 46. | Szebeni B, Veres G, Dezsõfi A, Rusai K, Vannay A, Mraz M, Majorova E, Arató A. Increased expression of Toll-like receptor (TLR) 2 and TLR4 in the colonic mucosa of children with inflammatory bowel disease. Clin Exp Immunol. 2008;151:34-41. |
| 47. | Xavier RJ, Podolsky DK. Unravelling the pathogenesis of inflammatory bowel disease. Nature. 2007;448:427-434. |
| 48. | Cho JH. The genetics and immunopathogenesis of inflammatory bowel disease. Nat Rev Immunol. 2008;8:458-466. |
| 49. | Watanabe T, Asano N, Murray PJ, Ozato K, Tailor P, Fuss IJ, Kitani A, Strober W. Muramyl dipeptide activation of nucleotide-binding oligomerization domain 2 protects mice from experimental colitis. J Clin Invest. 2008;118:545-559. |
| 50. | Netea MG, Kullberg BJ, de Jong DJ, Franke B, Sprong T, Naber TH, Drenth JP, Van der Meer JW. NOD2 mediates anti-inflammatory signals induced by TLR2 ligands: implications for Crohn’s disease. Eur J Immunol. 2004;34:2052-2059. |
| 51. | Strober W, Kitani A, Fuss I, Asano N, Watanabe T. The molecular basis of NOD2 susceptibility mutations in Crohn’s disease. Mucosal Immunol. 2008;1 Suppl 1:S5-S9. |
| 52. | Strober W. The multifaceted influence of the mucosal microflora on mucosal dendritic cell responses. Immunity. 2009;31:377-388. |
| 53. | Ryu JH, Kim SH, Lee HY, Bai JY, Nam YD, Bae JW, Lee DG, Shin SC, Ha EM, Lee WJ. Innate immune homeostasis by the homeobox gene caudal and commensal-gut mutualism in Drosophila. Science. 2008;319:777-782. |
| 54. | Nakazawa A, Dotan I, Brimnes J, Allez M, Shao L, Tsushima F, Azuma M, Mayer L. The expression and function of costimulatory molecules B7H and B7-H1 on colonic epithelial cells. Gastroenterology. 2004;126:1347-1357. |
| 55. | Duerkop BA, Vaishnava S, Hooper LV. Immune responses to the microbiota at the intestinal mucosal surface. Immunity. 2009;31:368-376. |
| 56. | Mizoguchi E. Chitinase 3-like-1 exacerbates intestinal inflammation by enhancing bacterial adhesion and invasion in colonic epithelial cells. Gastroenterology. 2006;130:398-411. |
| 57. | Darfeuille-Michaud A, Boudeau J, Bulois P, Neut C, Glasser AL, Barnich N, Bringer MA, Swidsinski A, Beaugerie L, Colombel JF. High prevalence of adherent-invasive Escherichia coli associated with ileal mucosa in Crohn’s disease. Gastroenterology. 2004;127:412-421. |
| 58. | Barnich N, Carvalho FA, Glasser AL, Darcha C, Jantscheff P, Allez M, Peeters H, Bommelaer G, Desreumaux P, Colombel JF. CEACAM6 acts as a receptor for adherent-invasive E. coli, supporting ileal mucosa colonization in Crohn disease. J Clin Invest. 2007;117:1566-1574. |
| 59. | Allez M, Brimnes J, Dotan I, Mayer L. Expansion of CD8+ T cells with regulatory function after interaction with intestinal epithelial cells. Gastroenterology. 2002;123:1516-1526. |
| 60. | Allez M, Brimnes J, Shao L, Dotan I, Nakazawa A, Mayer L. Activation of a unique population of CD8(+) T cells by intestinal epithelial cells. Ann N Y Acad Sci. 2004;1029:22-35. |
| 61. | Yio XY, Mayer L. Characterization of a 180-kDa intestinal epithelial cell membrane glycoprotein, gp180. A candidate molecule mediating t cell-epithelial cell interactions. J Biol Chem. 1997;272:12786-12792. |
| 62. | Campbell NA, Kim HS, Blumberg RS, Mayer L. The nonclassical class I molecule CD1d associates with the novel CD8 ligand gp180 on intestinal epithelial cells. J Biol Chem. 1999;274:26259-26265. |
| 63. | Campbell NA, Park MS, Toy LS, Yio XY, Devine L, Kavathas P, Mayer L. A non-class I MHC intestinal epithelial surface glycoprotein, gp180, binds to CD8. Clin Immunol. 2002;102:267-274. |
| 64. | Brimnes J, Allez M, Dotan I, Shao L, Nakazawa A, Mayer L. Defects in CD8+ regulatory T cells in the lamina propria of patients with inflammatory bowel disease. J Immunol. 2005;174:5814-5822. |
| 65. | Toy LS, Yio XY, Lin A, Honig S, Mayer L. Defective expression of gp180, a novel CD8 ligand on intestinal epithelial cells, in inflammatory bowel disease. J Clin Invest. 1997;100:2062-2071. |
| 66. | Dotan I, Allez M, Nakazawa A, Brimnes J, Schulder-Katz M, Mayer L. Intestinal epithelial cells from inflammatory bowel disease patients preferentially stimulate CD4+ T cells to proliferate and secrete interferon-gamma. Am J Physiol Gastrointest Liver Physiol. 2007;292:G1630-G1640. |
| 67. | Roda G, Dahan S, Mezzanotte L, Caponi A, Roth-Walter F, Pinn D, Mayer L. Defect in CEACAM family member expression in Crohn’s disease IECs is regulated by the transcription factor SOX9. Inflamm Bowel Dis. 2009;15:1775-1783. |
| 68. | Jay P, Berta P, Blache P. Expression of the carcinoembryonic antigen gene is inhibited by SOX9 in human colon carcinoma cells. Cancer Res. 2005;65:2193-2198. |
| 69. | Dahan S, Roda G, Pinn D, Roth-Walter F, Kamalu O, Martin AP, Mayer L. Epithelial: lamina propria lymphocyte interactions promote epithelial cell differentiation. Gastroenterology. 2008;134:192-203. |
| 70. | Dahan S, Roth-Walter F, Martin AP, Arnaboldi P, Mayer L. Lymphoepithelial interactions: a new paradigm. Ann N Y Acad Sci. 2009;1165:323-326. |