Published online Jun 7, 2009. doi: 10.3748/wjg.15.2679
Revised: April 13, 2009
Accepted: April 20, 2009
Published online: June 7, 2009
The author reports herein two cases of amelanotic malignant melanoma of the esophagus. Case 1 is an 87-year-old woman who was admitted to our hospital because of nausea and vomiting. Endoscopic examination revealed an ulcerated tumor of the distal esophagus, and a biopsy was taken. The biopsy showed malignant polygonal and spindle cells. No melanin pigment was recognized. Immunohistochemically, the tumor cells were positive for melanosome (HMB45), S100 protein, KIT and Platelet derived growth factor receptor-α (PDGFRA). The patient was treated by chemotherapy and radiation, but died of systemic metastasis 12 mo after the presentation. Case 2 is a 56-year-old man presenting with dysphagia. Endoscopic examination revealed a polypoid tumor in the middle esophagus, and a biopsy was obtained. The biopsy showed malignant spindle cells without melanin pigment. Immunohistochemically, the tumor cells were positively labeled for melanosome, S100 protein, KIT and PDGFRA. The patient refused operation, and was treated by palliative chemotherapy and radiation. He died of metastasis 7 mo after the admission. In both cases, molecular genetic analyses of KIT gene (exons 9, 11, 13 and 17) and PDGFRA gene (exons 12 and 18) were performed by the PCR direct sequencing method, which showed no mutations of KIT and PDGFRA genes. This is the first report of esophageal malignant melanoma with an examination of the expression of KIT and PDGFRA and the mutational status of KIT and PDGFRA genes.
-
Citation: Terada T. Amelanotic malignant melanoma of the esophagus: Report of two cases with immunohistochemical and molecular genetic study of
KIT andPDGFRA . World J Gastroenterol 2009; 15(21): 2679-2683 - URL: https://www.wjgnet.com/1007-9327/full/v15/i21/2679.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.2679
Primary malignant melanoma of the esophagus is very rare; only case reports and studies of small series have been reported[1–7]. Primary amelanotic malignant melanoma of the esophagus is extremely rare; only a few case reports have been published in the literature[8–13].
Melanoma is a highly aggressive tumor, and NRAS and BRAF mutations are mainly involved in the pathogenesis of melanoma[1415]. KIT gene, mapped to 4q12, encodes an oncogenic transmembranous receptor tyrosine kinase, KIT, whose ligand is stem cell factor[6–21]. The platelet derived growth factor receptor-α (PDGFRA) gene, also mapped to 4q12, additionally encodes an oncogenic transmembranous receptor tyrosine kinase, PDGFRA[6–21]. The KIT gene plays an important role in melanocyte migration, development, differentiation and tumorigenesis[22]. A few previous studies have shown that activating mutations of the KIT gene may lead to tumorigenesis of cutaneous melanoma[1423]. Since both KIT and PDGFRA genes are mapped to 4q12, it is anticipated that PDGFRA gene mutations are also involved in the tumorigenesis of melanoma, as in the case of gastrointestinal stromal tumors[16–21]. However, the incidence of PDGFRA gene mutations in melanoma has rarely been estimated[24].
The author herein reports two cases of esophageal amelanotic malignant melanoma with immunohistochemical and molecular genetic study of KIT and PDGFRA.
An 87-year-old woman was admitted to our hospital because of nausea and vomiting. Endoscopic examination revealed an ulcerated tumor of the distal esophagus, and a biopsy was taken. The biopsy was stained with HE. An immunohistochemical analysis was performed, using Dako’s Envision method, as previously described[25–30].
Genetic analyses of the KIT gene (exons 9, 11, 13 and 17) and the PDGFRA gene (exons 12 and 18) were performed by the PCR direct sequencing method, as previously reported[31–35]. The exons of both genes were selected because they are frequent mutation sites[16–21]. The primers are shown in Table 1. In brief, genomic DNA was extracted from paraffin blocks with proteinase K digestion and phenol/chloroform extraction, and subjected to PCR for 40 cycles (94°C for 1 min, 52°C for 1 min, 72°C for 1 min), using a thermal cycler (GeneAmp PCR system 9700, Applied Biosystems, ABI, CA). The annealing temperature was 53°C. PCR products were extracted, and subjected to computed automatic DNA sequencing (ABI PRIZM 3100 Genetic Analyzer, Applied Biosystems, ABI, CA).
| Forward | Reverse | |
| KIT exon 9 | 5’-TCCTAGAGTAAGCCAGGGCTT-3’ | 5’-TGGTAGACAGAGCCTAAACATCC-3’ |
| KIT exon11 | 5’-GATCTATTTTTCCCTTTCTC-3’ | 5’AGCCCCTGTTTCATACTGAC-3’ |
| KIT exon 13 | 5’-GCTTGACATCAGTTTGCCAG-3’ | 5’-AAAGGCAGCTTGGACACGGCTTTA-3’ |
| KIT exon 17 | 5’-CTCCTCCAACCTAATAGTGT-3’ | 5’-GTCAAGCAGAGAATGGGTAC-3’ |
| PDGFRA exon12 | 5’-TTGGATATTCACCAGTTACCTGTC-3’ | 5’-CAAGGGAAAAGCTCTTGG-3’ |
| PDGFRA exon 18 | 5’-ACCATGGATCAGCCAGTCTT-3’ | 5’-TGAAGGAGGATGAGCCTGACC-3’ |
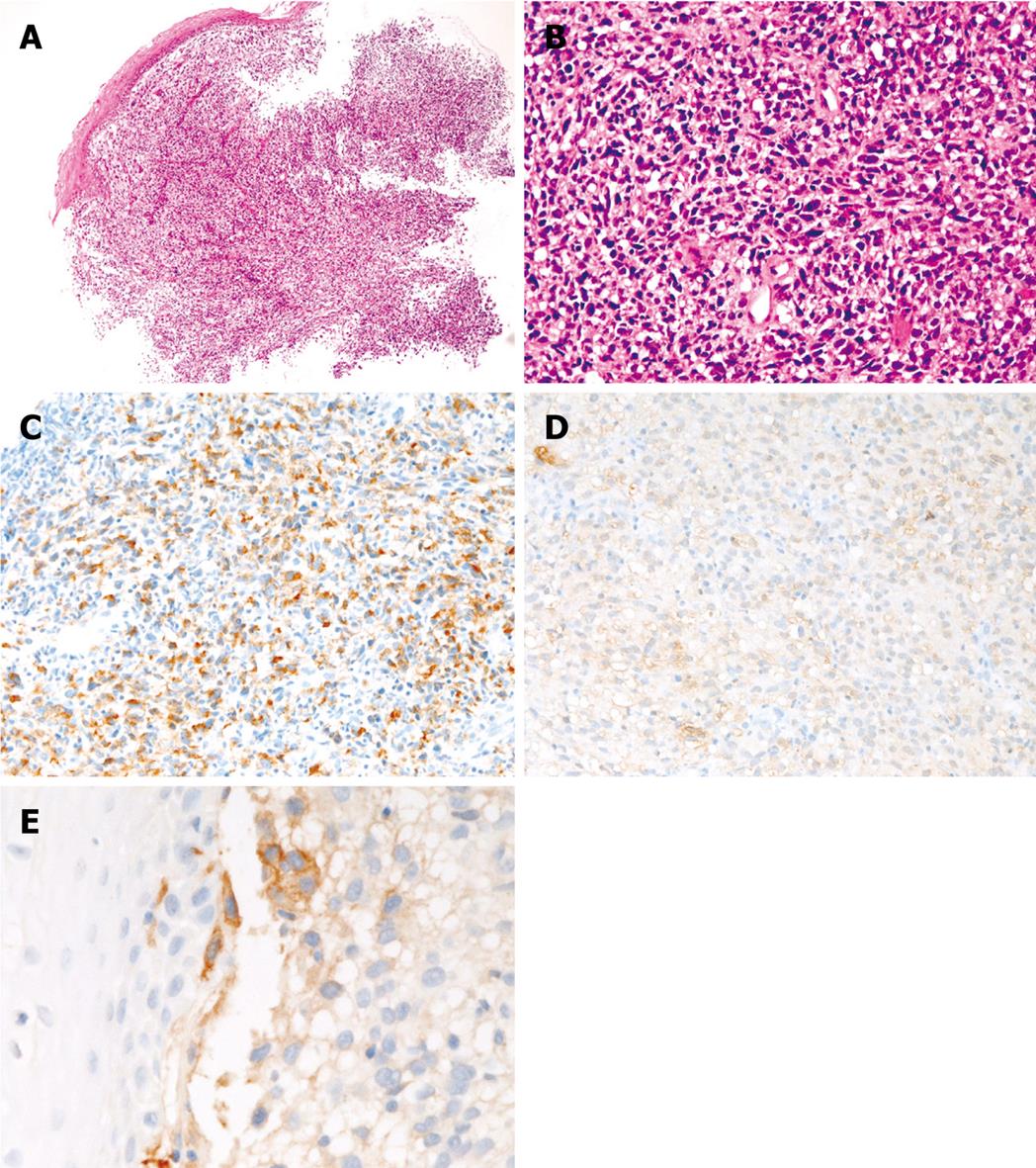
The biopsy showed malignant spindle and polygonal cells (Figure 1A and B) suspicious for sarcoma. No melanin pigment was recognized by HE and Masson-Fontana stains. The histology was relatively uniform in the biopsy. Immunohistochemically, the malignant cells were positive for melanosome (Figure 1C) (HMB45, Dako), S100 protein (Figure 1D) (polyclonal, Dako), vimentin (Vim 3B4, Dako), p53 protein (DO-7, Dako), neuron-specific enolase (BBS/NC/VI-H14, Dako), PDGFRA (Santa Cruz, CA, USA), and KIT (polyclonal, Dako) (Figure 1E). The KIT expression was focal. In contrast, the malignant cells were negative for cytokeratins (AE1/3 and polyclonal, Dako), CD3 (M7193, Dako), CD10 (M0727, Dako), CD15 (M0733, Dako), CD30 (M0751, Dako), CD45 (M0855, DAKO), CD45RO (UCHL-1, Dako, CD79α (M7050, Dako), CD20 (L26, Dako), desmin (D33, Dako), α-smooth muscle actin (1A4), CD34 (QBEND10, Dako), chromogranin (DAK-A3, Dako), synaptophysin (polyclonal, Dako), CD56 (MOC-1, Dako), and myoglobin (polyclonal, Dako). Ki-67 labeling (MIB1, Dako) was 80%. A pathologic diagnosis of amelanotic melanoma of the esophagus was made. The molecular genetic analysis showed no mutation of the KIT gene (exons 9, 11, 13 and 17) or the PDGFRA gene (exons 12 and 18). Examination of the skin, eye and intestine showed no tumors. Therefore, the esophageal melanoma was primary. The patient was inoperative because of weakness and old age, and chemotherapy and radiation were performed. The patient showed systemic metastasis, and died of respiratory failure 12 mo after the first presentation. One additional biopsy of the lung metastasis was obtained, and it showed amelanotic melanoma histology and immunohistochemistry results which were almost the same as those of the primary esophageal biopsy.
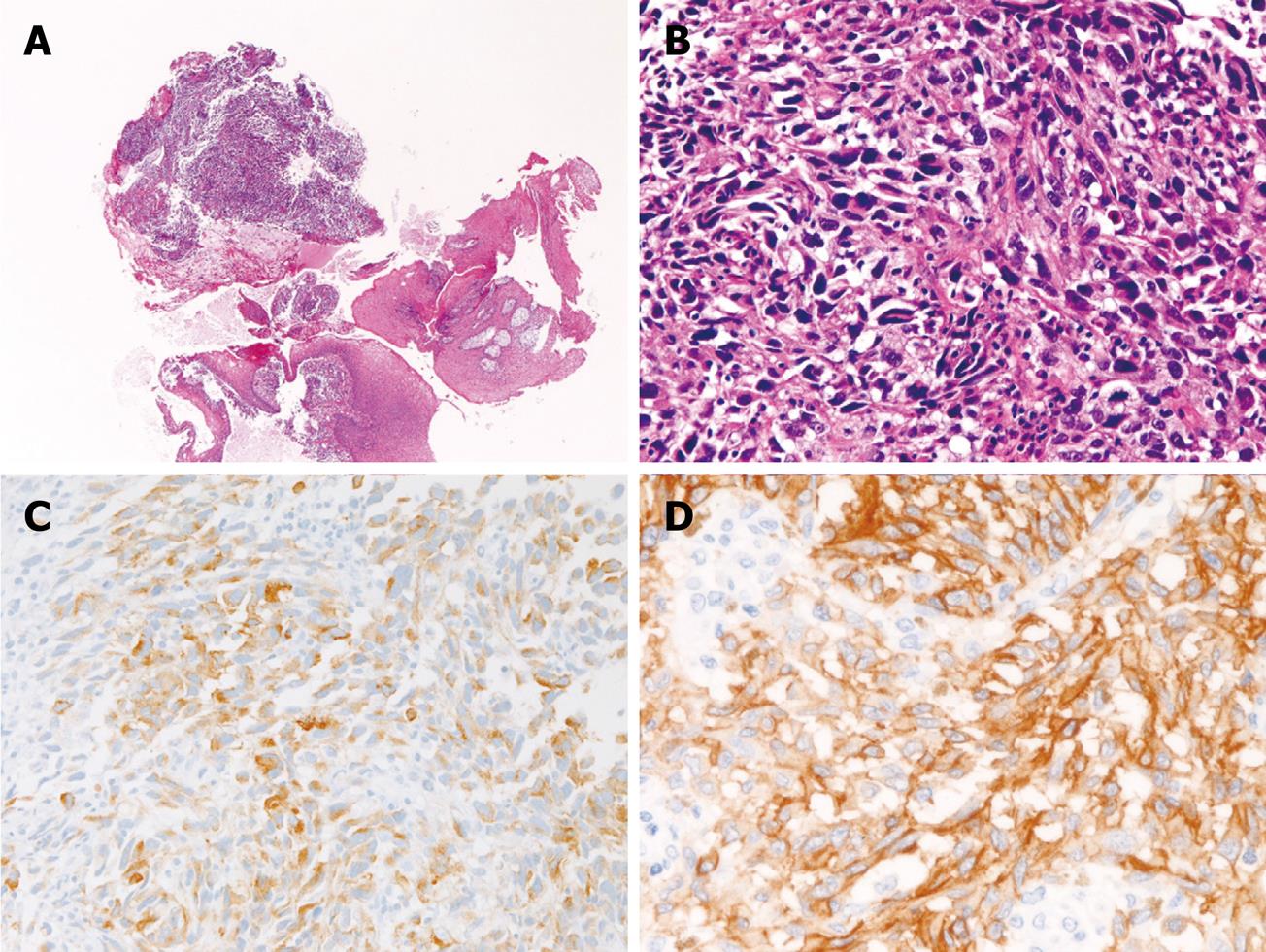
A 56-year-old man presented with dysphagia. Endoscopic examination revealed a polypoid tumor in the middle esophagus. A biopsy was taken (Figure 2A and B). Histologically, the biopsy showed proliferation of malignant spindle cells (Figure 2A and B). No melanin pigment was seen with HE and Masson-Fontana stains. The histology of the tumor was relatively uniform in the biopsy. Immunohistochemically, the tumor cells were positive for melanosome (Figure 2C), S100 protein, vimentin, p53 protein, PDGFRA, and KIT (Figure 2D). The KIT expression was diffuse. In contrast, the malignant cells were negative for cytokeratins, CD3, CD30, CD45, CD45RO, CD79α, CD20, desmin, α-smooth muscle actin, CD34, chromogranin, synaptophysin, CD56, and myoglobin. Ki-67 labeling was 95%. A pathological diagnosis of amelanotic malignant melanoma was made. The molecular genetic analysis identified no mutation of either KIT gene (exons 9, 11, 13 and 17) or PDGFRA gene (exons 12 and 18). No tumors were identified in the skin, eye and mucosal membrane. Therefore, the esophageal melanoma was considered primary. The patient refused operation, and was treated with palliative chemotherapy and radiation. The patient later showed systemic metastasis and died of melanoma 7 mo after the first presentation. No histological specimens were obtained from the metastatic sites.
Amelanotic melanoma of the esophagus is extremely rare, and the pathological diagnosis is very difficult because melanin pigment is absent. In fact, it has been frequently misdiagnosed as other sarcomas[1–13]. Immunohistochemistry of melanoma antigens such as melanosome (HMB45), Malan-A, and S100 protein are mandatory for the diagnosis of amelanotic melanoma. Ultrastructural demonstration of melanosome is also diagnostic. In the present study, both cases were positive for immunoreactive melanosome (HMB45) and S100 protein, confirming the diagnosis. In addition, positive KIT strongly suggests that the tumors are melanomas. Amelanotic melanoma can be diagnosed in biopsy specimens.
Malignant melanoma is a very aggressive tumor irrespective of its location. Malignant melanoma of the esophagus is therefore a very aggressive tumor, and its prognosis is very poor[1–13]. In the present study, the prognosis was indeed very poor in both patients. Operation followed by adjuvant chemotherapy and radiation is the best choice of treatment[1–13]. In the present cases, operation was impossible in one case and was not performed in another case because of the patient’s decision not to proceed.
The previously reported cases of esophageal melanoma were only case reports or clinical studies of very small series. There have been no reports of KIT and PDGFRA expression and mutations in esophageal melanomas. The present study is the first report of esophageal melanoma with an examination of KIT and PDGFRA protein expression and gene status of KIT and PDGFRA in esophageal melanoma. The tumors in the present cases expressed KIT and PDGFRA, but identified no mutations of KIT and PDGFRA. The positive expressions of KIT and PDGFRA suggest that these transmembranous oncoproteins are present in esophageal melanoma.
In cutaneous melanomas, the percentage of KIT expression varies amongst studies reported by researchers[36]. The percentage of KIT positive cutaneous melanomas in the literature is as follows; 35%[37], 21%[38], 87%[39], 90%[40], 50%[41] and 84%[42]. Sihto et al[36] reported that KIT expression in most human solid tumors, including cutaneous melanomas, was due to KIT gene amplification. Studies of KIT mutations in cutaneous melanoma are scant. Willmore-Payne et al[23] showed only 2% of KIT mutations in cutaneous melanomas. Sihto et al[36] showed no KIT mutations in 14 cutaneous melanomas. In contrast, Curtin et al[14] showed that KIT mutations are present in 39% of mucosal melanomas, in 36% of acral melanomas, in 28% of melanomas on sun-damaged skin, and in 0% of melanomas on non-sun-damaged skin. Beadling et al[37] recently reported that KIT mutations were present in 23% of acral melanomas, 15.6% of mucosal melanomas, 7.7% of conjunctival melanomas, 1.7% of cutaneous melanoma, and in 0% of choroidal melanomas.
PDGFRA protein expression in melanoma has not been performed, to the best of the author’s knowledge. As for PDGFRA mutations, Curtin et al[24] found no PDGFRA mutations in 26 cutaneous melanomas. Sihto et al[36] demonstrated no PDGFRA gene mutations in 14 cutaneous melanomas.
In summary, the author reported two extremely rare cases of amelanotic malignant melanoma of the esophagus with immunohistochemical and genetic analysis of KIT and PDGFRA.
| 1. | Sanchez AA, Wu TT, Prieto VG, Rashid A, Hamilton SR, Wang H. Comparison of primary and metastatic malignant melanoma of the esophagus: clinicopathologic review of 10 cases. Arch Pathol Lab Med. 2008;132:1623-1629. |
| 2. | Suzuki H, Nakanishi Y, Taniguchi H, Shimoda T, Yamaguchi H, Igaki H, Tachimori Y, Kato H. Two cases of early-stage esophageal malignant melanoma with long-term survival. Pathol Int. 2008;58:432-435. |
| 3. | Li B, Lei W, Shao K, Zhang C, Chen Z, Shi S, He J. Characteristics and prognosis of primary malignant melanoma of the esophagus. Melanoma Res. 2007;17:239-242. |
| 4. | Lohmann CM, Hwu WJ, Iversen K, Jungbluth AA, Busam KJ. Primary malignant melanoma of the oesophagus: a clinical and pathological study with emphasis on the immunophenotype of the tumours for melanocyte differentiation markers and cancer/testis antigens. Melanoma Res. 2003;13:595-601. |
| 5. | Kato H, Watanabe H, Tachimori Y, Watanabe H, Iizuka T, Yamaguchi H, Ishikawa T, Itabashi M. Primary malignant melanoma of the esophagus: report of four cases. Jpn J Clin Oncol. 1991;21:306-313. |
| 6. | Jawalekar K, Tretter P. Primary malignant melanoma of the esophagus: report of two cases. J Surg Oncol. 1979;12:19-25. |
| 7. | Stranks GJ, Mathai JT, Rowe-Jones DC. Primary malignant melanoma of the oesophagus: case report and review of surgical pathology. Gut. 1991;32:828-830. |
| 8. | Stringa O, Valdez R, Beguerie JR, Abbruzzese M, Lioni M, Nadales A, Iudica F, Venditti J, San Roman A. Primary amelanotic melanoma of the esophagus. Int J Dermatol. 2006;45:1207-1210. |
| 9. | Heidemann J, Lebiedz P, Herbst H, Spahn TW, Domagk D, Domschke W, Kucharzik T. Amelanotic malignant melanoma of the esophagus: case report. Z Gastroenterol. 2005;43:597-600. |
| 10. | De Simone P, Gelin M, El Nakadi I. Amelanotic malignant melanoma of the esophagus. Report of a case. Minerva Chir. 2006;61:45-49. |
| 11. | Suzuki Y, Aoyama N, Minamide J, Takata K, Ogata T. Amelanotic malignant melanoma of the esophagus: report of a patient with recurrence successfully treated with chemoendocrine therapy. Int J Clin Oncol. 2005;10:204-207. |
| 12. | Watanabe H, Yoshikawa N, Suzuki R, Hirai Y, Yoshie M, Ohshima H, Takahashi M, Takai M, Hosoda S, Asanuma K. Malignant amelanotic melanoma of the esophagus. Gastroenterol Jpn. 1991;26:209-212. |
| 13. | Taniyama K, Suzuki H, Sakuramachi S, Toyoda T, Matsuda M, Tahara E. Amelanotic malignant melanoma of the esophagus: case report and review of the literature. Jpn J Clin Oncol. 1990;20:286-295. |
| 14. | Curtin JA, Busam K, Pinkel D, Bastian BC. Somatic activation of KIT in distinct subtypes of melanoma. J Clin Oncol. 2006;24:4340-4346. |
| 15. | Curtin JA, Fridlyand J, Kageshita T, Patel HN, Busam KJ, Kutzner H, Cho KH, Aiba S, Brocker EB, LeBoit PE. Distinct sets of genetic alterations in melanoma. N Engl J Med. 2005;353:2135-2147. |
| 16. | Hirota S, Isozaki K. Pathology of gastrointestinal stromal tumors. Pathol Int. 2006;56:1-9. |
| 17. | Lasota J, Miettinen M. KIT and PDGFRA mutations in gastrointestinal stromal tumors (GISTs). Semin Diagn Pathol. 2006;23:91-102. |
| 18. | Miettinen M, Lasota J. Gastrointestinal stromal tumors: review on morphology, molecular pathology, prognosis, and differential diagnosis. Arch Pathol Lab Med. 2006;130:1466-1478. |
| 19. | Hirota S, Isozaki K, Moriyama Y, Hashimoto K, Nishida T, Ishiguro S, Kawano K, Hanada M, Kurata A, Takeda M. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279:577-580. |
| 20. | Hirota S, Ohashi A, Nishida T, Isozaki K, Kinoshita K, Shinomura Y, Kitamura Y. Gain-of-function mutations of platelet-derived growth factor receptor alpha gene in gastrointestinal stromal tumors. Gastroenterology. 2003;125:660-667. |
| 21. | Miettinen M, Lasota J. KIT (CD117): a review on expression in normal and neoplastic tissues, and mutations and their clinicopathologic correlation. Appl Immunohistochem Mol Morphol. 2005;13:205-220. |
| 22. | Alexeev V, Yoon K. Distinctive role of the cKit receptor tyrosine kinase signaling in mammalian melanocytes. J Invest Dermatol. 2006;126:1102-1110. |
| 23. | Willmore-Payne C, Holden JA, Tripp S, Layfield LJ. Human malignant melanoma: detection of BRAF- and c-kit-activating mutations by high-resolution amplicon melting analysis. Hum Pathol. 2005;36:486-493. |
| 24. | Curtin JA, Pinkel D, Bastian BC. Absence of PDGFRA mutations in primary melanoma. J Invest Dermatol. 2008;128:488-489. |
| 25. | Terada T, Kawaguchi M, Furukawa K, Sekido Y, Osamura Y. Minute mixed ductal-endocrine carcinoma of the pancreas with predominant intraductal growth. Pathol Int. 2002;52:740-746. |
| 26. | Terada T, Kawaguchi M. Primary clear cell adenocarcinoma of the peritoneum. Tohoku J Exp Med. 2005;206:271-275. |
| 27. | Terada T, Taniguchi M. Intraductal oncocytic papillary neoplasm of the liver. Pathol Int. 2004;54:116-123. |
| 28. | Terada T. Ductal adenoma of the breast: immunohisto-chemistry of two cases. Pathol Int. 2008;58:801-805. |
| 29. | Terada T. Gallbladder adenocarcinoma arising in Rokitansky-Aschoff sinus. Pathol Int. 2008;58:806-809. |
| 30. | Terada T. Intraductal tubular carcinoma, intestinal type, of the pancreas. Pathol Int. 2009;59:53-58. |
| 31. | Terada T. Gastrointestinal stromal tumor of the uterus: a case report with genetic analyses of c-kit and PDGFRA genes. Int J Gynecol Pathol. 2009;28:29-34. |
| 32. | Terada T. Primary multiple extragastrointestinal stromal tumors of the omentum with different mutations of c-kit gene. World J Gastroenterol. 2008;14:7256-7259. |
| 33. | Terada T. Primary small cell carcinoma of the mediastinum: a case report with immunohistochemical and molecular genetic analyses of KIT and PDGFRA genes. Med Oncol. 2009;26:247-250. |
| 34. | Terada T. Primary extragastrointestinal stromal tumor of the transverse mesocolon without c-kit mutations but with PDGFRA mutations. Med Oncol. 2009;26:233-237. |
| 35. | Terada T. Autopsy case of primary small cell carcinoma of the urinary bladder: KIT and PDGFRA expression and mutations. Pathol Int. 2009;59:247-250. |
| 36. | Sihto H, Sarlomo-Rikala M, Tynninen O, Tanner M, Andersson LC, Franssila K, Nupponen NN, Joensuu H. KIT and platelet-derived growth factor receptor alpha tyrosine kinase gene mutations and KIT amplifications in human solid tumors. J Clin Oncol. 2005;23:49-57. |
| 37. | Beadling C, Jacobson-Dunlop E, Hodi FS, Le C, Warrick A, Patterson J, Town A, Harlow A, Cruz F 3rd, Azar S. KIT gene mutations and copy number in melanoma subtypes. Clin Cancer Res. 2008;14:6821-6828. |
| 38. | Alexis JB, Martinez AE, Lutzky J. An immunohistochemical evaluation of c-kit (CD-117) expression in malignant melanoma, and results of imatinib mesylate (Gleevec) therapy in three patients. Melanoma Res. 2005;15:283-285. |
| 39. | Arber DA, Tamayo R, Weiss LM. Paraffin section detection of the c-kit gene product (CD117) in human tissues: value in the diagnosis of mast cell disorders. Hum Pathol. 1998;29:498-504. |
| 40. | Janku F, Novotny J, Julis I, Julisova I, Pecen L, Tomancova V, Kocmanova G, Krasna L, Krajsova I, Stork J. KIT receptor is expressed in more than 50% of early-stage malignant melanoma: a retrospective study of 261 patients. Melanoma Res. 2005;15:251-256. |
| 41. | Giehl KA, Nagele U, Volkenandt M, Berking C. Protein expression of melanocyte growth factors (bFGF, SCF) and their receptors (FGFR-1, c-kit) in nevi and melanoma. J Cutan Pathol. 2007;34:7-14. |
| 42. | Montone KT, van Belle P, Elenitsas R, Elder DE. Proto-oncogene c-kit expression in malignant melanoma: protein loss with tumor progression. Mod Pathol. 1997;10:939-944. |