Published online Jan 7, 2009. doi: 10.3748/wjg.15.38
Revised: October 2, 2008
Accepted: October 9, 2008
Published online: January 7, 2009
Pancreatic pseudocysts are complications of acute or chronic pancreatitis. Initial diagnosis is accomplished most often by cross-sectional imaging. Endoscopic ultrasound with fine needle aspiration has become the preferred test to help distinguish pseudocyst from other cystic lesions of the pancreas. Most pseudocysts resolve spontaneously with supportive care. The size of the pseudocyst and the length of time the cyst has been present are poor predictors for the potential of pseudocyst resolution or complications, but in general, larger cysts are more likely to be symptomatic or cause complications. The main two indications for some type of invasive drainage procedure are persistent patient symptoms or the presence of complications (infection, gastric outlet or biliary obstruction, bleeding). Three different strategies for pancreatic pseudocysts drainage are available: endoscopic (transpapillary or transmural) drainage, percutaneous catheter drainage, or open surgery. To date, no prospective controlled studies have compared directly these approaches. As a result, the management varies based on local expertise, but in general, endoscopic drainage is becoming the preferred approach because it is less invasive than surgery, avoids the need for external drain, and has a high long-term success rate. A tailored therapeutic approach taking into consideration patient preferences and involving multidisciplinary team of therapeutic endoscopist, interventional radiologist and pancreatic surgeon should be considered in all cases.
- Citation: Habashi S, Draganov PV. Pancreatic pseudocyst. World J Gastroenterol 2009; 15(1): 38-47
- URL: https://www.wjgnet.com/1007-9327/full/v15/i1/38.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.38
Pseudocyst of the pancreas is a localized fluid collection that is rich in amylase and other pancreatic enzymes and is surrounded by a wall of fibrous tissue that is not lined by epithelium[1]. Pseudocysts are connected with the pancreatic duct system, either as a direct communication or indirectly via the pancreatic parenchyma. They are caused by pancreatic ductal disruption following increased pancreatic ductal pressure, either due to stenosis, calculi or protein plugs obstructing the main pancreatic ductal system, or as a result of pancreatic necrosis following an attack of acute pancreatitis[23]. Pseudocysts are a common clinical problem and complicate the course of chronic pancreatitis in 30% to 40% of patients[4].
The occurrence of pseudocyst parallels that of pancreatitis and the etiology of pseudocysts resembles the causes of pancreatitis closely, although pseudocyst formation is less common after acute compared to chronic pancreatitis, and it is more common after alcohol-induced than after non-alcohol-related pancreatitis. Alcohol-related pancreatitis appears to be the major cause in studies from countries where alcohol consumption is high and accounts for 59%-78% of all pseudocysts[5].
Walt et al[6] reported data collected from Wayne State University Hospital in Detroit, USA. The causative factors in the 357 admissions for pancreatic pseudocysts included alcohol use in 251 cases (70%), biliary tract disease in 28 (8%), blunt trauma in 17 (5%), penetrating trauma in four (1%), operative trauma in one (0.3%), and idiopathic in 56 (16%). Most of the patients in the idiopathic group were thought to have been alcohol-related, but no definite evidence was recorded[6].
D’Egidio and Schein, in 1991, described a classification of pancreatic pseudocyst based on the underlying etiology of pancreatitis (acute or chronic), the pancreatic ductal anatomy, and the presence of communication between the cyst and the pancreatic duct[7]. They define three distinct types of pseudocysts[7]. Type I, or acute “post-necrotic” pseudocysts, that occur after an episode of acute pancreatitis and are associated with normal duct anatomy, and rarely communicate with the pancreatic duct. Type II, also post-necrotic pseudocysts, which occurs after an episode of acute-on-chronic pancreatitis (the pancreatic duct is diseased, but not strictured, and there is often a duct-pseudocyst communication). Type III, defined as “retention” pseudocysts, occur with chronic pancreatitis and are uniformly associated with duct stricture and pseudocystduct communication.
Another classification, based entirely on pancreatic duct anatomy, is proposed by Nealon and Walser[8]. Type I: normal duct/no communication with the cyst. Type II: normal duct with duct-cyst communication. Type III: otherwise normal duct with stricture and no duct-cyst communication. Type IV: otherwise normal duct with stricture and duct-cyst communication. Type V: otherwise normal duct with complete cut-off. Type VI: chronic pancreatitis, no duct-cyst communication. Type VII: chronic pancreatitis with duct-cyst communication[8].
Regardless of the etiology of pseudocyst, the incidence is low, 1.6%-4.5%, or 0.5-1 per 100 000 adults per year[910]. In a study by Imrie, pseudocysts developed after emergency hospital admission for an episode of acute pancreatitis in 86 patients[11]. Sixty-two of the 86 pseudocysts consequent to acute pancreatitis were derived from the local hospital population area, in which 879 patients with acute pancreatitis were admitted to hospital during the same time period. This resulted in a 7% overall incidence of pseudocysts as a complication of acute pancreatitis[11].
In a series of 926 patients with non-alcoholic acute pancreatitis, fluid collections were observed in 83 (9%). At the end of 6 wk, 48 (5%) still had a fluid collection consistent with a pseudocyst[12].
Kourtesis et al[13] followed prospectively with computed tomography (CT) 128 consecutive patients with acute pancreatitis (mostly alcohol-induced). Forty-eight patients (37%) developed fluid collection in the pancreatic region. The majority of these resolved spontaneously. In 15 (12%) patients, symptomatic pseudocysts developed.
Pseudocysts tend to be more common in chronic as compared to acute pancreatitis. Incidence figures of 30% to 40% have been reported in the literature[4]. However, there is a lack of precise data based on the long-term follow-up of patients with chronic pancreatitis, in contrast to acute pseudocysts where the patient with chronic pancreatitis may have had the disease for 10, 20 or more years giving him a high risk of developing a pseudocyst at least once over a long period of sickness[14].
The pathogenesis of pseudocysts seems to stem from disruptions of the pancreatic duct due to pancreatitis or trauma followed by extravasation of pancreatic secretions. Two thirds of patients with pseudocysts have demonstrable connections between the cyst and the pancreatic duct. In the other third, an inflammatory reaction most likely sealed the connection so that it is not demonstrable.
In case of pseudocyst following an episode of acute pancreatitis, only if the acute fluid collection persists more than 4-6 wk, and is well-defined by a wall of fibrous or granulation tissue, can one say that an acute pseudocyst has appeared. Such a pseudocyst usually contains enzymatic fluid and necrotic debris[15].
The pathogenesis of pseudocyst formation in chronic pancreatitis is less well understood but, at least two mechanisms may be involved, the cyst may develop as a consequence of an acute exacerbation of the underlying disease and/or blockage of a major branch of the pancreatic duct by a protein plug, calculus or localized fibrosis[15].
The clinical presentation of pancreatic pseudocyst can range from asymptomatic patient to major abdominal catastrophe due to complications[16–18]. Acute complications include bleeding (usually from splenic artery pseudoaneurysm), infection, and rupture.
Chronic complications include gastric outlet obstruction, biliary obstruction and thrombosis of the splenic or portal vein with development of gastric varices[18].
A variety of diseases can mimic the clinical presentation of pancreatic pseudocyst (Table 1). Once pancreatic cyst is identified by an imaging modality, the most important question is to differentiate pseudocyst from other cystic lesions of the pancreas (Table 2).
| Pancreatic diseases | Extrapancreatic diseases |
| Acute & chronic pancreatitis | Peptic ulcer disease & gastric cancer |
| Pancreatic necrosis & abscess | Acute cholecystitis & gallstones |
| Adenocarcinoma of the pancreas | Abdominal aortic aneurysm |
| Pancreatic cystic neoplasms | Intestinal ischemia |
| Pancreatic artery pseudoaneurysm | Ovarian cysts & cancers |
| Bowel obstruction | |
| Acute myocardial infarction | |
| Pneumonia |
| SCA | MCN | IPMN | SPN | Pseudocyst | |
| Prevalent age | Middle age | Middle age | Elderly | Young | Variable |
| Sex | Mostly female | Mostly female | Male > female | Mostly female | Male > female |
| Presentation | Mass/pain | Mass/pain | Pancreatitis | Mass/pain | Pain |
| Location | Evenly | Body/tail | Head | Evenly | Evenly |
| Malignant potential | Very low | Moderate to high | Low to high | Low | None |
No specific set of symptoms is specific for pseudocysts; however, one should consider the possibility of a pseudocyst in a patient who has persistent abdominal pain, anorexia, or abdominal mass after a case of pancreatitis. Rarely, patients present with jaundice or sepsis from an infected pseudocyst[16]. Occasionally, even patients with large pancreatic pseudocyst are asymptomatic. In patients presenting with pancreatic cyst incidentally discovered on imaging, a crucial point is to define whether the patient has had prior history of pancreatitis. The sensitivity of physical examination findings is limited. Patients frequently have a tender abdomen. They can occasionally have a palpable abdominal mass. Peritoneal signs suggest rupture of the cyst or infection. Other possible findings include fever, scleral icterus or pleural effusion[17].
Serum tests have limited utility. Amylase and lipase levels are often elevated, but may be within reference ranges. The serum bilirubin and liver chemistries may be elevated if the bile duct is obstructed from stone, extrinsic compression from the pseudocyst or from underlying liver disorder (e.g. alcoholic hepatitis). Some laboratory tests may provide clues to the underlying etiology of pancreatitis (e.g. elevated triglycerides or calcium level). Elevated liver chemistries raise the suspicion for biliary pancreatitis.
Pancreatic pseudocyst appears as an echoic structure associated with distal acoustic enhancement on US examination. They are well defined and round or oval, and they are contained within a smooth wall. During the early phases of their development, pseudocysts can appear more complex, with varying degrees of internal echoes. Usually, this appearance results from the presence of necrotic debris and is more common in pseudocysts that form as a result of acute necrotizing pancreatitis than in chronic pancreatitis related pseudocysts. The debris is cleared over time in most cases. The pseudocyst can appear more complex in two other instances: when hemorrhage occurs into the cyst or when infection of the cyst complicates the clinical course. Color Doppler or duplex scanning should always be performed in cystic lesions to ensure that the lesion in question is not a giant pseudoaneurysm. Sensitivity rates for US in the detection of pancreatic pseudocysts are 75% to 90%. Therefore, US is inferior to CT, which has a sensitivity of 90% to 100%. US has several limitations, as compared with CT, in the initial diagnosis of a pseudocyst: the presence of overlying bowel gas decreases the sensitivity of US, and unlike CT, US examinations are highly operator dependent[19].
The identification of a thick-walled, rounded, fluid-filled mass adjacent to the pancreas on an abdominal CT scan in a patient with a history of acute or chronic pancreatitis is virtually pathognomonic for pancreatic pseudocyst. Positive CT findings in this clinical situation do not require confirmation with another diagnostic modality. In the acute setting, a CT scan is the better choice because significant amounts of bowel gas resulting from ileus or obstruction decrease the sensitivity of US. In addition, CT scans provide more detailed information regarding the surrounding anatomy and can demonstrate additional pathology, including pancreatic duct dilatation and calcifications, common bile duct dilatation, and extension of the pseudocyst outside the lesser sac. The major weakness of CT scanning is the relative inability to differentiate pseudocyst from cystic neoplasm, especially mucinous cystadenomas and intraductal papillary mucinous neoplasm (IPMN)[20]. Furthermore, the intravenous contrast administered at the time of CT can precipitate or worsen kidney dysfunction.
MRI and magnetic resonance cholangiopancreatography (MRCP) are sensitive diagnostic modalities for pancreatic pseudocysts. They are generally not routinely used because CT scanning typically offers all the diagnostic information that is required. However, the increased contrast provides for better characterization of fluid collections. MRI or MRCP is superior to CT in depicting debris within fluid collections and pseudocysts. On T2-weighted images, a fluid-filled cystic mass produces high signal intensity and appears bright. The pancreatic duct and biliary systems are easily visualized in detail, although interpreting the status of pancreatic duct integrity may be difficult[21].
The ability of MRI/MRCP to depict choledocholithiasis is far superior to that of CT or US. Furthermore, MRCP techniques can also depict subtle branch-chain dilatation in chronic pancreatitis. MRI is also highly sensitive to detect bleeding with complex fluid collections.
ERCP is not necessary in diagnosing pseudocysts, but can provide definitive therapy in some cases. It also can be useful in planning possible drainage strategy. A study by Nealon et al[22] investigated the use of ERCP and the treatment of pseudocysts and acute pancreatitis and reported that ERCP findings may influence the treatment plan. Some authors, therefore, recommend performing an ERCP before contemplated surgical procedures. We believe that with the advent of alternative imaging technology [(CT, MRI, MRCP and endoscopic ultrasound (EUS)] ERCP is not necessary in most cases, but this has not been formally tested in a prospective study.
EUS is usually used as a secondary test to further evaluate pancreatic cyst detected by other imaging modality (US, CT or MRI). EUS is the test of choice when attempting to distinguish pancreatic pseudocyst from other cystic lesions of the pancreas. Visualization of the pancreas via EUS provides high quality images due to the close proximity of the ultrasound transducer to the area of interest. Criteria suggestive of cystic neoplasm include a cyst wall thickness of greater than 3 mm, macroseptation (all cystic components more than 10 mm), the presence of a mass or nodule, and cystic dilation of the main pancreatic duct[23–25]. Fine needle aspiration (FNA) of the cyst can be performed at the time of EUS and cyst fluid obtained for laboratory evaluation (see laboratory evaluation above). EUS can also be used to guide therapeutic endoscopic drainage.
Analysis of the cyst fluid may help differentiate pseudocysts from cystic tumors of the pancreas (Table 3). The preferred modality to obtain cystic fluid for analysis is EUS. Carcinoembryonic antigen (CEA) level in the cystic fluid is the marker most commonly used. It is low in pseudocysts and serous cystadenomas and elevated in mucinous cystadenomas. A CEA level of greater than 400 ng/mL within the cyst fluid strongly suggests mucinous lesion[232426]. Amylase levels are usually high in pseudocysts and low in serous cystadenoma. Cytology is occasionally helpful, but a negative result does not exclude malignancy.
| SCA | MCN | MCAC | Pseudocyst | |
| CEA | Low | High | High | Low |
| CA125 | Variable | Variable | High | Low |
| CA19-9 | Variable | Variable-high | Variable-high | Variable |
| Amylase | Low-high | Low-high | Low-high | High |
| Lipase | Low | Low | Low | High |
Hammel et al[27] published a study to assess the reliability of preoperative biochemical and tumor marker analysis in cyst fluids obtained by FNA for pathological diagnosis. Cyst fluid was obtained preoperatively by FNA, and biochemical and tumoral marker values were measured. The diagnosis of cystic tumors (seven serous cystadenomas and 12 mucinous tumors) was established by surgical specimen analysis. Thirty-one pancreatic pseudocysts complicating well-documented chronic pancreatitis were also studied. The results showed that carbohydrate antigen 19-9 levels of > 50 000 U/mL had a 75% sensitivity and a 90% specificity for distinguishing mucinous tumors from other cystic lesions. CEA levels of < 5 ng/mL had a 100% sensitivity and an 86% specificity for distinguishing serous cystadenomas from other cystic lesions. Amylase levels of > 5000 U/mL had a 94% sensitivity and a 74% specificity for distinguishing pseudocysts from other cystic lesions. His conclusion was: high carbohydrate antigen 19-9, low CEA, and high amylase levels in cyst fluid are very indicative of mucinous tumors, serous cystadenomas, and pseudocysts, respectively[27].
Sperti et al[28] published a study that was performed to evaluate the utility of serum and cyst fluid analysis for enzymes (amylase and lipase) and tumor markers (CEA, CA 19-9, CA 125, and CA 72-4) in the differential diagnosis of cystic pancreatic lesions. In the study, serum and cyst fluid were obtained from 48 patients with pancreatic cysts (21 pseudocysts, 14 mucinous cystic neoplasms, six ductal carcinomas, and seven serous cystadenomas), observed between 1989 and 1994. The results showed that serum CA 19-9 levels were significantly higher in ductal carcinomas (all > 100 U/mL) and mucinous cystic neoplasms (P < 0.05). CA 72-4 cyst fluid levels were significantly higher in mucinous cystic tumors (P < 0.005), with 95% specificity and 80% sensitivity in detecting mucinous or malignant cysts. A combined assay of serum CA 19-9 and cyst fluid CA 72-4 correctly identified 19 of 20 (pre-) malignant lesions (95%), with only one false-positive result (3.6%). Cytology showed a sensitivity of 48% and specificity of 100%. Their conclusion was that any pancreatic cyst with high serum CA 19-9 values, positive cytology, or high CA 72-4 in the fluid should be considered for resection[28].
Khalid et al[29] published a prospective study of the utility of molecular analysis of the pancreatic pseudocyst. In the study, endoscopic ultrasound-guided pancreatic cyst aspirates were prospectively collected during a period of 19 mo and studied for cytology, CEA level, and molecular analysis. Molecular evaluation incorporated DNA quantification (amount and quality), κ-ras point mutation, and broad panel tumor suppressor linked microsatellite marker allelic loss analysis by using fluorescent capillary electrophoresis. The sequence of mutation acquisition was also calculated on the basis of a clonal expansion model, and comparison was made to the final pathology. Thirty-six cysts with confirmed histology were analyzed. There were 11 malignant, 15 premalignant, and 10 benign cysts. Malignant cysts could be differentiated from premalignant cysts on the basis of fluid CEA level (P = 0.034), DNA quality (P = 0.009), number of mutations (P = 0.002), and on the sequence of mutations acquired (P < 0.001). Early κ-ras mutation followed by allelic loss was the most predictive of a malignant cyst (sensitivity, 91%; specificity, 93%). The study concluded that malignant cyst fluid contains adequate DNA to allow mutational analysis. A first hit κ-ras mutation followed by allelic loss is most predictive of the presence of malignancy in a pancreatic cyst. This approach should serve as an ancillary tool to the conventional work-up of pancreatic cysts. Cumulative amount and timing of detectable mutational damage can assist in diagnosis and clinical management[29].
Intravenous fluids, analgesics and antiemetics are routinely given. For patients that can tolerate oral intake, low fat diet is recommended. In patients that cannot tolerate oral nutrition, support can be provided via naso-enteral feeding or total parenteral nutrition (TPN). To date, no studies have compared these two approaches in the seating of pancreatic pseudocyst and choice is based on availability and local preferences. If one can extrapolate from studies comparing the two modalities in the seating of acute necrotizing pancreatitis, one can expect that jejunal feeding will be related with fewer complications (infection), but may not be able to provide as much calories as TPN.
The rationale of using octreotide as a therapy for pancreatic pseudocyst is that it will decrease pancreatic secretions and aid in pseudocyst resolution. Unfortunately, this strategy has not been rigorously tested and only a handful of case series have been published[3031].
Most pseudocysts resolve with supportive medical care. Vitas et al[32] followed over a period of 5 years 114 patients with the diagnosis of pancreatic pseudocyst. Forty-six patients underwent primary operative therapy, with 13% undergoing emergency operations for pseudocyst-related complications. Although no operative deaths occurred, significant morbidity occurred in 26% of patients (emergency operations, 67%; elective procedures, 10%). The remaining 68 patients were initially treated with a nonoperative, expectant approach. Severe, life-threatening complications in this group (follow-up for a mean of 46 mo) occurred in only six patients (9%); 19 patients eventually underwent elective operation directed at either the pseudocyst or other complications related to pancreatitis. Overall, in patients managed by a nonoperative approach, resolution of the pseudocyst occurred in 57% of the 24 patients with satisfactory radiographic follow-up, with 38% resolving more than 6 mo after diagnosis. Although patients eventually undergoing operation tended to have larger pancreatic pseudocysts than the patients managed successfully nonoperatively (6.9 cm vs 4.9 cm), no serious complications occurred in seven patients with pancreatic pseudocysts greater than 10 cm who were treated expectantly[32].
Several studies have indicated that the size of the cyst and the length of time the cyst has been present are poor predictors of potential for pseudocyst resolution or complications, but in general, larger cysts are more likely to become symptomatic or cause complications[33]. However, some patients with larger collections do well; therefore, size of the pseudocyst alone is not an indication for drainage[3435]. The two main indications for invasive intervention are the presence of symptoms or the presence of complications (infection, bleeding, gastric outlet or biliary obstruction).
Symptomatic pseudocysts or the presence of some complications (infected pseudocyst, gastric outlet or biliary obstruction) are the main two indications for some type of drainage procedure. To date, no prospective controlled studies have compared directly percutaneous, surgical and endoscopic drainage approaches. As a result, the management varies based on local expertise but in general endoscopic drainage is becoming the preferred approach.
External drainage can be achieved using CT or US guidance. With this technique, a drainage pigtail catheter is placed percutaneously into the fluid cavity and fluid is drained. Three-dimensional ultrasonography has been reported useful for the guidance of catheters into cyst cavities and avoiding vessels[36]. The fluid is collected over several weeks into an external collection system. When the drainage output becomes minimal, the catheter is removed. Contrast injection into the cyst cavity will demonstrate the size of the remaining cavity and this finding can be used to monitor the progress. This technique is successful at resolving pseudocysts, but it has a high risk of infections. The external drain tends to create significant patient discomfort. Furthermore, the catheter tends to clog and may require repositioning and exchange. The reported long-term success rate for pseudocyst resolution for US-guided pseudocyst drainage is around 50%. Unsuccessful drainages are usually caused by large ductal leaks or obstruction of the main pancreatic duct. Percutaneous catheter drainage is contraindicated in patients who are poorly compliant and cannot manage a catheter at home. It is also contraindicated in patients with strictures of the main pancreatic duct and in patients with cysts containing bloody or solid material[3738].
Surgical drainage of pseudocysts is accomplished by providing a communication between the pseudocyst cavity and the stomach or small bowel. This approach to drainage is often reserved for those patients that cannot tolerate or have failed percutaneous or endoscopic drainage. The surgical stoma should be placed in the most dependent portion of the cystic cavity in order to maximize the chances of complete drainage. The stoma usually remains patent and functional for several months.
Adams and Anderson published findings from a retrospective analysis of 94 patients[39]. The study population consisted of 42 patients undergoing internal surgical drainage and 52 patients undergoing percutaneous pseudocyst drainage. Significant complications occurred in 16.7% of the patients undergoing surgery and in 7.7% of the patients undergoing percutaneous drainage (P > 0.05). A subsequent operation was required in 9.5% of the surgical group and 19.2% of the percutaneous drainage group (P > 0.05). A significantly higher mortality rate was associated with surgical therapy (9%) than with percutaneous therapy (1%) (P < 0.05)[39].
Endoscopic drainage of pseudocysts is becoming the preferred therapeutic approach because it is less invasive than surgery, avoids the need for external drain and has a high long-term success rate. Drainage is accomplished with either a transpapillary approach with ERCP or direct drainage across the stomach or duodenal wall. A transpapillary approach is used when the pseudocyst communicates with the main pancreatic duct, usually in the genue of the pancreatic duct. This approach is also successful for patients with pancreatic duct disruption.
A transgastric or transduodenal approach is used when the pseudocyst is directly adjacent to the gastro-duodenal wall. To determine the size and location of the pseudocyst, and to measure the thickness of the pseudocyst wall, EUS has become the test of choice. A distance between the gastric or duodenal wall and cyst wall of more than 1 cm or the presence of large intervening vessels or varices are relative contraindications for endoscopic drainage[4041]. Transgastric or transduodenal stenting of pseudocysts may be performed using an endoscopic approach under fluoroscopic guidance or using EUS to introduce the guidewire into the pseudocyst cavity.
The endoscopic approach is dependent upon the presence of a bulge into the lumen of the stomach or duodenum in order to determine the entry site for catheterization. This approach has several inherent risks, including missing the pseudocyst, injuring intervening vessels, and sub-optimal placement of the drainage catheter[42]. Therapeutic echoendoscopes now make it possible to treat pseudocysts with EUS-guided transmural stenting[43]. Several series have described the deployment of a 7 Fr stent that is introduced with a needle knife catheter[44]. A new large-channel echoendoscope allows the use of 10 Fr stents across the stomach or duodenum[45].
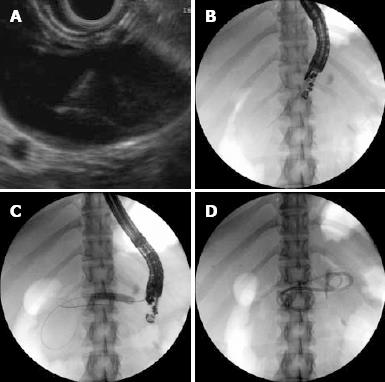
The exact technique for transmural pseudocyst drainage has not been standardized. In our institution, we prefer a combined EUS/fluoroscopy guided technique. The linear therapeutic channel EUS endoscope is used to detect an optimal site of apposition of pseudocyst and gut wall, free of intervening vascular structures (Figure 1A). The 19 Fr gauge EUS FNA needle is then advanced into the cyst cavity under real-time ultrasound guidance. The needle position is then located under fluoroscopy (Figure 1B). After the pseudocyst cavity has been entered, fluid is aspirated and a floppy-tip 0.035 guide wire is advanced via the needle and under fluoroscopic control is curled few times into the cyst cavity. The cyst-gastrostomy (duodenostomy) fistula tract is then pneumatically dilated, with 8 to 15 mm biliary balloon dilators (Figure 1C). The size of the balloon used for dilation is arbitrarily determined based on the size of the cyst, proximity of vessels, presence of necrotic debris in the cyst cavity, viscosity of the aspirated pseudocyst fluid and the presence of infection. In an attempt to decrease the risk of bleeding we try to avoid using electrocautery to create the fistulous tract. In a rare occasion, when the pseudocyst wall is very tick and the balloon dilator cannot be advanced, we use the Cystotome (Cook Medical, Winston-Salem, NC, USA). We will then stent the tract with two or more double pigtail stents (7F-10F) via the EUS scope (Figure 1D).
In a small series, the EUS approach has resulted in a success rate of more than 90% in patients with chronic pseudocysts[46]. The recurrence rate after endoscopic drainage is low, 4%, and the complication rate is less than 16%[47].
EUS is also capable of guiding the drainage of infected pseudocysts using naso-cystic drains[48]. It may even be possible to drain infected necrotic pancreatic tissue using EUS and endoscopic techniques[49].
Hookey et al[50] published a chart review and prospective follow-up for 116 patients with attempted endoscopic drainage of symptomatic pancreatic-fluid collections (pseudocysts and organized pancreatic necrosis). A total of 116 patients presented with fluid collections classified as acute fluid collection (n = 5), necrosis (n = 8), acute pseudocyst (n = 30), chronic pseudocyst (n = 64), and pancreatic abscess (n = 9). The median diameter of the collection drained was 60 mm (15-275 mm). Median follow-up after drainage was 21 mo. The drainage technique was transpapillary in 15 patients, transmural in 60, and both in 41. Successful resolution of symptoms and collection occurred in 87.9% of cases. No difference in success rates was observed between patients with acute pancreatitis and those with chronic pancreatitis. However, drainage of organized necrosis was associated with a significantly higher failure rate than other collections. No significant differences were observed regarding success when disease, drainage technique, or site of drainage was considered. Complications occurred in 13 patients (11%), and there were six deaths in the 30 d after drainage, including one that was procedure related. He concluded that endoscopic drainage of pancreatic-fluid collections is successful in the majority of patients and is accompanied by an acceptable complication rate[50].
Muscatiello et al[51] published a case report of alcohol use for the treatment of a pancreatic pseudocyst. In his report, aspiration of the pancreatic pseudocyst was started, and after an apparent reduction in the volume of the pseudocyst by about 30%, 30 mL of absolute ethanol diluted 1:1 with saline was injected and maintained for about 10 min. Aspiration then continued until EUS imaging showed that the cyst was completely empty. CT 24 h later demonstrated no complications and confirmed that the procedure had been successful. Culture of the aspiration fluid identified a Pseudomonas aeruginosa and Citrobacter freundii complex. Cytological examination did not show any neoplastic cells. The patient was discharged on the seventh day with no symptoms and with normal laboratory tests. It seems that, in addition to causing sclerosis of the cystic wall, ethanol contributes to sterilizing the infected fluid collection. In that case, a long follow-up period (18 mo) in which there was no recurrence of the pseudocyst confirms that this procedure may be useful in the treatment of organized necrotic abscesses and pancreatic abscesses when there is no communication with the pancreatic duct[51].
In a large retrospective analysis of 603 patients who were undergoing EUS-FNA of pancreatic cysts, possible infection developed in only a single patient. The majority of patients in this series (90%) received antibiotic prophylaxis, most commonly a fluoroquinolone given for 3 d after the procedure, and this may possibly explain the low infection rate. The benefit of prophylactic antibiotics before an FNA of cystic lesions has not been evaluated by prospective randomized studies[52].
The ASGE, in 2008, published the guidelines for prophylactic use of antibiotics for GI endoscopy. According to these guidelines, prophylaxis with an antibiotic, such as a fluoroquinolone administered before EUS-FNA of cystic lesions along the GI tract including pancreatic cyst. Antibiotics may be continued for 3 to 5 d after the procedure (supported by observational studies). When antibiotic prophylaxis is administered, a fluoroquinolone administered before the procedure and continued for 3 d after the procedure is a reasonable regimen[53].
Cahen et al[54] published a retrospective study to evaluates the short-term and long-term results with the endoscopic drainage of pancreatic pseudocyst and aimed to identify procedural modifications that may improve its safety and efficacy. A total of 92 patients were included (66 men, 26 women; median age 49 years). The technical success rate of the drainage procedure was 97% and the mortality rate was 1%. Complications occurred in 31 patients (34%), eight of which (9%) were major and required surgery: hemorrhage in four cases (three of which were caused by erosion of a straight endoprosthesis through the cyst wall), secondary infection in three, and perforation in one. During a median follow-up period of 43 mo, 10 patients (11%) underwent additional (nonendoscopic) treatment for a persistent cyst and five (5%) for a recurrent cyst. Overall, endoscopic drainage was successful in 65 patients (71%). He concluded that endoscopic drainage is an effective treatment for pancreatic pseudocysts and offers a definitive solution in almost three-quarters of the cases. The majority of major complications might have been prevented by using pigtail stents instead of straight stents and by taking a more aggressive approach to the prevention and treatment of secondary cyst infection[54].
Splenic complications of pseudocyst include massive hemorrhage into the pseudocyst, sepsis with splenic infarction, and splenic vein thrombosis. The diagnosis of intrasplenic pseudocyst, based on clinical findings alone, is difficult to arrive at but should be suggested by the presence of a mass in the left upper quadrant. Sonography and computerized axial tomography may be particularly helpful in confirming splenic involvement. Selective celiac arteriography should be performed whenever splenic involvement is suggested in order to confirm the diagnosis and to search for pseudoaneurysm formation. Urgent surgical intervention is usually warranted in view of the high incidence of serious complications and the propensity toward rapid clinical deterioration. Resection of the pseudocyst by splenectomy and distal pancreatectomy is the treatment of choice[55].
Rupture of a pseudocyst can have either a favorable or an unfavorable outcome and this depends on whether it ruptures into the gastrointestinal tract, into the general peritoneal cavity or into the vascular system[5657]. Rupture into the gastrointestinal tract either results in no symptoms or leads to melaena or hematemesis that usually requires urgent measures. Rupture into the general peritoneal cavity results in features of peritonitis and occasionally hemorrhagic shock. Emergent surgical exploration is usually required. While an internal drainage should always be aimed for, usually a thorough abdominal lavage and external drainage is all that can be achieved safely.
Hemorrhage can greatly complicate the course of a pseudocyst[58]. The morbidity and mortality is very high because it can appear without warning and is usually due to erosion of a major vessel in the vicinity of the pseudocyst. Interventional radiology can play an invaluable role both in locating the source of bleeding and in embolisation of the bleeding vessel[59]. Without prior information of the bleeding point, surgical exploration can be hazardous and challenging.
Infection occurs either spontaneously or after therapeutic or diagnostic manipulations. While infected pseudocyst can initially be treated with conservative means, a majority of patients will require intervention. Traditionally surgery has been the preferred modality but endoscopic treatment is gaining acceptance[4860]. An external drainage may be necessary in selected situations such as when there is evidence of gross sepsis and the patient is too unstable to undergo surgical or endoscopic drainage.
Biliary complications occur due to a large cyst in the pancreatic head region obstructing the common bile duct and resulting in obstructive jaundice[6162]. Therapeutic endoscopy with short-term biliary stenting is valuable in this situation. It can be retained until either the pseudocyst resolves or is treated by intervention.
Portal hypertension can result from compression or obstruction of the splenic vein/portal vein either by the cyst alone or in conjunction with underlying chronic pancreatitis[63]. In this situation, surgery appears to be the only treatment modality available and an appropriate surgical procedure can effectively treat this form of portal hypertension.
Pancreatic pseudocysts are the result of acute or chronic pancreatitis and are the most common cystic lesions of the pancreas, accounting for 75%-80% of such lesions. The most common symptoms are abdominal pain, nausea and vomiting, although they can be asymptomatic. Abdominal CT is an excellent choice for initial imaging. EUS plays an important role in differentiating pseudocyst from other cystic lesions of the pancreas and can greatly assist in transmural endoscopic drainage. Initial management consists of supportive care. Persistent symptoms and the development of complications warrant invasive intervention. The surgical, percutaneous and endoscopic pseudocyst drainage procedures have not been directly compared in high quality prospective randomized studies and the preferred approach varies based on patient preferences and local expertise. In recent years, the endoscopic approach has gained popularity with surgery reserved for patients who had failed endoscopic or percutaneous drainage. A tailored therapeutic approach taking into consideration patient preferences and involving multidisciplinary team of therapeutic endoscopist, interventional radiologist and pancreatic surgeon should be considered in all cases.
| 1. | Bradley EL 3rd. A clinically based classification system for acute pancreatitis. Summary of the International Symposium on Acute Pancreatitis, Atlanta, Ga, September 11 through 13, 1992. Arch Surg. 1993;128:586-590. |
| 2. | Sanfey H, Aguilar M, Jones RS. Pseudocysts of the pancreas, a review of 97 cases. Am Surg. 1994;60:661-668. |
| 3. | Gumaste VV, Pitchumoni CS. Pancreatic pseudocyst. Gastroenterologist. 1996;4:33-43. |
| 4. | Boerma D, Obertop H, Gouma DJ. Pancreatic pseudocysts in chronic pancreatitis. Surgical or interventional drainage? Ann Ital Chir. 2000;71:43-50. |
| 5. | Pitchumoni CS, Agarwal N. Pancreatic pseudocysts. When and how should drainage be performed? Gastroenterol Clin North Am. 1999;28:615-639. |
| 6. | Walt AJ, Bouwman DL, Weaver DW, Sachs RJ. The impact of technology on the management of pancreatic pseudocyst. Fifth annual Samuel Jason Mixter Lecture. Arch Surg. 1990;125:759-763. |
| 7. | D'Egidio A, Schein M. Pancreatic pseudocysts: a proposed classification and its management implications. Br J Surg. 1991;78:981-984. |
| 8. | Nealon WH, Walser E. Main pancreatic ductal anatomy can direct choice of modality for treating pancreatic pseudocysts (surgery versus percutaneous drainage). Ann Surg. 2002;235:751-758. |
| 9. | Sandy JT, Taylor RH, Christensen RM, Scudamore C, Leckie P. Pancreatic pseudocyst. Changing concepts in management. Am J Surg. 1981;141:574-576. |
| 10. | Wade JW. Twenty-five year experience with pancreatic pseudocysts. Are we making progress? Am J Surg. 1985;149:705-708. |
| 11. | Imrie CW, Buist LJ, Shearer MG. Importance of cause in the outcome of pancreatic pseudocysts. Am J Surg. 1988;156:159-162. |
| 12. | Maringhini A, Uomo G, Patti R, Rabitti P, Termini A, Cavallera A, Dardanoni G, Manes G, Ciambra M, Laccetti M. Pseudocysts in acute nonalcoholic pancreatitis: incidence and natural history. Dig Dis Sci. 1999;44:1669-1673. |
| 13. | Kourtesis G, Wilson SE, Williams RA. The clinical significance of fluid collections in acute pancreatitis. Am Surg. 1990;56:796-799. |
| 14. | Ammann RW, Akovbiantz A, Largiader F, Schueler G. Course and outcome of chronic pancreatitis. Longitudinal study of a mixed medical-surgical series of 245 patients. Gastroenterology. 1984;86:820-828. |
| 15. | Grace PA, Williamson RC. Modern management of pancreatic pseudocysts. Br J Surg. 1993;80:573-581. |
| 16. | Zdanyte E, Strupas K, Bubnys A, Stratilatovas E. [Difficulties of differential diagnosis of pancreatic pseudocysts and cystic neoplasms]. Medicina (Kaunas). 2004;40:1180-1188. |
| 17. | O'Malley VP, Cannon JP, Postier RG. Pancreatic pseudocysts: cause, therapy, and results. Am J Surg. 1985;150:680-682. |
| 18. | Gouyon P, Levy P, Ruszniewski P, Zins M, Hammel P, Vilgrain V, Sauvanet A, Belghiti J, Bernades P. Predictive factors in the outcome of pseudocysts complicating alcoholic chronic pancreatitis. Gut. 1997;41:821-825. |
| 19. | Pitchumoni CS, Agarwal N. Pancreatic pseudocysts. When and how should drainage be performed? Gastroenterol Clin North Am. 1999;28:615-639. |
| 20. | Siegelman SS, Copeland BE, Saba GP, Cameron JL, Sanders RC, Zerhouni EA. CT of fluid collections associated with pancreatitis. AJR Am J Roentgenol. 1980;134:1121-1132. |
| 21. | Morgan DE, Baron TH, Smith JK, Robbin ML, Kenney PJ. Pancreatic fluid collections prior to intervention: evaluation with MR imaging compared with CT and US. Radiology. 1997;203:773-778. |
| 22. | Nealon WH, Walser E. Surgical management of complications associated with percutaneous and/or endoscopic management of pseudocyst of the pancreas. Ann Surg. 2005;241:948-957; discussion 957-960. |
| 23. | Lewandrowski KB, Southern JF, Pins MR, Compton CC, Warshaw AL. Cyst fluid analysis in the differential diagnosis of pancreatic cysts. A comparison of pseudocysts, serous cystadenomas, mucinous cystic neoplasms, and mucinous cystadenocarcinoma. Ann Surg. 1993;217:41-47. |
| 24. | Linder JD, Geenen JE, Catalano MF. Cyst fluid analysis obtained by EUS-guided FNA in the evaluation of discrete cystic neoplasms of the pancreas: a prospective single-center experience. Gastrointest Endosc. 2006;64:697-702. |
| 25. | Sedlack R, Affi A, Vazquez-Sequeiros E, Norton ID, Clain JE, Wiersema MJ. Utility of EUS in the evaluation of cystic pancreatic lesions. Gastrointest Endosc. 2002;56:543-547. |
| 26. | Brugge WR, Lewandrowski K, Lee-Lewandrowski E, Centeno BA, Szydlo T, Regan S, del Castillo CF, Warshaw AL. Diagnosis of pancreatic cystic neoplasms: a report of the cooperative pancreatic cyst study. Gastroenterology. 2004;126:1330-1336. |
| 27. | Hammel P, Levy P, Voitot H, Levy M, Vilgrain V, Zins M, Flejou JF, Molas G, Ruszniewski P, Bernades P. Preoperative cyst fluid analysis is useful for the differential diagnosis of cystic lesions of the pancreas. Gastroenterology. 1995;108:1230-1235. |
| 28. | Sperti C, Pasquali C, Guolo P, Polverosi R, Liessi G, Pedrazzoli S. Serum tumor markers and cyst fluid analysis are useful for the diagnosis of pancreatic cystic tumors. Cancer. 1996;78:237-243. |
| 29. | Khalid A, McGrath KM, Zahid M, Wilson M, Brody D, Swalsky P, Moser AJ, Lee KK, Slivka A, Whitcomb DC. The role of pancreatic cyst fluid molecular analysis in predicting cyst pathology. Clin Gastroenterol Hepatol. 2005;3:967-973. |
| 30. | Gullo L, Barbara L. Treatment of pancreatic pseudocysts with octreotide. Lancet. 1991;338:540-541. |
| 31. | Suga H, Tsuruta O, Okabe Y, Saitoh F, Noda T, Yoshida H, Ono N, Kinoshita H, Toyonaga A, Sata M. A case of mediastinal pancreatic pseudocyst successfully treated with somatostatin analogue. Kurume Med J. 2005;52:161-164. |
| 32. | Vitas GJ, Sarr MG. Selected management of pancreatic pseudocysts: operative versus expectant management. Surgery. 1992;111:123-130. |
| 33. | Yeo CJ, Bastidas JA, Lynch-Nyhan A, Fishman EK, Zinner MJ, Cameron JL. The natural history of pancreatic pseudocysts documented by computed tomography. Surg Gynecol Obstet. 1990;170:411-417. |
| 34. | Cheruvu CV, Clarke MG, Prentice M, Eyre-Brook IA. Conservative treatment as an option in the management of pancreatic pseudocyst. Ann R Coll Surg Engl. 2003;85:313-316. |
| 35. | Andersson B, Nilsson E, Willner J, Andersson R. Treatment and outcome in pancreatic pseudocysts. Scand J Gastroenterol. 2006;41:751-756. |
| 36. | Gumaste VV, Pitchumoni CS. Pancreatic pseudocyst. Gastroenterologist. 1996;4:33-43. |
| 37. | Criado E, De Stefano AA, Weiner TM, Jaques PF. Long term results of percutaneous catheter drainage of pancreatic pseudocysts. Surg Gynecol Obstet. 1992;175:293-298. |
| 38. | Heider R, Meyer AA, Galanko JA, Behrns KE. Percutaneous drainage of pancreatic pseudocysts is associated with a higher failure rate than surgical treatment in unselected patients. Ann Surg. 1999;229:781-787; discussion 787-789. |
| 39. | Adams DB, Anderson MC. Percutaneous catheter drainage compared with internal drainage in the management of pancreatic pseudocyst. Ann Surg. 1992;215:571-576; discussion 576-578. |
| 40. | Weckman L, Kylanpaa ML, Puolakkainen P, Halttunen J. Endoscopic treatment of pancreatic pseudocysts. Surg Endosc. 2006;20:603-607. |
| 41. | Deviere J, Bueso H, Baize M, Azar C, Love J, Moreno E, Cremer M. Complete disruption of the main pancreatic duct: endoscopic management. Gastrointest Endosc. 1995;42:445-451. |
| 42. | Lo SK, Rowe A. Endoscopic management of pancreatic pseudocysts. Gastroenterologist. 1997;5:10-25. |
| 43. | Chak A. Endosonographic-guided therapy of pancreatic pseudocysts. Gastrointest Endosc. 2000;52:S23-S279. |
| 44. | Giovannini M, Bernardini D, Seitz JF. Cystogastrotomy entirely performed under endosonography guidance for pancreatic pseudocyst: results in six patients. Gastrointest Endosc. 1998;48:200-203. |
| 45. | Wiersema MJ, Baron TH, Chari ST. Endosonography-guided pseudocyst drainage with a new large-channel linear scanning echoendoscope. Gastrointest Endosc. 2001;53:811-813. |
| 46. | Norton ID, Clain JE, Wiersema MJ, DiMagno EP, Petersen BT, Gostout CJ. Utility of endoscopic ultrasonography in endoscopic drainage of pancreatic pseudocysts in selected patients. Mayo Clin Proc. 2001;76:794-798. |
| 47. | Libera ED, Siqueira ES, Morais M, Rohr MR, Brant CQ, Ardengh JC, Ferrari AP. Pancreatic pseudocysts transpapillary and transmural drainage. HPB Surg. 2000;11:333-338. |
| 48. | Giovannini M, Pesenti C, Rolland AL, Moutardier V, Delpero JR. Endoscopic ultrasound-guided drainage of pancreatic pseudocysts or pancreatic abscesses using a therapeutic echo endoscope. Endoscopy. 2001;33:473-477. |
| 49. | Fuchs M, Reimann FM, Gaebel C, Ludwig D, Stange EF. Treatment of infected pancreatic pseudocysts by endoscopic ultrasonography-guided cystogastrostomy. Endoscopy. 2000;32:654-657. |
| 50. | Hookey LC, Debroux S, Delhaye M, Arvanitakis M, Le Moine O, Deviere J. Endoscopic drainage of pancreatic-fluid collections in 116 patients: a comparison of etiologies, drainage techniques, and outcomes. Gastrointest Endosc. 2006;63:635-643. |
| 51. | Muscatiello N, Pietrini L, Gentile M, Tonti P, Ricciardelli C, Sorrentini I, Ierardi E. Endoscopic ultrasound-guided ethanol lavage of a pancreatic fluid collection. Endoscopy. 2006;38:951. |
| 52. | Lee LS, Saltzman JR, Bounds BC, Poneros JM, Brugge WR, Thompson CC. EUS-guided fine needle aspiration of pancreatic cysts: a retrospective analysis of complications and their predictors. Clin Gastroenterol Hepatol. 2005;3:231-236. |
| 53. | Banerjee S, Shen B, Baron TH, Nelson DB, Anderson MA, Cash BD, Dominitz JA, Gan SI, Harrison ME, Ikenberry SO. Antibiotic prophylaxis for GI endoscopy. Gastrointest Endosc. 2008;67:791-798. |
| 54. | Cahen D, Rauws E, Fockens P, Weverling G, Huibregtse K, Bruno M. Endoscopic drainage of pancreatic pseudocysts: long-term outcome and procedural factors associated with safe and successful treatment. Endoscopy. 2005;37:977-983. |
| 55. | Sitzmann JV, Imbembo AL. Splenic complications of a pancreatic pseudocyst. Am J Surg. 1984;147:191-196. |
| 56. | Yamamoto T, Hayakawa K, Kawakami S, Nishimura K, Katsuma Y, Hayashi N, Maeda M, Ishii Y. Rupture of a pancreatic pseudocyst into the portal venous system. Abdom Imaging. 1999;24:494-496. |
| 57. | Lesur G, Bernades P. [Pseudocysts of the pancreas. Diagnosis, course and principles of treatment]. Presse Med. 1996;25:939-943. |
| 58. | Ungania S, Panocchia N. [Splenic artery rupture in pancreatic pseudocyst]. Ann Ital Chir. 2000;71:251-255. |
| 59. | Gambiez LP, Ernst OJ, Merlier OA, Porte HL, Chambon JP, Quandalle PA. Arterial embolization for bleeding pseudocysts complicating chronic pancreatitis. Arch Surg. 1997;132:1016-1021. |
| 60. | Boerma D, van Gulik TM, Obertop H, Gouma DJ. Internal drainage of infected pancreatic pseudocysts: safe or sorry? Dig Surg. 1999;16:501-505. |
| 61. | Noda T, Ueno N, Tamada K, Ichiyama M, Fukuda M, Tomiyama T, Nishizono T, Tano S, Aizawa T, Iwao T. A case of chronic pancreatitis with pseudocysts complicated by infection and obstructive jaundice. Am J Gastroenterol. 1994;89:2066-2069. |
| 62. | Maema A, Kubota K, Bandai Y, Makuuchi M. Proximal bile duct stricture caused by a pancreatic pseudocyst: intra-operative placement of a metallic stent. Hepatogastroenterology. 1999;46:2020-2023. |
| 63. | Bernades P, Baetz A, Levy P, Belghiti J, Menu Y, Fekete F. Splenic and portal venous obstruction in chronic pancreatitis. A prospective longitudinal study of a medical-surgical series of 266 patients. Dig Dis Sci. 1992;37:340-346. |