Published online Sep 7, 2008. doi: 10.3748/wjg.14.5110
Revised: June 23, 2008
Accepted: June 30, 2008
Published online: September 7, 2008
Signal transducers and activators of transcription 3 (STAT3) play an important role in various autoimmune disorders including inflammatory bowel disease (IBD). Recent studies have revealed that STAT3 activation plays distinctly different roles between innate immune responses and acquired immune responses in colitis. STAT3-mediated activation of acquired immune responses plays a pathogenic role in colitis by enhancing the survival of pathogenic T cells. In contrast, STAT3-mediated activation of innate responses contributes to the suppression of colitis. This review will summarize the current understanding of the roles of STAT3 in IBD and the potential of targeting STAT3 for the treatment of IBD, emphasizing recent observations.
- Citation: Sugimoto K. Role of STAT3 in inflammatory bowel disease. World J Gastroenterol 2008; 14(33): 5110-5114
- URL: https://www.wjgnet.com/1007-9327/full/v14/i33/5110.htm
- DOI: https://dx.doi.org/10.3748/wjg.14.5110
Inflammatory bowel disease (IBD) is a group of idiopathic chronic relapsing immune-mediated inflammatory disorders, which is characterized by two major forms of intestinal inflammation: ulcerative colitis (UC) and Crohn’s disease (CD)[1]. Although the precise mechanisms of the pathogenesis of IBD still remain unclear, accumulating studies have shown that multiple cytokines, including interleukins (ILs) and interferons, are involved in the pathogenesis of IBD[2,3]. Most of these cytokines activate various members of a family of cytoplasmic transcription factors such as signal transducers and activators of transcription (STATs)[4,5]. Indeed, several recent studies have focused on the role of STATs on IBD. Among STATs, the role of STAT3 in IBD has been most well documented by recent studies obtained from human IBD studies[6-8] and experimental IBD models[9-12]. These studies have revealed that STAT3 is in a unique position for pathogenesis of IBD, because STAT3 plays distinct roles between innate immune cells and acquired immune cells. This review will focus on the roles of STAT3 in IBD from a different point of view, namely, innate immune systems versus acquired immune systems.
STAT3 was initially identified as an acute phase response factor, an inducible DNA binding protein that binds to the IL-6 responsive element within the promoters of hepatic acute phase protein genes[13]. Subsequent studies indicate that STAT3 becomes activated in response to a wide variety of cytokines and growth factors[4,5,14]. After ligation of their specific receptors by these cytokines and growth factors, STAT3 is immediately phosphorylated, resulting in their dimerization and migration to the nucleus to induce several gene expressions encoding molecules that play a role in a variety of biological functions such as cell growth, anti- and pro-apoptosis, cell motility, negative feedback loop (suppression of cytokine productions), regulatory cytokine production and anti-bacterial activity depending on cell types[4,5,14-17].
Although the role of STAT3 activation in the pathogenesis of IBD is still not very clear, the expression and activation of STAT3 protein in the mucosa of human IBD patients has been well studied. Lovato et al demonstrated constitutive activation of STAT3 in five out of seven CD patients in a small case series[6]. Musso et al demonstrated that STAT3 activation was confined to actively inflamed colons from not only CD, but also UC patients[7]. Accordingly, activated STAT3 was detected in isolated lamina propria mononuclear cells from inflamed IBD tissues, but not in peripheral blood mononuclear cells from control subjects or IBD patients. In this study, immunofluorescence demonstrated that the sources of activated STAT3 were macrophages and T lymphocytes, but not neutrophils. Mudter et al also showed that an increased amount of total STAT3 protein appeared in both UC and CD compared to noninflammatory control cells, and total STAT3 correlated with increased activated pSTAT3 in tissue sections from both UC and CD[8]. They also showed that increased pSTAT3 also directly correlated with the histological degree of inflammation of tissues. Importantly, this activation is observed in both acquired cells such as T cells and innate cells such as colonic epithelial cells (CECs).
Since classical knockout of the STAT3 gene in mice resulted in early embryonic lethality[18], various cell type-specific STAT3 knockout mice were generated to elucidate the functional relevance of STAT3[15,19-23]. Acquired immune cells, especially CD4+ T cells, have a considerable ability to induce the development and exacerbation of not only CD-like, but also UC-like experimental colitis[24-27]. Importantly, Takeda et al showed that T cell specific STAT3 knockout mice showed impaired T cell proliferation[19]. Moreover, they showed that STAT3 activation is involved in IL-6-dependent T cell proliferation through prevention of apoptosis. Atreya et al also showed that activation of IL-6/STAT3 cascade in lamina propria T cells can also induce prolonged survival of pathogenic T cells and, indeed, inactivation of this cascade contributes to the attenuation of chronic intestinal inflammation[9]. These observations indicate that activation of STAT3 in the acquired cells plays a pathogenic role in chronic colitis by inducing prolonged survival of pathogenic T cells and disruption of immune tolerance. Indeed, inactivation of IL-6 or leptin/STAT3 cascades results in the suppression of acquired immune (T cell) mediated colitis[9-12].
Dysregulated host/microbial interactions have been postulated to play a crucial role in the development of IBD by inducing uncontrolled activation of acquired immune responses[24,28-30]. Innate immune responses mediated by CECs and phagocytic cells such as macrophages play a crucial role in the initial defense to maintain the appropriate host/microbial interactions[15,22]. Accumulating studies have demonstrated that several mechanisms are involved in the maintenance and/or enhancement of the barrier function by CECs[15,31,32]. Among these mechanisms, activation of STAT3 is considered to be an important pathway that can enhance and maintain not only the barrier function of epithelial cells, but also the activation of macrophage[15,22]. Indeed, disruption of innate immune responses by genetically engineered either inactivation of STAT3 cascade in the CECs, or else macrophages induce intestinal inflammation[15,21-23]. This observation is further supported by a recent study using a conditional STAT3 knockout mouse strain in which STAT3 deletion is induced during the adult life[23]. This mouse strain shows efficient elimination of STAT3 protein in innate cells (macrophages and CECs), but not acquired cells (T and B cells), and develops a lethal colitis immediately after the deletion of STAT3 in the adult life. These findings indicate that, in contrast to acquired immune cells, activation of STAT3 in innate cells plays a regulatory role in colitis. Interestingly, inactivation of STAT3 in both CECs and macrophages results in lethal damage[18,23], whereas such fatal inflammation is not observed in mice in which the inactivation is specifically induced in either CECs or macrophages[15,22]. This raises a possibility that transient inactivation of STAT3 in both CEC and macrophage may cause lethal damage.
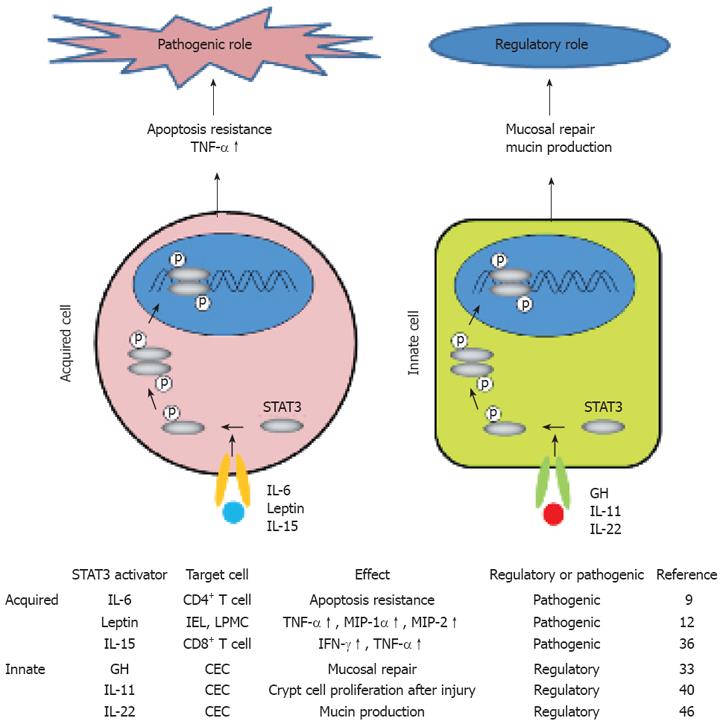
Several cytokines and growth factors including IL-6, IL-7, IL-9, IL-10, IL-11, IL-15, IFN-α/β, leukemia inhibitory factor (LIF), hepatocyte growth factor (HGF), oncostatin M (OSM), leptin and growth hormone (GH) have been identified to activate STAT3 cascade with a different efficiency depending on the cell types[4,5,14]. Among these, roles of IL-6, IL-11, IL-15, HGF, GH and leptin in experimental colitis have been demonstrated[9-12,33-36]. For example, IL-6, IL-15 and leptin play a pathogenic role in colitis by activating STAT3 predominantly in the acquired immune cells[9,10,12,36,37] (Figure 1).
In contrast, GH and IL-11, both of which activate STAT3 predominantly in CECs, contribute to the suppression of experimental colitis[33-35]. Of note, human trials utilizing GH, IL-11 and anti-IL-6R mAb have been conducted and have shown that the strategies using these products are beneficial to IBD patients[38-40] (Figure 1).
IL-22 has recently been shown to be preferentially expressed by the Th17 subset[41,42] and has been demonstrated as a strong activator of STAT3[43-45]. Recently, we have demonstrated that IL-22 contributes to rapid amelioration of local inflammation associated with a Th2-mediated colitis through activation of STAT3 in CECs[46] (Figure 1). Since IL-22 specifically targets innate immune pathways[16,45-47], selective activation of STAT3 in CECs, but not acquired immune cells by IL-22 may contribute to amelioration or chronic Th2-mediated colitis.
These findings suggest that it is possible to activate or inactivate STAT3 in specific cell populations for the treatment of human IBD.
STAT3 has been shown to induce anti-apoptotic genes such as bcl-2 and bcl-xl in T cells, and increase to resistance of lamina propria pathogenic T cells to apoptosis[9]. In contrast, STAT3 can induce several molecules involved in the regulation of inflammation in CECs. Recent studies have demonstrated that STAT3 is required for the induction of IL-10, a well-known regulatory cytokine that is involved in the suppression of several inflammatory processes[48] including experimental colitis[15,49,50]. Local, but not systemic administration of this cytokine is of benefit in human IBD[51,52]. Impaired IL-10 production in macrophage by the STAT3 deletion is involved in the development of colitis[15], and enhanced production of this cytokine by CEC contributes to the suppression of colitis[53]. Importantly, IL-22, a strong activator of STAT3, has been shown to induce IL-10 production by CEC lines[45]. In addition to IL-10, STAT3 activation can also induce the expression of suppressors of cytokine signaling (SOCS) 3 that has been shown to play an important role in the suppression of acute colitis[11,17]. Furthermore, STAT3 has been recently demonstrated to induce trefoil factors that regulate intestinal epithelial homeostasis[22]. Moreover, IL-22-mediated STAT3 activation contributes to the improvement of colitis-associated mucus layer destruction associated with goblet cell depletion by enhancing the production of membrane-bound mucins (MUC1, -3, -10, and -13)[46]. Membrane-bound mucins form a static external barrier at the epithelial surface and are stored in goblet cell vacuoles[54-56]. Therefore, our recent study suggests that the enhanced mucus barrier formation participates in the IL-22/STAT3-mediated attenuation of Th2-mediated colitis[46].
Recent data indicate that STAT3 is one of the crucial targets for the treatment of IBD. However, as the receptors of these cytokines and growth factors are present on both innate and acquired cells, activation of STAT3 is likely to occur in both cell types[5,14,34-36,57,58]. Therefore, as the function of STAT3 is a double-edged sword, careful attention should be directed toward the cell population that is being targeted when one contemplates STAT3 inhibition or activation in human IBD[59].
Peer reviewers: Atsushi Mizoguchi, Assistant Professor, Department of Experimental Pathology, Massachusetts General Hospital, Simches 8234, 185 Cambridge Street, Boston MA 02114, United States; Frank Hoentjen, PhD, Free University Medical Center, Sumatrastraat 16, Haarlem 2022XL, Netherlands; Beth A McCormick, PhD, Department of Pediatric Gastroenterology and Nutrition, Massachusetts General Hospital/Harvard Medical School, 114 16th Street (114-3503), Charlestown 02129, United States; Anthony J Bauer, Professor, PhD, University of Pittsburgh, Department of Medicine/Gastroenterology, S-849 Scaife Hall, 3550 Terrace Street, Pittsburgh 15261, United States
S- Editor Li DL L- Editor Alpini GD E- Editor Ma WH
| 1. | Podolsky DK. Inflammatory bowel disease. N Engl J Med. 2002;347:417-429. |
| 2. | Strober W, Fuss IJ, Blumberg RS. The immunology of mucosal models of inflammation. Annu Rev Immunol. 2002;20:495-549. |
| 3. | Mudter J, Neurath MF. Il-6 signaling in inflammatory bowel disease: pathophysiological role and clinical relevance. Inflamm Bowel Dis. 2007;13:1016-1023. |
| 4. | Rawlings JS, Rosler KM, Harrison DA. The JAK/STAT signaling pathway. J Cell Sci. 2004;117:1281-1283. |
| 5. | Levy DE, Darnell JE Jr. Stats: transcriptional control and biological impact. Nat Rev Mol Cell Biol. 2002;3:651-662. |
| 6. | Lovato P, Brender C, Agnholt J, Kelsen J, Kaltoft K, Svejgaard A, Eriksen KW, Woetmann A, Odum N. Constitutive STAT3 activation in intestinal T cells from patients with Crohn's disease. J Biol Chem. 2003;278:16777-16781. |
| 7. | Musso A, Dentelli P, Carlino A, Chiusa L, Repici A, Sturm A, Fiocchi C, Rizzetto M, Pegoraro L, Sategna-Guidetti C. Signal transducers and activators of transcription 3 signaling pathway: an essential mediator of inflammatory bowel disease and other forms of intestinal inflammation. Inflamm Bowel Dis. 2005;11:91-98. |
| 8. | Mudter J, Weigmann B, Bartsch B, Kiesslich R, Strand D, Galle PR, Lehr HA, Schmidt J, Neurath MF. Activation pattern of signal transducers and activators of transcription (STAT) factors in inflammatory bowel diseases. Am J Gastroenterol. 2005;100:64-72. |
| 9. | Atreya R, Mudter J, Finotto S, Mullberg J, Jostock T, Wirtz S, Schutz M, Bartsch B, Holtmann M, Becker C. Blockade of interleukin 6 trans signaling suppresses T-cell resistance against apoptosis in chronic intestinal inflammation: evidence in crohn disease and experimental colitis in vivo. Nat Med. 2000;6:583-588. |
| 10. | Yamamoto M, Yoshizaki K, Kishimoto T, Ito H. IL-6 is required for the development of Th1 cell-mediated murine colitis. J Immunol. 2000;164:4878-4882. |
| 11. | Suzuki A, Hanada T, Mitsuyama K, Yoshida T, Kamizono S, Hoshino T, Kubo M, Yamashita A, Okabe M, Takeda K. CIS3/SOCS3/SSI3 plays a negative regulatory role in STAT3 activation and intestinal inflammation. J Exp Med. 2001;193:471-481. |
| 12. | Siegmund B, Lehr HA, Fantuzzi G. Leptin: a pivotal mediator of intestinal inflammation in mice. Gastroenterology. 2002;122:2011-2025. |
| 13. | Wegenka UM, Buschmann J, Lutticken C, Heinrich PC, Horn F. Acute-phase response factor, a nuclear factor binding to acute-phase response elements, is rapidly activated by interleukin-6 at the posttranslational level. Mol Cell Biol. 1993;13:276-288. |
| 15. | Takeda K, Clausen BE, Kaisho T, Tsujimura T, Terada N, Forster I, Akira S. Enhanced Th1 activity and development of chronic enterocolitis in mice devoid of Stat3 in macrophages and neutrophils. Immunity. 1999;10:39-49. |
| 16. | Wolk K, Kunz S, Witte E, Friedrich M, Asadullah K, Sabat R. IL-22 increases the innate immunity of tissues. Immunity. 2004;21:241-254. |
| 17. | Yasukawa H, Sasaki A, Yoshimura A. Negative regulation of cytokine signaling pathways. Annu Rev Immunol. 2000;18:143-164. |
| 18. | Takeda K, Noguchi K, Shi W, Tanaka T, Matsumoto M, Yoshida N, Kishimoto T, Akira S. Targeted disruption of the mouse Stat3 gene leads to early embryonic lethality. Proc Natl Acad Sci USA. 1997;94:3801-3804. |
| 19. | Takeda K, Kaisho T, Yoshida N, Takeda J, Kishimoto T, Akira S. Stat3 activation is responsible for IL-6-dependent T cell proliferation through preventing apoptosis: generation and characterization of T cell-specific Stat3-deficient mice. J Immunol. 1998;161:4652-4660. |
| 20. | Sano S, Itami S, Takeda K, Tarutani M, Yamaguchi Y, Miura H, Yoshikawa K, Akira S, Takeda J. Keratinocyte-specific ablation of Stat3 exhibits impaired skin remodeling, but does not affect skin morphogenesis. EMBO J. 1999;18:4657-4668. |
| 21. | Welte T, Zhang SS, Wang T, Zhang Z, Hesslein DG, Yin Z, Kano A, Iwamoto Y, Li E, Craft JE. STAT3 deletion during hematopoiesis causes Crohn's disease-like pathogenesis and lethality: a critical role of STAT3 in innate immunity. Proc Natl Acad Sci USA. 2003;100:1879-1884. |
| 22. | Tebbutt NC, Giraud AS, Inglese M, Jenkins B, Waring P, Clay FJ, Malki S, Alderman BM, Grail D, Hollande F. Reciprocal regulation of gastrointestinal homeostasis by SHP2 and STAT-mediated trefoil gene activation in gp130 mutant mice. Nat Med. 2002;8:1089-1097. |
| 23. | Alonzi T, Newton IP, Bryce PJ, Di Carlo E, Lattanzio G, Tripodi M, Musiani P, Poli V. Induced somatic inactivation of STAT3 in mice triggers the development of a fulminant form of enterocolitis. Cytokine. 2004;26:45-56. |
| 24. | Mizoguchi A, Mizoguchi E, Bhan AK. Immune networks in animal models of inflammatory bowel disease. Inflamm Bowel Dis. 2003;9:246-259. |
| 25. | Powrie F. T cells in inflammatory bowel disease: protective and pathogenic roles. Immunity. 1995;3:171-174. |
| 26. | Blumberg RS, Saubermann LJ, Strober W. Animal models of mucosal inflammation and their relation to human inflammatory bowel disease. Curr Opin Immunol. 1999;11:648-656. |
| 27. | Neurath MF, Finotto S, Glimcher LH. The role of Th1/Th2 polarization in mucosal immunity. Nat Med. 2002;8:567-573. |
| 28. | Sartor RB. Therapeutic manipulation of the enteric microflora in inflammatory bowel diseases: antibiotics, probiotics, and prebiotics. Gastroenterology. 2004;126:1620-1633. |
| 29. | Dalwadi H, Wei B, Kronenberg M, Sutton CL, Braun J. The Crohn's disease-associated bacterial protein I2 is a novel enteric t cell superantigen. Immunity. 2001;15:149-158. |
| 30. | Lodes MJ, Cong Y, Elson CO, Mohamath R, Landers CJ, Targan SR, Fort M, Hershberg RM. Bacterial flagellin is a dominant antigen in Crohn disease. J Clin Invest. 2004;113:1296-1306. |
| 31. | Podolsky DK. Mucosal immunity and inflammation. V. Innate mechanisms of mucosal defense and repair: the best offense is a good defense. Am J Physiol. 1999;277:G495-G499. |
| 32. | Hooper LV, Gordon JI. Commensal host-bacterial relationships in the gut. Science. 2001;292:1115-1118. |
| 33. | Williams KL, Fuller CR, Dieleman LA, DaCosta CM, Haldeman KM, Sartor RB, Lund PK. Enhanced survival and mucosal repair after dextran sodium sulfate-induced colitis in transgenic mice that overexpress growth hormone. Gastroenterology. 2001;120:925-937. |
| 34. | Kiessling S, Muller-Newen G, Leeb SN, Hausmann M, Rath HC, Strater J, Spottl T, Schlottmann K, Grossmann J, Montero-Julian FA. Functional expression of the interleukin-11 receptor alpha-chain and evidence of antiapoptotic effects in human colonic epithelial cells. J Biol Chem. 2004;279:10304-10315. |
| 35. | Peterson RL, Wang L, Albert L, Keith JC Jr, Dorner AJ. Molecular effects of recombinant human interleukin-11 in the HLA-B27 rat model of inflammatory bowel disease. Lab Invest. 1998;78:1503-1512. |
| 36. | Ohta N, Hiroi T, Kweon MN, Kinoshita N, Jang MH, Mashimo T, Miyazaki J, Kiyono H. IL-15-dependent activation-induced cell death-resistant Th1 type CD8 alpha beta+NK1.1+ T cells for the development of small intestinal inflammation. J Immunol. 2002;169:460-468. |
| 37. | Hokama A, Mizoguchi E, Sugimoto K, Shimomura Y, Tanaka Y, Yoshida M, Rietdijk ST, de Jong YP, Snapper SB, Terhorst C. Induced reactivity of intestinal CD4(+) T cells with an epithelial cell lectin, galectin-4, contributes to exacerbation of intestinal inflammation. Immunity. 2004;20:681-693. |
| 38. | Ito H, Takazoe M, Fukuda Y, Hibi T, Kusugami K, Andoh A, Matsumoto T, Yamamura T, Azuma J, Nishimoto N. A pilot randomized trial of a human anti-interleukin-6 receptor monoclonal antibody in active Crohn's disease. Gastroenterology. 2004;126:989-996; discussion 947. |
| 39. | Slonim AE, Bulone L, Damore MB, Goldberg T, Wingertzahn MA, McKinley MJ. A preliminary study of growth hormone therapy for Crohn's disease. N Engl J Med. 2000;342:1633-1637. |
| 40. | Sands BE, Bank S, Sninsky CA, Robinson M, Katz S, Singleton JW, Miner PB, Safdi MA, Galandiuk S, Hanauer SB, Varilek GW, Buchman AL, Rodgers VD, Salzberg B, Cai B, Loewy J, DeBruin MF, Rogge H, Shapiro M, Schwertschlag US. Preliminary evaluation of safety and activity of recombinant human interleukin 11 in patients with active Crohn's disease. Gastroenterology. 1999;117:58-64. |
| 41. | Liang SC, Tan XY, Luxenberg DP, Karim R, Dunussi-Joannopoulos K, Collins M, Fouser LA. Interleukin (IL)-22 and IL-17 are coexpressed by Th17 cells and cooperatively enhance expression of antimicrobial peptides. J Exp Med. 2006;203:2271-2279. |
| 42. | Zheng Y, Danilenko DM, Valdez P, Kasman I, Eastham-Anderson J, Wu J, Ouyang W. Interleukin-22, a T(H)17 cytokine, mediates IL-23-induced dermal inflammation and acanthosis. Nature. 2007;445:648-651. |
| 43. | Radaeva S, Sun R, Pan HN, Hong F, Gao B. Interleukin 22 (IL-22) plays a protective role in T cell-mediated murine hepatitis: IL-22 is a survival factor for hepatocytes via STAT3 activation. Hepatology. 2004;39:1332-1342. |
| 44. | Brand S, Beigel F, Olszak T, Zitzmann K, Eichhorst ST, Otte JM, Diepolder H, Marquardt A, Jagla W, Popp A. IL-22 is increased in active Crohn's disease and promotes proinflammatory gene expression and intestinal epithelial cell migration. Am J Physiol Gastrointest Liver Physiol. 2006;290:G827-G838. |
| 45. | Nagalakshmi ML, Rascle A, Zurawski S, Menon S, de Waal Malefyt R. Interleukin-22 activates STAT3 and induces IL-10 by colon epithelial cells. Int Immunopharmacol. 2004;4:679-691. |
| 46. | Sugimoto K, Ogawa A, Mizoguchi E, Shimomura Y, Andoh A, Bhan AK, Blumberg RS, Xavier RJ, Mizoguchi A. IL-22 ameliorates intestinal inflammation in a mouse model of ulcerative colitis. J Clin Invest. 2008;118:534-544. |
| 47. | Laurence A, O'Shea JJ, Watford WT. Interleukin-22: a sheep in wolf's clothing. Nat Med. 2008;14:247-249. |
| 48. | Moore KW, de Waal Malefyt R, Coffman RL, O'Garra A. Interleukin-10 and the interleukin-10 receptor. Annu Rev Immunol. 2001;19:683-765. |
| 49. | Mizoguchi A, Mizoguchi E, Takedatsu H, Blumberg RS, Bhan AK. Chronic intestinal inflammatory condition generates IL-10-producing regulatory B cell subset characterized by CD1d upregulation. Immunity. 2002;16:219-230. |
| 50. | Davidson NJ, Leach MW, Fort MM, Thompson-Snipes L, Kuhn R, Muller W, Berg DJ, Rennick DM. T helper cell 1-type CD4+ T cells, but not B cells, mediate colitis in interleukin 10-deficient mice. J Exp Med. 1996;184:241-251. |
| 51. | Van Montfrans C, Hooijberg E, Rodriguez Pena MS, De Jong EC, Spits H, Te Velde AA, Van Deventer SJ. Generation of regulatory gut-homing human T lymphocytes using ex vivo interleukin 10 gene transfer. Gastroenterology. 2002;123:1877-1888. |
| 52. | Rogy MA, Beinhauer BG, Reinisch W, Huang L, Pokieser P. Transfer of interleukin-4 and interleukin-10 in patients with severe inflammatory bowel disease of the rectum. Hum Gene Ther. 2000;11:1731-1741. |
| 53. | De Winter H, Elewaut D, Turovskaya O, Huflejt M, Shimeld C, Hagenbaugh A, Binder S, Takahashi I, Kronenberg M, Cheroutre H. Regulation of mucosal immune responses by recombinant interleukin 10 produced by intestinal epithelial cells in mice. Gastroenterology. 2002;122:1829-1841. |
| 54. | Shirazi T, Longman RJ, Corfield AP, Probert CS. Mucins and inflammatory bowel disease. Postgrad Med J. 2000;76:473-478. |
| 55. | Ho SB, Dvorak LA, Moor RE, Jacobson AC, Frey MR, Corredor J, Polk DB, Shekels LL. Cysteine-rich domains of muc3 intestinal mucin promote cell migration, inhibit apoptosis, and accelerate wound healing. Gastroenterology. 2006;131:1501-1517. |
| 56. | Corfield AP, Carroll D, Myerscough N, Probert CS. Mucins in the gastrointestinal tract in health and disease. Front Biosci. 2001;6:D1321-D1357. |
| 57. | La Cava A, Matarese G. The weight of leptin in immunity. Nat Rev Immunol. 2004;4:371-379. |
| 58. | Reinecker HC, MacDermott RP, Mirau S, Dignass A, Podolsky DK. Intestinal epithelial cells both express and respond to interleukin 15. Gastroenterology. 1996;111:1706-1713. |
| 59. | Plevy S. A STAT need for human immunologic studies to understand inflammatory bowel disease. Am J Gastroenterol. 2005;100:73-74. |