INTRODUCTION
Inflammatory bowel disease (IBD) first presents in childhood and adolescence in approximately 20% of all cases. A European study reported that in the entire IBD population (children and adults) the incidence of Crohn’s disease (CD) increased significantly (+23%), while the incidence of ulcerative colitis (UC) decreased (-17%)[1]. One of the best North American studies reported the incidence of IBD in Wisconsin children to be 7.05 per 100 000, with the incidence for Crohn’s disease being 4.56, more than twice the rate of ulcerative colitis (2.14). An equal IBD incidence occurred among all ethnic groups, and children from sparsely and densely populated counties were equally affected and the majority (89%) of new IBD diagnoses were non-familial[2]. The median age of onset is 12 years and there appears to be a slight male predominance in the younger age group. Given the rise in incidence and the onset of disease coinciding with growth and development it is very important to highlight the considerations that should be taken into account when managing childhood onset IBD. This review with underscore the many unique facets involved in the presentation, diagnosis, treatment and psychosocial well being of the child with IBD.
ETIOPATHOGENESIS
The underlying pathogenesis of IBD in children appears to be similar to that in adult onset such that IBD results from a complex interaction of environmental, genetic, and immune factors. Genetics, however, may play an even greater role in disease onset and susceptibility in patients who present earlier in life. Despite research efforts, a gene specific to pediatric onset disease has not been identified. It does appear however that the NOD2 gene is similarly present in both 30%-35% of adult and pediatric CD patients. NOD2 was first identified in 2001. Although the true pathogenic role of NOD2 in CD remains unknown, it is an important gene involved in innate immunity which lends support to the notion that genetically determined defects in innate, and likely adaptive immunity alter the way our mucosal immune system interacts with our resident bacterial flora[3]. This dysregulated interaction leads to the adaptive immune response that is, in large part, responsible for the chronic inflammatory lesions, characteristic of patients with IBD. NOD2 has been well studied in the Caucasian population, however, the frequency of this gene among patients with CD of other racial/ethnic groups in the United States is not well known. Despite similar clinical characteristics significantly lower frequencies of read NOD2 mutations have been seen in African American and Hispanic children with CD compared with Caucasian children with CD[4]. There has been some evidence to suggest that the ethnic background of a child with CD may be associated with different genetic variants. A study from Israel suggested that G908R (SPN 12) allele-variant of the NOD2/CARD15 gene is closely related to the appearance of CD at a young age in Jewish Ashkenazi patients[5]. Research is ongoing to further examine the influence of ethnicity on disease susceptibility and disease modification. Initial genotype-phenotype correlation studies in children demonstrated that NOD2 is associated with fibrostenosing CD and more rapid progression to surgery[6]. It appears that genetics is only part of the story when it comes to understanding the influences or risk factors at predicting the natural history of disease in pediatric patients. Initial work suggested that there are specific immune markers present in the serum of children with IBD[7]. These markers were initially used to help differentiate CD from UC and to aid in the distinction between functional GI symptoms and those due to underlying IBD, CD in particular. The concept that sero-reactivity to specific microbial antigens seen in a subgroup of patients with IBD likely represents a surrogate measure of an individual’s adaptive immune response has resulted in this being an important area of investigation. Since reactivity to yeast oligomannan (anti-Saccharomyces cerevisiae antibody; ASCA) was first described in Crohn’s disease (CD), the number of CD-specific antigens for which reactivity can be measured has expanded to include the Pseudomonas fluorescens related protein (I2), Escherichia coli outer membrane-porin (OmpC), and CBir1 flagellin (CBir1)[8–10].
The evolution of serum immune response from diagnostic markers to markers of disease behavior and perhaps prognostic information has resulted in studies that have shown that the presence and magnitude of immune re-sponses in a given child is associated with more aggressive disease phenotypes and more rapid disease progression to complication and surgery. Larger scale studies are underway to confirm these preliminary findings and to further develop a risk assessment tool to identify children at risk of more aggressive disease and subsequent introduction of early intervention[1112].
DIAGNOSIS
The diagnostic approach to IBD in children is a two-tiered approach. The first diagnostic question is whether the presenting symptoms are compatible with a diagnosis of IBD. In the face of classic, alarm, symptoms the differentiation between IBD and diseases that mimic the symptoms of IBD can be relatively simple. Diarrhea and abdominal pain are the most common symptoms in both CD and UC with rectal bleeding more commonly seen in UC and weight loss and anorexia more characteristic of CD. Perianal disease, like in adults, remains characteristic of CD. However unlike adults, growth failure is an important presenting sign in pediatric onset IBD, most commonly in CD patients. The standard diagnostic approach is employed including routine labs looking for signs of inflammation (ESR, CRP, platelet count) complete blood count with a focus on the hemoglobin for anemia and other labs such as iron panel and albumin to look at nutritional/absorptive state. Stools studies should be done to rule out infection especially in the presence of a travel history. Endoscopic evaluation and tissue diagnosis remains the gold standard[13]. It is recommended that all children undergo both an upper endoscopy and colonoscopy at the time of initial investigation. The findings on upper endoscopy, although often non-specific, may provide additional information in a patient with indeterminate disease of the colon especially if granulomas are found. A recent study found upper gastrointestinal inflammation in 29 of 54 children (22 CD; 7 UC)[14]. Epigastric and abdominal pain, nausea and vomiting, weight loss and pan-ileocolitis were predictive of upper gastrointestinal involvement. However, 9 children with upper gastrointestinal involvement were asymptomatic at presentation (31%). Overall upper gastrointestinal tract inflammation was most common in the stomach (67%), followed by the esophagus (54%) and duodenum (22%).Thus, absence of specific upper gastrointestinal symptoms do not preclude presence of upper gastrointestinal inflammation. It is important to biopsy macroscopically involved and non-involved tissue so to maximize the yield on the initial invasive evaluation. Small bowel radiography is also part of the initial diagnostic evaluation of pediatric IBD patients especially in CD patients. This is especially so for patients in whom ileal intubation was not successful at the time of the colonoscopy or diagnosis is indeterminate. It has been suggested that a normal small bowel radiography alone should not be used to rule out pediatric inflammatory bowel disease when the symptoms suggest it. Colonoscopy with terminal ileal intubation is feasible and safe; it should be attempted in all children with symptoms consistent with inflammatory bowel disease[15]. Radiographic evaluation of the small bowel has evolved to include MRI enterography and CT enterography. Recent advances in gadolinium-enhanced magnetic resonance imaging (G-MRI) have been developed to enhance the resolution of the intestinal mucosa and facilitate the differentiation of UC from CD[16]. Until the MRI scanners are faster and the radiologists are more comfortable interpreting the scans, MRI remains experimental and should be limited to use in centers that have a radiologists who are trained in this technology. CT enterography on the other hand is evolving into the best method of assessing the small bowel and large bowel. The use of Volumen for oral contrast allows for accurate visualization and distension of the mucosa. The radiation is somewhat greater than a standard CT scan however is less than a small bowel follow through with prolonged fluoroscopy[17]. Experience is growing among pediatric gastroenterologists in this area. Positron emission tomography (PET) using fluorine-18-fluoro-deoxyglucose to identify metabolically active tissues was evaluated as a simple noninvasive alternative to conventional studies in identification and localization of active intestinal inflammation in children with IBD[18]. The authors concluded that PET may not be able to replace conventional studies; however, it may be useful when conventional studies cannot be performed or fail to be completed. The cost associated with this test is an important limiting factor. White cell scans have also been proposed as a noninvasive way of evaluating active inflammation, however there can be many false negatives and advances in technology such as the video capsule endoscopy (VCE) provides a unique way of evaluating the small bowel mucosa with increased sensitivity and specificity[19]. VCE may be helpful in patients with persistent small bowel symptoms despite what has been reported as a normal small bowel x-ray particularly those children with persistent growth failure. Moreover in cases of indeterminate colitis, children may benefit from more detailed small bowel visualization[20].
In the face of diagnostic uncertainty, such as in children presenting with symptoms compatible but not diagnostic of IBD, the pediatric approach tends to be less invasive then the approach to an adult patient presenting with similar symptoms. In this setting pediatricians tend to use non-invasive testing first to gather information that may increase the probability of disease and hence lead to more evidence to support invasive diagnostic testing. Other than routine laboratory tests as noted above, there are fecal markers, such as calprotectin and lactoferrin, and serological markers, such as ASCA and pANCA, that may aid in the differential diagnosis. Earlier reports demonstrated that fecal calprotectin correlated closely with the best invasive measures of colonic and small bowel inflammation in childhood inflammatory bowel disease and lends itself particularly to the monitoring and assessment of therapeutic interventions in children with inflammatory bowel disease[21]. Subsequent experience with calprotectin has yielded similar findings and in some practices is used frequently to differentiate functional from inflammatory GI disorders. Novel technologies have led to the development of a diagnostic algorithm to evaluate the sensitivity and specificity of serological immune markers as predictors of IBD vs non IBD. Historically these tests were accurate in differentiating IBD from non-IBD in 2/3 of cases[22]. The addition of novel immune responses and a change in technology may improve the diagnostic accuracy of non-invasive testing to close to 90%. Further validation studies in pediatric patients are needed to determine if indeed these tests will help pediatric gastroenterologist increase the likelihood of diagnosing IBD in the face of positive markers. In the adult cohort that these tests were validated in, it appears that disease was ruled out with approximately 90% certainty. This would be an important advance in pediatrics where invasive testing is avoided if possible.
The next tier in the diagnostic process of IBD is the differentiation between CD and UC. In the face of a classic CD presentation which includes small bowel localization, presence of granulomas, typical endoscopic and histological findings, the differentiation is simple. However there are some features of pediatric onset IBD that may pose more of a diagnostic dilemma when disease is confined to the colon. Despite the classic teachings of UC always involving the rectum, there are reports of rectal sparing in pediatric patients carrying a diagnosis of UC. The group from Boston reported that a significant proportion of children with new-onset UC had patchiness of microscopic features of chronicity (21% of patients), relative (23%), or absolute (3%), rectal sparing, and had little or no crypt architectural distortion in their rectal biopsies (8%). Of interest, these features were not observed in adult patients with UC. In addition, a higher proportion of children with UC initially presented with subtotal or with pancolitis compared with the adults[23]. Another study demonstrated that children < 10 years of age had significantly less crypt branching, plasma cells in the lamina propria, cryptitis, crypt abscesses, and epithelial injury than adults. Endoscopic rectal sparing was also seen in another study in 23% of children with newly diagnosed, untreated UC, and this feature did not correlate with presenting symptoms. However, the presence of rectal sparing may indicate more aggressive disease that is less responsive to medical treatment[24]. These studies must be interpreted with caution as these patients likely have what has become known as UC-like CD and although clinically present with UC symptoms, there are endoscopic and histological features more characteristic of CD. In these cases a more accurate evaluation of the small bowel with video capsule endoscopy may be warranted and perhaps serological immune responses will be helpful in differentiating between CD and UC. Studies have shown that pANCA is more commonly seen in patients with ulcerative colitis and ASCA and OmpC are prevalent in CD patients. It was then found that pANCA may be present in 25% of all CD patients, predominantly in patients with disease confined to the colon. A novel CD marker, anti-CBir1, has been shown to be present in approximately 50% of pANCA positive CD patients but rare (< 5%) of pANCA positive UC patients[25]. This marker may help differentiate CD from UC in indeterminate cases. This may be important as the efficacy of therapies may differ between the 2 pANCA subgroups. Further research is underway to delineate further the role of these markers in predicting response to therapy and post surgical prognosis[26].
TREATMENT
Modern approaches to IBD therapy calls upon the need for disease modifying targets with the goal of mucosal healing . It is hypothesized that mucosal healing should reduce disease related complications and in turn maximize the quality of life of IBD patients. This may be even more of an important issue when treating children with IBD given the long term consequences of early onset aggressive disease presentations. Currently there are few therapies approved for children with IBD, however, the concepts of targeting the full spectrum of the inflammatory cascade and the notion of mucosal healing can be applied. The main goals of targeting therapy in pediatric IBD are to: (1) Maximizing efficacy; (2) Maximize adherence; (3) Minimize toxicity; (4) Maximize quality of life; (5) Maintain physical and psychosocial growth; (6) Prevention of disease complications.
Like their adult counterparts, the current approach to the treatment of children with IBD is based on disease severity. In other words the traditional “step up” approach is applied in most cases. This approach also takes into account medication safety as the more milder/less toxic medications are often employed first letting patients declare themselves failures necessitating navigating up the pyramid to more aggressive anti-inflammatory agents. Pediatric gastroenterologists are limited in their ability to interpret whether this is the correct strategy given few studies have been done in children to support use of these medications especially the Mesalamine based therapies. Often we are forced to extrapolate information from adult study populations which may not be applicable to a child. Given the potential growth and development implications of persistent inflammation and corticosteroid dependency, efforts are made to maximize both anti-inflammatory and steroid sparing strategies. Aside from the potential growth effects, the esthetic changes associated with corticosteroid use can be devastating to a child. Nutritional therapy is typically unique to pediatric patients however compliance can be an obstacle to administration. A recent prospective, 10-week open-label trial in children with active naive CD randomized children to orally polymeric formula alone or oral corticosteroids. In this small study, children with active and recently diagnosed CD, a short course of polymeric diet was more effective than corticosteroids in inducing healing of gut inflammatory lesions[27]. Further large scaled studies are needed to further evaluate the short and long term benefits of this treatment strategy. In the USA, enteral nutrition is used more so as supplemental nutrition in the face of malnutrition and growth failure.
Recently, there has been a lot of discussion surrounding the notion of turning the therapeutic pyramid upside down, aka “top-down” therapy. In other words in patients who are candidates for corticosteroids, more potent biological therapies, such anti-Tumor Necrosis Factorα (anti-TNFα) therapies, may be considered as alternatives[28]. However the strategy of short term corticosteroids with 6-MP as bridge therapy may be a safer and equally effective strategy in this patient population. Markowitz et al demonstrated that a significant proportion of children were off steroids and in remission 600 days after the combination therapy was initiated[29]. If however the desired outcome of a steroid free remission is not achieved in the expected time frame (4-6 months) of this combination then at that time the introduction of a biological therapy should be considered. The hesitation to go directly to a biologic stems from the fact that the thiopurines work well in children and the serious safety concerns. There are definitely safety concerns associated with thiopurines as well, however, TPMT screening and metabolite monitoring enable clinicians to identify at risk patients and dose adjust so to minimize toxicity and dose escalate safely in patients with sub-therapeutic levels and not responding to their current dose[30]. The REACH trial was the first of its kind in pediatrics to evaluate in a multi-center fashion the efficacy and safety of infliximab in 113 patients[31]. The study was not powered for efficacy but the results do support its use in children with the response rate at 10 wk was close to 90% and a remission rate at 54 wk of approximately 50% which includes children off of corticosteroids when receiving drug every 8 wk as opposed to every 12 wk. The safety may have been more favorable among patients receiving the every 12 wk infusions, however the efficacy benefit of every 8 wk may outweigh its safety risks. The data lead to the approval of infliximab for children with luminal Crohn’s disease. Clinical trials are underway to evaluate the efficacy and safety of infliximab in children with ulcerative colitis as well as adalimumab for pediatric CD. Weighing the risks and benefits of each therapy must be considered and should be communicated with the child and the family. New safety information has emerged which has already started to alter the approach to patients receiving infliximab. There have been 18 confirmed cases of hepato-splenic T cell lymphoma (HSTCL) reported in patients receiving combination thiopurines and anti-TNFα therapy. Although rare, the majority of cases are fatal which has forced pediatricians to rethink their approach to this patient population. This calls into question of concomitant immunomodulation for the purpose of immunogenicity and perhaps improvement in response rates and how it relates to safety. In REACH all children had to be on concomitant therapy to be eligible so it is not known whether monotherapy would have been as effective. In the adult ACCENT1 trial only one-third of patients were on concomitant thiopurine or methotrexate and at the end of one year the remission rate off corticosteroids was only 24%[32]. Although the median duration of disease at the time of a patient’s first infliximab exposure was much lower in REACH as compared to ACCENT 1 and the patient populations are different, one must question as to whether there is an efficacy advantage of the combination as opposed to monotherapy. A significant proportion of children are being removed from thiopurines and continuing the infliximab and some clinicians are extrapolating data from the rheumatoid arthritis literature that suggests that low dose methotrexate (7.5 mg po weekly) is associated with increased through infliximab levels and decreased levels of antibodies to infliximab[33]. There is no data among pediatric patients and further research is needed to validate this strategy. Methotrexate in any form of administration has not been common place in pediatrics. There was always the notion that injections are traumatizing to children and that the oral form may be associated with treatment limiting nausea and the bioavailability was inferior to that when given subcutaneously or intramuscularly. A more recent report however showed that the bioavailability of methotrexate in patients with inflammatory bowel disease is no different from that observed in other disease states[34]. Perhaps the key to deciding the best strategy for an individual patient will be in identifying the at risk patient up front whose risk of untreated progressive disease outweighs the risk of the medications. A recent study reported that imunomodulators are used in approximately 60% of children with CD within 1 year of diagnosis and suggested that lower serum albumin levels and hematocrit, and elevated erythrocyte sedimentation rate at diagnosis may predict the need for immunomodulators earlier in the disease course[34].
Future research will evaluate the role genetics and immune responses as immunological risk factors for aggressive disease. In other words, stratify patients into high risk and low risk which will in turn modify treatment approaches.
SPECIAL CONSIDERATIONS
Bones and growth
Consideration must be given to those issues which are unique to children. Most adult and pediatric gastroenterologists would agree that the presence of growth and pubertal delay is a key factor in the management of pediatric IBD. Puberty is the most dynamic phase of growth in childhood. Maintaining adequate nutrition, minimizing inflammation and maximizing treatment off of corticosteroids remains an integral part of managing the potential growth stunting effects of active IBD, most specifically small bowel CD. Growth failure has been reported in up to 40% of children with CD and < 10% of UC. On occasion growth and pubertal delay is the only presenting sign of IBD and can precede any GI symptoms[36]. Growth failure is secondary to malnutrition with decreased intake contributing significantly to the malnutrition. Other factors such as increased GI losses, malabsorption, psychosocial factors and medication effects can certainly impact an individual’s nutritional state. Ongoing inflammation with release of specific cytokines that suppress growth factors has been shown to be a very important determinate of growth failure. Evidence suggests that IL-6 mediates growth failure in children with Crohn’s disease[37]. Nutritional supplementation and need for “catch-up” growth should be an important part of the evaluation of a pediatric IBD patient. A child’s self esteem can be significantly impacted by growth failure and every effort should be made to make this a priority in the management plan. Administration of growth hormone was examined in a pilot study (7 patients) and did not demonstrate any effect on growth[38]. However the combination of growth hormone with nutritional supplementation in an IBD patients would likely yield positive results given the important of adequate caloric intake in patients with ongoing inflammation.
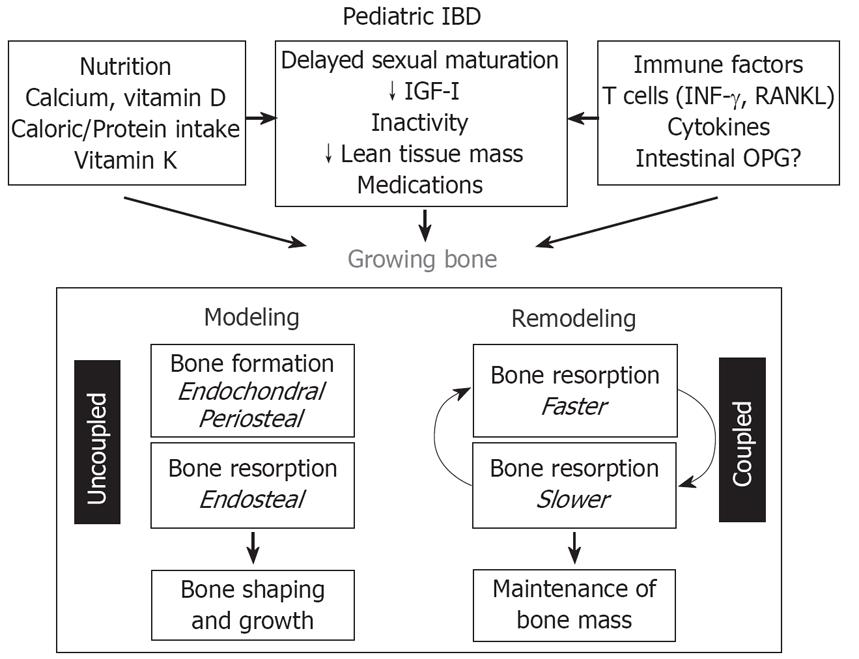
Defective bone mass accrual is another complication of chronic inflammatory disease and the major causative factor is growth failure. There are, however, other variables, such as physical activity, altered body composition, and disordered calcium and vitamin D metabolism which certainly play a role in bone mass. Persistent inflammation in the face of IBD can also impact the maintenance of bone mass. The method for assessment of bone mass in children is very important and what is currently used in adults does not apply to the pediatric age group. Bone mineral density (BMD) T-scores are appropriate for individuals that have reached skeletal maturity, but they should not be used in pediatric DEXA reports[39]. Instead, a Z-score should be calculated by subtracting the measured BMD from the expected BMD for individuals of the same age and sex, and dividing the result by the standard deviation. Additional adjustments may be needed in small or physically immature children (e.g., using height age or bone age instead of chronological age for Z-score calculation). It is important to note that a diagnosis of osteoporosis should not be made in children based on DEXA results alone. Experts in the field suggest that a Z-score of < -2.0 in children should be reported descriptively as ‘‘reduced bone mineral mass for age’’[40]. Because of these limitations, total body bone mineral content may be clinically more useful than BMD in children, especially when studying patients over a longitudinal time frame. Quantitative computed tomography (QCT) is an alternative to measure BMD in both adults and children[41]. QCT offers the advantage of providing a true volumetric BMD, and it can distinguish the individual contributions of cortical and trabecular bone. QCT can be performed in conventional CT scanners, usually in the lumbar vertebrae. It involves a higher radiation dose than DEXA. CT devices are available that measure BMD in the peripheral skeleton, with minimal radiation exposure, but the clinical significance of these measurements in children and adults with IBD is not known. Other experts in the field have shown that the apparent prevalence of osteopenia in children with IBD differs according to the method of data analysis used. Failure to account for bone age led to a label of moderate or severe osteopenia in 65% of cases. After adjustment for bone age, the proportion of children with osteopenia fell to 22%. Data suggests that children with IBD often have small bones for age because they have growth retardation. When DEXA data are interpreted with adjustment for bone size, most children were found to have adequate bone mass. Correct interpretation of DEXA is important for identifying children who may be at a real risk of osteoporosis[42] (Figure 1).
Figure 1 The effect of IBD on skeletal development.
Quality of life and psychosocial issues
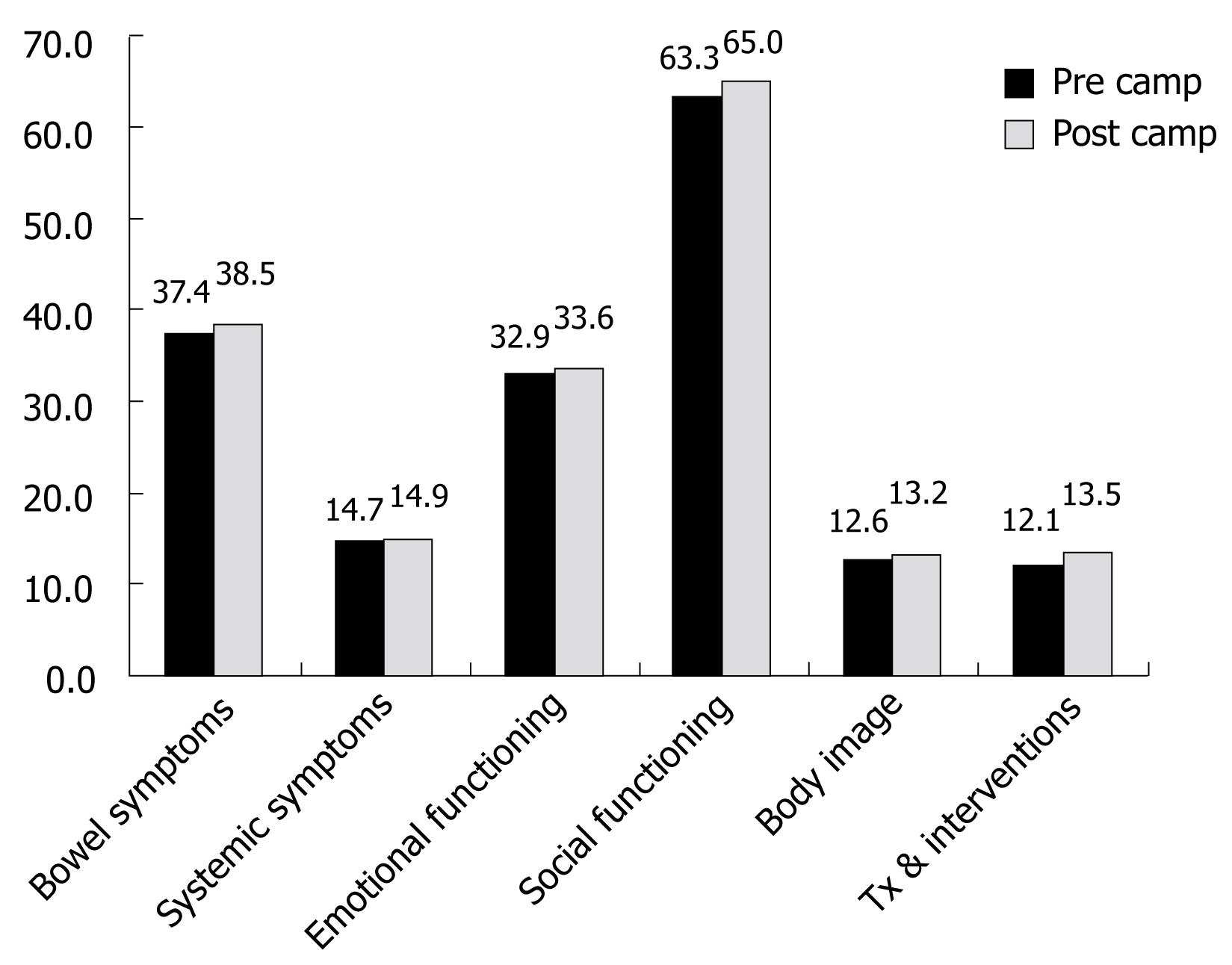
The burden of disease, both physical and psychosocial, imposed on children by Crohn’s disease (CD) and ulcerative colitis (UC) can be considerable. In order to asses this burden the IMPACT questionnaire, a disease-specific measure of health related quality of life (HRQOL), was developed and validated for use in children and youth with IBD ages 10 to 18 years inclusive[4344]. One study reported that the majority of patients perceived an improvement in HRQOL within 1 year of diagnosis and this improvement was in keeping with the overall improvement in disease severity[45]. Hester et al asked whether it may prove helpful to ask significant others or caregivers besides the patients themselves when evaluating health and the perception of health of patients[46]. This study reported that parents and children with IBD show high agreement when reporting observable aspects of the child’s HRQOL. On the other hand, agreement was lower when it concerns more subjective aspects of HRQOL, such as social functioning and emotions. It is worth noting that children reported fewer problems than their parents. Through the CCFA, summer camp programs are available for children with CD and UC. The goal of such camp programs is to allow children to participate in normal camp activities surrounded by their peers dealing with similar childhood issues. It has been suggested that camp may provide additional psychosocial benefits such as increased self esteem, altered perceptions and attitudes towards illness[47]. A prospective analysis of quality of life was completed at an overnight camp using the IMPACT II questionnaire (Canada and United States) and the State-Trait Anxiety Inventory for Children. These questionnaires were administered to the campers at the beginning and at the end of a 1-wk camp to assess HRQOL and anxiety. No difference was found between anxiety scores before and after camp on either the state or trait anxiety inventories. HRQOL, however, improved after the week of summer camp suggesting that camp may normalize the chronic illness experience[48] (Figure 2).
Figure 2 The impact of summer camp on HRQOL in children with IBD.
Psychosocial functioning and medication adherence
When comparing self-reported psychosocial functioning (behavioral/emotional functioning, social competence, self-esteem, stress coping strategies, and social support) of children with IBD to that of healthy children, it appears that most children with mild IBD report normal psychosocial functioning that is similar to that of healthy children[49]. Although rates range, adherence rates among chronic disease in children are typically reported to be approximately 50% with adherence being the lowest in adolescence and when maintenance medications are used even when the disease is in remission[50]. There are multiple factors that impact adherence in pediatric chronic illness which makes it potentially a more complex issue than in the adult population. Parents are often responsible for ensuring their children take their medication, so when evaluating adherence, both the patient and parent must be considered. One study examined the reports of adherence to oral medications, parent-child concordance in reports of adherence, and factors associated with poor adherence in adolescents with IBD. Mean parent- and child-reported adherence scores fell between the ‘‘most of the time’’ and ‘‘always’’ categories, although perfect adherence was low. Among IBD-specific medications < 50% of children and < 40% of parents reported being always adherent to all medications and parent-child concordance was high. Family dysfunction and poor child coping strategies were associated with worse adherence and there appeared to be a trend between more behavioral/emotional problems and lower adherence. Based on these results the authors concluded that adherence should be monitored in families that lack appropriate child discipline and in children who cope by simply wishing stressors would go away[51].
Transition of care
The transition from pediatric to adult medical care of patients with IBD can be difficult for the child and care givers. Clinicians must be sensitive to this transition and the barriers it may present. It has been recommended that the child be approached from the commonly accepted developmental stages, roughly defined chronology by ages 11-13, 14-16, 17-19, and 20-23[52]. These are certain traits needed to make a successful transition of care and allow for the ability to navigate through the adult healthcare milieu. Communication is critical in order for the child and caregiver to anticipate the new roles each member will play in this transition. The process of transition should be a gradual and if the process is delayed the transition may be less successful as the time to prepare and anticipate change has been limited. It is not unusual for the process to be more difficult on the family/primary caregivers than the child themselves. Family members need to relinquish responsibility and the weaning process should begin fairly early in adolescence so that when the time comes to meet with an adult gastroenterologist the patient and caregiver are prepared. In preparation patients may want to put a portfolio together whereby key documents can be brought to their visit to minimize duplication of history taking and perhaps procedures and tests. Examples would include, medical summaries, procedure reports, surgical reports, medication history, recent laboratory results and health insurance information.
Immunization status
Immunosuppressants/immunomodulators have become an integral part of the medical management of IBD both in adults and children. Guidelines have been proposed that take into account the interplay of host susceptibility to infection and the safety and efficacy of immunization for vaccine-preventable diseases. The guidelines suggest that with the exception of live agent vaccines [MMR (measles, mumps, rubella) and Varicella (chicken pox)], most immunizations (killed/attenuated) can be safely administered to patients with IBD even when immune compromised. Moreover, protection against vaccine-preventable illness may be of even greater benefit to those at risk for morbid or lethal complications of infections because of an immune compromised state. It was concluded that for most patients with IBD, recommendations for immunization do not deviate from recommended schedules for the general population[53]. Highlights of the recommendations from this report are the following: (1) Standard recommended immunization schedules for children and adults should be generally adhered to. (2) At diagnosis, children and adults should have complete review of immunization history for completeness. All patients with incomplete series should commence catch-up vaccination. (3) Children who are not immune by vaccination or acquired immunity through infection should receive varicella vaccine. (4) Live bacterial or viral vaccines should be avoided in immune compromised children with IBD: (a) Treatment with glucocorticoids (prednisone 20 mg/d equivalent, or 2 mg/kg per day if less than 10 kg, for 2 wk or more, and within 3 mo of stopping); (b) Treatment with effective doses of 6-mercaptopurine/azathioprine (effect on safety not established) and within 3 months of stopping; (c) Treatment with methotrexate (effect on safety not established) and within 3 months of stopping; (d) Treatment with infliximab (effect on safety not established) and within 3 months of stopping; (e) Significant protein-calorie malnutrition. (5) Whenever possible, adequate immune response (ie. Tetanus toxoid antibody, H influenza B antibody) should be ascertained for individuals who have required immunization while immune-suppressed. Repeat dosing may be considered when immune response to immunization is insufficient.
CONCLUSION
Approximately one-quarter of IBD presents in childhood, most commonly among the adolescent age group which corresponds to the most dynamic phase of growth. The disease can impact both physical and psychosocial development. Growth failure and bone health are important consideration in the pediatric age group. The diagnosis and treatment requires a comprehensive approach to improve the health related quality of life of children with IBD. Treatments are improving but individualized therapeutic strategies will be a critical part in the management of pediatric IBD so to maximize the therapeutic efficacy and minimize toxicity. Parents and patients must be prepared for the transition from pediatric to adult care and tools are available to improve the success of this process.
S- Editor Liu Y E- Editor Liu Y