Published online Jul 14, 2008. doi: 10.3748/wjg.14.4204
Revised: May 23, 2008
Accepted: May 30, 2008
Published online: July 14, 2008
AIM: To explore the prevalence of SEN virus (SENV) in patients with non A-E hepatitis and volunteer blood donors in Shanghai.
METHODS: According to the published gene sequences, primers from the conserved region were designed. Then, the prevalence of SEN virus in 30 samples from healthy voluntary blood donors and 30 samples from patients with non A-E hepatitis were detected by nested-PCR of SENV-D/H. Some PCR products were cloned and sequenced.
RESULTS: The specificity of genotype-specific PCR was confirmed by sequencing, the SENV DNA was detected in 53.3% of the patients with non A-E hepatitis and 10% of the blood donors. The prevalence of SENV-D/H viremia was significantly higher in patients with non A-E hepatitis than in blood donors (P = 0.0002). SENV-H subtype and SENV-D subtype were found in 2 and 1 samples, respectively from blood donors. SENV-H subtype, SENV D subtype, mixed SENV-D and SENV-H subtype were found in 8, 6 and 2 samples, respectively, from patients with non A-E hepatitis.
CONCLUSION: The gene type of SENV in patients with non A-E hepatitis and blood donors in shanghai is D or H subtype, and transfusion is not the only transmitting form of SENV.
- Citation: Tang ZH, Chen XH, Yu YS, Zang GQ. Prevalence and clinical significance of SEN virus infection in patients with non A-E hepatitis and volunteer blood donors in Shanghai. World J Gastroenterol 2008; 14(26): 4204-4208
- URL: https://www.wjgnet.com/1007-9327/full/v14/i26/4204.htm
- DOI: https://dx.doi.org/10.3748/wjg.14.4204
A DNA virus-designated SEN virus (SENV), which was discovered in the serum of a human immunodeficiency virus type 1 (HIV-1)-infected patient, has been described recently[1]. It is assumed to be transmitted parenterally and to cause posttransfusion hepatitis in humans. SENV is described as a small single-stranded, non-enveloped circular DNA virus containing a genome of approximately 3800 nucleotides, possibly belonging to the circoviridae family. To date, eight distinct strains of SENV (A-H) have been identified[2]. There are 15%-50% sequence diversities among them. Phylogenetically, SENV is distantly related to TTV with which it shares a similar structural organization but only about 40%- 60% sequence homologies[3]. According to previous studies, the prevalence of these eight different strains of SENV (A-H) is different in each infected group. The prevalence of SENV-D or SENV-H strains was 2.25% and 92.31% in different groups including healthy blood donors, patients with acute or chronic non A-E hepatitis, respectively, suggesting that SENV-D or SENV-H is significantly associated with the pathogenesis of non A-E hepatitis[3].
To date, the prevalence of SENV in patients with various forms of liver disease has been reported in many countries and several districts of China, but the results are not consistent to a certain extent, and the role of SENV infection in patients with non A-E hepatitis or other viral hepatitis, and the transmitting form of SENV are not very clear[4–7]. The purpose of this study was to investigate the prevalence and molecular biology characteristic of SENV in patients with non A-E hepatitis and volunteer blood donors in Shanghai, and the possible clinical significance of SENV infection in patients with non A-E hepatitis.
A total of 30 serum samples from volunteer blood donors and 30 serum samples from patients with non A-E hepatitis from January 2005 to June 2007 in sixth people’s Hospital affiliated to Shanghai Jiaotong University were studied. These cases included 42 men and 18 women, with a mean age of 37.2 ± 7.1 years (range, 19-65 years). All the serum samples were negative for both HBsAg and anti-HCV. The patients with non A-E hepatitis were defined as negative control for known serologic markers, including IgM anti-HAV, IgM antibody to hepatitis B core antigen (anti-HBc), hepatitis B surface antigen (HBsAg), HBV DNA, and antibodies to HCV, HDV and HEV. The patients had no history of HAV, HDV, HEV, EBV, CMV infection, or of adiposis hepatica and hepatic lesions induced by drugs, alcohol, cholestatic and autoimmunity, or of transfusion.
The primers were designed according to the sequences submitted to GenBank with computer analysis to determine the outer primer by inner primer. All primers were synthesized by Shanghai Sangon Biological Engineering Technology & Services Co. Ltd. The sequences of primers are as follows (W = A or T, Y = C or T, M = A or C). SENV common primers: P1, 5'-TWCYCMAACGACCAGCTAGACCT-3'; P2, 5'-GTTTGTGGTGAGCAGAACGGA-3'. SENV-D primers: P3, 5'-CTAAGCAGCCCTAACACTCATCCAG-3'; P4, 5'-GCAGTTGACCGCAAAGTTACAAGAG-3'. SENV-H primers: P5, 5'-TTTGGCTGCACCTTCTGGTT-3'; P6, 5'-AGAAATGATGGGTGAGTGTTAGGG-3'.
All blood samples were separated by centrifugation. The sera were stored at -80°C until SENV DNA analysis. Viral DNA was extracted from 200 &mgr;L serum with the QIAamp DNA blood mini kit (Qiagen) and resuspended in 100 &mgr;L elution buffer according to the manufacturer’s instructions.
PCR mixture of 50 &mgr;L contained 0.5 &mgr;L (50 pmol/L) sense primer SENV-P2, 0.5 &mgr;L (50 pmol/L) antisense primer P1, 250 &mgr;mol/L of each dNTP, 6 &mgr;L DNA sample, and 2.5 U Taq DNA polymerase (TaKaRa). The reactions consisted of preheating at 94°C for 4 min, 35 cycles of denaturation at 94°C for 40 s, annealing at 55°C for 50 s, extension at 72°C for 50 s, and a final at 72°C for 10 min.
The second PCR step was carried out with 50 &mgr;L PCR reaction mixture containing 10 &mgr;L of the first-step amplification product. The same PCR buffer was used for the first PCR step, 0.5 &mgr;L (50 pmol/L) sense primer P4 and antisense primer P3 for SENV-D, 0.5 &mgr;L (50 pmol/L) sense primer P6 and antisense primer P5 for SENV-H, 250 &mgr;mol/L of each dNTP, and 2.5 U Taq DNA polymerase. PCR consisted of preheating at 94°C for 30 s, annealing at 55°C for 50 s, extension at 72°C for 50 s, and a final incubation at 72°C for 10 min.
PCR amplified products were separated in 10 &mgr;L reaction mixture by electrophoresis on a 1.5% agarose gel, stained with ethidium bromide, and visualized using an ultraviolet transilluminator. Amplicons containing poly A tails and producing visible bands on agarose gel were excised from the gel and ligated to the pMD18-T vector (TaKaRa). DNA extracted from transformed Escherichia Coli was sequenced in Shanghai Sangon Biological Engineering Technology & Services Co, Ltd. The sequences excluding primer sequences were aligned with Cluster W to A-H SENV genotypes. The genotypes of SENV were determined by the phylogenetic trees.
Descriptive statistical data such as means and proportions were calculated. Frequency was compared between groups using the chi-square test or Fisher’s exact test, and group means were compared using Student’s t test. Stepwise logistic regression method was used to analyze the data. P < 0.05 was considered statistically significant.
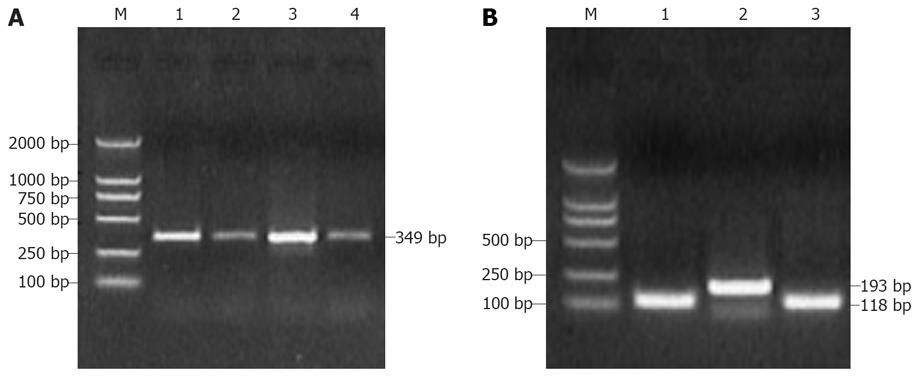
The nPCR results showed that 3 (10%) of the 30 volunteer blood donors and 16 (53.3%) of 30 patients with non A-E hepatitis were positive for SENV. Of 3 volunteer blood donors, 1 was positive for SENV-D and 2 were positive for SENV-H. Of the 16 patients with non A-E hepatitis, 6 were positive for SENV-D, 8 were positive for SENV-H, 2 were positive for both SENV-D and SENV-H DNA (SENV-D/H co-infection). The overall prevalence of SENV-D/H was 30%. After electrophoresis on 1.5% agarose gel stained with ethidium bromide on DNA product, the expected 349 bp, 118 bp and 193 bp bands were visualized on an UV transilluminator for SENV and SENV-D/-H, respectively (Figure 1). The positive rate of SENV infection was significantly higher in patients with non A-E hepatitis than in volunteer blood donors (P = 0.0002).
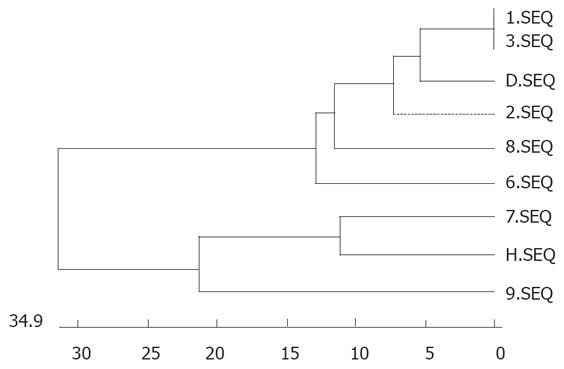
Randomly selected PCR products of P3 and P4 (118 bp) or P5 and P6 (193 bp) were sequenced. When the sequences of PCR positive products were compared with those of SENV by homology analysis, the homology was 98% between P3 and P4 amplification sequences of SENV-D,likewise, the homology was 98% between P5 and P6 amplification sequences of SENV-H. Some sequences of PCR positive products are shown in Figure 2. The phylogenic tree for certain sequences of PCR products from patients with non A-E hepatitis and SENV-D/ H subtype genes from the GenBank (http://www.ncbi.nlm.nih.gov/BLAST/) are shown in Figure 3.
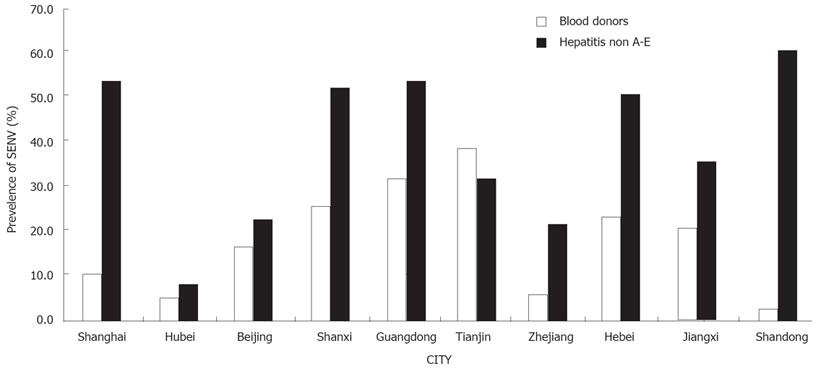
SENV infection in different regions of China has been reported elsewhere[8–13]. The detected population was concentrated in areas of high CLD infection and the prevalence of SENV was different in these regions of China (Figure 4).
Hepatitis viruses can be divided into five types: A-E and cause 80%-90% hepatitis cases. Some viruses such as TTV are correlated with non A-E hepatitis[14–16]. However, no pathogenicity of TTV has been found in different populations[17–20].
SENV has been recently identified as a candidate agent of non A-E hepatitis virus[21]. However, the exact role of this virus in the pathogenesis of chronic liver diseases, including chronic hepatitis and cirrhosis, and the development of hepatocellular carcinoma (HCC) remains to not be verified[22]. The prevalence of transfusion-associated non A-E hepatitis is high and the SENV-D and SENV-H strains are significantly associated with transfusion-associated non A-E hepatitis.
Pirovano et al[23] found that aminotransferase level is transitly elevated in infants with positive SENV. Umemura et al[24] showed that SENV-D/H infection is related with posttransfusion hepatitis and special RNA molecule of SENV in which non A-E hepatic tissue probably is an intermedium of SENV replication. Recent data indicate that SENV DNA can be found in hepatic tissue from acute non A-E hepatitis patients and SENV perhaps causes hepatic lesions induced by in situ PCR. In addition, the detectable SENV DNA rate is 21% in patients with chronic hepatitis C[25–27]. In this study, the detectable SENV DNA rate was 10% (3/30) in 30 volunteer blood donors and 53.3% (16/30) in 30 patients with non A-E hepatitis.
In the present study, SENV infection was found in blood donors and patients with non A-E hepatitis in Shanghai. We found that approximately 3.2%-38.9% of blood donors and 9.3%-59.6% of non A-E hepatitis had positive SENV (Figure 4) and the positive SENV DNA rate was significantly different in different areas, suggesting that we must continue the detection of SENV DNA prevalence in other provinces. Interestingly, the prevalence rate of SENV was higher in blood donors than in patients with non A-E hepatitis (38.9% vs 32.3%) in Tianjin.
Furthermore, SENV-H was found in 2 volunteer blood donors and SENV-D was found in 1 volunteer blood donor infected with SENV. SENV-H, SENV-D and mixed SENV-D/H were found in 8, 6, and 2 patients with hepatitis, indicating that the prevalence of SENV in Shanghai is relatively low. However, SENV infection was not associated with blood transfusion in 16 SENV positive non A-E hepatitis patients with no blood-transfusion history, indicating that blood transfusion transmission is not the only way for people to infect with SENV. Because of the limited number of serum samples in our study, we could not explain why SENV infection is related with blood transfusion or non A-E hepatitis.
In conclusion, SENV infection is higher in patients with non A-E hepatitis than in blood donors and the homology is 98%. Further study with a large number of samples is required to analyze the relationship between SENV infection and non A-E hepatitis.
SEN virus (SENV) is described as a small single-stranded, non-enveloped circular DNA virus, possibly belonging to the circoviridae family. To date, eight distinct strains of SENV (A-H) have been identified. The prevalence of these eight different strains SENV (A-H) is different in each infected group. In the present study, the prevalence of SENV-D or SENV–H strain was 2.25% and 92.31% in blood donors and patients with acute or chronic non A-E hepatitis, suggesting that SENV-D or SENV-H is significantly associated with the pathogenesis of non A-E hepatitis.
The prevalence of SENV in patients with various forms of liver disease has been reported in many countries and several regions of China, but the results are not consistent. The role of SENV infection in patients with non A-E hepatitis or other viral hepatitis and the transmitting form of SENV are not very clear. The clinical significance of SENV infection in patients with non A-E hepatitis is also not very clear.
SENV infection was studied in blood donors and patients with non A-E hepatitis in Shanghai. The results indicate that the prevalence of SENV in blood donors of Shanghai was relatively lower than that in other regions of China. SENV infection was not associated with blood transfusion in SENV positive patients with non A-E hepatitis with no blood-transfusion history, suggesting that blood transfusion transmission is not the only way to spread SENV.
The SENV infection rate is higher in non A-E hepatitis than in blood donors in Shanghai. Because of the limited number of serum samples in our study, we could not explain why SENV infection is associated with blood transfusion or non A-E hepatitis. Further study with a large number of samples is required to analyze the relationship between SENV infection and non A-E hepatitis.
This paper describes the prevalence of SEN virus in blood donors and hepatitis patients. The study is well designed and provides important information about the prevalence of SENV in hepatitis patients.
| 1. | Primi D, Fiordalisi G, Mantero GL; inventors. Diasorin, SRL, assignee. Identification of SENV genotypes. International patent number WO0028039 (international application published under the patent cooperation treaty; http://ep.espacenet.com/. May 18, 2000). . |
| 2. | Bowden S. New hepatitis viruses: contenders and pretenders. J Gastroenterol Hepatol. 2001;16:124-131. |
| 3. | Tanaka Y, Primi D, Wang RY, Umemura T, Yeo AE, Mizokami M, Alter HJ, Shih JW. Genomic and molecular evolutionary analysis of a newly identified infectious agent (SEN virus) and its relationship to the TT virus family. J Infect Dis. 2001;183:359-367. |
| 4. | Moriondo M, Resti M, Betti L, Indolfi G, Poggi GM, de Martino M, Vierucci A, Azzari C. SEN virus co-infection among HCV-RNA-positive mothers, risk of transmission to the offspring and outcome of child infection during a 1-year follow-up. J Viral Hepat. 2007;14:355-359. |
| 5. | Wang LY, Ho TY, Chen MC, Yi CS, Hu CT, Lin HH. Prevalence and determinants of SENV viremia among adolescents in an endemic area of chronic liver diseases. J Gastroenterol Hepatol. 2007;22:171-176. |
| 6. | Spataro P, Di Pietro A, Scoglio ME, Visalli G, Chirico C, Picerno I, Ferlazzo N, Campo S, Bellinghieri G, Savica V. Prevalence of SENV-H and SENV-D virus: epidemiological study in blood donors and dialysis patients. Ren Fail. 2006;28:441-448. |
| 7. | Dai CY, Chuang WL, Chang WY, Chen SC, Lee LP, Hsieh MY, Hou NJ, Lin ZY, Hsieh MY, Wang LY. Co-infection of SENV-D among chronic hepatitis C patients treated with combination therapy with high-dose interferon-alfa and ribavirin. World J Gastroenterol. 2005;11:4241-4245. |
| 8. | Zhu F, Cai XD, Qian JM, Fei GJ, Yang XM. The Relationship Between SEN Virus Infection and Chronic Liver Disease. J Clin digest DIS. 2004;16:3-5. |
| 9. | Gao YT, Chen RY, Song WQ, Qi ZL, Jing L, Qian SC. Prevalence of SENV in different groups. Chin J Infect Dis. 2004;22:206-208. |
| 10. | Yi DY, He B, Zhang LL, Chen SB, Huang JS, Wu XP, Zhao ZY. The Infection of SEN Virus (SENV) in Blood of Donators in Jiangxi Province. Acta Academiae Medicine Jiangxi. 2006;46:16-17. |
| 11. | Yu JG, Shang QH, Sun SC, An Y, Xu CZ, Pan W, Zhang YQ. Qi ZT, Zhang GS. Infective Condition of SEN Virus(SENV) in Different Groups of People in Shandong. Clin J Med Offic. 2004;32:85-87. |
| 12. | Tian DY, Xu D, Zhang ZG, SONG PH. Prevalence of SEN Virus Infection in Wuhan Area. Chin J Nosocomiol. 2003;13:913-915. |
| 13. | Mu SJ, Chen R, Li D, Zang XQ. Prevalence of SEN Virus D and H Subtype Infection in Different Type of Hepatitis and Blood Donors. J Mod Lab Med. 2006;21:10-12. |
| 14. | Emre S, Otlu B, Cankaya C, Doganay S, Durmaz R. Transfusion-transmitted virus DNA in serum, tear and aqueous humour of patients undergoing cataract operation. Clin Experiment Ophthalmol. 2007;35:759-762. |
| 15. | Hsu BG, Wang LY, Hu CT, Wang CH, Fang TC, Lin HH. TT virus infection in patients on peritoneal dialysis in Taiwan. Ren Fail. 2007;29:553-557. |
| 16. | Tomasiewicz K, Modrzewska R, Lyczak A, Krawczuk G. TT virus infection and pancreatic cancer: relationship or accidental coexistence. World J Gastroenterol. 2005;11:2847-2849. |
| 17. | Irshad M, Sharma Y, Dhar I, Singh J, Joshi YK. Transfusion-transmitted virus in association with hepatitis A-E viral infections in various forms of liver diseases in India. World J Gastroenterol. 2006;12:2432-2436. |
| 18. | Zhang JH, Chen SZ. An epidemiological study on transfusion transmitted virus infection in China. Shijie Huaren Xiaohua Zazhi. 2001;9:1384-1390. |
| 19. | Zhao Y, Wang JB. Epidemiological survey on TTV infection and the asso ciation with other viral liver diseases in Changchun area. Shijie Huaren Xiaohua Zazhi. 2001;9:747-750. |
| 20. | Fu EQ, Bai XF, Pan L, Li GY, Yang WS, Tang YM, Wang PZ, Sun JF. Investigation of TT virus infection in groups of different people in Xi’an. Shijie Huaren Xiaohua Zazhi. 1999;7:967-969. |
| 21. | Umemura T, Yeo AE, Sottini A, Moratto D, Tanaka Y, Wang RY, Shih JW, Donahue P, Primi D, Alter HJ. SEN virus infection and its relationship to transfusion-associated hepatitis. Hepatology. 2001;33:1303-1311. |
| 22. | Schruter M, Laufs R, Zollner B, Knodler B, Schafer P, Sterneck M, Fischer L, Feucht HH. Prevalence of SENV-H viraemia among healthy subjects and individuals at risk for parenterally transmitted diseases in Germany. J Viral Hepat. 2002;9:455-459. |
| 23. | Pirovano S, Bellinzoni M, Ballerini C, Cariani E, Duse M, Albertini A, Imberti L. Transmission of SEN virus from mothers to their babies. J Med Virol. 2002;66:421-427. |
| 24. | Umemura T, Tanaka E, Ostapowicz G, Brown KE, Heringlake S, Tassopoulos NC, Wang RY, Yeo AE, Shih JW, Orii K. Investigation of SEN virus infection in patients with cryptogenic acute liver failure, hepatitis-associated aplastic anemia, or acute and chronic non-A-E hepatitis. J Infect Dis. 2003;188:1545-1552. |
| 25. | Kojima H, Kaita KD, Zhang M, Giulivi A, Minuk GY. Genomic analysis of a recently identified virus (SEN virus) and genotypes D and H by polymerase chain reaction. Antiviral Res. 2003;60:27-33. |
| 26. | Lin JG, Goto T, Nakane K, Miura K, Mikami K, Ohshima S, Yoneyama K, Watanabe S. Clinical significance of SEN-virus on interferon response in chronic hepatitis C patients. J Gastroenterol Hepatol. 2003;18:1144-1149. |
| 27. | Schroter M, Laufs R, Zollner B, Knodler B, Schafer P, Feucht HH. A novel DNA virus (SEN) among patients on maintenance hemodialysis: prevalence and clinical importance. J Clin Virol. 2003;27:69-73. |