Published online May 7, 2008. doi: 10.3748/wjg.14.2767
Revised: March 9, 2008
Published online: May 7, 2008
AIM: To find the precautions against the safety in caudate lobe resection.
METHODS: The clinical data obtained from 11 cases of primary liver cancer in caudate lobe who received hepatectomy successfully were retrospectively analyzed. Four safe procedures were used in resection of primary liver cancer in caudate lobe: (1) selection of appropriate skin incision to obtain excellent exposure of operative field; (2) adequate mobilization of the liver to allow the liver to be displaced upwards to the left or to the right; (3) preparatory placement of tapes for total hepatic vascular exclusion, so that this procedure could be used to control the fatal bleeding of the liver when necessary; (4) selection of the ideal route for hepatectomy based on the condition of the tumor and the combined removal of multiple lobes if necessary. Among the 11 cases, simple occlusion of vessels of porta hepatis was used in caudate lobectomy for 6 cases, while in the other cases, the vessels were intermittently occluded several times or total hepatic vascular isolation was used in the caudate lobectomy. Combined partial right hepatectomy was done for 3 cases, combined left lateral lobectomy for 2 cases and caudate lobectomy alone for 6 cases.
RESULTS: Operation was successful for all the 11 cases. Intermittent inflow occlusion was performed for all patients for 15 min at 5-min intervals. Blockade was performed twice in 3 patients and total hepatic vascular exclusion was performed in one of the three patients. Blockade was performed three times in one patient, including a total hepatic vascular exclusion. Total hepatic vascular exclusion was performed only in one patient. The mean blood loss was 300 mL. Ascites and pleural effusion occurred in 4 patients, jaundice in 1 patient. Six patients died of tumor recurrence in 6, 11, 12, 13, 15, 19 mo after operation, respectively. The other 5 patients have survived more than 16 mo since the operation.
CONCLUSION: Caudate lobectomy for liver cancer in candidate lobe can be safely performed with the above procedures.
- Citation: Wen ZQ, Yan YQ, Yang JM, Wu MC. Precautions in caudate lobe resection: Report of 11 cases. World J Gastroenterol 2008; 14(17): 2767-2770
- URL: https://www.wjgnet.com/1007-9327/full/v14/i17/2767.htm
- DOI: https://dx.doi.org/10.3748/wjg.14.2767
Caudate lobe is the first segment of liver in the Couinaud’s classification[1–3], and because of its unique anatomical site, its resection has a high risk[4–7]. If appropriate surgical procedures are used, it can be safely and successfully resected[8].
From 2003 to 2005, four safe procedures were used in resection of primary liver cancer in caudate lobe. Eleven cases of primary liver cancer in caudate lobe received resection successfully at the First Department of Hepatic Surgery of the Eastern Hepatobiliary Surgery Hospital, Second Military Medical University. Among the 11 cases, 9 were males and 2 females (age range, 32-65 years, and mean age 53 years). All the patients were diagnosed with primary liver cancer. Nine patients were positive for hepatitis B surface antigen and their liver cirrhosis was confirmed by histologic examination of the resected specimens. The primary liver cancer was originated from the caudate lobe in 9 cases and from the adjacent lobe in 2 cases. Three cases received liver lobe resection ago. Nine cases were diagnosed with hepatocellular carcinoma and 2 cases with biliary cell carcinoma.
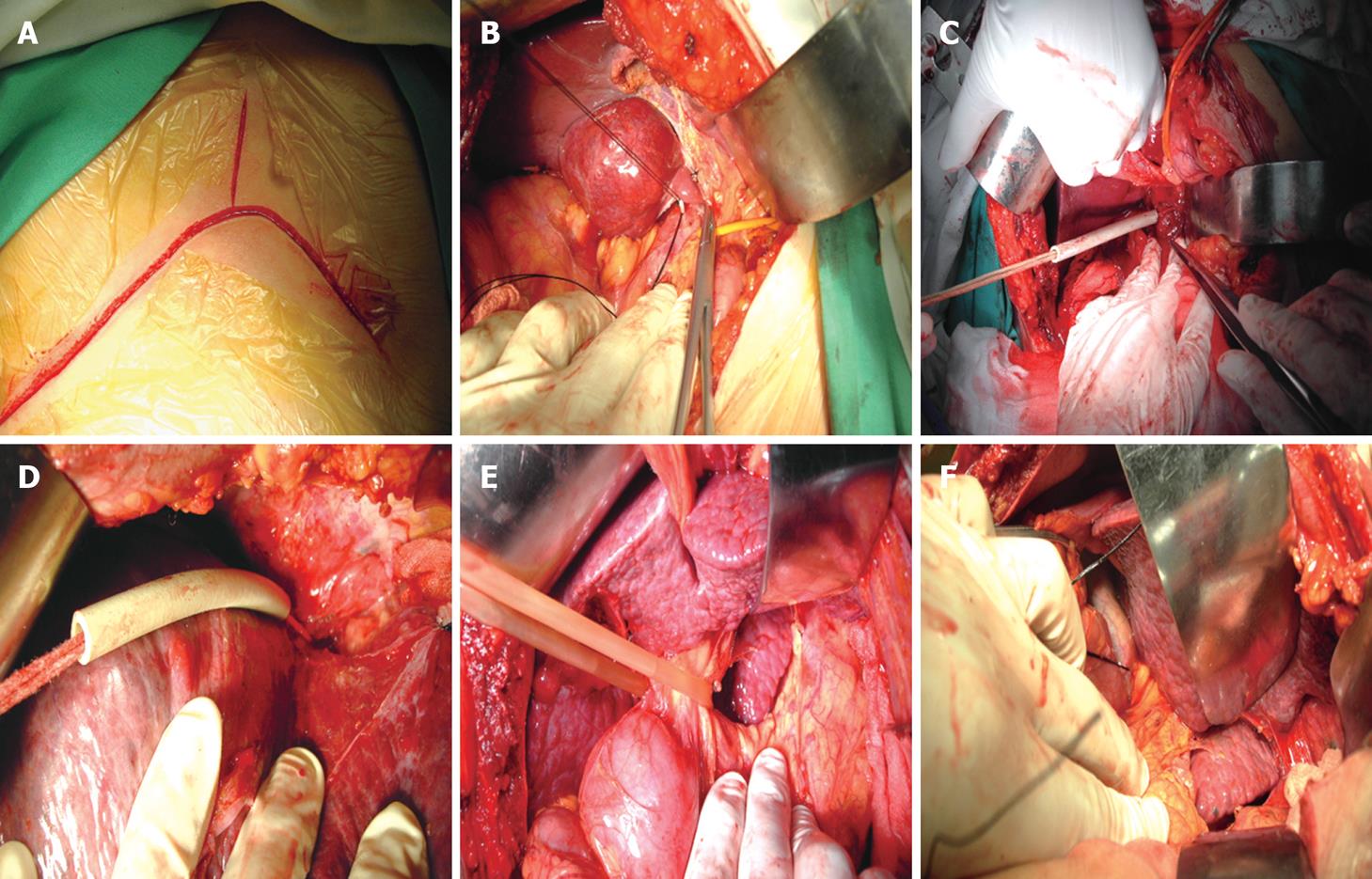
Skin incision: A reversed T-shaped skin incision was made (bilateral oblique incision under costal arch) (Figure 1A). The costal arch was hauled and secured with a special liver hook. The whole abdominal cavity was explored to rule out intra-abdominal metastasis.
Dividing ligaments: The falciform ligament was divided up to the front of the suprahepatic inferior vena cava (IVC), then the coronary, triangular, hepatorenal and hepatogastric ligaments were cut off, making the whole liver moveable.
Dissecting blood vessels of caudate lobe: The liver was pulled upward to exposure the infrahepatic IVC. The short hepatic veins of the caudate lobe were dissected and ligated (Figure 1B). Three thin and short hepatic veins were divided. The caudate portal triads were ligated and divided. Thus, the caudate lobe was isolated completely. The caudate lobe was detached from the neighboring liver parenchyma where large branches must be ligated if there are any.
Preparing tapes: As a safety precaution, tapes were preplaced around the infrahepatic and suprahepatic IVC (total hepatic vascular exclusion) (Figure 1C and D).
Pringle maneuver: The resection was begun with the occlusion of the first hepatic hilum (Pringle maneuver). The hepatic hilum was pulled aside to obtain the excellent exposure of caudate lobe (Figure 1E). Total vascular exclusion was used only when the patient had laceration of the infra vena cava or liver vein. Such a maneuver could achieve hemodynamic instability.
Resection: In our series, the left lobe was removed in 9 patients and the liver was inversed without removing the left lobe in 9 patients (Figure 1F), the right lobe was removed in 2 patients. The anterior entry approach was not used in any patients. The caudate lobe was resected in 6 patients. However, additional right hemihepatectomy was performed for 3 patients and left hemihepatectomy for 2 patients. The left lateral lobe or the right lateral lobe was resected to release the space, and then the caudate lobe was resected.
Intermittent inflow occlusion at the hepatoduodenal ligament was performed for all patients for 15 min at 5-min intervals. Six patients finished the operation. Blockade was performed twice in 3 patients and total hepatic vascular exclusion was performed in one of the three patients. Blockade was performed three times in one patient, including a total hepatic vascular exclusion. Total hepatic vascular exclusion was performed only in one patient. Among the patients undergoing total hepatic vascular exclusion, the infra vena cava was repaired in one patient, the liver vein was repaired in one patient, and the caudate lobe was resected with the right suprarenal gland in one patient.
It was reported that there are more intraoperative blood loss, longer operation time and more postoperative complications in resection of the caudate lobe than for other lobes[910]. In our study, blood loss varied from 100 to 800 mL. The mean blood loss was 300 mL. Ascites and pleural effusion occurred in 4 patients, jaundice in 1 patient. Two patients with severe cirrhosis were cured and drainage occurred in the right thoracic cavity of 2 patients with hydrothorax.
Intrahepatic recurrence was noted in 5 patients and lung metastasis was found in one patient. The 6 patients died of tumor recurrence 6, 11, 12, 13, 15, 19 mo after operation, respectively. The other 5 patients have survived more than 16 mo since the operation.
The first segment of caudate lobe is divided into three sub-segments: Spiegel lobe, paracaval portion, and caudate process[1112]. The Spiegel lobe is located behind the lesser omentum, just to the left of intrahepatic IVC. The paracaval portion is in front of the intrahepatic IVC, just to the right of the Spiegel lobe, and is closely attached to the right and middle hepatic veins. The caudate process is a tongue-like projection between the IVC and the adjacent portal vein, just to the right of the paracaval portion.
There are three porta hepatis in a liver[1314]. The first porta hepatis denotes the hilum in a general sense; the second porta hepatis denotes the confluence of major hepatic veins, and the third porta hepatis denotes the segment of retrohepatic IVC with a series of short hepatic veins. Caudate lobe is, thus, surrounded by the three porta hepatis, all of which consist of important and potentially dangerous structures in terms of performing operation[1516]. In view of the unique anatomical location, caudate lobe resection is technically challenging, especially for isolated caudactomy because it is easy to damage the hilum and bile duct in dissecting the anterior of caudate lobe, the inferior vena cava and cause uncontrolled bleeding when the posterior of caudate lobe is dissected as well as most difficult to remove the tumor near the protruded portion or inferior vena cava portion.
Carcinoma, originating from the caudate lobe, cannot be treated effectively with trans-arterial chemotherapy embolization (TACE)[1718], because the supply vessels of caudate lobe are different from those of other lobes. Caudate lobe has many supply blood vessels from the right and left portal veins and hepatic arteries, many veins outflow the caudate lobe into the inferior vena cava and hepatic veins. It is not easy to completely embolize so many arteries. Percutaneous ethanol injection therapy (PEIT)[19] and percutaneous radiofrequency ablation (PRFA)[20] are also limited in the treat ment of caudate lobe tumor, because injection or penetration can easily damage the adjacent blood vessels or other important structures. Therefore, caudate lobectomy may be the only effective method to cure the cancer.
Surgical approach to caudate lobe: The choice of procedures is essential to the success of caudate lobectomy[21]. There are different procedures of caudate lobectomy, but only three are effective. (1) Anterior procedure: resection of the hepatic parenchyma from the middle line with the right and left porta hepatis pulled aside to expose the caudate lobe. This method is complicated and causes huge injuries. We do not consider anterior procedure in caudate lobectomy resection. (2) Right procedure: resection of the 6 segments to expose the caudate lobe. This procedure resects more normal tissues. It is thus, not suitable for cirrhosis. If partial right lobe is not resected, it is difficult to dissect the protruded right portion of caudate lobe. (3) Left procedure: if the carcinoma is close to the left and upside of the lobe, resection of the left lateral lobe can expose the caudate lobe. It is better not to dissect the left lateral lobe. Otherwise, it causes liver failure if cirrhosis is severe. After the left lateral lobe is removed and the liver is inversed to the right and upwards, it is easy to perform the procedure. This procedure is commonly used to resect the caudate lobe.
Preparation of infra vena cava occlusion belt: Prepa-ration of infra vena cava belt is to block intraoperative vital bleeding caused by laceration of the infra vena cava and hepatic vein[22]. If bleeding occurs, the mortality would be high. We think that infra vena cava block belt should be prepared for all patients[23]. In practice, only few patients need infra vena cava block[24]. We stress that infra vena cava block belt secures the safety of operation. When total hepatic vascular exclusion is used, the block time should be controlled. The authors think that the preferable block time is less than 15 min in order to reduce complications after operation, because patients with liver cirrhosis have insufficient liver function.
Repair of laceration of infra vena cava or hepatic vein: Once much blood emerges through the wound, laceration of the infra vena cava is diagnosed[25]. We should make a prompt decision without any hesitation to perform total hepatic vascular exclusion, in order to remove the tumor quickly and repair the laceration. However, if the laceration is small and can be located, it is not necessary to perform total hepatic vascular exclusion. We can use the finger press method to stanch bleeding and repair the vessels quickly[26].
Primary liver cancer originating from the caudate lobe is not rare. It is difficult to remove it because of its unique anatomic location. We presented a safe technique of caudate lobectomy for liver cancer.
Caudate lobectomy can be safely performed when appropriate skin incision is made, liver ligaments are adequately cut off, tapes for total hepatic vascular exclusion are placed and the ideal section route is selected.
This is an interesting paper describing caudate lobe resection and the precautions against the safety in caudate lobe resection. The authors suggest that caudate lobectomy for liver cancer in caudate lobe can be safely performed when proper procedures are used.
| 1. | Wu MC. Liver surgery. Shanghai: Scientific and technical documents publishing house 2000; 14-15. |
| 2. | Niu CX, Li CL, Yang Y. Applied anatomy of the hepatic caudate lobectomy. Zhongguo Linchuang Jiepouxue Zazhi. 2001;19:151-154. |
| 3. | Couinaud C. Surgical Anatomy of the Liver Revisited. Paris, France: Couinaud 1989; 123-134. |
| 4. | Fan J, Wu ZQ, Tang ZY, Zhou J, Qiu SJ, Ma ZC, Zhou XD, Yu YQ. Complete resection of the caudate lobe of the liver with tumor: technique and experience. Hepatogastroenterology. 2001;48:808-811. |
| 5. | Pol B, Campan P, Hardwigsen J, Botti G, Pons J, Le Treut YP. Morbidity of major hepatic resections: a 100-case prospective study. Eur J Surg. 1999;165:446-453. |
| 6. | Hu JX, Miao XY, Zhong DW, Dai WD, Liu W. Anterior approach for complete isolated caudate lobectomy. Hepatogastroenterology. 2005;52:1641-1644. |
| 7. | Shimada M, Matsumata T, Maeda T, Yanaga K, Taketomi A, Sugimachi K. Characteristics of hepatocellular carcinoma originating in the caudate lobe. Hepatology. 1994;19:911-915. |
| 8. | Fukada T, Kimura F, Takayasiki T, Ito H, Shimizu H, Togawa A, Ohtsuka M, Yoshidome H, Kato A, Miyazaki M. Anterior transhepatic approach for hepatocellular carcinoma located in deep positions of segment VIII. Hepatogastroenterology. 2007;54:536-538. |
| 9. | Tanaka S, Shimada M, Shirabe K, Maehara S, Tsujita E, Taketomi A, Maehara Y. Surgical outcome of patients with hepatocellular carcinoma originating in the caudate lobe. Am J Surg. 2005;190:451-455. |
| 10. | Li AJ, Zhou WP, Wu MC, Luo XJ. Hepatectomy after primary repair of ruptured liver cancer. Hepatobiliary Pancreat Dis Int. 2007;6:267-270. |
| 11. | Bartlett D, Fong Y, Blumgart LH. Complete resection of the caudate lobe of the liver: technique and results. Br J Surg. 1996;83:1076-1081. |
| 12. | Kumon M. Anatomy of the caudate lobe with special reference to the portal vein and bile duct. Acta Hepatol Jpn. 1985;26:1193-1199. |
| 13. | Shibata T, Maetani Y, Ametani F, Kubo T, Itoh K, Konishi J. Efficacy of nonsurgical treatments for hepatocellular carcinoma in the caudate lobe. Cardiovasc Intervent Radiol. 2002;25:186-192. |
| 14. | Wang Y, Chen Han, Sun YF, Wei GT, Wu MC. Discussion of the surgical method of hepatic caudate lobe. Zhongguo Putong Waike Zazhi. 2005;20:36-40. |
| 15. | Xing X, Li H, Liu WG. Clinical studies on inferior right hepatic veins. Hepatobiliary Pancreat Dis Int. 2007;6:579-584. |
| 16. | Peng SY, Liu YB, Xu B, Cai XJ. Role and significance of extrahepatic control of hepatic vein and inferior vena cava in difficult hepatectomies for patients with liver tumor (in Chinese). Zhonghua Waike Zazhi. 2004;42:260-264. |
| 17. | Shibata T, Maetani Y, Ametani F, Kubo T, Itoh K, Konishi J. Efficacy of nonsurgical treatments for hepatocellular carcinoma in the caudate lobe. Cardiovasc Intervent Radiol. 2002;25:186-192. |
| 18. | Yamamoto T, Hirohashi K, Kubo S, Uenishi T, Ogawa M, Hai S, Sakabe K, Tanaka S, Shuto T, Tanaka H. Hepatectomy with transcatheter arterial embolization for large hepatoma in the caudate lobe. Hepatogastroenterology. 2003;50:2173-2175. |
| 19. | Matsumoto T, Iwaki K, Hagino Y, Kawano K, Kitano S, Tomonari K, Matsumoto S, Mori H. Ethanol injection therapy of an isolated bile duct associated with a biliary-cutaneous fistula. J Gastroenterol Hepatol. 2002;17:807-810. |
| 20. | Yamakado K, Nakatsuka A, Akeboshi M, Takaki H, Takeda K. Percutaneous radiofrequency ablation for the treatment of liver neoplasms in the caudate lobe left of the vena cava: electrode placement through the left lobe of the liver under CT-fluoroscopic guidance. Cardiovasc Intervent Radiol. 2005;28:638-640. |
| 21. | Peng SY, Li JT, Mou YP, Liu YB, Wu YL, Fang HQ, Cao LP, Chen L, Cai XJ, Peng CH. Different approaches to caudate lobectomy with “curettage and aspiration” technique using a special instrument PMOD: a report of 76 cases. World J Gastroenterol. 2003;9:2169-2173. |
| 22. | Smyrniotis V, Farantos C, Kostopanagiotou G, Arkadopoulos N. Vascular control during hepatectomy: review of methods and results. World J Surg. 2005;29:1384-1396. |
| 23. | Dixon E, Vollmer CM Jr, Bathe OF, Sutherland F. Vascular occlusion to decrease blood loss during hepatic resection. Am J Surg. 2005;190:75-86. |
| 24. | Zhou WP, Wu MC, Chen Han, Yao XP, Yang GS, Wu BW. Resection of the hepatic caudate lobe. Zhonghua Gandan Waike Zazhi. 2001;7:43-44. |
| 25. | Chao J, Tang WM, Ou QJ. Surgical management of posterohepatic vena cava injury during hepatectomy. Zhonghua Gandan Waike Zazhi. 2002;8:352-353. |
| 26. | Li A, Wu M, Yang G, Chen H, Shen F. [Management of retrohepatic inferior vena cava injuries in hepatectomy for neoplasm]. Zhonghua Waike Zazhi. 1999;37:14-17. |