Published online Dec 14, 2007. doi: 10.3748/wjg.v13.i46.6140
Revised: September 25, 2007
Accepted: October 26, 2007
Published online: December 14, 2007
Capsule endoscopy has revoluzionized the study of the small bowel by providing a reliable method to evaluate, endoscopically, the entire small bowel. In the last six years several papers have been published exploring the possible role of this examination in different clinical conditions. At the present time capsule endoscopy is generally recommended as a third examination, after negative bidirectional endoscopy, in patients with obscure gastrointestinal bleeding. A growing body of evidence suggests also an important role for this examination in other clinical conditions such as Crohn’s disease, celiac disease, small bowel polyposis syndromes or small bowel tumors. The main complication of this examination is the retention of the device at the site of a previously unknown small bowel stricture. However there are also some other open issues mainly due to technical limitations of this tool (which is not driven from remote control, is unable to take biopsies, to insufflate air, to suck fluids or debris and sometimes to correctly size and locate lesions).The recently developed double balloon enteroscope, owing to its capability to explore a large part of the small bowel and to take targeted biopsies, although being invasive and time consuming, can overcome some limitations of capsule endoscopy. At the present time, in the majority of clinical conditions (i.e. obscure GI bleeding), the winning strategy seems to be to couple these two techniques to explore the small bowel in a painless, safe and complete way (with capsule endoscopy) and to define and treat the lesions identified (with double balloon enteroscopy).
- Citation: Rondonotti E, Villa F, Mulder CJ, Jacobs MA, Franchis R. Small bowel capsule endoscopy in 2007: Indications, risks and limitations. World J Gastroenterol 2007; 13(46): 6140-6149
- URL: https://www.wjgnet.com/1007-9327/full/v13/i46/6140.htm
- DOI: https://dx.doi.org/10.3748/wjg.v13.i46.6140
The small bowel (SB) has been considered for a long time technically difficult to evaluate for many anatomical (i.e. distance from external orifices, length) and physiological (i.e. active peristalsis) reasons.
Until the introduction of Video Capsule Endoscopy (VCE) in clinical practice the small bowel was studied mostly with radiological or nuclear medicine techniques such as abdominal Computed Tomography (abdominal CT), abdominal Magnetic Resonance Imaging (abdominal MRI), small bowel follow through (SBFT), small bowel enteroclysis (SB enteroclysis) and 99mTC scan. Although CT scan and abdominal MRI are highly sensitive in recognizing the presence of abdominal masses and allow an accurate evaluation of solid organs, lymph nodes and vessels, they are able to provide limited information about the small bowel wall. On the other hand, small bowel follow-through and small bowel enteroclysis, although specifically designed to evaluate the small bowel, have low sensitivity and specificity in recognizing small and flat lesions[1].
Additionally these two techniques are often poorly tolerated by patients and sometimes difficult to interpret.
The endoscopic evaluation of the small bowel represents the best possible approach to small intestinal diseases, allowing a direct visualization of small bowel mucosa, the collection of targeted biopsies and sometimes an effective treatment. Sonde enteroscopy, introduced because of its theoretical capability to visualize the entire small bowel (achievable in about 80% of examinations in clinical practice)[2,3], had been abandoned at the end of the 90’s because of several technical limitations (angulation of the tip due to the presence of the balloon, duration of the examination, patient discomfort, inability to take biopsies)[4]. Push Enteroscopy (PE) is limited by the depth of insertion of the instrument to the proximal jejunum (about 90-150 cm from the oral route) and to the terminal ileum (50-80 cm in the retrograde way) and, despite sedation, is still poorly tolerated[5-7]. Intraoperative enteroscopy (IOE) is the most complete but also the most invasive means of examining the small bowel[7].
The introduction (in 2001)[8] and further continuous development of capsule endoscopy opened a new chapter in the study of small bowel diseases allowing, finally, to cross the frontier of the endoscopic examination of the small bowel. In fact this revolutionary technique made it possible, for the first time, to obtain high resolution endoscopic images of the entire small bowel (Figure 1) avoiding sedation, surgical intervention or radiation exposure. Capsule endoscopy showed, in everyday clinical practice, that the small bowel can be involved in several diseases (i.e. inflammatory, vascular, neoplastic, iatrogenic diseases). The knowledge of the large spectrum of lesions and diseases that can affect the small bowel stimulated the development and/or the implementation of other diagnostic and therapeutic techniques such as double balloon enteroscopy (DBE), MRI-enteroclysis and CT enteroclysis.
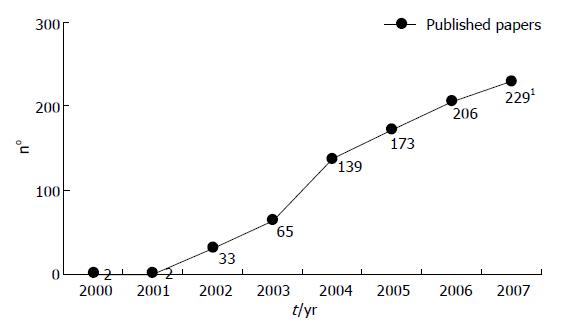
Performing a recursive search in the literature (by means of the most common search engine http://www.pubmed.org; using “capsule endoscopy OR capsule enteroscopy” as key words) we found a number of papers, increasing over the years, up to 754 (Figure 2). On the one hand this phenomenon certainly represents a proof of the revolutionary potential of this diagnostic tool in the field of small bowel endoscopy and, on the other hand, demonstrates the effort to establish the appropriate role of this device in different clinical conditions. Unfortunately about a quarter of published papers are case reports (187) or collections of small case series and 131 published papers are expert reviews. Following the rules of evidence based medicine[9] we can classify these papers at the lowest level of scientific evidence while, among the huge number of publications about capsule endoscopy, there are only 8 randomized controlled studies [five about bowel preparation, 2 about Nonsteroidal anti-inflammatory drugs (NSAIDs) induced damage and 1 about obscure GI bleeding (OGIB)][10-17] and 4 metanalyses (about OGIB or Crohn’s disease)[18-21]; all these papers can be ranked as evidence grade 1c.
Mainly on the ground of the former 12 mentioned papers three Practice Guidelines have been published so far, two (in 2004 and 2006) on behalf of the European Society of Gastrointestinal Endoscopy (ESGE) and one on behalf of the American Society of Gastrointestinal Endoscopy (ASGE)[22-24] in 2006.
The aim of the present paper is to briefly review the evidence, available to date, about the use of capsule endoscopy for the study of the small bowel (starting from the studies providing the highest grade of evidence), to highlight the benefits of this technique but also to highlight risks and limitations which have emerged in these six years of use of the device in clinical practice.
So far, OGIB is the main clinical indication for capsule endoscopy: about 70%-80%[25,26] of patients undergoing capsule endoscopy suffer from OGIB. The majority of studies published at the beginning of the experience with this new tool reported a high, although widely variable, diagnostic yield (ranging between 38% and 93%, about 75%-80% in most studies[27]). These studies, mainly performed in tertiary referral centres, collected highly selected patients with a long standing history of obscure GI bleeding, with low levels of haemoglobin at the time of the examination, who had undergone a huge number of prior examinations with negative results[28]. Subsequent studies performed on larger populations of patients, similar to those undergoing this examination in everyday clinical practice, showed a slightly lower diagnostic yield (about 50%)[29].
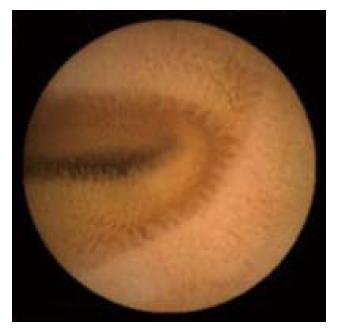
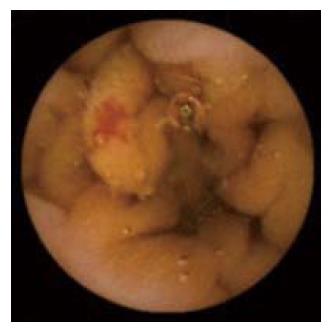
Although recent studies showed a decrease in the diagnostic yield of capsule endoscopy, two metanalyses[18,19] clearly demonstrated that capsule endoscopy in patients with obscure GI bleeding is superior to traditional radiological techniques (SBFT and SB enteroclysis) and PE. The latter comparison has been also recently confirmed in a specific prospective randomized controlled study[11]. The authors hypothesized that the high diagnostic yield of capsule endoscopy in this subgroup of patients may depend on the capability of the capsule to evaluate the mid-distal small bowel (particularly in comparison with PE) and/or to show small and flat lesions (vascular-Figure 3- or inflammatory-Figure 4) that are often missed by conventional radiological techniques.
As far as the possible factors potentially affecting the diagnostic yield of capsule endoscopy are concerned, the presence of active bleeding at the time of examination[30] or a short interval between the last episode of acute bleeding and capsule endoscopy[28-30], low levels of haemoglobin and high transfusion requirement have been found to be associated with a high diagnostic yield[31,32].
Since capsule endoscopy was introduced in clinical practice 6 years ago, some papers explored also the impact of this new technique on the outcome of patients with obscure GI bleeding. As expected, capsule endoscopy has been found to significantly modify the diagnostic and therapeutic work up immediately after the examination[33], decreasing the number of further examinations and reducing the length of hospital stay[10,34,35]. Nevertheless capsule endoscopy seems also to have a positive impact on long term follow up (mostly evaluated at 12-18 mo after the examination) in about 50%-66%[28,32] of patients and even patients with negative capsule endoscopy have a low probability of experiencing a new bleeding episode (the negative predictive value of capsule endoscopy ranges between 83% and 100%)[34,36].
On the ground of this large amount of published papers capsule endoscopy is now proposed by experts, in patients with obscure GI bleeding, as a third step after a negative bidirectional endoscopy[33], and scientific societies[22,23] define capsule endoscopy as a very valuable tool for investigating obscure gastrointestinal bleeding with the potential capability to improve outcomes.
Recently some Authors evaluated the possible role of capsule endoscopy in the diagnostic work up of patients with isolated iron deficiency anaemia. These studies[37,38], although small, reported that the diagnostic yield of capsule endoscopy in this clinical setting seems quite similar to that reported in patients with obscure GI bleeding (about 50%). These studies confirmed that capsule endoscopy is superior to conventional radiological techniques also in patients with iron deficiency anaemia.
We know that Crohn’s disease can affect the small bowel: in approximately 45% of Crohn’s disease patients the disease involves both the small bowel and the colon and in about 25% the disease is confined to the small bowel, primarily the ileum[39], that can be often difficult to evaluate with endoscopic (retrograde ileoscopy) or radiological methods. For these reasons and because of an increasing number of patients with ulcerative lesions suggesting Crohn’s disease[28] has been discovered among subjects undergoing capsule endoscopy for other indications, capsule endoscopy has been also proposed to evaluate the small bowel mucosa of patients with Crohn’s disease. However the possible presence of asymptomatic stenoses hampered, at least at the beginning of the experience, the use of this device in patients with a previously established Crohn’s disease.
In fact, the first published papers[40,41] on this topic (in 2003) evaluated the diagnostic yield of capsule endoscopy in patients with suspected Crohn’s disease (with negative traditional work up, including bidirectional endoscopy and mostly SBFT). The diagnostic yield of capsule endoscopy in this subset of patients (ranging between 33% and 70%)[18,19] has been found, in two independent metanalyses, to be higher when compared with other diagnostic techniques (such as SBFT, SB enteroclysis and retrograde ileoscopy). Marmo et al[18] comparing capsule endoscopy with radiological techniques also calculated that the number needed to diagnose (NND) for this subgroup is 2 (95% CI 2-3). Unfortunately the majority of studies aimed at evaluating the role of capsule endoscopy in patients with suspected Crohn’s disease included a heterogeneous group of patients, seldom verified over time the final diagnosis by means of other independent diagnostic techniques (i.e. histology), and often used different criteria to classify the lesions identified. A recently published paper[42] tried to overcome possible confounding factors by clearly defining patients with suspected Crohn’s disease and by verifying the diagnosis over time. In this paper Girelli et al[42]
confirmed that capsule endoscopy is an effective tool to diagnose (positive likelihood ratio: 5.8) or to rule out (negative likelihood ratio: 0.08) small bowel Crohn’s disease in this particular subset of patients. The authors also pointed out that, in patients with suspected Crohn’s disease, assuming a 50% pre-test probability of disease, a positive capsule endoscopy gives a post-test probability of 85%.
The low frequency of capsule retention in patients undergoing capsule endoscopy for suspected Crohn's disease (approximately 1.5%, quite comparable with that reported in patients with obscure GI bleeding)[25] encouraged the application of this new technique also in patients with established Crohn’s disease[43-45]. As expected, in these patients capsule endoscopy showed a high diagnostic yield, significantly superior to that of retrograde ileoscopy and conventional radiological techniques[18,19]. Initial reports comparing capsule endoscopy with CT enteroclysis[46] in patients with established Crohn’s disease, although small in size, seem to confirm that capsule endoscopy has a high capability of identifying small inflammatory lesions in the small bowel, and to significantly modify the subsequent management of the patients. However, Golder et al[47], using MRI enteroclysis to evaluate the small bowel in patient with Crohn’s disease highlighted that, although capsule endoscopy is able to identify a larger number of lesions in the proximal -mid small bowel, in the distal small bowel, which is mostly affected by Crohn’s disease, capsule endoscopy and MRI enteroclysis are closely comparable. The authors also pointed out that in these patients MRI enteroclysis identified significant extra intestinal findings in about 30% of cases.
When trying to compare different diagnostic tools for the study of the small bowel in patients with established Crohn’s disease, we must keep in mind that capsule endoscopy has been performed exclusively in patients with non stricturing-non penetrating Crohn’s disease. In fact all patients in whom a radiological technique showed a stenosis (or a fistula or an abscess) that must be considered as a positive finding of these examinations, were excluded from the comparative studies, leading to a significant and systematic underestimation of the true diagnostic yield of the radiological techniques. Nevertheless, although in the majority of cases, patients with strictures identified with radiological examinations were excluded from capsule endoscopy studies, capsule retention occurred in 5%-13% of cases[25,48]. Capsule retention in patients with established Crohn’s disease can be managed, and sometimes partially solved, by giving steroids[49] or using DBE (both for capsule retrieval and stricture dilation)[50,51]. In this subset of patients, capsule endoscopy can be considered as a major complication because it often requires surgical intervention. The development of a dissolvable capsule (see below) may represent, in the near future, the best way to test intestinal patency before capsule endoscopy, in order to avoid capsule retention, especially in patients with Crohn’s disease.
Practice guidelines from ESGE[22] suggested that capsule endoscopy, owing to its high diagnostic yield, should have a very important place in the diagnostic work up of patients with known or suspected Crohn’s disease, but more large prospective studies are needed to evaluate the specificity of inflammatory lesions, the impact on long term outcome, the clinical significance of the assessment of the extent and severity of small bowel involvement and the risk of capsule retention.
Recent published studies showed also that VCE may have a role in assessing tissue healing after therapy with biologics, relapse after surgical intervention and small bowel evaluation in patients with ulcerative colitis undergoing total colectomy[52].
Surprisingly two[13,15] of eight randomized controlled studies published on capsule endoscopy evaluated the role of this technique in assessing small bowel lesions due to NSAIDs consumption. This probably derived from the fact that these widely used drugs can induce small, spotty and superficial mucosal lesions (i.e. mucosal breaks) difficult to identify with other techniques.
Goldstein et al[15] clearly demonstrated that NSAIDs (i.e. naproxen), even if associated with proton pump inhibitors (omeprazole), caused more small bowel mucosal lesions than placebo, while Gomez et al[13], comparing different NSAIDs, showed that Ibuprofen seems to cause small bowel mucosal damage less frequently than other drugs (dexibuprofen and diclofenac). Another study by Goldstein et al[53] comparing COX2-inhibitors with naproxen plus omeprazole showed that among healthy subjects with no endoscopic lesions at baseline, celecoxib was significantly associated with fewer small bowel mucosal breaks than ibuprofen plus omeprazole.
Nevertheless the most important information in this field is the demonstration that small mucosal inflammatory lesions (such as mucosal breaks, small isolated erosion or superficial ulcers) have been detected in about 10%-13% of healthy subjects[15]. Although the clinical implications of these findings remain unclear, the occurrence of these lesions in young healthy subjects, define a new benchmark that must be considered in any further clinical study about capsule endoscopy.
As far as celiac disease is concerned two studies[54,55] explored the performance of capsule endoscopy compared with histological evaluation of small bowel biopsies taken during gastroscopy in patients with suspected celiac disease.
Although both studies showed a high agreement between these two techniques (capsule endoscopy sensitivity 85%-87.5%, specificity 90.9%-100%, positive predictive value 96.5%-100% and a negative predictive value of 71.4%-88.9%) the authors underlined that, at present, traditional gastroscopy with duodenal biopsies remains the method of choice to assess mucosal atrophy in patients with suspected celiac disease. However, capsule endoscopy can be a suitable tool in patients, with high clinical suspicion of celiac disease, unable or unwilling to undergo traditional endoscopy.
At the present time, the main obstacle to the extensive use of capsule endoscopy in the diagnosis of celiac disease remains the high costs of the procedure, but also, as highlighted by Biagi et al[55], the difficulty in the graduation of mucosal atrophy (see below).
Two studies[57,58], published in 2005 and 2007 respectively, evaluated the role of capsule endoscopy in patients with complicated/refractory celiac disease. In this particular subset of patients capsule endoscopy has been performed to rule out malignant neoplasms [primarily enteropathy associated T-cell lymphoma (EATL)] or other complications (i.e. ulcerative jejunitis). The study of Culliford[57] depicted for the first time the spectrum of findings (such as scalloping of folds, nodularity and villous atrophy, but also strictures, intussusceptions or submucosal masses), identified by capsule endoscopy in patients with complicated celiac disease, while Daum et al[58], demonstrated that capsule endoscopy adds significant clinical information affecting further management mostly in patients with refractory celiac disease type II. Nevertheless, both studies included a small number of patients with refractory/complicated celiac disease undergoing a huge number of examinations to exclude strictures; in fact, as previously mentioned for Crohn’s disease, refractory celiac disease can also result in a structuring disease. For these reasons, and mainly because of its capability to take targeted biopsies, double balloon enteroscopy can represent, in this field, a reasonable alternative to capsule endoscopy[59].
Small bowel tumours are still considered, particularly when compared with gastric or colonic neoplasms, a rare disease accounting for 1% to 3 % of all primary gastrointestinal tumours[60], however, since the introduction of capsule endoscopy in clinical practice, some small studies have been published reporting a frequency of small bowel tumours ranging between 6% and 9%[61-65]. These studies, including a series of patients undergoing capsule endoscopy in which this tool was able to identify the presence of small bowel tumours, showed a higher than expected frequency of these tumours. However, two recently presented studies[66,67], published to date only in abstract form, showed a frequency of small bowel tumours ranging between 1.6% and 2.4%. There are no obvious explanations for this discrepancy between studies but the huge number of patients enrolled in the last two studies (more than 6000), the histological confirmation of all reported cases and the substantial concordance with data coming from surgical series, strongly decrease the reliability of earlier data.
All published series about capsule endoscopy in the diagnosis of small bowel tumours underlined that the main clinical indication for capsule endoscopy in these patients is obscure GI bleeding.
In agreement with previously published surgical series, small bowel tumours have been described at capsule endoscopy mostly as polyps (or masses) and stenoses, leading to capsule retention in about 10%[63] to 25%[67] of cases. The most frequent treatment in patients with small bowel tumours is surgical intervention, which, at the same time, allows the retrieval of capsules in case of retention of the device. Therefore, capsule retention in patients with small bowel tumours is considered nowadays as a minor complication.
Capsule endoscopy has also been proposed for the diagnosis and surveillance over time of patients with hereditary polyposis syndromes. The main advantage of this technique in this setting is the capability of this system to inspect the entire small bowel, avoiding radiation exposure and increasing patients’ compliance, which is a key point in surveillance programs. Several studies evaluated the possible role of capsule endoscopy in patients with polyposis syndromes[68-70] confirming that, also in this field, capsule endoscopy is more accurate than conventional radiology (SBFT and SB enteroclysis)[71]. Nevertheless the same Authors underlined that the main limitation of this technique, particularly when compared with MRI-enteroclysis, is related to the estimation of size (see below) and location of polyps[68]. At the present time it is suggested that CE should be performed, instead of SBFT, at the time of diagnosis and, as a part of surveillance programs, every 2-3 years, but also as a first diagnostic step in patients with symptoms (i.e. abdominal pain or anaemia)[69,72]. Indeed, keeping in mind the limitations of capsule endoscopy in patients with polyposis syndromes, double balloon enteroscopy can become an important tool, to accurately size and locate lesions, but also to remove polyps identified by capsule endoscopy[73].
The role of CE is less established in patients affected by familial adenomatous polyposis. In fact the quick passage of the capsule through the proximal duodenum can hamper the accurate visualization of the periampullary area. For these reasons, at present, capsule endoscopy is not recommended when the diagnosis of FAP is already established, but may be considered as a part of surveillance for patients with severe duodenal polyposis[69,70]. In a recently published prospective study Wong et al[74] compared CE with push enteroscopy and lower endoscopy in 32 patients with FAP. They showed that, in a defined segment of the small bowel, CE diagnosed significantly fewer small-bowel polyps than standard endoscopy, showed only fair agreement with PE in determining polyp counts, and was fairly inaccurate in detecting large polyps and in sizing them.
Abdominal pain, as a possible indication for capsule endoscopy, is still largely debated. Although small bowel tumours have sometimes been identified in patients undergoing capsule endoscopy for unexplained abdominal pain[52], two studies[75,76] evaluating a group of 36 patients with chronic abdominal pain of unknown origin and previous negative diagnostic work-up, found that capsule endoscopy was negative or not clinically relevant in more than 85% of subjects. On the other hand May et al[77] clearly demonstrated that when chronic abdominal pain is associated with other signs or symptoms (weight loss > 10% of body weight, inflammation shown by laboratory tests, chronic anemia, or suspected mid-gastrointestinal bleeding) relevant, or potentially relevant, findings are diagnosed by capsule endoscopy in about 60% of cases.
Capsule endoscopy has also been used, with promising results, in other rare clinical conditions such as indeterminate colitis[78,79], small bowel transplantation[80], graft versus host disease[81,82], protein losing entheropathy[83], primitive lymphangectasia[84] (mostly in the pediatric population), Whipple disease[85] and irritable bowel syndrome (with clinical suspicion of celiac disease)[86].
The majority of published papers we mentioned pointed out that the results obtained using capsule endoscopy in clinical practice mainly depend on the revolutionary technical characteristics of this device; however the same technical characteristics can represent, from a certain point of view, limitations of capsule enoscopy. These technical limitations can also explain, in the majority of cases, the clinical limitations of this examination.
Lewis et al[87] analyzing a master database, provided by Given Imaging Ltd (Yoqneam, Israel), found that the global miss rate of capsule endoscopy is about 11% ranging between 0.5% for ulcerative disease and 18.9% for neoplastic disease. Despite the estimated miss rate, capsule endoscopy is significantly lower than that of conventional examinations (global miss rate: 73.3%, miss rate for ulcerative lesions and neoplastic disease: 78.7% and 63.2% respectively) these percentages, in some selected subgroups of patients (i.e. patients with small bowel tumour) are alarming.
Unfortunately there are no conclusive explanations for false negative capsule endoscopies but several factors such as the incompleteness of examination (that can occur in 15%-20% of cases), technical limitations (battery life duration, field of view) and the suboptimal cleanliness of the small bowel (mostly in distal segments) can play a role[88].
At present, although all published papers strongly underlined that small bowel cleanliness is a key point to ensure a complete and accurate examination, and several papers aimed at evaluating factors (dietary restrictions and/or laxatives and/or prokinetic and/or postural tricks) potentially affecting small bowel cleanliness[12,14,16,17,89-93] have been published, there are still no recommendations about small bowel preparation for capsule endoscopy.
This mainly depends on the fact that most studies are published in abstract form, the methodological quality of these studies is rather low, because randomized comparisons are only a small minority, different regimens (with different combinations of drugs) are compared in each study, and an accepted and validated scale to evaluate bowel cleanliness does not exist yet. Four[12,14,16,17] out of 8 controlled randomized studies published on the field of capsule endoscopy are aimed at identifying the best preparation regimen for VCE but, unfortunately, this is only another proof of the relevance of this point.
Despite the lack of any clinical study on this field, all Authors used an overnight fast. An agreement has been reached, basically on the ground of two studies[12,17], about the helpful role of simethicone, administered 20 min before the procedure, in reducing bubbles all along the small bowel, but, the main issue (the presence of liquid stools or fecal debris) which can affect the diagnostic yield of capsule endoscopy, remains to be solved.
In fact, although in 2004[24] the ESGE guidelines, on the ground of the study of Viazis et al[16], suggested 2-liters of a poly-ethylen-glycol (PEG) based solution the day before the examination, as small bowel preparation, the updated release of guidelines from the same scientific society (published in 2006)[22] does not recommend any particular schedule of preparation.
The absence of a remote control and of the capability of taking biopsies significantly decrease the specificity of capsule endoscopy findings, since the diagnosis can be based only on the endoscopic appearance. The low specificity of lesions observed at capsule endoscopy is an issue that affects all fields of application of this technique, especially regarding inflammatory lesions (i.e. erosions, ulcers-Figure 4) which can derive from acute and chronic inflammatory bowel diseases[94], ischemic[95], neoplastic[61-67], infectious[96,97] or iatrogenic[98] diseases. As previously mentioned, small and initial inflammatory lesions have also been described in healthy subjects[15].
Another clinical problem strictly dependent on the technical characteristics of the system is the problem of sizing and locating small bowel lesions. This problem, mainly highlighted in studies performed in patients with small bowel hereditary polyposis syndromes[68-70] has important clinical consequences. In fact the size and the location of the lesions are a key point to define, ultimately, the clinical significance of capsule endoscopy findings and to direct further management. In patients with obscure GI bleeding some Authors[99,100] suggested a possible three grade scale (from P0 to P3) to rank capsule endoscopy findings depending on the likelihood of these lesions to explain the reason for referral while, for patients with Crohn’s disease two possible scores[43,101] have been proposed but not yet validated. In the field of celiac disease Biagi et al[56] clearly demonstrated a large inter- and intra-observer variation in the evaluation of the grade of mucosal atrophy (compared with traditional histology).
In patients with hereditary polyposis syndromes capsule endoscopy tends to overestimate the number of polyps while MRI-enteroclysis seems to be more reliable to correctly estimate the size of polypoid lesions, particularly for polyps of 1-2 centimetres, generally considered clinically relevant[71,74,102].
To improve the capability to estimate the size of polyps Racz et al[103] suggested the ingestion, 20’ before the procedure, of mesalazine granules as “reference” while Greapler et al[104] demonstrated that training with a capsule with a graduated dome might be helpful.
Although awareness of this complication existed at the time of the introduction of this device in clinical practice, the risk of capsule retention at the site of a previously unknown small bowel stricture remains the main complication of capsule endoscopy. This complication seems to be seldom predictable by conventional radiology[105] but the development of a specific dissolvable capsule, even if its safety profile is still under discussion, seems to be a reliable test to screen patients at high risk for capsule retention[107,106-109]. As we know the frequency of capsule retention seems mostly dependent on the clinical indication for capsule endoscopy, ranging between 0% in healthy subjects and 21% in patients with intestinal obstruction[25]. Although capsule retention is the most feared complication of capsule endoscopy, in some selected patients (i.e. patients with small bowel tumor in which capsule retention has been described in 10%-25% of cases) it can be considered as “positive” or minor complication being a sort of “red flag” identifying the presence of the disease. On the contrary, capsule retention must be considered a serious and major complication in patients in whom surgical intervention must be avoided as long as possible (i.e. patients with Crohn’s disease, in which capsule retention has been described in about 5%-13% of cases).
Although some case reports described a possible acute obstruction[110] or a possible perforation[111], due to capsule retention, these complications are nowadays considered exceptional. Retained capsules can be retrieved by means of surgical interventions (possibly in a laparoscopic setting)[112] or by means of enteroscopy (PE or DBE, depending on the site of retention)[113].
Several studies demonstrated the safety of capsule endoscopy in patients with thoracic pace-makers or implanted defibrillators[114-116] and recently a capsule endoscopy has also been performed on a woman in her third trimester of pregnancy because of a life threatening haemorrhage showing a carcinoid tumor[117].
Last but not least, although recently published studies confirmed that this examination is cost-effective in patients with obscure GI bleeding[118], the cost of the procedure can prevent the use of this potentially helpful device in everyday clinical practice. To partially reduce costs of the procedure a possible “two steps” strategy (first step; revision of the video by the nurse and second validation of results by a physician) has been proposed[119,120].
Capsule endoscopy, introduced into clinical practice in 2001, revolutionized the study of the small bowel, providing for the first time, a reliable and painless method to evaluate this organ endoscopically. In this paper we critically evaluated the body of evidence produced in these last six years. Unfortunately the majority of published papers are case reports or expert reviews.
Capsule endoscopy has been proven to be significantly superior to conventional radiological techniques (SBFT or SB enteroclysis) for any clinical indication. However studies comparing capsule endoscopy with new imaging techniques (MRI-enteroclysis or CT-enteroclysis) are still few, small and mainly focused on some selected topic (i.e. polyposis syndromes). At the present time capsule endoscopy is recommended as the third examination, after negative bidirectional endoscopy, in patients with obscure GI bleeding. A growing body of evidence suggests also that capsule endoscopy can have a key role in other clinical conditions such as Crohn’s disease, celiac disease, small bowel polyposis syndromes or small bowel tumours.
Although awareness of this complication existed at the time of the introduction of this device in clinical practice, the risk of capsule retention at the site of a previously unknown small bowel stricture remains the main complication of capsule endoscopy today. This complication seems to be seldom predictable by means of conventional radiology but the development of a specific dissolvable capsule might, in the near future, provide a safe and reliable test to identify patients at high risk for capsule retention.
Capsule endoscopy still suffers from some technical limitations (there is no remote control, it cannot take biopsies, insufflate air, suck fluids or debris) which can partially explain the clinical limitations/complications of this device (i.e. the difficulty in interpreting inflammatory lesions, in sizing and locating polyps, in grading mucosal atrophy).
The recently developed double balloon enteroscope, owing to its capability to explore a large part of the small bowel and to take targeted biopsies, although invasive and time consuming, can overcome some limitations of capsule endoscopy. At the present time, in the majority of clinical conditions (i.e. obscure GI bleeding), the winning strategy seems to be to couple these two techniques to explore in the most painless, safe and complete way the small bowel (with capsule endoscopy) and to define and treat the lesions identified (with double balloon enteroscopy).
We would like to thank Erik Herdes for the tremendous, essential and continuous technical support.
S- Editor Liu Y L- Editor Alpini GD E- Editor Lu W
| 1. | Liangpunsakul S, Maglinte DD, Rex DK. Comparison of wireless capsule endoscopy and conventional radiologic methods in the diagnosis of small bowel disease. Gastrointest Endosc Clin N Am. 2004;14:43-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 38] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 2. | Berner JS, Mauer K, Lewis BS. Push and sonde enteroscopy for the diagnosis of obscure gastrointestinal bleeding. Am J Gastroenterol. 1994;89:2139-2142. [PubMed] |
| 3. | Lewis BS, Kornbluth A, Waye JD. Small bowel tumours: yield of enteroscopy. Gut. 1991;32:763-765. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 99] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 4. | Oates BC, Morris AI. Enteroscopy. Curr Opin Gastroenterol. 2000;16:121-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 5. | Gay GJ, Delmotte JS. Enteroscopy in small intestinal inflammatory diseases. Gastrointest Endosc Clin N Am. 1999;9:115-123. [PubMed] |
| 6. | Perez-Cuadrado E, Macenlle R, Iglesias J, Fabra R, Lamas D. Usefulness of oral video push enteroscopy in Crohn's disease. Endoscopy. 1997;29:745-747. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 22] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Delvaux M. Capsule endoscopy in 2005: facts and perspectives. Best Pract Res Clin Gastroenterol. 2006;20:23-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 52] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 8. | Iddan G, Meron G, Glukhovsky A, Swain P. Wireless capsule endoscopy. Nature. 2000;405:417. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1994] [Cited by in RCA: 1383] [Article Influence: 55.3] [Reference Citation Analysis (1)] |
| 9. | Available from: http: //www.cebm.net/downloads/Oxford_EBM_Levels_5.rtf. |
| 10. | Shiotani A, Opekun AR, Graham DY. Visualization of the small intestine using capsule endoscopy in healthy subjects. Dig Dis Sci. 2007;52:1019-1025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 35] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 11. | de Leusse A, Vahedi K, Edery J, Tiah D, Fery-Lemonnier E, Cellier C, Bouhnik Y, Jian R. Capsule endoscopy or push enteroscopy for first-line exploration of obscure gastrointestinal bleeding? Gastroenterology. 2007;132:855-862; quiz 1164-1165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 89] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 12. | Ge ZZ, Chen HY, Gao YJ, Hu YB, Xiao SD. The role of simeticone in small-bowel preparation for capsule endoscopy. Endoscopy. 2006;38:836-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 50] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 13. | Gómez BJ, Caunedo A, Redondo L, Esteban J, Sáenz-Dana M, Blasco M, Hergueta P, Rodríguez-Téllez M, Romero R, Pellicer FJ. Modification of pepsinogen I levels and their correlation with gastrointestinal injury after administration of dexibuprofen, ibuprofen or diclofenac: a randomized, open-label, controlled clinical trial. Int J Clin Pharmacol Ther. 2006;44:154-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 14. | Caddy GR, Moran L, Chong AK, Miller AM, Taylor AC, Desmond PV. The effect of erythromycin on video capsule endoscopy intestinal-transit time. Gastrointest Endosc. 2006;63:262-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 61] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 15. | Goldstein JL, Eisen GM, Lewis B, Gralnek IM, Zlotnick S, Fort JG. Video capsule endoscopy to prospectively assess small bowel injury with celecoxib, naproxen plus omeprazole, and placebo. Clin Gastroenterol Hepatol. 2005;3:133-141. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 499] [Cited by in RCA: 451] [Article Influence: 22.6] [Reference Citation Analysis (0)] |
| 16. | Viazis N, Sgouros S, Papaxoinis K, Vlachogiannakos J, Bergele C, Sklavos P, Panani A, Avgerinos A. Bowel preparation increases the diagnostic yield of capsule endoscopy: a prospective, randomized, controlled study. Gastrointest Endosc. 2004;60:534-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 129] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 17. | Albert J, Göbel CM, Lesske J, Lotterer E, Nietsch H, Fleig WE. Simethicone for small bowel preparation for capsule endoscopy: a systematic, single-blinded, controlled study. Gastrointest Endosc. 2004;59:487-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 98] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 18. | Marmo R, Rotondano G, Piscopo R, Bianco MA, Cipolletta L. Meta-analysis: capsule enteroscopy vs. conventional modalities in diagnosis of small bowel diseases. Aliment Pharmacol Ther. 2005;22:595-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 153] [Cited by in RCA: 142] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 19. | Triester SL, Leighton JA, Leontiadis GI, Gurudu SR, Fleischer DE, Hara AK, Heigh RI, Shiff AD, Sharma VK. A meta-analysis of the yield of capsule endoscopy compared to other diagnostic modalities in patients with non-stricturing small bowel Crohn's disease. Am J Gastroenterol. 2006;101:954-964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 392] [Cited by in RCA: 368] [Article Influence: 19.4] [Reference Citation Analysis (1)] |
| 20. | Leighton JA, Triester SL, Sharma VK. Capsule endoscopy: a meta-analysis for use with obscure gastrointestinal bleeding and Crohn's disease. Gastrointest Endosc Clin N Am. 2006;16:229-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 75] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 21. | Triester SL, Leighton JA, Leontiadis GI, Fleischer DE, Hara AK, Heigh RI, Shiff AD, Sharma VK. A meta-analysis of the yield of capsule endoscopy compared to other diagnostic modalities in patients with obscure gastrointestinal bleeding. Am J Gastroenterol. 2005;100:2407-2418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 419] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 22. | Rey JF, Ladas S, Alhassani A, Kuznetsov K. European Society of Gastrointestinal Endoscopy (ESGE). Video capsule endoscopy: update to guidelines (May 2006). Endoscopy. 2006;38:1047-1053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 94] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 23. | Mishkin DS, Chuttani R, Croffie J, Disario J, Liu J, Shah R, Somogyi L, Tierney W, Song LM, Petersen BT. ASGE Technology Status Evaluation Report: wireless capsule endoscopy. Gastrointest Endosc. 2006;63:539-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 169] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 24. | Rey JF, Gay G, Kruse A, Lambert R. European Society of Gastrointestinal Endoscopy guideline for video capsule endoscopy. Endoscopy. 2004;36:656-658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 47] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 25. | Pennazio M. Capsule endoscopy: where are we after 6 years of clinical use? Dig Liver Dis. 2006;38:867-878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 63] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 26. | Tatar EL, Shen EH, Palance AL, Sun JH, Pitchumoni CS. Clinical utility of wireless capsule endoscopy: experience with 200 cases. J Clin Gastroenterol. 2006;40:140-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 27. | Tang SJ, Haber GB. Capsule endoscopy in obscure gastrointestinal bleeding. Gastrointest Endosc Clin N Am. 2004;14:87-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 51] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 28. | Pennazio M, Santucci R, Rondonotti E, Abbiati C, Beccari G, Rossini FP, De Franchis R. Outcome of patients with obscure gastrointestinal bleeding after capsule endoscopy: report of 100 consecutive cases. Gastroenterology. 2004;126:643-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 637] [Cited by in RCA: 604] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 29. | Sturniolo GC, Di Leo V, Vettorato MG, De Boni M, Lamboglia F, De Bona M, Bellumat A, Martines D, D'Inca R. Small bowel exploration by wireless capsule endoscopy: results from 314 procedures. Am J Med. 2006;119:341-347. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 30. | Pennazio M. Bleeding update. Gastrointest Endosc Clin N Am. 2006;16:251-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 31. | May A, Wardak A, Nachbar L, Remke S, Ell C. Influence of patient selection on the outcome of capsule endoscopy in patients with chronic gastrointestinal bleeding. J Clin Gastroenterol. 2005;39:684-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 32. | Estévez E, González-Conde B, Vázquez-Iglesias JL, de Los Angeles Vázquez-Millán M, Pértega S, Alonso PA, Clofent J, Santos E, Ulla JL, Sánchez E. Diagnostic yield and clinical outcomes after capsule endoscopy in 100 consecutive patients with obscure gastrointestinal bleeding. Eur J Gastroenterol Hepatol. 2006;18:881-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 60] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 33. | Pennazio M, Eisen G, Goldfarb N. ICCE consensus for obscure gastrointestinal bleeding. Endoscopy. 2005;37:1046-1050. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 117] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 34. | Delvaux M, Fassler I, Gay G. Clinical usefulness of the endoscopic video capsule as the initial intestinal investigation in patients with obscure digestive bleeding: validation of a diagnostic strategy based on the patient outcome after 12 months. Endoscopy. 2004;36:1067-1073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 136] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 35. | Leighton JA, Sharma VK, Hentz JG, Musil D, Malikowski MJ, McWane TL, Fleischer DE. Capsule endoscopy versus push enteroscopy for evaluation of obscure gastrointestinal bleeding with 1-year outcomes. Dig Dis Sci. 2006;51:891-899. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 36. | Hartmann D, Schmidt H, Bolz G, Schilling D, Kinzel F, Eickhoff A, Huschner W, Möller K, Jakobs R, Reitzig P. A prospective two-center study comparing wireless capsule endoscopy with intraoperative enteroscopy in patients with obscure GI bleeding. Gastrointest Endosc. 2005;61:826-832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 201] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 37. | Apostolopoulos P, Liatsos C, Gralnek IM, Giannakoulopoulou E, Alexandrakis G, Kalantzis C, Gabriel P, Kalantzis N. The role of wireless capsule endoscopy in investigating unexplained iron deficiency anemia after negative endoscopic evaluation of the upper and lower gastrointestinal tract. Endoscopy. 2006;38:1127-1132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 71] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 38. | Fireman Z, Kopelman Y. The role of video capsule endoscopy in the evaluation of iron deficiency anaemia. Dig Liver Dis. 2004;36:97-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 39. | Baumgart DC, Sandborn WJ. Inflammatory bowel disease: clinical aspects and established and evolving therapies. Lancet. 2007;369:1641-1657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1247] [Cited by in RCA: 1353] [Article Influence: 75.2] [Reference Citation Analysis (0)] |
| 40. | Fireman Z, Mahajna E, Broide E, Shapiro M, Fich L, Sternberg A, Kopelman Y, Scapa E. Diagnosing small bowel Crohn's disease with wireless capsule endoscopy. Gut. 2003;52:390-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 270] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 41. | Herrerías JM, Caunedo A, Rodríguez-Téllez M, Pellicer F, Herrerías JM. Capsule endoscopy in patients with suspected Crohn's disease and negative endoscopy. Endoscopy. 2003;35:564-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 167] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 42. | Girelli CM, Porta P, Malacrida V, Barzaghi F, Rocca F. Clinical outcome of patients examined by capsule endoscopy for suspected small bowel Crohn's disease. Dig Liver Dis. 2007;39:148-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 45] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 43. | Mow WS, Lo SK, Targan SR, Dubinsky MC, Treyzon L, Abreu-Martin MT, Papadakis KA, Vasiliauskas EA. Initial experience with wireless capsule enteroscopy in the diagnosis and management of inflammatory bowel disease. Clin Gastroenterol Hepatol. 2004;2:31-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 193] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 44. | Marmo R, Rotondano G, Piscopo R, Bianco MA, Siani A, Catalano O, Cipolletta L. Capsule endoscopy versus enteroclysis in the detection of small-bowel involvement in Crohn's disease: a prospective trial. Clin Gastroenterol Hepatol. 2005;3:772-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 96] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 45. | Voderholzer WA. The role of PillCam endoscopy in Crohn's disease: the European experience. Gastrointest Endosc Clin N Am. 2006;16:287-97, ix. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 46. | Voderholzer WA, Beinhoelzl J, Rogalla P, Murrer S, Schachschal G, Lochs H, Ortner MA. Small bowel involvement in Crohn's disease: a prospective comparison of wireless capsule endoscopy and computed tomography enteroclysis. Gut. 2005;54:369-373. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 243] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 47. | Gölder SK, Schreyer AG, Endlicher E, Feuerbach S, Schölmerich J, Kullmann F, Seitz J, Rogler G, Herfarth H. Comparison of capsule endoscopy and magnetic resonance (MR) enteroclysis in suspected small bowel disease. Int J Colorectal Dis. 2006;21:97-104. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 102] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 48. | Barkin JS, Friendman S. Wireless endoscopy requiring surgical intervention: the world's experience. Am J Gastroenterol. 2002;97:S298. [RCA] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 96] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 49. | Cave D, Legnani P, de Franchis R, Lewis BS. ICCE consensus for capsule retention. Endoscopy. 2005;37:1065-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 255] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 50. | May A, Nachbar L, Ell C. Extraction of entrapped capsules from the small bowel by means of push-and-pull enteroscopy with the double-balloon technique. Endoscopy. 2005;37:591-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 61] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 51. | Sunada K, Yamamoto H, Kita H, Yano T, Sato H, Hayashi Y, Miyata T, Sekine Y, Kuno A, Iwamoto M. Clinical outcomes of enteroscopy using the double-balloon method for strictures of the small intestine. World J Gastroenterol. 2005;11:1087-1089. [PubMed] |
| 52. | Lo SK. Capsule endoscopy in the diagnosis and management of inflammatory bowel disease. Gastrointest Endosc Clin N Am. 2004;14:179-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 53. | Goldstein JL, Eisen GM, Lewis B, Gralnek IM, Aisenberg J, Bhadra P, Berger MF. Small bowel mucosal injury is reduced in healthy subjects treated with celecoxib compared with ibuprofen plus omeprazole, as assessed by video capsule endoscopy. Aliment Pharmacol Ther. 2007;25:1211-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 140] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 54. | Hopper AD, Sidhu R, Hurlstone DP, McAlindon ME, Sanders DS. Capsule endoscopy: an alternative to duodenal biopsy for the recognition of villous atrophy in coeliac disease? Dig Liver Dis. 2007;39:140-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 69] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 55. | Rondonotti E, Spada C, Cave D, Pennazio M, Riccioni ME, De Vitis I, Schneider D, Sprujevnik T, Villa F, Langelier J. Video capsule enteroscopy in the diagnosis of celiac disease: a multicenter study. Am J Gastroenterol. 2007;102:1624-1631. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 123] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 56. | Biagi F, Rondonotti E, Campanella J, Villa F, Bianchi PI, Klersy C, De Franchis R, Corazza GR. Video capsule endoscopy and histology for small-bowel mucosa evaluation: a comparison performed by blinded observers. Clin Gastroenterol Hepatol. 2006;4:998-1003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 58] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 57. | Culliford A, Daly J, Diamond B, Rubin M, Green PH. The value of wireless capsule endoscopy in patients with complicated celiac disease. Gastrointest Endosc. 2005;62:55-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 132] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 58. | Daum S, Wahnschaffe U, Glasenapp R, Borchert M, Ullrich R, Zeitz M, Faiss S. Capsule endoscopy in refractory celiac disease. Endoscopy. 2007;39:455-458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 78] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 59. | Hadithi M, Al-toma A, Oudejans J, van Bodegraven AA, Mulder CJ, Jacobs M. The value of double-balloon enteroscopy in patients with refractory celiac disease. Am J Gastroenterol. 2007;102:987-996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 84] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 60. | DiSarjo JA, Burt RW, Vargas H, McWhorter WP. Small bowel cancer: epidemiological and clinical characteristics from a population-based registry. Am J Gastroenterol. 1994;89:699-701. [PubMed] |
| 61. | de Franchis R, Rondonotti E, Abbiati C, Beccari G, Signorelli C. Small bowel malignancy. Gastrointest Endosc Clin N Am. 2004;14:139-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 33] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 62. | Cobrin GM, Pittman RH, Lewis BS. Increased diagnostic yield of small bowel tumors with capsule endoscopy. Cancer. 2006;107:22-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 127] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 63. | Bailey AA, Debinski HS, Appleyard MN, Remedios ML, Hooper JE, Walsh AJ, Selby WS. Diagnosis and outcome of small bowel tumors found by capsule endoscopy: a three-center Australian experience. Am J Gastroenterol. 2006;101:2237-2243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 124] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 64. | Estévez E, González-Conde B, Vázquez-Iglesias JL, Alonso PA, Vázquez-Millán Mde L, Pardeiro R. Incidence of tumoral pathology according to study using capsule endoscopy for patients with obscure gastrointestinal bleeding. Surg Endosc. 2007;21:1776-1780. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 65. | Urbain D, De Looze D, Demedts I, Louis E, Dewit O, Macken E, Van Gossum A. Video capsule endoscopy in small-bowel malignancy: a multicenter Belgian study. Endoscopy. 2006;38:408-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 52] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 66. | Rondonotti E, Pennazio M. Small bowel tumor detected by video-capsule endoscopy (VCE): preliminary results from European Capsule Endoscopy Group database. Gastrointestinal Endoscopy. 2007;65:AB5. [DOI] [Full Text] |
| 67. | Pasha SF, Sharma VK, Carey EJ, Shiff AD, Heigh RI, Gurudu SR, Erickson PJ, Post JK, Hara AK, Fleischer DE. Utility of video capsule endoscopy in the detection of small bowel tumors. A single center experience of 1000 consecutive patients. Proceedings of the 6th International Conference on Capsule Endoscopy; 2007 June 8-10; Madrid, Spain, 2007: 45. . |
| 68. | Brown G, Fraser C, Schofield G, Taylor S, Bartram C, Phillips R, Saunders B. Video capsule endoscopy in peutz-jeghers syndrome: a blinded comparison with barium follow-through for detection of small-bowel polyps. Endoscopy. 2006;38:385-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 69] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 69. | Schulmann K, Hollerbach S, Kraus K, Willert J, Vogel T, Möslein G, Pox C, Reiser M, Reinacher-Schick A, Schmiegel W. Feasibility and diagnostic utility of video capsule endoscopy for the detection of small bowel polyps in patients with hereditary polyposis syndromes. Am J Gastroenterol. 2005;100:27-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 150] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 70. | Burke CA, Santisi J, Church J, Levinthal G. The utility of capsule endoscopy small bowel surveillance in patients with polyposis. Am J Gastroenterol. 2005;100:1498-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 125] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 71. | Caspari R, von Falkenhausen M, Krautmacher C, Schild H, Heller J, Sauerbruch T. Comparison of capsule endoscopy and magnetic resonance imaging for the detection of polyps of the small intestine in patients with familial adenomatous polyposis or with Peutz-Jeghers' syndrome. Endoscopy. 2004;36:1054-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 145] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 72. | Latchford A, Greenhalf W, Vitone LJ, Neoptolemos JP, Lancaster GA, Phillips RK. Peutz-Jeghers syndrome and screening for pancreatic cancer. Br J Surg. 2006;93:1446-1455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 37] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 73. | Ohmiya N, Taguchi A, Shirai K, Mabuchi N, Arakawa D, Kanazawa H, Ozeki M, Yamada M, Nakamura M, Itoh A. Endoscopic resection of Peutz-Jeghers polyps throughout the small intestine at double-balloon enteroscopy without laparotomy. Gastrointest Endosc. 2005;61:140-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 111] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 74. | Wong RF, Tuteja AK, Haslem DS, Pappas L, Szabo A, Ogara MM, DiSario JA. Video capsule endoscopy compared with standard endoscopy for the evaluation of small-bowel polyps in persons with familial adenomatous polyposis (with video). Gastrointest Endosc. 2006;64:530-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 72] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 75. | Spada C, Pirozzi GA, Riccioni ME, Iacopini F, Marchese M, Costamagna G. Capsule endoscopy in patients with chronic abdominal pain. Dig Liver Dis. 2006;38:696-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 76. | Bardan E, Nadler M, Chowers Y, Fidder H, Bar-Meir S. Capsule endoscopy for the evaluation of patients with chronic abdominal pain. Endoscopy. 2003;35:688-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 72] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 77. | May A, Manner H, Schneider M, Ipsen A, Ell C. Prospective multicenter trial of capsule endoscopy in patients with chronic abdominal pain, diarrhea and other signs and symptoms (CEDAP-Plus Study). Endoscopy. 2007;39:606-612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 66] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 78. | Viazis N, Karamanolis DG. Indeterminate colitis--the role of wireless capsule endoscopy. Aliment Pharmacol Ther. 2007;25:859; author reply 860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 79. | Maunoury V, Savoye G, Bourreille A, Bouhnik Y, Jarry M, Sacher-Huvelin S, Ben Soussan E, Lerebours E, Galmiche JP, Colombel JF. Value of wireless capsule endoscopy in patients with indeterminate colitis (inflammatory bowel disease type unclassified). Inflamm Bowel Dis. 2007;13:152-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 94] [Cited by in RCA: 87] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 80. | de Franchis R, Rondonotti E, Abbiati C, Beccari G, Merighi A, Pinna A, Villa E. Capsule enteroscopy in small bowel transplantation. Dig Liver Dis. 2003;35:728-731. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 81. | Neumann S, Schoppmeyer K, Lange T, Wiedmann M, Golsong J, Tannapfel A, Mossner J, Niederwieser D, Caca K. Wireless capsule endoscopy for diagnosis of acute intestinal graft-versus-host disease. Gastrointest Endosc. 2007;65:403-409. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 82. | Silbermintz A, Sahdev I, Moy L, Vlachos A, Lipton J, Levine J. Capsule endoscopy as a diagnostic tool in the evaluation of graft-vs.-host disease. Pediatr Transplant. 2006;10:252-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 83. | Pungpapong S, Stark ME, Cangemi JR. Protein-losing enteropathy from eosinophilic enteritis diagnosed by wireless capsule endoscopy and double-balloon enteroscopy. Gastrointest Endosc. 2007;65:917-918; discussion 918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 84. | Vignes S, Bellanger J. [Videocapsule endoscopy as a useful tool to diagnose primary intestinal lymphangiectasia]. Rev Med Interne. 2007;28:173-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 85. | Fritscher-Ravens A, Swain CP, von Herbay A. Refractory Whipple's disease with anaemia: first lessons from capsule endoscopy. Endoscopy. 2004;36:659-662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 86. | Adler SN, Jacob H, Lijovetzky G, Mulder CJ, Zwiers A. Positive coeliac serology in irritable bowel syndrome patients with normal duodenal biopsies: Video capsule endoscopy findings and HLA-DQ typing may affect clinical management. J Gastrointestin Liver Dis. 2006;15:221-225. [PubMed] |
| 87. | Lewis BS, Eisen GM, Friedman S. A pooled analysis to evaluate results of capsule endoscopy trials. Endoscopy. 2005;37:960-965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 187] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 88. | Rondonotti E, Herrerias JM, Pennazio M, Caunedo A, Mascarenhas-Saraiva M, de Franchis R. Complications, limitations, and failures of capsule endoscopy: a review of 733 cases. Gastrointest Endosc. 2005;62:712-76; quiz 752, 754. [PubMed] |
| 89. | Niv Y, Niv G, Wiser K, Demarco DC. Capsule endoscopy - comparison of two strategies of bowel preparation. Aliment Pharmacol Ther. 2005;22:957-962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 90. | Ben-Soussan E, Savoye G, Antonietti M, Ramirez S, Ducrotté P, Lerebours E. Is a 2-liter PEG preparation useful before capsule endoscopy? J Clin Gastroenterol. 2005;39:381-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 74] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 91. | Dai N, Gubler C, Hengstler P, Meyenberger C, Bauerfeind P. Improved capsule endoscopy after bowel preparation. Gastrointest Endosc. 2005;61:28-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 99] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 92. | Niv Y, Niv G. Capsule endoscopy: role of bowel preparation in successful visualization. Scand J Gastroenterol. 2004;39:1005-1009. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 59] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 93. | Fireman Z, Kopelman Y, Fish L, Sternberg A, Scapa E, Mahaina E. Effect of oral purgatives on gastric and small bowel transit time in capsule endoscopy. Isr Med Assoc J. 2004;6:521-523. [PubMed] |
| 94. | Bar-Meir S. Review article: capsule endoscopy - are all small intestinal lesions Crohn's disease? Aliment Pharmacol Ther. 2006;24 Suppl 3:19-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 22] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 95. | Liatsos C, Goulas S, Karagiannis S, Patelaros E, Sabaziotis D, Mavrogiannis C. Diagnosis of small-bowel ischemic necrosis by capsule endoscopy. Gastrointest Endosc. 2005;62:439-440; discussion 440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 96. | Hirata M, Yamaguchi Y, Ikei Y, Koyama G, Matsui T, Ishida H, Takahashi S. A case of Diphyllobothrium latum/nihonkaiense infection identified by capsule endoscopy in small intestine. Gastrointest Endosc. 2006;64:129; discussion 130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 97. | Cello JP. Capsule endoscopy features of human immunodeficiency virus and geographical diseases. Gastrointest Endosc Clin N Am. 2004;14:169-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 98. | Jazwinski A, Palazzo J, Kastenberg D. Capsule endoscopy diagnosis of radiation enteritis in a patient previously considered to have celiac sprue. Endoscopy. 2007;39 Suppl 1:E66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 99. | Saurin JC, Delvaux M, Gaudin JL, Fassler I, Villarejo J, Vahedi K, Bitoun A, Canard JM, Souquet JC, Ponchon T. Diagnostic value of endoscopic capsule in patients with obscure digestive bleeding: blinded comparison with video push-enteroscopy. Endoscopy. 2003;35:576-584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 314] [Article Influence: 14.3] [Reference Citation Analysis (0)] |
| 100. | Costamagna G, Shah SK, Riccioni ME, Foschia F, Mutignani M, Perri V, Vecchioli A, Brizi MG, Picciocchi A, Marano P. A prospective trial comparing small bowel radiographs and video capsule endoscopy for suspected small bowel disease. Gastroenterology. 2002;123:999-1005. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 565] [Cited by in RCA: 517] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 101. | Kornbluth A, Legnani P, Lewis BS. Video capsule endoscopy in inflammatory bowel disease: past, present, and future. Inflamm Bowel Dis. 2004;10:278-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 54] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 102. | Soares J, Lopes L, Vilas Boas G, Pinho C. Wireless capsule endoscopy for evaluation of phenotypic expression of small-bowel polyps in patients with Peutz-Jeghers syndrome and in symptomatic first-degree relatives. Endoscopy. 2004;36:1060-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 67] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 103. | Rácz I, Jánoki M, Kovács V. Measurement of small-bowel polyp size in patients with Peutz-Jeghers syndrome by using reference granules during video capsule endoscopy. Endoscopy. 2007;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 104. | Greapler F, Wolter M, Vonthein R, Gregor . Accuracy of size estimation in wireless capsule endoscopy. Gastrointestinal Endoscopy. 2007;65:AB160. |
| 105. | Ho KK, Joyce AM. Complications of capsule endoscopy. Gastrointest Endosc Clin N Am. 2007;17:169-178, viii-ix. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 49] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 106. | Signorelli C, Rondonotti E, Villa F, Abbiati C, Beccari G, Avesani EC, Vecchi M, de Franchis R. Use of the Given Patency System for the screening of patients at high risk for capsule retention. Dig Liver Dis. 2006;38:326-330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 68] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 107. | Spada C, Riccioni ME, Costamagna G. Patients with known small bowel stricture or with symptoms of small bowel obstruction secondary to Crohn's disease should not perform video capsule endoscopy without being previously tested for small bowel patency. Am J Gastroenterol. 2007;102:1542-1543; author reply 1543-1544;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 108. | Banerjee R, Bhargav P, Reddy P, Gupta R, Lakhtakia S, Tandan M, Rao VG, Reddy ND. Safety and efficacy of the M2A patency capsule for diagnosis of critical intestinal patency: results of a prospective clinical trial. J Gastroenterol Hepatol. 2007;22:2060-2063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 109. | Delvaux M, Ben Soussan E, Laurent V, Lerebours E, Gay G. Clinical evaluation of the use of the M2A patency capsule system before a capsule endoscopy procedure, in patients with known or suspected intestinal stenosis. Endoscopy. 2005;37:801-807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 102] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 110. | Lin OS, Brandabur JJ, Schembre DB, Soon MS, Kozarek RA. Acute symptomatic small bowel obstruction due to capsule impaction. Gastrointest Endosc. 2007;65:725-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 44] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 111. | Picazo-Yeste J, González-Carro P, Moreno-Sanz C, Seoane-González J. [Intestinal perforation secondary to impaction of a retained endoscopic capsule]. Cir Esp. 2006;79:316-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 112. | Dominguez EP, Choi Y, Raijman IL, Sweeney JF. Laparoscopic approach for the retrieval of retained video capsule endoscopy. JSLS. 2006;10:496-498. [PubMed] |
| 113. | Tanaka S, Mitsui K, Shirakawa K, Tatsuguchi A, Nakamura T, Hayashi Y, Sakamoto C, Terano A. Successful retrieval of video capsule endoscopy retained at ileal stenosis of Crohn's disease using double-balloon endoscopy. J Gastroenterol Hepatol. 2006;21:922-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 32] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 114. | Payeras G, Piqueras J, Moreno VJ, Cabrera A, Menéndez D, Jiménez R. Effects of capsule endoscopy on cardiac pacemakers. Endoscopy. 2005;37:1181-1185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 115. | Guyomar Y, Vandeville L, Heuls S, Coviaux F, Graux P, Cornaert P, Filoche B. Interference between pacemaker and video capsule endoscopy. Pacing Clin Electrophysiol. 2004;27:1329-1330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 116. | Girelli CM, Tartara P, Vitali E. Lack of reciprocal interference between capsule endoscope and left ventricular assist device. Endoscopy. 2006;38:94-95; discussion 95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 117. | Hogan RB, Ahmad N, Hogan RB 3rd, Hensley SD, Phillips P, Doolittle P, Reimund E. Video capsule endoscopy detection of jejunal carcinoid in life-threatening hemorrhage, first trimester pregnancy. Gastrointest Endosc. 2007;66:205-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 118. | Marmo R, Rotondano G, Rondonotti E, de Franchis R, D'Incà R, Vettorato MG, Costamagna G, Riccioni ME, Spada C, D'Angella R. Capsule enteroscopy vs. other diagnostic procedures in diagnosing obscure gastrointestinal bleeding: a cost-effectiveness study. Eur J Gastroenterol Hepatol. 2007;19:535-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 119. | Bossa F, Cocomazzi G, Valvano MR, Andriulli A, Annese V. Detection of abnormal lesions recorded by capsule endoscopy. A prospective study comparing endoscopist's and nurse's accuracy. Dig Liver Dis. 2006;38:599-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 120. | Niv Y, Niv G. Capsule endoscopy examination--preliminary review by a nurse. Dig Dis Sci. 2005;50:2121-2124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 40] [Article Influence: 2.0] [Reference Citation Analysis (0)] |