INTRODUCTION
A polypoidal lesion may be defined as an elevation on the mucosa. Such polypoid lesions in the gallbladder are present in about 5% of the adult population[1]. As ultrasound technology improves, an increasing number of polypoid lesions of the gallbladder are being detected[2]. Although some patients are symptomatic, others are found incidentally. The potential range of pathological lesions is large, and the differentiation between benign, malignant and potentially malignant lesions is a major diagnostic dilemma. Christensen and Ishak[3] categorized polypoid lesions of the gallbladder into: true tumors (adenoma, leiomyoma, lipoma and others), pseudotumors (cholesterol polyp, adenomyomatosis, heterotopias and inflammatory lesions), and malignant lesions.
Adenomas of the biliary tract are uncommon. Gallbladder adenomas are found in 0.5% of cholecystectomy specimens[4]. The vast majority of gallbladder adenomas have a tubular structure while tubulovillous adenomas are exceedingly rare. The incidence of pure villous adenomas of the gallbladder is only 0.08% in autopsy series[5].
CASE REPORT
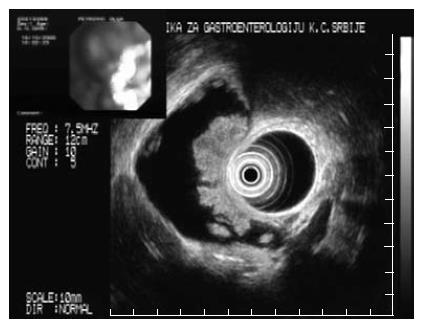
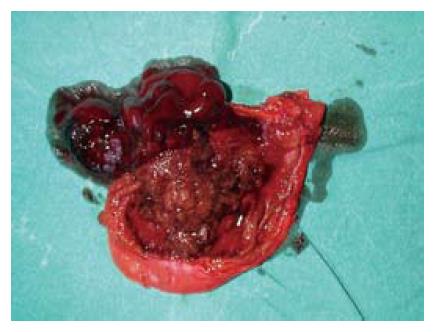
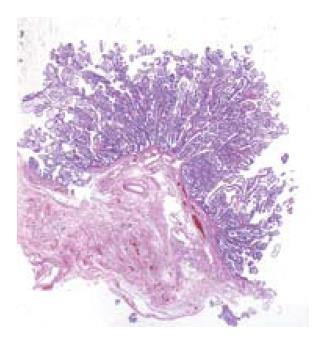
A middle aged female with acromegaly diagnosed 5 years earlier, was admitted to the hospital because of nonspecific gastrointestinal complaints. Conventional ultrasonography (US) revealed a large heterogeneous polypoid lesion in the gallbladder. Endoscopic ultrasonography (EUS) of the upper GI tract and biliary system was performed, using an Olympus radial 7.5/12 MHz switchable probe. The lesion was described as a large sessile polyp, measuring 4.5 cm, and consisting of numerous pedicles which were moving independently during the examination (Figure 1). The tumor was hyperechoic and hetrogenous, with an irregular surface, and did not show any penetration of the muscularis propria layer. There were several small polyps surrounding the large lesion. No polyps were identi-fied in the gastrointestinal tract on colonoscopy, push enteroscopy and small bowel follow through examination. The patient underwent an open cholecystectomy which confirmed the findings of EUS (Figure 2). Histopathology revealed a villous adenoma with regular arborization of the villo-papillar adenomatous epithelium and peripheral stromal edema of the villi (Figure 3). Most of the microscopic fields showed low grade epithelial dysplasia, with a few small foci of prominent atypia consistent with intraepithelial carcinoma. Numerous small polyps located in the periphery of the larger one were tubular adenomas, with only low grade epithelial atypia. The muscularis propria layer of the gallbladder was clearly preserved and the local lymph nodes were unremarkable. After five years, the patient reports no complaints and repeated clinical examinations, laboratory testing, and imaging studies have been within the normal range.
Figure 1 Endosonography picture of the gallbladder polyp.
Figure 2 Resected specimen of the gallbladder.
Figure 3 Low power light microscopy (HE) of the villous adenoma.
DISCUSSION
Villous adenomas are benign epithelial lesions that can occur anywhere in the gastrointestinal tract. These tumors are usually found in the rectum and colon, less frequently in the small intestine, and rarely in the biliary tree[6,7]. This case report describes a patient with acromegaly and a villous adenoma of the gallbladder, the first such presentation seen in our region.
Seven hereditary syndromes associated with hamar-tomatous polyps have been described. These include familial juvenile polyposis syndrome, Cowden’s syndrome, Bannayan-Ruvalcaba-Riley syndrome, Peutz-Jeghers syndrome, basal cell nevus syndrome, neurofibromatosis 1, and multiple endocrine neoplastic syndrome 2B. Hereditary mixed polyposis syndrome is a variant of juvenile polyposis characterized by both hamartomatous and adenomatous polyps. The hamartomatous syndromes occur at appro-ximately 1/10th of the frequency of the adenomatous syndromes and account for < 1% of colorectal cancers in North America[8]. In these inherited diseases, there is a higher incidence of gallbladder polyps, but there is also a higher risk of extraintestinal malignancy[4]. While the diagnosis of these inherited syndromes is primarily clinical, genetic testing is now available for the six syndromes noted above. However, a significant number of spontaneous mutations have been described in each of these syndromes.
Patients with acromegaly are at an increased risk of developing neoplastic lesions, especially of the large bowel secondary to an increased incidence of colonic polyps and carcinomas. Histological examination of the polyps shows both adenomas as well as hyperplastic polyps. The pathogenesis is multifactorial. The major mechanism is believed to be IGF-I hypersecretion, as well as mutation of the tumor suppressor genes, such as adenomatous polyposis coli and p53, DDC (deleted in colon cancer), and DNA mismatch repair genes[9,10]. Nevertheless, despite several analyses the findings remain controversial[11-13]. To our knowledge, there is no link between gallbladder polyps and acromegaly. In our patient, the gallbladder polyp was the only neoplasm identified, and polyps were not detect-ed in the gastrointestinal tract, and no other neoplastic diseases were observed.
A review of the literature revealed ten cases of bile duct villous adenomas[6]. The most common location of these tumors was at the ampulla of Vater[14]. Most patients presented with obstructive jaundice and were treated surgically, supporting the importance of meticulous preoperative assessment and judicious use of surgical exploration.
In our patient, cholecystectomy was the ideal procedure. Laparoscopic cholecystectomy is generally the preferred operative choice. However, if the chances of malignancy are considered high, for example in polyps larger than 2 cm, open surgery is preferable in order to reduce the risk of tumor seeding associated with laparoscopic surgery[15]. This is why an open cholecystectomy was performed in our patient. Besides the clinical features, certain US and EUS findings are highly indicative of malignancy. Choi[16] presented a scoring system based on five variables: layer structure, echo pattern, margin of the polyp stalk, and number of polyps. Based on this scoring system, the appearance of the polyp in our patient (heterogeneous echo pattern, lobulated margin, sessile, multiple), despite preserved layer structure was highly suspicious for a malignant lesion. Thus, the scoring system is more accurate if the polypoid lesion measures between 5-15 mm.
Based on an analysis of surgical specimens of gallbladder polyps, it has been suggested that in symptomatic patients there should be no reservation about the need for surgery, regardless of the size of the lesion. Factors associated with an increased risk for malignancy are: age of the patient (> 60 years), size of the polyp (> 10 mm) and coexistence of gallstones. Other authors have suggested the following features as suspicious for malignancy and therefore an indication for surgical treatment: wide base lesion, lesion tending to enlarge in a short period, long pedicle, polyps at the neck preventing emptying of the gallbladder, and polyps associated with irregular thickening of the gallbladder wall[2,17,18].
A causal relation between the acromegaly and villous adenoma of the gallbladder cannot be established on the basis of a single case. Although the vast majority of gallbladder polyps are benign, these patients need careful evaluation and appropriate follow-up. In the present case, the polypoid lesion in the gallbladder was a villous adenoma, which has a significant potential for malignant transformation.