Published online Jun 7, 2007. doi: 10.3748/wjg.v13.i21.2960
Revised: April 3, 2007
Accepted: April 18, 2007
Published online: June 7, 2007
AIM: To study the (functional) relevance of single nucleotide polymorphisms (SNPs) in genes encoding matrix metalloproteinases (MMP)-1, -2, -3, -9, tissue inhibitors of metalloproteinases (TIMP)-1, -2 and tumor necrosis factor (TNF)-α in the etiopathogenesis of inflammatory bowel diseases (IBD), that may enhance susceptibility and/or disease severity.
METHODS: Genomic DNA from 134 Crohn’s disease (CD), 111 ulcerative colitis (UC) patients and 248 control subjects was isolated from resected intestinal tissue or blood. Allelic composition at SNP loci was determined by PCR-RFLP or tetra primer ARMS PCR.
RESULTS: The TIMP-1 genotype TT in women and T in men at SNP +372 T/C was found to increase CD susceptibility (39% vs 23.8%, P = 0.018 and 67.9% vs 51.6%, P = 0.055, respectively), while women with this genotype were less prone to development of fistulae during follow-up (41.4% vs 68.3%, P = 0.025). Male IBD or CD patients carrying the TIMP-1 +372 T-allele expressed lower levels of TIMP-1 in surgically resected macroscopically inflamed tissue (0.065 < P < 0.01). The 5T5T genotype at MMP-3 SNP -1613 5T/6T increased the chance of stenotic complications in CD during follow-up (91.2% vs 71.8%, P = 0.022) but seemed to protect against colonic involvement of this disease at first endoscopic/radiologic examination (35.3% vs 59.5%, P = 0.017).
CONCLUSION: Allelic composition at the examined SNPs in genes coding for TIMP-1 and MMP-3 affect CD susceptibility and/or phenotype, i.e., fistulizing disease, stricture pathogenesis and first disease localisation. These findings reinforce the important role of these proteins in IBD.
- Citation: Meijer MJ, Mieremet-Ooms MA, Hogezand RAV, Lamers CB, Hommes DW, Verspaget HW. Role of matrix metalloproteinase, tissue inhibitor of metalloproteinase and tumor necrosis factor-α single nucleotide gene polymorphisms in inflammatory bowel disease. World J Gastroenterol 2007; 13(21): 2960-2966
- URL: https://www.wjgnet.com/1007-9327/full/v13/i21/2960.htm
- DOI: https://dx.doi.org/10.3748/wjg.v13.i21.2960
Crohn’s disease (CD) is characterised by chronic, patchy, transmural inflammation of the gastrointestinal tract, predominantly in the ileocecal area, while ulcerative colitis (UC) is manifested by chronic, continuous, rather superficial inflammation of the mucosal layers of the colon[1,2]. The incidence and prevalence of both CD and UC have increased in the Western population since the second World War[3,4], and lately also increased in developing industrialising countries. Although there has been much controversy regarding etiology and pathogenesis of both forms of inflammatory bowel disease (IBD), recent evidence points to an exaggerated immune response to enteric bacterial flora in genetically susceptible individuals. Based on a higher disease concordance in monozygotic vs dizygotic twins[5], a higher frequency of IBD in certain families and ethnic groups[6,7], the association of IBD with genetic disorders like Turner’s and Hermansky-Pudlak syndrome[8,9], the presence of a genetic component in IBD is evident. Indeed, large-scale genome-wide linkage studies have mapped several regions of the human genome to IBD, i.e., 16q12 (IBD1), 12q13 (IBD2), 6p21 (IBD3), 14q11 (IBD4), 19p13 (IBD5), 5q31-q33 (IBD6) and Xq21.3[10-15] and subsequent research has identified several CD predisposing mutations in the IBD1 gene encoding NOD2[16]. However, the different chromosomal locations found to be associated with IBD in these studies suggest disease heterogeneity: different sets of disease predisposing mutations may lead to a similar clinical outcome. This is corroborated by evidence obtained from animal models, where distinct genetic manipulations, for instance deletion of the DNA encoding TCRα, IL-10 or TNF-α 3’UTR AU repeat motifs, all lead to ileitis and/or colitis[17-19]. Therefore, genes on other loci, not identified in the studies mentioned above, may also contribute to IBD susceptibility and worthy considering in this respect are the matrix metalloproteinases (MMPs) and their natural inhibitors, the tissue inhibitors of metalloproteinases (TIMPs). The MMPs constitute a group of neutral, Ca- and Zn-activated endoproteinases and are involved in physiological matrix turnover during embryogenesis, angiogenesis, etc[20]. Production is tightly regulated at the transcriptional and post-transcriptional levels, and excessive MMP-mediated tissue destruction is prevented by strictly regulated activation mechanisms of the latent pro-enzyme and inhibition of the active enzyme in a 1:1 stoichiometry by TIMPs. Recently, several functional single nucleotide polymorphisms (SNPs) in the genes encoding MMPs and TIMPs have been described. The insertion of an additional guanosine residue at -1607 in the promoter of MMP-1 creates a PEA3 consensus sequence next to an AP-1 binding site up-regulating promoter activity, while the insertion of an additional thymidine at -1613 of the MMP-3 promoter results in decreased mRNA transcription[21,22]. The -1306 C/T transition in the promoter of MMP-2 results in decreased binding affinity for stimulating protein Sp1, leading to decreased mRNA transcription[23]. In contrast, the -1562 C/T transition in the promoter of MMP-9 results in the removal of a binding site for an unknown repressor protein, thus elevating transcription[24]. In TIMP-1 and -2 SNPs have been found in the exon part of the genes (+372 T/C and +303 G/A, respectively). Although no effect on transcriptional activity/mRNA stability was observed, these SNPs might serve as markers in association studies[25]. Both MMP and TIMP expression are affected by TNF-α and this pro-inflammatory cytokine is known to play a pivotal role in IBD, particularly CD but also UC, as demonstrated by impressive clinical improvement following anti-TNF-α antibody infliximab administration[26,27]. The G/A transition at -308 in the TNF-α promoter might result in increased levels of circulating TNF-α protein[28], thus inducing extra MMPs and/or TIMPs. Of note, the gene encoding TNF-α is mapped to the 6p21 IBD3 region, while the MMP and TIMP genes have not been mapped to any known IBD region (MMP-1, -3: 11q22-q23; MMP-2: 16q13; MMP-9: 20q11.2-q13.1; TIMP-1: Xp11.3-p11.23 and TIMP-2: 17q25). Conceivably, direct and indirect SNP-linked overproduction of MMPs and/or down-regulation of TIMPs, would result in net destruction of tissue, impairment of intestinal barrier function, influx of bacteria and consequently excessive immune response, thus predisposing to or worsening IBD. Therefore, we analysed the genotype distributions at these SNP loci of the genes encoding MMP-1, -2, -3, -9, TIMP-1, -2 and TNF-α in CD, UC and controls. Recently, we measured MMP and TIMP protein/activity levels in a large group of resected intestinal IBD tissues (Meijer et al[32], submitted) and here the expression data in a subgroup of which we also had DNA, are correlated to MMP, TIMP and TNF genotypes.
Surgically resected intestinal mucosa from predominantly Dutch Caucasian patients with CD (n = 134, 40% male, median age at surgery 36.3 years, range 11.6-78.7 years) or UC [n = 111, 42% male, 37.8 (15.9-81.9) years], was collected in the period 1983-2002 at the department of Pathology, LUMC and stored at -70°C. The control group consisted of 79 patients with colorectal carcinoma [CRC, macroscopically normal tissue obtained at least 10 cm away from evident neoplasia, 43% male, median age at surgery 56.4 (19.0-85.0) years] and 169 healthy volunteers [37% male, age at blood collection date 33.3 (18.2-72.9) years], recruited among spouses of patients from the out-patient clinic and through advertisement. Informed consent from participants and approval of the LUMC ethics committee for the study protocol was obtained[29]. In both the IBD and control groups, more than 95% of the participants were of Caucasian origin. Resected tissue was homogenised with a Turrax device, blood was centrifuged and genomic DNA was isolated using the salting out method[30] and reconstituted to 10 ng/μL in 0.01 mol/L Tris/0.1 mmol/L EDTA, pH = 7.5. Differential diagnosis of CD or UC was established by routine clinical, radiological and histological findings. Age at onset, localisation at first endoscopy/radiology and development of fistulae and stenotic processes in a subset of CD patients (n = 123) were recorded in medical files. The measurement of myeloperoxidase was according to the procedure described by Kruidenier et al[31], while the MMP and TIMP protein/activity levels in IBD and CRC control tissue were measured previously by our group (Meijer et al[32] and submitted). In brief, homogenates obtained from surgically resected tissue were appropriately diluted. The MMPs and TIMPs antigen levels were measured by ELISAs (MMP-2, -9, TIMP-1, -2) or by highly sensitive bio-immuno activity assays (BIA) involving the conversion of chromogenic peptide S-2444 by MMP-activated pro-urokinase (MMP-1, -3), with all BIAs performed in the presence of APMA to account for total MMP antigen levels. Allelic composition at the SNPs of interest was determined by PCR-RFLP (MMP-1, -3, -9, TIMP-1, -2) or tetra primer ARMS PCR (MMP-2), as described previously[32,33]. Differences between groups were assessed by Chi-square, Kruskal-Wallis or Mann-Whitney U tests, as indicated. Statistical significance was reached if two-tailed P value ≤ 0.05.
Between IBD and controls, no significant differences in genotype distribution were found at -1607 1G/2G and -1306 C/T of MMP-1 and -2 promoters, respectively (Table 1). Also, 1G MMP-1 (55.4 vs 51.4%) and C wild-type (75.5 vs 76.8%) MMP-2 allelic frequencies were similar in both groups. The MMP-3 and MMP-9 genotype distribution at -1613 5T/6T and -1562 C/T, respectively, were also similar. The TIMP-1 gene is located on the X-chromosome, thus the results are presented according to gender. In both men and women the T (T) genotype seems relatively abundant in IBD (men T 61.6 vs 51.6%; women TT 31.2 vs 23.8%) and especially in CD (men T 67.9 vs 51.6%, P = 0.055; women TT 39.0 vs 23.8%, P = 0.018, Table 2). No differences in genotype distribution were observed for TIMP-2 and TNF-α at +303 G/A and -308 G/A, respectively (Table 1). For all SNPs, genotype frequencies in the control group are similar to what was expected from the Hardy-Weinberg equilibrium, except for MMP-2 (CC, CT, TT: 61.9, 29.9, 8.2 observed vs 59.0, 35.7, 5.3% expected, χ2 = 6.36, P < 0.05). Genotype and allelic frequencies for all SNPs examined were similar in CD vs UC and also in the healthy volunteers vs the carcinoma controls. As MMPs and TIMPs are involved in cancer and metastasis, all analyses were repeated with a control group consisting only of the healthy volunteers (n = 169), yielding similar results as mentioned above.
| Protein | SNP | Genotype | IBD(% of patients) | Controls(% of individuals) | Allele | IBD(% of total) | Controls(% of total) |
| MMP-1 | -1607 1G/2G | 1G1G | 31 | 26.6 | 1G | 55.4 | 51.4 |
| 11q22-q23 | 1G2G | 49 | 49.6 | 2G | 44.6 | 48.6 | |
| 2G2G | 20.1 | 23.8 | |||||
| MMP-2 | -1306 C/T | CC | 57.7 | 61.9 | C | 75.5 | 76.8 |
| 16q13 | CT | 35.6 | 29.9 | T | 24.5 | 23.2 | |
| TT | 6.7 | 8.2 | |||||
| MMP-3 | -1613 5T/6T | 5T5T | 29.2 | 27.9 | 5T | 52.3 | 52 |
| 11q23 | 5T6T | 46.3 | 48.4 | 6T | 47.7 | 48 | |
| 6T6T | 24.6 | 23.8 | |||||
| MMP-9 | -1562 C/T | CC | 72.4 | 69.7 | C | 85.6 | 84 |
| 20q11.2-q13.1 | CT + TT | 27.6 | 30.3 | T | 14.4 | 16 | |
| ♂, TIMP-11 | +372 T/C | T | 61.6 | 51.6 | T | 61.6 | 51.6 |
| Xp11.3-p11.23 | C | 38.4 | 48.4 | C | 38.4 | 48.4 | |
| ♀, TIMP-11 | +372 T/C | TT | 31.2 | 23.8 | T | 52.8 | 50.7 |
| Xp11.3-p11.23 | TC | 43.3 | 53.6 | C | 47.2 | 49.3 | |
| CC | 25.5 | 22.5 | |||||
| TIMP-2 | +303 G/A | GG | 77.5 | 78.7 | G | 88.3 | 89.1 |
| 17q25 | GA + AA | 22.5 | 21.3 | A | 11.7 | 10.9 | |
| TNF-α | -308 G/A | GG | 68.8 | 69.7 | G | 82.0 | 83.0 |
| 6p21 | GA + AA | 31.4 | 30.3 | A | 18.0 | 17.0 |
| Protein | Genotype | CD (% of patients) | Controls (% of individuals) | P1 | Allele | CD (% of total) | Controls (% of total) | P-value |
| TIMP-1, ♂ | T | 67.9 | 51.6 | 0.055 | T | 67.9 | 51.6 | 0.055 |
| C | 32.1 | 48.4 | C | 32.1 | 48.4 | |||
| TIMP-1, ♀ | TT | 39 | 23.8 | 0.018 | T | 56.5 | 50.7 | 0.238 |
| TC | 35.1 | 53.6 | C | 43.5 | 49.3 | |||
| CC | 26 | 22.5 |
Effect of MMP and TNF-αSNPs on CD phenotype
The median age at onset of disease in 123 CD patients with a full medical record was 21.5 (range 0.3-61.5) years. Patients stratified according to genotype at the SNPs examined had similar ages at onset (Table 3). At first endoscopic/radiologic examination, in 53.3% of the patients colonic w/wo ileal involvement was evident. The MMP-3 genotype was associated with disease localisation (P = 0.04 for all three groups) and further analysis revealed a lower chance of colonic involvement at first endoscopy/radiology in patients with the 5T5T MMP-3 genotype (P = 0.017, 5T5T vs 5T6T and 6T6T combined). However, this genotype also conferred a major risk to development of stenotic complications: 91.2% of patients carrying the 5T5T genotype suffered from stenotic complications compared to 71.8% for the other genotypes (P = 0.022). The allelic polymorphisms at other SNP loci were not associated with disease localisation or stricture involvement. Of all CD patients, 80/123 or 65.0% developed peri-anal, entero-entero or entero-cutaneous fistulae during follow-up. Female patients with the TT genotype at +372 SNP of TIMP-1 appeared less prone to develop fistulae (P = 0.08), and when the TT group was compared with the combined TC and CC group, this result was statistically significant (P = 0.025).
| Protein | SNP | Genotype | Number ofpatients | Age (yr) at onsetof disease,median (range) | Colonic involvementat first examination(% of patients) | Fistulae duringfollow-up(% of patients) | Strictures duringfollow-up(% of patients) |
| MMP-1 | -1607 1G/2G | 1G1G | 38 | 20.7 (0.3-45.6) | 52.6 | 71.1 | 84.2 |
| 1G2G | 54 | 22.4 (7.0-61.5) | 50 | 63 | 75.9 | ||
| 2G2G | 26-27 | 21.0 (3.4-40.2) | 57.7 | 63 | 70.4 | ||
| MMP-2 | -1306 C/T | CC | 66-67 | 20.1 (5.8-61.5) | 57.6 | 64.2 | 74.6 |
| CT+TT | 521 | 22.4 (0.3-50.1) | 46.2 | 67.3 | 80.8 | ||
| MMP-3 | -1613 5T/6T | 5T5T | 34 | 22.4 (0.3-53.6) | 35.3a | 70.6 | 91.2a |
| 5T6T | 54 | 19.3 (3.4-61.5) | 63 | 66.7 | 74.1 | ||
| 6T6T | 30-31 | 23.5 (7.2-49.1) | 53.3 | 58.1 | 67.7 | ||
| MMP-9 | -1562 C/T | CC | 88-89 | 21.0 (0.3-61.5) | 53.4 | 68.5 | 75.3 |
| CT+TT | 301 | 22.7 (7.2-45.5) | 50 | 56.7 | 83.3 | ||
| TIMP-1, ♂ | +372 T/C | T | 32 | 18.9 (0.3-48.3) | 53.1 | 78.1 | 87.5 |
| C | 17 | 18.5 (5.9-61.5) | 52.9 | 76.5 | 94.1 | ||
| TIMP-1, ♀ | +372 T/C | TT | 29 | 24.3 (7.0-56.8) | 55.2 | 41.4c | 69.0 |
| TC | 22-23 | 23.5 (3.4-39.6) | 45.5 | 69.6 | 78.3 | ||
| CC | 18 | 22.2 (10.4-49.1) | 55.6 | 66.7 | 55.6 | ||
| TIMP-2 | +303 G/A | GG | 91-92 | 21.5 (0.3-61.5) | 56.0 | 68.5 | 77.2 |
| GA+AA | 271 | 22.4 (3.4-56.8) | 40.7 | 55.6 | 77.8 | ||
| TNF-α | -308 G/A | GG | 79-80 | 22.6 (0.3-61.5) | 50.6 | 68.8 | 78.8 |
| GA+AA | 391 | 18.9 (3.4-45.5) | 56.4 | 59.0 | 74.4 |
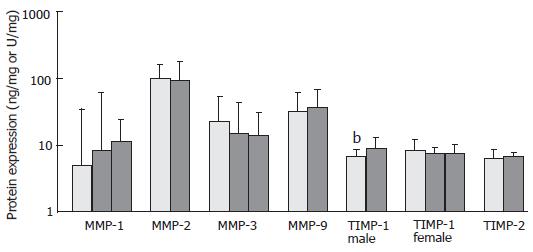
Male IBD patients carrying the T allele at SNP +372 expressed lower levels of TIMP-1 in inflamed tissue compared to those carrying the C allele, P = 0.009 (Figure 1), with similar MPO levels in both groups [median 24.2 (range 9.1-80.4) vs 28.6 (2.5-75.9) U/g, P = 0.194]. In male CD patients a similar pattern in TIMP-1 expression was observed [6.8 (1.7-18.6) vs 9.2 (1.8-19.9) ng/mg TIMP-1, n = 46 vs 19, T vs C allele, respectively], although not longer statistically significant (P = 0.065). However, female IBD or CD patients carrying the TT, TC or CC genotype expressed similar levels of TIMP-1 in inflamed tissue. The respective protein expression was not affected by genotype at other MMP and TIMP SNPs in inflamed intestinal tissue. In non-inflamed IBD and control CRC tissue, no differences in protein levels were observed between patients stratified to genotype. Finally, allelic composition at TNF-α-308 G/A was not associated with higher or lower levels of MMPs or TIMPs in inflamed and non-inflamed IBD or control tissue.
In this study we found increased susceptibility to CD in men and women carrying the T and TT genotype, respectively, at TIMP-1 SNP +372. The X chromosome region p11.3-p11.23 might thus represent a novel linkage marker in IBD, extending the results obtained in previous genome-wide linkage studies[14,34]. Women with this genotype also appear less prone to the development of fistulae. The direct or indirect involvement of the X-chromosome in CD etiopathogenesis is further corroborated by a higher incidence of CD in women compared to men[35], the association of CD with X-linked Turner’s syndrome[8] and the higher incidence of extra-intestinal complications and surgery recurrence rates in female compared to male CD patients[36]. Importantly, in men the T allele at SNP +372 was accompanied with a lower TIMP-1 protein expression in inflamed tissue. The lower TIMP-1 protein levels relative to MMP in susceptible individuals might shift the balance to a more proteolytic mucosal Crohn’s disease phenotype. The TIMP-1 SNP might also be linked to other markers on the X-chromosome increasing CD susceptibility and conferring protection against fistulae pathogenesis thus explaining the observed results in women. We observed no association between allelic composition at MMP-3 SNP-1613 and susceptibility to IBD. Our findings in UC confirm previous publications on primary sclerosing cholangitis and UC[22,37], but those on CD are different from the results obtained by the group of Pender et al[38], who noted increased susceptibility to sporadic, but not familial CD in individuals carrying the 5T allele. These contrasting results might arise from a different proportion of sporadic versus familial cases in our study. We also found a decreased chance of colonic involvement at first endoscopic/radiologic examination and a higher incidence of stenotic complications in patients carrying the 5T5T MMP-3 genotype at SNP-1613. Previously, over transmission of the 5T allele was associated with ileal localisation and stenosis in CD CARD15 mutation carriers[38] and the group of Warnaar et al[39] reported increased levels of MMP-3 in stenotic and pre-stenotic resected CD ileum, pointing to an MMP-3 mediated altered clinical course of CD patients by an, as yet, unidentified mechanism. The 5T5T genotype was reported to both increase[40,41] and decrease[42] MMP-3 protein expression, but in our study patients stratified according to MMP-3 genotype expressed similar MMP-3 total activity. Previously, the A allele at TNF-α SNP-308 was reported to increase susceptibility to UC[43], CD[44] and the incidence of fistulae in CD[45], possibly mediated by an increased promoter activity[46,47]. In contrast, we found no effect of allelic composition at this SNP on disease risk and phenotype, in line with other reports[48,49], adding further complexity to this matter. As mentioned before, the patient populations might differ dependent on the genetic (ethnic) background, thus explaining the contrasting results. We could not demonstrate an association of MMP-1, -2, -9 and TIMP-2 SNPs with disease susceptibility or clinical course of disease, in line with previous (genome-wide) linkage reports[11-13,37,50]. As other studies have clearly shown the involvement of these proteins in IBD pathology[51,52], it seems that they primarily function as mediators/effectors instead of initiators during IBD etiopathogenesis. However, the regulation of these proteins by immuno-suppressive medication, such as infliximab, might be dependent on the allelic composition at the SNPs examined, as previously shown by ex vivo explant studies from our group[32]. In principle, enhanced MMP expression might also be associated with SNPs in other genes, for instance with those encoding cytokines regulating MMP expression, e.g., IL-1β[44] and TNF-α. Dependent on the presence of relevant cis-acting elements in the promoter sequence, especially MMP-1 and MMP-9 would be affected[53,54], but we found no effect.
In summary, several studies reported associations between SNPs in diverse genes and IBD[16,48,55-59]. We have focused on the SNPs in genes coding for matrix remodeling proteins, i.e., MMPs and TIMPs, and believe the T allele at SNP +372 T/C in TIMP-1 might be involved in CD susceptibility in both sexes and in men by down-regulating TIMP-1 expression, while the 5T5T genotype at MMP-3-1613 might protect for colonic disease localization but also confers a major risk to stenotic complications. These findings reinforce the potential role of MMP and TIMPs in IBD and should be confirmed in larger prospective follow-up studies.
The technical assistance of M van den Berg, J J van der Reijden and W van Duijn is greatly appreciated.
S- Editor Liu Y L- Editor Alpini GD E- Editor Lu W
| 1. | Farrell RJ, Peppercorn MA. Ulcerative colitis. Lancet. 2002;359:331-340. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 207] [Cited by in RCA: 186] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 2. | Shanahan F. Crohn's disease. Lancet. 2002;359:62-69. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 358] [Cited by in RCA: 353] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 3. | Loftus EV, Silverstein MD, Sandborn WJ, Tremaine WJ, Harmsen WS, Zinsmeister AR. Crohn's disease in Olmsted County, Minnesota, 1940-1993: incidence, prevalence, and survival. Gastroenterology. 1998;114:1161-1168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 433] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 4. | Loftus EV, Silverstein MD, Sandborn WJ, Tremaine WJ, Harmsen WS, Zinsmeister AR. Ulcerative colitis in Olmsted County, Minnesota, 1940-1993: incidence, prevalence, and survival. Gut. 2000;46:336-343. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 360] [Cited by in RCA: 360] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 5. | Tysk C, Lindberg E, Järnerot G, Flodérus-Myrhed B. Ulcerative colitis and Crohn's disease in an unselected population of monozygotic and dizygotic twins. A study of heritability and the influence of smoking. Gut. 1988;29:990-996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 543] [Cited by in RCA: 518] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 6. | Kurata JH, Kantor-Fish S, Frankl H, Godby P, Vadheim CM. Crohn's disease among ethnic groups in a large health maintenance organization. Gastroenterology. 1992;102:1940-1948. [PubMed] |
| 7. | Russel MG, Pastoor CJ, Janssen KM, van Deursen CT, Muris JW, van Wijlick EH, Stockbrügger RW. Familial aggregation of inflammatory bowel disease: a population-based study in South Limburg, The Netherlands. The South Limburg IBD Study Group. Scand J Gastroenterol Suppl. 1997;223:88-91. [PubMed] |
| 8. | Arulanantham K, Kramer MS, Gryboski JD. The association of inflammatory bowel disease and X chromosomal abnormality. Pediatrics. 1980;66:63-67. [PubMed] |
| 9. | Gahl WA, Brantly M, Kaiser-Kupfer MI, Iwata F, Hazelwood S, Shotelersuk V, Duffy LF, Kuehl EM, Troendle J, Bernardini I. Genetic defects and clinical characteristics of patients with a form of oculocutaneous albinism (Hermansky-Pudlak syndrome). N Engl J Med. 1998;338:1258-1264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 196] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 10. | Hugot JP, Laurent-Puig P, Gower-Rousseau C, Olson JM, Lee JC, Beaugerie L, Naom I, Dupas JL, Van Gossum A, Orholm M. Mapping of a susceptibility locus for Crohn's disease on chromosome 16. Nature. 1996;379:821-823. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 649] [Cited by in RCA: 598] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 11. | Ma Y, Ohmen JD, Li Z, Bentley LG, McElree C, Pressman S, Targan SR, Fischel-Ghodsian N, Rotter JI, Yang H. A genome-wide search identifies potential new susceptibility loci for Crohn's disease. Inflamm Bowel Dis. 1999;5:271-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 110] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 12. | Rioux JD, Silverberg MS, Daly MJ, Steinhart AH, McLeod RS, Griffiths AM, Green T, Brettin TS, Stone V, Bull SB. Genomewide search in Canadian families with inflammatory bowel disease reveals two novel susceptibility loci. Am J Hum Genet. 2000;66:1863-1870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 348] [Cited by in RCA: 361] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 13. | Satsangi J, Parkes M, Louis E, Hashimoto L, Kato N, Welsh K, Terwilliger JD, Lathrop GM, Bell JI, Jewell DP. Two stage genome-wide search in inflammatory bowel disease provides evidence for susceptibility loci on chromosomes 3, 7 and 12. Nat Genet. 1996;14:199-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 476] [Cited by in RCA: 458] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 14. | Vermeire S, Satsangi J, Peeters M, Parkes M, Jewell DP, Vlietinck R, Rutgeerts P. Evidence for inflammatory bowel disease of a susceptibility locus on the X chromosome. Gastroenterology. 2001;120:834-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Yang H, Plevy SE, Taylor K, Tyan D, Fischel-Ghodsian N, McElree C, Targan SR, Rotter JI. Linkage of Crohn's disease to the major histocompatibility complex region is detected by multiple non-parametric analyses. Gut. 1999;44:519-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 75] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 16. | Hugot JP, Chamaillard M, Zouali H, Lesage S, Cézard JP, Belaiche J, Almer S, Tysk C, O'Morain CA, Gassull M. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn's disease. Nature. 2001;411:599-603. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4223] [Cited by in RCA: 3901] [Article Influence: 162.5] [Reference Citation Analysis (0)] |
| 17. | Kontoyiannis D, Pasparakis M, Pizarro TT, Cominelli F, Kollias G. Impaired on/off regulation of TNF biosynthesis in mice lacking TNF AU-rich elements: implications for joint and gut-associated immunopathologies. Immunity. 1999;10:387-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1030] [Cited by in RCA: 1054] [Article Influence: 40.5] [Reference Citation Analysis (0)] |
| 18. | Kühn R, Löhler J, Rennick D, Rajewsky K, Müller W. Interleukin-10-deficient mice develop chronic enterocolitis. Cell. 1993;75:263-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3189] [Cited by in RCA: 3224] [Article Influence: 100.8] [Reference Citation Analysis (0)] |
| 19. | Mombaerts P, Mizoguchi E, Grusby MJ, Glimcher LH, Bhan AK, Tonegawa S. Spontaneous development of inflammatory bowel disease in T cell receptor mutant mice. Cell. 1993;75:274-282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 524] [Cited by in RCA: 505] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 20. | Visse R, Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res. 2003;92:827-839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3186] [Cited by in RCA: 3278] [Article Influence: 149.0] [Reference Citation Analysis (0)] |
| 21. | Fujimoto T, Parry S, Urbanek M, Sammel M, Macones G, Kuivaniemi H, Romero R, Strauss JF. A single nucleotide polymorphism in the matrix metalloproteinase-1 (MMP-1) promoter influences amnion cell MMP-1 expression and risk for preterm premature rupture of the fetal membranes. J Biol Chem. 2002;277:6296-6302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 114] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 22. | Satsangi J, Chapman RW, Haldar N, Donaldson P, Mitchell S, Simmons J, Norris S, Marshall SE, Bell JI, Jewell DP. A functional polymorphism of the stromelysin gene (MMP-3) influences susceptibility to primary sclerosing cholangitis. Gastroenterology. 2001;121:124-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 61] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 23. | Price SJ, Greaves DR, Watkins H. Identification of novel, functional genetic variants in the human matrix metalloproteinase-2 gene: role of Sp1 in allele-specific transcriptional regulation. J Biol Chem. 2001;276:7549-7558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 294] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 24. | Zhang B, Ye S, Herrmann SM, Eriksson P, de Maat M, Evans A, Arveiler D, Luc G, Cambien F, Hamsten A. Functional polymorphism in the regulatory region of gelatinase B gene in relation to severity of coronary atherosclerosis. Circulation. 1999;99:1788-1794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 418] [Cited by in RCA: 421] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 25. | Krex D, Röhl H, König IR, Ziegler A, Schackert HK, Schackert G. Tissue inhibitor of metalloproteinases-1, -2, and -3 polymorphisms in a white population with intracranial aneurysms. Stroke. 2003;34:2817-2821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | Present DH, Rutgeerts P, Targan S, Hanauer SB, Mayer L, van Hogezand RA, Podolsky DK, Sands BE, Braakman T, DeWoody KL. Infliximab for the treatment of fistulas in patients with Crohn's disease. N Engl J Med. 1999;340:1398-1405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1969] [Cited by in RCA: 1838] [Article Influence: 70.7] [Reference Citation Analysis (0)] |
| 27. | Targan SR, Hanauer SB, van Deventer SJ, Mayer L, Present DH, Braakman T, DeWoody KL, Schaible TF, Rutgeerts PJ. A short-term study of chimeric monoclonal antibody cA2 to tumor necrosis factor alpha for Crohn's disease. Crohn's Disease cA2 Study Group. N Engl J Med. 1997;337:1029-1035. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2328] [Cited by in RCA: 2266] [Article Influence: 80.9] [Reference Citation Analysis (0)] |
| 28. | Allen RD. Polymorphism of the human TNF-alpha promoter--random variation or functional diversity? Mol Immunol. 1999;36:1017-1027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 157] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 29. | van der Veek PP, van den Berg M, de Kroon YE, Verspaget HW, Masclee AA. Role of tumor necrosis factor-alpha and interleukin-10 gene polymorphisms in irritable bowel syndrome. Am J Gastroenterol. 2005;100:2510-2516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 146] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 30. | Miller SA, Dykes DD, Polesky HF. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988;16:1215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13387] [Cited by in RCA: 14446] [Article Influence: 390.4] [Reference Citation Analysis (0)] |
| 31. | Kruidenier L, Kuiper I, Van Duijn W, Mieremet-Ooms MA, van Hogezand RA, Lamers CB, Verspaget HW. Imbalanced secondary mucosal antioxidant response in inflammatory bowel disease. J Pathol. 2003;201:17-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 166] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 32. | Meijer MJ, Mieremet-Ooms MA, van Duijn W, van der Zon AM, Hanemaaijer R, Verheijen JH, van Hogezand RA, Lamers CB, Verspaget HW. Effect of the anti-tumor necrosis factor-alpha antibody infliximab on the ex vivo mucosal matrix metalloproteinase-proteolytic phenotype in inflammatory bowel disease. Inflamm Bowel Dis. 2007;13:200-210. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 54] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 33. | Kubben FJ, Sier CF, Meijer MJ, van den Berg M, van der Reijden JJ, Griffioen G, van de Velde CJ, Lamers CB, Verspaget HW. Clinical impact of MMP and TIMP gene polymorphisms in gastric cancer. Br J Cancer. 2006;95:744-751. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 85] [Cited by in RCA: 92] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 34. | Hampe J, Schreiber S, Shaw SH, Lau KF, Bridger S, Macpherson AJ, Cardon LR, Sakul H, Harris TJ, Buckler A. A genomewide analysis provides evidence for novel linkages in inflammatory bowel disease in a large European cohort. Am J Hum Genet. 1999;64:808-816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 251] [Cited by in RCA: 258] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 35. | Gollop JH, Phillips SF, Melton LJ, Zinsmeister AR. Epidemiologic aspects of Crohn's disease: a population based study in Olmsted County, Minnesota, 1943-1982. Gut. 1988;29:49-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 176] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 36. | Wagtmans MJ, Verspaget HW, Lamers CB, van Hogezand RA. Gender-related differences in the clinical course of Crohn's disease. Am J Gastroenterol. 2001;96:1541-1546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 60] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 37. | Wiencke K, Louka AS, Spurkland A, Vatn M, Schrumpf E, Boberg KM. Association of matrix metalloproteinase-1 and -3 promoter polymorphisms with clinical subsets of Norwegian primary sclerosing cholangitis patients. J Hepatol. 2004;41:209-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 38. | Pender SL, Croucher PJ, Mascheretti S, Prothero JD, Fisher SA, MacDonald TT, Schreiber S, Ye S. Transmission disequilibrium test of stromelysin-1 gene variation in relation to Crohn's disease. J Med Genet. 2004;41:e112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 39. | Warnaar N, Hofker HS, Maathuis MH, Niesing J, Bruggink AH, Dijkstra G, Ploeg RJ, Schuurs TA. Matrix metalloproteinases as profibrotic factors in terminal ileum in Crohn's disease. Inflamm Bowel Dis. 2006;12:863-869. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 40. | Borghaei RC, Rawlings PL, Javadi M, Woloshin J. NF-kappaB binds to a polymorphic repressor element in the MMP-3 promoter. Biochem Biophys Res Commun. 2004;316:182-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 56] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 41. | Medley TL, Kingwell BA, Gatzka CD, Pillay P, Cole TJ. Matrix metalloproteinase-3 genotype contributes to age-related aortic stiffening through modulation of gene and protein expression. Circ Res. 2003;92:1254-1261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 121] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 42. | Samnegård A, Silveira A, Lundman P, Boquist S, Odeberg J, Hulthe J, McPheat W, Tornvall P, Bergstrand L, Ericsson CG. Serum matrix metalloproteinase-3 concentration is influenced by MMP-3 -1612 5A/6A promoter genotype and associated with myocardial infarction. J Intern Med. 2005;258:411-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 94] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 43. | Sashio H, Tamura K, Ito R, Yamamoto Y, Bamba H, Kosaka T, Fukui S, Sawada K, Fukuda Y, Tamura K. Polymorphisms of the TNF gene and the TNF receptor superfamily member 1B gene are associated with susceptibility to ulcerative colitis and Crohn's disease, respectively. Immunogenetics. 2002;53:1020-1027. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 112] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 44. | Ferreira AC, Almeida S, Tavares M, Canedo P, Pereira F, Regalo G, Figueiredo C, Trindade E, Seruca R, Carneiro F. NOD2/CARD15 and TNFA, but not IL1B and IL1RN, are associated with Crohn's disease. Inflamm Bowel Dis. 2005;11:331-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 45. | Louis E, Peeters M, Franchimont D, Seidel L, Fontaine F, Demolin G, Croes F, Dupont P, Davin L, Omri S. Tumour necrosis factor (TNF) gene polymorphism in Crohn's disease (CD): influence on disease behaviour? Clin Exp Immunol. 2000;119:64-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 67] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 46. | Bouma G, Crusius JB, Oudkerk Pool M, Kolkman JJ, von Blomberg BM, Kostense PJ, Giphart MJ, Schreuder GM, Meuwissen SG, Peña AS. Secretion of tumour necrosis factor alpha and lymphotoxin alpha in relation to polymorphisms in the TNF genes and HLA-DR alleles. Relevance for inflammatory bowel disease. Scand J Immunol. 1996;43:456-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 292] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 47. | Wilson AG, Symons JA, McDowell TL, McDevitt HO, Duff GW. Effects of a polymorphism in the human tumor necrosis factor alpha promoter on transcriptional activation. Proc Natl Acad Sci USA. 1997;94:3195-3199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1608] [Cited by in RCA: 1700] [Article Influence: 60.7] [Reference Citation Analysis (0)] |
| 48. | Cantor MJ, Nickerson P, Bernstein CN. The role of cytokine gene polymorphisms in determining disease susceptibility and phenotype in inflammatory bowel disease. Am J Gastroenterol. 2005;100:1134-1142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 66] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 49. | Zipperlen K, Peddle L, Melay B, Hefferton D, Rahman P. Association of TNF-alpha polymorphisms in Crohn disease. Hum Immunol. 2005;66:56-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 50. | Nohara H, Saito Y, Higaki S, Okayama N, Hamanaka Y, Okita K, Hinoda Y. Polymorphisms of the IL-1beta and IL-1beta-inducible genes in ulcerative colitis. J Gastroenterol. 2002;37 Suppl 14:107-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 51. | Baugh MD, Perry MJ, Hollander AP, Davies DR, Cross SS, Lobo AJ, Taylor CJ, Evans GS. Matrix metalloproteinase levels are elevated in inflammatory bowel disease. Gastroenterology. 1999;117:814-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 255] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 52. | von Lampe B, Barthel B, Coupland SE, Riecken EO, Rosewicz S. Differential expression of matrix metalloproteinases and their tissue inhibitors in colon mucosa of patients with inflammatory bowel disease. Gut. 2000;47:63-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 255] [Cited by in RCA: 290] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 53. | Huhtala P, Tuuttila A, Chow LT, Lohi J, Keski-Oja J, Tryggvason K. Complete structure of the human gene for 92-kDa type IV collagenase. Divergent regulation of expression for the 92- and 72-kilodalton enzyme genes in HT-1080 cells. J Biol Chem. 1991;266:16485-16490. [PubMed] |
| 54. | Rutter JL, Benbow U, Coon CI, Brinckerhoff CE. Cell-type specific regulation of human interstitial collagenase-1 gene expression by interleukin-1 beta (IL-1 beta) in human fibroblasts and BC-8701 breast cancer cells. J Cell Biochem. 1997;66:322-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 55. | Moehle C, Ackermann N, Langmann T, Aslanidis C, Kel A, Kel-Margoulis O, Schmitz-Madry A, Zahn A, Stremmel W, Schmitz G. Aberrant intestinal expression and allelic variants of mucin genes associated with inflammatory bowel disease. J Mol Med (Berl). 2006;84:1055-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 133] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 56. | Brand S, Hofbauer K, Dambacher J, Schnitzler F, Staudinger T, Pfennig S, Seiderer J, Tillack C, Konrad A, Göke B. Increased expression of the chemokine fractalkine in Crohn's disease and association of the fractalkine receptor T280M polymorphism with a fibrostenosing disease Phenotype. Am J Gastroenterol. 2006;101:99-106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 81] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 57. | Pierik M, Joossens S, Van Steen K, Van Schuerbeek N, Vlietinck R, Rutgeerts P, Vermeire S. Toll-like receptor-1, -2, and -6 polymorphisms influence disease extension in inflammatory bowel diseases. Inflamm Bowel Dis. 2006;12:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 219] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 58. | Borm ME, van Bodegraven AA, Mulder CJ, Kraal G, Bouma G. A NFKB1 promoter polymorphism is involved in susceptibility to ulcerative colitis. Int J Immunogenet. 2005;32:401-405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 59. | Xia B, Crusius JB, Wu J, Zwiers A, van Bodegraven AA, Peña AS. Signal transducer and activator of transcription 6 gene G2964A polymorphism and inflammatory bowel disease. Clin Exp Immunol. 2003;131:446-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.1] [Reference Citation Analysis (0)] |