Published online May 21, 2007. doi: 10.3748/wjg.v13.i19.2743
Revised: January 15, 2007
Accepted: January 31, 2007
Published online: May 21, 2007
AIM: To investigate the anticancer activity of a chinese medical mixture, WRCP (warming and relieving Cold Phlegm), on hepatocarcinoma Bel-7402 cells.
METHODS: Fingerprints of WRCP, which were composed of aqueous extracts of Aconitum carmichaeli, Rhizoma bolbostemmatis, Phytolacca acinosa, Panax notoginseng and Gekko swinhonis Gūenther, and aconitine, which could be isolated from Aconitum carmichaeli and have the potential toxicity, were identified by high pressure liquid chromatography. Bel-7402 cells were grown in the presence of WRCP, As2O3 or all-trans-retinoic acid (ATRA). Cell proliferation and viability were determined by trypan blue stain. Apoptosis and cell cycle of Bel-7402 cells were detected by flow cytometry. Morphologic and ultrastructural variations were determined under optic and electronic microscopy. The secretion of alpha-fetoprotein and albumin was detected by radioimmunoassay.
RESULTS: The average quality of aconitine is 1.15 ± 0.10 μg per 7.5 g extracts. WRCP could suppress the proliferation and viability of Bel-7402 cells. The percentage of apoptosis cells and S phase cells increased on WRCP-treated cells. Treated with WRCP, Bel-7402 cells showed ultrastructural features of differentiation. The alpha-fetoprotein secretion decreased while the albumin secretion increased (P < 0.001, P < 0.001, respectively) markedly in WRCP-treated cells.
CONCLUSION: WRCP can affect the proliferation, differentiation and apoptosis of Bel-7402 cells. It can arrest cells in S phase and has strong cytotoxicity to Bel-7402 cells.
-
Citation: Yan ZC, Chen D, Wu XZ, Xie GR, Ba Y, Yan Z. Effects of aqueous extracts of
Aconitum carmichaeli, Rhizoma bolbostemmatis, Phytolacca acinosa, Panax notoginseng andGekko swinhonis Gūenther on Bel-7402 cells. World J Gastroenterol 2007; 13(19): 2743-2746 - URL: https://www.wjgnet.com/1007-9327/full/v13/i19/2743.htm
- DOI: https://dx.doi.org/10.3748/wjg.v13.i19.2743
Hepatocellular carcinoma (HCC) is one of the most common malignant tumors worldwide, with an annual incidence of approximately one million cases[1,2]. Despite the high mortality and frequency of this cancer, surgical resection is an option for only a small proportion of patients, because metastases are often present when the cancer is discovered. Because HCC is most resistant to medicinal treatment, chemotherapy adds little to overall survival of the HCC patients[3,4]. Tumor is a cell cycle disease that has the abnormal interface of high proliferation, low differentiation and apoptosis block. A Chinese medical mixture, WRCP (warming and relieving Cold Phlegm), was composed of the aqueous extracts of Aconitum carmichaeli, Rhizoma bolbostemmatis, Phytolacca acinosa, Panax notoginseng and Gekko swinhonis Gūenther. This study aims to investigate the anticancer activity of WRCP by regulating tumor biological action and its cytotoxic activity on hepatocarcinoma Bel-7402 cells.
Plants and animal were obtained from Sichuan Chinese Herb Company, Flora Manufacturing and Distributing Ltd., Chengdu, Sichuan, China. The plants and animal were identified by Prof. Duan from the Department of Basic Traditional Chinese Medicine, Chengdu University of Traditional Chinese Medicine. All voucher specimens have been deposited in the Center of Integrative Oncology, Tianjin Medical University Cancer Institute and Hospital.
The following air-dried and powdered plants and animal species were studied: rhizomes of Aconitum carmichaeli, Rhizoma bolbostemmatis, Phytolacca acinosa, Panax notoginseng and whole animal of Gekko swinhonis Gūenther. After being dried and crushed, individual samples of each part utilized were percolated with hot distilled water twice at 90°C-95°C for 1 h, and the solutions were combined in vacuo to obtain residues. The powder was then dissolved in double distilled water (DD water) to prepare the stock solution at 75 g/L extract concentration and stored at -20°C. It was further diluted with RPMI 1640 medium to obtain 250 mg/L and 75 mg/L of working solution.
Hepatocarcinoma Bel-7402 cells were cultured in RPMI 1640 medium (Gibco, USA) supplemented with 100 mL/L fetal bovine serum (Hani, Chengdu), 100 g/L penicillin and 100 mg/mL streptomycin were used at 37°C in a humidified 50 mL/L CO2 incubator. As2O3 (Sigma, USA) was kept in sterile 1 × PBS as a 10 mmol/L stock solution and all-trans-retinoic acid (ATRA; Sigma, USA) was kept in 990 mL/L alcohol as a 1 g/L stock solution.
High pressure liquid chromatography (HPLC) was carried out on an LC-6A HPLC system (Shimabzu, Ltd. Japan) using a C18 column (4.6 mm × 250 mm, Kromaisl, Ltd., Sweden), and the mobile phase was a mixture of water and acetonitrile (containing 3 mL/L triethylamine) (65: 35, V/V) in 5.0 column volumes at a flow rate of 1.0 mL/min. The detection wavelength was set at 235 nm.
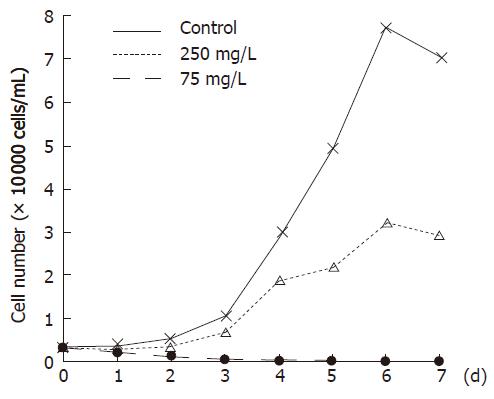
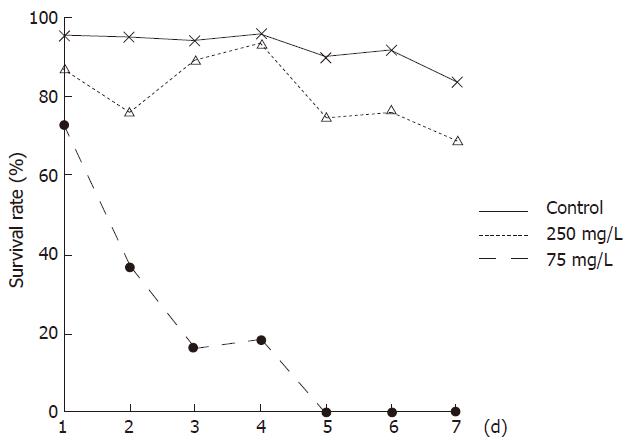
Bel-7402 cells were seeded in 12-well plates (0.3 × 101 cells/L) and allowed to grow for 24 h before being exposed to 250 mg/L and 75 mg/L concentrations of WRCP, respectively. For the negative control group, an equal volume of sterile 1 × PBS was added. Cells were collected respectively at 0.5, 24, 48, 72, 96, 120 and 144 h after drugs were added. The cells were then trypan blue stained and an hemocytometer was used to determine the total cell count and viable cell number. The growth curve of Bel-7402 cells was marked. Viability of cells were computed as follows: viability (%) = viable cell number /total cell count × 100%.
Bel-7402 cells were cultured in 6-well plates (0.5 × 101 cells/L and allowed to grow for 24 h before being exposed to 250 mg/L and 75 mg/L concentrations of WRCP respectively. As2O3 at a concentration of 0.1 mg/L was added in positive control group and an equal volume of sterile 1 × PBS was added in negative control group. Cells were collected at d 4 after drug was added and fixed in 700 mL/L ice-cold EtOH/PBS for 20 min on ice, and then washed with PBS and incubated in propidium iodide (PI) solution (69 mmol/L PI, 388 mmol/L sodium citrate, 100 mg/L RNase A) for 15 min at 37°C. Then cell DNA was analyzed by flow cytometry (Becton Dickinson Com, USA). Raw data for the distribution of DNA content were expressed as the percentage of G0/G1 through G2/M populations. Multicycle AV software (Phoenix Flow Systems, San Diago, California, USA) was used for data analysis.
Bel-7402 cells were seeded in 50 mL bottle (0.4 × 102 cells/L) and allowed to grow for 24 h before being exposed to 250 mg/L and 75 mg/L concentrations of WRCP respectively. ATRA at a concentration of 3 mg/L was added in positive control group and an equal volume of sterile 1 × PBS was added in negative control group. The morphologic variations of Bel-7402 cells were observed under optical microscope at d 4 after adding drugs. In succession, cells were harvested and placed immediately in 25 g/L glutaraldehyde, then in 10 g/L OsO4, dehydrated in ethanol series, and replaced in propene oxide. The samples were examined under transmission electronic microscope.
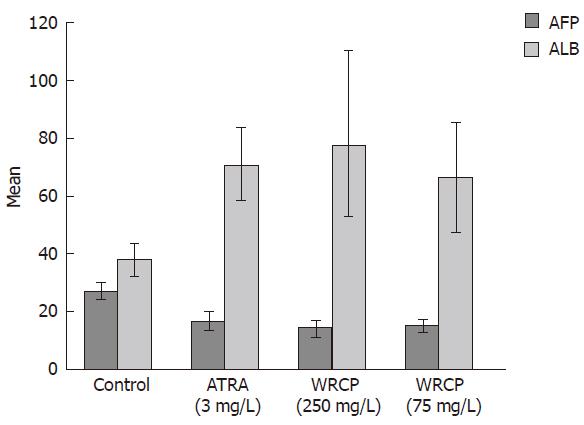
Bel-7402 cells were seeded in 50 mL bottle (2.5 × 102 cells/L) and allowed to grow for 24 h before being exposed to 250 mg/L and 75 mg/L concentrations of WRCP, respectively. ATRA at a concentration of 3 mg/L was added in positive control group and an equal volume of sterile 1 × PBS was added in negative control group. At d 4 after treatment with drugs, culture medium was harvested respectively. AFP and Alb were determined by double-antibody radioimmunoassay (RIA).
Analysis of variance (ANOVA and General Linear Model) was used to determine whether there were significant differences among these groups. Statistical analysis was performed by SPSS (Version: 13.0, Chicago, USA).
Aconitine, which could be isolated from Aconitum carmichaeli and is potentially toxic[5], was identified as “Aco peak” in chromatogram. The average quality of aconitine was 1.15 ± 0.10 μg per 7.5 g extracts (max dose of one day). Bel-7402 cells all died after treatment with WRCP at a concentration of 250 mg/L for 96 h. Even at a concentration of 75 mg/L, WRCP could strongly inhibit the proliferation and viability of Bel-7402 cells. The inhibitory ratio of cell growth at a concentration of 75 mg/L was more than 50% (Figures 1 and 2).
The percentage of apoptosis cells in the negative control group was 8.9%, and 22.5% in As2O3-treated group. However, the percentages of apoptosic cells in WRCP-treated group (250 mg/L and 75 mg/L) were 35.9% and 19.9%, respectively. Furthermore, concentrated chromatins were detected under electronic microscope. The percentages of G0/G1 phase cells, S phase cells and G2/M phase cells were 57.9%, 28.6% and 13.6% respectively in negative control group. The G2/M phase cells achieved 16.1% in As2O3-treated group, whereas the percentage of S phase cells increased and the G0/G1 phase cells decreased in WRCP-treated group. In the group treated with WRCP at a concentration of 250 mg/L, the percentages of G0/G1 phase cells and S phase cells were 49.4% and 37.8%, respectively.
Bel-7402 cells showed abnormal features with a round shape under optical microscopy. Modifications of cell shape were induced by ATRA and WRCP. Bel-7402 cells changed to polygon shape after treatment with ATRA, but they appeared in spindle shape after treated with WRCP. Examined under electronic microscope, Bel-7402 cells exhibited irregular nuclear envelope, poor euchromatin and abundant mitochondrion and ribosome. The malignant shape of nucleus was slightly reversed by ATRA. However, with WRCP treatment, cells showed ultrastructural features of better differentiation, e.g. abundant heterochromatin, poor euchromatin and low necleo-cytoplasmic ratio. The AFP level in the culture medium of the WRCP-treated group was significantly lower than that of the PBS-treated group. However, the ALB level in the culture medium was remarkably increased with WRCP treatment. There was significant difference between negative control group and WRCP group (P < 0.001, Figure 3).
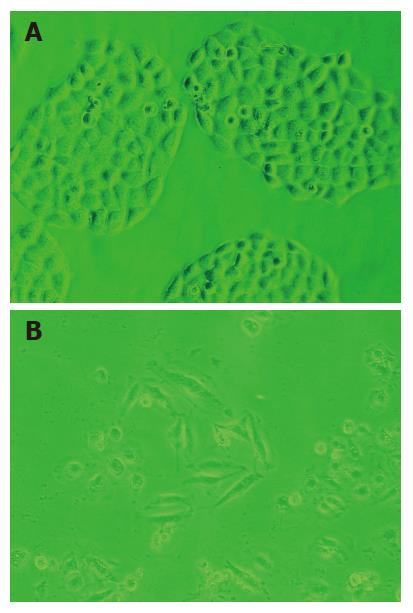
After WRCP treatment, some Bel-7402 cells changed to round shape and could not adhere to the bottom. Many survived cells were tumid, and abundant vacuolations in cytoplasm were detected under optical and electronic microscope (Figure 4A and B).
Disturbances of the equilibrium between cellular replication, growth, differentiation and apoptosis favor the growth of preneoplastic and tumoral lesions in the liver. There is decreased cell death and increased cell proliferation in HCC[6]. WRCP suppressed the proliferation of Bel-7402 cells in a dose- and time-dependent manner. Even at a concentration of 75 mg/L, WRCP inhibited the proliferation and viability of Bel-7402 cells strongly. Because cells were arrested by WRCP at S phase, we inferred that WRCP suppressed the proliferation of Bel-7402 cells through inhibiting the replication of DNA. Furthermore, WRCP induced the apoptosis of Bel-7402 cells. The percentage of apoptosis cells was 35.9% in Bel-7402 cells after treatment with WRCP at concentration of 250 mg/L.
The development of malignancies can result from the change of the normal process of cell differentiation[7]. The induction of terminal differentiation in tumor cells represents a possible therapeutic strategy for HCC. Retinoids and compounds involved in the vitamin A metabolic pathway are known to be differentiation-inducing agents with hypoproliferative effects. Although almost 95% of patients with acute promyelocytic leukemia (APL) undergo complete remission when treated with all-trans-retinoic acid (ATRA), the effect of retinoids on most tumors is disappointing[8,9]. A study using retinoid showed a 20% reduction in the second tumor development in patients who had been treated with percutaneous alcohol injection[10]. Retinoids have been used in the prevention of second tumor development after initial tumor resection or ablation[11,12].
Cell differentiation includes the morphologic variation and functional maturation. Bel-7402 cells showed abnormal features with a round shape seen under optical microscopy, but cells changed to spindle shape after exposure to WRCP. In addition, WRCP-treated cells showed ultrastructural features of better differentiation, such as abundant heterochromatin, poor euchromatin and low nucleo-cytoplasmic ratio. AFP is recognized as a malignant phenotype protein of human hepatoma cells. On the contrary, ALB is a protein secreted by mature hepatocytes. The AFP secretion decreased while the ALB secretion increased markedly in WRCP-treated cells. These results suggest that WRCP could induce the differentiation in human hepatoma Bel-7402 cells. We tested the effects of Gekko sulfated polysaccharides on the proliferation, apoptosis and differentiation of Bel-7402 cells simultaneously, and found that Gekko sulfated polysaccharides suppressed the proliferation and induced differentiation of Bel-7402 cells, but the toxicity was negligible[13]. Thus Gekko sulfated polysaccharides may be the main compound of WRCP, which induce differentiation of HCC. Tumor is a cell cycle disease that has the abnormal interface of high proliferation, low differentiation and apoptosis block. WRCP could make tumor cells renew the normal interface mechanism by suppressing proliferation, inducing differentiation and inducing apoptosis.
Hepatocarcinoma is one of the most frequently occurring cancers worldwide. However, few effective chemotherapeutic agents for this disease have not been developed. Hepatocarcinoma cells can resist chemotherapeutic agents especially by expressing multi-drug resistance gene[3,4]. In addition, many hepatocarcinoma cells are in G0/G1 phase, which are insensitive to cytotoxin[14]. In this study, the ratio of G0/G1 phase cells is 57.9%. WRCP could inhibit the viability of Bel-7402 cells significantly. The survived cells were tumid, and abundant vacuolations occurred in cytoplasm. These results demonstrated that WRCP had a strong cytotoxic effect in Bel-7402 cells. However, WRCP is significantly different from chemotherapeutic agents, because it could enhance its cytotoxicity by cell cycle regulation. WRCP increased the S phase cells and decreased the G0/G1 phase cells. The ratios of G0/G1 phase cells and S phase cells are 57.9% and 28.6%, respectively in negative control group. After treated with a 250 mg/L WRCP, 49.4% of the cells were in G0/G1 phase and 37.8% of the cells were in S phase. Therefore, WRCP may enhance its cytotoxic effect in tumor cells through cell cycle regulation.
In conclusion, WRCP could effectively inhibit proliferation, induce apoptosis and differentiation and arrest cells in S phase in human hepatocarcinoma cells. In addition, WRCP had a strong cytotoxicity in hepatocarcinoma cells. The reversion of the abnormal biological action of carcinoma may enhance its cytotoxic effect. These results indicate that WRCP has the potential value of development of supplementary therapies against cancer. However, the identification of the active compounds and the exact mechanism involved remains to be further investigated.
S-Editor Zhu LH L-Editor Ma JY E- Editor Chen GJ
| 1. | Parker SL, Tong T, Bolden S, Wingo PA. Cancer statistics, 1997. CA Cancer J Clin. 1997;47:5-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1494] [Cited by in RCA: 1410] [Article Influence: 50.4] [Reference Citation Analysis (0)] |
| 2. | Chen CJ, Yu MW, Liaw YF. Epidemiological characteristics and risk factors of hepatocellular carcinoma. J Gastroenterol Hepatol. 1997;12:S294-S308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 341] [Cited by in RCA: 338] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 3. | Petraccia L, Onori P, Sferra R, Lucchetta MC, Liberati G, Grassi M, Gaudio E. MDR (multidrug resistance) in hepatocarcinoma clinical-therapeutic implications. Clin Ter. 2003;154:325-335. [PubMed] |
| 4. | Cheng SC, Zhou J, Xie Y. P-glycoprotein expression induced by glucose depletion enhanced the chemosensitivity in human hepatocellular carcinoma cell-lines. Cell Biol Int. 2005;29:269-275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 5. | Chan TY, Tomlinson B, Tse LK, Chan JC, Chan WW, Critchley JA. Aconitine poisoning due to Chinese herbal medicines: a review. Vet Hum Toxicol. 1994;36:452-455. [PubMed] |
| 6. | Kountouras J, Zavos C, Chatzopoulos D. Apoptosis in hepatocellular carcinoma. Hepatogastroenterology. 2003;50:242-249. [PubMed] |
| 7. | Sell S. The role of determined stem-cells in the cellular lineage of hepatocellular carcinoma. Int J Dev Biol. 1993;37:189-201. [PubMed] |
| 8. | Sirulnik LA, Stone RM. Acute promyelocytic leukemia: current strategies for the treatment of newly diagnosed disease. Clin Adv Hematol Oncol. 2005;3:391-397, 429. [PubMed] |
| 9. | Tallmann MS. Curative therapeutic approaches to APL. Ann Hematol. 2004;83 Suppl 1:S81-S82. [PubMed] |
| 10. | Muto Y, Moriwaki H, Saito A. Prevention of second primary tumors by an acyclic retinoid in patients with hepatocellular carcinoma. N Engl J Med. 1999;340:1046-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 180] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 11. | Okuno M, Kojima S, Moriwaki H. Chemoprevention of hepatocellular carcinoma: concept, progress and perspectives. J Gastroenterol Hepatol. 2001;16:1329-1335. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 12. | Ryder SD. Guidelines for the diagnosis and treatment of hepatocellular carcinoma (HCC) in adults. Gut. 2003;52 Suppl 3:iii1-iii8. [PubMed] |
| 13. | Wu X, Chen D, Xie GR. Effects of Gekko sulfated polysaccharide on the proliferation and differentiation of hepatic cancer cell line. Cell Biol Int. 2006;30:659-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 28] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | Hui AM, Makuuchi M, Li X. Cell cycle regulators and human hepatocarcinogenesis. Hepatogastroenterology. 1998;45:1635-1642. [PubMed] |