Published online Mar 21, 2006. doi: 10.3748/wjg.v12.i11.1786
Revised: July 29, 2005
Accepted: August 3, 2005
Published online: March 21, 2006
The gallbladder is an unusual location of pancreatic heterotopia, defined as the presence of pancreatic tissue lacking anatomical and vascular continuity with the main body of the gland. A 28-year-old man presented with anorexia, nausea and pain in the right upper abdomen. On physical examination, the abdomen was tender to palpation and Murphy sign was positive. The patient underwent a cholecystecomy. This case, in our opinion, is very interesting since it permits to consider a controversial issue in the pathology of the gallbladder. The histological appearance of ductal structure in pancreatic heterotopia resembles the histological picture of both Aschoff-Rokitansky (AR) sinuses and adenomyomas. This finding suggests that these lesions are linked by a common histogenetic origin. We suggest that the finding of an adenomyoma in the gallbladder should prompt an extensive sampling of the organ in order to verify the coexistence of pancreatic rests.
- Citation: Pilloni L, Cois A, Uccheddu A, Ambu R, Coni P, Faa G. Complete pancreatic heterotopia of gallbladder with hypertrophic duct simulating an adenomyoma. World J Gastroenterol 2006; 12(11): 1786-1787
- URL: https://www.wjgnet.com/1007-9327/full/v12/i11/1786.htm
- DOI: https://dx.doi.org/10.3748/wjg.v12.i11.1786
The gallbladder is an unusual location of pancreatic heterotopia, defined as the presence of pancreatic tissue lacking anatomical and vascular continuity with the main body of the gland[1]. About 75% of all pancreatic rests are located in the stomach, duodenum and jejunum, the appendix, diverticulum of Meckel and the ampulla of Vater[2] tract, ectopic pancreas has been found in the umbilicus, fallopian tube, mediastinum[2], spleen[3] and omentum[4]. Here, we describe a case of gallbladder pancreatic heterotopia associated with a hypertrophic duct simulating a small intrapancreatic adenomyoma.
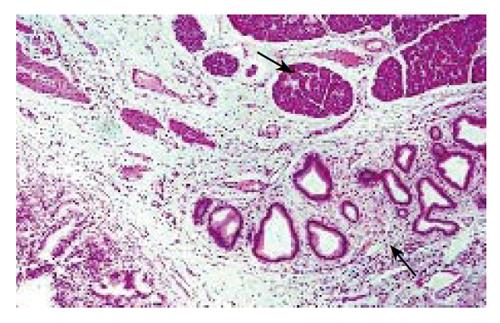
A 28-year-old man presented with anorexia, nausea and pain in the right upper abdomen. On physical examination, the abdomen was tender to palpation and Murphy sign was positive. The patient underwent surgery, and a cholecystectomy was performed. The gallbladder was 10.5 cm in length and 3.5 cm in width, with a mural thickness of 0.3 cm. No stones were present. An intramural, firm, yellow nodule measuring 0.6 × 0.5 cm was seen in the gallbladder body: it was well circumscribed and showed a central umbilication on the mucosal surface. Light microscopy revealed diffuse proliferation of the surface epithelium, resulting in numerous invaginations into a thickened wall and numerous Aschoff-Rokitansky (AR) sinuses. The muscular layers appeared hypertrophied and possibly hyperplastic, with frequent foci of adenomyomatosis. No inflammatory infiltrate was seen. The epithelial component had typical features of pancreatic tissue. Acini were composed of polygonal cells with basal nuclei and apical granular cytoplasm surrounding a minute lumen. Endocrine islands were occasionally detected. The ductal system consisted of intercalated, intralobular and interlobular ducts: the major duct, surrounded by bundles of smooth muscle, appeared to drain into the gallbladder lumen. Ductal epithelium was surrounded by a smooth muscle component and displayed a typical pancreatic duct-like immunophenotype, characterized by immunoreactivity for cytokeratins 7, 8, 18, 19 and CA19-9. No reactivity against CEA and cytokeratin 20 was found. Acinar cells arranged in a single layer with typical exocrine differentiation were focally positive for α1-antitrypsin, chimotrypsin and cytokeratins 8 and 18. They were negative for CEA, CA19-9 and cytokeratins 7, 19 and 20. The patient, after cholecystectomy, had an uneventful recovery and normal development at follow-up.
This case, in our opinion, is very interesting since it permits to consider a controversial issue in the pathology of the gallbladder. The histological appearance of ductal structures in pancreatic heterotopia resembles the histological picture of both AR sinuses and adenomyomas. This finding suggests that these lesions are linked by a common histogenetic origin.
In fact, AR sinuses are structures consisting of down growth of the gallbladder mucosa into the wall[5]. They are thought to be the consequence of increased intraluminal pressure, analogous to colonic diverticula[5]. Sometimes, mucosal proliferation is so exuberant as to result in formation of branching sinuses and cystic structures. In many cases, the muscular layer is hypertrophic and hyperplastic, thus resembling focal adenomyomatosis[5]. The latter feature is seen in our case (Figure 1), where the major duct, surrounded by a thickened muscular layer, shows morphological features overlapping those of adenomyomas. Analogously, it is not precisely defined when florid AR sinuses become adenomyomatosis[5].
Likewise, adenomyomas consist of glandular formations lined with cuboidal-to-columnar epithelium and surrounded by bundles of smooth muscle[6,7], similar to the prominent duct seen in our case.
Also heterotopic pancreas, when it includes only ducts, without acinar cells (canalicular or abortive form), shows the same morphological appearance[5].
On the basis of such morphological observations, we believe that AR sinuses, adenomyomas (when focal), adenomyomatosis (when diffuse), and abortive forms of pancreatic heterotopia can be considered variable manifestations of the same pathological process and be grouped into the same nosological category.
Consistent with this hypothesis, several authors have suggested that adenomyomas associated with pancreatic heterotopia found in other organs (stomach and duodenum) should be considered as an abortive variant of pancreatic heterotopia missing an acinar component[7,8]. This hypothesis emphazises the concept of a common pathogenetic origin of these lesions[7,8].
In contrast, Ryan et al believed that the adenomyomas of the gallbladder, in spite of histology, arise from diverticular disease of the gallbladder and are not pathogenetically related to heterotopic pancreas[9].
To our knowledge, only 30 cases of ectopic pancreas in the gallbladder have been reported; if the association between adenomyosis and pancreatic rests is true, as we propose, this finding should be more frequently observed. Therefore, we suggest that the finding of an adenomyoma in the gallbladder should prompt an extensive sampling of the organ in order to verify the coexistence of pancreatic rests. Unfortunately, it is not possible to provide immunohistochemical support to our hypothesis, since the gallbladder and the pancreas have the same embryologic origin, and thus there are no specific markers allowing unequivocal differentiation between pancreatic tissue and gallbladder epithelium. Recently, Ko et al[10] reported that proliferating duct cells of the pancreas express vimentin. Accordingly, we used immunohistochemistry to detect the expression of vimentin in order to verify their observations, but in our hands duct cells of both normal pancreas and pancreatic adenocarcinoma were negative for this marker. Further studies are thus required, apart from morphology, to support an unequivocal link between adenomyomas and pancreatic heterotopia in the gallbladder.
S- Editor Guo SY L- Editor Zhang JZ E- Editor Ma WH
| 1. | Harold KL, Sturdevant M, Matthews BD, Mishra G, Heniford BT. Ectopic pancreatic tissue presenting as submucosal gastric mass. J Laparoendosc Adv Surg Tech A. 2002;12:333-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 2. | Lai EC, Tompkins RK. Heterotopic pancreas. Review of a 26 year experience. Am J Surg. 1986;151:697-700. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 130] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 3. | Mourra N, Balladur P, Parc R, Flejou JF. Intrasplenic mucinous cystadenoma with mesenchymal stroma arising in pancreatic heterotopia. Histopathology. 2003;42:616-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 4. | Tornoczky T, Kalman E, Jakso P, Mehes G, Pajor L, Kajtar GG, Battyany I, Davidovics S, Sohail M, Krausz T. Solid and papillary epithelial neoplasm arising in heterotopic pancreatic tissue of the mesocolon. J Clin Pathol. 2001;54:241-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 46] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 5. | Owen DA, Kelly JK. Pathology of the gallbladder, biliary tract and pancreas. Eds WB Saunders company. 2001;247-250. |
| 6. | Serour F, Gorenstein A, Lipnitzky V, Zaidel L. Adenomyoma of the small bowel: a rare cause of intussusception in childhood. J Pediatr Gastroenterol Nutr. 1994;18:247-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 7. | Babal P, Zaviacic M, Danihel L. Evidence that adenomyoma of the duodenum is ectopic pancreas. Histopathology. 1998;33:487-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 8. | Erberich H, Handt S, Mittermayer C, Tietze L. Simultaneous appearance of an adenomyoma and pancreatic heterotopia of the stomach. Virchows Arch. 2000;436:172-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 20] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 9. | Ryan A, Lafnitzegger JR, Lin DH, Jakate S, Staren ED. Myoepithelial hamartoma of the duodenal wall. Virchows Arch. 1998;432:191-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Ko SH, Suh SH, Kim BJ, Ahn YB, Song KH, Yoo SJ, Son HS, Cha BY, Lee KW, Son HY. Expression of the intermediate filament vimentin in proliferating duct cells as a marker of pancreatic precursor cells. Pancreas. 2004;28:121-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 35] [Article Influence: 1.7] [Reference Citation Analysis (0)] |