Published online Apr 21, 2005. doi: 10.3748/wjg.v11.i15.2364
Revised: July 9, 2004
Accepted: August 12, 2004
Published online: April 21, 2005
AIM: An inherited deficiency of human lysosomal acid lipase (LAL) results in the rare conditions of Wolman disease and cholesteryl ester storage disease (CESD). We want to present the rare case of CESD in an adult.
METHODS: We report about an adult female patient with severe chronic diarrhea and weight loss as a consequence of CESD. Clinical examination revealed signs of malabsorption and slightly elevated liver enzymes.
RESULTS: Histopathologic changes in the liver tissue and DNA sequence analysis confirmed the diagnosis of CESD due to homozygosity for the most common CESD mutation, a G934A splice site defect encoded by exon 8 of the lysosomal acid lipase (LIPA) gene.
CONCLUSION: It is the first case in the literature with diarrhea as a putative symptom of CESD in adult patients.
- Citation: Drebber U, Andersen M, Kasper HU, Lohse P, Stolte M, Dienes HP. Severe chronic diarrhea and weight loss in cholesteryl ester storage disease: A case report. World J Gastroenterol 2005; 11(15): 2364-2366
- URL: https://www.wjgnet.com/1007-9327/full/v11/i15/2364.htm
- DOI: https://dx.doi.org/10.3748/wjg.v11.i15.2364
Wolman disease and cholesteryl ester storage disease (CESD) are very rare inherited disorders of lipid metabolism. Both are the consequences of autosomal recessive mutations which result in a deficient or severely diminished activity of human lysosomal acid lipase (LAL) acid esterase and cholesteryl ester hydrolase, respectively[1]. The mature 378-amino acid protein with molecular masses of approximately 43-54 ku is encoded by the lysosomal acid lipase (LIPA) gene on chrom-osome 10q23.2-q23.3 which contains 10 exons dispersed over a 38.8 kb region[2-4].
LAL is essential for the intralysosomal breakdown of cholesteryl esters and triglycerides which are taken up by receptor-mediated endocytosis. An inherited deficiency or low activity results in the intralysosomal storage of the respective lipid substrates. As the enzyme is synthesized by all nucleated cells, lipid-laden cells are found in all organs, particularly in liver, spleen, the adrenal and the hemopoietic system, and in the intestine as well as in the lymph nodes, lungs, testes, and ovaries[5-8].
While Wolman disease has an infaust prognosis and leads to hydrops fetalis and congenital ascites as well as an abnormal neurological development, CESD presents as the milder form due to a residual enzymatic activity of approximately 3% which results in widespread lipid deposition and can induce symptoms at any age[9-13].
CESD is rarely diagnosed. We report about the unusual case of a 41-year-old woman who had been suffering from recurrent diarrhea since early childhood and was referred to the hospital during a severe episode of diarrhea and ensuing weight loss. During the clinical examination, a liver biopsy was taken which led to the tentative diagnosis of CESD. This was confirmed by molecular genetic analysis.
A 41-year-old female patient, who had been brought up as a foster-child, was referred to the hospital because of persistent chronic diarrhea which caused deterioration by inducing a weight loss of 10 kg in the last 5 mo (body weight 44 kg, height 168 cm at the time of hospital admission). Clinical examination revealed a reduced level of lipid-soluble vitamins as well as a pathologic lactose tolerance. Abdominal ultrasound showed edema of the small bowel mucosa. Celiac disease was excluded by normal antibody levels against endomysium, tissue-transglutaminase, and gliadin. Other causes of diarrhea-like infection or laxative abuse were exclu-ded as well. The patient had elevated liver enzymes (AST 47 U/L, ALT 57 U/L). There were, however, no signs of a viral hepatitis (HbsAg- and anti-HBc-). Immunoglobulin values were also normal. In order to find a cause for the raised liver enzymes and the diarrhea, biopsies of liver and small intestines were taken.
Formalin-fixed, paraffin-embedded liver tissue and small bowel biopsies were prepared for histopathologic and immunhistochemical analyses. Ultrastuctural examination was performed on glutaraldehyde-fixed liver tissue under the electron microscope.
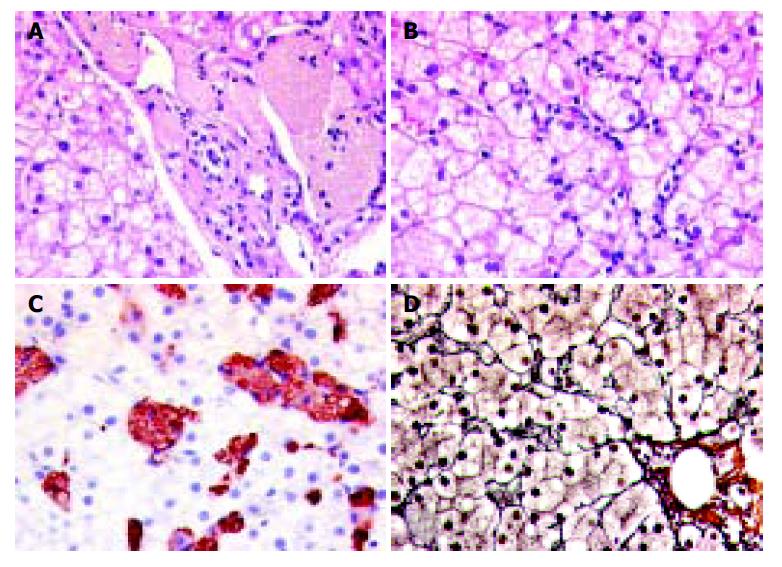
Light-microscopy in liver showed an increase of Kupffer cells and macrophages which were found within the sinusoids as well as in the portal tracts. The macrophages were conspicuous with the cytoplasmatic storage of foamy, tan-colored, PAS-positive material. In immunohistochemistry, the described cell population expressed the macrophage-specific marker CD68. The distribution of the CD68-positive population was abundant over the whole acinus without zonal accentuation. Additionally, a discrete parenchymal inflammatory activity with single apoptotic bodies as well as a fine vesicular steatosis in about 40% of the hepatocytes. A discrete pericellular fibrosis was seen in the reticulin stain (Figure 1).
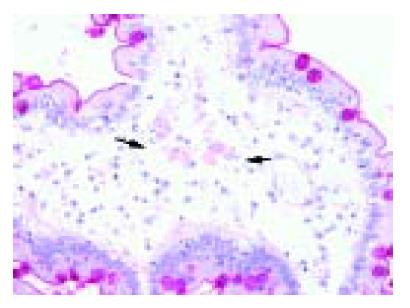
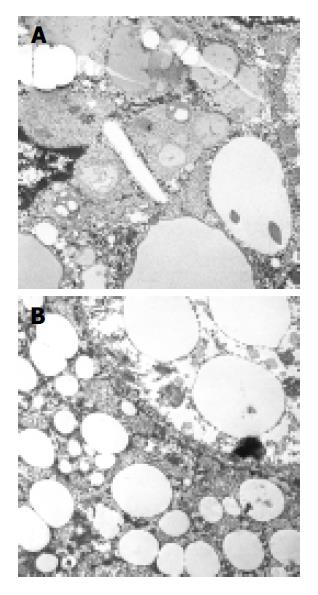
The small bowel biopsies revealed single macrophages with cytoplasmatic storage of PAS-positive foamy, tan-colored material within the apical part of the villi in the lamina submucosa (Figure 2). Ultrastructural examination demonstrated lysosomal lipid storage in the cytoplasm of hepatocytes and macrophages as well as the presence of single cholesteryl ester crystals (Figure 3).
Overall, the histopathologic changes in the liver biopsy were suggestive of CESD. Therefore, a DNA sequence analysis of the proband’s LIPA gene was performed. The patient was found to be a homozygous carrier of the most common CESD mutation, a G934A exchange affecting the last coding base of exon 8, thereby confirming the diagnosis. This genetic defect results in the skipping of exon 8 from the mRNA transcript and in the loss of the internal amino acids 254-277 from the mature enzyme. Despite the presence of the mutation, approximately 3% of normal splicing occurs, which ensures survival of the affected individual[1,3].
We present the unusual case of CESD in an adult woman. CESD is a rare condition, which is usually diagnosed during the first or second decade of life due to the accompanying hepatomegaly, often in combination with hyperchol-esterolemia, but only seldom in later adulthood[7]. Although there is no specific treatment, alleviation can be induced by 3-hydroxy-3-methylglutaryl coenzyme A (HMG CoA) reductase inhibitors such as lovostatin[14-16]. In case of liver cirrhosis, a liver transplantation is indicated[17-19].
The clinical symptoms of our patient, who suffered from severe diarrhea and weight loss, represent a remarkable difference from the typical disease pattern of CESD which normally shows a liver symptomatology with early progression toward cirrhosis[1]. The patient reported about chronic diarrhea with the onset during early childhood, so that CESD seems to play a causal role. Accordingly, laboratory findings showed a reduced level of lipid-soluble vitamins as a sign of chronic malabsorption. The liver symptomatology, in contrast, was discrete. A liver biopsy was taken because of slightly elevated liver enzymes of unknown etiology. Histopathologic changes in the liver were characteristic for CESD, however, a substantial fibrosis had not developed which is reflected by the laboratory findings. This is especially remarkable, because of the long time-period of disease symptoms, and in comparison with other reported cases, where severe fibrosis has been reported to be present as early as childhood[20].
DNA sequence analysis revealed homozygosity for a G934A mutation in exon 8 of the LIPA gene. This nucleotide substitution results in exon skipping and the subsequent loss of 24 amino acids from the mature enzyme. It is the most frequent genetic defect seen in CESD so that the mutation does not explain the unusual clinical presentation[3].
In conclusion, CESD is a very rare metabolic error and presents with an impressing variety of clinical symptoms which make it difficult to establish the correct diagnosis. The patient reported here is the first case in the literature with CESD and chronic diarrhea as the putative symptom of CESD. We therefore conclude that CESD should be taken into account in the differential diagnosis of chronic diarrhea when other causes for diarrhea have been excluded.
Science Editor Li WZ Language Editor Elsevier HK
| 1. | Anderson RA, Bryson GM, Parks JS. Lysosomal acid lipase mutations that determine phenotype in Wolman and cholesterol ester storage disease. Mol Genet Metab. 1999;68:333-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 58] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 2. | D'Agostino D, Bay L, Gallo G, Chamoles N. Cholesterol ester storage disease: clinical, biochemical, and pathological studies of four new cases. J Pediatr Gastroenterol Nutr. 1988;7:446-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 3. | Aslanidis C, Ries S, Fehringer P, Büchler C, Klima H, Schmitz G. Genetic and biochemical evidence that CESD and Wolman disease are distinguished by residual lysosomal acid lipase activity. Genomics. 1996;33:85-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 110] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 4. | Lohse P, Maas S, Sewell AC, van Diggelen OP D. Molecular defects underlying Wolman disease appear to be more heterogeneous than those resulting in cholesteryl ester storage disease. J Lipid Res. 1999;40:221-228. [PubMed] |
| 5. | Edelstein RA, Filling-Katz MR, Pentchev P, Gal A, Chandra R, Shawker T, Guzzetta P, Comly M, Kaneski C, Brady RO. Cholesteryl ester storage disease: a patient with massive splenomegaly and splenic abscess. Am J Gastroenterol. 1988;83:687-692. [PubMed] |
| 6. | Elleder M, Chlumská A, Hyánek J, Poupĕtová H, Ledvinová J, Maas S, Lohse P. Subclinical course of cholesteryl ester storage disease in an adult with hypercholesterolemia, accelerated atherosclerosis, and liver cancer. J Hepatol. 2000;32:528-534. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 54] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 7. | Elleder M, Chlumska A, Ledvinová J, Poupetová H. Testis - a novel storage site in human cholesteryl ester storage disease. Autopsy report of an adult case with a long-standing subclinical course complicated by accelerated atherosclerosis and liver carcinoma. Virchows Arch. 2000;436:82-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 8. | Hanák J, Elleder M. Cholesterol ester storage disease (CESD). Cesk Pediatr. 1984;39:721-725. [PubMed] |
| 9. | Klima H, Ullrich K, Aslanidis C, Fehringer P, Lackner KJ, Schmitz G. A splice junction mutation causes deletion of a 72-base exon from the mRNA for lysosomal acid lipase in a patient with cholesteryl ester storage disease. J Clin Invest. 1993;92:2713-2718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 76] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 10. | Muntoni S, Wiebusch H, Funke H, Ros E, Seedorf U, Assmann G. Homozygosity for a splice junction mutation in exon 8 of the gene encoding lysosomal acid lipase in a Spanish kindred with cholesterol ester storage disease (CESD). Hum Genet. 1995;95:491-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 39] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 11. | Aslanidis C, Klima H, Lackner KJ, Schmitz G. Genomic organization of the human lysosomal acid lipase gene (LIPA). Genomics. 1994;20:329-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 47] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 12. | Pagani F, Pariyarath R, Garcia R, Stuani C, Burlina AB, Ruotolo G, Rabusin M, Baralle FE. New lysosomal acid lipase gene mutants explain the phenotype of Wolman disease and cholesteryl ester storage disease. J Lipid Res. 1998;39:1382-1388. [PubMed] |
| 13. | Lohse P, Maas S, Lohse P, Elleder M, Kirk JM, Besley GT, Seidel D. Compound heterozygosity for a Wolman mutation is frequent among patients with cholesteryl ester storage disease. J Lipid Res. 2000;41:23-31. [PubMed] |
| 14. | Levy R, Ostlund RE, Schonfeld G, Wong P, Semenkovich CF. Cholesteryl ester storage disease: complex molecular effects of chronic lovastatin therapy. J Lipid Res. 1992;33:1005-1015. [PubMed] |
| 15. | Gasche C, Aslanidis C, Kain R, Exner M, Helbich T, Dejaco C, Schmitz G, Ferenci P. A novel variant of lysosomal acid lipase in cholesteryl ester storage disease associated with mild phenotype and improvement on lovastatin. J Hepatol. 1997;27:744-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 44] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 16. | Rassoul F, Richter V, Lohse P, Naumann A, Purschwitz K, Keller E. Long-term administration of the HMG-CoA reductase inhibitor lovastatin in two patients with cholesteryl ester storage disease. Int J Clin Pharmacol Ther. 2001;39:199-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 17. | Leone L, Ippoliti PF, Antonicelli R, Balli F, Gridelli B. Treatment and liver transplantation for cholesterol ester storage disease. J Pediatr. 1995;127:509-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 18. | Ferry GD, Whisennand HH, Finegold MJ, Alpert E, Glombicki A. Liver transplantation for cholesteryl ester storage disease. J Pediatr Gastroenterol Nutr. 1991;12:376-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 19. | Kale AS, Ferry GD, Hawkins EP. End-stage renal disease in a patient with cholesteryl ester storage disease following successful liver transplantation and cyclosporine immunosuppression. J Pediatr Gastroenterol Nutr. 1995;20:95-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 20. | Akçören Z, Göğüş S, Koçak N, Gürakan F, Ozen H, Yüce A. Cholesteryl ester storage disease: case report during childhood. Pediatr Dev Pathol. 1999;2:574-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |