Published online Oct 15, 2004. doi: 10.3748/wjg.v10.i20.3011
Revised: January 26, 2004
Accepted: March 2, 2004
Published online: October 15, 2004
AIM: To construct a pEgr-IFNγ plasmid and to investigate its expression properties of interferon-γ (INF-γ ) induced by irradiation and the effect of gene-radiotherapy on the growth of melanoma.
METHODS: A recombined plasmid, pEgr-IFNγ , was constructed and transfected into B16 cell line with lipofectamine. The expression properties of pEgr-IFNγ were investigated by ELISA. Then, a B16 melanoma-bearing model was established in mice, and the plasmid was injected into the tumor tissue. The tumor received 20 Gy X-ray irradiation 36 h after injection, and IFN-γ expression was detected from the tumor tissue. A tumor growth curve at different time points was determined.
RESULTS: The eukaryotic expression vector, pEgr-IFNγ , was successfully constructed and transfected into B16 cells. IFN-γ expression was significantly increased in transfected cells after X-ray irradiation in comparison with 0 Gy group (77.73-94.60 pg/mL, P < 0.05-0.001), and was significantly higher at 4 h and 6 h than that of control group after 2 Gy X-ray irradiation (78.90-90.00 pg/mL, P < 0.01-0.001). When the transfected cells were given 2 Gy irradiation 5 times at an interval of 24 h, IFN-γ expression decreased in a time-dependent manner. From d 3 to d 15 after IFNγ gene-radiotherapy, the tumor growth was significantly slower than that after irradiation or gene therapy alone.
CONCLUSION: The anti-tumor effect of pEgr-IFNγ gene-radiotherapy is better than that of genetherapy or radiotherapy alone for melanoma. These results may establish an important experimental basis for gene-radiotherapy of cancer.
-
Citation: Wu CM, Li XY, Huang TH. Anti-tumor effect of
pEgr-IFNγ gene-radiotherapy in B16 melanoma-bearing mice. World J Gastroenterol 2004; 10(20): 3011-3015 - URL: https://www.wjgnet.com/1007-9327/full/v10/i20/3011.htm
- DOI: https://dx.doi.org/10.3748/wjg.v10.i20.3011
Radiotherapy is one of the treatments for cancer. However, its therapeutic effect is still unsatisfactory, and thus new therapeutic strategy must be adopted. Gene therapy in combination with radiotherapy is one of the most important advances[1-4]. The introduction of Egr-1 promoter induced by irradiation has provided a possible approach to this combination therapy[5-6].
IFNγ is the first cytokine produced by gene engineering and used for treatment of carcinoma, and has anti-tumor effects. Its antitumor mechanism includes direct inhibition of tumor cell proliferation, and indirect action by activating cytotoxic activities[7-18]. In the present study we constructed the pEgr-IFNγ plasmid by connecting IFNγ cDNA to Egr-1 promoter to investigate its expression properties in B16 cells and its antitumor effect in mice.
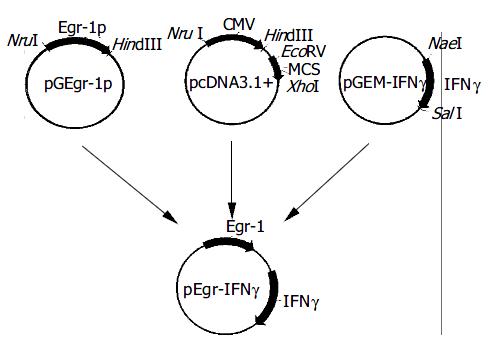
The expression vector for pEgr-IFNγ is shown in Figure 1.
B16 cell line was cultured in MH Radiobiology Research Unit of Jilin University and maintained in RPMI 1640 (Life Technologies) with 100 mL/L fetal bovine serum (Hyclone Laboratories), L-glutamine, 100 μg/mL of streptomycin, and 100 U/mL of penicillin. The cell line was incubated at 37 °C in 50 mL/L CO2.
B16 cells were transfected in a 6-well plate when the cells reached 70% confluence. Solution A was prepared by addition of 10 μg of pEgr-IFNγ or pcDNA3.1+ to 100 μL serum-free medium (SFM), and solution B by addition of 10 μL liposome to 100 μL SFM. Solutions A and B were mixed at room temperature for 30 min, then mixed with 0.8 mL SFM, the mixture was added to the rinsed cells. The medium was replaced with fresh and complete medium 6 h after transfection.
Supernatants from different groups were collected for detection of the IFNγ expression with ELISA kit (Genzyme).
Adult female Kunming mice were provided by the Experimental Animal Center of Jilin University, with an average weight of 18 ± 2 g.
A melanoma-bearing model was established by subcutaneous injection at right hind limb with 0.1 mL B16 cells (5 × 10 6 /mL), 10 d later, tumor tissue received multi-focus injection of plasmids packaged with liposome (20 μg plasmid and 0.1 mL liposome per mouse) for the experimental groups.
Tumor size was measured. Then, tumor volume (V) was calculated according to the formula: V (mm3) = L × W 2/2, where, L: the longest diameter of tumor; W: the diameter at right angles on the largest horizontal section. Tumor growth rate (f) was the ratio of the volume at different time points over the initial volume (V0).
X-rays of 200 kV and 10 mA with 0.5 mm copper and 1.0 mm aluminum filter were given at a dose-rate of 0.8639 Gy/min for a total dose of 2--20 Gy.
B16 cells were seeded in a 6-well plate and randomly divided into different groups. The experiment groups received X-ray irradiation of various doses or at different time points, control groups received sham irradiation simultaneously.
Mice bearing B16 xenografts (n = 40) were randomly divided into five groups: control group, 20 Gy group, pcDNA3.1 + 20 Gy group, pEgr-IFNγ group and pEgr-IFNγ + 20 Gy group. Thirty-six hours before irradiation, melanoma tissue was injected with plasmids or buffer at 5 separate sites. Tumor beds were given 20 Gy X-ray irradiation. Animals of 20 Gy group, pcDNA3.1 + 20 Gy group and pEgr-IFNγ + 20 Gy group were shielded with lead except for the tumor-bearing hind limb, animals in the other 2 groups were given sham-irradiation at the same time. Tumors were measured and recorded as previously described.
Total RNA were extracted from EC9706 cells and tumor tissue for RT-PCR. GAPDH was used as an internal reference. Primers were as follows: GAPDH, forward primer 5’-TGCACCACCAAC TGCTTAGC -3’ and reverse one 5’-GGCATGGACTGTGG TCATGAG-3’, mouse IFNγ cDNA, forward primer 5’-GATCCT TTGGACCCTCTG ACTT-3’ and reverse one 5’-AGACAGTGA TAAACTATAAATGAGCG-3’.
RT-PCR was performed as following: denaturation at 95 °C for 3 min, 30 cycles at 95 °C for 45 s, at 56 °C for 45 s, at 72 °C for 40 s and extension at 72 °C for 10 min.
Student’s t test was used to determine comparability between groups. P values less than 0.05 were considered statistically significant.
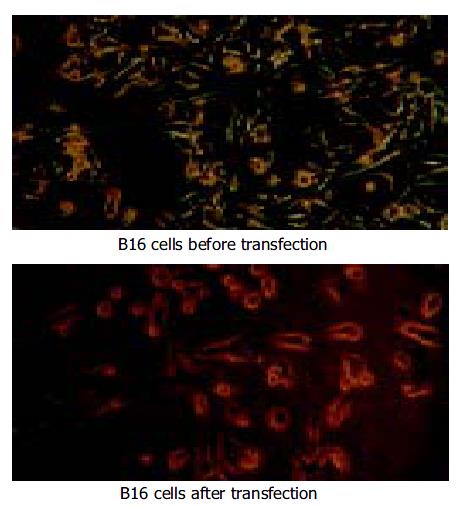
Pre- and post-transfection of B16 cells are shown in Figure 2.
After transfection B16 cells received different doses of X-ray irradiation. The cells of control group were transfected with pcDNA3.1+ plasmid. Six hours after irradiation IFNγ expression and mRNA level were detected.
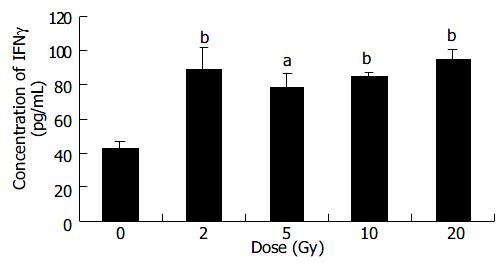
The results showed that IFNγ expression in 2-20 Gy groups was significantly higher than that in 0 Gy group (P < 0.05-0.01) (Figure 3).
After irradiation IFNγ mRNA could be detected in B16 cells (Figure 4). The level of IFNγ mRNA was compared with that of GAPDH, and their ratios are shown in Table 1. The IFNγ mRNA levels in 2-20 Gy groups were higher than that of 0 Gy group.
| Dose (Gy) | Ratio of IFNγ mRNA level |
| 0 | 0.819 |
| 2 | 0.972 |
| 5 | 1.347 |
| 10 | 1.950 |
| 20 | 2.144 |
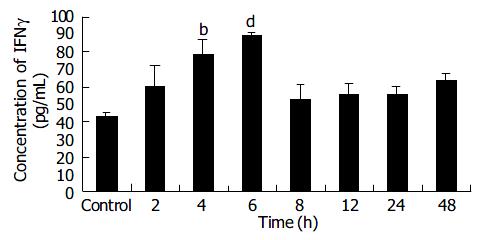
After transfection B16 cells received 2 Gy of X-ray irradiation while the control group received sham irradiation. IFNγ protein was detected at different time points after irradiation. ELISA results showed that the IFNγ expression increased with time from 2 h to 6 h in a time-dependent manner, and peaked at 6 h, about 1.8 times of that in control group (P < 0.001). However, from 8 h to 48 h post-radiation IFNγ expressions were not significantly different from that in control group (Figure 5).
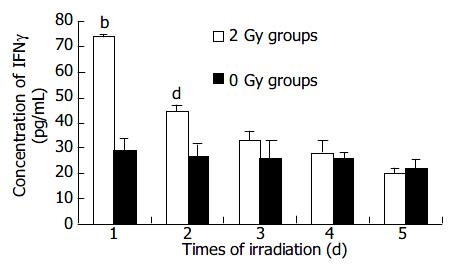
After transfection B16 cells received 2 Gy irradiation while the control group received sham irradiation. IFNγ expression was detected 6 h later. Irradiation and detection were repeated 5 times at an interval of 24 h.
The result showed that the IFNγ expression after the first irradiation was the highest, then decreased in a time-dependent manner. The expressions after the first 2 times of irradiation were higher than that in control group (P < 0.01-0.001) (Figure 6).
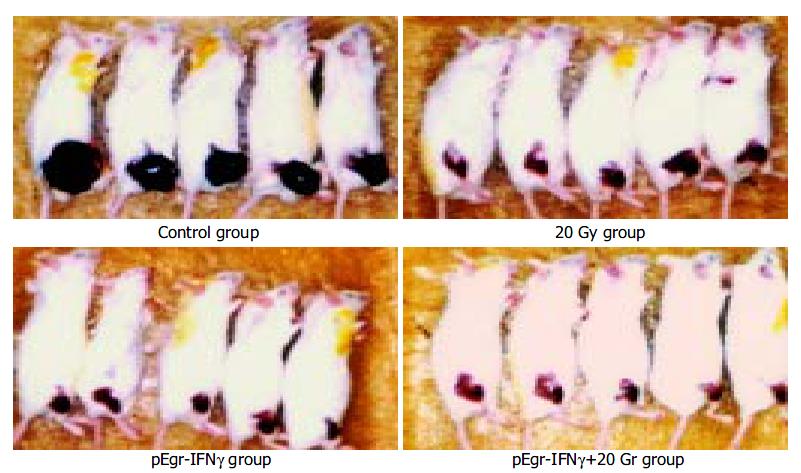
Melanoma-bearing mice of different groups were shown in Figure 7.
Tumor growth rate of pEgr-IFNγ group was significantly slower than that of control group (P < 0.001) between 6 d and 15 d after irradiation (Table 2), so was pEgr-IFNγ plus 20 Gy group compared with control and 20 Gy groups between 3 d and 15 d after irradiation (P < 0.001).
| Group | f (V/V0) on days after irradiation | ||||
| 3 d | 6 d | 9 d | 2 d | 15 d | |
| Control | 1.23 ± 0.37 | 3.11 ± 1.5 | 12.29 ± 4.83 | 20.21 ± 7.62 | 22.80 ± 8.50 |
| 20 Gy | 1.57 ± 0.19 | 2.34 ± 0.40 | 3.28 ± 0.68b | 4.18 ± 0.66b | 6.18 ± 1.40b |
| PcDNA3.1 + 20 Gy | 1.86 ± 0.54 | 1.67 ± 0.40ad | 2.26 ± 0.50bd | 2.86 ± 0.58bd | 5.19 ± 0.66b |
| PEgr-IFNγ | 1.38 ± 0.23 | 1.76 ± 0.56b | 2.61 ± 0.75b | 5.23 ± 0.98b | 8.03 ± 2.14b |
| PEgr-IFNγ + 20 Gy | 0.48 ± 0.10bf | 0.34 ± 0.11bf | 0.38 ± 0.14bf | 0.43 ± 0.11bf | 0.35 ± 0.10bf |
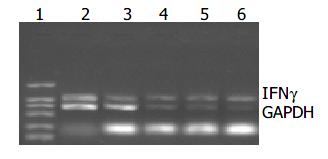
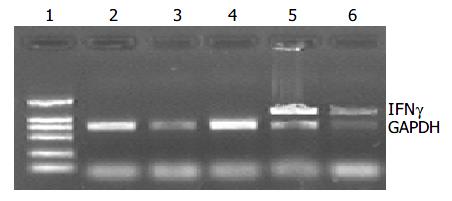
Melanoma-bearing mice were injected with plasmids, and the tumor received 20 Gy X-ray irradiation, 3 d later total RNA from tumor tissue was extracted for RT-PCR.
GAPDH bands were shown in all groups, but IFNγ cDNA bands were shown only in pEgr-IFNγ and pEgr-IFNγ + 20 Gy groups (Figure 8).
In 1992 Weichselbaum put forward the new therapeutic strategy that took advantage of the dual tumor-killing effects of genetherapy and radiotherapy, namely, to choose certain exogenous genes that could be activated by irradiation and then transcript some cytotoxic proteins to kill the tumor cells. They also established the techniques that might be used for target gene therapy of carcinomas[19-25].
It had been reported that Egr-1 was transcriptionally induced by exposure to irradiation, and its induction by irradiation was conferred by serum response or CC (A/T) rGG elements in its promoter region[26-30]. Based on this finding, we firstly connected IFNγ cDNA with Egr-1 promoter to construct pEgr-IFNγ plasmid to investigate the expression properties in B16 melanoma cells. Furthermore, a melanoma model was established by subcutaneous injection of B16 cells, and then plasmids were injected to observe its antitumor effect in vivo.
Firstly, B16 cells transfected with pEgr-IFNγ received different doses of X-ray irradiation and the IFNγ expression was detected. The results showed that the IFNγ expression level in B16 cells post-transfection induced by irradiation was higher than that of sham-irradiation group (P < 0.05-0.01). Time-course studies revealed that IFNγ expression reached its peak at 6 h after 2Gy irradiation, and the maximal level was 1.8 times of that in control group (P < 0.01). Furthermore, after repeated irradiation the IFNγ expression in B16 cells post-transfection reached the peak level just after the first irradiation, and then decreased in time-dependent manner. All of these demonstrated that pEgr-IFNγ plasmid could enhance IFNγ expression.
Secondly, the results of in vivo experiments showed that the proliferation of melanoma was significantly inhibited in pEgr-IFNγ group in comparison with control group between 6 and 15 d after irradiation (P < 0.001). So was pEgr-IFNγ gene-radiotherapy group compared with control and 20 Gy groups between 3 and 15 d after irradiation (P < 0.001). IFNγ expression was detected in melanoma tissue having receivied injection of pEgr-IFNγ plasmid. These results demonstrated that injection of pEgr-IFNγ improved antitumor effect, and combined pEgr-IFNγ and irradiation showed the most optimal effect.
This study combined pEgr-IFNγ plasmid and irradiation, and demonstrated much more enhanced antitumor efficacy than either one in the melanoma model. It was very easy to administer directly the plasmid into melanoma tissue, and therapeutic dose could also be administered as required. The tumor could be effectively exposed to radiation with external beam or intratumoral sources, or both, to enhance local IFNγ expression and boost local tumor control. Increased IFNγ levels might elicit systemic mediators, such as cytokines and matrix proteinases, which target occult distant metastases and thereby further enhance the therapeutic ratio. The absence of systemic toxicities with intratumoral administration of IFNγ supports the safe addition of pEgr-IFNγ gene radiotherapy to current antitumor protocols.
Edited by Ren SY and Wang XL Proofread by Xu FM
| 1. | Weichselbaum RR, Hallahan DE, Beckett MA, Mauceri HJ, Lee H, Sukhatme VP, Kufe DW. Gene therapy targeted by radiation preferentially radiosensitizes tumor cells. Cancer Res. 1994;54:4266-4269. [PubMed] |
| 2. | Liu XF, Zou SQ, Qiu FZ. Construction of HCV-core gene vector and its expression in cholangiocarcinoma. World J Gastroenterol. 2002;8:135-138. [PubMed] |
| 3. | Guo WJ, Yu EX, Liu LM, Li J, Chen Z, Lin JH, Meng ZQ, Feng Y. Comparison between chemoembolization combined with radiotherapy and chemoembolization alone for large hepatocellular carcinoma. World J Gastroenterol. 2003;9:1697-1701. [PubMed] |
| 4. | Ido A, Uto H, Moriuchi A, Nagata K, Onaga Y, Onaga M, Hori T, Hirono S, Hayashi K, Tamaoki T. Gene therapy targeting for hepatocellular carcinoma: selective and enhanced suicide gene expression regulated by a hypoxia-inducible enhancer linked to a human alpha-fetoprotein promoter. Cancer Res. 2001;61:3016-3021. [PubMed] |
| 5. | Datta R, Rubin E, Sukhatme V, Qureshi S, Hallahan D, Weichselbaum RR, Kufe DW. Ionizing radiation activates transcription of the EGR1 gene via CArG elements. Proc Natl Acad Sci U S A. 1992;89:10149-10153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 152] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 6. | Tsai-Morris CH, Cao XM, Sukhatme VP. 5' flanking sequence and genomic structure of Egr-1, a murine mitogen inducible zinc finger encoding gene. Nucleic Acids Res. 1988;16:8835-8846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 128] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 7. | Lokshin A, Mayotte JE, Levitt ML. Mechanism of interferon beta-induced squamous differentiation and programmed cell death in human non-small-cell lung cancer cell lines. J Natl Cancer Inst. 1995;87:206-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 8. | Shiau AL, Lin CY, Tzai TS, Wu CL. Postoperative immuno-gene therapy of murine bladder tumor by in vivo administration of retroviruses expressing mouse interferon-gamma. Cancer Gene Ther. 2001;8:73-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 9. | Siesjö P, Visse E, Sjögren HO. Cure of established, intracerebral rat gliomas induced by therapeutic immunizations with tumor cells and purified APC or adjuvant IFN-gamma treatment. J Immunother Emphasis Tumor Immunol. 1996;19:334-345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 46] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 10. | Saleh M, Jonas NK, Wiegmans A, Stylli SS. The treatment of established intracranial tumors by in situ retroviral IFN-gamma transfer. Gene Ther. 2000;7:1715-1724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 11. | Li XM, Chopra RK, Chou TY, Schofield BH, Wills-Karp M, Huang SK. Mucosal IFN-gamma gene transfer inhibits pulmonary allergic responses in mice. J Immunol. 1996;157:3216-3219. [PubMed] |
| 12. | Fujinami K, Ikeda I, Miura T, Kondo I. [Combination therapy with 5-fluorouracil (5-FU), cisplatin (CDDP) and interferon alpha-2B (IFN alpha-2B) for advanced renal cell carcinoma]. Gan To Kagaku Ryoho. 1996;23:1689-1691. [PubMed] |
| 13. | Yeow WS, Lawson CM, Beilharz MW. Antiviral activities of individual murine IFN-alpha subtypes in vivo: intramuscular injection of IFN expression constructs reduces cytomegalovirus replication. J Immunol. 1998;160:2932-2939. [PubMed] |
| 14. | Ahn EY, Pan G, Vickers SM, McDonald JM. IFN-gammaupregulates apoptosis-related molecules and enhances Fas-mediated apoptosis in human cholangiocarcinoma. Int J Cancer. 2002;100:445-451. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 61] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 15. | Blanck G. Components of the IFN-gamma signaling pathway in tumorigenesis. Arch Immunol Ther Exp (Warsz). 2002;50:151-158. [PubMed] |
| 16. | Nayak SK, McCallister T, Han LJ, Gangavalli R, Barber J, Dillman RO. Transduction of human renal carcinoma cells with human gamma-interferon gene via retroviral vector. Cancer Gene Ther. 1996;3:143-150. [PubMed] |
| 17. | Tada H, Maron DJ, Choi EA, Barsoum J, Lei H, Xie Q, Liu W, Ellis L, Moscioni AD, Tazelaar J. Systemic IFN-beta gene therapy results in long-term survival in mice with established colorectal liver metastases. J Clin Invest. 2001;108:83-95. [PubMed] |
| 18. | Paradis TJ, Floyd E, Burkwit J, Cole SH, Brunson B, Elliott E, Gilman S, Gladue RP. The anti-tumor activity of anti-CTLA-4 is mediated through its induction of IFN gamma. Cancer Immunol Immunother. 2001;50:125-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 29] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 19. | Weichselbaum RR, Hallahan DE, Sukhatme VP, Kufe DW. Gene therapy targeted by ionizing radiation. Int J Radiat Oncol Biol Phys. 1992;24:565-567. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 20. | Weichselbaum RR, Kufe DW, Advani SJ, Roizman B. Molecular targeting of gene therapy and radiotherapy. Acta Oncol. 2001;40:735-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 21. | Khodarev NN, Park JO, Yu J, Gupta N, Nodzenski E, Roizman B, Weichselbaum RR. Dose-dependent and independent temporal patterns of gene responses to ionizing radiation in normal and tumor cells and tumor xenografts. Proc Natl Acad Sci U S A. 2001;98:12665-12670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 22. | Gupta VK, Park JO, Jaskowiak NT, Mauceri HJ, Seetharam S, Weichselbaum RR, Posner MC. Combined gene therapy and ionizing radiation is a novel approach to treat human esophageal adenocarcinoma. Ann Surg Oncol. 2002;9:500-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 23. | Hanna NN, Seetharam S, Mauceri HJ, Beckett MA, Jaskowiak NT, Salloum RM, Hari D, Dhanabal M, Ramchandran R, Kalluri R. Antitumor interaction of short-course endostatin and ionizing radiation. Cancer J. 2000;6:287-293. [PubMed] |
| 24. | Takahashi T, Namiki Y, Ohno T. Induction of the suicide HSV-TK gene by activation of the Egr-1 promoter with radioisotopes. Hum Gene Ther. 1997;8:827-833. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 34] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 25. | Griscelli F, Li H, Cheong C, Opolon P, Bennaceur-Griscelli A, Vassal G, Soria J, Soria C, Lu H, Perricaudet M. Combined effects of radiotherapy and angiostatin gene therapy in glioma tumor model. Proc Natl Acad Sci U S A. 2000;97:6698-6703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 80] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 26. | Christy B, Nathans D. DNA binding site of the growth factor-inducible protein Zif268. Proc Natl Acad Sci U S A. 1989;86:8737-8741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 352] [Cited by in RCA: 419] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 27. | Seyfert VL, Sukhatme VP, Monroe JG. Differential expression of a zinc finger-encoding gene in response to positive versus negative signaling through receptor immunoglobulin in murine B lymphocytes. Mol Cell Biol. 1989;9:2083-2088. [PubMed] |
| 28. | Joseph LJ, Le Beau MM, Jamieson GA, Acharya S, Shows TB, Rowley JD, Sukhatme VP. Molecular cloning, sequencing, and mapping of EGR2, a human early growth response gene encoding a protein with "zinc-binding finger" structure. Proc Natl Acad Sci U S A. 1988;85:7164-7168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 208] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 29. | Sukhatme VP. Early transcriptional events in cell growth: the Egr family. J Am Soc Nephrol. 1990;1:859-866. [PubMed] |
| 30. | Cao XM, Koski RA, Gashler A, McKiernan M, Morris CF, Gaffney R, Hay RV, Sukhatme VP. Identification and characterization of the Egr-1 gene product, a DNA-binding zinc finger protein induced by differentiation and growth signals. Mol Cell Biol. 1990;10:1931-1939. [PubMed] |