Published online Apr 26, 2016. doi: 10.13105/wjma.v4.i2.10
Peer-review started: November 25, 2015
First decision: December 28, 2015
Revised: January 19, 2016
Accepted: March 9, 2016
Article in press: March 14, 2016
Published online: April 26, 2016
Processing time: 141 Days and 4.8 Hours
AIM: To review evidence relating passive smoking to lung cancer risk in never smokers, considering various major sources of bias.
METHODS: Epidemiological prospective or case-control studies were identified which provide estimates of relative risk (RR) and 95%CI for never smokers for one or more of seven different indices of exposure to environmental tobacco smoke (ETS): The spouse; household; workplace; childhood; travel; social and other; and total. A wide range of study details were entered into a database, and the RRs for each study, including descriptions of the comparisons made, were entered into a linked database. RRs were derived where necessary. Results were entered, where available, for all lung cancer, and for squamous cell cancer and adenocarcinoma. “Most adjusted” results were entered based on results available, adjusted for the greatest number of potential confounding variables. “Least adjusted” results were also entered, with a preference for results adjusted at least for age for prospective studies. A pre-planned series of fixed-effects and random-effects meta-analyses were conducted. Overall analyses and analyses by continent were run for each exposure index, with results for spousal smoking given by sex, and results for childhood exposure given by source of ETS exposure. For spousal exposure, more extensive analyses provide results by various aspects of study design and definition of the RR. For smoking by the husband (or nearest equivalent), additional analyses were carried out both for overall risk, and for risk per 10 cigarettes per day smoked by the husband. These adjusted for uncontrolled confounding by four factors (fruit, vegetable and dietary fat consumption, and education), and corrected for misclassification of smoking status of the wife. For the confounding adjustment, estimates for never smoking women were derived from publications on the relationship of the four factors to both lung cancer risk and at home ETS exposure, and on the correlations between the factors. The bias due to misclassification was calculated on the basis that the proportion of ever smokers denying smoking is 10% in Asian studies and 2.5% elsewhere, and that those who deny smoking have the same risk as those who admit it. This approach, justified in previous work, balances higher true denial rates and lower risk in deniers compared to non-deniers.
RESULTS: One hundred and two studies were identified for inclusion, published in 1981 onwards, 45 in Asia, 31 in North America, 21 in Europe, and five elsewhere. Eighty-five were of case-control design and 17 were prospective. Significant (P < 0.05) associations were noted, with random-effects of (RR = 1.22, 95%CI: 1.14-1.31, n = 93) for smoking by the husband (RR = 1.14, 95%CI: 1.01-1.29, n = 45) for smoking by the wife (RR = 1.22, 95%CI: 1.15-1.30, n = 47) for workplace exposure (RR = 1.15, 95%CI: 1.02-1.29, n = 41) for childhood exposure, and (RR = 1.31, 95%CI: 1.19-1.45, n = 48) for total exposure. No significant association was seen for ETS exposure in travel (RR = 1.34, 95%CI: 0.94-1.93, n = 8) or in social situations (RR = 1.01, 95%CI: 0.82-1.24, n = 15). A significant negative association (RR = 0.78, 95%CI: 0.64-0.94, n = 8) was seen for ETS exposure in childhood, specifically from the parents. Significant associations were also seen for spousal smoking for both squamous cell carcinoma (RR = 1.44, 95%CI: 1.15-1.80, n = 24) and adenocarcinoma (RR = 1.33, 95%CI: 1.17-1.51, n = 30). Results generally showed marked heterogeneity between studies. For smoking by either the husband or wife, where 119 RR estimates gave an overall estimate of (RR = 1.21, 95%CI: 1.14-1.29), the heterogeneity was highly significant (P < 0.001), with evidence that the largest RRs were seen in studies published in 1981-89, in small studies (1-49 cases), and for estimates unadjusted by age. For smoking by the husband, the additional analyses showed that adjustment for the four factors reduced the overall (RR = 1.22, 95%CI: 1.14-1.31) based on 93 estimates to (RR = 1.14, 95%CI: 1.06-1.22), implying bias due to uncontrolled confounding of 7%. Further correction for misclassification reduced the estimate to a marginally non-significant (RR = 1.08, 95%CI: 0.999-1.16). In the fully adjusted and corrected analyses, there was evidence of an increase in Asia (RR = 1.18, 95%CI: 1.07-1.30, n = 44), but not in other regions (RR = 0.96, 95%CI: 0.86-1.07, n = 49). Studies published in the 1980’s, studies providing dose-response data, and studies only providing results unadjusted for age showed elevated RRs, but later published studies, studies not providing dose-response data, and studies adjusting for age did not. The pattern of results for RRs per 10 cigs/d was similar, with no significant association in the adjusted and corrected results (RR = 1.03, 95%CI: 0.994-1.07).
CONCLUSION: Most, if not all, of the ETS/lung cancer association can be explained by confounding adjustment and misclassification correction. Any causal relationship is not convincingly demonstrated.
Core tip: We present an up-to-date meta-analysis of the evidence relating non-smoker lung cancer to environmental tobacco smoke (ETS) exposure. We demonstrate a clear risk increase for spousal, at-home, workplace and total exposure, but not childhood exposure. For husband smoking, the relative risk (RR) is estimated as (RR = 1.22, 95%CI: 1.14-1.31). However, adjustment for confounding by education and dietary variables, and correction for misclassified wife’s smoking reduces it to (RR = 1.08, 95%CI: 0.999-1.16). Given the other data limitations and biases we discuss, one cannot reliably conclude that any true ETS effect on lung cancer risk exists. Our results suggest caution in drawing inferences from weak epidemiological associations where known biases exist.
- Citation: Lee PN, Fry JS, Forey BA, Hamling JS, Thornton AJ. Environmental tobacco smoke exposure and lung cancer: A systematic review. World J Meta-Anal 2016; 4(2): 10-43
- URL: https://www.wjgnet.com/2308-3840/full/v4/i2/10.htm
- DOI: https://dx.doi.org/10.13105/wjma.v4.i2.10
It has been widely accepted that environmental tobacco smoke (ETS) exposure increases lung cancer risk, based on various authoritative reviews[1]. However, it was suggested some years ago[2] that a substantial part, if not all, of the relationship may be due to bias resulting from confounding by other lung cancer risk factors, and misclassification of smoking habits, with some self-reported never smokers actually being smokers. While there have been various meta-analyses of the evidence in the last 20 years[3-10]. these are often limited to specific indices of exposure or regions, and typically do not include formal adjustments for potential biases. They also do not take into account all the more recent studies, with over 100 studies published by now, many relatively recent.
The objective of this review, therefore, is to present an up-to-date comprehensive meta-analysis of the available evidence which relates ETS exposure to lung cancer risk among never smokers, considering exposure from various sources, and illustrating the potential magnitude of the bias that can arise from confounding and misclassification of smoking.
The analyses presented were conducted in three stages. First, results of meta-analysis are presented relating a range of indices of ETS exposure to risk of lung cancer. Second, for two indices (spousal smoking and amount smoked by the spouse), individual study estimates for females are adjusted for the effects of confounding for selected variables (fruit consumption, vegetable consumption, dietary fat consumption and education) and revised meta-analyses conducted. Third, further adjustments are made for the biasing effects of misclassification of smoking status. The materials and methods section is therefore divided accordingly.
Attention was restricted to epidemiological prospective or case-control studies published up to and including July 2015, which involved five or more lung cancers, and which provided relative risk (RR) estimates for never (or virtually never) smokers for one or more defined ETS exposure types or dose-related ETS indices. The “exposure types” compare subjects exposed and unexposed to ETS from seven different sources: Spouse; household; workplace; childhood; travel; social and other; and total, the final category including biochemical assessments of exposure. The “dose-related indices” concern ETS exposure in terms of amount smoked, duration of smoking and the number of smokers the subject was exposed to. ETS exposure from pipe/cigar only was ignored. Note that the term “relative risk” is taken to include estimates of it, such as the odds ratio or hazard ratio.
Studies using near equivalent definitions of “never smokers” were accepted when stricter definitions were unavailable, so never smokers could include occasional smokers, those with a minimal lifetime duration of smoking or number smoked, or ex-smokers who had quit at least 20 years previously.
Up until July 2015 potentially relevant papers were regularly sought from MEDLINE searches (using search terms “tobacco smoke pollution” and “lung neoplasm”), from files on smoking and health which were collected for many years within our company, and from references which were cited in the papers obtained. At the end of the process no paper examined cited a possibly relevant paper which had not been previously examined.
Relevant papers were separated into studies, noting where there were multiple papers per study or multiple studies per paper, and any overlaps between studies. Each study was uniquely referenced by a ≤ 6 character code, based on the name of the principal author, with a suffix indicating where the same author had reported on multiple studies.
Data were entered on a study database, and also on a linked RR database. The structure and content of the databases are described in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%201.pdf.
In brief, a study database record describes the study design, the available data and a previously described index of study quality[11]. Typically there are multiple records per study on the RR database, each record holding a detailed description of a specific comparison made and the corresponding RR and its 95%CI.
When available, adjusted RRs and CIs were entered. Unadjusted estimates were derived from the 2 × 2 table, using standard methodology[12], any differences between calculated and author-provided estimates being noted. Other methodologies were used where required to derive estimates, those more commonly used[13,14] being described in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%201.pdf.
RRs were entered, if available, relating to various pre-defined combinations of type of lung cancer, index of smoking, confounders considered, and strata. The combinations are described in the following sections.
Type of lung cancer: Results were entered for overall lung cancer, squamous cell carcinoma and adenocarcinoma, or their nearest equivalents for which data were available.
Smoking indices: The intention was to enter RRs comparing subjects exposed and unexposed to the various indices of ETS defined above. Though RRs for exposure to smoking by the spouse should ideally be derived from data only for married subjects, we also allowed RRs from studies where unmarried subjects were included in the reference group. Similarly, RRs for workplace exposure could include non-working subjects. For the “household” and “childhood” categories, RRs were entered for all possible sources recorded by the studies, but for the “travel” and “social” categories, if more than one index of exposure was available, only that representing the greatest number of exposed subjects was entered. RRs were entered for all available timings of adult exposure, but for childhood, only RRs for the earliest exposure were entered. “Total” exposure was defined as exposure to two or more types of exposure, or biochemical assessment of overall exposure. For dose-related exposure indices, RRs were entered for each level of exposure relative to a common base level. RRs were entered, where available, using denominators representing both “no exposure to the specific type of ETS” and “no exposure to any ETS”.
Confounders: For case-control studies, we entered results adjusted for the most potential confounders available, and also adjusted for fewest. For prospective studies, we entered results adjusted for age and the most confounders, and for age and the fewest, and unadjusted results were entered only where there were no age-adjusted results. We describe these alternative RRs as “most-adjusted” and “least-adjusted”.
Strata: We only entered results stratified by sex or age. Combined sex results were only entered if results by sex were unavailable. We entered results for all ages and for separate age groups. Specifically for spousal exposure (or nearest equivalent - see “analyses conducted” below), where an adjusted RR was available only for combined sexes but numbers of cases and controls were given by sex, split-sex estimates were entered, assuming that the RR applied to each sex, with separate CIs estimated for males and females.
Analyses conducted: The series of meta-analyses conducted was pre-planned. For a given exposure type, a set of up to 20 analyses was conducted. Meta-analyses 1 and 2 used the overall data available, while meta-analyses 3 and 4 were separated by region (North America, Europe, Asia or other regions), with meta-analyses 1 and 3 using most-adjusted and 2 and 4 least-adjusted data. Analyses 5-20 were based on most-adjusted data only and studied variation by other factors, as described in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%201.pdf.
The primary index of exposure used was “spousal smoking (or nearest equivalent)” where, for studies which provided no results for spousal exposure, results for household, total or both spousal/home and other exposure were chosen instead. This identified a single exposure definition for each study. For overall lung cancer, the full set of 20 meta-analyses was carried out restricted to females, and unrestricted on sex. Further meta-analyses for the principal index of exposure corresponded to meta-analyses 1 to 4 only. These included analyses for spousal smoking (or nearest equivalent) for males, spousal smoking (specifically) for females, males and unrestricted on sex, and analyses for spousal smoking (or nearest equivalent) for squamous cell carcinoma and for adenocarcinoma, each for females, males and unrestricted on sex.
Analyses for the other types of exposure were run only for overall lung cancer, without restriction on sex, and were equivalent to meta-analyses 1-4 only. The childhood and household exposure analyses were run using alternative indices, depending on the available data, as described in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%201.pdf.
Selecting RRs for the meta-analyses: In selecting RRs to include we tried to include all relevant data once only. Where a study had multiple RRs, that used is chosen by an order of preference specific to the meta-analysis. Order of preference may be needed for exposure status and timing, and for the unexposed base. As RR definitions may be sex-specific, the RRs selected may differ by sex. Results for sexes combined are only considered in the absence of sex-specific results.
Conducting the meta-analyses for exposure indices: We conducted fixed-effect and random-effects meta-analysis of study-specific data for the various exposure indices studied as described elsewhere[13]. Heterogeneity was measured by H, the ratio of heterogeneity χ2 to degrees of freedom. H relates to I2 statistic[15] by I2 = 100 (H - 1)/H. For all meta-analyses, results of publication bias tests using the Egger method[16] were also given.
Results are displayed in forest plots. Within each plot, studies are identified by their reference code, and listed in order of RR. Most of the plots are also grouped by region. The study estimates are shown both as numbers and in graphical form logarithmically. In the latter representation an RR is shown as a square, the area of which is proportional to its weight, its inverse-variance. Arrows warn if the CI goes outside the range of the plot. Random-effects estimates are also presented, overall or by region, shown by a diamond whose width indicates the 95%CI.
Carrying out meta-analyses for number of cigarettes smoked by the husband: The methods used are as described elsewhere[17], and are summarized here. The underlying model assumes that, when comparing two groups differing in exposure by dose d, log RR is estimated by βd. For each study, given data at each level of exposure consisting of the dose level, the number of cases, and the number of controls (or subjects at risk), β and its standard error (SEβ) are estimated by the method of Greenland and Longnecker[18], This can be applied to studies with only two levels (unexposed and exposed), and also to confounder-corrected RRs and 95%CIs, by estimating pseudo-counts using the method of Hamling[14]. Estimates of β and SEβ from each study are then meta-analysed as described above. The method of estimating midpoint doses for intervals such as 1-19 or 20+ cigarettes per day is as described previously[17].
The series of meta-analyses conducted for the estimates of β was similar to that for the exposure indices as described above.
The potential confounding variables considered (consumption of fruit, consumption of vegetables, consumption of dietary fat, and education) and the methods used to adjust for them are as described in a previous publication[19] and in an unpublished updated analysis conducted in 2006[20]. The methods are summarized briefly below.
Estimating the relationship between the four potential confounding variables considered and the risk of lung cancer in never smoking females: The database of studies relating lung cancer risk in never smokers to the four potential confounding variables used in 2001[19] and updated in 2006[20] was used, restricting attention to never smoking females. Exp(β), the increase in risk per dose unit (SDs for dietary variables, years for education), was estimated using methodology equivalent to that described in the previous section. Methods for assigning midpoint dose values for grouped dietary data (e.g., by quintiles), and for education groups were as before[19].
Estimating the differences in fruit, vegetable and fat consumption and in education associated with ETS exposure: The database of studies comparing the distribution of the four potential confounding variables set up in 2001[19] and updated in 2006[20] was used, with attention restricted to never smoking females. For each potential confounding variable, we estimated the difference, δ, in relation to marriage to, or living with, a smoker, in units of SDs for the dietary variables and years for education, using the methodology described earlier[19]. These study-specific estimates of δ were then combined using random-effects meta-analysis.
The basic method for confounder adjustment: As described earlier[19] we assume that the logarithm of L, the lung cancer risk, is linearly related to n explanatory factors xi by:
log L = βo* + ∑inβi*xi (1)
Exp(βo*) is the background risk that is expected for zero exposure to each factor. Exp (βi*) is the multiplicative risk increase expected per unit increase in exposure to the ith factor.
Should data relating lung cancer to the factors be available only univariately the relationship with each factor would be formulated as:
log L = βo + βixi (2)
where exp(βi) is the RR for a unit dose increase associated with factor i that is not adjusted for the other risk factors.
The βi and the βi* are related by the matrix equation:
B* = S-1C-1SB (3)
Here B* and B are the n × 1 vectors of and βi, S the n x n standard deviation (SD) matrix, si, and C the n x n correlation matrix cij.
Given B, C and S, we can estimate B*. In our context, there are five factors. i = 1 represents ETS with I = 2…5 the three dietary variables and education. Thus exp(βi) is the unadjusted risk increase for each unit of increase in ETS exposure, and exp(βi*) the adjusted risk increase. The joint confounding effect of the four variables is estimated as exp(βi)/exp(βi*).
Relationship of the factors to lung cancer risk: Estimates of βi are generally those described in the sub-section “carrying out meta-analyses for number smoked by the husband”. However, the basic method assumes that βi is unadjusted for any of the four potential confounding variables. Where βi is adjusted for one or more of the variables, we first back-corrected it in order to take out the effect of the adjustment as described earlier[19]. This back-correction procedure avoids double-adjustment for the same factor. Back-correction was also carried out in the following cases: For fruit consumption, where the RR estimate had already been adjusted for vitamin C; for dietary fat, where the RR estimate had already been adjusted for energy intake, for meat, or for cholesterol; and for education, where the RR estimate had already been adjusted for income, for socioeconomic status, or for ownership of a colour TV.
Standard deviations: We estimated s1, the SD for ETS, directly for each study from the population data by level of exposure as described elsewhere[17]. s2, s3 and s4, the SDs for the dietary variables are 1, since they are measured in units of SD. We took the SD for education as 2.435 years[20].
Correlations: If i = j, cij = 1. To quantify other correlations, we used the formula:
Cij = δjs1/d1(average)sj (4)
Here δj is a common estimate of the difference in exposure to variable j for living with a smoker (see the sub-section “estimating the differences in fruit…”). s1 and sj are as described above, and d1(average) is the mean ETS exposure for exposed never smokers. Where studies have more than one exposure level, we estimated d1(average) by weighting on the number of exposed subjects.
To quantify the correlations between the potential confounding variables we used averaged data from seven databases, the five used in 2001[19] and two additional US databases (NHIS2000, NHANES III), as described in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%202.pdf.
How the bias arises and what it depends on: Estimates of the RR of lung cancer in self-reported never smoking women associated with marriage to a smoker may be biased if a proportion of the women are actually current or ex-smokers. This bias arises because smokers marry smokers more often than is expected by chance. Misclassified smokers are therefore commoner among those married to a smoker. As shown by Lee and Forey[21], the bias depends mainly on the rate of misclassification, the active smoking risk, the degree to which smoking by spouses is concordant, and the proportions of smokers among subjects and their spouses.
Correction method used: We use the Lee and Forey[21] method for bias correction, assuming joint effects of active smoking and ETS exposure are additive, and the published extension of the method[22].
Concordance ratio: The concordance ratio is defined as the odds of the husband smoking if the wife ever smoked divided by the odds if the wife never smoked. From an earlier review[23] we used an estimate of 3.0.
Study-specific data on active smoking RRs: For each study, estimates were made of the active smoking RR, derived if possible from the source paper itself or another paper using the same study population. Otherwise they were derived from studies in that country, from estimates presented by the EPA[24] or by other methods, as described in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%202.pdf.
Misclassification rates: Misclassified smokers have a lower lung cancer risk than non-misclassified smokers. To take this into account, we followed precedent[21,22] in carrying out the misclassification correction on the basis that those who deny smoking have the same risk as those who admit it, but using lower misclassification rates (10% for Asia, and 2.5% elsewhere) than are observed. Support for the use of these rates is provided elsewhere[21-23,25,26].
Application of the method: RRs for spousal smoking and for amount smoked by the spouse, were calculated: (1) with no adjustment for confounding or correction for misclassification; (2) with adjustment for confounding and no correction for misclassification; and (3) with adjustment for confounding and correction for misclassification.
There were 102 studies which met the inclusion criteria. Some studies were noted to have overlaps with other studies. However, as all overlaps were minor and could not be disentangled, it was decided to ignore them. Tables 1 and 2 give study details including reference(s), location, design, dates, numbers of cases in never smokers, definition of never smoking, ETS exposure measures considered, adjustment variables used, extent of histological confirmation of cases, whether results are available by histological type, and availability of dose-response data. http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%203.pdf describes why other publications which could be thought possibly relevant are not considered in our analyses.
| Study ref | Main ref | Other ref | Location | Study design1 | Study dates2 | Total cases | Never smoker criteria3 | ||
| GARFI1 | [75] | [104] | United States | P | 1959 - | 1960, | 1971 | 153 | Dur 6 mo |
| CHAN | [105] | [106,107] | Hong Kong | CC | 1976 - | 1977 | 84 | ||
| CORREA | [67] | United States | CC | 1979 - | 1982 | 32 | |||
| TRICHO | [27] | [108,109] | Greece | CC | 1978 - | 1982 | 77 | ||
| BUFFLE | [110] | United States | CC | 1976 - | 1980 | 52 | |||
| HIRAYA | [28] | [111-120] | Japan | P | 1965 - | 1966, | 1981 | 264 | |
| KABAT1 | [53] | United States | CC | 1971 - | 1980 | 76 | |||
| GARFI2 | [121] | [122] | United States | CC | 1971 - | 1981 | 134 | ||
| LAMW | [29] | Hong Kong | CC | 1981 - | 1984 | 75 | |||
| WU | [123] | United States | CC | 1981 - | 1982 | 31 | |||
| AKIBA | [68] | Japan | CC | 1971 - | 1980 | 113 | |||
| LEE | [124] | United Kingdom | CC | 1979 - | 1982 | 47 | |||
| BROWN1 | [125] | United States | CC | 1979 - | 1982 | 19 | |||
| GAO | [126] | China | CC | 1984 - | 1986 | 246 | |||
| HUMBL1 | [127] | United States | CC | 1980 - | 1984 | 28 | Dur 6 mo | ||
| KOO | [128] | [129-133] | Hong Kong | CC | 1981 - | 1983 | 88 | N20 | |
| LAMT | [30] | Hong Kong | CC | 1983 - | 1986 | 199 | Occ (1 yr) | ||
| PERSHA | [134] | [135] | Sweden | CC | 1961 - | 1980 | 77 | ||
| BUTLER | [136] | United States | P | 1976 - | 1976, | 1982 | 8 | ||
| GENG | [31] | [137] | China | CC | 1983 - | 1983 | 54 | ||
| INOUE | [138] | Japan | CC | 1973 - | 1983 | 28 | |||
| SHIMIZ | [70] | Japan | CC | 1982 - | 1985 | 90 | |||
| CHOI | [139] | South Korea | CC | 1985 - | 1988 | 88 | |||
| HOLE | [140] | [141] | Scotland | P | 1972 - | 1976, | 1985 | 9 | |
| SCHOEN | [142] | United States | CC | 1982 - | 1983 | 116 | |||
| SVENSS | [143] | [144] | Sweden | CC | 1983 - | 1985 | 34 | Occ | |
| JANERI | [72] | [145] | United States | CC | 1982 - | 1985 | 191 | N100 | |
| KALAND | [32] | Greece | CC | 1987 - | 1989 | 91 | |||
| SOBUE | [146] | [147] | Japan | CC | 1986 - | 1988 | 144 | ||
| WU-WIL | [46] | China | CC | 1985 - | 1987 | 417 | |||
| LIUZ | [148] | [149,150] | China | CC | 1985 - | 1986 | 54 | 1sm | |
| BROWN2 | [151] | [152-155] | United States | CC | 1986 - | 1991 | 432 | ||
| STOCKW | [58] | [156] | United States | CC | 1987 - | 1991 | 210 | N100, Dur 6 mo | |
| DU | [52] | [157-159] | China | CC | 1986 - | 1986 | 75 | ||
| LIUQ | [160] | China | CC | 1983 - | 1984 | 38 | |||
| FONTHA | [33] | [161-164] | United States | CC | 1986 - | 1988 | 653 | N100, Dur 6 mo | |
| LAYARD | [165] | United States | CC | 1986 - | 1986 | 60 | |||
| DEWAAR | [166] | [167] | Netherlands | CC | 1977 - | 1991 | 23 | Cot | |
| KABAT2 | [168] | [169,170] | United States | CC | 1983 - | 1990 | 110 | ||
| SCHWAR | [57] | United States | CC | 1984 - | 1987 | 257 | |||
| SUN | [60] | China | CC | NA | 230 | ||||
| WANGS | [34] | China | CC | NA | 82 | ||||
| WANGT | [171] | China | CC | 1992 - | 1994 | 135 | |||
| CARDEN | [73] | [172,173] | United States | P | 1982 - | 1982, | 1989 | 362 | |
| ZHENG | [35] | China | CC | 1990 - | 1993 | 94 | Non | ||
| AUVINE | [174] | Finland | CC | 1986 - | 1992 | 44 | |||
| BOFFET | [66] | [175-180] | West Europe | CC | 1988 - | 1994 | 640 | N400 | |
| SHEN | [181] | [182-185] | China | CC | 1993 - | 1993 | 70 | ||
| ZARIDZ | [36] | [186-188] | Russia | CC | 1991 - | 1993 | 189 | ||
| BOFFE2 | [189] | Europe | CC | 1994 - | 1996 | 70 | N400 | ||
| JEE | [190] | South Korea | P | 1992 - | 1994, | 1997 | 79 | Occ, Dur 1 yr | |
| RAPITI | [61] | India | CC | 1991 - | 1992 | 58 | N400 | ||
| SPEIZE | [191] | United States | P | 1982 - | 1982, | 1992 | 35 | ||
| ZHONG | [54] | China | CC | 1992 - | 1994 | 504 | Dur 6 mo | ||
| LEECH | [37] | [192-194] | Taiwan | CC | 1992 - | 1998 | 268 | N400 | |
| MALATS | [195] | [196] | Europe/Brazil | CC | NA | 122 | Occ | ||
| WANGL | [62] | [197] | China | CC | 1994 - | 1998 | 233 | Dur 6 mo | |
| JOHNSO | [198] | [199-201] | Canada | CC | 1994 - | 1997 | 71 | N100 | |
| LAGARD | [202] | [203] | Sweden | CC | 1980 - | 1995 | 433 | Occ (1 yr) | |
| NISHIN | [204] | Japan | P | 1984 - | 1984, | 1992 | 24 | ||
| OHNO | [205] | Japan | CC | NA | 191 | N365 | |||
| RACHTA | [63] | [206] | Poland | CC | 1991 - | 1997 | 54 | Dur 6 mo | |
| ENSTRO | [207] | [208] | United States | P | 1959 - | 1960, | 1998 | 256 | |
| ZATLOU | [64] | [209,210] | Czech Republic | CC | 1998 - | 2002 | 84 | N100 | |
| IARCKR | [1] | [180] | Germany | CC | 1990 - | 1996 | 123 | Dur 6 mo, N400 | |
| MCGHEE | [211] | Hong Kong | CC | 1998 - | 1998 | 324 | |||
| EPICA | [212] | [213-216] | Western Europe | P | 1993 - | 1998, | 2000 | 59 | |
| FANG | [38] | [216] | China | CC | 2001 - | 2004 | 157 | ||
| FRANCO | [71] | [216] | Mexico | CC | 2000 - | 2002 | 94 | ||
| GORLOV | [55] | [217-222] | United States | CC | 1995 - | 2003 | 193 | N100 | |
| NEUBER | [49] | United States | CC | 1994 - | 1997 | 56 | N100, Dur 6 mo | ||
| RYLAND | [223] | Sweden | CC | 1989 - | 1994 | 49 | |||
| WEN | [56] | [224-227] | China | P | 1997 - | 2000, | 2004 | 106 | |
| YU | [228] | [228-230] | Hong Kong | CC | 2002 - | 2004 | 213 | N400, Dur 1 yr | |
| ZEKA | [59] | East Europe, United Kingdom | CC | 1998 - | 2002 | 223 | N100 | ||
| HILL1 | [231] | New Zealand | P | 1981 - | 1981, | 1984 | 147 | Occ | |
| HILL2 | [231] | New Zealand | P | 1996 - | 1996, | 1999 | 234 | Occ | |
| LOPEZC | [232] | Spain | CC | 2000 - | 2005 | 36 | N100 | ||
| ASOMAN | [233] | United States | CC | 1992 - | NA | 138 | Occ (1 yr) | ||
| GALLEG | [51] | Mexico | CC | 2003 - | 2007 | 32 | |||
| KURAHA | [234] | Japan | P | 1990 - | 1993, | 2004 | 109 | ||
| PANDEY | [50] | Nepal | CC | NA | 268 | ||||
| YANG | [39] | [65,221,235,236] | United States | CC | 1997 - | 2008 | 297 | N100 | |
| OLIVOM | [65] | [237] | United States | CC | NA | 45 | N100 | ||
| TSE | [238] | [239] | China | CC | 2004 - | 2006 | 132 | N400, Dur 1 yr | |
| LIANG | [40] | China | CC | 2004 - | 2007 | 226 | |||
| BRENNE | [47] | Canada | CC | 1997 - | 2002 | 156 | N100 | ||
| JIANG | [41] | China | CC | 2009 - | 2009 | 145 | |||
| EPICC | [240] | [212] | Western Europe | P | 1992 - | 1998, | 2006 | 98 | |
| KIYOHA | [241] | Japan | CC | 1996 - | 2008 | 153 | |||
| HE | [242] | [243] | China | P | 1976 - | 1994, | 2011 | 16 | N100 |
| LIM | [74] | [244-246] | China | CC | 1996 - | 2008 | 433 | Occ (1 yr) | |
| LIN | [42] | China | CC | 2006 - | 2010 | 226 | |||
| FERREC | [247] | Chile | CC | 2007 - | 2010 | 59 | |||
| ALZOUG | [48] | [248,249] | Canada | CC | 1996 - | 2000 | 44 | N100 | |
| GELAC | [43] | Taiwan | CC | 2002 - | 2009 | 1540 | Occ | ||
| MASJED | [44] | [250] | Iran | CC | 2002 - | 2005 | 81 | Dur 6 mo | |
| REN | [251] | China | CC | 2002 - | 2012 | 764 | |||
| SEKI | [252] | Japan | CC | 1997 - | 2009 | 431 | |||
| WHIOS | [253] | [254] | United States | P | 1993 - | 1998, | 2009 | 200 | |
| ILCCO | [45] | [69] | International | CC | 1984 - | 2014 | 2504 | N100 | |
| TORRES | [255] | [256] | Spain | CC | 2011 - | 2013 | 192 | N100 | |
| Ref. | ETS exposures1 | Adjustment variables used2 | Extent (%) of histological confirmation | Results by histological type | Dose-response results available3 |
| GARFI1 | s | 7 | NA | Yes | |
| CHAN | s | 0 | 80 | ||
| CORREA | s c4 | 1 | 97 | Yes | |
| TRICHO | s | 0 | 27 | Yes | Yes |
| BUFFLE | h | 0 | 100 | Yes | |
| HIRAYA | s | 2 | NA | Yes | |
| KABAT1 | s h w | 4 | 100 | ||
| GARFI2 | s h w c o tot | 4 | 100 | Yes | Yes |
| LAMW | s tot | 1 | 100 | Yes | |
| WU | s w c tot4 | 2 | 100 | Yes | |
| AKIBA | s c | 6 | 53 | Yes | |
| LEE | s h w tr o tot | 3 | 38 | Yes | |
| BROWN1 | tot | 3 | 100 | Yes | |
| GAO | s h c tot | 2 | 43 | Yes | |
| HUMBL1 | s | 2 | 100 | Yes | |
| KOO | s h w c tot | 5 | 97 | Yes | Yes |
| LAMT | s | 0 | 100 | Yes | Yes |
| PERSHA | s c | 2 | 83 | Yes | Yes |
| BUTLER | s | 2 | 100 | ||
| GENG | s | 0 | 85 | Yes | |
| INOUE | s | 3 | NA | Yes | |
| SHIMIZ | s h w | 3 | 100 | ||
| CHOI | s | 0 | 100 | Yes | |
| HOLE | h | 2 | NA | Yes | |
| SCHOEN | s | 6 | 100 | ||
| SVENSS | h c tot | 1 | 70 | ||
| JANERI | s h w c o | 3 | 100 | Yes | Yes |
| KALAND | s h w | 5 | 48 | Yes | Yes |
| SOBUE | s h c | 3 | 100 | ||
| WU-WIL | s h w c | 5 | 42 | ||
| LIUZ | h | 3 | 17 | ||
| BROWN2 | s h w c | 2 | 76 | Yes | Yes |
| STOCKW | s h w4 c o4 tot | 3 | 100 | Yes | Yes |
| DU | s | 2 | NA | Yes | |
| LIUQ | s | 3 | 32 | Yes | |
| FONTHA | s h w c o tot | 10 | 100 | Yes | Yes |
| LAYARD | s | 3 | NA | Yes | |
| DEWAAR | tot | 0 | 71 | Yes | |
| KABAT2 | s h w c tr o tot | 6 | 100 | Yes | |
| SCHWAR | h w | 2 | 100 | ||
| SUN | s h w c hw tot | 2 | 100 | Yes | |
| WANGS | tot | 0 | 100 | ||
| WANGT | s w c | 1 | 57 | Yes | |
| CARDEN | s h w o tot | 8 | NA | Yes | |
| ZHENG | h | 2 | 82 | Yes | Yes |
| AUVINE | tot | 1 | NA | ||
| BOFFET | s h w c tr o tot | 7 | 96 | Yes | Yes |
| SHEN | tot | 9 | 100 | Yes | Yes |
| ZARIDZ | s h w c | 2 | 100 | Yes | Yes |
| BOFFE2 | s w c tot | 5 | 100 | Yes | Yes |
| JEE | s | 5 | 0 | Yes | |
| RAPITI | s w c tr | 3 | 100 | Yes | Yes |
| SPEIZE | tot | 1 | 100 | ||
| ZHONG | s h w c tot | 7 | 57 | Yes | Yes |
| LEECH | s h w c tot | 7 | 100 | Yes | |
| MALATS | s tot | 2 | 100 | Yes | |
| WANGL | h c | 6 | 32 | Yes | |
| JOHNSO | h w c tot | 4 | 100 | Yes | |
| LAGARD | h tot | 6 | 100 | ||
| NISHIN | s h | 7 | NA | ||
| OHNO | s w c tr o tot | 2 | 100 | Yes | |
| RACHTA | c | 21 | 100 | Yes | |
| ENSTRO | s | 8 | NA | ||
| ZATLOU | c tot | 3 | 100 | Yes | |
| IARCKR | s w c | 2 | 100 | Yes | |
| MCGHEE | h | 2 | 0 | Yes | |
| EPICA | h w tot | 7 | NA | ||
| FANG | tot | 8 | 100 | Yes | |
| FRANCO | h | 2 | 100 | ||
| GORLOV | h w hw tot | 4 | 100 | ||
| NEUBER | tot4 | 3 | 100 | ||
| RYLAND | h w | 3 | 98 | ||
| WEN | s w c tot | 9 | NA | Yes | |
| YU | tot | 20 | 100 | Yes | |
| ZEKA | w | 4 | NA | Yes | |
| HILL1 | h | 9 | NA | ||
| HILL2 | h | 9 | NA | ||
| LOPEZC | tot | 0 | 100 | ||
| ASOMAN | h w o | 0 | 100 | ||
| GALLEG | tot | 0 | 100 | ||
| KURAHA | s w c hw tot | 5 | 90 | Yes | Yes |
| PANDEY | c tot | 0 | NA | Yes | |
| YANG | c tot | 5 | NA | ||
| OLIVOM | c | 4 | 100 | Yes | |
| TSE | h w tot | 9 | 100 | Yes | Yes |
| LIANG | c tot | 0 | 100 | ||
| BRENNE | h w c tot | 3 | 100 | Yes | |
| JIANG | tot | 17 | 100 | Yes | |
| EPICC | c | 10 | NA | Yes | |
| KIYOHA | s | 0 | 100 | ||
| HE | tot | 9 | 88 | Yes | |
| LIM | h | 0 | 96 | ||
| LIN | tot | 9 | 100 | Yes | |
| FERREC | c tot4 | 3 | 72 | ||
| ALZOUG | s h w c tot | 3 | NA | Yes | Yes |
| GELAC | s h w hw tot | 6 | 100 | Yes | |
| MASJED | s h w o tot | 4 | 100 | Yes | |
| REN | tot | 5 | 100 | ||
| SEKI | s | 7 | 94 | Yes | |
| WHIOS | h4 w4 c4 tot | 0 | 0 | 4 | |
| ILCCO | h w c tot | 3 | 100 | Yes | Yes |
| TORRES | h | 2 | 99 | Yes |
Of the 102 studies, 31 were conducted in North America (including 26 in United States), 45 in Asia (including 23 in China, 10 in Japan and 6 in Hong Kong), 21 in Europe (4 in Sweden being the most for any country), and 5 in other locations (including two international studies).
Eighty-five studies were of case-control design and 17 were prospective. Twenty-six studies were published in 1981-1989, 28 in 1990-1999, 32 in 2000-2009 and 16 in or after 2010.
In general, the total number of cases per study was small, with 20 studies based on less than 50 cases, and 29 considering 50-99. Twenty-four studies examined 100-199 cases, 18 200-399 cases, with only 11 based on 400 or more cases.
The most commonly studied index was smoking by the spouse, considered by 55 studies. Smoking by a cohabitant was considered by 47 studies, workplace smoking by 40, and childhood exposure by 41. Travel and social exposures were considered by 5 and 11 studies respectively, and total exposure by 51.
In what follows, meta-analysis RRs referred to in the text, tables and figures are based on “most-adjusted” estimates, meta-analysis RRs based on “least-adjusted” estimates usually being very similar. The results highlighted are drawn from more detailed analyses for all the exposure indices made available in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%204.pdf, which also shows the “preferences” used in each analysis. This includes some analyses based on “least-adjusted” estimates, and also gives estimates for each individual study included in an analysis. Significance is taken to be at P < 0.05 unless otherwise stated. RRs and 95%CIs are normally shown to 2 decimal places. Exceptionally, they are shown to 3 decimal places for the analyses investigating bias due to confounding and misclassification, to show the effects of adjustment and correction more clearly.
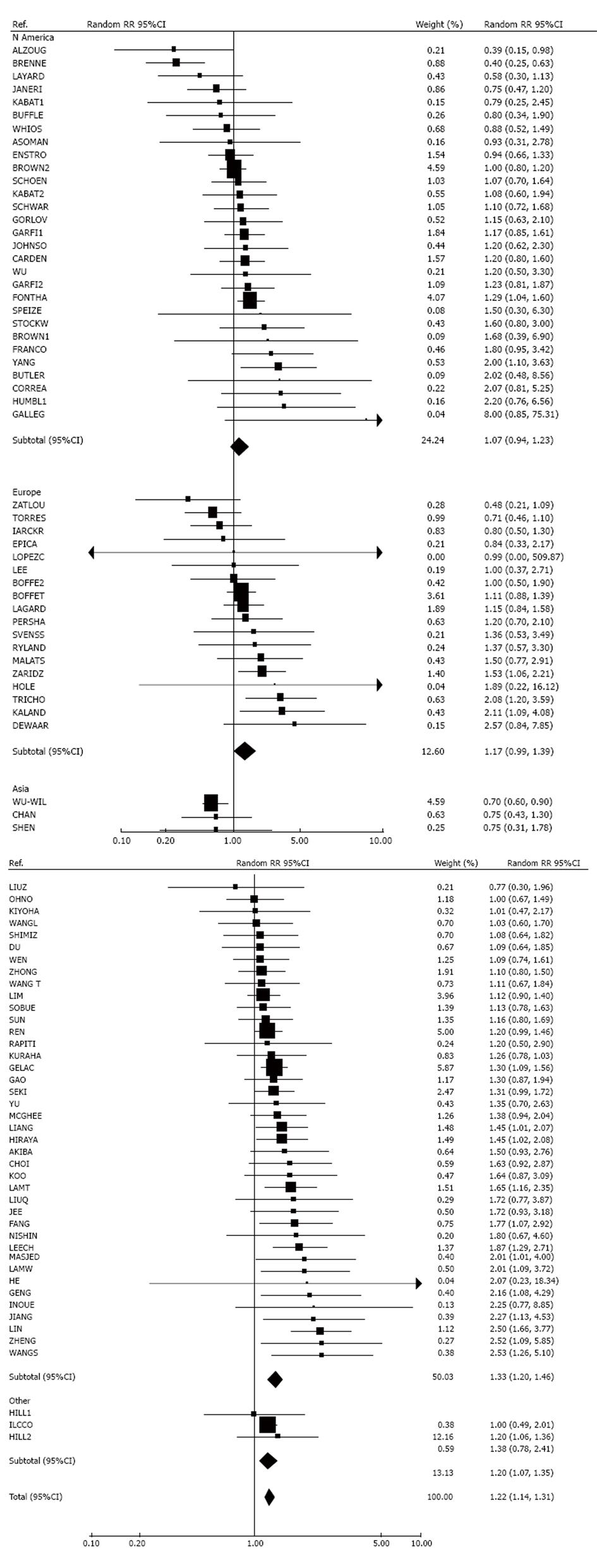
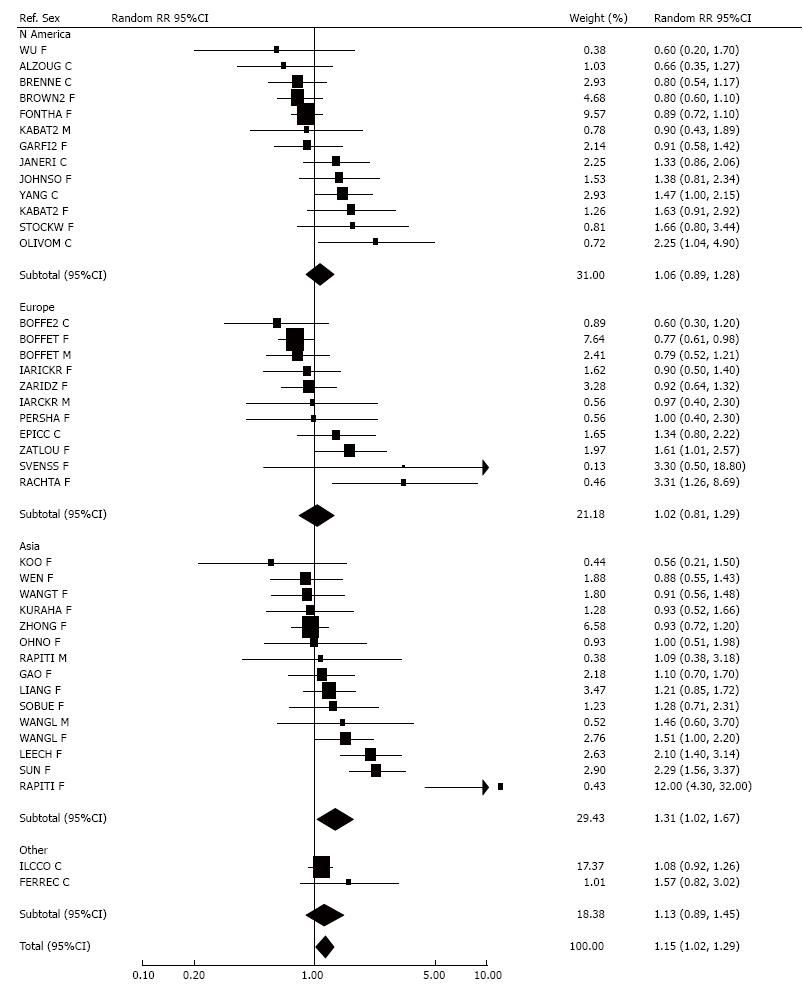
Ninety-three studies provided results relating lung cancer in women to husband’s smoking (or nearest equivalent), with 19[27-45] giving significantly raised RRs and 51 non-significantly raised RRs. In contrast 18 studies showed a negative relationship, significant in three studies[46-48]. Five studies gave a RR of 1.00. Two studies could not be included in the meta-analysis, one study[49] reporting no significant effect of passive smoking but giving no further details, with another[50] only giving an odds ratio of 2.2 (1.4-3.7) for greater than 40 smoker-years exposure to passive smoking. There was marked heterogeneity (P < 0.001) between the individual study estimates. However, fixed-effect (1.19, 95%CI: 1.14-1.24) and random-effects estimates (1.22, 1.14-1.31) were similar. Based on the Egger test there was no clear evidence of publication bias (0.05 < P < 0.1). Further analyses of these data are given in the section “smoking by the husband - detailed analyses” below.
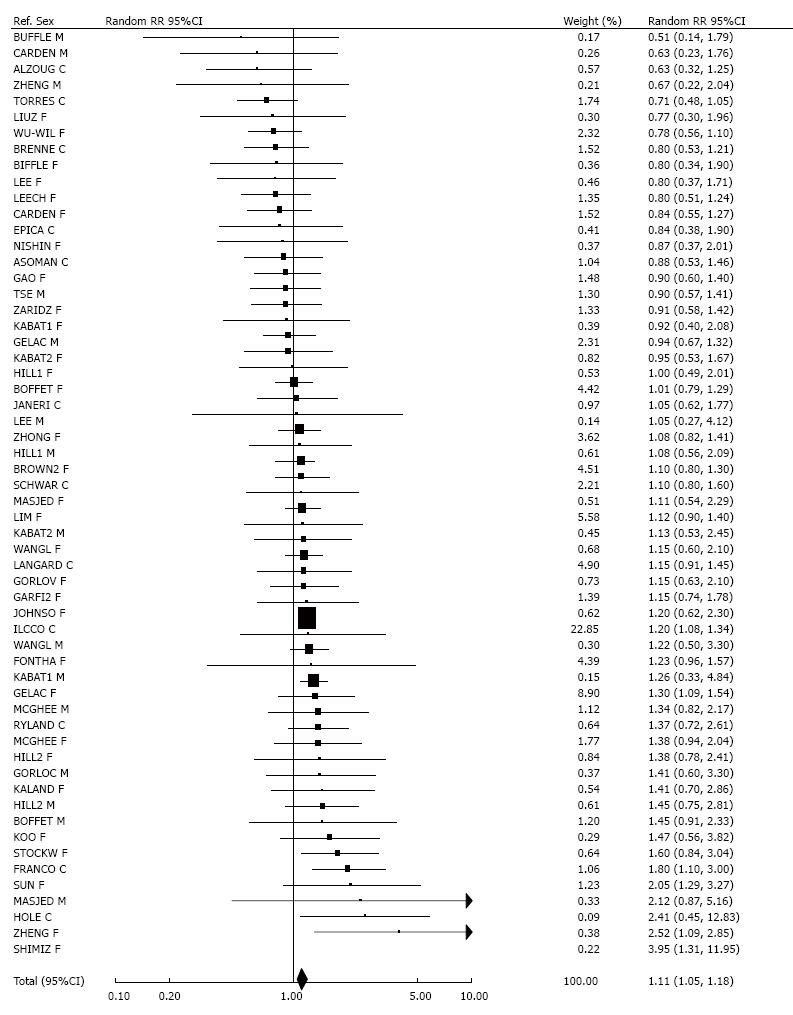
Forty-five studies considered smoking by the wife (or nearest equivalent). Twenty-eight RRs were raised, three[28,39,51] significantly. Fifteen studies reported a negative association, significant in one study[47]. One study found no association. One study[52] reported ETS was not statistically associated with lung cancer, but gave no further details and could not be included in the meta-analysis. Heterogeneity between studies was not significant, and fixed-effect (1.15, 1.03-1.28) and random-effects estimates (1.14, 1.01-1.29) were similar. There was no evidence of publication bias (P > 0.1).
Results by sex, separated by region, are given as forest plots in Figure 1 (husband smoking) and Figure 2 (wife smoking).
Further meta-analyses were carried out on results for smoking by either the husband or the wife (or nearest equivalent), based on 119 RR estimates. Details are given in Table 3, along with estimates split by various other factors. Overall, a fixed-effect RR (1.18, 1.14-1.23) and a random-effects RR (1.21, 1.14-1.29) were estimated, with marked heterogeneity between studies (P < 0.001). When the studies were examined according to various factors, there was evidence of heterogeneity between factor levels for publication date (P < 0.01), study size (P < 0.01) and age adjustment (P < 0.05), with the largest RRs seen for early (1981-1989) studies, small studies (1-49 cases) and estimates unadjusted for age. There was no significant heterogeneity by location, study type, reporting of dose-response results, or use of spouse as the index of exposure. There was no clear evidence of publication bias (0.05 < P < 0.1).
| Estimates considered | No. of estimates | Relative risk (95% confidence limits) | Heterogeneity1 | |
| Fixed-effects meta-analysis | Random-effects meta-analysis | |||
| All | 119 | 1.18 (1.14-1.23) | 1.21 (1.14-1.29) | +++ |
| N America | 38 | 1.08 (1.00-1.17) | 1.09 (0.95-1.26) | +++ |
| Europe | 22 | 1.15 (1.03-1.28) | 1.16 (1.00-1.35) | (+) |
| Asia | 54 | 1.24 (1.17-1.32) | 1.31 (1.20-1.44) | +++ |
| Asia - Japan | 13 | 1.26 (1.11-1.45) | 1.26 (1.11-1.45) | NS |
| Asia - Hong Kong | 8 | 1.32 (1.12-1.57) | 1.31 (1.06-1.63) | NS |
| Asia - China | 23 | 1.16 (1.06-1.27) | 1.29 (1.08-1.54) | +++ |
| Asia - Other | 10 | 1.34 (1.19-1.51) | 1.37 (1.19-1.57) | NS |
| Heterogeneity between Asian countries | NS | |||
| Other continents | 5 | 1.20 (1.09-1.33) | 1.20 (1.09-1.33) | NS |
| Heterogeneity between continents | (+) | |||
| Published in 1981-1989 | 34 | 1.38 (1.24-1.54) | 1.38 (1.24-1.54) | NS |
| Published in 1990-1999 | 33 | 1.09 (1.01-1.17) | 1.15 (1.02-1.28) | ++ |
| Published in 2000-2009 | 34 | 1.22 (1.12-1.33) | 1.21 (1.08-1.36) | + |
| Published in 2010 onwards | 18 | 1.17 (1.10-1.26) | 1.13 (0.94-1.36) | +++ |
| Heterogeneity by publication date | ++ | |||
| 1-49 cases | 23 | 1.44 (1.14-1.81) | 1.47 (1.15-1.88) | NS |
| 50-99 | 31 | 1.30 (1.14-1.47) | 1.27 (1.08-1.50) | + |
| 100-199 | 29 | 1.09 (1.00-1.19) | 1.10 (0.96-1.26) | +++ |
| 200-399 | 22 | 1.33 (1.21-1.46) | 1.32 (1.16-1.50) | + |
| 400+ | 14 | 1.14 (1.07-1.20) | 1.13 (1.02-1.25) | ++ |
| Heterogeneity by study size | ++ | |||
| Case-control | 97 | 1.18 (1.13-1.23) | 1.22 (1.13-1.31) | +++ |
| Prospective | 22 | 1.18 (1.05-1.33) | 1.18 (1.05-1.33) | NS |
| Heterogeneity by study type | NS | |||
| Not age adjusted | 21 | 1.34 (1.19-1.50) | 1.42 (1.18-1.71) | + |
| Age adjusted | 98 | 1.16 (1.11-1.21) | 1.18 (1.10-1.26) | +++ |
| Heterogeneity by age adjustment | NS | |||
| Dose-response results not reported | 46 | 1.13 (1.06-1.21) | 1.18 (1.06-1.31) | ++ |
| Only no dose-response stated | 2 | 0.95 (0.60-1.50) | 0.95 (0.60-1.50) | NS |
| Dose-response results reported | 71 | 1.21 (1.15-1.28) | 1.24 (1.14-1.35) | +++ |
| Heterogeneity by dose response reporting | NS | |||
| Spouse the index | 71 | 1.18 (1.11-1.24) | 1.21 (1.12-1.31) | ++ |
| Spouse not the index | 48 | 1.19 (1.12-1.27) | 1.20 (1.07-1.35) | +++ |
| Heterogeneity by index definition | NS | |||
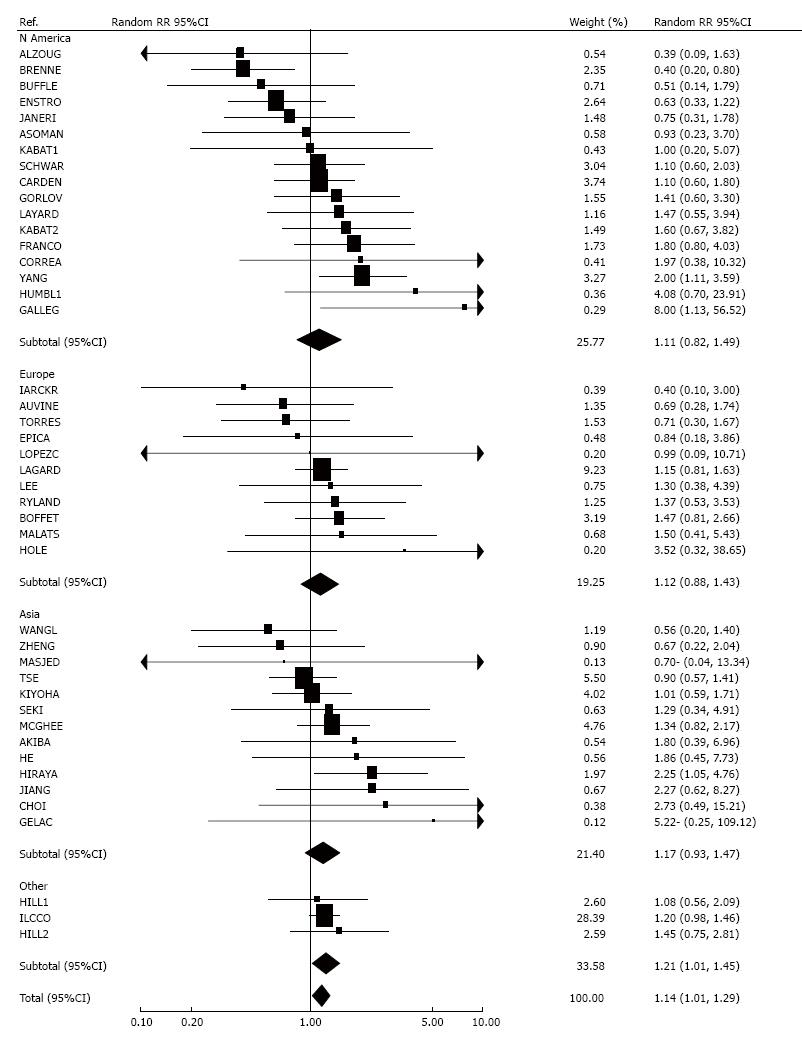
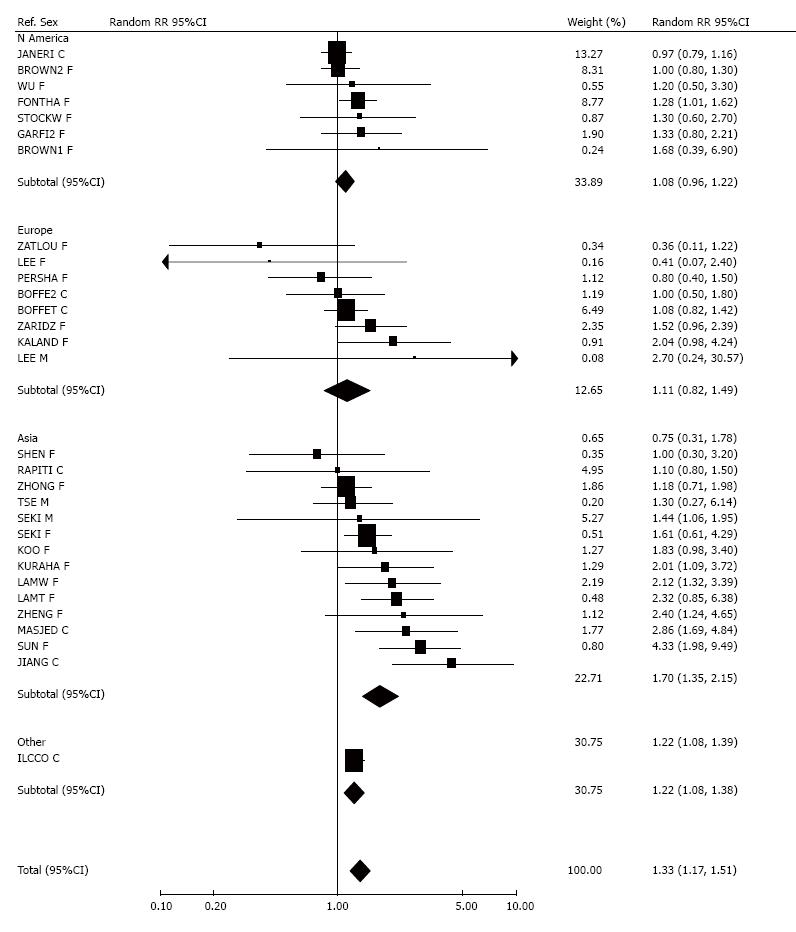
Results for smoking by the spouse (or nearest equivalent) were also examined by histological type of cancer, with Figure 3 (squamous cell carcinoma) and Figure 4 (adenocarcinoma) showing forest plots by region. The analysis of squamous cell carcinoma, based on 24 RR estimates, showed a significant (P < 0.001) positive association and heterogeneity (P < 0.001), overall estimates being 1.41 (1.24-1.59, fixed-effect) and 1.44 (1.15-1.80, random-effects). No significant variation by region was seen. For adenocarcinoma, the 30 RR estimates were again heterogeneous (P < 0.01), with the meta-analysis showing significantly raised RRs, of 1.23 (1.15-1.32, fixed-effect) and 1.33 (1.17-1.51, random-effects). The heterogeneity was partly due to differences (P < 0.001) by region, with little increase seen in North American and European studies (random-effects RRs 1.08, 0.96-1.22 for North America; 1.11, 0.82-1.49 for Europe), but a clear increase for Asia (random-effects RR 1.70, 1.35-2.15).
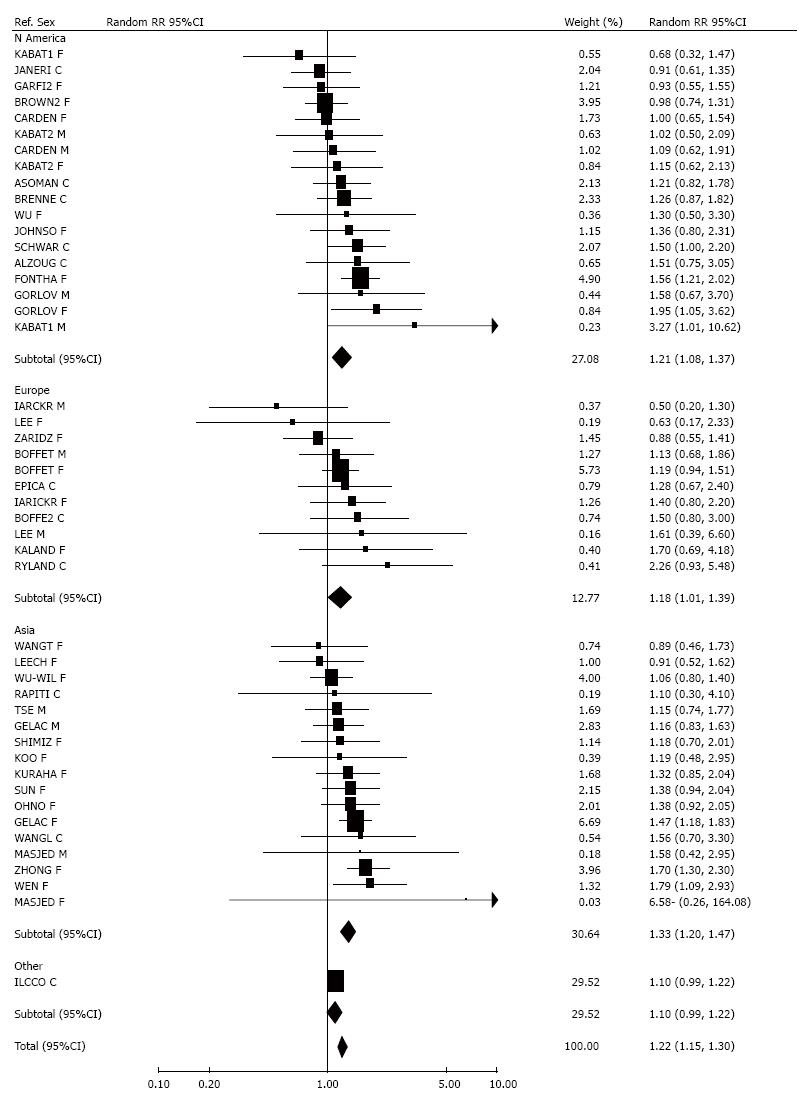
For lung cancer and workplace ETS exposure, 47 RR estimates were available (Figure 5). Of these, 37 were raised, with estimates from six studies[33,45,53-56] being significant, and another[57] of borderline significance. This contrasted with nine studies, where RRs were non-significantly below 1.00, and one showing no association. Two other studies[58,59], neither of which reported an association, could not be included in the meta-analysis, due to providing insufficient detail. Overall, there was a significant positive relationship, whether based on fixed-effect (1.21, 1.14-1.28) or random-effects RRs (1.22, 1.15-1.30). There was no evidence of heterogeneity or publication bias. Studies conducted in North America (1.21, 1.08-1.37), Europe (1.18, 1.01-1.39) and Asia (1.33, 1.20-1.47) all showed a significantly increased random-effects RR.
Results for childhood ETS exposure are given, by region, in Figure 6, with further meta-analyses given in Table 4. For childhood exposure from any cohabitant, 41 RR estimates were available. Of these, 21 were raised, eight significantly[37,39,60-65]. In contrast 18 RR estimates were below 1.00, one[66] significantly so, while two were equal to 1.00. In addition, three studies[67-69] found no relationship but provided insufficient detail for inclusion in the meta-analysis. Although meta-analysis suggested a positive relationship with the risk of lung cancer, this only just reached statistical significance (fixed-effect RR = 1.08, 1.01-1.15; random-effects RR = 1.15, 1.02-1.29). There was significant heterogeneity between the studies (P < 0.001), and heterogeneity between the continents (P < 0.05), with a significant increase seen in Asia (random-effects RR = 1.31, 1.02-1.67), but not in North America (RR = 1.06, 0.89-1.28) or Europe (RR = 1.02, 0.81-1.29).
| Estimates considered | No. of estimates | Relative risk (95% confidence limits) | Heterogeneity1 | |
| Fixed-effects meta-analysis | Random-effects meta-analysis | |||
| From any cohabitant | 41 | 1.08 (1.01-1.15) | 1.15 (1.02-1.29) | +++ |
| N America | 13 | 1.00 (0.89-1.13) | 1.06 (0.89-1.28) | + |
| Europe | 11 | 0.94 (0.81-1.08) | 1.02 (0.81-1.29) | + |
| Asia | 15 | 1.26 (1.11-1.42) | 1.31 (1.02-1.67) | +++ |
| Other | 2 | 1.10 (0.95-1.28) | 1.13 (0.89-1.45) | NS |
| Heterogeneity between continents | + | |||
| From mother specifically | 9 | 0.96 (0.77-1.20) | 0.98 (0.77-1.25) | NS |
| From father specifically | 11 | 0.90 (0.80-1.00) | 1.00 (0.78-1.29) | +++ |
| From parents specifically | 8 | 0.78 (0.64-0.94) | 0.78 (0.64-0.94) | NS |
Based on nine RR estimates, meta-analysis showed no evidence of any relationship specifically with maternal smoking in childhood, with the fixed-effect estimate 0.96 (0.77-1.20) and the random-effects estimate 0.98 (0.77-1.25). There was also no association specifically with paternal smoking in childhood (fixed-effect model 0.90, 0.80-1.00; random-effects model 1.00, 0.78-1.29), based on 11 estimates. However, meta-analysis of the eight estimates specifically considering parental smoking during childhood gave a reduced RR (0.78, 0.64-0.94 for both models). There was no significant heterogeneity for maternal smoking or parental smoking, but there was for paternal smoking (P < 0.001) due to an atypically high estimate of 12.64 (4.89-32.68) for females in one study[61].
None of the analyses of childhood exposure showed any significant evidence of publication bias.
A total of 58 RR estimates were available for household ETS exposure from any source, as shown in Figure 7. Thirty-six RRs were above 1.00, statistically significant in six studies[35,43,45,60,70,71]. Twenty-one non-significantly negative RRs were also reported, while one study found no association. Overall RRs were 1.13 (1.07-1.19, fixed-effect) and 1.11 (1.05-1.18, random-effects). There was marked heterogeneity (P < 0.001) between the estimates, but no significant variation by study location, or evidence of publication bias.
Restricting attention to sources of ETS other than the spouse, only 13 RRs were available, and the overall RR, although raised, was not significant (1.04, 0.89-1.21, fixed-effect) or (1.12, 0.87-1.44, random-effects).
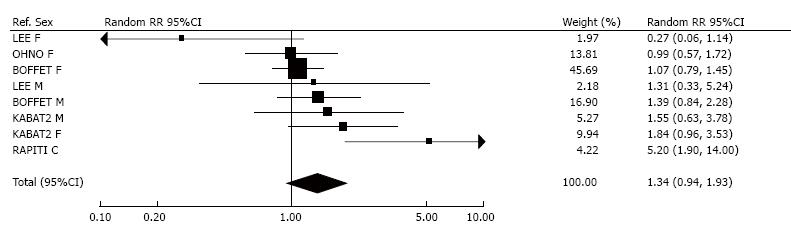
Figure 8 shows the eight RRs for ETS exposure during travel. Six were above 1.00, and two were below 1.00. Only one estimate[61] was significant, and its high RR of 5.20 was the major contributor to the significant (P < 0.05) heterogeneity. Fixed effect meta-analysis gave a RR which was just significant (1.24, 1.01-1.53), but the random-effects RR was not (1.34, 0.94-1.93).
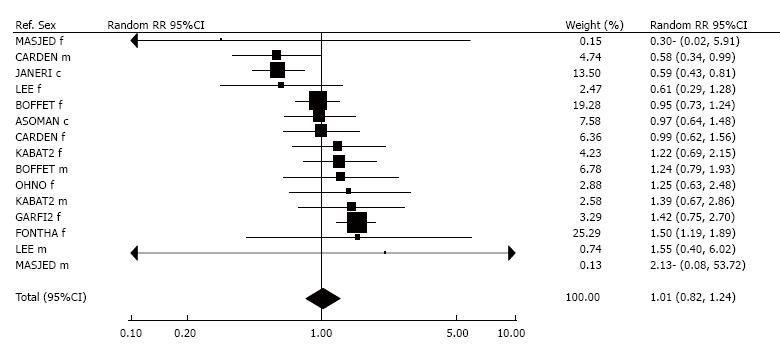
Analysis of the relationship of lung cancer to ETS exposure in social situations was based on 15 RR estimates, shown in Figure 9. One[33] study provided a significantly increased RR, with seven studies giving non-significantly raised estimates. Seven RRs were below 1.00, significantly so in two[72,73] studies. Overall, there was no evidence of an increased risk, for either fixed-effect (1.03, 0.92-1.16) or random-effects RRs (1.01, 0.82-1.24).
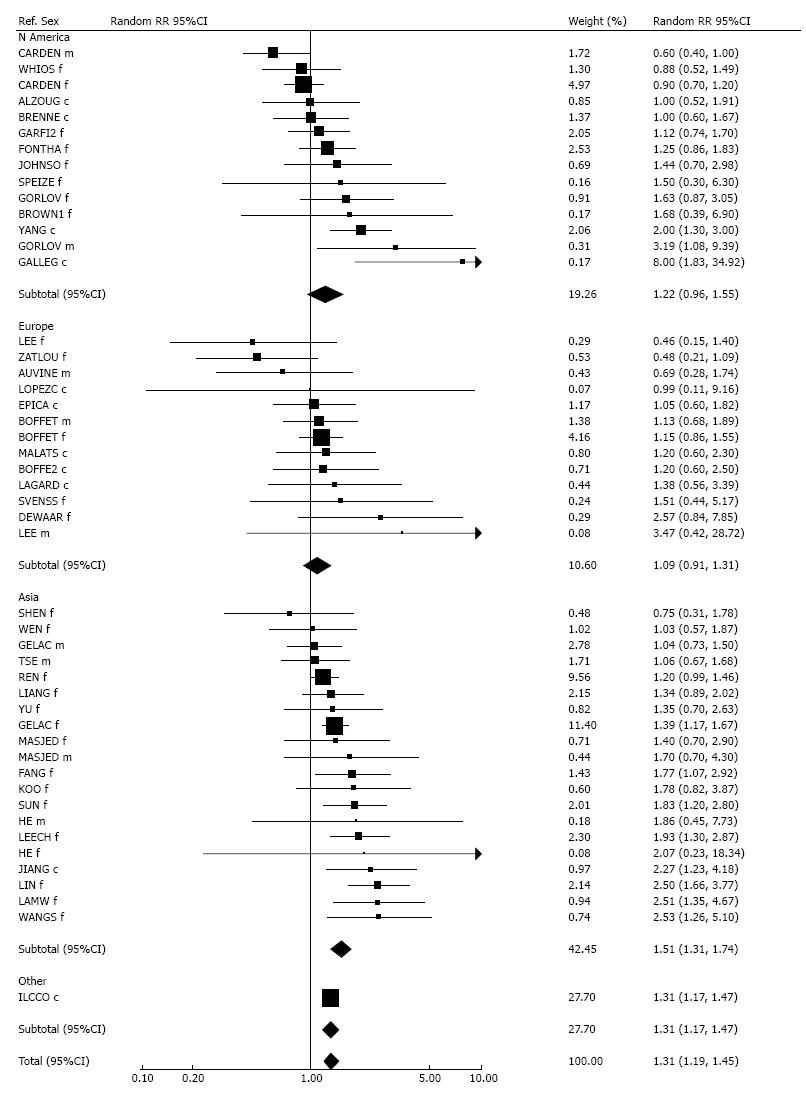
The 48 RRs for total ETS exposure are shown, by region, in Figure 10. Thirty-eight were above 1.00, significantly so for 12 studies[29,34,37-39,41-43,45,51,55,60]. Eight non-significantly reduced RRs were also reported, while two studies reported RRs of 1.00. Although there was marked heterogeneity (P < 0.001), fixed-effect RRs (1.30, 1.22-1.38), and random-effects RRs were quite similar (1.31, 1.19-1.45). Heterogeneity between the continents was statistically significant (P < 0.01), with random-effects RRs higher for Asia (1.51, 1.31-1.74), than for North America (1.22, 0.96-1.55) or Europe (1.09, 0.91-1.31). There was no evidence (P > 0.1) of publication bias.
Smoking by the husband (or nearest equivalent) is now considered in more detail, with results presented both for overall exposure and per 10 cigarettes per day smoked by the husband. A fuller report which includes adjustment for confounding and for misclassification of exposure, is available in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%202.pdf, with only the main findings presented here.
For overall exposure, the RRs considered are those shown in Figure 1 and briefly referred to in the section “smoking by the spouse”. As noted there, combining estimates from 93 studies gave (RR = 1.19, 95%CI: 1.14-1.24, fixed-effects) and (RR =1.22, 95%CI: 1.14-1.31, random-effects).
Of the 93 studies, 29 were in North America, 18 in Europe, 26 in China or Hong Kong, 18 in the rest of Asia, and two in New Zealand. One Asian study[74] was of Chinese women in Singapore, and has been included in the subset of China studies. As the studies in New Zealand were principally of people of European descent, they have been included in the European subset of studies. One of the studies[45] was international, but due to a high proportion of Asian subjects has been included in the Rest of Asia subset.
The first study appeared in 1981[75], a further 25, 27, 26 and 14 being published in, respectively, 1982-89, 1990-99, 2000-09 and 2010-2014. Sixteen were prospective (cohort) studies and 77 case-control. Twenty-two studies involved less than 50 cases in lifelong non-smokers, and nine over 400 cases.
Nine studies adjusted for fruit consumption, 11 for vegetables, and 4 for dietary fat. Less than half (32 studies) adjusted for an index of education.
Twenty-four of the studies provided data on lung cancer risk by amount smoked by the husband specifically, with the remainder only providing results for overall exposure. Table 1 of http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%202.pdf gives the data used for each study and the fitted estimates of β and SEβ. Based on these data, it was estimated that each 10 cigarettes per day smoked by the husband multiplied risk by an estimated 1.09 (95%CI: 1.07-1.11) based on a fixed-effects analysis and 1.10 (1.07-1.14) using a random-effects analysis.
In order to adjust for the uncontrolled effects of confounding by diet (by fruit, vegetables and dietary fat) and education, summary estimates were required of the relationships of the four potential factors to both risk of lung cancer and ETS exposure, and of the correlations between the four factors. The estimates used are presented in Table 5, and show that, in non-smoking females, both risk of lung cancer and ETS exposure at home are associated with reduced fruit and vegetable consumption and education, and increased dietary fat consumption. All these associations are significant at least at P < 0.05, and for education at P < 0.001, with the data based on analysis of results from at least 10 studies (with one exception - dietary fat and lung cancer, based on 6 studies). Table 5 also shows the inter-correlations between the four confounding variables, based on combined estimates from seven studies. These show that fruit and vegetable consumption are strongly correlated with each other (P < 0.01). Other correlations are weaker and not significant at P < 0.05.
| Statistic | Fruit consumption | Vegetable consumption | Dietary fat consumption | Education | |
| Lung cancer risk | N studies | 14 | 16 | 6 | 12 |
| RR2 | 0.86 | 0.88 | 1.22 | 0.91 | |
| (95%CI) | (0.78-0.96)8 | (0.80-0.97)8 | (1.09-1.36)8 | (0.88-0.95)6 | |
| per | SD | SD | SD | Year3 | |
| ETS exposure | N studies | 11 | 16 | 12 | 13 |
| at home | Difference24 | -0.0737 | -0.0568 | 0.1317 | -0.5346 |
| (SE) | -0.02 | -0.021 | -0.032 | -0.063 | |
| unit | SD | SD | SD | Year3 | |
| Correlations5 | Fruit consumption | 1 | +0.3147 | -0.104NS | +0.143NS |
| Vegetable consumption | 1 | -0.054NS | -0.1309 | ||
| Dietary fat consumption | 1 | -0.039NS | |||
| Education | 1 |
As described in the methods, we used misclassification rates of 10% for Asian studies and 2.5% elsewhere, these rates accounting for the lower rates of lung cancer seen among misclassified smokers than among non-misclassified smokers.
Table 6 presents results of analyses adjusting for confounding and misclassification based on RRs for smoking by the husband, while Table 7 similarly presents results based on RRs per 10 cigarettes smoked by the husband. Each table presents three sets of results: (1) unadjusted; (2) adjusted for confounders; and (3) adjusted for confounders and corrected for smoking misclassification. They each give overall estimates and results subdivided by various aspects of the studies considered.
| Studies | n | Unadjusted for confoundingUncorrected for misclassificationRR (95%CI) | Adjusted for confounding2Uncorrected for misclassificationRR (95%CI) | Adjusted for confounding2Corrected for misclassification3RR (95%CI) |
| All | 93 | 1.219 (1.138-1.305) | 1.139 (1.062-1.221) | 1.077 (0.999-1.162) |
| North America | 29 | 1.074 (0.937-1.232) | 1.004 (0.873-1.154) | 0.898 (0.775-1.039) |
| Europe and New Zealand | 20 | 1.174 (1.007-1.369) | 1.092 (0.934-1.277) | 1.062 (0.899-1.254) |
| China (including Hong Kong and study LIM) | 27 | 1.321 (1.144-1.524) | 1.239 (1.071-1.433) | 1.175 (1.005-1.374) |
| Rest of Asia (including study ILCCO) | 17 | 1.284 (1.187-1.389) | 1.194 (1.103-1.291) | 1.164 (1.072-1.262) |
| North America, Europe and New Zealand | 49 | 1.112 (1.004-1.231) | 1.037 (0.935-1.150) | 0.959 (0.858-1.072) |
| Asia | 44 | 1.314 (1.199-1.439) | 1.229 (1.121-1.348) | 1.181 (1.070-1.304) |
| Published in 1980s | 26 | 1.361 (1.216-1.522) | 1.267 (1.132-1.417) | 1.194 (1.059-1.347) |
| Published in 1990s | 27 | 1.152 (1.016-1.305) | 1.077 (0.948-1.225) | 1.005 (0.871-1.160) |
| Published in 2000s | 26 | 1.240 (1.105-1.392) | 1.163 (1.034-1.308) | 1.115 (0.987-1.260) |
| Published in 2010s | 14 | 1.139 (0.945-1.372) | 1.059 (0.877-1.277) | 1.026 (0.844-1.247) |
| < 100 cases | 49 | 1.339 (1.178-1.521) | 1.249 (1.098-1.422) | 1.192 (1.038-1.370) |
| 100-199 cases | 22 | 1.117 (0.973-1.284) | 1.042 (0.904-1.200) | 0.978 (0.846-1.131) |
| 200-399 cases | 13 | 1.363 (1.190-1.561) | 1.275 (1.114-1.460) | 1.226 (1.051-1.429) |
| 400+ cases | 9 | 1.101 (0.973-1.247) | 1.027 (0.905-1.166) | 0.957 (0.826-1.108) |
| With dose-response data4 | 24 | 1.308 (1.181-1.449) | 1.226 (1.105-1.359) | 1.170 (1.052-1.302) |
| Without dose-response data | 69 | 1.182 (1.088-1.286) | 1.104 (1.014-1.201) | 1.040 (0.948-1.141) |
| With age adjustment5 | 75 | 1.184 (1.100-1.274) | 1.106 (1.027-1.191) | 1.048 (0.966-1.136) |
| Without age adjustment | 18 | 1.437 (1.194-1.728) | 1.340 (1.110-1.618) | 1.264 (1.026-1.556) |
| Case-control studies | 77 | 1.226 (1.133-1.326) | 1.144 (1.057-1.239) | 1.080 (0.990-1.177) |
| Prospective studies | 16 | 1.187 (1.043-1.350) | 1.111 (0.977-1.264) | 1.064 (0.928-1.220) |
| Studies | n | Unadjusted for confoundingUncorrected for misclassificationRR (95%CI) | Adjusted for confounding2Uncorrected for misclassificationRR (95%CI) | Adjusted for confounding2Corrected for misclassification3RR (95%CI) |
| All | 93 | 1.102 (1.065-1.140) | 1.062 (1.027-1.099) | 1.032 (0.994-1.071) |
| North America | 29 | 1.037 (0.977-1.101) | 1.006 (0.946-1.070) | 0.957 (0.896-1.022) |
| Europe and New Zealand | 20 | 1.060 (0.995-1.128) | 1.020 (0.956-1.088) | 1.003 (0.938-1.073) |
| China (including Hong Kong and study LIM) | 27 | 1.169 (1.082-1.263) | 1.127 (1.041-1.219) | 1.094 (1.006-1.191) |
| Rest of Asia (including study ILCCO) | 17 | 1.142 (1.095-1.191) | 1.094 (1.050-1.141) | 1.079 (1.033-1.127) |
| North America, Europe and New Zealand | 49 | 1.046 (1.001-1.094) | 1.012 (0.967-1.059) | 0.974 (0.928-1.023) |
| Asia | 44 | 1.158 (1.104-1.216) | 1.113 (1.060-1.170) | 1.089 (1.033-1.147) |
| Published in 1980s | 26 | 1.148 (1.092-1.207) | 1.105 (1.052-1.162) | 1.075 (1.019-1.134) |
| Published in 1990s | 27 | 1.063 (1.004-1.125) | 1.025 (0.967-1.087) | 0.988 (0.926-1.053) |
| Published in 2000s | 26 | 1.123 (1.056-1.194) | 1.085 (1.020-1.155) | 1.061 (0.995-1.132) |
| Published in 2010s | 14 | 1.073 (0.970-1.188) | 1.032 (0.932-1.143) | 1.014 (0.912-1.128) |
| < 100 cases | 49 | 1.143 (1.077-1.213) | 1.101 (1.036-1.169) | 1.072 (1.005-1.144) |
| 100-199 cases | 22 | 1.062 (0.993-1.137) | 1.025 (0.957-1.098) | 0.994 (0.926-1.066) |
| 200-399 cases | 13 | 1.176 (1.097-1.261) | 1.134 (1.058-1.216) | 1.111 (1.027-1.202) |
| 400+ cases | 9 | 1.041 (0.976-1.111) | 1.002 (0.938-1.070) | 0.966 (0.895-1.042) |
| With dose-response data4 | 24 | 1.123 (1.072-1.176) | 1.082 (1.032-1.134) | 1.053 (1.005-1.103) |
| Without dose-response data | 69 | 1.091 (1.044-1.139) | 1.053 (1.007-1.100) | 1.021 (0.973-1.071) |
| With age adjustment5 | 75 | 1.084 (1.046-1.123) | 1.044 (1.008-1.082) | 1.015 (0.976-1.056) |
| Without age adjustment | 18 | 1.211 (1.101-1.331) | 1.168 (1.061-1.285) | 1.131 (1.018-1.256) |
| Case-control studies | 77 | 1.106 (1.064-1.150) | 1.066 (1.025-1.109) | 1.034 (0.991-1.080) |
| Prospective studies | 16 | 1.081 (1.021-1.145) | 1.043 (0.985-1.105) | 1.018 (0.957-1.083) |
As shown in Table 6, adjustment for confounding variables reduces the overall RR for smoking by the husband from 1.219 (1.138 to 1.305) to 1.139 (1.062-1.221), implying bias due to failure to control for the four variables is 1.219/1.139 = 1.070. Further correction for misclassification reduced the estimate to a marginally nonsignificant 1.077 (0.999-1.162), implying a further bias of 1.139/1.077 = 1.058. In the fully adjusted and corrected analyses, there is no evidence of an association in North America, Europe and New Zealand (RR 0.959, 0.858-1.072) but there is an association in Asia (1.181, 1.070-1.304).
RRs are higher for studies providing dose-response data (1.170, 1.052-1.302) than for other studies (1.040, 0.948-1.141), and higher for studies which did not adjust for age (1.264, 1.026-1.556) than for those which did (1.048, 0.966-1.136). However, neither difference is statistically significant (P = 0.10 and P = 0.08 respectively).
The pattern of results shown in Table 7, where RRs are per amount smoked by the husband, is similar, though the RRs themselves are lower. Thus, the unadjusted/uncorrected overall RR of 1.102 (1.065-1.140) reduces to 1.062 (1.027-1.099) after adjustment for confounding (bias = 1.038), and to a nonsignificant 1.032 (0.994-1.071) after further correction for misclassification (additional bias = 1.030). Patterns of variation by study factors are very similar to those for overall smoking by the husband in Table 6.
Additional material presented in http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%202.pdf shows that the effect of confounder adjustment was greatest for education, and least for fruit and vegetables. Thus, in the analysis of RRs per amount smoked by the husband, the biases due to uncontrolled confounding were estimated as 1.024 for education, 1.013 for dietary fat, 1.005 for fruit, and 1.004 for vegetables.
We have demonstrated, as other reviews before us[6,76], a weak but significant (P < 0.05) association of ETS exposure with never smoker lung cancer risk. This can be seen for various indices of exposure, including spousal, household, workplace and total exposure. It is less clearly evident for exposure in travel and in social situations, where data are quite limited, and for childhood exposure where the results shown in Table 4 are rather conflicting. There is also clear heterogeneity between study-specific estimates for many of the indices of exposure. Meta-analyses for smoking by the spouse (or nearest equivalent) shown in Table 3 indicate that estimates are higher in early studies (published in 1981-89), in small studies (1-49 cases), and where estimates are not age-adjusted.
Do these quite weak associations provide good evidence of a causal relationship? To gain insight into this we carried out additional analyses for smoking by the husband investigating biases due to uncontrolled confounding by education and three aspects of diet (fruit, vegetables and dietary fat) and due to failure to adjust for misclassification of smoking by the subject. Based on 93 studies, confounder adjustment and misclassification correction substantially reduced the magnitude of the association with smoking by the husband, the RR (95%CI) estimate of 1.22 (1.14-1.31) reducing to 1.14 (1.06-1.22) after confounder adjustment, and further reducing to 1.08 (0.999-1.16) after additional correction for misclassification. The adjusted and corrected estimate is not quite significant, the same being true for analyses based on the RR per 10 cigs/day smoked by the spouse, where the overall RR reduced from 1.10 (1.07-1.14) to 1.06 (1.03-1.10) after adjustment for confounding and to 1.03 (0.994-1.07) after the further correction for misclassification.
Below we discuss some aspects of the evidence relevant to consideration of causality. Parts of the discussion are quite brief, the interested reader being referred to our publication[2] describing our earlier analyses.
Since active smoking causes lung cancer, and since ETS contains many of the carcinogens in tobacco smoke, it can be argued that some causal effect of ETS exposure is to be expected, though this argument depends on there being no threshold dose of exposure. If there is no threshold, what effect might one expect? Certainly, exposure from ETS is much less than that from active smoking, with studies based on cotinine indicating relative exposure factors of 0.4%[77], 0.2%[78] or 0.06%[79] and studies based on particulate matter[80-88] suggesting a lower factor, of order 0.005%-0.02%. Given an RR for current vs never smoking of 8.43, as reported in a recent meta-analysis[89] and assuming a linear dose-response relationship, even a relative exposure factor as high as 0.5% would only suggest that the RR for ETS exposure would be about 1.04, while a relative exposure factor of 0.1% would suggest a RR of about 1.008. These RRs are much less than the unadjusted/uncorrected RR of 1.22 for smoking by the husband (or nearest equivalent) shown in Table 6. Either the relationship between dose and risk is distinctly non-linear (and the evidence does not suggest this for active smoking[89]) or a substantial part, if not all, of the observed association is due to bias.
Based on the evidence we collected, we have demonstrated a clear tendency for increased dietary fat consumption, reduced fruit and vegetable consumption and fewer years of education to be associated both with increased lung cancer risk and with increased at home ETS exposure. Given that relatively few of the studies adjusted for the dietary variables or education, it was to be expected that adjustment for these four factors would reduce the RR for smoking by the husband, and so it proved. While there is uncertainty in this adjustment, as discussed elsewhere[19], it is clear that there is a considerable potential for bias. Among other things it should be noted that these are not the only potential sources of bias. We considered various other candidate confounding factors, including income, occupation, and socioeconomic factors, obesity, physical activity, air pollution, alcohol and tea drinking, but concluded that for none of these were there data adequate to provide any sort of reliable qualitative estimate of their relationship to lung cancer risk in non-smokers. That said, the general tendency for smoking and marriage to a smoker to be associated with lifestyle factors generally considered associated with adverse health[90-93], suggests that our adjustments may well have been conservative.
Some current or former smokers are known to deny having smoked, so being wrongly described as never smokers[26,94]. Also, marital partners’ smoking habits are correlated, with smokers tending to marry smokers[3,23]. Taken together, these two tendencies, if ignored, will bias the observed association of smoking by the husband to never smoker lung cancer risk[3,21,95]. There are many difficulties in accurately estimating the extent of bias due to misclassification. These include the misclassification rates being dependent on the circumstances in which the questions were asked, as well as the fact that smokers who deny smoking are unrepresentative of all smokers, tending to be more often occasional smokers and long-term ex-smokers and so have lower lung cancer risks than non-misclassified smokers[23]. Here we have assumed, as earlier[22], that misclassification correction can be carried out assuming that, among women, the percentage of average-risk ever-smokers who deny smoking is 10.0% in Asia and 2.5% elsewhere, these misclassification rates taking account of the lower lung cancer rates in misclassified compared to non-misclassified smokers.
While the misclassification correction is clearly open to question, and we have not formally updated the extensive work we did some years ago on estimating rates[23,26], we still believe that the rates we have used are not unreasonable. Indeed given recent estimates of substantial denial of smoking in recent studies[94,96,97], our correction may be somewhat conservative.
We now briefly comment on other sources of bias.
Publication bias occurs if the data that are published are not representative of all the data that exist on a topic. For many exposures, positive findings are published more often than negative findings[98-100], so meta-analyses of data drawn from the literature overestimate true relationships. We have not attempted to quantify the extent of publication bias, though our detailed tables (http://www.pnlee.co.uk/downloads/etslc/23482-supplementary%20file%204.pdf) do include results of Egger tests[16], a number showing some evidence that smaller studies are more likely to produce above average than below average RRs. This is consistent with the higher RRs reported in small studies seen in Table 3 for spousal smoking. We believe that some publication bias exists but given that the larger studies seem likely to publish regardless of the findings, and that these contribute most to the overall estimates, such bias is probably unimportant.
There is some evidence (P = 0.10) that RRs are higher for those studies which provide dose-response results than for those which do not so. If this represents a true effect, it is suggestive of a different form of publication bias, with authors tending to be more likely to report dose-response results where there is a strong association in the first place.
Misdiagnosis of lung cancer certainly exists, especially when based on X-rays or sputum cytology[101-103]. The extent, and direction, to which it might have biased the RR estimate for ETS and lung cancer is difficult to determine. While randomly misdiagnosing as lung cancer diseases which are unassociated with ETS would tend to dilute any true RR, misdiagnosis might not be random and may be correlated with ETS exposure or factors associated with it. Since random-effects estimates for spousal smoking proved to be quite similar for studies that did or did not require full histological confirmation, this seems unlikely to be an important source of bias.
Case-control studies collect exposure data after the disease has occurred, and the presence of the disease itself, or knowledge of it, may distort responses about past exposure. Such recall bias is not an issue for prospective studies. Given that our analyses for spousal smoking found little difference in RRs by study type, we feel that recall bias is unlikely to be a major problem.
Random misclassification of smoking spouses as non-smokers will not create a false effect if no true risk exists, but will underestimate a true relationship. It has been clearly shown[21] that such misclassification causes much less bias effect than does misclassification of the subject’s smoking, so for practical purposes it can be ignored.
When considering the relationship of lung cancer risk to smoking by the husband, three categories of women are relevant: A - never smokers married to ever smokers; B - never smokers married to never smokers; and C - never smokers without any ETS exposure. Group C is a subset of group B. In the analysis of the effect of husband’s smoking, the RR estimate is based on comparison of groups A and B, but it has been argued[3] that a better estimate RR* is based on comparison of groups A and C. If a marker of ETS exposure, such as cotinine, is Z times higher in group A than group B, RR* can be estimated by RR* = RR(Z-1) / (Z-RR)[2,3].
Some comments can be made on this revised estimate. First, and most noteworthy, to conduct background correction only makes sense when the original association, with marriage to a smoker, derives from a causal effect of ETS. Where adjustment for confounding and correction for smoker misclassification bias explains the whole of the observed association, background correction will have no effect. If such adjustment and correction explains most of the association, the correction will have a small effect. Thus, assuming Z = 3, as estimated by Hackshaw et al[3], this correction has quite a substantial effect on the unadjusted association for husband’s smoking, increasing it from 1.22 to 1.37. However, applying it to the confounder adjusted and misclassification corrected estimate of 1.08 only increases it to 1.12. In any case, the validity of the background-corrected estimate of 1.12 is dubious, given that the 1.08 was not statistically significant in the first place, and could itself be an overestimate due to the limitations in confounder adjustment and misclassification correction discussed above.
Second, background correction only applies to the simple comparison of risk in the exposed and comparison groups, and does not apply to estimates of the increase in risk for amount smoked by the husband. Also, background correction is only an indirect method for estimating lung cancer risk from sources of ETS exposure other than the spouse, using data only relating to spousal exposure. This method ignores existing data on risk from these other sources.
Coming to reliable conclusions regarding a weak association based on non-randomized epidemiological studies is difficult at the best of times. When, as here, there is evidence that adjustment for confounding and correction for misclassification substantially weakens the association most usually considered (smoking by the husband) and renders it nonsignificant, and when these adjustments and corrections may themselves be somewhat limited, one cannot reliably conclude that a true effect of ETS exposure on lung cancer risk has been demonstrated. While one cannot prove a negative, and while the clear relationship of smoking to lung cancer suggests that some association might exist, the only conclusion that seems valid is that there may be a relationship of ETS to lung cancer risk (with the evidence stronger for Asian studies), but if it exists, it is certainly much weaker than suggested by meta-analyses that do not adjust for confounding and misclassification.
Most, if not all, of the weak association of ETS with risk of lung cancer is explicable by confounding and smoking misclassification. A causal relationship is not demonstrated.
We thank Japan Tobacco International S.A. for supporting publication of this paper. The opinions and conclusions of the authors are their own, and do not necessarily reflect the position of Japan Tobacco International S.A. We also thank the United Kingdom Tobacco Manufacturers Association, Imperial Tobacco Ltd, British-American Tobacco Limited, and Philip Morris Products S.A. for earlier support in developing the databases used. Finally we also thank Pauline Wassell, Diana Morris and Yvonne Cooper for assistance in typing various drafts of the paper and obtaining relevant literature, and all the researchers who published the reports which formed the basis of our work.
The authors address the widely held claim that environmental tobacco smoke (ETS) exposure causes lung cancer by presenting an up-to-date comprehensive meta-analysis of the available evidence, considering exposure from various sources, and illustrating the potential magnitude of bias from confounding and misclassification of smoking.
Based on all 102 studies providing relevant data, the authors demonstrate a significant (P < 0.05) increase in never smoker lung cancer risk for various exposure indices - from the spouse, at home, at work and overall, though the evidence for childhood exposure is less clear. Based on smoking by the husband, the most studied ETS exposure index, the RR is estimated as 1.22 (95%CI: 1.14-1.31). However, adjustment for confounding by education and by consumption of fruit, vegetables and dietary fat, and correction for misclassification of active smoking by the wife, markedly reduces this association, which becomes a nonsignificant 1.08 (95%CI: 0.999-1.16). Since these adjustments and corrections may not fully correct for the bias from these sources, and given the existence of other biases, one cannot conclude with any certainty that a true effect of ETS exposure on lung cancer risk exists.
The new feature of the paper is the extent of the evidence considered, and the adjustments and corrections made.
The authors analysis should engender caution in drawing inferences from weak associations seen in non-randomized epidemiological studies, particularly where biases are known to exist.
It is an interesting paper.
P- Reviewer: Kawai H, Pereira-Vega A S- Editor: Qiu S L- Editor: A E- Editor: Lu YJ
| 1. | International Agency for Research on Cancer. Tobacco smoke and involuntary smoking. IARC Monographs on the evaluation of carcinogenic risks to humans Lyon, France: IARC 2004; Available from: http//monographs.iarc.fr/ENG/Monographs/vol83/mono83.pdf. |
| 2. | Lee PN, Fry JS, Forey BA. Revisiting the association between environmental tobacco smoke exposure and lung cancer risk. Indoor Built Environ. 2002;11:59-82. [DOI] [Full Text] |
| 3. | Hackshaw AK, Law MR, Wald NJ. The accumulated evidence on lung cancer and environmental tobacco smoke. BMJ. 1997;315:980-988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 379] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 4. | Zhong L, Goldberg MS, Parent ME, Hanley JA. Exposure to environmental tobacco smoke and the risk of lung cancer: a meta-analysis. Lung Cancer. 2000;27:3-18. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 104] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 5. | Taylor R, Cumming R, Woodward A, Black M. Passive smoking and lung cancer: a cumulative meta-analysis. Aust N Z J Public Health. 2001;25:203-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 45] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 6. | Boffetta P. Involuntary smoking and lung cancer. Scand J Work Environ Health. 2002;28 Suppl 2:30-40. [PubMed] |
| 7. | Stayner L, Bena J, Sasco AJ, Smith R, Steenland K, Kreuzer M, Straif K. Lung cancer risk and workplace exposure to environmental tobacco smoke. Am J Public Health. 2007;97:545-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 71] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 8. | Taylor R, Najafi F, Dobson A. Meta-analysis of studies of passive smoking and lung cancer: effects of study type and continent. Int J Epidemiol. 2007;36:1048-1059. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 9. | Zhao H, Gu J, Xu H, Yang B, Han Y, Li L, Liu S, Yao H. [Meta-analysis of the relationship between passive smoking population in China and lung cancer]. Zhongguo Fei Ai Za Zhi. 2010;13:617-623. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 10. | Wang X, Qin Y, Gu J, Wang F, Jia P, Wang H, Yao Q, Zhu S. [Systematic review of studies of workplace exposure to environmental tobacco smoke and lung cancer risk]. Zhongguo Fei Ai Za Zhi. 2011;14:345-350. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 11. | Lee PN. An assessment of the epidemiological evidence relating lung cancer risk in never smokers to environmental tobacco smoke exposure. Environmental tobacco smoke, Discussion on ETS, Tokyo. New York: Springer-Verlag 1993; 28-70. |
| 12. | Gardner MJ, Altman DG, editors . Statistics with confidence. Confidence intervals and statistical guidelines. London: British Medical Journal 1989; . |
| 13. | Fleiss JL, Gross AJ. Meta-analysis in epidemiology, with special reference to studies of the association between exposure to environmental tobacco smoke and lung cancer: a critique. J Clin Epidemiol. 1991;44:127-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 261] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 14. | Hamling J, Lee P, Weitkunat R, Ambühl M. Facilitating meta-analyses by deriving relative effect and precision estimates for alternative comparisons from a set of estimates presented by exposure level or disease category. Stat Med. 2008;27:954-970. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 438] [Cited by in RCA: 531] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 15. | Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557-560. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39087] [Cited by in RCA: 46097] [Article Influence: 2095.3] [Reference Citation Analysis (3)] |
| 16. | Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. 1997;315:629-634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34245] [Cited by in RCA: 40150] [Article Influence: 1433.9] [Reference Citation Analysis (2)] |
| 17. | Fry JS, Lee PN. Revisiting the association between environmental tobacco smoke exposure and lung cancer risk. I. The dose-response relationship with amount and duration of smoking by the husband. Indoor Built Environ. 2000;9:303-316. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 18. | Greenland S, Longnecker MP. Methods for trend estimation from summarized dose-response data, with applications to meta-analysis. Am J Epidemiol. 1992;135:1301-1309. [PubMed] |
| 19. | Fry JS, Lee PN. Revisiting the association between environmental tobacco smoke exposure and lung cancer risk. II. Adjustment for the potential confounding effects of fruit, vegetables, dietary fat and education. Indoor Built Environ. 2001;10:20-39. [DOI] [Full Text] |
| 20. | Lee PN, Fry JS. The relationship between lung cancer and ETS exposure: adjustment for the potential confounding effects of multiple risk factors and for misclassification of active smoking status. Updated analyses. Sutton, Surrey: P N Lee Statistics and Computing Ltd 2006; Available from: http//www.pnlee.co.uk/Reports.htm. |
| 21. | Lee PN, Forey BA. Misclassification of smoking habits as a source of bias in the study of environmental tobacco smoke and lung cancer. Stat Med. 1996;15:581-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 22. | Lee PN, Forey BA, Fry JS. Revisiting the association between environmental tobacco smoke exposure and lung cancer risk. III. Adjustment for the biasing effect of misclassification of smoking habits. Indoor Built Environ. 2001;10:384-398. [DOI] [Full Text] |
| 24. | National Cancer Institute. Shopland DR, editor. Respiratory health effects of passive smoking: lung cancer and other disorders. The report of the US Environmental Protection Agency. Bethesda, MD: US Department of Health and Human Services, Public Health Service, National Institutes of Health 1993; Available from: http//cancercontrol.cancer.gov/tcrb/monographs/. |
| 25. | Lee P. Passive smoking and lung cancer. Strength of evidence on passive smoking and lung cancer is overstated. BMJ. 1998;317:346-347; author reply 348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 26. | Lee PN, Forey BA. Misclassification of smoking habits as determined by cotinine or by repeated self-report - a summary of evidence from 42 studies. J Smoking-Related Dis. 1995;6:109-129. |
| 27. | Trichopoulos D, Kalandidi A, Sparros L. Lung cancer and passive smoking: conclusion of Greek study. Lancet. 1983;2:677-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 52] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 28. | Hirayama T. Lung cancer in Japan: effects of nutrition and passive smoking. Lung cancer: causes and prevention, Proceedings of the International Lung Cancer Update Conference, New Orleans, Louisiana, March 3-5, 1983. Deerfield Beach, Florida: Verlag Chemie International, Inc 1984; 175-195. |
| 29. | Lam WK. A clinical and epidemiological study of carcinoma of lung in Hong Kong [Thesis]. Hong Kong: University of Hong Kong 1985; Available from: http//hub.hku.hk/handle/10722/28030. |
| 30. | Lam TH, Kung IT, Wong CM, Lam WK, Kleevens JW, Saw D, Hsu C, Seneviratne S, Lam SY, Lo KK. Smoking, passive smoking and histological types in lung cancer in Hong Kong Chinese women. Br J Cancer. 1987;56:673-678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 76] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 31. | Geng GY, Liang ZH, Zhang AY, Wu GL. On the relationship between cigarette smoking and female lung cancer. Smoking and health 1987, Proceedings of the 6th World Conference on Smoking and Health, Tokyo, 9-12 November 1987. Amsterdam: Elsevier Science Publishers B.V. (Biomedical Division) 1988; 483-486 International Congress Series No. 780. |
| 32. | Kalandidi A, Katsouyanni K, Voropoulou N, Bastas G, Saracci R, Trichopoulos D. Passive smoking and diet in the etiology of lung cancer among non-smokers. Cancer Causes Control. 1990;1:15-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 75] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 33. | Fontham ET, Correa P, Reynolds P, Wu-Williams A, Buffler PA, Greenberg RS, Chen VW, Alterman T, Boyd P, Austin DF. Environmental tobacco smoke and lung cancer in nonsmoking women. A multicenter study. JAMA. 1994;271:1752-1759. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 83] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 34. | Wang SY, Hu YL, Wu YL, Li X, Chi GB, Chen Y, Dai WS. A comparative study of the risk factors for lung cancer in Guangdong, China. Lung Cancer. 1996;14 Suppl 1:S99-105. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 40] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 35. | Zheng S, Fan R, Wu Z. [Studies on relationship between passive smoking and lung cancer in non-smoking women]. Zhonghua Yu Fang Yi Xue Za Zhi. 1997;31:163-165. [PubMed] |
| 36. | Zaridze D, Maximovitch D, Zemlyanaya G, Aitakov ZN, Boffetta P. Exposure to environmental tobacco smoke and risk of lung cancer in non-smoking women from Moscow, Russia. Int J Cancer. 1998;75:335-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 37. | Lee CH, Ko YC, Goggins W, Huang JJ, Huang MS, Kao EL, Wang HZ. Lifetime environmental exposure to tobacco smoke and primary lung cancer of non-smoking Taiwanese women. Int J Epidemiol. 2000;29:224-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 75] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Fang J, Gan DK, Zheng SH, Zhang HW. [A case-control study of the risk factors for lung cancer among Chinese women who have never smoked]. Wei Sheng Yan Jiu. 2006;35:464-467. [PubMed] |
| 39. | Yang P, Sun Z, Krowka MJ, Aubry MC, Bamlet WR, Wampfler JA, Thibodeau SN, Katzmann JA, Allen MS, Midthun DE. Alpha1-antitrypsin deficiency carriers, tobacco smoke, chronic obstructive pulmonary disease, and lung cancer risk. Arch Intern Med. 2008;168:1097-1103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 122] [Cited by in RCA: 116] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 40. | Liang H, Guan P, Yin Z, Li X, He Q, Zhou B. Risk of lung cancer following nonmalignant respiratory conditions among nonsmoking women living in Shenyang, Northeast China. J Womens Health (Larchmt). 2009;18:1989-1995. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 41. | Jiang T, Song H, Peng X, Yan L, Yu M, Liu Y, Liu H, Liu F, Lu Y. [A case-control study on non-smoking primary lung cancers in Sichuan, China]. Zhongguo Fei Ai Za Zhi. 2010;13:511-516. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 42. | Lin Y, Cai L. Environmental and dietary factors and lung cancer risk among Chinese women: a case-control study in southeast China. Nutr Cancer. 2012;64:508-514. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 43. | Lo YL, Hsiao CF, Chang GC, Tsai YH, Huang MS, Su WC, Chen YM, Hsin CW, Chang CH, Yang PC. Risk factors for primary lung cancer among never smokers by gender in a matched case-control study. Cancer Causes Control. 2013;24:567-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 44] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 44. | Masjedi MR, Naghan PA, Taslimi S, Yousefifard M, Ebrahimi SM, Khosravi A, Karimi S, Hosseini M, Mortaz E. Opium could be considered an independent risk factor for lung cancer: a case-control study. Respiration. 2013;85:112-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 45. | Kim CH, Lee YC, Hung RJ, McNallan SR, Cote ML, Lim WY, Chang SC, Kim JH, Ugolini D, Chen Y. Exposure to secondhand tobacco smoke and lung cancer by histological type: a pooled analysis of the International Lung Cancer Consortium (ILCCO). Int J Cancer. 2014;135:1918-1930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 89] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 46. | Wu-Williams AH, Dai XD, Blot W, Xu ZY, Sun XW, Xiao HP, Stone BJ, Yu SF, Feng YP, Ershow AG. Lung cancer among women in north-east China. Br J Cancer. 1990;62:982-987. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 224] [Cited by in RCA: 200] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 47. | Brenner DR, Hung RJ, Tsao MS, Shepherd FA, Johnston MR, Narod S, Rubenstein W, McLaughlin JR. Lung cancer risk in never-smokers: a population-based case-control study of epidemiologic risk factors. BMC Cancer. 2010;10:285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 51] [Cited by in RCA: 62] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 48. | Al-Zoughool M, Pintos J, Richardson L, Parent MÉ, Ghadirian P, Krewski D, Siemiatycki J. Exposure to environmental tobacco smoke (ETS) and risk of lung cancer in Montreal: a case-control study. Environ Health. 2013;12:112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 49. | Neuberger JS, Mahnken JD, Mayo MS, Field RW. Risk factors for lung cancer in Iowa women: implications for prevention. Cancer Detect Prev. 2006;30:158-167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 50. | Pandey A, Sharma S. Abstracts of the ISEE (International Society of Environmental Epidemiology) 20th Annual Conference. Pasadena, California, USA. October 12-16, 2008. Epidemiology. 2008;19 Suppl 6:S13-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 51. | Gallegos-Arreola MP, Figuera-Villanueva LE, Troyo-Sanroman R, Morgán-Villela G, Puebla-Pérez AM, Flores-Marquez MR, Zúniga-González GM. CYP1A1 *2B and *4 polymorphisms are associated with lung cancer susceptibility in Mexican patients. Int J Biol Markers. 2008;23:24-30. [PubMed] |
| 52. | Du YX, Cha Q, Chen YZ, Wu JM. Exposure to environmental tobacco smoke and female lung cancer in Guangzhou, China. In: Proceedings of Indoor 1993; 511-516. |
| 53. | Kabat GC, Wynder EL. Lung cancer in nonsmokers. Cancer. 1984;53:1214-1221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 54. | Zhong L, Goldberg MS, Gao YT, Jin F. A case-control study of lung cancer and environmental tobacco smoke among nonsmoking women living in Shanghai, China. Cancer Causes Control. 1999;10:607-616. [PubMed] |
| 55. | Gorlova OY, Zhang Y, Schabath MB, Lei L, Zhang Q, Amos CI, Spitz MR. Never smokers and lung cancer risk: a case-control study of epidemiological factors. Int J Cancer. 2006;118:1798-1804. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 82] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 56. | Wen W, Shu XO, Gao YT, Yang G, Li Q, Li H, Zheng W. Environmental tobacco smoke and mortality in Chinese women who have never smoked: prospective cohort study. BMJ. 2006;333:376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 65] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 57. | Schwartz AG, Yang P, Swanson GM. Familial risk of lung cancer among nonsmokers and their relatives. Am J Epidemiol. 1996;144:554-562. [PubMed] |
| 58. | Stockwell HG, Goldman AL, Lyman GH, Noss CI, Armstrong AW, Pinkham PA, Candelora EC, Brusa MR. Environmental tobacco smoke and lung cancer risk in nonsmoking women. J Natl Cancer Inst. 1992;84:1417-1422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 66] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 59. | Zeka A, Mannetje A, Zaridze D, Szeszenia-Dabrowska N, Rudnai P, Lissowska J, Fabiánová E, Mates D, Bencko V, Navratilova M. Lung cancer and occupation in nonsmokers: a multicenter case-control study in Europe. Epidemiology. 2006;17:615-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 37] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 60. | Sun XW, Dai XD, Lin CY, Shi YB, Ma YY, Li W. Environmental tobacco smoke (ETS) and lung cancer among nonsmoking women in Harbin. Lung Cancer. 1996;14:S237. |
| 61. | Rapiti E, Jindal SK, Gupta D, Boffetta P. Passive smoking and lung cancer in Chandigarh, India. Lung Cancer. 1999;23:183-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 32] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 62. | Wang L, Lubin JH, Zhang SR, Metayer C, Xia Y, Brenner A, Shang B, Wang Z, Kleinerman RA. Lung cancer and environmental tobacco smoke in a non-industrial area of China. Int J Cancer. 2000;88:139-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 63. | Rachtan J. Smoking, passive smoking and lung cancer cell types among women in Poland. Lung Cancer. 2002;35:129-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 64. | Zatloukal P, Kubík A, Pauk N, Tomásek L, Petruzelka L. Adenocarcinoma of the lung among women: risk associated with smoking, prior lung disease, diet and menstrual and pregnancy history. Lung Cancer. 2003;41:283-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 43] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 65. | Olivo-Marston SE, Yang P, Mechanic LE, Bowman ED, Pine SR, Loffredo CA, Alberg AJ, Caporaso N, Shields PG, Chanock S. Childhood exposure to secondhand smoke and functional mannose binding lectin polymorphisms are associated with increased lung cancer risk. Cancer Epidemiol Biomarkers Prev. 2009;18:3375-3383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 45] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 66. | Boffetta P, Agudo A, Ahrens W, Benhamou E, Benhamou S, Darby SC, Ferro G, Fortes C, Gonzalez CA, Jöckel KH. Multicenter case-control study of exposure to environmental tobacco smoke and lung cancer in Europe. J Natl Cancer Inst. 1998;90:1440-1450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 148] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 67. | Correa P, Pickle LW, Fontham E, Lin Y, Haenszel W. Passive smoking and lung cancer. Lancet. 1983;2:595-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 128] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 68. | Akiba S, Kato H, Blot WJ. Passive smoking and lung cancer among Japanese women. Cancer Res. 1986;46:4804-4807. [PubMed] |
| 69. | Lee YA, Hung R, Boffetta P, Brennan P, Christiani D, Duell EJ, Field JK, Kiyohara C, Lan Q, Lazarus P. A pooled analysis on the associations between involuntary smoking and lung cancer risk by histological types [Abstract]. Cancer Epidemiol Biomarkers Prev. 2010;19:892-893. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 70. | Shimizu H, Morishita M, Mizuno K, Masuda T, Ogura Y, Santo M, Nishimura M, Kunishima K, Karasawa K, Nishiwaki K. A case-control study of lung cancer in nonsmoking women. Tohoku J Exp Med. 1988;154:389-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 30] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 71. | Franco-Marina F, Villalba Caloca J, Corcho-Berdugo A. Role of active and passive smoking on lung cancer etiology in Mexico City. Salud Publica Mex. 2006;48 Suppl 1:S75-S82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 72. | Janerich DT, Thompson WD, Varela LR, Greenwald P, Chorost S, Tucci C, Zaman MB, Melamed MR, Kiely M, McKneally MF. Lung cancer and exposure to tobacco smoke in the household. N Engl J Med. 1990;323:632-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 181] [Cited by in RCA: 141] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 73. | Cardenas VM, Thun MJ, Austin H, Lally CA, Clark WS, Greenberg RS, Heath CW. Environmental tobacco smoke and lung cancer mortality in the American Cancer Society’s Cancer Prevention Study. II. Cancer Causes Control. 1997;8:57-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 74. | Lim WY, Chen Y, Chuah KL, Eng P, Leong SS, Lim E, Lim TK, Ng A, Poh WT, Tee A. Female reproductive factors, gene polymorphisms in the estrogen metabolism pathway, and risk of lung cancer in Chinese women. Am J Epidemiol. 2012;175:492-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 75. | Garfinkel L. Time trends in lung cancer mortality among nonsmokers and a note on passive smoking. J Natl Cancer Inst. 1981;66:1061-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 161] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 76. | A review of human carcinogens: Part E: Personal habits and indoor combustions; International Agency for Research on Cancer. Second-hand tobacco smoke. Volume 100. Lyon, France: IARC 2012; 215-265. |
| 77. | Office of Population Censuses and Surveys. Colhoun H, Prescott-Clarke P, editors. Health survey for England 1994. Volume I: Findings. Volume II: Survey methodology & documentation. London: HMSO 1996; . |
| 78. | Ziegler RG, Mason TJ, Stemhagen A, Hoover R, Schoenberg JB, Gridley G, Virgo PW, Altman R, Fraumeni JF, Jr . Dietary carotene and vitamin A and risk of lung cancer among white men in New Jersey. J Natl Cancer Inst. 1984;73:1429-1435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 79. | Pirkle JL, Flegal KM, Bernert JT, Brody DJ, Etzel RA, Maurer KR. Exposure of the US population to environmental tobacco smoke: the Third National Health and Nutrition Examination Survey, 1988 to 1991. JAMA. 1996;275:1233-1240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 93] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 80. | Benowitz NL, Bernert JT, Caraballo RS, Holiday DB, Wang J. Optimal serum cotinine levels for distinguishing cigarette smokers and nonsmokers within different racial/ethnic groups in the United States between 1999 and 2004. Am J Epidemiol. 2009;169:236-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 466] [Cited by in RCA: 538] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 81. | Phillips K, Bentley MC, Howard DA, Alván G. Assessment of air quality in Stockholm by personal monitoring of nonsmokers for respirable suspended particles and environmental tobacco smoke. Scand J Work Environ Health. 1996;22 Suppl 1:1-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 82. | Phillips K, Bentley MC, Howard DA, Alván G, Huici A. Assessment of air quality in Barcelona by personal monitoring of nonsmokers for respirable suspended particles and environmental tobacco smoke. Environ Int. 1997;23:173-196. [DOI] [Full Text] |
| 83. | Phillips K, Howard DA, Bentley MC, Alván G. Assessment of air quality in Kuala Lumpur by personal monitoring of nonsmokers at home and in the workplace by reference to respirable suspended particles (RSP) and environmental tobacco smoker (ETS). Indoor and built environment problems in Asia, Proceedings of a conference held in Kuala Lumpur, Malaysia on 4th & 5th September 1997. Rothenfluh, Switzerland: The International Society of the Built Environment 1997; 151-159. |
| 84. | Phillips K, Howard DA, Bentley MC, Alván G. Assessment by personal monitoring of respirable suspended particles and environmental tobacco smoke exposure for non-smokers in Sydney, Australia. Indoor Built Environ. 1998;7:188-203. [DOI] [Full Text] |
| 85. | Phillips K, Howard DA, Bentley MC. Assessment of environmental tobacco smoke and respirable suspended particle exposures for nonsmokers in Lisbon by personal monitoring. Environ Int. 1998;24:301-324. [DOI] [Full Text] |
| 86. | Phillips K, Howard DA, Bentley MC, Alván G. Measured exposures by personal monitoring for respirable suspended particles and environmental tobacco smoke of housewives and office workers resident in Bremen, Germany. Int Arch Occup Environ Health. 1998;71:201-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 87. | Phillips K, Bentley MC, Howard DA, Alván G. Assessment of environmental tobacco smoke and respirable suspended particle exposures for nonsmokers in Prague using personal monitoring. Int Arch Occup Environ Health. 1998;71:379-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 88. | Phillips K, Howard DA, Bentley MC, Alván G. Environmental tobacco smoke and respirable suspended particle exposures for non-smokers in Beijing. Indoor Built Environ. 1998;7:254-269. [DOI] [Full Text] |
| 89. | Lee PN, Forey BA, Coombs KJ. Systematic review with meta-analysis of the epidemiological evidence in the 1900s relating smoking to lung cancer. BMC Cancer. 2012;12:385. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 196] [Cited by in RCA: 191] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 90. | Thornton A, Lee P, Fry J. Differences between smokers, ex-smokers, passive smokers and non-smokers. J Clin Epidemiol. 1994;47:1143-1162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 90] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 91. | Dallongeville J, Marécaux N, Fruchart JC, Amouyel P. Cigarette smoking is associated with unhealthy patterns of nutrient intake: a meta-analysis. J Nutr. 1998;128:1450-1457. [PubMed] |
| 92. | Forastiere F, Mallone S, Lo Presti E, Baldacci S, Pistelli F, Simoni M, Scalera A, Pedreschi M, Pistelli R, Corbo G. Characteristics of nonsmoking women exposed to spouses who smoke: epidemiologic study on environment and health in women from four Italian areas. Environ Health Perspect. 2000;108:1171-1177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 93. | Iribarren C, Friedman GD, Klatsky AL, Eisner MD. Exposure to environmental tobacco smoke: association with personal characteristics and self reported health conditions. J Epidemiol Community Health. 2001;55:721-728. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 55] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 94. | Connor Gorber S, Schofield-Hurwitz S, Hardt J, Levasseur G, Tremblay M. The accuracy of self-reported smoking: a systematic review of the relationship between self-reported and cotinine-assessed smoking status. Nicotine Tob Res. 2009;11:12-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 674] [Cited by in RCA: 786] [Article Influence: 49.1] [Reference Citation Analysis (0)] |
| 95. | Lehnert G, Garfinkel L, Hirayama T, Schmähl D, Uberla K, Wynder EL, Lee P. Roundtable discussion: symposium: medical perspectives on passive smoking. Prev Med. 1984;13:730-746. [PubMed] |
| 96. | England LJ, Grauman A, Qian C, Wilkins DG, Schisterman EF, Yu KF, Levine RJ. Misclassification of maternal smoking status and its effects on an epidemiologic study of pregnancy outcomes. Nicotine Tob Res. 2007;9:1005-1013. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 104] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 97. | Jung-Choi KH, Khang YH, Cho HJ. Hidden female smokers in Asia: a comparison of self-reported with cotinine-verified smoking prevalence rates in representative national data from an Asian population. Tob Control. 2012;21:536-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 186] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 98. | Sterling TD. Publication decisions and their possible effects on inferences drawn from tests of significance - or vice versa. J Am Stat Assoc. 1959;54:30-34. [RCA] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 209] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 99. | Begg CB, Mazumdar M. Operating characteristics of a rank correlation test for publication bias. Biometrics. 1994;50:1088-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10586] [Cited by in RCA: 12110] [Article Influence: 403.7] [Reference Citation Analysis (0)] |
| 100. | Easterbrook PJ, Berlin JA, Gopalan R, Matthews DR. Publication bias in clinical research. Lancet. 1991;337:867-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2020] [Cited by in RCA: 2001] [Article Influence: 58.9] [Reference Citation Analysis (0)] |
| 101. | Lee PN. Comparison of autopsy, clinical and death certificate diagnosis with particular reference to lung cancer. A review of the published data. APMIS. 1994;102 Suppl 45:4. |
| 102. | Faccini JM. The role of histopathology in the evaluation of risk of lung cancer from environmental tobacco smoke. Exp Pathol. 1989;37:177-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 5] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 103. | Sterling TD, Rosenbaum WL, Weinkam JJ. Bias in the attribution of lung cancer as cause of death and its possible consequences for calculating smoking-related risks. Epidemiology. 1992;3:11-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 104. | Hammond EC. Smoking in relation to the death rates of one million men and women. Epidemiological approaches to the study of cancer and other chronic diseases. Bethesda, Maryland: U.S. Department of Health, Education, and Welfare. Public Health Service National Cancer Institute 1966; 127-204. |
| 105. | Chan WC, Fung SC. Lung cancer in non-smokers in Hong Kong. Cancer Epidemiology. Volume 6. Stuttgart, New York: Gustav Fischer Verlag 1982; 199-202. |
| 106. | Chan WC, Colbourne MJ, Fung SC, Ho HC. Bronchial cancer in Hong Kong 1976-1977. Br J Cancer. 1979;39:182-192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 43] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 107. | Chan WC. Zahlen aus Hongkong. Munch Med Wochenschr. 1982;124:16. |
| 108. | Trichopoulos D, Kalandidi A, Sparros L, MacMahon B. Lung cancer and passive smoking. Int J Cancer. 1981;27:1-4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 269] [Cited by in RCA: 211] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 109. | Trichopoulos D, Kalandidi A, Tzonou A. Incidence and distribution of lung cancer in Greece. Excerpta Medica International Congress Series. 1982;558:10-17. |
| 110. | Buffler PA, Pickle LW, Mason TJ, Contant C. The causes of lung cancer in Texas. Lung cancer: causes and prevention, Proceedings of the International Lung Cancer Update Conference, New Orleans, Louisiana, March 3-5, 1983. Deerfield Beach, Florida: Verlag Chemie International, Inc 1984; 83-99. |
| 111. | Hirayama T. Non-smoking wives of heavy smokers have a higher risk of lung cancer: a study from Japan. Br Med J (Clin Res Ed). 1981;282:183-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 501] [Cited by in RCA: 425] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 112. | Hirayama T. Cancer mortality in nonsmoking women with smoking husbands based on a large-scale cohort study in Japan. Prev Med. 1984;13:680-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 97] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 113. | Hirayama T. Passive smoking--a new target of epidemiology. Tokai J Exp Clin Med. 1985;10:287-293. [PubMed] |
| 114. | Hirayama T. Passive smoking and cancer: an epidemiological review. Changing cancer patterns and topics in cancer epidemiology. In Memory of Professor Mitsuo Segi. Volume 33. Basel, Switzerland: S. Karger AG 1987; 127-135. |
| 115. | Hirayama T. Health effects of active and passive smoking. Smoking and health 1987, Proceedings of the 6th World Conference on Smoking and Health, Tokyo, 9-12 November 1987. Amsterdam: Elsevier Science Publishers B.V. (Biomedical Division) 1988; 75-86. |
| 116. | Hirayama T. A large-scale cohort study on risk factors for primary liver cancer, with special reference to the role of cigarette smoking. Cancer Chemother Pharmacol. 1989;23 Suppl:S114-S117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 39] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 117. | Hirayama T. Dietary habits are of limited importance in influencing the lung cancer risk among Japanese females who never smoked. Present and future of indoor air quality, Proceedings of the Brussels Conference, 14-16 February 1989. Amsterdam: Elsevier Science Publishers BV (Biomedical Division) 1989; 77-82. |
| 118. | Hirayama T. Life-style and mortality: A large scale census based cohort study in Japan. Volume 6. Basel, Switzerland: Karger 1990; . |
| 119. | Hirayama T. Passive smoking and cancer: The association between husbands smoking and cancer in the lung of non-smoking wives. Indoor air quality, International Conference on Indoor Air Quality, Tokyo, November 4-6, 1987. Berlin Heidelberg: Springer-Verlag 1990; 299-311. |
| 120. | Hirayama T. Lung cancer and other diseases related to passive smoking: a large-scale cohort study. Control of tobacco-related cancers and other diseases. Bombay: Oxford University Press 1992; 129-127. |
| 121. | Garfinkel L, Auerbach O, Joubert L. Involuntary smoking and lung cancer: a case-control study. J Natl Cancer Inst. 1985;75:463-469. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 122. | Stellman SD, Garfinkel L. Passive smoking and lung cancer: An American Cancer Society study. Indoor air quality, International Conference on Indoor Air Quality, Tokyo, November 4-6, 1987. Berlin Heidelberg: Springer-Verlag 1990; 283-289. |
| 123. | Wu AH, Henderson BE, Pike MC, Yu MC. Smoking and other risk factors for lung cancer in women. J Natl Cancer Inst. 1985;74:747-751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 124. | Lee PN, Chamberlain J, Alderson MR. Relationship of passive smoking to risk of lung cancer and other smoking-associated diseases. Br J Cancer. 1986;54:97-105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 129] [Cited by in RCA: 105] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 125. | Brownson RC, Reif JS, Keefe TJ, Ferguson SW, Pritzl JA. Risk factors for adenocarcinoma of the lung. Am J Epidemiol. 1987;125:25-34. [PubMed] |
| 126. | Gao YT, Blot WJ, Zheng W, Ershow AG, Hsu CW, Levin LI, Zhang R, Fraumeni JF. Lung cancer among Chinese women. Int J Cancer. 1987;40:604-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 268] [Cited by in RCA: 244] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 127. | Humble CG, Samet JM, Pathak DR. Marriage to a smoker and lung cancer risk. Am J Public Health. 1987;77:598-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 52] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 128. | Koo LC, Ho JH, Saw D, Ho CY. Measurements of passive smoking and estimates of lung cancer risk among non-smoking Chinese females. Int J Cancer. 1987;39:162-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 48] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 129. | Koo LC, Ho JH-C, Saw D. Active and passive smoking among female lung cancer patients and controls in Hong Kong. J Exp Clin Cancer Res. 1983;4:367-375. |
| 130. | Koo LC, Ho JH-C, Saw D. Is passive smoking an added risk factor for lung cancer in Chinese women? J Exp Clin Cancer Res. 1984;3:277-283. |
| 131. | Koo LC, Ho JH, Lee N. An analysis of some risk factors for lung cancer in Hong Kong. Int J Cancer. 1985;35:149-155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 68] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 132. | Koo LC, Ho JH, Rylander R. Life-history correlates of environmental tobacco smoke: a study on nonsmoking Hong Kong Chinese wives with smoking versus nonsmoking husbands. Soc Sci Med. 1988;26:751-760. [PubMed] |
| 133. | Koo LC, Ho JH. Diet as a confounder of the association between air pollution and female lung cancer: Hong Kong studies on exposures to environmental tobacco smoke, incense, and cooking fumes as examples. Lung Cancer. 1996;14 Suppl 1:S47-S61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 35] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 134. | Pershagen G, Hrubec Z, Svensson C. Passive smoking and lung cancer in Swedish women. Am J Epidemiol. 1987;125:17-24. [PubMed] |
| 135. | Pershagen G, Svensson C, Hrubec Z. Environmental tobacco smoke and lung cancer in Swedish women. Indoor Air 87, Volume 2, Proceedings of the 4th International Conference on Indoor Air Quality and Climate, Berlin (West), 17-21 August 1987. Berlin: Institute for Water, Soil and Air Hygiene 1988; 34-38. |
| 136. | Butler TL. The relationship of passive smoking to various health outcomes among Seventh day Adventists in California [Thesis]. Los Angeles: University of California 1988; . |
| 137. | Geng G, Liang Z, Xu R, Liu J, Shi P. The relationship between smoking and lung cancer in humans. In: International symposium on lifestyle factors and human lung cancer. Dec 12-16 1994, Guangzhou, People’s Republic of China 1994; 1-8. |
| 138. | Inoue R, Hirayama T. Passive smoking and lung cancer in women. Smoking and health 1987, Proceedings of the 6th World Conference on Smoking and Health, Tokyo, 9-12 November 1987. Amsterdam: Elsevier Science Publishers B.V. (Biomedical Division) 1988; 283-285. |
| 139. | Choi SY, Lee KH, Lee TO. A case-control study on risk factors in lung cancer. Korean J Epidemiol. 1989;11:66-80. |
| 140. | Hole DJ, Gillis CR, Chopra C, Hawthorne VM. Passive smoking and cardiorespiratory health in a general population in the west of Scotland. BMJ. 1989;299:423-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 92] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 141. | Gillis CR, Hole DJ, Hawthorne VM, Boyle P. The effect of environmental tobacco smoke in two urban communities in the west of Scotland. Eur J Respir Dis Suppl. 1984;133:121-126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 142. | Schoenberg J, Klotz J. A case-control study of radon and lung cancer among New Jersey women. USA: New Jersey State Department of Health 1989; Technical report-phase I Available from: http//www.state.nj.us/health/eohs/regional_state/air/case_control_women.pdf. |
| 143. | Svensson C, Pershagen G, Klominek J. Smoking and passive smoking in relation to lung cancer in women. Acta Oncol. 1989;28:623-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 40] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 144. | Svensson C. Lung cancer etiology in women [Thesis]. Stockholm: Departments of Oncology and Environmental Hygiene, Karolinska Institute 1988; . |
| 145. | Varela LR. Assessment of the association between passive smoking and lung cancer [Thesis]. New Haven, Conn: Faculty of the Graduate School of Yale University 1987; . |
| 146. | Sobue T. Association of indoor air pollution and lifestyle with lung cancer in Osaka, Japan. Int J Epidemiol. 1990;19 Suppl 1:S62-S66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 147. | Sobue T, Suzuki R, Nakayama N, Inubuse C, Matsuda M, Doi O, Mori T, Furuse K, Fukuoka M, Yasumitsu T. Passive smoking smong nonsmoking women and the relationship between indoor air pollution and lung cancer incidence - results of a multicenter case controlled study. Gan No Rinsho. 1990;36:329-333. |
| 148. | Liu ZY, He XZ, Chapman RS. Smoking and other risk factors for lung cancer in Xuanwei, China. Int J Epidemiol. 1991;20:26-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 149. | Liu Z. [A case-control study of lung cancer risk factors in Xuanwei]. Zhonghua Liu Xing Bing Xue Za Zhi. 1990;11:79-83. [PubMed] |
| 150. | He XZ, Chen W, Liu ZY, Chapman RS. An epidemiological study of lung cancer in Xuan Wei County, China: current progress. Case-control study on lung cancer and cooking fuel. Environ Health Perspect. 1991;94:9-13. [PubMed] [DOI] [Full Text] |
| 151. | Brownson RC, Alavanja MC, Hock ET, Loy TS. Passive smoking and lung cancer in nonsmoking women. Am J Public Health. 1992;82:1525-1530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 75] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 152. | Butler WJ. Workplace exposure to ETS and lung cancer: a more detailed presentation of the data from a negative study, Brownson et al (1992). Washington DC: Occupational Safety and Health Administration 1994; . |
| 153. | Alavanja MCR, Brownson RC, Benichou J, Swanson C, Boice JD, Jr . Attributable risk of lung cancer in nonsmoking women. In: International symposium on lifestyle factors and human lung cancer. Guangzhou, People’s Republic of China 1994; 1-13. |
| 154. | Alavanja MC, Brownson RC, Benichou J, Swanson C, Boice JD. Attributable risk of lung cancer in lifetime nonsmokers and long-term ex-smokers (Missouri, United States). Cancer Causes Control. 1995;6:209-216. [PubMed] |
| 155. | Bennett WP, Alavanja MC, Blomeke B, Vähäkangas KH, Castrén K, Welsh JA, Bowman ED, Khan MA, Flieder DB, Harris CC. Environmental tobacco smoke, genetic susceptibility, and risk of lung cancer in never-smoking women. J Natl Cancer Inst. 1999;91:2009-2014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 118] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 156. | Stockwell HG, Candelora EC, Armstrong AW, Pinkham PA. Environmental tobacco smoke and lung cancer in never smoking women. Am J Epidemiol. 1991;134:724. |
| 157. | Du Y, Cha Q, Chen X, Chen Y, Lei Y, Xue S. Exposure to environmental tobacco smoke and female lung cancer. Indoor Air. 1995;5:231-236. [DOI] [Full Text] |
| 158. | Laux G, Madirazza D. [Follow-up studies of surgery for varicoses veins. Psychological and surgical aspects]. ZFA (Stuttgart). 1977;53:741-744. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 52] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 159. | Lei YX, Cai WC, Chen YZ, Du YX. Some lifestyle factors in human lung cancer: a case-control study of 792 lung cancer cases. Lung Cancer. 1996;14 Suppl 1:S121-S136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 160. | Liu Q, Sasco AJ, Riboli E, Hu MX. Indoor air pollution and lung cancer in Guangzhou, People’s Republic of China. Am J Epidemiol. 1993;137:145-154. [PubMed] |
| 161. | Fontham ET, Correa P, WuWilliams A, Reynolds P, Greenberg RS, Buffler PA, Chen VW, Boyd P, Alterman T, Austin DF. Lung cancer in nonsmoking women: a multicenter case-control study. Cancer Epidemiol Biomarkers Prev. 1991;1:35-43. [PubMed] |
| 162. | Fontham ETH, Correa P, Buffler PA, Greenberg R, Reynolds P, Wu-Williams A. Environmental tobacco smoke and lung cancer. Cancer Bull. 1993;45:92-94. |
| 163. | Fontham ET, Correa P, Chen VW. Passive smoking and lung cancer. J La State Med Soc. 1993;145:132-136. [PubMed] |
| 164. | Reynolds P, von Behren J, Fontham ET, Correa P, Wu A, Buffler PA, Greenberg RS. Occupational exposure to environmental tobacco smoke. JAMA. 1996;275:441-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 165. | Layard MW. Ischemic heart disease, lung cancer, and spousal smoking in the National Mortality Followback Survey. Submitted to OSHA re Proposed Rules. Federal Register. 1994;59:65, Docket No H-122. |
| 166. | de Waard F, Kemmeren JM, van Ginkel LA, Stolker AA. Urinary cotinine and lung cancer risk in a female cohort. Br J Cancer. 1995;72:784-787. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 22] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 167. | Ellard GA, de Waard F, Kemmeren JM. Urinary nicotine metabolite excretion and lung cancer risk in a female cohort. Br J Cancer. 1995;72:788-791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 168. | Kabat GC, Stellman SD, Wynder EL. Relation between exposure to environmental tobacco smoke and lung cancer in lifetime nonsmokers. Am J Epidemiol. 1995;142:141-148. [PubMed] |
| 169. | Kabat GC. Epidemiologic studies of the relationship between passive smoking and lung cancer. In: Presented at the 1990; 187-199. |
| 170. | Kabat GC. Aspects of the epidemiology of lung cancer in smokers and nonsmokers in the United States. In: International symposium on lifestyle factors and human lung cancer. Dec 12-16 1994, Guangzhou, People’s Republic of China 1994; 1-27. |
| 171. | Wang TJ, Zhou BS, Shi JP. Lung cancer in nonsmoking Chinese women: a case-control study. Lung Cancer. 1996;14 Suppl 1:S93-S98. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 64] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 172. | Cardenas VM. Environmental tobacco smoke and lung cancer mortality in the American Cancer Society’s Cancer Prevention Study II [Thesis]. Atlanta, Georgia: Emory University 1994; . |
| 173. | Cardenas VM. Environmental tobacco smoke and lung cancer mortality in the American Cancer Society’s Cancer Prevention Study II. Diss Abstr Int. 1995;56/07-B:3714. |
| 174. | Auvinen A, Mäkeläinen I, Hakama M, Castrén O, Pukkala E, Reisbacka H, Rytömaa T. Indoor radon exposure and risk of lung cancer: a nested case-control study in Finland. J Natl Cancer Inst. 1996;88:966-972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 73] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 175. | Boffetta P, Brennan S, Lea S, Ferro G. Lung cancer and exposure to environmental tobacco smoke. IARC biennial report. In: IARC 1998; 76-77. |
| 176. | Boffetta P, Garcia-Gómez M, Pompe-Kirn V, Zaridze D, Bellander T, Bulbulyan M, Caballero JD, Ceccarelli F, Colin D, Dizdarevic T. Cancer occurrence among European mercury miners. Cancer Causes Control. 1998;9:591-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 177. | Boffetta P, Agudo A, Ahrens W, Benhamou E, Benhamou S, Darby SC, Ferro G, Fortes C, Gonzalez CA, Jöckel KH. European multicentre case-control study of lung cancer in non-smokers. Detailed results on exposure to environmental tobacco smoke. Lyon: International Agency for Research on Cancer 1998; . |
| 178. | Boffetta P, Nyberg F, Agudo A, Benhamou E, Jockel KH, Kreuzer M, Merletti F, Pershagen G, Pohlabeln H, Simonato L. Risk of lung cancer from exposure to environmental tobacco smoke from cigars, cigarillos and pipes. Int J Cancer. 1999;83:805-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 179. | Brennan P, Butler J, Agudo A, Benhamou S, Darby S, Fortes C, Jöckel KH, Kreuzer M, Nyberg F, Pohlabeln H. Joint effect of diet and environmental tobacco smoke on risk of lung cancer among nonsmokers. J Natl Cancer Inst. 2000;92:426-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 180. | Kreuzer M, Krauss M, Kreienbrock L, Jöckel KH, Wichmann HE. Environmental tobacco smoke and lung cancer: a case-control study in Germany. Am J Epidemiol. 2000;151:241-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 31] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 181. | Shen XB, Wang GX, Zhou BS. Relation of exposure to environmental tobacco smoke and pulmonary adenocarcinoma in non-smoking women: a case control study in Nanjing. Oncol Rep. 1998;5:1221-1223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 182. | Shen X, Wang G, Xiang L, Huang Y. Analyses of sex differentials in risk factors for primary lung adenocarcinoma. In: International symposium on lifestyle factors and human lung cancer, International symposium on lifestyle factors and human lung cancer. Guangzhou, People’s Republic of China 1994; 1-6. |
| 183. | Shen XB, Wang GX, Huang YZ, Xiang LS, Wang XH. Analysis and estimates of attributable risk factors for lung cancer in Nanjing, China. Lung Cancer. 1996;14 Suppl 1:S107-S112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 184. | Shen XB, Wang GX, Xiang LS, Huang YZ. International Symposium on Lifestyle Factors and Human Lung Cancer. Guangzhou, China, 12-16 December 1994. Proceedings and abstracts. Lung Cancer. 1996;14 Suppl 1:S1-245. [PubMed] |
| 185. | Shen X, Wang G, Xiang L, Wu JM. Relationship of passive smoking and pulmonary adenocarcinoma in non-smoking women - a case control study in Nanjing, P.R. China. Epidemiology. 1996;7:S20. [RCA] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 186. | Zaridze DG, Zemlyanaya GM. Indoor air pollution and lung cancer risk in non-smoking women in Moscow. Exp Oncol. 1994;16:441-445. |
| 187. | Zaridze D. Indoor and outdoor air pollution and the risk of lung cancer. Epidemiology. 1998;9:S90. |
| 188. | Zemlianaja GM, Zaridze DG. Lung cancer in non-smoking women in Moscow. Epidemiology. 1998;9:S89. |
| 189. | Boffetta P, Ahrens W, Nyberg F, Mukeria A, Brüske-Hohlfeld I, Fortes C, Constantinescu V, Simonato L, Batura-Gabryel H, Lea S. Exposure to environmental tobacco smoke and risk of adenocarcinoma of the lung. Int J Cancer. 1999;83:635-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 190. | Jee SH, Ohrr H, Kim IS. Effects of husbands’ smoking on the incidence of lung cancer in Korean women. Int J Epidemiol. 1999;28:824-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 69] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 191. | Speizer FE, Colditz GA, Hunter DJ, Rosner B, Hennekens C. Prospective study of smoking, antioxidant intake, and lung cancer in middle-aged women (USA). Cancer Causes Control. 1999;10:475-482. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 192. | Ko YC, Lee CH, Chen MJ, Huang CC, Chang WY, Lin HJ, Wang HZ, Chang PY. Risk factors for primary lung cancer among non-smoking women in Taiwan. Int J Epidemiol. 1997;26:24-31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 192] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 193. | Ko YC, Cheng LS, Lee CH, Huang JJ, Huang MS, Kao EL, Wang HZ, Lin HJ. Chinese food cooking and lung cancer in women nonsmokers. Am J Epidemiol. 2000;151:140-147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 168] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 194. | Lee PN. A review of the epidemiology of heart disease related to active smoking. The role of amount smoked, age and time quit smoking. Sutton, Surrey: P N Lee Statistics and Computing Ltd 2001; Available from: http//www.pnlee.co.uk/Reports.htm. |
| 195. | Malats N, Camus-Radon AM, Nyberg F, Ahrens W, Constantinescu V, Mukeria A, Benhamou S, Batura-Gabryel H, Bruske-Hohlfeld I, Simonato L. Lung cancer risk in nonsmokers and GSTM1 and GSTT1 genetic polymorphism. Cancer Epidemiol Biomarkers Prev. 2000;9:827-833. [PubMed] |
| 196. | Cohet C, Borel S, Nyberg F, Mukeria A, Brüske-Hohlfeld I, Constantinescu V, Benhamou S, Brennan P, Hall J, Boffetta P. Exon 5 polymorphisms in the O6-alkylguanine DNA alkyltransferase gene and lung cancer risk in non-smokers exposed to second-hand smoke. Cancer Epidemiol Biomarkers Prev. 2004;13:320-323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 54] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 197. | Kleinerman RA, Wang ZY, Lubin JH, Zhang SZ, Metayer C, Brenner AV. Lung cancer and indoor air pollution in rural China. Ann Epidemiol. 2000;10:469. |
| 198. | Johnson KC, Hu J, Mao Y. 1-800-AIDS-NIH. Science. 1992;257:1207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 199. | Johnson KC, Hu J, Mao Y. Passive and active smoking and breast cancer risk in Canada, 1994-97. Cancer Causes Control. 2000;11:211-221. [PubMed] |
| 200. | Johnson KC, Hu J, Mao Y. Lifetime workplace and residential exposure to environmental tobacco smoke and lung cancer in never-smoking women. Am J Epidemiol. 2000;151:S28. |
| 201. | Hu J, Mao Y, Dryer D, White K. Risk factors for lung cancer among Canadian women who have never smoked. Cancer Detect Prev. 2002;26:129-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 53] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 202. | Lagarde F, Axelsson G, Damber L, Mellander H, Nyberg F, Pershagen G. Residential radon and lung cancer among never-smokers in Sweden. Epidemiology. 2001;12:396-404. [PubMed] |
| 203. | Hou SM, Yang K, Nyberg F, Hemminki K, Pershagen G, Lambert B. Hprt mutant frequency and aromatic DNA adduct level in non-smoking and smoking lung cancer patients and population controls. Carcinogenesis. 1999;20:437-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 204. | Nishino Y, Tsubono Y, Tsuji I, Komatsu S, Kanemura S, Nakatsuka H, Fukao A, Satoh H, Hisamichi S. Passive smoking at home and cancer risk: a population-based prospective study in Japanese nonsmoking women. Cancer Causes Control. 2001;12:797-802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 205. | Ohno Y, Wakai K, Ando M, Shimokata K, Saka H, Yamamoto M, Shima K, Sugiura S, Sakai S, Nomura F. Studies on health effects of passive smoking - multicancer case-control study of the relationship between passive smoking and lung cancer in lifetime nonsmokers. In: SRF Annual Report 2001 2002; 857-861. |
| 206. | Rachtan J. A case-control study of lung cancer in Polish women. Neoplasma. 2002;49:75-80. [PubMed] |
| 207. | Enstrom JE, Kabat GC. Environmental tobacco smoke and tobacco related mortality in a prospective study of Californians, 1960-98. BMJ. 2003;326:1057. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 115] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 208. | Enstrom JE, Heath CW. Smoking cessation and mortality trends among 118,000 Californians, 1960-1997. Epidemiology. 1999;10:500-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 209. | Kubík A, Zatloukal P, Boyle P, Robertson C, Gandini S, Tomásek L, Gray N, Havel L. A case-control study of lung cancer among Czech women. Lung Cancer. 2001;31:111-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 210. | Kubík AK, Zatloukal P, Tomásek L, Petruzelka L. Lung cancer risk among Czech women: a case-control study. Prev Med. 2002;34:436-444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 211. | McGhee SM, Ho SY, Schooling M, Ho LM, Thomas GN, Hedley AJ, Mak KH, Peto R, Lam TH. Mortality associated with passive smoking in Hong Kong. BMJ. 2005;330:287-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 47] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 212. | Vineis P, Airoldi L, Veglia F, Olgiati L, Pastorelli R, Autrup H, Dunning A, Garte S, Gormally E, Hainaut P. Environmental tobacco smoke and risk of respiratory cancer and chronic obstructive pulmonary disease in former smokers and never smokers in the EPIC prospective study. BMJ. 2005;330:277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 188] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 213. | Vineis P, Anttila S, Benhamou S, Spinola M, Hirvonen A, Kiyohara C, Garte SJ, Puntoni R, Rannug A, Strange RC. Evidence of gene gene interactions in lung carcinogenesis in a large pooled analysis. Carcinogenesis. 2007;28:1902-1905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 214. | Vineis P, Hoek G, Krzyzanowski M, Vigna-Taglianti F, Veglia F, Airoldi L, Overvad K, Raaschou-Nielsen O, Clavel-Chapelon F, Linseisen J. Lung cancers attributable to environmental tobacco smoke and air pollution in non-smokers in different European countries: a prospective study. Environ Health. 2007;6:7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 89] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 215. | Veglia F, Vineis P, Overvad K, Boeing H, Bergmann M, Trichopoulou A, Trichopoulos D, Palli D, Krogh V, Tumino R. Occupational exposures, environmental tobacco smoke, and lung cancer. Epidemiology. 2007;18:769-775. [PubMed] |
| 216. | International Agency for Research on Cancer. IARC Handbooks of Cancer Prevention, Tobacco Control, Vol 13: Evaluating the effectiveness of smoke-free policies. Lyon, France: IARC 2009; . |
| 217. | Hudmon KS, Honn SE, Jiang H, Chamberlain RM, Xiang W, Ferry G, Gosbee W, Hong WK, Spitz MR. Identifying and recruiting healthy control subjects from a managed care organization: a methodology for molecular epidemiological case-control studies of cancer. Cancer Epidemiol Biomarkers Prev. 1997;6:565-571. [PubMed] |
| 218. | Gorlova OY, Weng SF, Zhang Y, Amos CI, Spitz MR, Wei Q. DNA repair capacity and lung cancer risk in never smokers. Cancer Epidemiol Biomarkers Prev. 2008;17:1322-1328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 25] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 219. | Spitz MR, Wei Q, Dong Q, Amos CI, Wu X. Genetic susceptibility to lung cancer: the role of DNA damage and repair. Cancer Epidemiol Biomarkers Prev. 2003;12:689-698. [PubMed] |
| 220. | Spitz MR, Hong WK, Amos CI, Wu X, Schabath MB, Dong Q, Shete S, Etzel CJ. A risk model for prediction of lung cancer. J Natl Cancer Inst. 2007;99:715-726. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 297] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 221. | Spitz MR, Gorlov IP, Amos CI, Dong Q, Chen W, Etzel CJ, Gorlova OY, Chang DW, Pu X, Zhang D. Variants in inflammation genes are implicated in risk of lung cancer in never smokers exposed to second-hand smoke. Cancer Discov. 2011;1:420-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 222. | Gorlova OY, Weng SF, Hernandez L, Spitz MR, Forman MR. Dietary patterns affect lung cancer risk in never smokers. Nutr Cancer. 2011;63:842-849. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 223. | Rylander R, Axelsson G. Lung cancer risks in relation to vegetable and fruit consumption and smoking. Int J Cancer. 2006;118:739-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 224. | Zheng W, Chow WH, Yang G, Jin F, Rothman N, Blair A, Li HL, Wen W, Ji BT, Li Q. The Shanghai Women’s Health Study: rationale, study design, and baseline characteristics. Am J Epidemiol. 2005;162:1123-1131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 319] [Cited by in RCA: 378] [Article Influence: 18.9] [Reference Citation Analysis (0)] |
| 225. | Weiss JM, Lacey JV, Shu XO, Ji BT, Hou L, Yang G, Li H, Rothman N, Blair A, Gao YT. Menstrual and reproductive factors in association with lung cancer in female lifetime nonsmokers. Am J Epidemiol. 2008;168:1319-1325. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 65] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 226. | Pronk A, Coble J, Ji BT, Shu XO, Rothman N, Yang G, Gao YT, Zheng W, Chow WH. Occupational risk of lung cancer among lifetime non-smoking women in Shanghai, China. Occup Environ Med. 2009;66:672-678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 227. | Fowke JH, Gao YT, Chow WH, Cai Q, Shu XO, Li HL, Ji BT, Rothman N, Yang G, Chung FL. Urinary isothiocyanate levels and lung cancer risk among non-smoking women: a prospective investigation. Lung Cancer. 2011;73:18-24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 228. | Yu IT, Chiu YL, Au JS, Wong TW, Tang JL. Dose-response relationship between cooking fumes exposures and lung cancer among Chinese nonsmoking women. Cancer Res. 2006;66:4961-4967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 149] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 229. | Wang XR, Chiu YL, Qiu H, Au JS, Yu IT. The roles of smoking and cooking emissions in lung cancer risk among Chinese women in Hong Kong. Ann Oncol. 2009;20:746-751. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 47] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 230. | Chiu YL, Wang XR, Qiu H, Yu IT. Risk factors for lung cancer: a case-control study in Hong Kong women. Cancer Causes Control. 2010;21:777-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 231. | Hill SE, Blakely T, Kawachi I, Woodward A. Mortality among lifelong nonsmokers exposed to secondhand smoke at home: cohort data and sensitivity analyses. Am J Epidemiol. 2007;165:530-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 232. | López-Cima MF, González-Arriaga P, García-Castro L, Pascual T, Marrón MG, Puente XS, Tardón A. Polymorphisms in XPC, XPD, XRCC1, and XRCC3 DNA repair genes and lung cancer risk in a population of northern Spain. BMC Cancer. 2007;7:162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 116] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 233. | Asomaning K, Miller DP, Liu G, Wain JC, Lynch TJ, Su L, Christiani DC. Second hand smoke, age of exposure and lung cancer risk. Lung Cancer. 2008;61:13-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 73] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 234. | Kurahashi N, Inoue M, Liu Y, Iwasaki M, Sasazuki S, Sobue T, Tsugane S. Passive smoking and lung cancer in Japanese non-smoking women: a prospective study. Int J Cancer. 2008;122:653-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 69] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 235. | Yang P, Wentzlaff KA, Katzmann JA, Marks RS, Allen MS, Lesnick TG, Lindor NM, Myers JL, Wiegert E, Midthun DE. Alpha1-antitrypsin deficiency allele carriers among lung cancer patients. Cancer Epidemiol Biomarkers Prev. 1999;8:461-465. [PubMed] |
| 236. | Yang P, Allen MS, Aubry MC, Wampfler JA, Marks RS, Edell ES, Thibodeau S, Adjei AA, Jett J, Deschamps C. Clinical features of 5,628 primary lung cancer patients: experience at Mayo Clinic from 1997 to 2003. Chest. 2005;128:452-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 308] [Cited by in RCA: 329] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 237. | Zheng YL, Loffredo CA, Yu Z, Jones RT, Krasna MJ, Alberg AJ, Yung R, Perlmutter D, Enewold L, Harris CC. Bleomycin-induced chromosome breaks as a risk marker for lung cancer: a case-control study with population and hospital controls. Carcinogenesis. 2003;24:269-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 238. | Tse LA, Yu IT, Au JS, Yu KS, Kwok KP, Qiu H, Wong TW. Environmental tobacco smoke and lung cancer among Chinese nonsmoking males: might adenocarcinoma be the culprit? Am J Epidemiol. 2009;169:533-541. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 239. | Tse LA, Yu IS, Au JS, Qiu H, Wang XR. Silica dust, diesel exhaust, and painting work are the significant occupational risk factors for lung cancer in nonsmoking Chinese men. Br J Cancer. 2011;104:208-213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 240. | Chuang SC, Gallo V, Michaud D, Overvad K, Tjønneland A, Clavel-Chapelon F, Romieu I, Straif K, Palli D, Pala V. Exposure to environmental tobacco smoke in childhood and incidence of cancer in adulthood in never smokers in the European Prospective Investigation into Cancer and Nutrition. Cancer Causes Control. 2011;22:487-494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 241. | Kiyohara C, Horiuchi T, Takayama K, Nakanishi Y. Methylenetetrahydrofolate reductase polymorphisms and interaction with smoking and alcohol consumption in lung cancer risk: a case-control study in a Japanese population. BMC Cancer. 2011;11:459. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 242. | He Y, Jiang B, Li LS, Li LS, Ko L, Wu L, Sun DL, He SF, Liang BQ, Hu FB. Secondhand smoke exposure predicted COPD and other tobacco-related mortality in a 17-year cohort study in China. Chest. 2012;142:909-918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 243. | Lam TH, He Y, Li LS, Li LS, He SF, Liang BQ. Mortality attributable to cigarette smoking in China. JAMA. 1997;278:1505-1508. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 51] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 244. | Zhao B, Seow A, Lee EJ, Poh WT, Teh M, Eng P, Wang YT, Tan WC, Yu MC, Lee HP. Dietary isothiocyanates, glutathione S-transferase -M1, -T1 polymorphisms and lung cancer risk among Chinese women in Singapore. Cancer Epidemiol Biomarkers Prev. 2001;10:1063-1067. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 142] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 245. | Seow A, Poh WT, Teh M, Eng P, Wang YT, Tan WC, Chia KS, Yu MC, Lee HP. Diet, reproductive factors and lung cancer risk among Chinese women in Singapore: evidence for a protective effect of soy in nonsmokers. Int J Cancer. 2002;97:365-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 86] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 246. | Tang L, Lim WY, Eng P, Leong SS, Lim TK, Ng AW, Tee A, Seow A. Lung cancer in Chinese women: evidence for an interaction between tobacco smoking and exposure to inhalants in the indoor environment. Environ Health Perspect. 2010;118:1257-1260. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 49] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 247. | Ferreccio C, Yuan Y, Calle J, Benítez H, Parra RL, Acevedo J, Smith AH, Liaw J, Steinmaus C. Arsenic, tobacco smoke, and occupation: associations of multiple agents with lung and bladder cancer. Epidemiology. 2013;24:898-905. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 79] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 248. | Lo YL, Hsiao CF, Jou YS, Chang GC, Tsai YH, Su WC, Chen YM, Huang MS, Chen HL, Yang PC. ATM polymorphisms and risk of lung cancer among never smokers. Lung Cancer. 2010;69:148-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 249. | Lo YL, Hsiao CF, Jou YS, Chang GC, Tsai YH, Su WC, Chen KY, Chen YM, Huang MS, Hsieh WS. Polymorphisms of MLH1 and MSH2 genes and the risk of lung cancer among never smokers. Lung Cancer. 2011;72:280-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 250. | Hosseini M, Naghan PA, Karimi S, SeyedAlinaghi S, Bahadori M, Khodadad K, Mohammadi F, Kaynama K, Masjedi MR. Environmental risk factors for lung cancer in Iran: a case-control study. Int J Epidemiol. 2009;38:989-996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 38] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 251. | Ren YW, Yin ZH, Wan Y, Guan P, Wu W, Li XL, Zhou BS. P53 Arg72Pro and MDM2 SNP309 polymorphisms cooperate to increase lung adenocarcinoma risk in Chinese female non-smokers: a case control study. Asian Pac J Cancer Prev. 2013;14:5415-5420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 252. | Seki T, Nishino Y, Tanji F, Maemondo M, Takahashi S, Sato I, Kawai M, Minami Y. Cigarette smoking and lung cancer risk according to histologic type in Japanese men and women. Cancer Sci. 2013;104:1515-1522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 253. | Wang A, Kubo J, Luo J, Desai M, Henderson MT, Chlebowski RT, Tindle H, Chen C, Manson JE, Schwartz AG. Active and passive smoking in relation to lung cancer incidence in the Women’s Health Initiative prospective cohort study. J Clin Oncol. 2013;31 suppl:abstr 1504. |
| 254. | Kabat GC, Kim M, Hunt JR, Chlebowski RT, Rohan TE. Body mass index and waist circumference in relation to lung cancer risk in the Women’s Health Initiative. Am J Epidemiol. 2008;168:158-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 77] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 255. | Torres-Durán M, Ruano-Ravina A, Parente-Lamelas I, Leiro-Fernández V, Abal-Arca J, Montero-Martínez C, Pena-Álvarez C, González-Barcala FJ, Castro-Añón O, Golpe-Gómez A. Lung cancer in never-smokers: a case-control study in a radon-prone area (Galicia, Spain). Eur Respir J. 2014;44:994-1001. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 69] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 256. | Ruano-Ravina A, García-Lavandeira JA, Torres-Durán M, Prini-Guadalupe L, Parente-Lamelas I, Leiro-Fernández V, Montero-Martínez C, González-Barcala FJ, Golpe-Gómez A, Martínez C. Leisure time activities related to carcinogen exposure and lung cancer risk in never smokers. A case-control study. Environ Res. 2014;132:33-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |