Published online Feb 15, 2003. doi: 10.3748/wjg.v9.i2.368
Revised: September 4, 2002
Accepted: September 12, 2002
Published online: February 15, 2003
AIM: To investigate the expression of E-cadherin and alpha-catenin and beta-catenin in pancreatic carcinoma and its relationship with the clinicopathologic characteristics, and clarify the mechanism of invasion and metastasis of pancreatic cancer.
METHODS: The expression of E-cadherin and alpha-, beta-catenin was examined in 47 cases of infiltrative ductal adenocarcinoma of pancreas and 12 adult normal pancreatic tissues by immunohistochemical technique.
RESULTS: The immunoreactivity of E-cadherin and alpha-, beta-catenin was expressed by normal ductal and acinar cells with strong membranous staining at the intercellular border in 12 cases of adult normal pancreatic tissues. Abnormal expression of E-cadherin and alpha-, beta-catenin in 47 pancreatic carcinoma tissues was demonstrated in 53.2%, 61.7% and 68.1%, respectively. Both abnormal expression of E-cadherin and alpha-catenin significantly correlated with differentiation, lymph node and liver metastases (P < 0.05, respectively), whereas aberrant beta-catenin expression only correlated with lymph node and liver metastases (P < 0.001). Abnormal E-cadherin and alpha-, beta-catenin expression was not associated with tumor size, invasion and survival time of patients (P > 0.05, all).
CONCLUSION: Pancreatic cancer likely occurs in case of E-cadherin-catenin complex genes mutations or deletions and abnormal expression of proteins, which significantly correlate with the biologic character of the tumor and lymph node and liver metastases. It is suggested that the abnormal E-cadherin-catenin complex expression plays an important role in the development and progression of tumor, and thus may become a new marker in pancreatic cancer metastasis.
- Citation: Li YJ, Ji XR. Relationship between expression of E-cadherin-catenin complex and clinicopathologic characteristics of pancreatic cancer. World J Gastroenterol 2003; 9(2): 368-372
- URL: https://www.wjgnet.com/1007-9327/full/v9/i2/368.htm
- DOI: https://dx.doi.org/10.3748/wjg.v9.i2.368
Pancreatic cancer is a highly malignant tumor, and is the fifth leading cause of death among the malignant diseases in Western societies, and its incidence has been increasing year by year. However, there is no valid diagnosis and treatment for this disease. Only 15% of patients can undergo resection, and the overall 5-year survival rate is about 10%. One reason why the prognosis of pancreatic cancer is so poor is that many tumors remain silent until a late stage in the disease, and at the time of diagnosis 80%-90% of patients will have extensive invasion and metastasis[1-5]. So studying the mechanism of invasion and metastasis of pancreatic cancer is double valued. A recent study demonstrated that the invasion and metastasis of tumors as a continuous process, include three steps: A reduced cell-cell adhesion, alterations in the interaction of the tumor cell with the extracellular matrix, and invasion into surrounding tissues including blood vessels and lymphduct; thus the first and critical step is that the tumor cells detach from the primary focus and re-adhere to metastatic position[6-8]. E-cadherin is a transmembrane gloycoprotein which maintains normal epithelial polarity and intercellular adhesion[9]. It is almost present in all normal epithelial cell surface, and has been recognized as an important suppression gene[10,11]. Catenins serve not only as associated cytoplasmic proteins of E-cadherin, but also as molecules of intracellular signal transduction[12-15]. They link E-cadherin to the intracellular peptide segment to form E-cadherin-catenin complex, mediating homotypic cell-cell adhesion and playing an essential role in the control of epithelial cell architecture and differentiation[10,11,16]. The abnormal structure and dysfunction of one or several molecules in E-cadherin-catenin complex correlate with tumorigenesis, differentiation, invasion, metastasis and prognosis[17-20]. We detected the expression of E-cadherin and alpha-, beta-catenin in normal pancreas and pancreatic cancer by immunohistochemical technique, and investigated the action of abnormal expression of E-cadherin and alpha-, beta-catenin on invasion and metastasis of pancreatic cancer.
Specimens of 47 pancreatic carcinoma collected between 1995 to 1999 were selected from the Department of Pathology, Affiliated Hospital of Medical College, Qingdao University. These included 29 male and 18 female patients with a mean age of 56.7 years (range 27-75 years). No patient received radiotherapy or chemotherapy before surgery. The tumor size was assessed by CT scan: smaller than 3 cm in 9 patients, 3-5 cm in 15 patients and larger than 5 cm in 23. Histologically, all cases were infiltrative ductal adenocarcinoma. According to the Modified Kloppel Histological Grading System[21], there were 10 in grade I (well differentiated), 16 in grade II (moderately differentiated) and 21 in grade III (poorly differentiated). Based on the tumor being resected successfully or not, we divided 47 pancreatic carcinomas into two groups: non-invasion (17 cases) and invasion (30 cases). There were 19 cases with local lymph node metastasis, 12 cases with liver metastasis and 22 without metastasis. All patients were followed for more than 1 year; 9 patients survived more than 12 mo and 38 patients died within 12 mo. The 1-year survival rate was 19.1%. 12 normal adult pancreatic tissues were obtained from healthy young men who died of traffic accident.
All specimens were fixed in 100 mL·SL-1 neutral buffered formalin for 24-48 h, paraffin-embedded, and cut into 4 um thick sections for immunohistochemical staining. PicTureTM two-step method was used (Zmed, USA). Each step was practised according to the manual. The primary antibodies were E-cadherin monoclonal antibody (ready for use, Zmed, USA) and alpha-, beta-catenin monoclonal antibodies (diluted 1:100, Santa Cruz, USA), respectively. Before staining, the sections were pretreated with microwave (4 min × 4 at 900W) in 0.1 mol·L-1 citrate buffer for antigen retrieval. DAB was used for chromogen. PBS was substituted for primary antibodies as negative control.
The slides were reviewed by two independent observers who had no knowledge of patients' outcome. The positive stainings of E-cadherin and alpha-, beta-catenin appeared in pale brown, and located in the cell membrance. Staining grade standard based on the method described by Jawhari et al[22]. 0 score: no expression; 1 score: Expression in cytoplasm; 2 score: decreased expression; 3 score: Expression at cell membrance, namely normal expression. A score of 0 to 2 was abnormal expression.
Statistical analysis was performed using the χ2 test or Fisher's exact test. P < 0.05 was considered significant.
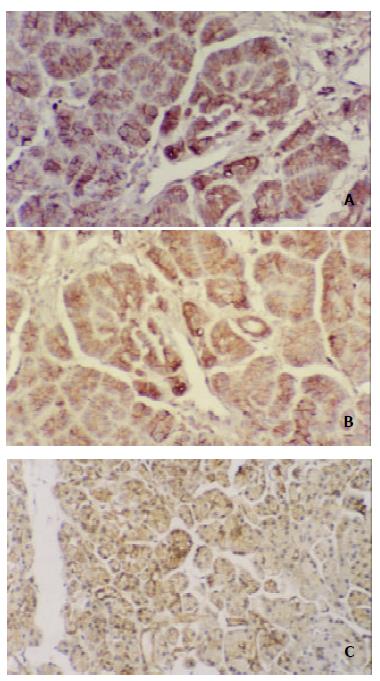
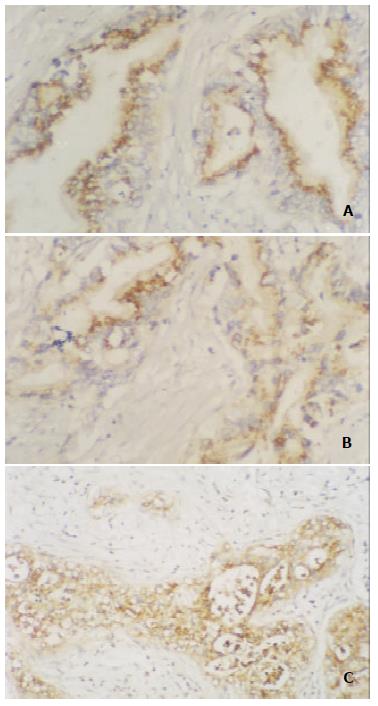
The immunoreactivity of E-cadherin and alpha-, beta-catenin was expressed by normal ductal and acinar cells with strong membranous staining at the intercellular border in 12 cases of adult normal pancreases (Figure 1). The rates of abnormal expression of E-cadherin and alpha-, beta-catenin in 47 pancreatic carcinoma tissue specimens were 53.2% (25/47), 61.7% (29/47) and 68.1% (32/47), respectively. The expression of E-cadherin-catenin complex in most cases was in cytoplasm, whereas membranous staining reduced or disappeared (Figure 2).
The abnormal expression of E-cadherin and alpha-, beta-catenin was not associated with tumor size, invasion and survival time of patients (P = 0.12-0.926, P > 0.05, all). The significant difference of abnormal E-cadherin and alpha-catenin expression was seen between histological grade I, II and II (χ2 = 12.02, 7.74; P = 0.002, 0.021; P < 0.01, 0.05). The tumors with lymph node (χ2 = 4.19, 4.45, 6.60; P = 0.0406, 0.035, 0.0102; P < 0.05) and liver metastases (P = 0.0105, 0.0005, 0.0025; P < 0.05, 0.01) showed a relatively higher frequency of abnormal expression of E-cadherin and alpha-, beta-catenin than those without metastasis (Table 1).
| Parameter | n | E-cadherin (%) | α-catenin (%) | β-catenin (%) | |||
| Normal | Abnormal | Normal | Abnormal | Normal | Abnormal | ||
| Mass size (cm) | |||||||
| < 3 | 9 | 5 (55.6) | 4 (44.4) | 4 (44.4) | 5 (55.6) | 3 (33.3) | 6 (66.7) |
| 3-5 | 15 | 7 (46.7) | 8 (53.3) | 5 (33.3) | 10 (66.7) | 4 (26.7) | 11 (73.3) |
| > 5 | 23 | 10 (43.5) | 13 (56.5) | 9 (39.1) | 14 (60.9) | 8 (34.8) | 15 (65.2) |
| Histologic grade | |||||||
| I | 10 | 9 (90.0) | 1 (10.0) | 7 (70.0) | 3 (30.0) | 5 (50.0) | 5 (50.0) |
| II | 16 | 8 (50.0) | 8 (50.0) | 7 (43.8) | 9 (56.2) | 6 (37.5) | 10 (62.5) |
| III | 21 | 5 (23.8) | 16 (76.2) | 4 (19.0) | 17 (81.0) | 4 (19.0) | 17 (81.0) |
| Invasive state | |||||||
| Invasion | 30 | 12 (40.0) | 18 (60.0) | 9 (30.0) | 21 (70.0) | 8 (26.7) | 22 (73.3) |
| No invasion | 17 | 10 (58.8) | 7 (41.2) | 9 (52.9) | 8 (47.1) | 7 (41.2) | 10 (58.8) |
| Metastases | 19 | 6 (31.6) | 13 (68.4)a | 5 (26.3) | 14 (73.7)a | 3 (15.8) | 16 (84.2)a |
| Lymph node | |||||||
| Liver | 12 | 2 (16.7) | 10 (83.3)a | 0 | 12 (100.0)b | 0 | 12 (100.0)b |
| No | 22 | 14 (63.6) | 8 (36.4) | 13 (59.1) | 9 (40.9) | 12 (54.5) | 10 (45.5) |
| Survival time (month) | |||||||
| > 12 | 9 | 6 (66.7) | 3 (33.3) | 5 (55.6) | 4 (44.4) | 4 (44.4) | 5 (55.6) |
| ≤ 12 | 38 | 16 (42.1) | 22 (57.9) | 13 (34.2) | 25 (65.8) | 11 (28.9) | 27 (71.1) |
E-cadherin-catenin complex is a major mediator of maintaining intercellular adhesion and epithelial integration[16,19], and its down regulation may occur in several ways amongst which are gene mutations or deletions, transcriptional or posttranscriptional alterations, methylation of 5'CpG dinucleotides within the promoter region of E-cadherin, tyrosine phosphorylation of beta-catenin[14,23-26]. The abnormal structure and dysfunction of one or several molecules in E-cadherin-catenin complex correlate with the tumorigenesis, differentiation, invasion, metastasis and prognosis[17-20]. Now the E-cadherin-catenin complex has been regarded as a suppressor of tumor invasion and metastasis, and its reduced expression has been observed in diverse types of human cancer and plays an important role in the process of tumor invasion and metastasis[27-30]. Dai et al[31] found that the positive rates of E-cadherin and alpha-catenin expression were 38.6% and 28.6% respectively and they were significantly reduced in gastric cancer with more prominent malignant phenotype, such as tumor in stage III/IV with poor cell differentiation, invasion through serosa, lymph node and liver metastases, and peritoneal implantation of cancer cells. So they considered that down regulation of E-cadherin and/or alpha-catenin correlates with the degree of malignancy of gastric cancer and that examination of E-cadherin and alpha-catenin expression in gastric cancer helps evaluate the intensity of lymph node metastasis, TNM staging and prognosis. Joo et al[32] reported that the immunoreactivity of E-cadherin and alpha-, beta-, and gamma-catenin was expressed by normal gastric epithelial cells with strong membranous staining at the intercellular border, and reduced expression of them occurred in a considerable proportion of both early and advanced cancer groups and correlated with the diffuse type of cancer with poor differentiation, but it was not related with depth of invasion or lymph node metastases.
Till now, there have been only a few articles about the relationship between expression of E-cadherin-catenin complex and biological behavior of pancreatic cancer, and the results were not consistent. Karayiannakis et al[33,34] observed that in non-cancerous pancreatic cells E-cadherin, alpha-, beta- and gamma-catenin immunoreactivity was localized on the cell membrane, particularly at the intercellular junctions, but in 43 pancreatic carcinomas abnormal E-cadherin, alpha-, beta- and gamma-catenin expressions were found in 42%, 37%, 44% and 40%, respectively; moreover, E-cadherin, alpha-catenin and gamma-catenin expressions correlated with poor differentiation, disease stage and with lymph node and distant metastases, whereas aberrant beta-catenin expression only correlated with the presence of lymph node metastases. Some authors[35-37] also reported that loss of E-cadherin played a causative role in peritoneal dissemination, invasion and metastasis of pancreatic carcinoma, and altered expression of adhesion molecules correlated with dedifferentiating change and aggressive biological behavior of pancreatic cancer. But Gunji et al[38] concluded that reduced E-cadherin and alpha-catenin expression in primary pancreatic cancer had no significant predictive value regarding the presence of liver metastasis.
In this study, we found that the immunoreactivity of E-cadherin and alpha-, beta-catenin was expressed by normal ductal and acinar cells with strong membranous staining at the intercellular border in 12 cases of adult normal pancreases, but over 50% pancreatic carcinomas showed abnormal expression of E-cadherin (53.2%) and alpha-, beta-catenin (61.7%, 68.1%), and the three adhesion molecules in most cases were present in cytoplasm, whereas membranous staining was reduced or disappeared. The results pointed out that there was significant difference in protein level of E-cadherin-catenin complex expression between normal pancreas and pancreatic carcinoma tissues, and E-cadherin and alpha-, beta-catenin genes mutations or deletions likely were related with tumorigenesis of pancreas. Our data also demonstrated that both abnormal expression of E-cadherin and alpha-catenin significantly correlated with differentiation, lymph node and liver metastases (P < 0.05, 0.01, respectively), whereas aberrant beta-catenin expression only correlated with lymph node and liver metastases (P < 0.01), and all 12 cases with liver metastasis showed abnormal expression of alpha-catenin and beta-catenin. It can be inferred from our findings that the changes of E-cadherin-catenin complex genes happen in early pancreatic carcinoma, and become more serious with the progression of tumor. Moreover, because of the gene inactivation and dysfunction of E-cadherin-catenin complex, carcinoma cells are “set free” for invasion and metastases. Therefore, these results suggest that abnormal change of E-cadherin and alpha-, beta-catenin plays a conjoint role in the development and progression of tumor, and combined examination of E-cadherin-catenin complex expression has very important clinical value for evaluating the dedifferentiation, lymph node and liver metastases of pancreatic cancer.
Some authors reported[39-42] that the level of E-cadherin-catenin complex expression could be regarded as a prognostic marker of tumors. Karayiannakis et al[33] also regarded that all three catenins and E-cadherin were associated with a poor prognosis of pancreatic cancer, but only E-cadherin and alpha-catenin were independent prognostic factors for cancer-specific survival. However, in this study our data showed the expression of E-cadherin and alpha-, beta-catenin had no significant relationship with survival time of patients (P > 0.05), and was not associated with invasion and size of pancreatic cancer as well (P > 0.05, all). Perhaps this was related to the criteria of invasion adopted in our research and different operative method and different postoperative therapy. All these factors can bring about influence on our results. A similar conclusion was reported in non-small cell lung cancer by Pirinen et al[43].
In summary, our findings suggested that the change of E-catenin complex genes and down regulation and abnormal distribution of their encoding proteins happened in the early phase of pancreatic carcinoma and finally the tumor completely lost E-cadherin and alpha-, beta-catenin, resulting in a loss of adhesive mechanism in maintaining intercellular attachment, a dedifferentiated state and an increased metastatic ability. This is a key link of metastatic mechanism of the late pancreatic cancer.
Edited by Lu HM
| 1. | Wagman R, Grann A. Adjuvant therapy for pancreatic cancer: current treatment approaches and future challenges. Surg Clin North Am. 2001;81:667-681. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 11] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 2. | Gebhardt C, Meyer W, Reichel M, Wünsch PH. Prognostic factors in the operative treatment of ductal pancreatic carcinoma. Langenbecks Arch Surg. 2000;385:14-20. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 70] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 3. | Kayahara M, Nagakawa T, Ohta T, Kitagawa H, Ueno K, Tajima H, Elnemr A, Miwa K. Analysis of paraaortic lymph node involvement in pancreatic carcinoma: A significant indication for surgery. Cancer. 1999;85:583-590. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 3] [Reference Citation Analysis (0)] |
| 4. | Carr JA, Ajlouni M, Wollner I, Wong D, Velanovich V. Adenocarcinoma of the head of the pancreas: Effects of surgical and nonsurgical therapy on survival--a ten-year experience. Am Surg. 1999;65:1143-1149. [PubMed] [Cited in This Article: ] |
| 5. | Benassai G, Mastrorilli M, Mosella F, Mosella G. Significance of lymph node metastases in the surgical management of pancreatic head carcinoma. J Exp Clin Cancer Res. 1999;18:23-28. [PubMed] [Cited in This Article: ] |
| 6. | Beavon IR. The E-cadherin-catenin complex in tumour metastasis: structure, function and regulation. Eur J Cancer. 2000;36:1607-1620. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 259] [Cited by in F6Publishing: 262] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 7. | Korn WM. Moving toward an understanding of the metastatic process in hepatocellular carcinoma. World J Gastroenterol. 2001;7:777-778. [PubMed] [Cited in This Article: ] |
| 8. | Stamenkovic I. Matrix metalloproteinases in tumor invasion and metastasis. Semin Cancer Biol. 2000;10:415-433. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 542] [Cited by in F6Publishing: 547] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 9. | Bair EL, Massey CP, Tran NL, Borchers AH, Heimark RL, Cress AE, Bowden GT. Integrin- and cadherin-mediated induction of the matrix metalloprotease matrilysin in cocultures of malignant oral squamous cell carcinoma cells and dermal fibroblasts. Exp Cell Res. 2001;270:259-267. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 24] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Hirohashi S. Inactivation of the E-cadherin-mediated cell adhesion system in human cancers. Am J Pathol. 1998;153:333-339. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 620] [Cited by in F6Publishing: 653] [Article Influence: 25.1] [Reference Citation Analysis (0)] |
| 11. | Christofori G, Semb H. The role of the cell-adhesion molecule E-cadherin as a tumour-suppressor gene. Trends Biochem Sci. 1999;24:73-76. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 525] [Cited by in F6Publishing: 531] [Article Influence: 21.2] [Reference Citation Analysis (0)] |
| 12. | Van Aken E, De Wever O, Correia da Rocha AS, Mareel M. Defective E-cadherin/catenin complexes in human cancer. Virchows Arch. 2001;439:725-751. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 177] [Cited by in F6Publishing: 188] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 13. | Wijnhoven BP, Dinjens WN, Pignatelli M. E-cadherin-catenin cell-cell adhesion complex and human cancer. Br J Surg. 2000;87:992-1005. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 313] [Cited by in F6Publishing: 317] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 14. | Debruyne P, Vermeulen S, Mareel M. The role of the E-cadherin/catenin complex in gastrointestinal cancer. Acta Gastroenterol Belg. 1999;62:393-402. [PubMed] [Cited in This Article: ] |
| 15. | Behrens J. Cadherins and catenins: role in signal transduction and tumor progression. Cancer Metastasis Rev. 1999;18:15-30. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 209] [Cited by in F6Publishing: 205] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 16. | El-Bahrawy MA, Pignatelli M. E-cadherin and catenins: molecules with versatile roles in normal and neoplastic epithelial cell biology. Microsc Res Tech. 1998;43:224-232. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 2] [Reference Citation Analysis (0)] |
| 17. | Bremnes RM, Veve R, Hirsch FR, Franklin WA. The E-cadherin cell-cell adhesion complex and lung cancer invasion, metastasis, and prognosis. Lung Cancer. 2002;36:115-124. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 163] [Cited by in F6Publishing: 165] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 18. | Berx G, Van Roy F. The E-cadherin/catenin complex: An important gatekeeper in breast cancer tumorigenesis and malignant progression. Breast Cancer Res. 2001;3:289-293. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 305] [Cited by in F6Publishing: 287] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 19. | Nollet F, Berx G, van Roy F. The role of the E-cadherin/catenin adhesion complex in the development and progression of cancer. Mol Cell Biol Res Commun. 1999;2:77-85. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 144] [Cited by in F6Publishing: 142] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 20. | Kitagawa T, Matsumoto K, Nagafuchi A, Tsukita S, Suzuki H. Co-expression of E-cadherin and alpha-catenin molecules in colorectal cancer. Surg Today. 1999;29:511-518. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 21. | Giulianotti PC, Boggi U, Fornaciari G, Bruno J, Rossi G, Giardino D, Di Candio G, Mosca F. Prognostic value of histological grading in ductal adenocarcinoma of the pancreas. Klöppel versus TNM grading. Int J Pancreatol. 1995;17:279-289. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 21] [Cited by in F6Publishing: 14] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 22. | Jawhari A, Jordan S, Poole S, Browne P, Pignatelli M, Farthing MJ. Abnormal immunoreactivity of the E-cadherin-catenin complex in gastric carcinoma: relationship with patient survival. Gastroenterology. 1997;112:46-54. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 161] [Cited by in F6Publishing: 171] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 23. | Hirohashi S. Molecular aspects of adhesion-epigenetic mechanisms for inactivation of the E-Cadherin-mediated cell adhesion system in cancers. Verh Dtsch Ges Pathol. 2000;84:28-32. [PubMed] [Cited in This Article: ] |
| 24. | Maeno Y, Moroi S, Nagashima H, Noda T, Shiozaki H, Monden M, Tsukita S, Nagafuchi A. alpha-catenin-deficient F9 cells differentiate into signet ring cells. Am J Pathol. 1999;154:1323-1328. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 32] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 25. | Chalmers IJ, Höfler H, Atkinson MJ. Mapping of a cadherin gene cluster to a region of chromosome 5 subject to frequent allelic loss in carcinoma. Genomics. 1999;57:160-163. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 26. | Li LC, Chui RM, Sasaki M, Nakajima K, Perinchery G, Au HC, Nojima D, Carroll P, Dahiya R. A single nucleotide polymorphism in the E-cadherin gene promoter alters transcriptional activities. Cancer Res. 2000;60:873-876. [PubMed] [Cited in This Article: ] |
| 27. | Maruyama K, Ochiai A, Nakamura S, Baba S, Hirohashi S. [Dysfunction of E-cadherin-catenin system in invasion and metastasis of colorectal cancer]. Nihon Geka Gakkai Zasshi. 1998;99:402-408. [PubMed] [Cited in This Article: ] |
| 28. | Mareel M, Van Roy F. [The human E-cadherin/catenin complex: A potent invasion and tumor suppressor]. Verh K Acad Geneeskd Belg. 1998;60:567-598. [PubMed] [Cited in This Article: ] |
| 29. | Tanaka M, Kitajima Y, Edakuni G, Sato S, Miyazaki K. Abnormal expression of E-cadherin and beta-catenin may be a molecular marker of submucosal invasion and lymph node metastasis in early gastric cancer. Br J Surg. 2002;89:236-244. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 43] [Cited by in F6Publishing: 46] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 30. | Fujioka T, Takebayashi Y, Kihana T, Kusanagi Y, Hamada K, Ochi H, Uchida T, Fukumoto M, Ito M. Expression of E-cadherin and beta-catenin in primary and peritoneal metastatic ovarian carcinoma. Oncol Rep. 2001;8:249-255. [PubMed] [Cited in This Article: ] |
| 31. | Dai D, Chen J, Xu H. [The clinical significance of E-cadherin and alpha-catenin expression in human gastric cancer]. Zhonghua Zhongliu Zazhi. 2001;23:35-38. [PubMed] [Cited in This Article: ] |
| 32. | Joo YE, Rew JS, Kim HS, Choi SH, Park CS, Kim SJ. Changes in the E-cadherin-catenin complex expression in early and advanced gastric cancers. Digestion. 2001;64:111-119. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 18] [Cited by in F6Publishing: 19] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 33. | Karayiannakis AJ, Syrigos KN, Polychronidis A, Simopoulos C. Expression patterns of alpha-, beta- and gamma-catenin in pancreatic cancer: correlation with E-cadherin expression, pathological features and prognosis. Anticancer Res. 2001;21:4127-4134. [PubMed] [Cited in This Article: ] |
| 34. | Karayiannakis AJ, Syrigos KN, Chatzigianni E, Papanikolaou S, Alexiou D, Kalahanis N, Rosenberg T, Bastounis E. Aberrant E-cadherin expression associated with loss of differentiation and advanced stage in human pancreatic cancer. Anticancer Res. 1998;18:4177-4180. [PubMed] [Cited in This Article: ] |
| 35. | Furuyama H, Arii S, Mori A, Imamura M. Role of E-cadherin in peritoneal dissemination of the pancreatic cancer cell line, panc-1, through regulation of cell to cell contact. Cancer Lett. 2000;157:201-209. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 12] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 36. | Yonemasu H, Takashima M, Nishiyama KI, Ueki T, Yao T, Tanaka M, Tsuneyoshi M. Phenotypical characteristics of undifferentiated carcinoma of the pancreas: A comparison with pancreatic ductal adenocarcinoma and relevance of E-cadherin, alpha catenin and beta catenin expression. Oncol Rep. 2001;8:745-752. [PubMed] [Cited in This Article: ] |
| 37. | Takao S, Che X, Fukudome T, Natsugoe S, Ozawa M, Aikou T. Down-regulation of E-cadherin by antisense oligonucleotide enhances basement membrane invasion of pancreatic carcinoma cells. Hum Cell. 2000;13:15-21. [PubMed] [Cited in This Article: ] |
| 38. | Gunji N, Oda T, Todoroki T, Kanazawa N, Kawamoto T, Yuzawa K, Scarpa A, Fukao K. Pancreatic carcinoma: correlation between E-cadherin and alpha-catenin expression status and liver metastasis. Cancer. 1998;82:1649-1656. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 1] [Reference Citation Analysis (0)] |
| 39. | Aaltomaa S, Lipponen P, Ala-Opas M, Eskelinen M, Kosma VM. Alpha-catenin expression has prognostic value in local and locally advanced prostate cancer. Br J Cancer. 1999;80:477-482. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 43] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 40. | Inagawa S, Itabashi M, Adachi S, Kawamoto T, Hori M, Shimazaki J, Yoshimi F, Fukao K. Expression and prognostic roles of beta-catenin in hepatocellular carcinoma: correlation with tumor progression and postoperative survival. Clin Cancer Res. 2002;8:450-456. [PubMed] [Cited in This Article: ] |
| 41. | Bremnes RM, Veve R, Gabrielson E, Hirsch FR, Baron A, Bemis L, Gemmill RM, Drabkin HA, Franklin WA. High-throughput tissue microarray analysis used to evaluate biology and prognostic significance of the E-cadherin pathway in non-small-cell lung cancer. J Clin Oncol. 2002;20:2417-2428. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 230] [Cited by in F6Publishing: 237] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 42. | Hugh TJ, Dillon SA, Taylor BA, Pignatelli M, Poston GJ, Kinsella AR. Cadherin-catenin expression in primary colorectal cancer: A survival analysis. Br J Cancer. 1999;80:1046-1051. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 85] [Cited by in F6Publishing: 92] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 43. | Pirinen RT, Hirvikoski P, Johansson RT, Hollmén S, Kosma VM. Reduced expression of alpha-catenin, beta-catenin, and gamma-catenin is associated with high cell proliferative activity and poor differentiation in non-small cell lung cancer. J Clin Pathol. 2001;54:391-395. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 76] [Article Influence: 3.3] [Reference Citation Analysis (0)] |