Copyright
©The Author(s) 2021.
World J Gastroenterol. Nov 7, 2021; 27(41): 7190-7206
Published online Nov 7, 2021. doi: 10.3748/wjg.v27.i41.7190
Published online Nov 7, 2021. doi: 10.3748/wjg.v27.i41.7190
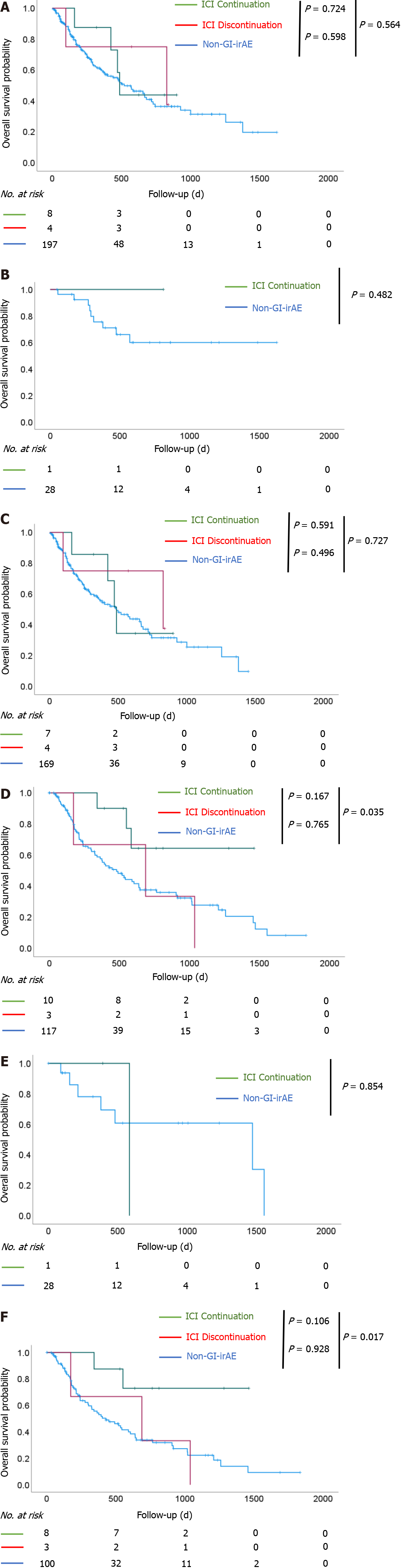
Figure 3 Overall survival after initiation of immune checkpoint inhibitor treatment.
Overall survival (OS) of patients with non-small cell lung cancer (NSCLC) and OS of patients with malignant melanoma (MM). A: In patients with NSCLC, there was no significant difference in OS among the three groups; B: The results were similar when stratified in stage III; C: The results were similar when stratified in stage IV; D: In patients with MM, there was a significant prolongation of OS in the immune checkpoint inhibitor (ICI) continuation group compared to the non-gastrointestinal immune-related adverse event (non-GI-irAE) group (P = 0.035); E: There was no significant difference in OS between the ICI continuation group and the non-GI-irAE group among patients with stage III disease; F: Among patients with stage IV disease, there was a significant prolongation of OS in the ICI continuation group compared to the non-GI-irAE group (P = 0.017). ICI continuation group: Patients who continued ICI treatment after developing GI-irAEs; ICI discontinuation group: Patients who discontinued ICI treatment after developing GI-irAEs; non-GI-irAE group: Patients with no GI-irAEs.
- Citation: Yamada K, Sawada T, Nakamura M, Yamamura T, Maeda K, Ishikawa E, Iida T, Mizutani Y, Kakushima N, Ishikawa T, Furukawa K, Ohno E, Honda T, Kawashima H, Ishigami M, Furune S, Hase T, Yokota K, Maeda O, Hashimoto N, Akiyama M, Ando Y, Fujishiro M. Clinical characteristics of gastrointestinal immune-related adverse events of immune checkpoint inhibitors and their association with survival. World J Gastroenterol 2021; 27(41): 7190-7206
- URL: https://www.wjgnet.com/1007-9327/full/v27/i41/7190.htm
- DOI: https://dx.doi.org/10.3748/wjg.v27.i41.7190