INTRODUCTION
Endoscopic ultrasound (EUS) represents a valuable addition to imaging modalities in digestive diseases, fostering a wide range of diagnostic and therapeutic applications for gastrointestinal (GI) and pancreatico-biliary diseases. Provided with superior resolution compared to other cross-sectional imaging techniques and the added possibility to perform fine-needle aspiration for pathological confirmation, EUS may trigger changes to both patients’ diagnosis and management, displaying a considerable impact upon clinical decision[1]. Several additional techniques have been developed in recent years for enhanced imaging with EUS, including contrast enhancement, elastography, and three-dimensional reconstructions. Such techniques can provide a better characterisation of lesions and improve diagnostic accuracy while possibly diminishing the operator dependency of EUS[2]. Under the circumstance, a series of therapeutic applications have emerged for EUS, some already established, such as drainage of a variety of extraluminal fluid collections, celiac plexus neurolysis, and other rather experimental indications[3]. Hence, EUS is improving steadily as a result of both technical developments and the ever growing interest on behalf of GI endoscopists, who are continuously searching for novel applications.
For the lower GI tract, EUS has played a noteworthy role in staging rectal cancer, representing a key imaging technique for the pretherapeutic evaluation of these patients. However, in the light of recent developments and image-enhancement techniques, other EUS indications have been investigated as well. Consequently, the purpose of the present review is to highlight recent evidence for the application of EUS in evaluating colorectal cancer (CRC), as well as other possible indications for colorectal diseases, such as in the diagnosis and follow-up of inflammatory bowel disease (IBD) patients and in guiding therapeutic procedures.
EUS FOR IMAGING COLORECTAL CANCER
EUS is an already established imaging technique used for the initial evaluation of rectal cancer patients, being considered a fast, well tolerated procedure that enables accurate local staging[4]. The therapeutic strategy is defined for each patient based on accurate assessment of the disease local extent into the rectal wall and the surrounding structures (T stage), lymph nodes (N stage), location and possible involvement of the mesorectal fascia[5]. EUS can assess the depth of tumour penetration with accuracies ranging between 70% and 95%, performing best in the diagnosis of early lesions and in the hands of experienced examiners. With its high resolution that enables reliable demarcations between histological layers of the rectal wall, EUS has been recommended as a highly accurate method for the selection of early rectal lesions, appropriate for endoscopic resection or transanal endoscopic microsurgery, an alternative to conventional surgery. Current research data has confirmed previous results showing a 90% accuracy of EUS in identifying early rectal tumours. However, despite its excellent performance, EUS hardly seems to make a change in the management of patients, especially when clinical features and other imaging findings from computed tomography (CT) or magnetic resonance imaging (MRI) examination are combined before therapy[6].
Results have proven less favourable for EUS N staging, with reported accuracies of 70% to 75%. Nevertheless, evaluation of lymph node involvement has been challenging so far by means of other imaging methods as well, for it is an area with yet unmet needs in rectal cancer staging[4]. The performance of EUS-guided fine needle aspiration (EUS-FNA) was thought to bring additional benefits by providing samples for cytological confirmation of metastases, and thus increasing N staging accuracy. Recent studies showing good diagnostic accuracy of EUS-FNA in staging CRC and assessing nodal involvement were retrospective, included only a limited number of patients[7,8] and although specificity was as high as 100% in predicting nodal metastases, negative predictive value was moderate (77%)[8], meaning that lymphatic metastases cannot be ruled out by a negative FNA, which could result from sampling errors. Therefore, whether the performing of EUS-FNA has a significant impact on the initial management of rectal cancer patients or not, as compared to EUS alone, is still debatable. EUS-FNA seems however justified for perirectal lesions, in patients with previous cancer history, for whom it can confirm or rule out recurrence[9]. Perirectal recurrences can be diagnosed by EUS-FNA and cytological examination with 97% sensitivity, 100% specificity, 100% positive predictive value, 94% negative predictive value, and an overall accuracy of 98%, as recently reported[10].
One of the disadvantages displayed by EUS in the evaluation of rectal tumours, due to its limited in-depth imaging, has been the low accuracy degree in assessing the involvement of the mesorectal fascia, an important factor to predict local recurrence and which is better evaluated by MRI[11]. Findings postulated by a recent research study put forward that for low anterior rectal tumours, EUS can predict the involvement of the mesorectal fascia with an increased accuracy and a high negative predictive value, thus performing similarly to MRI[12].
Staging beyond rectal cancer
In staging cancers beyond the rectum, cross-sectional imaging with CT is widely used, for as it can provide reliable information regarding the distal spread, though it does not ensure an ideal accuracy for local staging[13]. While surgery was previously considered the golden standard in treating CRC patients, nowadays an adequate therapy involves a multidisciplinary plan set up by oncologists, gastroenterologists, surgeons. Consequently, the number of primary tumour resections for CRC patients has decreased, while survival rates have improved ever since the introduction of new chemotherapeutic regimens and biologic agents[14]. Therefore imaging techniques improvement is also required for a better evaluation of patients, especially since CRC is one of the most frequent malignancies worldwide.
EUS examination of the colon was previously attempted with a forward-oblique-viewing echoendoscope, but its design enabled only the imaging of the sigmoid and of the left colon[15]. Mini-probe EUS has also been used for colonic tumours and based on its reduced diameter, which enables passage though the working channel of regular colonoscopes, it can evaluate lesions throughout the entire colon. Although mini-probe EUS can accurately stage earlier tumours, its performance is hampered by the limited in-depth acoustic penetration resulting in less accurate staging of larger and more invasive cancers[16,17].
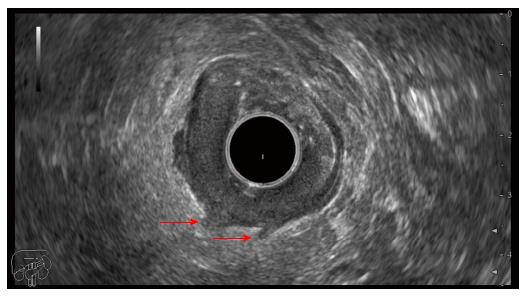
One of the improvements in EUS technology was the development of the forward-viewing radial echoendoscope which was recently tested and proved to be feasible for staging CRC beyond the rectum. The forward-viewing echoendoscope was able to safely reach all colonic lesions and within time frames similar to standard colonoscopy procedures[18]. Although the accuracy was 81.0% for tumour staging and only 52.4% for lymph node staging, overall, EUS was more accurate compared to CT (81.0% vs 68.4%) and these results can undoubtedly be improved through further studies and an ever-increasing experience in assessing peri-colonic anatomy. Within our institute we have also performed EUS examination by means of a forward-viewing radial-array echoendoscope for staging beyond the rectum, which proved to be feasible and accurate (Figure 1). The design of the new echoendoscope makes both diagnosis, by colonoscopy with biopsy, and staging of the local extension possible during the same procedure, and it can be used for the preoperative evaluation of CRC patients.
Figure 1 Endoscopic ultrasonography image in a T3 sigmoid cancer showing hypoechoic infiltration beyond the muscularis propria (arrows).
EUS image enhancement techniques for colorectal cancer evaluation
Contrast-enhanced EUS: The use of intravenous ultrasound contrast agents has been a major development for the practice of ultrasonography, enabling better characterisation of lesions based on their vascular enhancement. Furthermore, perfusion quantification is possible with additional post-processing software applications that enable objective measurements of several parameters, being thus useful in monitoring early changes of vascularization following the anti-angiogenic treatment[19]. Consequently, further clinical studies have been conducted proving that contrast-enhanced ultrasonography (CE-US) can assess early response for several tumour sites, including hepatocellular carcinoma, colorectal metastases and renal carcinoma[20]. Potential applications are also expanding for contrast-enhanced EUS (CE-EUS). However, for colorectal diseases, there is only limited information on the role of CE-US and initial published data on CRC was the result of transabdominal examination[21-23]. Zhuang et al[22] put forward the possibility of using CE-US in quantifying colorectal tumour angiogenesis based on time-intensity curve (TIC) analysis. One of the computed parameters, i.e. AUC (area under the curve), as an indirect indicator of blood volume, was significantly higher in adenocarcinomas compared to adenomas, and was also positively correlated with MVD, thus suggesting possible prognostic value for colorectal tumours[22].
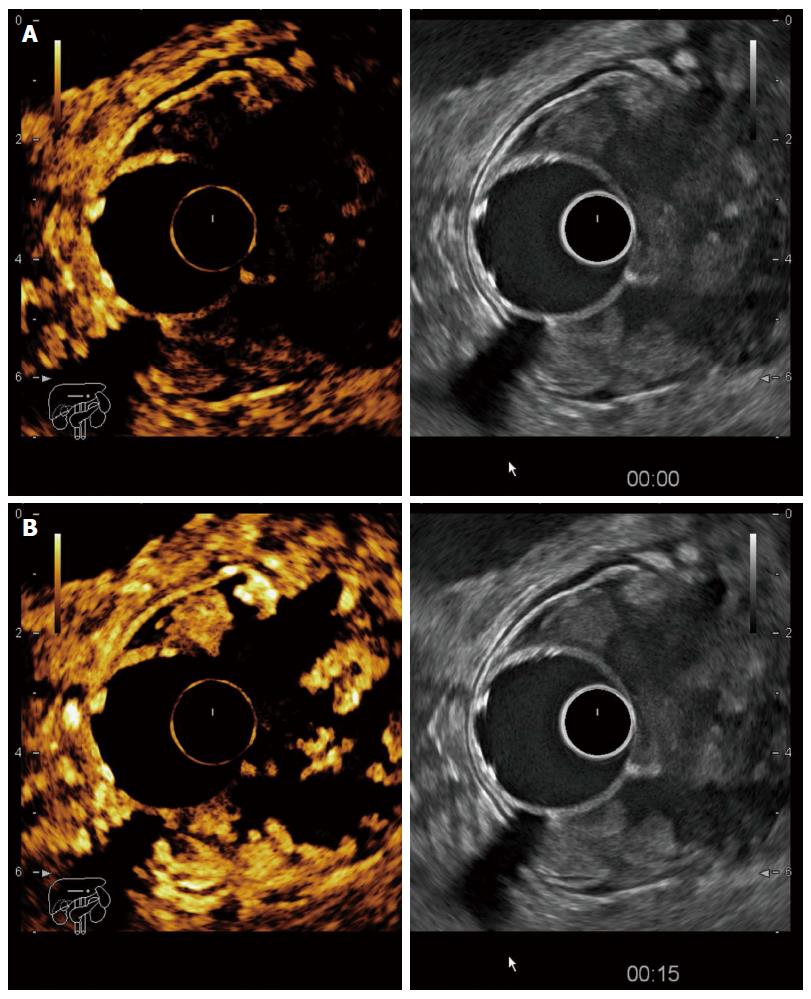
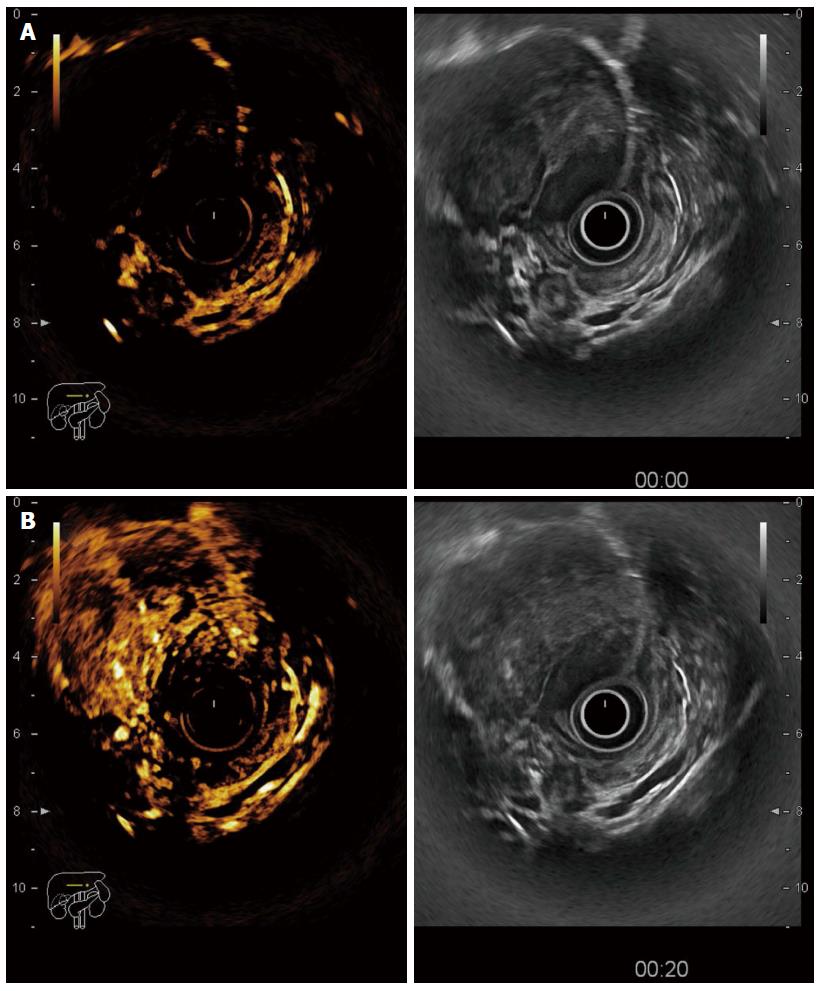
Although EUS exhibits the advantage of an increased imaging resolution, as it has direct access for the examination of GI lesions, avoiding artefacts associated with transabdominal ultrasound, only one study was so far published on the possibility to apply CE-EUS in rectal cancer[24]. CE-EUS examination of rectal tumours was performed by using a rigid probe and TIC analysis was used to determine several parameters of perfusion, while searching for correlations with MVD and other clinical and pathologic prognostic factors. Only a weak positive correlation was validated between the enhanced intensity parameter and the MVD (r = 0.295, P = 0.016). The same TIC parameter was inversely correlated with the histologic differentiation grade (r = -0.264, P = 0.007). However, these are only initial studies that suggest the possibility to use CE-US as a safe and a more convenient imaging technique for dynamic measurements of tumour angiogenesis in CRC and are likely to be followed by further validation research (Figure 2).
Figure 2 Contrast-enhanced endoscopic ultrasonography in a T3 tumour of the recto-sigmoid junction.
A: Before contrast arrival (left side contrast harmonic imaging mode, right side B mode); B: Maximal enhancement of the tumour 15 s after contrast injection with hyperenhanced areas alternating with avascular (necrotic) areas.
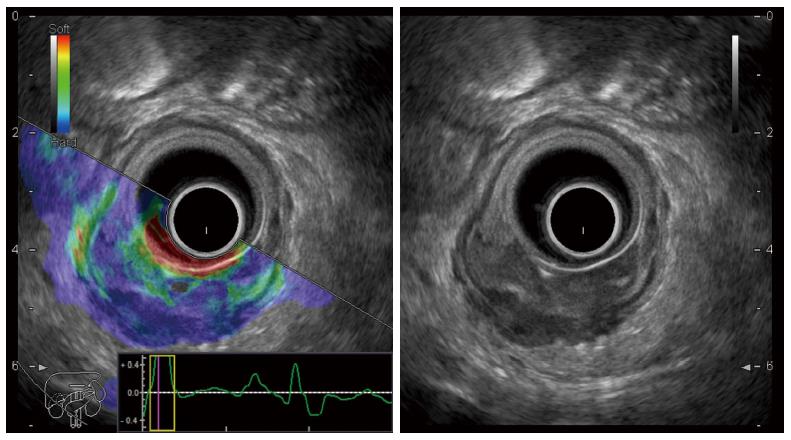
EUS elastography: EUS elastography displays the differences between tissues hardness by adding a colour overlay coding for different elasticity values to the conventional grey-scale images. Tissue strain is measured based on an integrated software application that analyses backscattered ultrasound signals and thus it is possible to evaluate elastic properties of tissues, a feature that can better characterize and differentiate benign from malignant tumours[2]. Quantitative characterization of tissue hardness is also possible. Hue histogram analysis implies a graphical representation of the colours distribution in a region of interest and the establishing of the histogram average value as a measure of the lesion hardness. Strain ratio calculation reports the elastographic properties of the target lesion to a selected area from the surrounding normal structures[25]. For GI imaging, elastography seems more technically demanding, and especially for the normal bowel, as it is impaired by its thin wall and peristaltic movements[26]. Studies have therefore been limited. For colorectal tumours, the main indication, so far, has been for the differentiation of benign from malignant lesions (Figure 3). Transrectal elastography was used in the evaluation of 69 patients with rectal tumours enabling a distinction between adenocarcinomas and adenomas of high accuracy (0.94), based on a strain-ratio (SR) cut-off value of 1.25[27]. A validation study on 120 patients followed which reproduced the same high accuracy when using the initially proposed SR cut-off value to differentiate benign from malignant tumours. Furthermore, the use of SR calculation proved to be more accurate compared to both EUS and MRI examination[28]. It seems that EUS elastography can improve staging of rectal adenomas and early cancers compared to EUS alone and may thus enable more accurate selection of patients suitable for local resection[29]. Visual evaluation of EUS elastography sequences was highly reproducible between blinded observers with reference to differentiating benign and malignant rectal tumours[30].
Figure 3 Endoscopic ultrasonography elastography image of a rectal adenocarcinoma with a predominantly blue pattern indicating a low strain mass (left side real-time sono-elastography mode, right side B mode).
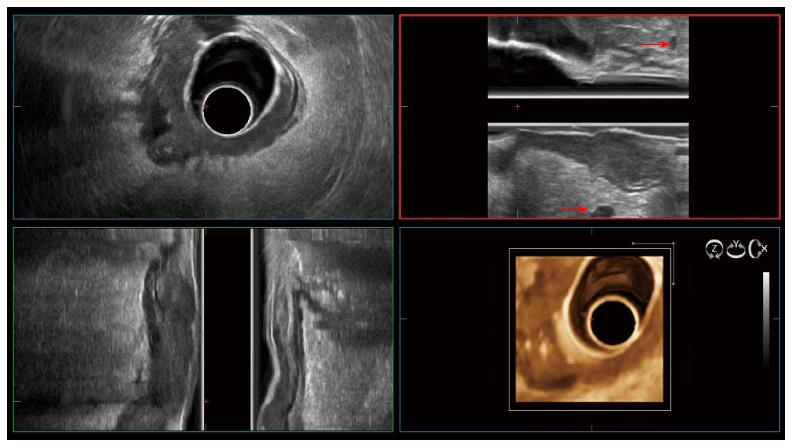
Three-dimensional EUS: Three-dimensional (3D) reconstructions have increased imaging resolution for EUS, allowing multi-planar display of rectal and perirectal anatomy[11] and consequently have improved staging accuracy for both T and N evaluation compared to conventional EUS and CT[31]. Recently published data show excellent results for 3D-EUS which was able to correctly classify T1 tumours in 97.1% of cases, T2 in 94.3%, T3 in 95.7%, and T4 in 98.5% of cases, with postoperative morphologic evaluation used as control. Favourable results were also obtained in determining lymph nodes metastatic involvement with a 87.3% diagnostic accuracy[32]. Such accurate volumetric measurements can be used to assess tumour response following radiation and chemotherapy[33] and possibly to enhance the diagnostic performance of EUS in this setting. Subsequently, the role of 3D-EUS should be further evaluated for the management of CRC patients as it can compensate for some of the limitations imposed by conventional EUS and strengthen the diagnostic accuracy (Figure 4).
Figure 4 Three-dimensional endoscopic ultrasonography in a T3 rectal cancer with peritumoral lymph nodes (red arrows).
EUS IN INFLAMMATORY BOWEL DISEASES
In IBD patients the main indication of EUS has been for the evaluation of perianal and perirectal complications of Crohn’s disease. Fistulas are usually complex in these patients, with a high recurrence rate, thus accurate anatomic assessment is essential in planning the most appropriate surgical therapy in order to prevent faecal incontinence and impairment of patients’ quality of life. For this particular indication transrectal EUS has proved to be superior to other examination methods including CT, fistulography, and is comparable to MRI examination[34]. A recent meta-analysis that compared the performance of MRI and endoanal ultrasound for the examination of perianal fistulas indicated similar sensitivities (0.87), though poor specificity for both techniques (0.69 for MRI vs 0.43 for endoanal US)[35]. However, the studies included in this meta-analysis proved a high degree of heterogeneity. In contrast to pelvic MRI examination, transrectal EUS can also visualize the rectal mucosa and asses the severity of inflammation in predicting the outcome[36]. In order to enhance EUS examination, hydrogen peroxide can be used, which enables a significantly increased accuracy in the identification of the fistulous main tract and its ramifications, as well as classification in relation to the sphincteric apparatus[37,38]. 3D-EUS provides more accurate information on the fistulous tract in relation to anorectal anatomy, by indicating also longitudinal and sagittal imaging planes[38].
EUS examination is well tolerated by patients, safer, and less expensive compared to MRI, and therefore can be easily repeated and used to follow-up on therapeutic response[39]. In a small prospective study including Crohn’s patients with perianal fistulas, EUS performed during combined medical and surgical treatment was associated with better outcomes, reducing the need for additional surgery[40].
Novel technologies for enhanced ultrasound imaging have also been applied to the evaluation of IBD, but mainly during transabdominal examination which, as a non-invasive and low-cost imaging procedure, has been increasingly used for the diagnosis and follow-up of patients. CE-US can be used to assess disease activity based on quantification of bowel wall vascularization and to monitor changes in vascularity following an anti-inflammatory therapy[41]. Other potential applications include differentiation between inflammatory and fibrotic strictures and the characterization of inflammatory masses in Crohn’s disease[42]. The possibility of using strain elastography for discriminating active inflammatory stenosis from fibrotic ones has also been tested[43]. Moreover, a pilot study using transrectal EUS, also investigated the role of elastography in classifying IBD phenotypes. The study included 30 patients with Crohn’s disease, 25 with ulcerative colitis and 28 controls and found a significantly higher SR in Crohn’s disease compared to both ulcerative colitis patients and controls. Although the rectal wall thickness was different between ulcerative colitis patients and controls, there was no difference in the calculated SR[44]. Admittedly, the use of EUS and additional technologies for IBD patients is still a field that needs further investigation with possible translation of transabdominal ultrasound findings or delineation of novel potential indications that could impact upon patient management.
EUS FOR COLORECTAL SUBMUCOSAL LESIONS
Subepithelial lesions that may be found during colonoscopy encompass a broad spectrum of intramural lesions and extrinsic compressions. EUS represents an ideal investigation method to provide an accurate classification of these lesions throughout the entire colon by using either flexible mini-probes[45] or the more recently introduced forward-viewing radial echoendoscope as previously described. Features identified during EUS, such as the originating wall layer and echostructure are indicative of the nature of the lesion and cytological or histological confirmation may be provided by performing EUS-FNA or EUS-Trucut biopsy (EUS-TCB)[46]. EUS can also be used to safely guide endoscopic resection of selected neuroendocrine tumours[47,48].
Differentiating between benign and malignant lesions, especially for gastrointestinal stromal tumours (GIST) is particularly important. The diagnostic value of EUS-FNA for this purpose has been moderate and EUS-TCB does not seem to be superior, as immunostaining and mitotic counts are often difficult to perform on the provided samples[49]. Consequently, EUS-guided sampling is only recommended when it can alter the management of patients[9]. Recent studies have investigated the possibility to use CE-EUS for the characterization and differentiation of submucosal tumours[50]. While leiomyomas and GIST, originating from the same layer can be hard to differentiate based on EUS features alone, a recent study has reported distinct patterns at CE-EUS, with GIST showing hyperenhancement as opposed to leiomyomas which were hypoenhanced[51]. Furthermore, CE-EUS may be able to predict the malignant potential of GIST by observing tumour vascularization. Heterogeneous enhancement seems to be indicative for the presence of neoangiogenesis and represents a feature more frequently encountered in GIST of intermediate and high-risk, as recently suggested[52,53]. Although these studies included upper GI tumours, their findings could be translated to colorectal lesions, pending on further investigation needed to validate initial results (Figure 5). Reports on the role of elastography for the diagnosis of submucosal tumours should also follow to investigate their associated patterns of tissue hardness, which might differentiate benign from malignant lesions[54].
Figure 5 Contrast-enhanced endoscopic ultrasonography in a large rectal gastrointestinal stromal tumour.
A: Before contrast uptake; B: Heterogeneous enhancement after contrast injection.
EUS-GUIDED THERAPY IN COLORECTAL DISEASES - CURRENT AND FUTURE DIRECTIONS
EUS-guided drainage of abdominal and pelvic collections
EUS-guided therapy has emerged from an increasing need to develop less invasive alternatives to radiologic and surgical interventions, provided that expertize in EUS and interventional GI endoscopy is available to ensure the success of such complex procedures. Thus, despite extensive research studies for bilio-pancreatic indications, with high level of evidence data already available, colorectal EUS-guided procedures have been less frequently reported, including mainly EUS-guided drainage of abdominal and pelvic collections[55]. A recent retrospective study that included 38 patients confirmed that draining abdomino-pelvic abscesses under EUS guidance were safe and effective procedures for both the transrectal and transcolonic routes[56]. No significant difference was noted in terms of technical success (100% for each route), treatment success or complications, nor in outcomes measured during follow-ups when comparing transcolonic with transrectal drainages. Accordingly, transcolonic EUS-guided drainage can be successfully performed as an alternative to the percutaneous route when the latter seems technically demanding. While previously published literature reports have used plastic stents and catheters, in more recent case studies valuable results were also obtained with fully covered metal stents[57].
EUS-guided insertion of fiducial markers
Fiducial markers are used to delineate the target lesion and guide radiotherapy for more precision and less toxicity. EUS-guided insertion of fiducial markers appears to be safe and with high technical success rates, ranging between 85% and 100%, as reported by studies that included mainly pancreatic cancer patients[55]. However, a series of case reports validated successful EUS-guided fiducials placement in rectal cancer[58] and recent studies that included oesophageal cancer patients, demonstrated the feasibility of the procedure and the stability of the markers during image-guided radiotherapy[59,60]. Hence, considering the significant number of patients with rectal cancer that undergo radiation therapy, this would probably remain a particular research area where the role of EUS in guiding fiducials insertion needs to be further investigated.
Targeted microbubble drug delivery
Significant progress has led to the development of new microbubble ultrasound contrast agents that can be used for selective delivery of drugs and gene therapies with a wide variety of indications, including oncologic patients[50]. Different bioactive substances can be either incorporated into the microbubbles or attached to their surface, resulting in variable designs for drug-delivery vehicles[61]. Interaction of the loaded microbubbles with the ultrasound represents the triggering factor that enhances cellular uptake of the chemotherapeutic agent at the site of delivery by causing simultaneous destabilization of the microbubble encapsulating shell and increased cell membrane permeability at the targeted tissue. A recent experiment targeted tumour angiogenesis in mouse models with subcutaneous colon tumours by using endostatin microbubbles combined with focused ultrasound, which resulted in significant damage to tumour vascularization and size reduction[62]. By encouraging data from preclinical studies, microbubble drug delivery has the potential to significantly improve therapeutic strategies based on further research and validation.