Copyright
©The Author(s) 2015.
World J Gastroenterol. May 14, 2015; 21(18): 5685-5694
Published online May 14, 2015. doi: 10.3748/wjg.v21.i18.5685
Published online May 14, 2015. doi: 10.3748/wjg.v21.i18.5685
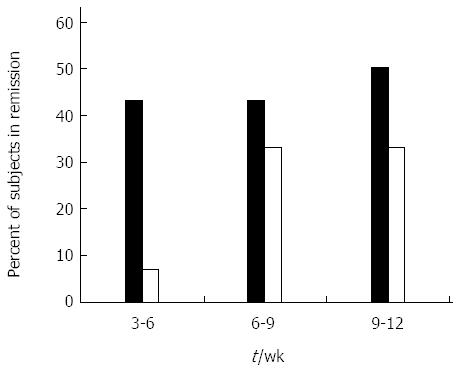
Figure 1 Effect of oral administration of AlequelTM on clinical remission.
Percent of subjects in clinical remission (Crohn’s disease activity index < 150) during the course of the study. Black bars represent the AlequelTM-treated group and open bars represent the placebo group. The evaluable number of patients in each group was too small to reach a statistical significance.
- Citation: Israeli E, Zigmond E, Lalazar G, Klein A, Hemed N, Goldin E, Ilan Y. Oral mixture of autologous colon-extracted proteins for the Crohn’s disease: A double-blind trial. World J Gastroenterol 2015; 21(18): 5685-5694
- URL: https://www.wjgnet.com/1007-9327/full/v21/i18/5685.htm
- DOI: https://dx.doi.org/10.3748/wjg.v21.i18.5685