Published online Nov 21, 2014. doi: 10.3748/wjg.v20.i43.16159
Revised: May 21, 2014
Accepted: June 26, 2014
Published online: November 21, 2014
Gastric cancer is the second most frequent cause of cancer death worldwide. Patients infected with Helicobacter pylori (H. pylori) are at increased risk of gastric cancer. H. pylori induces genomic instability in both nuclear and mitochondrial (mt) DNA of gastric epithelial cells. Changes in mtDNA represent an early event during gastric tumorigenesis, and thus may serve as potential biomarkers for early detection and prognosis in gastric carcinoma.This review article summarizes the mtDNA mutations that have been reported in gastric carcinomas and their precancerous conditions. Unexplored research topics, such as the role of mtDNA alterations in an alternative pathway of gastric carcinogenesis, are identified and directions for future research are suggested.
Core tip: Gastric cancer is a heterogeneous disease with multiple environmental etiologies and alternative pathways of carcinogenesis. Somatic mitochondrial DNA (mtDNA) mutations and variable mtDNA copy number are involved in gastric tumorigenesis. The aim of this article is to review the growing literature on the mtDNA changes in gastric carcinomas and in their precancerous conditions. Furthermore, the authors describe which research questions remain unexplored, and suggest future research directions.
- Citation: Rigoli L, Caruso RA. Mitochondrial DNA alterations in the progression of gastric carcinomas: Unexplored issues and future research needs. World J Gastroenterol 2014; 20(43): 16159-16166
- URL: https://www.wjgnet.com/1007-9327/full/v20/i43/16159.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i43.16159
Mitochondria are cytoplasmic organelles that play an essential role in numerous biological processes such as ATP production, iron and calcium homeostasis, production of reactive oxygen species, autophagic cell death and apoptosis[1]. Mitochondrial (mt) DNA was initially considered to be naked, unprotected, and vulnerable to injuries. However, recently several works have shown that mtDNA is protein-coated and packaged into aggregates called nucleoids[2-3]. Nucleoids are also important for the biogenesis of mtDNA, as they contain proteins that mediate DNA replication, repair, and recombination[4-5]. Human mtDNA is a 16.6-kb double-stranded closed-circular DNA molecule, and a few hundreds to several thousand copies of mtDNA are present in each cell[6-7]. It contains 37 genes, including the structural genes for 13 polypeptides of the electron transport chain involved in oxidative phosphorylation, two ribosomal RNAs, and a complete set of 22 tRNAs that are required for translation of the mtDNA-encoded mRNAs[8]. In addition, mtDNA contains a non-coding region: the displacement loop (D-loop) that controls both replication and transcription[9]. Generally, each human cell contains several hundred to 1000 mitochondria, and each mitochondrion has 2-10 copies of mtDNA. Somatic mtDNA mutations occur randomly, both in the non-coding D-loop region and the coding genes, increase with age, and can affect all mtDNA copies within a cell (homoplasmy) or only some (heteroplasmy)[10,11]. Homoplasmy or a high degree of heteroplasmy are needed so that a mutation results in an observable mutated cellular phenotype[10]. How mtDNA regulates the tumorigenesis process has not been clearly defined, but current evidence suggests that mutation, reduction, or deletion of mtDNA lead to defective oxidative phosphorylation, increased reactive oxygen species production, induction of the glycolytic pathway, and increased expression of prosurvival proteins, which ultimately results in cancer proliferation and tumorigenesis[12]. Therefore, modulation of mtDNA content in cancer is important for understanding the disease process.
Gastric cancer is the second leading cause of cancer death and the fourth most common malignant tumour in the world. The reason for fatality of gastric cancer is mainly due to late diagnosis and lack of programs for early detection; thus, identification of early events in gastric carcinomas is a challenging task[13,14]. Gastric carcinoma is a heterogeneous disease with several epidemiological and histopathological characteristics. This tumour is classified anatomically as proximal (also known as cardia) and distal (also known as noncardia). This classification seems to distinguish two clinicopathologic entities[15-17]. The risk factors positively associated with cardial adenocarcinoma include obesity, hiatal hernia and reflux gastroesophagitis, whereas Helicobacter pylori (H. pylori) infection appears to be the main causative agent for distal gastric adenocarcinomas[15,18,19]. Pathologically, distal gastric carcinoma may be distinguished according to the Laurèn classification[20] as intestinal or diffuse subtypes. Recently, a gastric-type differentiation has been demonstrated in some cases of intestinal-type adenocarcinomas[21-24], but the introduction of these new terms may be a source of semantic confusion, particularly with clinicians.
This review article discusses controversies regarding histogenesis and classification of distal gastric cancer. In addition, it summarizes the mtDNA changes that have been reported in gastric carcinomas and their precancerous conditions. Future research directions on the role of mtDNA in gastric carcinogenesis are suggested.
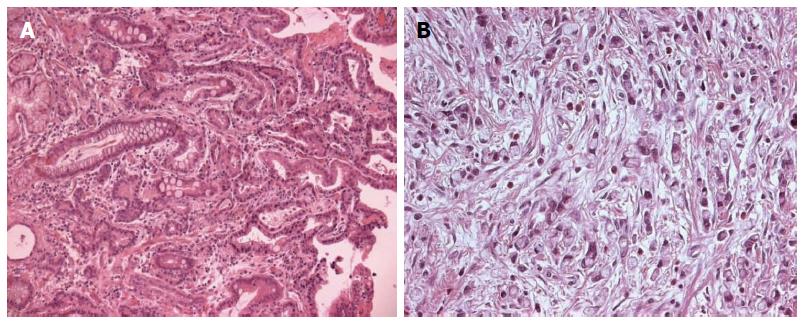
Based on histopathological features, several classification systems of gastric cancer have been proposed. The two most commonly used classifications are the Lauren’s[20] and the World Health Organization (WHO) systems[25]. The WHO classification distinguishes five major types of gastric carcinoma. This is based on the predominant morphologic component of the tumour and includes: papillary, tubular, mucinous, poorly cohesive (including signet-ring cells and other variants) and mixed carcinomas. In Lauren’s classification, gastric adenocarcinomas are divided into two main types: intestinal (Figure 1A) and diffuse (Figure 1B). Intestinal adenocarcinomas usually arise in an older population with an increased incidence in men (male/female ratio of 2:1)[15]. These tumours have the gross appearance of an exophytic mass, and histologically show a glandular structure resembling the glandular pattern of the intestine, although some solid or papillary areas are often present. Diffuse-type carcinomas do not show gender predominance, tend to develop in younger subjects, and have a poorer prognosis than intestinal-type tumours. Grossly, these tumours appear as ulcerative lesions or involve the entire thickness of the stomach wall, causing the thickening and increased firmness that has been called “linitis plastic”. Histologically, they are made up either of separated single cells with or without signet ring cell configuration or small aggregates of malignant cells with little or no gland formation[15,26]. It is thought that diffuse-type gastric carcinomas develop through the loss of function of E-cadherin, as germline mutations of the CDH1 gene (encoding E-cadherin) have been found in 30%-40% of hereditary diffuse gastric cancer cases. Furthermore, CDH1 is also frequently inactivated in sporadic diffuse-type gastric cancers through genetic and epigenetic alterations[27-33]. A neoplastic precursor lesion associated with the development of diffuse-type gastric cancer, and familial gastric cancer related to E-cadherin mutations, is usually referred to as “tubule neck dysplasia” and consists of signet ring cells that line the deep foveolar pits in a pagetoid fashion without mucosal involvement[34-36]. However, this lesion is rarely found and is not readily recognizable. Distinctive clinicopathological features of intestinal and diffuse type of gastric carcinoma are shown in Table 1.
| Intestinal-type adenocarcinoma | Gastric-type adenocarcinoma | Diffuse-type carcinoma | |
| Age | Old age | Old age | Young age |
| Sex | |||
| (Male: Female) | 2:1 | Unknown data | 1:1 |
| Precancerous condition | Corpus-predominant | Corpus-predominant | Pangastritis |
| gastritis with intestinal | Gastritis with pseudopyloric | ||
| metaplasia | metaplasia | ||
| Precancerous lesion | Intestinal-type adenoma | Pyloric-gland adenoma | Tubular-neck dysplasia: |
| signet-ring cell in situ | |||
| Gross feature | Exophytic lesion | Exophytic lesion | Ulcerative lesion and |
| linitis plastic | |||
| Microscopy | Tubulopapillary glands | Tubulopapillary glands lined by columnar cells with clear mucinous cytoplasm | Discohesive cells or signet ring cells |
| lined by columnar cells | |||
| with eosinophilic cytoplasm | |||
| Immunohistochemistry | CD10 and MUC2 | MUC5AC | Variable positivity for MUC1, MUC2, MUC5AC, MUC6 |
| immunoreactivity | immunoreactivity | ||
| Liver metastasis | Frequent | Rare | Rare |
| Peritoneal spread | Rare | Frequent | Frequent |
| Malignant potential | Low | High | High |
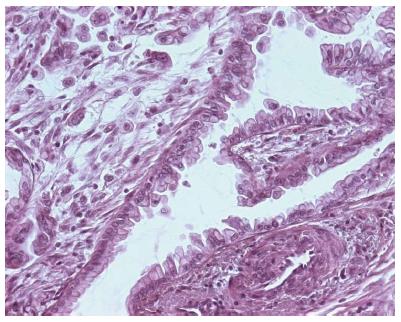
Recently, a new classification of gastric carcinomas based on mucin expression has been proposed[21-24]. Intestinal gastric carcinomas were reclassified as gastric or intestinal phenotype on the basis of mucin expression by surface mucous cells, glandular mucous cells, and intestinal columnar and goblet cells[21-24,37]. Histologically, gastric-type adenocarcinoma shows a papillary growth pattern in the upper portion and irregular branching/fusion in the deeper portion. Papillary projections are lined by columnar cells with clear mucinous cytoplasm and basally oriented enlarged vesicular nuclei with prominent nucleoli (Figure 2). Tajima et al[22] showed that gastric-type adenocarcinomas were significantly associated with a high risk of peritoneal recurrence and a poorer outcome after surgical resection compared with those with intestinal phenotype adenocarcinoma. Immunohistochemically, gastric type adenocarcinoma is positive for MUC5AC, and negative for CD10 and MUC2. Instead, intestinal-type adenocarcinoma is positive for CD10 and MUC2 and negative for MUC5AC[23,24]. Diffuse-type carcinoma shows a variable positivity for MUC1, MUC2, MUC5AC and MUC6[26].
The main clinicopathologic features of gastric-type adenocarcinoma compared to intestinal and diffuse type carcinomas are shown in Table 1.
Several studies show that both intestinal and diffuse types of gastric cancer are equally associated with H. pylori infection[15]: a Gram-negative bacterium classified as a Class I carcinogen by the WHO[38]. However, only a subset, 1%-2% of infected individuals develop gastric malignancies[15]. Clinical outcome of H. pylori infection may be correlated with specific virulence-associated bacterial genotypes such as cagA and VacA s1/m1. This genetic variability of H. pylori has been extensively studied in numerous laboratories and results have been summarized in previous publications[39-41].
Experimental studies investigating the role of H. pylori on the mitochondrial genome of gastric epithelial cells have recently been reviewed by Strickertsson et al[42]. H. pylori infection has been associated with an increase of mtDNA mutations both in the mitochondrial D-loop region and in several genes encoding subunits of the electron transport chain[43,44]. Deletion/insertion mutations have been described in the D-loop region[43,45,46]. The increase in the number of mutations was mainly attributed to a rise of transitions, possibly a consequence of oxidative damage, and was correlated with bacterial virulence-associated cagA and vacA s1/m1 genotypes[43]. mtDNA D-loop mutations may provoke a decrease in the copy number of the mitochondrial genome and alteration in gene expression. mtDNA depletion is a common event in gastric cancers[47,48]. Over 55% of gastric cancers have a lower mtDNA copy number than their corresponding non-tumoural gastric mucosa[47,48]. These results suggest that the mtDNA mutations in the D-loop region, due to H. pylori infection, contribute to the decrease in the mtDNA copy number in gastric cancer. Recently, Zhang et al[49] demonstrated that variable mtDNA content (either decreased or increased mtDNA content) markedly increased the risk of lymph node metastasis and high mortality in patients with advanced gastric carcinomas. These observations suggest that copy number variations of mtDNA may be involved in gastric cancer progression. However, the disparity of these findings in the alteration of mtDNA copy number among gastric carcinomas needs further study.
The most widely used grading system for gastritis is the Update Sydney System[50]. The system classifies chronic gastritis on the basis of topography, morphology, and, when possible, etiology. Topographic information provides further opportunities for assessing the risk of H. pylori gastritis. These are: (1) the predominance or restriction of H pylori-related gastritis in the antrum strongly correlates with an increased risk of peptic ulcer disease, and of duodenal ulcer in particular; and (2) the occurrence of corpus-predominant or pangastritis is associated with a high risk of gastric cancer[30]. In particular, patients with pangastritis are at high risk of diffuse-type gastric cancer, whereas those with corpus-predominant gastritis are at high risk of intestinal type gastric cancer (Table 1)[51].
An international group of gastroenterologists and pathologists [the Operative Link for Gastritis Assessment (OLGA)] has proposed a system for reporting gastritis in terms of stage (the OLGA Staging System)[52]. The OLGA system considers gastric atrophy as the lesion that indicates disease progression. Atrophy is distinguished in a non-metaplastic (shrinkage or complete disappearance of glandular units, replaced by expanded (fibrotic) lamina propria) and a metaplastic form including intestinal metaplasia and pseudopyloric metaplasia also known as spasmolytic polypeptide-expressing metaplasia. The OLGA staging system ranks gastric cancer risk according to the extent and severity of gastric atrophy and includes 5 stages: 0, I, and II, or low-grade atrophy associated with a low risk of gastric cancer, and III and IV, or high-grade atrophy associated with a high risk of gastric cancer[52]. The histopathological diagnosis of pseudopyloric metaplasia requires the endoscopist to communicate a correct identification of the location of the biopsy specimen in the body mucosa otherwise the pathologist considers antral-like mucosa as non-metaplastic[53]. As atrophic gastritis and pseudopyloric metaplasia remain difficult histopathologic diagnoses with low interobserver agreement, a gastritis staging system has recently been proposed as an alternative to the OLGA (OLGIM system)[54]. In the OLGIM system only intestinal metaplasia is considered as the key lesion to score for staging purposes[54]. Although replacement of atrophic gastritis by intestinal metaplasia in the staging of gastritis considerably increases interobserver agreement, the OLGIM system disregards pseudopyloric metaplasia that is now recognized as an important step in the tumorigenesis of gastric-type adenocarcinoma. By focusing on intestinal metaplasia only, the OLGIM system might be less sensitive in identifying patients with high-risk gastritis[55].
According to the Correa model, histogenesis of intestinal type gastric cancer follows a pathway of chronic active gastritis due to H. pylori infection leading to multifocal atrophy, intestinal metaplasia, followed by gastric dysplasia and finally invasive adenocarcinoma[15]. Previous studies[56-58] showed a sequential accumulation of mitochondrial microsatellite instability (MSI) in the histological progression from chronic gastritis to cancer via intestinal metaplasia and dysplasia. These findings suggested an important role of mtMSI in the progression of gastric carcinogenesis. Recent studies[59] using mtDNA mutations as a marker of clonal expansion demonstrated that intestinal metaplastic epithelium shares a common mtDNA mutation and spreads by fission: a process characterized by a bud arising from the isthmus/neck region that continues until a new gland and foveolus is formed. Furthermore, they showed that dysplasia can arise from a single clone of mutated intestinal metaplastic glands and expand to form the entire dysplastic lesion[60]. These morphologic and mtDNA findings strongly support Correa’s hypothesis of intestinal-type gastric carcinogenesis[15].
However, recent studies based on minute EGC less than 3 mm in diameter have not confirmed the association between intestinal metaplasia and intestinal type gastric cancer[61]. Some authors consider intestinal metaplasia a paracancerous lesion rather than a precancerous condition, a withered branch in the histogenetic evolution of gastric carcinoma[62,63]. Detailed mapping studies of resected stomachs from patients with intestinal-type gastric cancer have shown that atrophic gastritis, but not intestinal metaplasia, is present in every case[64,65]. Gastric atrophy therefore appears to be a better indicator of gastric cancer risk than intestinal metaplasia. Atrophy is generally present as either a multifocal or a diffuse pattern in gastric tissue and is, by definition, associated with the presence of pseudopyloric metaplasia[66,67]. This type of metaplasia may be a consequence not only of H. pylori corpus-dominant gastritis, but also of autoimmune gastritis, where disruption of oxyntic glands is due to lymphocytes. Pseudopyloric metaplasia is more strongly associated with gastric cancer than intestinal metaplasia and might be the precursor to the cancerous lesion[64-66]. In some mouse models of gastric cancer, classic intestinal metaplasia seen in humans is not observed, whereas pseudopyloric metaplasia clearly precedes and gives rise to gastric cancer[68]. The precancerous condition of pseudopyloric metaplasia has also been confirmed in patients who developed remnant carcinomas 16-20 years after a previous gastrectomy[69]. In experimental models, where chronic inflammation is absent, pseudopyloric metaplasia does not progress to dysplasia or neoplasia, suggesting that pseudopyloric metaplasia per se may not be precancerous in the absence of inflammation. To our knowledge, there are no biomolecular studies regarding the role of mtDNA mutation in pseudopyloric metaplasia.
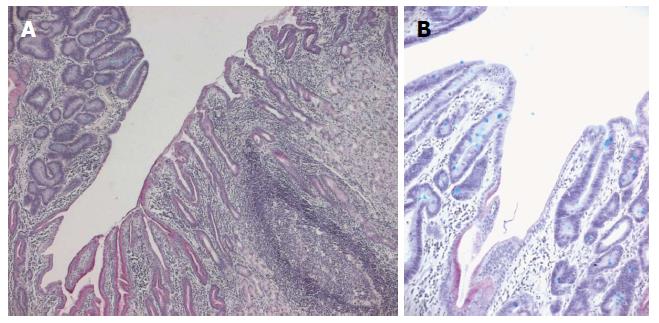
To elucidate the role of mtDNA mutations in gastric carcinogenesis, we analyzed mutations in the D-loop region of mtDNA in 24 paraffin-embedded gastric adenomas from a high gastric cancer risk area in northern Italy[46]. H. pylori infection was assessed by histological examination (Giemsa staining). The gastric adenomas were divided into two groups by their association with H. pylori gastritis. Group A with lesions arising on a background of H. pylori-positive gastritis contained 7 patients, and group B with lesions associated with H. pylori-negative gastritis contained 17 patients. Group A had a larger proportion of high-grade lesions than group B and showed a foveolar phenotype. Group B had a larger proportion of cases with mtDNA D-loop mutations than group A and exhibited an intestinal phenotype. Our data provide evidence for the morphologic and mtDNA biomolecular heterogeneity of gastric adenomas[46]. Further studies confirmed clinical and morphologic heterogeneity in gastric adenomas[37,70-76]. Phenotypically, the majority of gastric adenomas belong to the intestinal type (containing goblet cells, absorptive cells, Paneth cells, and/or columnar cells with various degrees of differentiation) (Figure 3). Gastric-type adenomas are predominantly composed of pyloric gland mucous cells (pyloric gland adenomas) or foveolar-like cells (foveolar-type adenomas)[37,70-75]. Pyloric gland adenomas occur predominantly in old women; they tend to arise in the corpus mucosa of the stomach, showing close association with pseudopyloric metaplasia of fundic glands[70-75]. Histologically, pyloric gland adenomas are composed of pyloric glands-type tubules that are not fused and are lined by a monolayer of cuboidal to low columnar epithelial cells containing clear cytoplasm or pale eosinophilic cytoplasm (showing a ground glass appearance) without an apical mucin cap[69-70,73-74]. The nuclei tend to be round and usually lack prominent nucleoli. Immunohistochemically, pyloric gland adenomas are positive for MUC6 (pyloric gland marker) with variable MUC5AC (foveolar cell marker) stain. MUC2 and CD10 are generally negative[74]. Taken together, these studies confirm that intestinal-type adenomas may represent a step towards malignant transformation of intestinal-type adenocarcinoma, according to Correa’s cascade, but suggest that gastric-type adenomas represent a distinct evolutionary pathway of gastric carcinogenesis. Thus, it is plausible that each metaplasia gives rise to a distinct type of differentiated gastric adenocarcinoma; e.g., classic intestinal metaplasia could evolve into intestinal-type gastric adenocarcinoma, according to Correa’s hypothesis, whereas it is possible to suggest a histogenetic sequence: pseudopyloric metaplasia- gastric-type adenoma -gastric-type adenocarcinoma.
Gastric adenocarcinoma is a heterogeneous disease with alternative pathways of carcinogenesis. This review article reveals that most research efforts regarding mtDNA alterations focus on gastric carcinogenesis according to the Correa model. Further studies are needed to define with greater clarity the possible role of mtDNA mutations in alternative pathways of gastric carcinogenesis, such as pseudopyloric metaplasia-gastric type adenocarcinoma.
P- Reviewer: Greco L, Marrelli D, Welling TH S- Editor: Qi Y L- Editor: O’Neill M E- Editor: Liu XM
| 1. | Raimundo N. Mitochondrial pathology: stress signals from the energy factory. Trends Mol Med. 2014;20:282-292. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 93] [Cited by in F6Publishing: 95] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 2. | Chen XJ, Butow RA. The organization and inheritance of the mitochondrial genome. Nat Rev Genet. 2005;6:815-825. [PubMed] [Cited in This Article: ] |
| 3. | Spelbrink JN. Functional organization of mammalian mitochondrial DNA in nucleoids: history, recent developments, and future challenges. IUBMB Life. 2010;62:19-32. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 81] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 4. | Friedman JR, Nunnari J. Mitochondrial form and function. Nature. 2014;505:335-343. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 972] [Cited by in F6Publishing: 1113] [Article Influence: 111.3] [Reference Citation Analysis (0)] |
| 5. | Bogenhagen DF, Rousseau D, Burke S. The layered structure of human mitochondrial DNA nucleoids. J Biol Chem. 2008;283:3665-3675. [PubMed] [Cited in This Article: ] |
| 6. | Anderson S, Bankier AT, Barrell BG, de Bruijn MH, Coulson AR, Drouin J, Eperon IC, Nierlich DP, Roe BA, Sanger F. Sequence and organization of the human mitochondrial genome. Nature. 1981;290:457-465. [PubMed] [Cited in This Article: ] |
| 7. | Attardi G. Animal mitochondrial DNA: an extreme example of genetic economy. Int Rev Cytol. 1985;93:93-145. [PubMed] [Cited in This Article: ] |
| 8. | Shadel GS, Clayton DA. Mitochondrial DNA maintenance in vertebrates. Annu Rev Biochem. 1997;66:409-435. [PubMed] [Cited in This Article: ] |
| 9. | Clayton DA. Replication of animal mitochondrial DNA. Cell. 1982;28:693-705. [PubMed] [Cited in This Article: ] |
| 10. | DiMauro S, Schon EA. Mitochondrial DNA mutations in human disease. Am J Med Genet. 2001;106:18-26. [PubMed] [Cited in This Article: ] |
| 11. | Chatterjee A, Dasgupta S, Sidransky D. Mitochondrial subversion in cancer. Cancer Prev Res (Phila). 2011;4:638-654. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 137] [Cited by in F6Publishing: 142] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 12. | Ishikawa K, Takenaga K, Akimoto M, Koshikawa N, Yamaguchi A, Imanishi H, Nakada K, Honma Y, Hayashi J. ROS-generating mitochondrial DNA mutations can regulate tumor cell metastasis. Science. 2008;320:661-664. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 980] [Cited by in F6Publishing: 1005] [Article Influence: 62.8] [Reference Citation Analysis (0)] |
| 13. | de Martel C, Forman D, Plummer M. Gastric cancer: epidemiology and risk factors. Gastroenterol Clin North Am. 2013;42:219-240. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 256] [Cited by in F6Publishing: 262] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 14. | Caruso RA, Irato E, Branca G, Finocchiaro G, Fedele F, Arnese A. Gastric adenocarcinoma incidence in the province of Messina (Insular Italy): A cancer registry study. Oncol Lett. 2014;7:861-865. [PubMed] [Cited in This Article: ] |
| 15. | Correa P. Gastric cancer: overview. Gastroenterol Clin North Am. 2013;42:211-217. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 382] [Cited by in F6Publishing: 370] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 16. | Guggenheim DE, Shah MA. Gastric cancer epidemiology and risk factors. J Surg Oncol. 2013;107:230-236. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 296] [Cited by in F6Publishing: 350] [Article Influence: 29.2] [Reference Citation Analysis (0)] |
| 17. | Shah MA, Khanin R, Tang L, Janjigian YY, Klimstra DS, Gerdes H, Kelsen DP. Molecular classification of gastric cancer: a new paradigm. Clin Cancer Res. 2011;17:2693-2701. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 225] [Cited by in F6Publishing: 227] [Article Influence: 17.5] [Reference Citation Analysis (0)] |
| 18. | Navarro Silvera SA, Mayne ST, Gammon MD, Vaughan TL, Chow WH, Dubin JA, Dubrow R, Stanford JL, West AB, Rotterdam H. Diet and lifestyle factors and risk of subtypes of esophageal and gastric cancers: classification tree analysis. Ann Epidemiol. 2014;24:50-57. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 42] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 19. | Carr JS, Zafar SF, Saba N, Khuri FR, El-Rayes BF. Risk factors for rising incidence of esophageal and gastric cardia adenocarcinoma. J Gastrointest Cancer. 2013;44:143-151. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 52] [Cited by in F6Publishing: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 20. | Lauren P. The two histological main types of gastric carcinoma: diffuse and so-called intestinal-type carcinoma. An attempt at a histo-clinical classification. Acta Pathol Microbiol Scand. 1965;64:31-49. [PubMed] [Cited in This Article: ] |
| 21. | Yamazaki K, Tajima Y, Makino R, Nishino N, Aoki S, Kato M, Sakamoto M, Morohara K, Kaetsu T, Kusano M. Tumor differentiation phenotype in gastric differentiated-type tumors and its relation to tumor invasion and genetic alterations. World J Gastroenterol. 2006;12:3803-3809. [PubMed] [Cited in This Article: ] |
| 22. | Tajima Y, Yamazaki K, Makino R, Nishino N, Aoki S, Kato M, Morohara K, Kaetsu T, Kusano M. Gastric and intestinal phenotypic marker expression in early differentiated-type tumors of the stomach: clinicopathologic significance and genetic background. Clin Cancer Res. 2006;12:6469-6479. [PubMed] [Cited in This Article: ] |
| 23. | Morohara K, Tajima Y, Nakao K, Nishino N, Aoki S, Kato M, Sakamoto M, Yamazaki K, Kaetsu T, Suzuki S. Gastric and intestinal phenotypic cell marker expressions in gastric differentiated-type carcinomas: association with E-cadherin expression and chromosomal changes. J Cancer Res Clin Oncol. 2006;132:363-375. [PubMed] [Cited in This Article: ] |
| 24. | Namikawa T, Hanazaki K. Mucin phenotype of gastric cancer and clinicopathology of gastric-type differentiated adenocarcinoma. World J Gastroenterol. 2010;16:4634-4639. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 30] [Cited by in F6Publishing: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 25. | Lauwers GY, Carneiro F, Graham DY, Curado MP, Franceschi S, Montgomery E, Tatematsu M, Hattori T. Gastric carcinoma. WHO Classification of Tumors of the Digestive System. 4th Edition. Lyon: IARC 2010; 48-68. [Cited in This Article: ] |
| 26. | Nguyen MD, Plasil B, Wen P, Frankel WL. Mucin profiles in signet-ring cell carcinoma. Arch Pathol Lab Med. 2006;130:799-804. [PubMed] [Cited in This Article: ] |
| 27. | Mimata A, Fukamachi H, Eishi Y, Yuasa Y. Loss of E-cadherin in mouse gastric epithelial cells induces signet ring-like cells, a possible precursor lesion of diffuse gastric cancer. Cancer Sci. 2011;102:942-950. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 28. | Yamamoto E, Suzuki H, Takamaru H, Yamamoto H, Toyota M, Shinomura Y. Role of DNA methylation in the development of diffuse-type gastric cancer. Digestion. 2011;83:241-249. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 29. | Nobili S, Bruno L, Landini I, Napoli C, Bechi P, Tonelli F, Rubio CA, Mini E, Nesi G. Genomic and genetic alterations influence the progression of gastric cancer. World J Gastroenterol. 2011;17:290-299. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 82] [Cited by in F6Publishing: 92] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 30. | Fitzgerald RC, Hardwick R, Huntsman D, Carneiro F, Guilford P, Blair V, Chung DC, Norton J, Ragunath K, Van Krieken JH. Hereditary diffuse gastric cancer: updated consensus guidelines for clinical management and directions for future research. J Med Genet. 2010;47:436-444. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 380] [Cited by in F6Publishing: 368] [Article Influence: 26.3] [Reference Citation Analysis (0)] |
| 31. | Guilford P, Humar B, Blair V. Hereditary diffuse gastric cancer: translation of CDH1 germline mutations into clinical practice. Gastric Cancer. 2010;13:1-10. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 113] [Cited by in F6Publishing: 114] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 32. | Barber ME, Save V, Carneiro F, Dwerryhouse S, Lao-Sirieix P, Hardwick RH, Caldas C, Fitzgerald RC. Histopathological and molecular analysis of gastrectomy specimens from hereditary diffuse gastric cancer patients has implications for endoscopic surveillance of individuals at risk. J Pathol. 2008;216:286-294. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 90] [Cited by in F6Publishing: 93] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 33. | Carneiro F, Huntsman DG, Smyrk TC, Owen DA, Seruca R, Pharoah P, Caldas C, Sobrinho-Simões M. Model of the early development of diffuse gastric cancer in E-cadherin mutation carriers and its implications for patient screening. J Pathol. 2004;203:681-687. [PubMed] [Cited in This Article: ] |
| 34. | Ghandur-Mnaymneh L, Paz J, Roldan E, Cassady J. Dysplasia of nonmetaplastic gastric mucosa. A proposal for its classification and its possible relationship to diffuse-type gastric carcinoma. Am J Surg Pathol. 1988;12:96-114. [PubMed] [Cited in This Article: ] |
| 35. | Kumarasinghe MP, Lim TK, Ooi CJ, Luman W, Tan SY, Koh M. Tubule neck dysplasia: precursor lesion of signet ring cell carcinoma and the immunohistochemical profile. Pathology. 2006;38:468-471. [PubMed] [Cited in This Article: ] |
| 36. | Lauwers GY, Srivastava A. Gastric preneoplastic lesions and epithelial dysplasia. Gastroenterol Clin North Am. 2007;36:813-829, vi. [PubMed] [Cited in This Article: ] |
| 37. | Kushima R, Vieth M, Borchard F, Stolte M, Mukaisho K, Hattori T. Gastric-type well-differentiated adenocarcinoma and pyloric gland adenoma of the stomach. Gastric Cancer. 2006;9:177-184. [PubMed] [Cited in This Article: ] |
| 38. | Schistosomes , liver flukes and Helicobacter pylori. IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Lyon, 7-14 June 1994. IARC Monogr Eval Carcinog Risks Hum. 1994;61:1-241. [PubMed] [Cited in This Article: ] |
| 39. | Rudi J, Kolb C, Maiwald M, Kuck D, Sieg A, Galle PR, Stremmel W. Diversity of Helicobacter pylori vacA and cagA genes and relationship to VacA and CagA protein expression, cytotoxin production, and associated diseases. J Clin Microbiol. 1998;36:944-948. [PubMed] [Cited in This Article: ] |
| 40. | Galmiche A, Rassow J, Doye A, Cagnol S, Chambard JC, Contamin S, de Thillot V, Just I, Ricci V, Solcia E. The N-terminal 34 kDa fragment of Helicobacter pylori vacuolating cytotoxin targets mitochondria and induces cytochrome c release. EMBO J. 2000;19:6361-6370. [PubMed] [Cited in This Article: ] |
| 41. | Atherton JC, Peek RM, Tham KT, Cover TL, Blaser MJ. Clinical and pathological importance of heterogeneity in vacA, the vacuolating cytotoxin gene of Helicobacter pylori. Gastroenterology. 1997;112:92-99. [PubMed] [Cited in This Article: ] |
| 42. | Strickertsson JA, Desler C, Rasmussen LJ. Impact of bacterial infections on aging and cancer: impairment of DNA repair and mitochondrial function of host cells. Exp Gerontol. 2014;56:164-174. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 43. | Machado AM, Figueiredo C, Touati E, Máximo V, Sousa S, Michel V, Carneiro F, Nielsen FC, Seruca R, Rasmussen LJ. Helicobacter pylori infection induces genetic instability of nuclear and mitochondrial DNA in gastric cells. Clin Cancer Res. 2009;15:2995-3002. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 98] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 44. | Machado AM, Desler C, Bøggild S, Strickertsson JA, Friis-Hansen L, Figueiredo C, Seruca R, Rasmussen LJ. Helicobacter pylori infection affects mitochondrial function and DNA repair, thus, mediating genetic instability in gastric cells. Mech Ageing Dev. 2013;134:460-466. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 31] [Cited by in F6Publishing: 36] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 45. | Hiyama T, Tanaka S, Shima H, Kose K, Tuncel H, Ito M, Kitadai Y, Sumii M, Yoshihara M, Shimamoto F. Somatic mutation in mitochondrial DNA and nuclear microsatellite instability in gastric cancer. Oncol Rep. 2003;10:1837-1841. [PubMed] [Cited in This Article: ] |
| 46. | Rigoli L, Di Bella C, Verginelli F, Falchetti M, Bersiga A, Rocco A, Nardone G, Mariani-Costantini R, Caruso RA. Histological heterogeneity and somatic mtDNA mutations in gastric intraepithelial neoplasia. Mod Pathol. 2008;21:733-741. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 47. | Yu M. Generation, function and diagnostic value of mitochondrial DNA copy number alterations in human cancers. Life Sci. 2011;89:65-71. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 142] [Cited by in F6Publishing: 147] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 48. | Wu CW, Yin PH, Hung WY, Li AF, Li SH, Chi CW, Wei YH, Lee HC. Mitochondrial DNA mutations and mitochondrial DNA depletion in gastric cancer. Genes Chromosomes Cancer. 2005;44:19-28. [PubMed] [Cited in This Article: ] |
| 49. | Zhang G, Qu Y, Dang S, Yang Q, Shi B, Hou P. Variable copy number of mitochondrial DNA (mtDNA) predicts worse prognosis in advanced gastric cancer patients. Diagn Pathol. 2013;8:173. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 27] [Cited by in F6Publishing: 29] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 50. | Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996;20:1161-1181. [PubMed] [Cited in This Article: ] |
| 51. | Uemura N, Okamoto S, Yamamoto S, Matsumura N, Yamaguchi S, Yamakido M, Taniyama K, Sasaki N, Schlemper RJ. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784-789. [PubMed] [Cited in This Article: ] |
| 52. | Rugge M, Meggio A, Pennelli G, Piscioli F, Giacomelli L, De Pretis G, Graham DY. Gastritis staging in clinical practice: the OLGA staging system. Gut. 2007;56:631-636. [PubMed] [Cited in This Article: ] |
| 53. | Rugge M, Pennelli G, Pilozzi E, Fassan M, Ingravallo G, Russo VM, Di Mario F, Gruppo Italiano Patologi Apparato Digerente (GIPAD), Società Italiana di Anatomia Patologica e Citopatologia Diagnostica/International Academy of Pathology, Italian division (SIAPEC/IAP). Gastritis: the histology report. Dig Liver Dis. 2011;43 Suppl 4:S673-S684. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 90] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 54. | Capelle LG, de Vries AC, Haringsma J, Ter Borg F, de Vries RA, Bruno MJ, van Dekken H, Meijer J, van Grieken NC, Kuipers EJ. The staging of gastritis with the OLGA system by using intestinal metaplasia as an accurate alternative for atrophic gastritis. Gastrointest Endosc. 2010;71:1150-1158. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 301] [Cited by in F6Publishing: 320] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 55. | Rugge M, Fassan M, Pizzi M, Farinati F, Sturniolo GC, Plebani M, Graham DY. Operative link for gastritis assessment vs operative link on intestinal metaplasia assessment. World J Gastroenterol. 2011;17:4596-4601. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 89] [Cited by in F6Publishing: 88] [Article Influence: 6.8] [Reference Citation Analysis (1)] |
| 56. | Ling XL, Fang DC, Wang RQ, Yang SM, Fang L. Mitochondrial microsatellite instability in gastric cancer and its precancerous lesions. World J Gastroenterol. 2004;10:800-803. [PubMed] [Cited in This Article: ] |
| 57. | Sui G, Zhou S, Wang J, Canto M, Lee EE, Eshleman JR, Montgomery EA, Sidransky D, Califano JA, Maitra A. Mitochondrial DNA mutations in preneoplastic lesions of the gastrointestinal tract: a biomarker for the early detection of cancer. Mol Cancer. 2006;5:73. [PubMed] [Cited in This Article: ] |
| 58. | Jeong CW, Lee JH, Sohn SS, Ryu SW, Kim DK. Mitochondrial microsatellite instability in gastric cancer and gastric epithelial dysplasia as a precancerous lesion. Cancer Epidemiol. 2010;34:323-327. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 12] [Cited by in F6Publishing: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 59. | McDonald SA, Greaves LC, Gutierrez-Gonzalez L, Rodriguez-Justo M, Deheragoda M, Leedham SJ, Taylor RW, Lee CY, Preston SL, Lovell M. Mechanisms of field cancerization in the human stomach: the expansion and spread of mutated gastric stem cells. Gastroenterology. 2008;134:500-510. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 188] [Cited by in F6Publishing: 189] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 60. | Gutierrez-Gonzalez L, Graham TA, Rodriguez-Justo M, Leedham SJ, Novelli MR, Gay LJ, Ventayol-Garcia T, Green A, Mitchell I, Stoker DL. The clonal origins of dysplasia from intestinal metaplasia in the human stomach. Gastroenterology. 2011;140:1251-1260.e1-e6. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 60] [Cited by in F6Publishing: 70] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 61. | Kawachi H, Takizawa T, Eishi Y, Shimizu S, Kumagai J, Funata N, Koike M. Absence of either gastric or intestinal phenotype in microscopic differentiated gastric carcinomas. J Pathol. 2003;199:436-446. [PubMed] [Cited in This Article: ] |
| 62. | Meining A, Morgner A, Miehlke S, Bayerdörffer E, Stolte M. Atrophy-metaplasia-dysplasia-carcinoma sequence in the stomach: a reality or merely an hypothesis? Best Pract Res Clin Gastroenterol. 2001;15:983-998. [PubMed] [Cited in This Article: ] |
| 63. | Goldenring JR, Nam KT, Wang TC, Mills JC, Wright NA. Spasmolytic polypeptide-expressing metaplasia and intestinal metaplasia: time for reevaluation of metaplasias and the origins of gastric cancer. Gastroenterology. 2010;138:2207-2210, 2210.e1. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 127] [Cited by in F6Publishing: 142] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 64. | El-Zimaity HM, Ota H, Graham DY, Akamatsu T, Katsuyama T. Patterns of gastric atrophy in intestinal type gastric carcinoma. Cancer. 2002;94:1428-1436. [PubMed] [Cited in This Article: ] |
| 65. | Halldórsdóttir AM, Sigurdardóttrir M, Jónasson JG, Oddsdóttir M, Magnússon J, Lee JR, Goldenring JR. Spasmolytic polypeptide-expressing metaplasia (SPEM) associated with gastric cancer in Iceland. Dig Dis Sci. 2003;48:431-441. [PubMed] [Cited in This Article: ] |
| 66. | Schmidt PH, Lee JR, Joshi V, Playford RJ, Poulsom R, Wright NA, Goldenring JR. Identification of a metaplastic cell lineage associated with human gastric adenocarcinoma. Lab Invest. 1999;79:639-646. [PubMed] [Cited in This Article: ] |
| 67. | El-Zimaity H. Gastritis and gastric atrophy. Curr Opin Gastroenterol. 2008;24:682-686. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 20] [Cited by in F6Publishing: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 68. | Fox JG, Li X, Cahill RJ, Andrutis K, Rustgi AK, Odze R, Wang TC. Hypertrophic gastropathy in Helicobacter felis-infected wild-type C57BL/6 mice and p53 hemizygous transgenic mice. Gastroenterology. 1996;110:155-166. [PubMed] [Cited in This Article: ] |
| 69. | Yamaguchi H, Goldenring JR, Kaminishi M, Lee JR. Identification of spasmolytic polypeptide expressing metaplasia (SPEM) in remnant gastric cancer and surveillance postgastrectomy biopsies. Dig Dis Sci. 2002;47:573-578. [PubMed] [Cited in This Article: ] |
| 70. | Abraham SC, Montgomery EA, Singh VK, Yardley JH, Wu TT. Gastric adenomas: intestinal-type and gastric-type adenomas differ in the risk of adenocarcinoma and presence of background mucosal pathology. Am J Surg Pathol. 2002;26:1276-1285. [PubMed] [Cited in This Article: ] |
| 71. | Vieth M, Kushima R, Borchard F, Stolte M. Pyloric gland adenoma: a clinico-pathological analysis of 90 cases. Virchows Arch. 2003;442:317-321. [PubMed] [Cited in This Article: ] |
| 72. | Park do Y, Srivastava A, Kim GH, Mino-Kenudson M, Deshpande V, Zukerberg LR, Song GA, Lauwers GY. Adenomatous and foveolar gastric dysplasia: distinct patterns of mucin expression and background intestinal metaplasia. Am J Surg Pathol. 2008;32:524-533. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 73] [Cited by in F6Publishing: 63] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 73. | Chen ZM, Scudiere JR, Abraham SC, Montgomery E. Pyloric gland adenoma: an entity distinct from gastric foveolar type adenoma. Am J Surg Pathol. 2009;33:186-193. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 94] [Cited by in F6Publishing: 75] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 74. | Vieth M, Kushima R, Mukaisho K, Sakai R, Kasami T, Hattori T. Immunohistochemical analysis of pyloric gland adenomas using a series of Mucin 2, Mucin 5AC, Mucin 6, CD10, Ki67 and p53. Virchows Arch. 2010;457:529-536. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 42] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 75. | Robinson JC, Hoffman B, Sun S. Gastric adenomas: Subtypes and their clinical significance. Pathol Case Rev. 2013;18:70-74. [DOI] [Cited in This Article: ] [Cited by in Crossref: 2] [Cited by in F6Publishing: 2] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 76. | Rugge M, Capelle LG, Cappellesso R, Nitti D, Kuipers EJ. Precancerous lesions in the stomach: from biology to clinical patient management. Best Pract Res Clin Gastroenterol. 2013;27:205-223. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 79] [Cited by in F6Publishing: 79] [Article Influence: 7.2] [Reference Citation Analysis (0)] |