Published online Dec 7, 2013. doi: 10.3748/wjg.v19.i45.8168
Revised: October 18, 2013
Accepted: November 3, 2013
Published online: December 7, 2013
Therapeutic management of Helicobacter pylori (H. pylori) remains an unsolved issue. Indeed, no therapeutic regimen is able to cure the infection in all treated patients, and in many the infection persists despite the administration of several consecutive standard therapies. Although antibiotic resistance reports describe alarming results, the outcome of therapeutic regimens does not seem to parallel this scenario in most cases, since a successful performance is often reached in more than 80% of cases. However, the phenomenon of increasing antibiotic resistance is being closely studied, and the results show controversial aspects even in the same geographic area. For the continents of Europe, America, Asia, Africa, and Oceania, minimal and maximal values of resistance to the main antibiotics (clarithromycin, amoxicillin, metronidazole, and levofloxacin) feature wide ranges in different countries. The real enigma is therefore linked to the several different therapeutic regimens, which show results that often do not parallel the in vitro findings even in the same areas. A first aspect to be emphasized is that some regimens are limited by their use in very small geographic districts. Moreover, not all therapeutic trials have considered bacterial and host factors affecting the therapeutic outcome. The additional use of probiotics may help to reduce adverse events, but their therapeutic impact is doubtful. In conclusion, the “ideal therapy”, paradoxically, appears to be a “utopia”, despite the unprecedented volume of studies in the field and the real breakthrough in medical practice made by the discovery and treatment of H. pylori. The ample discrepancies observed in the different areas do not encourage the development of therapeutic guidelines that could be valid worldwide. On these bases, one of the main challenges for the future might be identifying a successful solution to overcome antibiotic resistances. In this context, geography must be considered a relevant matter.
Core tip: The present topic outlines the main data regarding antibiotic resistances, paying particular attention to the discrepant results obtained in different geographic areas worldwide, and even in the same districts. Discordances between in vitro and in vivo studies are detailed and the possible factors explaining this phenomenon are analyzed. Finally, the challenge for the future of devising a successful solution to overcome antibiotic resistances is highlighted, and geography is suggested as a relevant matter.
-
Citation: Ierardi E, Giorgio F, Losurdo G, Di Leo A, Principi M. How antibiotic resistances could change
Helicobacter pylori treatment: A matter of geography? World J Gastroenterol 2013; 19(45): 8168-8180 - URL: https://www.wjgnet.com/1007-9327/full/v19/i45/8168.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i45.8168
Helicobacter pylori (H. pylori) eradication has undoubted benefits. The bacterium’s diffusion worldwide, even if it is decreasing, especially in developed countries, still dictates the treatment of all infected subjects, and the possible legal implications that denial of treatment could have must also be considered. In Table 1, epidemiological studies of the last five years from five continents of the world (Europe, America, Asia, Africa, and Oceania) are reported[1-14]. Different infection rates depend on the type of study population (pediatric, adult, or geriatric patients), since increasing age, as well as poor hygienic environmental factors, are well known to have a strong influence. Arresting the bacterium’s diffusion still has not been achieved.
| Country | Ref. | Helicobacter pylori positivity | Test | Population |
| Southern Europe | ||||
| Italy | Dore et al[1], 2012 | 13.3% | Serology | Children |
| Portugal | Bastos et al[2], 2013 | 84.2% | Serology | Adults |
| Northern Europe | ||||
| Sweden | Thjodleifsson et al[3], 2007 | 11% | Serology | Adults |
| Norway | Bakkevold[4], 2010 | 51% | Serology | Adults |
| Eastern Europe | ||||
| Czech Republic | Bureš[5], 2012 | 23.5% | Urea breath test | Combined data |
| Russia | Svarval’ et al[6], 2011 | 40.48% | Serology | Children-adolescents |
| America | ||||
| United States | Patterson et al[7], 2012 | 17.1% | Serology | Adults |
| South America (combined data) | Porras et al[8], 2013 | 79.4% | Urea breath test | Adults |
| Asia | ||||
| Saudi Arabia | Hanafi et al[9], 2013 | 28.3% | Serology | Children, adolescents |
| South Korea | Baik et al[10], 2012 | 55.7% | Serology | Adults |
| Africa | ||||
| Nigeria | Etukudo et al[11], 2012 | 30.9% | Serology | Children |
| Morocco | Benajah et al[12], 2013 | 75.5% | Biopsy | Adults |
| Oceania | ||||
| Australia | Pandeya et al[13], 2011 | 15.5% | Serology | Combined data |
| New Zealand | Fawcett et al[14], 2005 | 6.2% | Serology | Children, adolescents |
Treatment of the infection nowadays is both a simple and, at the same time, complex problem. Alongside the conventional first-line regimen (triple therapy), others have been proposed (sequential, concomitant, quadruple, and miscellaneous) to face the growing problem of antibiotic resistance. For no other infection have so large a number of therapeutic proposals been reported by different working groups. The results, however, often appear conflicting and the same regimen may be extremely effective in one geographic area and deliver disappointing results in another. Finally, although many experts believe that there is no such thing as untreatable H. pylori, only ill-treated H. pylori, no clinical trial has yielded a successful 100% eradication. We must therefore conclude that “the infallible therapy” does not exist at present. Therefore, the aim of this review is to analyze the different results of therapeutic schemes in different geographic regions, as well as their relationship with the diffusion of antibiotic resistances in the same areas.
H. pylori antibiotic resistances are defined as primary (no previous treatment for bacterium eradication) and secondary (a susceptible strain acquires resistance during a treatment)[15]. The main reason for this phenomenon is point mutations of H. pylori DNA, often associated with inappropriate antibiotic use[15]. Heteroresistance is a condition characterized by the coexistence of susceptible and resistant strains in the same patient. Resistances are currently detected by culture-based and molecular methods. Different tests for both techniques have been described. The main culture-based techniques are the agar dilution method, E-test, breakpoint susceptibility test, and modified disk diffusion method. Molecular techniques include polymerase chain reaction (PCR), restriction fragment length polymorphism, allele-specific PCR, sequencing, real-time PCR, and fluorescent in situ hybridization[16-22]. Although these tests require sophisticated tools, their performance before starting a treatment for H. pylori will undoubtedly significantly improve the therapeutic outcome. However, this strategy is hard to apply in clinical practice owing to the long period necessary before obtaining results, as well as the high costs of routine performance of such methods.
A large number of reports from different geographic areas are available in the literature, showing heterogeneous results. Firstly, we will consider the geographic rates of resistance to each antibiotic, thus underlining differences and consequent clinical implications.
Clarithromycin is a drug that belongs to the macrolide family, and its mechanism of action is the inhibition of protein synthesis by binding and slowing down the activity of the bacterial ribosomal unit[23]. The mutations that may cause resistances are point mutations in the 23SrRNA component of ribosomes[24]: the three most frequent mutations that may occur are A2143G, A2142G, and A2142C, which are responsible for 90% of cases of primary clarithromycin resistance in H. pylori strains isolated in Western countries[25,26]. In particular, the A2143G mutation has a much stronger impact on conferring resistance than the other two[27]. In Eastern countries (e.g., South Korea) additional mutations, such as T2183C and A2223G, have been frequently found to justify the observed clarithromycin resistance, while A2143G accounted only for 23% of resistant strains[26]. This finding, if confirmed in other Asian countries, suggests that point mutations inducing clarithromycin resistance might differ in this continent as compared to those in Europe and North America. In conclusion, resistance to clarithromycin is considered as the cause of most eradication regimen failures[28], as its prevalence is continuously increasing. Moreover, new point mutations have also emerged in South America[29]. However, isolated reports may not reflect the real scenario; therefore, it would be more useful to consider epidemiological data about resistance region by region.
In Eastern Asian countries, very high clarithromycin resistances have been recorded. The highest rate of resistance was found in Japan (86.4%) in a study on the efficacy of this antibiotic in a third line regimen[30]. Interestingly, in the same country, a lower percentage of 15.2% was detected in another study of first line therapy[31]. In any case, in every region of the Far East, the rates of resistance were at least 15%, a percentage that often determines the failure of clarithromycin-based therapy. In another two Japanese studies the rates were 32.4%[32] and 18.9%[33], respectively. In China, resistances ranged between 21.5% and 23.8%[34,35], while in Vietnam they were considerably higher (33%)[36]. A South Korean study revealed, in a pediatric population, tripled resistance rates within 20 years. Surprisingly, a Malaysian study carried out on 90 gastric samples did not show any strain resistant to clarithromycin: this is the only discordant value in this geographical area[37].
A similar pattern is observed in the Near East, where the percentages range between 14.3% (Iran)[38] and 37% (Pakistan)[39]. Interestingly, it is noticeable that in the same area, another study found a much lower resistance (17.1%)[40], emphasizing that even in the same geographical region, relevant differences may occur.
In Southern America, the most recent studies report a resistance rate that ranges between 13.6% and 19.5%, illustrating a more homogeneous distribution of resistant strains[41,42]. In the United States the continuous migration flows from Mexico and other Latin American countries are causing rapid changes in local H. pylori strains, as witnessed by the need for new regimens[43]. If we consider that in a 2011 randomized study the success rate of a therapy including levofloxacin, amoxicillin, and clarithromycin was only 73.3%[44], whereas in 1995 a clarithromycin-based therapy achieved eradication in more than 90%[45], this is a remarkable difference.
In Europe, there are ample variations between Northern Europe and Mediterranean countries, where the resistances to clarithromycin are considerably more widespread. Finland and Sweden recorded a rate of 2% and 1.5%[46,47], respectively, whilst in Germany and Norway the rates were 7.5% and 5.9%, respectively[48,49].
Resistance is higher in Central/Eastern Europe [9.3% (95%CI: 0-22)], and is at its highest in Southern Europe [18% (95%CI: 2.1-34.8)][50]. In Italy, the trend is continually on the increase; while in the year 2000 the percentages ranged between 1.8% and 14%[51-53], a few years later resistance had increased up to 24.1%, and will likely have doubled within 15 years[54,55]. As in all of Europe, primary clarithromycin resistance in Italy is highly variable in different geographic areas: 0%-6% in the north, 7%-15% in central areas, and 10%-25% in the south[56]. Other Mediterranean countries with high rates of clarithromycin resistance are Greece (40%), Spain (15%-20%), France (17.5%), and Portugal (34.7%)[57-60]. In Eastern Europe, the situation is similar to that in Southern Europe; several Bulgarian studies have reported a resistance rate of 18.4%-23.4%[15,61,62]. An isolated phenomenon has been observed in Poland; in 2013, resistance is around 22%, with a trend toward a perceptible decrease (it was 34% in 2008)[63]. Another strong diffusion of resistant strains has been shown in Lithuania (24.7%)[64].
In Oceania, resistance rates range between 8.7%[65] and 15.7%[66], suggesting that there is still a fair option for the use of the antibiotic in those areas.
Finally, the main data about the prevalence of resistance to clarithromycin in Africa show very low values in Gambia and Senegal (0% and 1%, respectively)[67,68], but not in South Africa (15.3%)[69].
Metronidazole is a nitroimidazole antibiotic used particularly against anaerobic bacteria and protozoa. It works as a pro-drug: it is non-enzymatically reduced by reacting with reduced ferredoxin, which is generated by pyruvate oxidoreductase, and then the reduced molecule is taken up into bacterial DNA and forms unstable molecules that cause the death of the organism[70]. The resistance mechanism to metronidazole is not entirely straightforward[71,72]. Clearly, alterations of the rdxA gene are of primary relevance, but it has not been possible to identify a clear panel of point mutations which could explain the phenomenon. Moreover, other genes such as frxA seem to be involved[73].
Eastern Asian countries are the geographical area where it is possible to detect the highest percentages of resistance: 56.6%-95.4% in China[34,35], 57% in Japan[30], 27.3%-52.9% in South Korea[74,75], 69.9% in Vietnam[36], and 75.5% in Malaysia[37]. A very similar pattern has been detected in Africa, where the rates vary between 68.8% and 85%[67,68]. Indeed, it is well known that the prevalence is much higher in developing countries (50%-80%) like Mexico (76.3%)[76], Colombia (75.5%)[41], and Brazil (40%)[42]. In the Near East, a 69.5% resistance was reported in Saudi Arabia[77], 64.5% in Pakistan [40], and 76.8% in Iran[38].
The scenario appears to be slightly different in Europe, where in a multicenter study the global resistance rate to metronidazole was 33.1% (95%CI: 7.5-58.9), with no significant difference between the north [33% (95%CI: 7.1-69.2)] and south [40.8% (95%CI: 27.3-54.3)], but with a significantly lower prevalence in central and eastern areas [29.2% (95%CI: 17.9-41.5)][78]. However, this report was dated 2001, and in the last ten years the situation appears to have changed surprisingly, featuring a decreasing rate of resistance in northern countries (22.5% in Norway[49], 1.1% in Lithuania[63], 19.9% in the Netherlands[79], and 13% in the United Kingdom[51]). In Southern and Central-Eastern Europe however, rates of resistance are much higher (34.9% in France[59], 63.6% in Croatia[80], 37.2% in Germany[48], and 23.3% in Bulgaria[15]).
Several Italian studies[81] have described a resistance rate of 20%-23.9%[82-84], but the resistant strain could be undergoing a dizzying growth, if we consider that only five years later a single group reported a more than doubled rate (59.3%)[85].
In Oceania, three recent studies reported values of 20%[86], 36%[87], and 43.5%[65], suggesting an overall high rate of resistances.
Amoxicillin is a β-lactam antibiotic included in all current therapeutic regimens for H. pylori eradication[88]. Amoxicillin acts by interfering with peptidoglycan synthesis, in particular by blocking transporters named penicillin binding proteins[16]. This drug was the first antibiotic used for H. pylori therapy due to a presumed absence of resistance[89]. In almost all studies, percentages of resistance are quite low, and these data seem homogeneous worldwide. No resistances have been detected in Croatia, France, Germany, the Netherlands, Portugal, Spain, or Sweden[90-96]. In Italy, resistances range from 0% to 0.2%[97], while in the United Kingdom they range from 0% to 0.4%[98,99]. Almost negligible resistances have also been reported in America and Oceania. Data in contrast with this trend have been described in Iran and Japan, with a resistance prevalence of 28.6%[38] and 8.2%-15.2%[30,33], respectively. Surprisingly, an extremely high resistance rate (85.6%) has been observed in Cameroon[81].
Levofloxacin is a broad spectrum antibiotic of the fluoroquinolone drug class which is active against both Gram-positive and Gram-negative bacteria[100,101]. It acts by inhibiting DNA gyrase, type II topoisomerase, and topoisomerase IV, an enzyme which is necessary to separate replicated DNA and block cell division[102]. Resistance of H. pylori to fluoroquinolones is due to point mutations in the quinolone resistance determining regions of gyrA[103].
Levofloxacin has recently appeared in therapeutic regimens for H. pylori eradication: in the Maastricht-Florence IV consensus for H. pylori treatment, a levofloxacin-containing regimen was proposed as second-line treatment when classical first-line therapy containing clarithromycin failed[88]. However, in the last three years, resistant strains are increasing, because of plasmid-mediated horizontally transferable genes encoding quinolone resistance[104], so that more and more levofloxacin-based treatments will likely be ineffective in the future.
An example of this unfavorable trend is evident in Asian countries, where the rates of resistance exceed 10%: 18.4% in Vietnam[36], 20.6% in China[34], and are as high as 63.3% in Pakistan[39]. Only Malaysia registered 0%[37], although Japan was also low at 8.2%[30].
In Europe, the overall resistance to levofloxacin, detected in a recent multicentric epidemiologic study, is 14.1%[105], with values ranging between 11.7% in Ireland[106] and 29.1% in Germany[107]: these last percentages must set the clinician on guard, if we consider that only a few years before, in 2003, a resistance rate of 3.3% was detected in France[108]. In Italy, a single study found resistance in 10.6% of strains[81], data confirmed by a recent overview that noted a rate of 11.8% in already treated patients[109]. In Africa, resistant strains are also not very widespread: 15% in Senegal and 10.2% in South Africa[68,69].
In America, a rate of 19% was found in Alaska[110], while in South America percentages are higher (23% in Brazil, where a clarithromycin resistance of only 8% means that this last drug is still a good therapeutic option)[111]. Surprisingly, no report about levofloxacin resistance in Oceania has yet been made, to the best of our knowledge.
Resistance to tetracycline is very low, or even absent, in most countries. Very low rates have been reported in Spain (0.7%[95]), the United Kingdom (0.5%[98]), and Hong Kong (0.5%[112]). Values lower than 5% are recorded in Germany[48] and Lithuania[64]. The highest prevalence rates are found in Korea (5.3%[113]), Iran[38] (18.7%), and Vietnam[36] (5.8%). The resistance mechanism has been described as a change in three contiguous nucleotides in the 16S rRNA gene (AGA 926-928RTTC)[114,115].
Rifabutin is a bactericidal antibiotic drug primarily used in the treatment of tuberculosis, and its effect on bacteria is based on DNA-dependent RNA polymerase blockage[116]. When it was firstly used in the late ’90s, the prevalence of H. pylori resistance to this group of antibiotics was extremely low, as these drugs were used only in a limited number of patients to treat mycobacterial infections. For example, Heep et al[117] did not find a single resistant strain among 81 German patients tested in 1999, nor did Fujimura et al[118] among 52 strains in Japan. Even today in some regions such as Brazil, Ireland, and Senegal, no resistant strain has been detected[42,68,106], but data from Malaysia[37], Germany[48], and Iran[38] report a resistance rate of 2.2%, less than 5%, and 28.6%, respectively. Resistance is due to point mutations in the rpoB gene, as for other bacteria, and occurs in all rifamycin drugs, suggesting a potential risk of cross-resistances between antibiotics of the same family[119,120].
Furazolidone is a nitrofuran antibiotic. As a veterinary medicine, it is used to treat salmonids for Myxobolus cerebralis infections. In the past, it has been used in humans to treat diarrhea and enteritis caused by bacteria or protozoan infections, but since 1991 it has been recognized by the FDA as a carcinogenic agent and so is no longer used, except in a few developing countries that allow its use for human diseases[121]. It was previously used to treat traveler’s diarrhea, cholera, and bacteremic salmonellosis. Its use to treat H. pylori infections was also proposed[122]. Primary furazolidone resistance is rare. In Spain[123], the rates were less than 2%, and in Bulgaria 1.8%[124]. Lower values have been described in China[34] (0.1%) and Brazil[42] (no resistant strains detected). A higher furazolidone resistance rate (9%) was found in Iranian children[125]. As the high cost of some drugs, such as clarithromycin and quinolones, prevents their use in developing countries, where a high prevalence of primary metronidazole resistance is also present, to overcome these limitations, furazolidone-based treatments have been suggested. On the other hand, the low rate of primary H. pylori resistance to furazolidone in developed countries may make the use of this drug attractive. In this case, however, it is imperative to consider that furazolidone-based first-line therapy achieves H. pylori eradication rates of 75.7% and 79.6%[126], and its use cannot ignore obvious ethical considerations in view of the carcinogenic evidence.
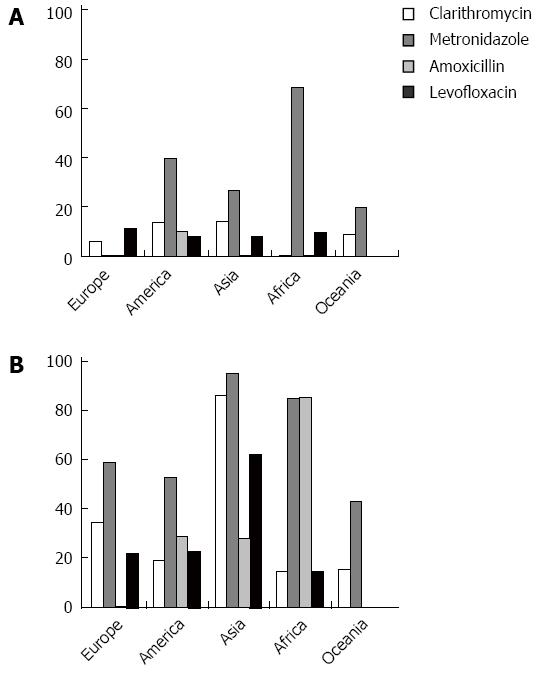
Figure 1 illustrates the simultaneous minimal and maximal resistance rates in five world continents (Europe, America, Asia, Africa, and Oceania). It is evident that clarithromycin shows a good prospect of success in Africa, Oceania, and few Northern European countries (e.g., Norway), whilst in Southern Europe (e.g., Italy, Spain, Portugal, and Greece) and even South Africa, the risk of failure is high. Metronidazole might be very effective in Lithuania, but its use should be strongly discouraged in Italy and Croatia as well as most American, Asian, and African countries. Amoxicillin appears to be a reliable option in many countries, but it is almost ineffective in Iran and Cameroon. Finally, levofloxacin, which has been proposed as an alternative to clarithromycin in the areas where this last key antibiotic shows a high resistance rate, has been shown to be moderately ineffective worldwide, aside from Oceania, thus confirming the rapid trend toward therapeutic failure. Therefore, proposals for wider use could induce a counterproductive effect.
Among the other antibiotics, tetracycline and rifabutin appear to have low resistance rates, even if the use of the latter drug is limited by the regulations of many countries, where it is not indicated in H. pylori infection care, even apart from its high cost. Finally, furazolidone, despite its effectiveness, has been forbidden in many countries.
An obvious point is that, in each country, an ideal specific therapy should be identified. Nevertheless, all the studies on resistances in vitro might lack positive feedback in vivo. In fact, therapy failure may depend on several factors, of both bacterial and host origin. Not infrequently, different factors act simultaneously in reducing antibiotic therapy efficacy in the same patient. Indeed, a poor compliance to the H. pylori eradication regimen is inversely associated with the probability of therapeutic success. Unfortunately, the approved eradication regimens require the combination of 3-4 different drugs in multiple daily doses. Therapy regimen complexity and the onset of side effects are associated with reduced patient compliance. The in vitro activity of various antibiotics is greatly reduced or eliminated in vivo by the very low pH values encountered in gastric juices. This explains the need to include a proton pump inhibitor in H. pylori eradication regimens. However, a significant variability in gastric acid secretion among different subjects has been reported. A small proportion of subjects show a higher basal acid output in association with normal gastrin values. These hyper-secretor subjects probably have a large parietal cell mass and a low eradication rate[127].
These considerations may partially account for the wide discrepancies demonstrated between studies on resistances in vitro and the results of clinical trials in vivo. A final factor which might limit the reliability of in vitro resistance studies is the different methods used for detection. The methods are complex, expensive, and may often fail even in expert hands, and for this reason, their use is predominantly confined to research purposes.
Triple therapy is one of the oldest schemes for H. pylori eradication. In Europe, it was used successfully in the United Kingdom until 5 years ago[128], achieving an eradication rate of 92%. However, it is characterized by an enormous variability, if we consider the poor rate of 50% calculated in a German study in 2011[129]. The possibility of failure was very high in a Turkish study[130], where only 32.7% of eradication occurred. In Asia, a high percentage of eradication was seen in India (82.9%)[131], with a lower percentage in Korea (67.7)[132]. In the American continent, eradication rates range from 78% to 97%[133,134]. Data from Africa are more homogeneous, but the weight of resistances affects the possibility of the eradication achieved in 71%-78.2% of cases, in a multicentric and in a Moroccan study, respectively[135,136]. The only recent available data from Oceania derive from two trials in which rifampicin was used instead of clarithromycin as second-line regimen, which achieved eradication rates of 95% and 96.6%, respectively[137,138].
Quadruple therapy appears to be more effective than triple therapy, if we consider that in United States, Laine et al[139] reported a success rate of 87.7%, and a lower value was seen only in a Canadian study[140] (70.8%). However, in Europe, failures are more frequent: quadruple therapy was effective only in 64.8% of cases in a Greek study[141], despite a success rate of 91% in a British trial[128]. In Asian countries, the rates seem to be even more discouraging, ranging between 47.1% and 89.5% in Turkey[130] and China[142], respectively. No data are available from Africa to the best of our knowledge. Only one Australian study investigated the effectiveness of quadruple therapy, but using a novel combination of a proton pump inhibitor, bismuth subcitrate, rifabutin, and ciprofloxacin as a first-line regimen for patients allergic to penicillin, and achieving an eradication rate of 94.2%[143].
Concomitant therapy is a combination of antibiotics including amoxicillin, metronidazole, clarithromycin, and a proton pump inhibitor (PPI) for a period of five or seven days. It has proven very effective in Japan, where an eradication rate of 98.1%[144] was achieved, but in South Korea the percentage was much lower (63.2%)[145]. In two different European studies, the same author reported the minimal and the maximal eradication rates of concomitant therapy as 85.5% and 95.5%, respectively[146,147]. In a multicentric Southern American study, this therapy achieved only 78.7% eradication[148]. We did not find any results from Africa for this regimen. No further data are available from Oceania.
Sequential therapy is a ten-day therapy that consists of a PPI plus amoxicillin in the first 5 d and a PPI, clarithromycin, and metronidazole in the following 5 d. In Italy it has proven to be very useful compared to other combinations[149]; it achieved eradication rates that range between 97.3% in a pediatric population[150], 97% in an elderly population[151], and 89% in a multicentric study involving more than 1000 patients[83]. The data from other European countries, however, are very poor. A good performance of this therapy was demonstrated in Africa, with a positive outcome ranging between 89.9% and 94.2%[136,152]. However, in South America this scheme appears to be less effective, if we consider the Peruvian percentage of success of 73%[153] and overall rate of 81.1%[148]. In Asia, a good result was achieved in South Korea[154] (92.6% maximum), whilst in China[155] only 78.3% eradicated the bacterium. No data are available from Oceania.
Miscellaneous therapy has been recently introduced and includes sequences of different combinations of antibiotics. The main four studies are from the four continents that show promising results: Colombia 94%[156], Italy 85.7%[157], Iran 92.9%[158], and Taiwan 97.4%[159]. This regimen requires further confirmation of these excellent results, as well as an accurate evaluation of patient compliance owing to the risk of a large number of side effects and consequent drop-outs.
Although resistance reports describe alarming results, the outcomes of therapeutic schemes do not seem to parallel this scenario in most cases, since a success of more than 80% is often reached. Despite some of the factors that may explain this discrepancy between in vitro and in vivo results that have been mentioned previously, it is possible that other factors may elucidate this controversial point: (1) most studies are performed in single centers and include populations selected from geographic areas of irrelevant dimensions. This may be an important handicap for the reproducibility of the therapeutic regimen in other areas; (2) the selection of patients is limited to bacterial positivity, often based on non-invasive tests, and does not take into account some bacterial factors such as the intra-gastric load[160], the possibility of primary resistances (which may even be different in the body and antrum of the same subject)[161], heteroresistance status, and CagA status[162,163]. Finally, the presence of coccoid forms[164] in the stomach may have clinical relevance, due to the potential reactivation of H. pylori in its spiral form following therapy; (3) the results of therapeutic studies may be affected by host factors such as PPI metabolism[165], parietal cell mass[166], and related gastric pH, which is strictly related to antibiotic MIC values, the mucus layer (which affects bacterium/antibiotic contact), the frequent patchy distribution of H. pylori in the stomach, and even its persistence in small areas (e.g., cardiac) after apparently successful eradication; and (4) the already outlined technical problems related to resistance detections in vitro.
The possibility of probiotics interfering with H. pylori gastric colonization has been postulated by many authors and, therefore, many studies are available in the literature about the treatment of infected patients with beneficial bacteria supplementation. However, conflicting data have been obtained.
A review of available data showed that clinical trials can be divided into two groups: those using probiotics in association with antibiotic therapy and those using probiotics alone. In the first group, the efficacy of a single strain of probiotics associated with antibiotic triple or quadruple therapy generally resulted in a decrease of side effects such as diarrhea, bloating, nausea, and taste disturbances during treatment[167,168], as well as an improvement of the eradication rate. Some studies have tested the association of a multi-strain probiotic mixture associated with antibiotic therapy. Two of these[169,170] showed a reduction in the side effects of antibiotic therapy and a higher eradication rate than that obtained with a single strain, whilst the third study[171] did not obtain any significant result.
Among the second group of clinical trials (only probiotics) we found that most studies tested a single probiotic strain, especially Lactobacillus species, obtaining a H. pylori load reduction as expressed by the urea breath test delta value. Experience by our group confirmed this finding after oral administration of Lactobacillus reuteri ATC 55730, not only with a delta value reduction, but also with a semiquantitative fecal antigen decrease[172].
Recently, Szajewska et al[173] reported a very interesting meta-analysis of the effects of Saccharomyces boulardii supplementation in standard triple therapy, showing a significant effect in both increasing the eradication rate and reducing side effects, in a total of 1307 patients from five randomized controlled trials.
Therapeutic management of H. pylori remains an unsolved issue. Indeed, no therapy regimen is able to cure the infection in all treated patients, and a definite number remain infected despite several consecutive standard therapies. This therapeutic failure is often considered to be the consequence of incorrect treatment rather than treatment limitation, since this appears unacceptable in the antibiotic era. However, no clinical trial has reported an eradication rate of 100% to the best of our knowledge.
Therapeutic failures are attributed to increasing antibiotic resistance. However, this phenomenon has been widely studied and the results show controversial findings even in the same geographic area. For each world continent, minimal and maximal values of resistance to different antibiotics have been reported for different countries, although some regimens appear to be almost unknown in some areas.
Another enigma is the outcomes of several different therapeutic schemes, which often do not parallel in vitro findings even in the same areas. Moreover, some schemes are limited by their use in very small geographic districts. Finally, not all therapeutic trials have considered bacterial and host factors affecting the therapeutic outcome.
In conclusion, the “ideal therapy”, paradoxically, appears to be a “utopia”, despite the unprecedented volume of studies in the field and the real breakthrough in medical practice made by the discovery and the treatment of H. pylori. A key point could be the possibility, in the near future, to group in vivo and in vitro studies by geographic areas in order to identify the best therapy, which is certainly related to the local habitat. Indeed, the ample discrepancies observed in the different areas do not encourage the development of therapeutic guidelines that could be valid worldwide. The main challenge for the future might be identifying a successful solution for overcoming antibiotic resistances and, in this context, geography must be considered a relevant matter.
P- Reviewers: Abenavoli L, Chuah SK, Day AS, Kim BW S- Editor: Gou SX L- Editor: Rutherford A E- Editor: Liu XMs
| 1. | Dore MP, Fanciulli G, Tomasi PA, Realdi G, Delitala G, Graham DY, Malaty HM. Gastrointestinal symptoms and Helicobacter pylori infection in school-age children residing in Porto Torres, Sardinia, Italy. Helicobacter. 2012;17:369-373. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 2. | Bastos J, Peleteiro B, Barros R, Alves L, Severo M, de Fátima Pina M, Pinto H, Carvalho S, Marinho A, Guimarães JT. Sociodemographic Determinants of Prevalence and Incidence of Helicobacter pylori Infection in Portuguese Adults. Helicobacter. 2013;18:413-422. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 58] [Cited by in F6Publishing: 61] [Article Influence: 5.5] [Reference Citation Analysis (1)] |
| 3. | Thjodleifsson B, Asbjörnsdottir H, Sigurjonsdottir RB, Gíslason D, Olafsson I, Cook E, Gíslason T, Jogi R, Janson C. Seroprevalence of Helicobacter pylori and cagA antibodies in Iceland, Estonia and Sweden. Scand J Infect Dis. 2007;39:683-689. [PubMed] [Cited in This Article: ] |
| 4. | Bakkevold KE. Time trends in incidence of peptic ulcer bleeding and associated risk factors in Norway 1985-2008. Clin Exp Gastroenterol. 2010;3:71-77. [PubMed] [Cited in This Article: ] |
| 5. | Bureš J, Kopáčová M, Koupil I, Seifert B, Skodová Fendrichová M, Spirková J, Voříšek V, Rejchrt S, Douda T, Král N. Significant decrease in prevalence of Helicobacter pylori in the Czech Republic. World J Gastroenterol. 2012;18:4412-4418. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 32] [Cited by in F6Publishing: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 6. | Svarval' AV, Ferman RS, Zhebrun AB. [Prevalence of Helicobater pylori infection among population of Northwestern federal district of Russian Federation]. Zh Mikrobiol Epidemiol Immunobiol. 2011;84-88. [PubMed] [Cited in This Article: ] |
| 7. | Patterson T, Straten E, Jimenez S. The prevalence of Helicobacter pylori antibody in different age groups in Central Texas. Clin Lab Sci. 2012;25:102-106. [PubMed] [Cited in This Article: ] |
| 8. | Porras C, Nodora J, Sexton R, Ferreccio C, Jimenez S, Dominguez RL, Cook P, Anderson G, Morgan DR, Baker LH. Epidemiology of Helicobacter pylori infection in six Latin American countries (SWOG Trial S0701). Cancer Causes Control. 2013;24:209-215. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 75] [Cited by in F6Publishing: 82] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 9. | Hanafi MI, Mohamed AM. Helicobacter pylori infection: seroprevalence and predictors among healthy individuals in Al Madinah, Saudi Arabia. J Egypt Public Health Assoc. 2013;88:40-45. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 10. | Baik SJ, Yi SY, Park HS, Park BH. Seroprevalence of Helicobacter pylori in female Vietnamese immigrants to Korea. World J Gastroenterol. 2012;18:517-521. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 7] [Cited by in F6Publishing: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 11. | Etukudo OM, Ikpeme EE, Ekanem EE. Seroepidemiology of Helicobacter pylori infection among children seen in a tertiary hospital in Uyo, southern Nigeria. Pan Afr Med J. 2012;12:39. [PubMed] [Cited in This Article: ] |
| 12. | Benajah DA, Lahbabi M, Alaoui S, El Rhazi K, El Abkari M, Nejjari C, Amarti A, Bennani B, Mahmoud M, Ibrahimi SA. Prevalence of Helicobacter pylori and its recurrence after successful eradication in a developing nation (Morocco). Clin Res Hepatol Gastroenterol. 2013;37:519-526. [PubMed] [Cited in This Article: ] |
| 13. | Pandeya N, Whiteman DC. Prevalence and determinants of Helicobacter pylori sero-positivity in the Australian adult community. J Gastroenterol Hepatol. 2011;26:1283-1289. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 14. | Fawcett JP, Barbezat GO, Poulton R, Milne BJ, Xia HH, Talley NJ. Helicobacter pylori serology in a birth cohort of New Zealanders from age 11 to 26. World J Gastroenterol. 2005;11:3273-3276. [PubMed] [Cited in This Article: ] |
| 15. | Boyanova L. Prevalence of multidrug-resistant Helicobacter pylori in Bulgaria. J Med Microbiol. 2009;58:930-935. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 36] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 16. | Mégraud F, Lehours P. Helicobacter pylori detection and antimicrobial susceptibility testing. Clin Microbiol Rev. 2007;20:280-322. [PubMed] [Cited in This Article: ] |
| 17. | Mégraud F. H pylori antibiotic resistance: prevalence, importance, and advances in testing. Gut. 2004;53:1374-1384. [PubMed] [Cited in This Article: ] |
| 18. | Cerqueira L, Fernandes RM, Ferreira RM, Oleastro M, Carneiro F, Brandão C, Pimentel-Nunes P, Dinis-Ribeiro M, Figueiredo C, Keevil CW. Validation of a fluorescence in situ hybridization method using peptide nucleic acid probes for detection of Helicobacter pylori clarithromycin resistance in gastric biopsy specimens. J Clin Microbiol. 2013;51:1887-1893. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 44] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | Schmitt BH, Regner M, Mangold KA, Thomson RB, Kaul KL. PCR detection of clarithromycin-susceptible and -resistant Helicobacter pylori from formalin-fixed, paraffin-embedded gastric biopsies. Mod Pathol. 2013;26:1222-1227. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 20. | Rimbara E, Sasatsu M, Graham DY. PCR detection of Helicobacter pylori in clinical samples. Methods Mol Biol. 2013;943:279-287. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 37] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 21. | Vacher S, Ménard A, Bernard E, Mégraud F. PCR-restriction fragment length polymorphism analysis for detection of point mutations associated with macrolide resistance in Campylobacter spp. Antimicrob Agents Chemother. 2003;47:1125-1128. [PubMed] [Cited in This Article: ] |
| 22. | Monno R, Giorgio F, Carmine P, Soleo L, Cinquepalmi V, Ierardi E. Helicobacter pylori clarithromycin resistance detected by Etest and TaqMan real-time polymerase chain reaction: a comparative study. APMIS. 2012;120:712-717. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 23. | Hao H, Yuan Z, Shen Z, Han J, Sahin O, Liu P, Zhang Q. Mutational and transcriptomic changes involved in the development of macrolide resistance in Campylobacter jejuni. Antimicrob Agents Chemother. 2013;57:1369-1378. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 24. | Hwang TJ, Kim N, Kim HB, Lee BH, Nam RH, Park JH, Lee MK, Park YS, Lee DH, Jung HC. Change in antibiotic resistance of Helicobacter pylori strains and the effect of A2143G point mutation of 23S rRNA on the eradication of H. pylori in a single center of Korea. J Clin Gastroenterol. 2010;44:536-543. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 103] [Cited by in F6Publishing: 110] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 25. | Matsumura M, Hikiba Y, Ogura K, Togo G, Tsukuda I, Ushikawa K, Shiratori Y, Omata M. Rapid detection of mutations in the 23S rRNA gene of Helicobacter pylori that confers resistance to clarithromycin treatment to the bacterium. J Clin Microbiol. 2001;39:691-695. [PubMed] [Cited in This Article: ] |
| 26. | Oleastro M, Ménard A, Santos A, Lamouliatte H, Monteiro L, Barthélémy P, Mégraud F. Real-time PCR assay for rapid and accurate detection of point mutations conferring resistance to clarithromycin in Helicobacter pylori. J Clin Microbiol. 2003;41:397-402. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 155] [Cited by in F6Publishing: 152] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 27. | De Francesco V, Margiotta M, Zullo A, Hassan C, Troiani L, Burattini O, Stella F, Di Leo A, Russo F, Marangi S. Clarithromycin-resistant genotypes and eradication of Helicobacter pylori. Ann Intern Med. 2006;144:94-100. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 138] [Cited by in F6Publishing: 155] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 28. | Giorgio F, Principi M, De Francesco V, Zullo A, Losurdo G, Di Leo A, Ierardi E. Primary clarithromycin resistance to Helicobacter pylori: Is this the main reason for triple therapy failure? World J Gastrointest Pathophysiol. 2013;4:43-46. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 26] [Cited by in F6Publishing: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 29. | Barile KA, Silva AL, Xavier JN, Assumpção MB, Corvelo TC. Characterization of 23S rRNA domain V mutations in gastric biopsy patients from the eastern Amazon. Mem Inst Oswaldo Cruz. 2010;105:314-317. [PubMed] [Cited in This Article: ] |
| 30. | Murakami K, Furuta T, Ando T, Nakajima T, Inui Y, Oshima T, Tomita T, Mabe K, Sasaki M, Suganuma T. Multi-center randomized controlled study to establish the standard third-line regimen for Helicobacter pylori eradication in Japan. J Gastroenterol. 2013;48:1128-1135. [PubMed] [Cited in This Article: ] |
| 31. | Horiki N, Omata F, Uemura M, Suzuki S, Ishii N, Iizuka Y, Fukuda K, Fujita Y, Katsurahara M, Ito T. Annual change of primary resistance to clarithromycin among Helicobacter pylori isolates from 1996 through 2008 in Japan. Helicobacter. 2009;14:86-90. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 70] [Cited by in F6Publishing: 67] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 32. | Kato S, Fujimura S. Primary antimicrobial resistance of Helicobacter pylori in children during the past 9 years. Pediatr Int. 2010;52:187-190. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 32] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 33. | Kobayashi I, Murakami K, Kato M, Kato S, Azuma T, Takahashi S, Uemura N, Katsuyama T, Fukuda Y, Haruma K. Changing antimicrobial susceptibility epidemiology of Helicobacter pylori strains in Japan between 2002 and 2005. J Clin Microbiol. 2007;45:4006-4010. [PubMed] [Cited in This Article: ] |
| 34. | Su P, Li Y, Li H, Zhang J, Lin L, Wang Q, Guo F, Ji Z, Mao J, Tang W. Antibiotic resistance of Helicobacter pylori isolated in the Southeast Coastal Region of China. Helicobacter. 2013;18:274-279. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 106] [Cited by in F6Publishing: 108] [Article Influence: 9.8] [Reference Citation Analysis (1)] |
| 35. | Gao W, Cheng H, Hu F, Li J, Wang L, Yang G, Xu L, Zheng X. The evolution of Helicobacter pylori antibiotics resistance over 10 years in Beijing, China. Helicobacter. 2010;15:460-466. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 100] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 36. | Binh TT, Shiota S, Nguyen LT, Ho DD, Hoang HH, Ta L, Trinh DT, Fujioka T, Yamaoka Y. The incidence of primary antibiotic resistance of Helicobacter pylori in Vietnam. J Clin Gastroenterol. 2013;47:233-238. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 34] [Cited by in F6Publishing: 45] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 37. | Goh KL, Navaratnam P. High Helicobacter pylori resistance to metronidazole but zero or low resistance to clarithromycin, levofloxacin, and other antibiotics in Malaysia. Helicobacter. 2011;16:241-245. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Milani M, Ghotaslou R, Akhi MT, Nahaei MR, Hasani A, Somi MH, Rafeey M, Sharifi Y. The status of antimicrobial resistance of Helicobacter pylori in Eastern Azerbaijan, Iran: comparative study according to demographics. J Infect Chemother. 2012;18:848-852. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 42] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 39. | Rajper S, Khan E, Ahmad Z, Alam SM, Akbar A, Hasan R. Macrolide and fluoroquinolone resistance in Helicobacter pylori isolates: an experience at a tertiary care centre in Pakistan. J Pak Med Assoc. 2012;62:1140-1144. [PubMed] [Cited in This Article: ] |
| 40. | Zendedel A, Moradimoghadam F, Almasi V, Zivarifar H. Antibiotic resistance of Helicobacter pylori in Mashhad, Iran. J Pak Med Assoc. 2013;63:336-339. [PubMed] [Cited in This Article: ] |
| 41. | Trespalacios AA, Otero W, Caminos JE, Mercado MM, Avila J, Rosero LE, Arévalo A, Poutou-Piñales RA, Graham DY. Phenotypic and genotypic analysis of clarithromycin-resistant Helicobacter pylori from Bogotá D.C., Colombia. J Microbiol. 2013;51:448-452. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 42. | Ogata SK, Godoy AP, da Silva Patricio FR, Kawakami E. High Helicobacter pylori resistance to metronidazole and clarithromycin in Brazilian children and adolescents. J Pediatr Gastroenterol Nutr. 2013;56:645-648. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 35] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 43. | Salazar CO, Cardenas VM, Reddy RK, Dominguez DC, Snyder LK, Graham DY. Greater than 95% success with 14-day bismuth quadruple anti- Helicobacter pylori therapy: a pilot study in US Hispanics. Helicobacter. 2012;17:382-390. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 44. | Basu PP, Rayapudi K, Pacana T, Shah NJ, Krishnaswamy N, Flynn M. A randomized study comparing levofloxacin, omeprazole, nitazoxanide, and doxycycline versus triple therapy for the eradication of Helicobacter pylori. Am J Gastroenterol. 2011;106:1970-1975. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 67] [Cited by in F6Publishing: 75] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 45. | Graham DY. Clarithromycin for treatment of Helicobacter pylori infections. Eur J Gastroenterol Hepatol. 1995;7 Suppl 1:S55-S58. [PubMed] [Cited in This Article: ] |
| 46. | Koivisto TT, Rautelin HI, Voutilainen ME, Niemelä SE, Heikkinen M, Sipponen PI, Färkkilä MA. Primary Helicobacter pylori resistance to metronidazole and clarithromycin in the Finnish population. Aliment Pharmacol Ther. 2004;19:1009-1017. [PubMed] [Cited in This Article: ] |
| 47. | Storskrubb T, Aro P, Ronkainen J, Wreiber K, Nyhlin H, Bolling-Sternevald E, Talley NJ, Engstrand L, Agréus L. Antimicrobial susceptibility of Helicobacter pylori strains in a random adult Swedish population. Helicobacter. 2006;11:224-230. [PubMed] [Cited in This Article: ] |
| 48. | Selgrad M, Meissle J, Bornschein J, Kandulski A, Langner C, Varbanova M, Wex T, Tammer I, Schlüter D, Malfertheiner P. Antibiotic susceptibility of Helicobacter pylori in central Germany and its relationship with the number of eradication therapies. Eur J Gastroenterol Hepatol. 2013;25:1257-1260. [PubMed] [Cited in This Article: ] |
| 49. | Larsen AL, Ragnhildstveit E, Moayeri B, Eliassen L, Melby KK. Resistance rates of metronidazole and other antibacterials in Helicobacter pylori from previously untreated patients in Norway. APMIS. 2013;121:353-358. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 50. | Glupczynski Y, Mégraud F, Lopez-Brea M, Andersen LP. European multicentre survey of in vitro antimicrobial resistance in Helicobacter pylori. Eur J Clin Microbiol Infect Dis. 2001;20:820-823. [PubMed] [Cited in This Article: ] |
| 51. | Perri F, Villani MR, Festa V, Quitadamo M, Andriulli A. Predictors of failure of Helicobacter pylori eradication with the standard ‘Maastricht triple therapy’. Aliment Pharmacol Ther. 2001;15:1023-1029. [PubMed] [Cited in This Article: ] |
| 52. | Pilotto A, Rassu M, Leandro G, Franceschi M, Di Mario F. Prevalence of Helicobacter pylori resistance to antibiotics in Northeast Italy: a multicentre study. GISU. Interdisciplinary Group for the Study of Ulcer. Dig Liver Dis. 2000;32:763-768. [PubMed] [Cited in This Article: ] |
| 53. | Savarino V, Zentilin P, Pivari M, Bisso G, Raffaella Mele M, Bilardi C, Borro P, Dulbecco P, Tessieri L, Mansi C. The impact of antibiotic resistance on the efficacy of three 7-d regimens against Helicobacter pylori. Aliment Pharmacol Ther. 2000;14:893-900. [PubMed] [Cited in This Article: ] |
| 54. | De Francesco V, Margiotta M, Zullo A, Hassan C, Giorgio F, Burattini O, Stoppino G, Cea U, Pace A, Zotti M. Prevalence of primary clarithromycin resistance in Helicobacter pylori strains over a 15 year period in Italy. J Antimicrob Chemother. 2007;59:783-785. [PubMed] [Cited in This Article: ] |
| 55. | De Francesco V, Margiotta M, Zullo A, Hassan C, Valle ND, Burattini O, D’Angelo R, Stoppino G, Cea U, Giorgio F. Claritromycin resistance and Helicobacter pylori genotypes in Italy. J Microbiol. 2006;44:660-664. [PubMed] [Cited in This Article: ] |
| 56. | De Francesco V, Giorgio F, Ierardi E, Zotti M, Neri M, Milano A, Varasano V, Luzza F, Suraci E, Marmo R. Primary clarithromycin resistance in Helicobacter pylori: the Multicentric Italian Clarithromycin Resistance Observational (MICRO) study. J Gastrointestin Liver Dis. 2011;20:235-239. [PubMed] [Cited in This Article: ] |
| 57. | Georgopoulos SD, Xirouchakis E, Martinez-Gonzalez B, Sgouras DN, Spiliadi C, Mentis AF, Laoudi F. Clinical Evaluation of a Ten-Day Regimen with Esomeprazole, Metronidazole, Amoxicillin, and Clarithromycin for the Eradication of Helicobacter pylori in a High Clarithromycin Resistance Area. Helicobacter. 2013;18:459-467. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 47] [Cited by in F6Publishing: 48] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 58. | Molina-Infante J, Gisbert JP. [Update on the efficacy of triple therapy for Helicobacter pylori infection and clarithromycin resistance rates in Spain (2007-2012)]. Gastroenterol Hepatol. 2013;36:375-381. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 59. | Mégraud F. Current recommendations for Helicobacter pylori therapies in a world of evolving resistance. Gut Microbes. 2013;4:Epub ahead of print. [PubMed] [Cited in This Article: ] |
| 60. | Oleastro M, Cabral J, Ramalho PM, Lemos PS, Paixão E, Benoliel J, Santos A, Lopes AI. Primary antibiotic resistance of Helicobacter pylori strains isolated from Portuguese children: a prospective multicentre study over a 10 year period. J Antimicrob Chemother. 2011;66:2308-2311. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 38] [Cited by in F6Publishing: 44] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 61. | Boyanova L, Ilieva J, Gergova G, Spassova Z, Nikolov R, Davidkov L, Evstatiev I, Kamburov V, Katsarov N, Mitov I. Evaluation of clinical and socio-demographic risk factors for antibacterial resistance of Helicobacter pylori in Bulgaria. J Med Microbiol. 2009;58:94-100. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 12] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 62. | Boyanova L, Mitov I. Geographic map and evolution of primary Helicobacter pylori resistance to antibacterial agents. Expert Rev Anti Infect Ther. 2010;8:59-70. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 78] [Cited by in F6Publishing: 84] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 63. | Karczewska E, Klesiewicz K, Skiba I, Wojtas-Bonior I, Sito E, Czajecki K, Zwolińska-Wcisło M, Budak A. Variability in Prevalence of Helicobacter pylori Strains Resistant to Clarithromycin and Levofloxacin in Southern Poland. Gastroenterol Res Pract. 2012;2012:418010. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 13] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 64. | Kupcinskas L, Rasmussen L, Jonaitis L, Kiudelis G, Jørgensen M, Urbonaviciene N, Tamosiunas V, Kupcinskas J, Miciuleviciene J, Kadusevicius E. Evolution of Helicobacter pylori susceptibility to antibiotics during a 10-year period in Lithuania. APMIS. 2013;121:431-436. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 18] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 65. | Rerksuppaphol S, Hardikar W, Midolo PD, Ward P. Antimicrobial resistance in Helicobacter pylori isolates from children. J Paediatr Child Health. 2003;39:332-335. [PubMed] [Cited in This Article: ] |
| 66. | Grove DI, Koutsouridis G. Increasing resistance of Helicobacter pylori to clarithromycin: is the horse bolting? Pathology. 2002;34:71-73. [PubMed] [Cited in This Article: ] |
| 67. | Secka O, Berg DE, Antonio M, Corrah T, Tapgun M, Walton R, Thomas V, Galano JJ, Sancho J, Adegbola RA. Antimicrobial susceptibility and resistance patterns among Helicobacter pylori strains from The Gambia, West Africa. Antimicrob Agents Chemother. 2013;57:1231-1237. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 36] [Cited by in F6Publishing: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 68. | Seck A, Burucoa C, Dia D, Mbengue M, Onambele M, Raymond J, Breurec S. Primary antibiotic resistance and associated mechanisms in Helicobacter pylori isolates from Senegalese patients. Ann Clin Microbiol Antimicrob. 2013;12:3. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 39] [Cited by in F6Publishing: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 69. | Tanih NF, Ndip RN. Molecular Detection of Antibiotic Resistance in South African Isolates of Helicobacter pylori. Gastroenterol Res Pract. 2013;2013:259457. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 22] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 70. | Edwards DI. Mechanism of antimicrobial action of metronidazole. J Antimicrob Chemother. 1979;5:499-502. [PubMed] [Cited in This Article: ] |
| 71. | Mendz GL, Mégraud F. Is the molecular basis of metronidazole resistance in microaerophilic organisms understood? Trends Microbiol. 2002;10:370-375. [PubMed] [Cited in This Article: ] |
| 72. | Chisholm SA, Owen RJ. Mutations in Helicobacter pylori rdxA gene sequences may not contribute to metronidazole resistance. J Antimicrob Chemother. 2003;51:995-999. [PubMed] [Cited in This Article: ] |
| 73. | Marais A, Bilardi C, Cantet F, Mendz GL, Mégraud F. Characterization of the genes rdxA and frxA involved in metronidazole resistance in Helicobacter pylori. Res Microbiol. 2003;154:137-144. [PubMed] [Cited in This Article: ] |
| 74. | Seo JH, Jun JS, Yeom JS, Park JS, Youn HS, Ko GH, Baik SC, Lee WK, Cho MJ, Rhee KH. Changing pattern of antibiotic resistance of Helicobacter pylori in children during 20 years in Jinju, South Korea. Pediatr Int. 2013;55:332-336. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 26] [Cited by in F6Publishing: 30] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 75. | Kim JM, Kim JS, Jung HC, Kim N, Kim YJ, Song IS. Distribution of antibiotic MICs for Helicobacter pylori strains over a 16-year period in patients from Seoul, South Korea. Antimicrob Agents Chemother. 2004;48:4843-4847. [PubMed] [Cited in This Article: ] |
| 76. | Torres J, Camorlinga-Ponce M, Pérez-Pérez G, Madrazo-De la Garza A, Dehesa M, González-Valencia G, Muñoz O. Increasing multidrug resistance in Helicobacter pylori strains isolated from children and adults in Mexico. J Clin Microbiol. 2001;39:2677-2680. [PubMed] [Cited in This Article: ] |
| 77. | Marie MAM. Patterns of Helicobacter pylori resistance to metronidazole, clarithromycin and amoxicillin in Saudi Arabia. J Bacteriol Virol. 2008;38:173-178. [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 78. | Loffeld RJ, Werdmuller BF. Changes in Antibiotic Susceptibility of Helicobacter pylori in the Course of Eight Years in the Zaanstreek Region in The Netherlands. Gastroenterol Res Pract. 2013;2013:625937. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3] [Cited by in F6Publishing: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 79. | Elviss NC, Owen RJ, Xerry J, Walker AM, Davies K. Helicobacter pylori antibiotic resistance patterns and genotypes in adult dyspeptic patients from a regional population in North Wales. J Antimicrob Chemother. 2004;54:435-440. [PubMed] [Cited in This Article: ] |
| 80. | Hojsak I, Kos T, Dumančić J, Mišak Z, Jadrešin O, Jaklin Kekez A, Lukić Grlić A, Kolaček S. Antibiotic resistance of Helicobacter pylori in pediatric patients -- 10 years’ experience. Eur J Pediatr. 2012;171:1325-1330. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 17] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 81. | De Francesco V, Giorgio F, Hassan C, Manes G, Vannella L, Panella C, Ierardi E, Zullo A. Worldwide H. pylori antibiotic resistance: a systematic review. J Gastrointestin Liver Dis. 2010;19:409-414. [PubMed] [Cited in This Article: ] |
| 82. | Zullo A, Perna F, Hassan C, Ricci C, Saracino I, Morini S, Vaira D. Primary antibiotic resistance in Helicobacter pylori strains isolated in northern and central Italy. Aliment Pharmacol Ther. 2007;25:1429-1434. [PubMed] [Cited in This Article: ] |
| 83. | Vaira D, Zullo A, Vakil N, Gatta L, Ricci C, Perna F, Hassan C, Bernabucci V, Tampieri A, Morini S. Sequential therapy versus standard triple-drug therapy for Helicobacter pylori eradication: a randomized trial. Ann Intern Med. 2007;146:556-563. [PubMed] [Cited in This Article: ] |
| 84. | Berrutti M, Pellicano R, Astegiano M, Smedile A, Saracco G, Morgando A, De Angelis C, Repici A, Fagoonee S, Leone N. Helicobacter pylori eradication: metronidazole or tinidazole? Data from Turin, Italy. Minerva Gastroenterol Dietol. 2008;54:355-358. [PubMed] [Cited in This Article: ] |
| 85. | Saracino IM, Zullo A, Holton J, Castelli V, Fiorini G, Zaccaro C, Ridola L, Ricci C, Gatta L, Vaira D. High prevalence of primary antibiotic resistance in Helicobacter pylori isolates in Italy. J Gastrointestin Liver Dis. 2012;21:363-365. [PubMed] [Cited in This Article: ] |
| 86. | Mollison LC, Stingemore N, Wake RA, Cullen DJ, McGechie DB. Antibiotic resistance in Helicobacter pylori. Med J Aust. 2000;173:521-523. [PubMed] [Cited in This Article: ] |
| 87. | Ahmed D, Brooks H, McConnell M, Barbezat G. Antibiotic resistance in Helicobacter pylori: is it a problem in New Zealand? N Z Med J. 2004;117:U1022. [PubMed] [Cited in This Article: ] |
| 88. | Malfertheiner P, Megraud F, O’Morain CA, Atherton J, Axon AT, Bazzoli F, Gensini GF, Gisbert JP, Graham DY, Rokkas T. Management of Helicobacter pylori infection--the Maastricht IV/ Florence Consensus Report. Gut. 2012;61:646-664. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1541] [Cited by in F6Publishing: 1494] [Article Influence: 124.5] [Reference Citation Analysis (3)] |
| 89. | Francesco VD, Zullo A, Hassan C, Giorgio F, Rosania R, Ierardi E. Mechanisms of Helicobacter pylori antibiotic resistance: An updated appraisal. World J Gastrointest Pathophysiol. 2011;2:35-41. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 92] [Cited by in F6Publishing: 91] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 90. | Bago J, Halle ZB, Strinić D, Kućisec N, Jandrić D, Bevanda M, Tomić M, Bilić A. The impact of primary antibiotic resistance on the efficacy of ranitidine bismuth citrate- vs. omeprazole-based one-week triple therapies in H. pylori eradication--a randomised controlled trial. Wien Klin Wochenschr. 2002;114:448-453. [PubMed] [Cited in This Article: ] |
| 91. | Mégraud F. Surveillance de la résistance de Helicobacter pylori aux antibiotiques. St Maurice, France: Institut de Veille Sanitaire 2003; 327-329. [Cited in This Article: ] |
| 92. | Wolle K, Leodolter A, Malfertheiner P, König W. Antibiotic susceptibility of Helicobacter pylori in Germany: stable primary resistance from 1995 to 2000. J Med Microbiol. 2002;51:705-709. [PubMed] [Cited in This Article: ] |
| 93. | Debets-Ossenkopp YJ, Herscheid AJ, Pot RG, Kuipers EJ, Kusters JG, Vandenbroucke-Grauls CM. Prevalence of Helicobacter pylori resistance to metronidazole, clarithromycin, amoxycillin, tetracycline and trovafloxacin in The Netherlands. J Antimicrob Chemother. 1999;43:511-515. [PubMed] [Cited in This Article: ] |
| 94. | Cabrita J, Oleastro M, Matos R, Manhente A, Cabral J, Barros R, Lopes AI, Ramalho P, Neves BC, Guerreiro AS. Features and trends in Helicobacter pylori antibiotic resistance in Lisbon area, Portugal (1990-1999). J Antimicrob Chemother. 2000;46:1029-1031. [PubMed] [Cited in This Article: ] |
| 95. | Cuchí Burgos E, Forné Bardera M, Quintana Riera S, Lite Lite J, Garau Alemany J. [Evolution of the sensitivity of 235 strains of Helicobacter pylori from 1995 to 1998 and impact of antibiotic treatment]. Enferm Infecc Microbiol Clin. 2002;20:157-160. [PubMed] [Cited in This Article: ] |
| 96. | Mégraud F, Lehn N, Lind T, Bayerdörffer E, O’Morain C, Spiller R, Unge P, van Zanten SV, Wrangstadh M, Burman CF. Antimicrobial susceptibility testing of Helicobacter pylori in a large multicenter trial: the MACH 2 study. Antimicrob Agents Chemother. 1999;43:2747-2752. [PubMed] [Cited in This Article: ] |
| 97. | Toracchio S, Marzio L. Primary and secondary antibiotic resistance of Helicobacter pylori strains isolated in central Italy during the years 1998-2002. Dig Liver Dis. 2003;35:541-545. [PubMed] [Cited in This Article: ] |
| 98. | Parsons HK, Carter MJ, Sanders DS, Winstanley T, Lobo AJ. Helicobacter pylori antimicrobial resistance in the United Kingdom: the effect of age, sex and socio-economic status. Aliment Pharmacol Ther. 2001;15:1473-1478. [PubMed] [Cited in This Article: ] |
| 99. | Teare L, Peters T, Saverymuttu S, Owen R, Tiwari I. Antibiotic resistance in Helicobacter pylori. Lancet. 1999;353:242. [PubMed] [Cited in This Article: ] |
| 100. | Nelson JM, Chiller TM, Powers JH, Angulo FJ. Fluoroquinolone-resistant Campylobacter species and the withdrawal of fluoroquinolones from use in poultry: a public health success story. Clin Infect Dis. 2007;44:977-980. [PubMed] [Cited in This Article: ] |
| 101. | Kawahara S. [Chemotherapeutic agents under study]. Nihon Rinsho. 1998;56:3096-3099. [PubMed] [Cited in This Article: ] |
| 102. | Drlica K, Zhao X. DNA gyrase, topoisomerase IV, and the 4-quinolones. Microbiol Mol Biol Rev. 1997;61:377-392. [PubMed] [Cited in This Article: ] |
| 103. | Moore RA, Beckthold B, Wong S, Kureishi A, Bryan LE. Nucleotide sequence of the gyrA gene and characterization of ciprofloxacin-resistant mutants of Helicobacter pylori. Antimicrob Agents Chemother. 1995;39:107-111. [PubMed] [Cited in This Article: ] |
| 104. | Robicsek A, Jacoby GA, Hooper DC. The worldwide emergence of plasmid-mediated quinolone resistance. Lancet Infect Dis. 2006;6:629-640. [PubMed] [Cited in This Article: ] |
| 105. | Megraud F, Coenen S, Versporten A, Kist M, Lopez-Brea M, Hirschl AM, Andersen LP, Goossens H, Glupczynski Y. Helicobacter pylori resistance to antibiotics in Europe and its relationship to antibiotic consumption. Gut. 2013;62:34-42. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 588] [Cited by in F6Publishing: 607] [Article Influence: 55.2] [Reference Citation Analysis (2)] |
| 106. | O'Connor A, Taneike I, Nami A, Fitzgerald N, Ryan B, Breslin N, O’Connor H, McNamara D, Murphy P, O’Morain C. Helicobacter pylori resistance rates for levofloxacin, tetracycline and rifabutin among Irish isolates at a reference centre. Ir J Med Sci. 2013;182:693-695. [PubMed] [Cited in This Article: ] |
| 107. | Wueppenhorst N, Stueger HP, Kist M, Glocker EO. High secondary resistance to quinolones in German Helicobacter pylori clinical isolates. J Antimicrob Chemother. 2013;68:1562-1566. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 37] [Cited by in F6Publishing: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 108. | Tankovic J, Lascols C, Sculo Q, Petit JC, Soussy CJ. Single and double mutations in gyrA but not in gyrB are associated with low- and high-level fluoroquinolone resistance in Helicobacter pylori. Antimicrob Agents Chemother. 2003;47:3942-3944. [PubMed] [Cited in This Article: ] |
| 109. | Monno R, Capolongo C, Giorgio F, Losurdo G, Di Leo A, Ierardi E. An overview of antibiotic phenotypic resistances to Helicobacter pylori (Hp) in Southern Italy: may tigecycline be the “drug of the future?”. Madrid: Proceedings European Helicobacter Study Group Meeting 2013; 12-14 (abstract A156). [Cited in This Article: ] |
| 110. | Tveit AH, Bruce MG, Bruden DL, Morris J, Reasonover A, Hurlburt DA, Hennessy TW, McMahon B. Alaska sentinel surveillance study of Helicobacter pylori isolates from Alaska Native persons from 2000 to 2008. J Clin Microbiol. 2011;49:3638-3643. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 30] [Cited by in F6Publishing: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 111. | Eisig JN, Silva FM, Barbuti RC, Navarro-Rodriguez T, Moraes-Filho JP, Pedrazzoli Jr J. Helicobacter pylori antibiotic resistance in Brazil: clarithromycin is still a good option. Arq Gastroenterol. 2011;48:261-264. [PubMed] [Cited in This Article: ] |
| 112. | Ling TK, Leung WK, Lee CC, Ng EK, Yung MY, Chung SS, Sung JJ, Cheng AF. The antimicrobial susceptibility of Helicobacter pylori in Hong Kong (1997-2001). Helicobacter. 2002;7:327-328. [PubMed] [Cited in This Article: ] |
| 113. | Kim JJ, Reddy R, Lee M, Kim JG, El-Zaatari FA, Osato MS, Graham DY, Kwon DH. Analysis of metronidazole, clarithromycin and tetracycline resistance of Helicobacter pylori isolates from Korea. J Antimicrob Chemother. 2001;47:459-461. [PubMed] [Cited in This Article: ] |
| 114. | Trieber CA, Taylor DE. Mutations in the 16S rRNA genes of Helicobacter pylori mediate resistance to tetracycline. J Bacteriol. 2002;184:2131-2140. [PubMed] [Cited in This Article: ] |
| 115. | Gerrits MM, de Zoete MR, Arents NL, Kuipers EJ, Kusters JG. 16S rRNA mutation-mediated tetracycline resistance in Helicobacter pylori. Antimicrob Agents Chemother. 2002;46:2996-3000. [PubMed] [Cited in This Article: ] |
| 116. | Pinheiro M, Pereira-Leite C, Arêde M, Nunes C, Caio JM, Moiteiro C, Giner-Casares JJ, Lúcio M, Brezesinski G, Camacho L. Evaluation of the structure-activity relationship of rifabutin and analogs: a drug-membrane study. Chemphyschem. 2013;14:2808-2816. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 117. | Heep M, Beck D, Bayerdörffer E, Lehn N. Rifampin and rifabutin resistance mechanism in Helicobacter pylori. Antimicrob Agents Chemother. 1999;43:1497-1499. [PubMed] [Cited in This Article: ] |
| 118. | Fujimura S, Kato S, Kawamura T, Watanabe A. In vitro activity of rifampicin against Helicobacter pylori isolated from children and adults. J Antimicrob Chemother. 2002;49:541-543. [PubMed] [Cited in This Article: ] |
| 119. | Tan Y, Hu Z, Zhao Y, Cai X, Luo C, Zou C, Liu X. The beginning of the rpoB gene in addition to the rifampin resistance determination region might be needed for identifying rifampin/rifabutin cross-resistance in multidrug-resistant Mycobacterium tuberculosis isolates from Southern China. J Clin Microbiol. 2012;50:81-85. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 40] [Cited by in F6Publishing: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 120. | Heep M, Rieger U, Beck D, Lehn N. Mutations in the beginning of the rpoB gene can induce resistance to rifamycins in both Helicobacter pylori and Mycobacterium tuberculosis. Antimicrob Agents Chemother. 2000;44:1075-1077. [PubMed] [Cited in This Article: ] |
| 121. | World Gastroenterology Organisation. World Gastroenterology Organisation Global Guideline: Helicobacter pylori in developing countries. J Clin Gastroenterol. 2011;45:383-388. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 65] [Cited by in F6Publishing: 73] [Article Influence: 5.6] [Reference Citation Analysis (1)] |
| 122. | Machado RS, Silva MR, Viriato A. Furazolidone, tetracycline and omeprazole: a low-cost alternative for Helicobacter pylori eradication in children. J Pediatr (Rio J). 2008;84:160-165. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 123. | Alarcón T, de la Obra P, Domingo D, García-Campos JA, Díaz-Regañón J, López-Brea M. [In vitro activity of furazolidone and nitrofurantoin in Helicobacter pylori clinical isolates and study of mutation rate]. Rev Esp Quimioter. 2005;18:313-318. [PubMed] [Cited in This Article: ] |
| 124. | Boyanova L, Gergova G, Nikolov R, Davidkov L, Kamburov V, Jelev C, Mitov I. Prevalence and evolution of Helicobacter pylori resistance to 6 antibacterial agents over 12 years and correlation between susceptibility testing methods. Diagn Microbiol Infect Dis. 2008;60:409-415. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 57] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 125. | Rafeey M, Ghotaslou R, Nikvash S, Hafez AA. Primary resistance in Helicobacter pylori isolated in children from Iran. J Infect Chemother. 2007;13:291-295. [PubMed] [Cited in This Article: ] |
| 126. | Zullo A, Ierardi E, Hassan C, De Francesco V. Furazolidone-based therapies for Helicobacter pylori infection: a pooled-data analysis. Saudi J Gastroenterol. 2012;18:11-17. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 30] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 127. | De Francesco V, Ierardi E, Hassan C, Zullo A. Helicobacter pylori therapy: Present and future. World J Gastrointest Pharmacol Ther. 2012;3:68-73. [PubMed] [DOI] [Cited in This Article: ] [Cited by in CrossRef: 35] [Cited by in F6Publishing: 35] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 128. | Ching SS, Sabanathan S, Jenkinson LR. Treatment of Helicobacter pylori in surgical practice: a randomised trial of triple versus quadruple therapy in a rural district general hospital. World J Gastroenterol. 2008;14:3855-3860. [PubMed] [Cited in This Article: ] |
| 129. | Malfertheiner P, Bazzoli F, Delchier JC, Celiñski K, Giguère M, Rivière M, Mégraud F. Helicobacter pylori eradication with a capsule containing bismuth subcitrate potassium, metronidazole, and tetracycline given with omeprazole versus clarithromycin-based triple therapy: a randomised, open-label, non-inferiority, phase 3 trial. Lancet. 2011;377:905-913. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 350] [Cited by in F6Publishing: 346] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 130. | Songür Y, Senol A, Balkarli A, Baştürk A, Cerçi S. Triple or quadruple tetracycline-based therapies versus standard triple treatment for Helicobacter pylori treatment. Am J Med Sci. 2009;338:50-53. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 23] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 131. | Pai CG, Thomas CP, Biswas A, Rao S, Ramnarayan K. Quadruple therapy for initial eradication of Helicobacter pylori in peptic ulcer: comparison with triple therapy. Indian J Gastroenterol. 2003;22:85-87. [PubMed] [Cited in This Article: ] |
| 132. | Hong J, Yang HR. Efficacy of Proton Pump Inhibitor-based Triple Therapy and Bismuth-based Quadruple Therapy for Helicobacter pylori Eradication in Korean Children. Pediatr Gastroenterol Hepatol Nutr. 2012;15:237-242. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 10] [Cited by in F6Publishing: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 133. | Veldhuyzen van Zanten S, Chiba N, Barkun A, Fallone C, Farley A, Cockeram A, Dallaire C, Simms L, Nicholls B. A randomized trial comparing seven-day ranitidine bismuth citrate and clarithromycin dual therapy to seven-day omeprazole, clarithromycin and amoxicillin triple therapy for the eradication of Helicobacter pylori. Can J Gastroenterol. 2003;17:533-538. [PubMed] [Cited in This Article: ] |
| 134. | Lind T, Veldhuyzen van Zanten S, Unge P, Spiller R, Bayerdörffer E, O’Morain C, Bardhan KD, Bradette M, Chiba N, Wrangstadh M. Eradication of Helicobacter pylori using one-week triple therapies combining omeprazole with two antimicrobials: the MACH I Study. Helicobacter. 1996;1:138-144. [PubMed] [Cited in This Article: ] |
| 135. | Wong BC, Chang FY, Abid S, Abbas Z, Lin BR, Van Rensburg C, Chen PC, Schneider H, Simjee AE, Hamid SS. Triple therapy with clarithromycin, omeprazole, and amoxicillin for eradication of Helicobacter pylori in duodenal ulcer patients in Asia and Africa. Aliment Pharmacol Ther. 2000;14:1529-1535. [PubMed] [Cited in This Article: ] |
| 136. | Lahbabi M, Alaoui S, El Rhazi K, El Abkari M, Nejjari C, Amarti A, Bennani B, Mahmoud M, Ibrahimi A, Benajah DA. Sequential therapy versus standard triple-drug therapy for Helicobacter pylori eradication: result of the HPFEZ randomised study. Clin Res Hepatol Gastroenterol. 2013;37:416-421. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 15] [Cited by in F6Publishing: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 137. | Van der Poorten D, Katelaris PH. The effectiveness of rifabutin triple therapy for patients with difficult-to-eradicate Helicobacter pylori in clinical practice. Aliment Pharmacol Ther. 2007;26:1537-1542. [PubMed] [Cited in This Article: ] |
| 138. | Borody TJ, Pang G, Wettstein AR, Clancy R, Herdman K, Surace R, Llorente R, Ng C. Efficacy and safety of rifabutin-containing ‘rescue therapy’ for resistant Helicobacter pylori infection. Aliment Pharmacol Ther. 2006;23:481-488. [PubMed] [Cited in This Article: ] |
| 139. | Laine L, Hunt R, El-Zimaity H, Nguyen B, Osato M, Spénard J. Bismuth-based quadruple therapy using a single capsule of bismuth biskalcitrate, metronidazole, and tetracycline given with omeprazole versus omeprazole, amoxicillin, and clarithromycin for eradication of Helicobacter pylori in duodenal ulcer patients: a prospective, randomized, multicenter, North American trial. Am J Gastroenterol. 2003;98:562-567. [PubMed] [Cited in This Article: ] |
| 140. | Rodgers C, van Zanten SV. A meta-analysis of the success rate of Helicobacter pylori therapy in Canada. Can J Gastroenterol. 2007;21:295-300. [PubMed] [Cited in This Article: ] |
| 141. | Mantzaris GJ, Petraki K, Archavlis E, Amberiadis P, Christoforidis P, Kourtessas D, Chiotakakou E, Triantafyllou G. Omeprazole triple therapy versus omeprazole quadruple therapy for healing duodenal ulcer and eradication of Helicobacter pylori infection: a 24-month follow-up study. Eur J Gastroenterol Hepatol. 2002;14:1237-1243. [PubMed] [Cited in This Article: ] |
| 142. | Zheng Q, Chen WJ, Lu H, Sun QJ, Xiao SD. Comparison of the efficacy of triple versus quadruple therapy on the eradication of Helicobacter pylori and antibiotic resistance. J Dig Dis. 2010;11:313-318. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 44] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 143. | Tay CY, Windsor HM, Thirriot F, Lu W, Conway C, Perkins TT, Marshall BJ. Helicobacter pylori eradication in Western Australia using novel quadruple therapy combinations. Aliment Pharmacol Ther. 2012;36:1076-1083. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 49] [Cited by in F6Publishing: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 144. | Nagahara A, Miwa H, Ogawa K, Kurosawa A, Ohkura R, Iida N, Sato N. Addition of metronidazole to rabeprazole-amoxicillin-clarithromycin regimen for Helicobacter pylori infection provides an excellent cure rate with five-day therapy. Helicobacter. 2000;5:88-93. [PubMed] [Cited in This Article: ] |
| 145. | Choi C, Lee D, Chon I. Concomitant therapy was more effective than ppi-base triple therapy in Korea: a preliminary report. Helicobacter. 2011;16:136. [Cited in This Article: ] |
| 146. | Treiber G, Wittig J, Ammon S, Walker S, van Doorn LJ, Klotz U. Clinical outcome and influencing factors of a new short-term quadruple therapy for Helicobacter pylori eradication: a randomized controlled trial (MACLOR study). Arch Intern Med. 2002;162:153-160. [PubMed] [Cited in This Article: ] |
| 147. | Treiber G, Ammon S, Schneider E, Klotz U. Amoxicillin/metronidazole/omeprazole/clarithromycin: a new, short quadruple therapy for Helicobacter pylori eradication. Helicobacter. 1998;3:54-58. [PubMed] [Cited in This Article: ] |
| 148. | Greenberg ER, Anderson GL, Morgan DR, Torres J, Chey WD, Bravo LE, Dominguez RL, Ferreccio C, Herrero R, Lazcano-Ponce EC. 14-day triple, 5-day concomitant, and 10-day sequential therapies for Helicobacter pylori infection in seven Latin American sites: a randomised trial. Lancet. 2011;378:507-514. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 181] [Cited by in F6Publishing: 209] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 149. | Gatta L, Vakil N, Vaira D, Scarpignato C. Global eradication rates for Helicobacter pylori infection: systematic review and meta-analysis of sequential therapy. BMJ. 2013;347:f4587. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 184] [Cited by in F6Publishing: 193] [Article Influence: 17.5] [Reference Citation Analysis (95)] |
| 150. | Francavilla R, Lionetti E, Castellaneta SP, Magistà AM, Boscarelli G, Piscitelli D, Amoruso A, Di Leo A, Miniello VL, Francavilla A. Improved efficacy of 10-Day sequential treatment for Helicobacter pylori eradication in children: a randomized trial. Gastroenterology. 2005;129:1414-1419. [PubMed] [Cited in This Article: ] |
| 151. | Zullo A, Gatta L, De Francesco V, Hassan C, Ricci C, Bernabucci V, Cavina M, Ierardi E, Morini S, Vaira D. High rate of Helicobacter pylori eradication with sequential therapy in elderly patients with peptic ulcer: a prospective controlled study. Aliment Pharmacol Ther. 2005;21:1419-1424. [PubMed] [Cited in This Article: ] |
| 152. | Seddik H, Ahid S, El Adioui T, El Hamdi FZ, Hassar M, Abouqal R, Cherrah Y, Benkirane A. Sequential therapy versus standard triple-drug therapy for Helicobacter pylori eradication: a prospective randomized study. Eur J Clin Pharmacol. 2013;69:1709-1715. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 153. | Zegarra Chang A, Bravo Paredes E, Piscoya Rivera A, De Los Ríos Senmache R, Pinto Valdivia JL, Huerta-Mercado Tenorio J. [“Pilot study: sequential therapy in erradicating Helicobacter pylori in the Cayetano Heredia Hospital”]. Rev Gastroenterol Peru. 2011;31:21-25. [PubMed] [Cited in This Article: ] |
| 154. | Kim YS, Kim SJ, Yoon JH, Suk KT, Kim JB, Kim DJ, Kim DY, Min HJ, Park SH, Shin WG. Randomised clinical trial: the efficacy of a 10-day sequential therapy vs. a 14-day standard proton pump inhibitor-based triple therapy for Helicobacter pylori in Korea. Aliment Pharmacol Ther. 2011;34:1098-1105. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 49] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 155. | Qian J, Ye F, Zhang J, Yang YM, Tu HM, Jiang Q, Shang L, Pan XL, Shi RH, Zhang GX. Levofloxacin-containing triple and sequential therapy or standard sequential therapy as the first line treatment for Helicobacter pylori eradication in China. Helicobacter. 2012;17:478-485. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 33] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 156. | Sierra F, Forero JD, Rey M, Botero ML, Cárdenas A. Pilot study: miscellaneous therapy is highly successful for Helicobacter pylori eradication. Aliment Pharmacol Ther. 2013;37:1165-1171. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 7] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 157. | Zullo A, Scaccianoce G, De Francesco V, Ruggiero V, D'Ambrosio P, Castorani L, Bonfrate L, Vannella L, Hassan C, Portincasa P. Concomitant, sequential, and hybrid therapy for H. pylori eradication: A pilot study. Clin Res Hepatol Gastroenterol. 2013;pii:S2210-7401(13)00090-9. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 63] [Cited by in F6Publishing: 71] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 158. | Sardarian H, Fakheri H, Hosseini V, Taghvaei T, Maleki I, Mokhtare M. Comparison of hybrid and sequential therapies for Helicobacter pylori eradication in Iran: a prospective randomized trial. Helicobacter. 2013;18:129-134. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 77] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 159. | Hsu PI, Wu DC, Wu JY, Graham DY. Modified sequential Helicobacter pylori therapy: proton pump inhibitor and amoxicillin for 14 days with clarithromycin and metronidazole added as a quadruple (hybrid) therapy for the final 7 days. Helicobacter. 2011;16:139-145. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 125] [Cited by in F6Publishing: 122] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 160. | Molnar B, Szoke D, Ruzsovics A, Tulassay Z. Significantly elevated Helicobacter pylori density and different genotype distribution in erosions as compared with normal gastric biopsy specimen detected by quantitative real-time PCR. Eur J Gastroenterol Hepatol. 2008;20:305-313. [PubMed] [Cited in This Article: ] |
| 161. | De Francesco V, Giorgio F, Ierardi E, D’Ercole C, Hassan C, Zullo A. Helicobacter pylori clarithromycin resistance assessment: are gastric antral biopsies sufficient? Gastroenterology Insights. 2012;4:e2. [Cited in This Article: ] |
| 162. | van Doorn LJ, Schneeberger PM, Nouhan N, Plaisier AP, Quint WG, de Boer WA. Importance of Helicobacter pylori cagA and vacA status for the efficacy of antibiotic treatment. Gut. 2000;46:321-326. [PubMed] [Cited in This Article: ] |
| 163. | De Francesco V, Zullo A, Margiotta M, Marangi S, Burattini O, Berloco P, Russo F, Barone M, Di Leo A, Minenna MF. Sequential treatment for Helicobacter pylori does not share the risk factors of triple therapy failure. Aliment Pharmacol Ther. 2004;19:407-414. [PubMed] [Cited in This Article: ] |
| 164. | Kusters JG, Gerrits MM, Van Strijp JA, Vandenbroucke-Grauls CM. Coccoid forms of Helicobacter pylori are the morphologic manifestation of cell death. Infect Immun. 1997;65:3672-3679. [PubMed] [Cited in This Article: ] |
| 165. | Furuta T, Shirai N, Xiao F, Ohashi K, Ishizaki T. Effect of high-dose lansoprazole on intragastic pH in subjects who are homozygous extensive metabolizers of cytochrome P4502C19. Clin Pharmacol Ther. 2001;70:484-492. [PubMed] [Cited in This Article: ] |
| 166. | Hirschowitz BI, Haber MM. Helicobacter pylori effects on gastritis, gastrin and enterochromaffin-like cells in Zollinger-Ellison syndrome and non-Zollinger-Ellison syndrome acid hypersecretors treated long-term with lansoprazole. Aliment Pharmacol Ther. 2001;15:87-103. [PubMed] [Cited in This Article: ] |
| 167. | Gill HS. Probiotics to enhance anti-infective defences in the gastrointestinal tract. Best Pract Res Clin Gastroenterol. 2003;17:755-773. [PubMed] [Cited in This Article: ] |
| 168. | Rosania R, Minenna MF, Giorgio F, Facciorusso A, De Francesco V, Hassan C, Panella C, Ierardi E. Probiotic multistrain treatment may eradicate Helicobacter pylori from the stomach of dyspeptics: a placebo-controlled pilot study. Inflamm Allergy Drug Targets. 2012;11:244-249. [PubMed] [Cited in This Article: ] |
| 169. | Cremonini F, Di Caro S, Covino M, Armuzzi A, Gabrielli M, Santarelli L, Nista EC, Cammarota G, Gasbarrini G, Gasbarrini A. Effect of different probiotic preparations on anti-helicobacter pylori therapy-related side effects: a parallel group, triple blind, placebo-controlled study. Am J Gastroenterol. 2002;97:2744-2749. [PubMed] [Cited in This Article: ] |
| 170. | Myllyluoma E, Veijola L, Ahlroos T, Tynkkynen S, Kankuri E, Vapaatalo H, Rautelin H, Korpela R. Probiotic supplementation improves tolerance to Helicobacter pylori eradication therapy--a placebo-controlled, double-blind randomized pilot study. Aliment Pharmacol Ther. 2005;21:1263-1272. [PubMed] [Cited in This Article: ] |
| 171. | Yoon H, Kim N, Kim JY, Park SY, Park JH, Jung HC, Song IS. Effects of multistrain probiotic-containing yogurt on second-line triple therapy for Helicobacter pylori infection. J Gastroenterol Hepatol. 2011;26:44-48. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 172. | Francavilla R, Lionetti E, Castellaneta SP, Magistà AM, Maurogiovanni G, Bucci N, De Canio A, Indrio F, Cavallo L, Ierardi E. Inhibition of Helicobacter pylori infection in humans by Lactobacillus reuteri ATCC 55730 and effect on eradication therapy: a pilot study. Helicobacter. 2008;13:127-134. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 99] [Cited by in F6Publishing: 102] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 173. | Szajewska H, Horvath A, Piwowarczyk A. Meta-analysis: the effects of Saccharomyces boulardii supplementation on Helicobacter pylori eradication rates and side effects during treatment. Aliment Pharmacol Ther. 2010;32:1069-1079. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 149] [Cited by in F6Publishing: 153] [Article Influence: 10.9] [Reference Citation Analysis (0)] |