Published online May 28, 2013. doi: 10.3748/wjg.v19.i20.3077
Revised: March 23, 2013
Accepted: March 28, 2013
Published online: May 28, 2013
Processing time: 153 Days and 0.4 Hours
AIM: To introduce robotic cholecystectomy (RC) using new port sites on the low abdominal area.
METHODS: From June 2010 to June 2011, a total of 178 RCs were performed at Ajou University Medical Center. We prospectively collected the set-up time (working time and docking time) and console time in all robotic procedures.
RESULTS: Eighty-three patients were male and 95 female; the age ranged from 18 to 72 years of age (mean 54.6 ± 15.0 years). All robotic procedures were successfully completed. The mean operation time was 52.4 ± 17.1 min. The set-up time and console time were 11.9 ± 5.4 min (5-43 min) and 15.1 ± 8.0 min (4-50 min), respectively. The conversion rate to laparoscopic or open procedures was zero. The complication rate was 0.6% (n = 1, bleeding). There was no bile duct injury or mortality. The mean hospital stay was 1.4 ± 1.1 d. There was a significant correlation between the console time and white blood cell count (r = 0.033, P = 0.015). In addition, the higher the white blood cell count (more than 10000), the longer the console time.
CONCLUSION: Robotic cholecystectomy using new port sites on the low abdominal area can be safely and efficiently performed, with sufficient patient satisfaction.
Core tip: The robotic procedure is safe; however, it is not acceptable as a standard operation for gallbladder disease because of its lack of benefits for patients as a result of the high cost and prolonged operating time. In the previous studies, port sites of robotic cholecystectomy were located on the supraumbilical area, similar to laparoscopic surgery. In this study, we changed the port placements from the upper abdominal area to the lower abdominal area.
- Citation: Kim JH, Baek NH, Li G, Choi SH, Jeong IH, Hwang JC, Kim JH, Yoo BM, Kim WH. Robotic cholecystectomy with new port sites. World J Gastroenterol 2013; 19(20): 3077-3082
- URL: https://www.wjgnet.com/1007-9327/full/v19/i20/3077.htm
- DOI: https://dx.doi.org/10.3748/wjg.v19.i20.3077
Laparoscopic cholecystectomy is a standard technique for treatment of gallbladder diseases[1]. However, there are some disadvantages to using laparoscopic techniques[2], and laparoscopic surgery can have a steep learning curve[3]. To overcome these limitations of laparoscopic techniques, the robotic-assisted procedure developed, the 3-dimensional view, magnification, tremor suppression, and flexibility of the instruments[4,5] have allowed precise operating techniques in general surgery[6-8]. Since the first robotic-assisted cholecystectomy was performed in 1997, many reports have been published, including comparative[8-10] and non-comparative studies[6,11-15]. All authors agreed on the safety and feasibility of the robotic procedure. However, most of them concluded that this procedure is not acceptable as a standard operation because of the lack of benefits for patients due to the high cost and prolonged operating time. In this regard, the benefits of the robotic procedure in gallbladder diseases have not yet been established.
Based on the above reports, port sites of robotic cholecystectomy were located on the supraumbilical area, similar to laparoscopic surgery. As a result, we performed robotic cholecystectomy, changing the port placements from the upper abdominal area to the lower abdominal area. In this study, we examined robotic cholecystectomy using port sites located on the low abdominal area and evaluated its surgical outcomes.
From June 2010 to June 2011, a total of 178 robotic cholecystectomies were performed at Ajou University Medical Center. We prospectively collected the set-up time (working time and docking time) and console time in all robotic procedures. The initial indications of surgery included gallbladder polyp or symptomatic gallstones. Exclusion criteria were the presence of acute cholecystitis and previous history of extensive upper abdominal surgery. Informed consent was obtained for the robotic cholecystectomy. We retrospectively reviewed the medical records of all patients and analyzed data, including demographic information, clinical presentation, results of laboratory studies, operative records, postoperative complications, and postoperative hospital stay.
In this study, the operating time was defined as the time from skin incision to wound closure. The working time extended from the first skin incision until the decision to bring the da vinci into place was made. The docking time spanned the setup of the robot onto the surgical field. The set-up time was defined as the time from skin incision until the start the dissection. The console time was defined as the time from the start of dissection until the moment the gallbladder was completely freed from the liver.
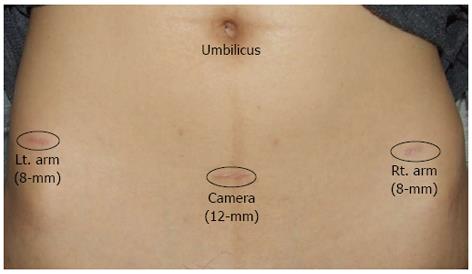
The robotic-assisted operations were performed with the 4-arm da Vinci robot system (Intuitive Surgical, CA, United States). The operating team consisted of one operating surgeon and one assisting resident. The assisting resident replaced instruments and paced clips during cholecystectomy. Robotic cholecystectomy was performed using a three or four port technique. A total of three trocars were utilized as shown in Figure 1. First, a 12-mm trocar was inserted through a vertical incision below the umbilicus using an open method. CO2 gas was introduced through this trocar to obtain an intraperitoneal pressure of 12 mm Hg. All other ports were placed under direct visualization. The 8-mm ports were placed 7 to 10 cm distant from the endoscope. An additional fourth trocar (3 or 5 mm) was placed in the right anterior axillary line in the upper quadrant in cases of severe inflammation of gallbladder and thickening of the gallbladder wall and used for retraction and clip/loop placement.
The patient was then placed in reverse Trendelenburg position with the right side up. The da vinci surgical robot was then brought into position and docked. The maryland forceps was inserted into the right robotic positioner, and a cadiere grasper was placed into the left positioner. The dissection was performed according to the critical view method as described by Strasberg et al[16]. After clear identification of the cystic duct and cystic artery, only the cystic duct was ligated manually with clips. In contrast, the cystic artery was coagulated just around the gallbladder and not ligated. The gallbladder was dissected from the fossa and placed in an endoscopic retrieval bag. Once fully dissected, the gallbladder was removed through the umbilical port in an endopouch. The robot was then withdrawn, and the 12-mm port site was closed with absorbable sutures.
Patients were discharged 1 d after surgery if sufficiently recovered and if pain and nausea had receded. All patients were seen for examination and reassessment at the outpatient clinics 1 wk after surgery. Laboratory tests were performed only if indicated.
Statistical analysis was performed with independent t-test, and Spearman’s correlation. P-value < 0.05 was considered statistically significant.
Eighty-three patients were male and 95 female; the age ranged from 18 to 72 years of age (mean 54.6 ± 15.0 years). Table 1 shows the clinical characteristics of patients who underwent robotic cholecystectomy. The associated diseases were hypertension (n = 29), diabetes mellitus (n = 5), hypothyroidism (n = 2), and hepatitis (n = 2). The previous operations were appendectomy (n = 10), C-section (n = 10), and hysterectomy (n = 1). Endoscopic retrograde cholangiopancreatogram was performed in 18 patients. After surgery, two patients in robotic cholecystectomy (RC) group were diagnosed with gallbladder (GB) cancer. A 53-year-old man who was diagnosed with a gallbladder mass underwent cholecystectomy with lymph node dissection. Unfortunately, tumor penetrated the serosa layer of the gallbladder (T3N0). We recommended re-operation; however, he refused the surgery in our hospital and did not follow up. The other patient was a 60-year-old man who had undergone surgery for gallbladder polyp and was diagnosed with T1a gallbladder cancer. There was no recurrence for 8 mo after surgery.
| RC (n = 178) | |
| Age (yr) | 40.1 ± 9.8 |
| Gender (male/female) | 83/95 |
| Laboratory findings | |
| White blood cell count (/m3) | 7483.4 ± 2670.8 |
| AST (IU/L) | 62.9 ± 124.0 |
| ALT (IU/L) | 68.9 ± 134.4 |
| Total bilirubin (mg/dL) | 1.0 ± 0.9 |
| Combined diseases | |
| Diabetes mellitus | 5 |
| Hypertension | 29 |
| Ischemic heart disease | 0 |
| COPD | 0 |
All robotic procedures were successfully completed. The mean operation time was 52.4 ± 17.1 min. The set-up time and console time were 11.9 ± 5.4 min (5-43 min) and 15.1 ± 8.0 min (4-50 min), respectively (Table 2). The conversion rate to laparoscopic or open procedures was zero. The complication rates was 0.6% (n = 1, bleeding) (Table 3). The patient who had complications was a 34-year-old female who underwent re-operation on postoperative day 1; previous incisions on the low abdominal area were employed during the surgery. We identified the focus of bleeding on the gallbladder bed and coagulated the bleeder. She was finally discharged from the hospital without any symptoms. There was no bleeding associated with the cystic artery. There was no bile duct injury and mortality. The mean postoperative hospital stay was 1.43 ± 1.16 d.
| RC (n = 178) | |
| Operation time (min) | 52.4 ± 17.1 |
| Set-up time (min) | 11.9 ± 5.4 |
| Working time (min) | 7.6 ± 4.2 |
| Docking time (min) | 4.3 ± 2.5 |
| Console time (min) | 15.1 ± 8.0 |
| RC (n = 178) | |
| Complications | |
| Bleeding | 1 |
| Bile duct injury | 0 |
| Open conversion | 0 |
| Total hospital stay (d) | 2.9 ± 1.8 |
| Postoperative hospital stay (d) | 1.4 ± 1.1 |
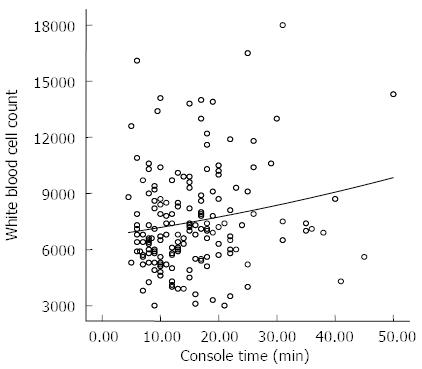
We analyzed the relationship between console time and other factors. The results showed that the only significant correlation was between console time and white blood cell count (r = 0.182, P = 0.015) (Figure 2). In addition, the higher the white blood cell count (more than 10000), the longer the console time (Table 4).
| WBC (< 10000) | WBC (≥10000) | P value | |
| (n = 151) | (n = 27) | ||
| Console time (min) | 14.6 ± 7.5 | 18.3 ± 9.9 | 0.025 |
Since 2005, when the robotic system was first introduced in South Korea, other investigators have also reported experience with surgical robotics in a variety of surgical procedures, including cholecystectomy, gastrectomy, and thyroidectomy[17-19]. However, hepato-biliary surgeons in South Korea stopped performing robotic-assisted cholecystectomy because of the lack of advantages for patients compared to its high cost[8,9,20]. In our hospital, we experienced the first fully robotic procedure in a patient who was diagnosed with choledochal cyst; the patient underwent resection of the cyst and intracorporeal hepaticojejunostomy. After that, we decided to change the trocar placements when using the robotic system in gallbladder diseases. Before beginning robotic surgery, Professor Kim (Kim WH) also enrolled in the Intuitive Surgical da Vinci training course.
In the present study, we used only three of four arms of the da Vinci system to reduce the instrument-related costs. Most importantly, port sites were also much lower than the umbilical line. The 12-mm camera port site was located almost 10 cm from the umbilicus. The other two 8-mm port sites were located around the right and left anterior superior iliac spine. This line can be called the “Panty line” or “Bikini line”. All patients were satisfied with both the degree of postoperative pain and scarring. However, we experienced a problem in that the length of the robotic arms were too short to dissect the gallbladder, especially in big or obese patients. Indeed, Cadière et al[21]. Previously suggested that the robotic approach requires new operative strategies and a change in the pattern of trocar placement. Most surgeons will likely agree that, although Nio et al[9]. Reported on altered positioning of trocars in robot-assisted laparoscopic cholecystectomy, their locations were not different from the laparoscopic technique. In the early period of the study, we included patients who were diagnosed with gallbladder polyp or minimal symptomatic gallstones. As the number of cases increased, we attempted robotic procedures in cases of inflamed gallbladder, such as acute cholecystitis, empyematous cholecystitis, and gangrenous cholecystitis. According to previous reports, some authors did not perform robotic procedures in acute cholecystitis[11,22,23]. In study by Ruurda et al[14], the rates of acute cholecystitis were 17%, and there was one conversion to an open procedure, caused by the surgeons’ inability to expose the gallbladder sufficiently because of severe cholecystitis. In the present study, there was no conversion to laparoscopic or open procedures after including cases of acute cholecystitis.
Thus far, most authors have reported that the operation time of robotic procedures was much longer than cases that used laparoscopic technique. The operation time varied, with a range of 55-152 min[13,14,21,23-28]. The reason for the various results in those studies was that the definition of the operation time was heterogeneous; the operation time was defined as the time from skin incision to skin closure, or it included anesthesia or time in the robotic room. In the present study, the operation time was 81.3 ± 19.0, similar to other results[14,21,24-28]. Marescaux et al[24] reported that the median time for dissection was 25 min (range 14-109 min) and the overall operative time was 108 min, similar to times of conventional laparoscopy. In this study, the console time was 15.1 ± 8.0 min, similar to another report[24]. The console time in cases of inflamed gallbladder was longer than that of cases of non-inflamed gallbladder.
The conversion rates of robotic procedures in other studies have been almost zero[9,13,21,22,25,27]; our data showed similar results. However, Miller et al[28] reported that conversion to conventional laparoscopic techniques was necessary due to malfunction of graspers in three consecutive procedures. The other reasons for conversion were in cases of acute cholecystitis[24], presence of severe adhesions, and poor visualization[26]. In this study, we found that the fourth 5-mm instrument was effective in grasping and retracting the edematous gallbladder. Until now, we have experienced no conversion after the indications were expanded to include severe acute cholecystitis. There are two published reports of 8-mm port-site hernias, so it should be recommended that any port greater than 5 mm in diameter should be routinely sutured closed[22,29]. We did not experience an 8-mm port-site hernia, likely due to the short follow-up period after surgery.
To our knowledge, this study is the first large series of robotic cholecystectomy in South Korea. Some advantages of robotic cholecystectomy from this study are as follows. First, patient satisfaction regarding a lower lying wound was very high because of the absence of a scar on the upper abdominal area. Second, the subjective perception of the surgeons is that the robotic system makes dissection easier at Callot’s triangle. Furthermore, the use of the robotics allowed the surgeon to remain in an ergonomic position throughout the procedure. This could reduce the fatigue experienced during prolonged or difficult operations, especially in cases of severe acute cholecystitis. Third, the robotic procedure is safely performed in patients who underwent upper abdominal surgery, because an adhesiolysis can be easily performed throughout the lower lying port sites.
In summary, robotic cholecystectomy using the new port sites on the low abdominal area can be safely and efficiently performed, with sufficient patient satisfaction. However, we recommend that the more difficult cases (due to acute inflammation) were likely excluded from participation in an early period.
To overcome the limitations of laparoscopic techniques, the robotic-assisted procedure developed, the 3-dimensional view, magnification, tremor suppression, and flexibility of the instruments have allowed precise operating techniques in general surgery. The robotic procedure is safe; however, it is not acceptable as a standard operation for gallbladder disease because of the lack of advantages for patients compared to its high cost.
Since the first robotic-assisted cholecystectomy was performed in 1997, the benefits of the robotic procedure in gallbladder diseases have not yet been established. In the previous studies, port sites of robotic cholecystectomy were located on the supraumbilical area, similar to laparoscopic surgery. As a result, we performed robotic cholecystectomy, changing the port placements.
Recent reports have highlighted the importance of changing the port placements from the upper abdominal area to the lower abdominal area. This is the first study to report that robotic cholecystectomy with lower lying ports can be safely and efficiently performed.
This study may represent a new strategy for surgical intervention in the treatment of patients with gallbladder diseases.
Da vinci robot system (Intuitive Surgical, CA, United States) is composed of the surgeon’s viewing and control console and a movable cart with four articulated robot arms.
This study is a feasibility study that demonstrates that robotically assisted cholecystectomy may be performed. The technical details of port placement by the authors will make it helpful to the general surgeon who wishes to use the robot to assist with cholecystectomy.
P- Reviewers Kumar A, Leitman IM S- Editor Huang XZ L- Editor A E- Editor Zhang DN
| 1. | Brasesco OE, Rosin D, Rosenthal RJ. Laparoscopic surgery of the liver and biliary tract. J Laparoendosc Adv Surg Tech A. 2002;12:91-100. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 9] [Cited by in F6Publishing: 10] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 2. | Lanfranco AR, Castellanos AE, Desai JP, Meyers WC. Robotic surgery: a current perspective. Ann Surg. 2004;239:14-21. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 761] [Cited by in F6Publishing: 640] [Article Influence: 32.0] [Reference Citation Analysis (0)] |
| 3. | Voitk AJ, Tsao SG, Ignatius S. The tail of the learning curve for laparoscopic cholecystectomy. Am J Surg. 2001;182:250-253. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 66] [Cited by in F6Publishing: 69] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 4. | Dakin GF, Gagner M. Comparison of laparoscopic skills performance between standard instruments and two surgical robotic systems. Surg Endosc. 2003;17:574-579. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 110] [Cited by in F6Publishing: 96] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 5. | Jourdan IC, Dutson E, Garcia A, Vleugels T, Leroy J, Mutter D, Marescaux J. Stereoscopic vision provides a significant advantage for precision robotic laparoscopy. Br J Surg. 2004;91:879-885. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 68] [Cited by in F6Publishing: 71] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 6. | Giulianotti PC, Coratti A, Angelini M, Sbrana F, Cecconi S, Balestracci T, Caravaglios G. Robotics in general surgery: personal experience in a large community hospital. Arch Surg. 2003;138:777-784. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 749] [Cited by in F6Publishing: 704] [Article Influence: 33.5] [Reference Citation Analysis (0)] |
| 7. | Talamini M, Campbell K, Stanfield C. Robotic gastrointestinal surgery: early experience and system description. J Laparoendosc Adv Surg Tech A. 2002;12:225-232. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 76] [Cited by in F6Publishing: 78] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 8. | Kornprat P, Werkgartner G, Cerwenka H, Bacher H, El-Shabrawi A, Rehak P, Mischinger HJ. Prospective study comparing standard and robotically assisted laparoscopic cholecystectomy. Langenbecks Arch Surg. 2006;391:216-221. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 32] [Cited by in F6Publishing: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 9. | Nio D, Bemelman WA, Busch OR, Vrouenraets BC, Gouma DJ. Robot-assisted laparoscopic cholecystectomy versus conventional laparoscopic cholecystectomy: a comparative study. Surg Endosc. 2004;18:379-382. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 50] [Cited by in F6Publishing: 46] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 10. | Mühlmann G, Klaus A, Kirchmayr W, Wykypiel H, Unger A, Höller E, Nehoda H, Aigner F, Weiss HG. DaVinci robotic-assisted laparoscopic bariatric surgery: is it justified in a routine setting? Obes Surg. 2003;13:848-854. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 55] [Cited by in F6Publishing: 49] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Chitwood WR, Nifong LW, Chapman WH, Felger JE, Bailey BM, Ballint T, Mendleson KG, Kim VB, Young JA, Albrecht RA. Robotic surgical training in an academic institution. Ann Surg. 2001;234:475-84; discussion 484-6. [PubMed] [Cited in This Article: ] |
| 12. | Hanisch E, Markus B, Gutt C, Schmandra TC, Encke A. [Robot-assisted laparoscopic cholecystectomy and fundoplication--initial experiences with the Da Vinci system]. Chirurg. 2001;72:286-288. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 28] [Cited by in F6Publishing: 32] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 13. | Perez A, Zinner MJ, Ashley SW, Brooks DC, Whang EE. What is the value of telerobotic technology in gastrointestinal surgery? Surg Endosc. 2003;17:811-813. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 30] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 14. | Ruurda JP, Broeders IA, Simmermacher RP, Borel Rinkes IH, Van Vroonhoven TJ. Feasibility of robot-assisted laparoscopic surgery: an evaluation of 35 robot-assisted laparoscopic cholecystectomies. Surg Laparosc Endosc Percutan Tech. 2002;12:41-45. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 64] [Cited by in F6Publishing: 71] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 15. | Talamini MA, Chapman S, Horgan S, Melvin WS. A prospective analysis of 211 robotic-assisted surgical procedures. Surg Endosc. 2003;17:1521-1524. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 208] [Cited by in F6Publishing: 172] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 16. | Strasberg SM, Hertl M, Soper NJ. An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg. 1995;180:101-125. [PubMed] [Cited in This Article: ] |
| 17. | Lee S, Ryu HR, Park JH, Kim KH, Kang SW, Jeong JJ, Nam KH, Chung WY, Park CS. Excellence in robotic thyroid surgery: a comparative study of robot-assisted versus conventional endoscopic thyroidectomy in papillary thyroid microcarcinoma patients. Ann Surg. 2011;253:1060-1066. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 83] [Cited by in F6Publishing: 82] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 18. | Woo Y, Hyung WJ, Pak KH, Inaba K, Obama K, Choi SH, Noh SH. Robotic gastrectomy as an oncologically sound alternative to laparoscopic resections for the treatment of early-stage gastric cancers. Arch Surg. 2011;146:1086-1092. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 147] [Cited by in F6Publishing: 162] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 19. | Kang CM, Chi HS, Hyeung WJ, Kim KS, Choi JS, Lee WJ, Kim BR. The first korean experience of telemanipulative robot-assisted laparoscopic cholecystectomy using the da vinci system. Yonsei Med J. 2007;48:540-545. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 17] [Cited by in F6Publishing: 20] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 20. | Müller-Stich BP, Reiter MA, Wente MN, Bintintan VV, Köninger J, Büchler MW, Gutt CN. Robot-assisted versus conventional laparoscopic fundoplication: short-term outcome of a pilot randomized controlled trial. Surg Endosc. 2007;21:1800-1805. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 95] [Cited by in F6Publishing: 82] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 21. | Cadière GB, Himpens J, Germay O, Izizaw R, Degueldre M, Vandromme J, Capelluto E, Bruyns J. Feasibility of robotic laparoscopic surgery: 146 cases. World J Surg. 2001;25:1467-1477. [PubMed] [Cited in This Article: ] |
| 22. | Jayaraman S, Davies W, Schlachta CM. Getting started with robotics in general surgery with cholecystectomy: the Canadian experience. Can J Surg. 2009;52:374-378. [PubMed] [Cited in This Article: ] |
| 23. | Heemskerk J, van Dam R, van Gemert WG, Beets GL, Greve JW, Jacobs MJ, Bouvy ND. First results after introduction of the four-armed da Vinci Surgical System in fully robotic laparoscopic cholecystectomy. Dig Surg. 2005;22:426-431. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 41] [Cited by in F6Publishing: 44] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 24. | Marescaux J, Smith MK, Fölscher D, Jamali F, Malassagne B, Leroy J. Telerobotic laparoscopic cholecystectomy: initial clinical experience with 25 patients. Ann Surg. 2001;234:1-7. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 125] [Cited by in F6Publishing: 134] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 25. | Kim VB, Chapman WH, Albrecht RJ, Bailey BM, Young JA, Nifong LW, Chitwood WR. Early experience with telemanipulative robot-assisted laparoscopic cholecystectomy using da Vinci. Surg Laparosc Endosc Percutan Tech. 2002;12:33-40. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 90] [Cited by in F6Publishing: 102] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 26. | Vidovszky TJ, Smith W, Ghosh J, Ali MR. Robotic cholecystectomy: learning curve, advantages, and limitations. J Surg Res. 2006;136:172-178. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 81] [Cited by in F6Publishing: 89] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 27. | Breitenstein S, Nocito A, Puhan M, Held U, Weber M, Clavien PA. Robotic-assisted versus laparoscopic cholecystectomy: outcome and cost analyses of a case-matched control study. Ann Surg. 2008;247:987-993. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 133] [Cited by in F6Publishing: 133] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 28. | Miller DW, Schlinkert RT, Schlinkert DK. Robot-assisted laparoscopic cholecystectomy: initial Mayo Clinic Scottsdale experience. Mayo Clin Proc. 2004;79:1132-1136. [PubMed] [Cited in This Article: ] |
| 29. | Tonouchi H, Ohmori Y, Kobayashi M, Kusunoki M. Trocar site hernia. Arch Surg. 2004;139:1248-1256. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 273] [Cited by in F6Publishing: 298] [Article Influence: 15.7] [Reference Citation Analysis (1)] |