Published online Jan 14, 2011. doi: 10.3748/wjg.v17.i2.207
Revised: May 5, 2010
Accepted: May 12, 2010
Published online: January 14, 2011
AIM: To compare the natural history and course of acute diverticulitis in a younger age group with an older population and to evaluate whether younger patients should be managed differently.
METHODS: This study was a retrospective review of 157 patients treated with acute diverticulitis between January 1, 2004 and December 31, 2007. Diverticulitis was stratified according to the Hinchey classification. Patients were divided into 2 populations: group A ≤ 50 years (n = 31); group B > 50 years (n = 126). Mean patient follow-up was 15 mo.
RESULTS: The median age was 60 years. A significantly higher proportion of patients in group B presented with complicated diverticulitis (36.5% vs 12.9%, P = 0.01). Recurrence was more frequent in group A (25.8% vs 11.1%, P = 0.03) and the mean time-to-recurrence was shorter (12 mo vs 28 mo, P = 0.26). The most severe recurrent episodes of acute diverticulitis were classified as Hinchey stage I and none of the patients required emergency surgery. In multivariate analysis, only age (P = 0.024) was identified as an independent prognostic factor for recurrence.
CONCLUSION: Based on the results of this study, the authors recommend that diverticulitis management should be based on the severity of the disease and not on the age of the patient.
- Citation: Faria GR, Almeida AB, Moreira H, Pinto-de-Sousa J, Correia-da-Silva P, Pimenta AP. Acute diverticulitis in younger patients: Any rationale for a different approach? World J Gastroenterol 2011; 17(2): 207-212
- URL: https://www.wjgnet.com/1007-9327/full/v17/i2/207.htm
- DOI: https://dx.doi.org/10.3748/wjg.v17.i2.207
Diverticulosis of the colon is an acquired condition that results from herniation of the mucosa through defects in the muscular layer. Epidemiological and anatomic studies have revealed that diverticular formation of the colon occurs primarily in industrialized and Westernized countries[1,2]. The true incidence of diverticulosis is difficult to measure because most individuals are asymptomatic. It is believed that approximately 60% of individuals over the age of 60, living in Westernized countries, will develop colonic diverticula[3]. Postmortem studies revealed that the disorder is rare prior to the fourth decade, but increases progressively, so that at age 80 approximately 65% of patients will have diverticula of the colon[4,5]. Several epidemiological studies have demonstrated an association between diverticulosis and diets low in dietary fibers and high in refined carbohydrates[6-9].
Most patients with colonic diverticula remain asymptomatic; only 10%-25% will develop diverticulitis and 1% will actually require surgery[1,3,10,11]. The clinical presentation of acute diverticulitis varies widely from mild inflammation to full-blown perforation and peritonitis. In most patients however, the clinical presentation of diverticulitis is mild and medical treatment will achieve successful management of the acute episode[12].
When treated medically, the risk of recurrent diverticulitis ranges from 7% to 45%[13-15], with most recurrences (90%) occurring in the first 5 years[13-16]. The fact that the severity of recurrent attacks is usually considered to be higher than the first episode[13] has been questioned by several authors[16-21].
In recent years, there has been conflicting information regarding the incidence and natural history of diverticulitis in younger patients (aged 50 years or younger). While earlier studies found that the incidence ranged between 2% and 7%[22-26], more recent studies reported a higher incidence ranging between 18% and 34%[17,27]. The natural course and outcome of diverticulitis in the younger patient has also been a matter of discussion. While earlier studies reported a more severe course with a higher rate of complications and recurrent episodes in younger patients with diverticulitis[7,8,22,25,28], some recent reports suggested a milder course, not different from that observed in older patients[12,19,21,27]. The aim of this retrospective study was to compare the natural history and the course of acute diverticulitis in a younger age group with that of an older population. Our purpose was to evaluate whether younger patients should be treated according to the same criteria as their older counterparts.
One hundred fifty seven consecutive patients aged 24-93 years (median, 60 years) with documented acute sigmoid diverticulitis were treated at the University Hospital São João at Porto, with a catchment area of roughly 1 million patients, between 2004 and 2007.
For the patients diagnosed with diverticulitis, who had prior attacks, the clinical, radiological and analytical data of the first episode was retrieved and analyzed. Patients were stratified for age, according to the age at first episode.
The diagnosis of acute diverticulitis was based on lower abdominal pain (usually on the left side), fever and an increase in serum inflammatory parameters. Emergency surgical treatment was undertaken in patients with diffuse peritonitis or septic shock. Patients not undergoing emergency surgery had an abdominal computed tomography (CT) scan and only those with characteristic findings were included in this study. CT scan findings were classified as uncomplicated (localized colonic wall thickening and/or infiltration of pericolonic fat) or complicated (pericolonic or abdominal abscess, localized or free extra luminal air or contrast, bowel obstruction, and fistula formation). The severity of complicated diverticulitis was graded according to Hinchey’s criteria[29]. Stage I patients had small or confined pericolic abscesses, Stage II patients had larger pelvic or distant intra-abdominal abscesses, Stage III patients had purulent diverticulitis and in Stage IV patients had fecal peritonitis.
After resolution of acute inflammation, all patients who had no previous endoscopic diagnosis of diverticular disease underwent elective colonoscopy to confirm the presence of colonic diverticula and to rule out the presence of other diseases, such as cancer or inflammatory bowel disease.
The patients were stratified into 2 groups, according to age at first acute episode: group A included patients aged 50 years or younger (n = 31) and group B included patients older than 50 years (n = 126).
Demographic (age, gender, ethnicity), clinical (presentation signs and symptoms, endoscopic findings), laboratory [white blood cell count and C-reactive protein (CRP) measurements] and radiological (CT scan) features were analyzed and compared between the 2 groups.
Emergency surgery was undertaken if patients had diffuse peritonitis, septic shock, or if the clinical course did not improve after 48-72 h of conservative medical management. In some patients, elective surgery was performed after resolution of the acute episode and all of these had bowel preparation and primary anastomosis. CT-guided percutaneous drainage was performed in patients with abscesses larger than 4 cm in diameter.
Follow-up (range, 6-54 mo; median, 15 mo) was available for 98% of the population and completed in June 2008.
Recurrence of diverticulitis was diagnosed if the patient presented with similar symptoms and clinical findings, confirmed by laboratory and radiological investigations. The rate and severity of recurrences was compared between both groups, as well as the need of emergency or elective surgery after an episode of recurrent diverticulitis.
For statistical analysis, the program SPSS (version 15.0 for Mac) was used. The differences in the distribution of cases according to several parameters being analyzed were compared by χ2 test for categorical variables and the Student t-test for continuous variables. Multivariate analysis was performed using the logistic regression method in order to identify independent factors associated with recurrence. P < 0.05 were considered significant.
The median age of the 157 patients with acute sigmoid diverticulitis was 60 years (range, 24-93 years). The mean age of patients in group A was 40 years (median, 41 years) and for group B was 65 years (median, 62 years). Sixty-eight patients (43.3%) were male and 89 (56.7%) were female (M/F: 0.76/1). Thirty-one patients (19.7%) were 50 years old or younger. There was a striking male predominance in the younger age group (group A) with a male:female ratio of 1.8:1. The older age group (group B) showed a female predominance with a male:female ratio of 0.61:1. The difference between both groups according to gender was significant (P = 0.008).
The most common symptom present on admission was left lower quadrant abdominal pain, which was observed in 147 (93.6%) patients. No significant differences were observed between groups A and B regarding the symptoms and signs most commonly observed in acute episodes (Table 1).
| Group A ( ≤50 yr) | Group B (> 50 yr) | P | |
| Signs and symptoms | |||
| Abdominal pain | 30 (96.7) | 117 (92.8) | NS |
| Fever | 9 (29.0) | 30 (23.8) | NS |
| Acute abdomen | 10 (32.3) | 30 (23.8) | NS |
| Vomiting | 5 (16.1) | 26 (20.6) | NS |
| Constipation | 3 (9.7) | 23 (18.2) | NS |
| Diarrhea | 2 (6.4) | 13 (10.3) | NS |
| Laboratory | |||
| Leucocytosis (> 11 × 109) | 24 (82.7) | 67 (55.8) | 0.01 |
| Mean WBC count (× 109) | 15.0 | 13.1 | 0.01 |
| Mean CRP (mg/dL) | 151.5 | 133.5 | 0.005 |
In this series, younger patients had a significantly higher percentage of leucocytosis (82.7% vs 55.8%, P = 0.01) than patients older than 50 years. The mean value of leucocytosis was significantly higher in patients of group A (P = 0.012) as well as the mean value of CRP elevation (P = 0.005) (Table 1).
One hundred and seven patients (68.2%) had a mild course of diverticulitis with no complications. Fifty patients (31.8%) had a more severe and complicated course. The most common complications were abdominal abscesses, which were observed in 33 cases (21.0%). Five patients required guided percutaneous drainage of abdominal abscesses. These resolved in all patients with no need for further urgent surgery. There were 17 patients (10.8%) who developed free perforation and all underwent emergency surgery (Table 2). No patient died during hospitalization.
| Group A ( ≤50 yr) | Group B (> 50 yr) | P | |
| Disease severity | 0.01 | ||
| Uncomplicated | 27 (87.1) | 80 (63.5) | |
| Complicated | 4 (12.9) | 46 (36.5) | |
| Complication grading | 0.04 | ||
| Grade I/II | 3 (9.7) | 30 (23.8) | |
| Grade III/IV | 1 (3.2) | 16 (12.7) | |
| Surgical treatment | |||
| Emergency surgery | 1 (3.2) | 18 (14.3) | 0.03 |
| Elective surgery | 4 (16.1) | 5 (4.0) | 0.07 |
One hundred and forty seven patients underwent an abdominal CT scan. Ten patients presented with diffuse peritonitis and septic shock, and underwent emergency surgery without further radiological investigation. In these cases, diagnosis of complicated diverticulitis was done intraoperatively.
A significantly lower proportion of patients of group A presented with complicated diverticulitis, compared to patients of group B (12.9% vs 36.5%, P = 0.01), namely abdominal abscesses (Hinchey stages I/II, 9.7% vs 23.8%) and free perforation (Hinchey stages III/IV, 3.2% vs 12.7%) (P = 0.04) (Table 2).
A significantly lower proportion of patients of group A underwent an emergency operation after the first episode of acute diverticulitis (3.2% vs 14.3%, P = 0.026) (Table 2). Seventeen patients undergoing emergency surgery had free perforation with diffuse peritonitis, and 2 patients had pericolonic abscesses.
A significantly higher proportion of patients of group A developed recurrence of diverticulitis (25.8% vs 11.1%, P = 0.03) (Table 3). The mean interval between the primary episode and the recurrent disease was 12 mo (range, 1-40 mo; median, 4 mo) in group A and 28 mo (range, 1-150 mo; median, 6 mo) in group B (P = 0.26). No significant differences were observed between the 2 groups regarding the severity of recurrent episodes (P = 0.1) (Table 3). The most severe recurrent episodes of acute diverticulitis were classified as Hinchey stage I and none of the patients required emergency surgery.
| Group A ( ≤50 yr) | Group B (> 50 yr) | P | |
| Disease recurrence | 0.03 | ||
| Non recurrent | 23 (74.2) | 112 (88.9) | |
| Recurrent | 8 (25.8) | 14 (11.1) | |
| Severity of recurrence | 0.10 | ||
| Non complicated | 6 (75) | 10 (71) | |
| Complicated (Grade I) | 2 (25) | 4 (29) |
In multivariate analysis of this series, age (P = 0.024) was identified as an independent prognostic factor of recurrence of acute diverticulitis (Table 4).
| Factor | Binary regression logistics | |
| Exp (B) | P | |
| Male gender | 1.13 | 0.827 |
| Age ≤ 50 yr | 0.26 | 0.024 |
| White blood cell count (× 109) | 0.99 | 0.830 |
| Severity index | 1.08 | 0.798 |
For many years, diverticulosis was considered an old patient’s disease with an incidence of 5% in patients younger than 50 years of age[13]. Recently, the incidence of diverticulosis in younger patients has increased and has been reported to range from 18% to 34%[17,19,21,27]. In our study, 19.7% of patients with diverticulitis were aged 50 years or younger.
We report a male predominance in younger patients with acute diverticulitis, which is in agreement with previous studies[19,30].
Patients presenting with clinical signs, symptoms and laboratory findings suggesting acute diverticulitis underwent a CT scan of the abdomen at initial presentation. Abdominal CT is the diagnostic test of choice in acute diverticulitis. It has high sensitivity (approximately 93%-97%) and specificity approaching 100% for the diagnosis[31,32]. It is the best method for grading the severity of inflammation, and it also enables the classification of diverticulitis into complicated or uncomplicated disease[12,33].
In the present series, 17 patients (10.8%) with diffuse peritonitis and septic shock underwent emergency surgery. This observation is in agreement with other reports suggesting that 10% of patients admitted with acute diverticulitis will require surgical treatment during the same admission[34].
The clinical course and severity of diverticulitis in younger patients have not yet been clearly defined. The most appropriate management of patients whose acute diverticulitis resolves after medical treatment is still controversial. Diverticulitis in younger patients has been reported to have a more aggressive course or to require emergency surgery more frequently than in older patients[7,8,13,21,24,28,35-37]. As such, it has been argued that all patients younger than 50 should undergo elective colon resection after an initial episode of acute diverticulitis, with the intention to prevent a recurrent attack, which could present with perforation and require a stoma[11,17,36,37]. However, other studies published in the last decade failed to support this conclusion and suggest that the course of diverticulitis in younger patients is not as aggressive as once thought[12,19,27,38-41]. These studies reported a course of disease in younger patients not different from the older age group and with a similar rate of complications. Our study found that complicated episodes of acute diverticulitis were significantly less frequent in younger patients, suggesting that diverticulitis in younger patients does not have a more aggressive course.
In our study, younger patients were significantly more prone to recurrent episodes, despite the relatively short follow-up period. Our data is consistent with earlier studies that reported a higher recurrence rate of diverticulitis among younger patients[21,30].
The most appropriate timing for elective surgery following an episode of acute diverticulitis remains controversial. Parks first described medical management of diverticulitis in 1969[13]. He stated that mortality rate for each subsequent attack of diverticulitis increased from 4.7% during the first admission to 7.8% during each subsequent episode. He also reported that diverticulitis was less likely to respond to medical therapy as the number of acute episodes increased. Some retrospective studies suggested that the number of recurrences is associated with an increased need for a subsequent emergency surgical procedure[19]. It was suggested[42] that the likelihood of emergency surgery is increased by a factor of at least 2 with each subsequent hospitalization for diverticulitis. Thereafter, the American Society of Colon and Rectal Surgeons published a practice guideline for uncomplicated diverticulitis, which recommended definitive surgical treatment after 2 episodes of diverticulitis[11] based on the data provided by Parks. In recent years, this finding has been questioned and in its most recent guidelines, the American Society of Colon and Rectal Surgeons, no longer considers the number of attacks as a definite indication for surgery[41].
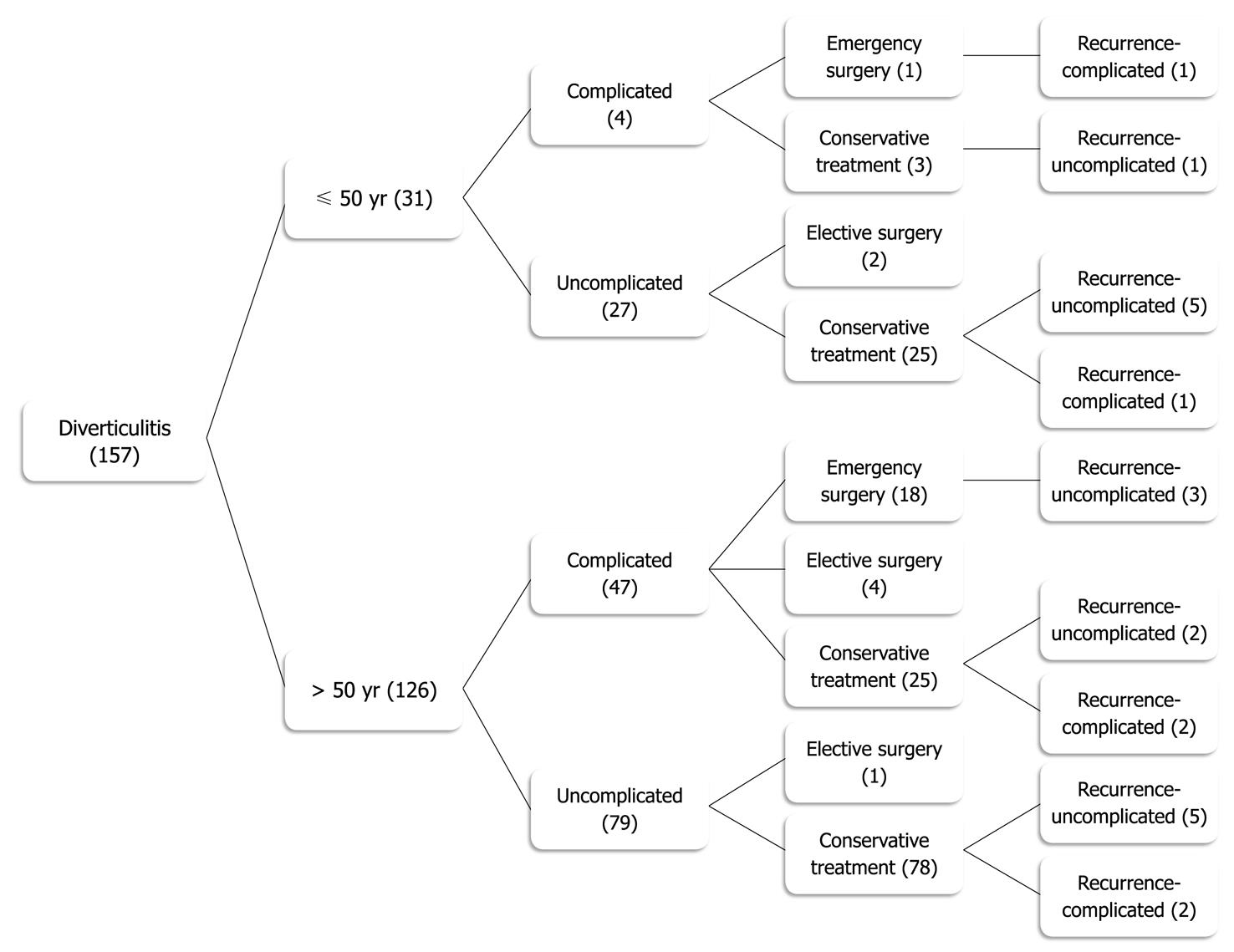
In this study, we have observed that in the subgroup of 18 patients, which developed acute recurrence after nonoperative treatment (Figure 1), only 5 (27.8%) had complicated findings on CT scan, classified as Hinchey stage I. All patients that developed acute recurrence were successfully treated nonoperatively. These results suggest that recurrence is not associated with an increased rate of either complications or less successful medical management, and are in agreement with several recent publications that have questioned the practice of surgical resection after 2 attacks, as well as after complicated disease[16-21,43,44].
According to our data, the great majority of younger patients (87.1%) had uncomplicated diverticulitis according to the CT scan criteria. Twenty-five of the younger patients (81%), after a first episode of uncomplicated diverticulitis, did not require surgery during follow-up. Although a significant amount of these patients developed recurrent episodes (24.0%), only one developed a recurrence with a pericolonic abscess (Hinchey stage I) and none required emergency surgery. This data suggests that younger patients with uncomplicated diverticulitis may be managed safely without an operation after an initial episode of diverticulitis, which is in agreement with other recent reports[45]. Three patients of group A (< 50 years old) with complicated diverticulitis did not require surgery during follow-up (Figure 1). One patient developed a noncomplicated recurrence and none required an emergency operation. Although this may suggest that complicated diverticulitis may also be managed without an operation, the number of patients was too small to allow for any recommendation. These results are in agreement with another recent report[46].
In conclusion, our study shows that acute diverticulitis is a mild disease with a low complication rate. In younger patients, there is a male predominance and the disease tends to be less severe than in the older age group, in spite of the higher recurrence rate. According to our results, the clinical course of uncomplicated acute diverticulitis is similar both in younger and older patients. Therefore the same guidelines should be used in the treatment of both groups of patients. The number of younger patients with complicated diverticulitis evaluated in this study was small but, should our results be confirmed in larger or even prospective studies, it seems reasonable to recommend the adoption of a similar strategy in both age groups.
The management of patients with diverticular disease after resolution of an attack of acute diverticulitis, has been a matter of debate by colon and rectal surgeons. The reports of a more severe and virulent disease in younger patients has been questioned by several authors and although the incidence has increased in the last decades, these patients are increasingly managed conservatively.
To compare the natural history and the course of acute diverticulitis in a younger age group with an older population and to evaluate whether younger patients should be managed differently.
In this retrospective study, the patients younger than 50 years at first presentation of diverticulitis had less severe disease but a higher recurrence rate (25.8% vs 11.1%). All the patients with recurrent disease were managed with conservative treatment and for the majority of patients, the most severe attack of diverticulitis was the first. On multivariate analysis, age was the only factor predictive of recurrence.
Based on the results of this study, we recommend that diverticulitis management should be based on the severity of the disease and not on the age of the patient.
Overall this is a good observational study, with interesting conclusions. Its strength is that all patients had computed tomography-documented episodes of diverticulitis and good follow-up.
Peer reviewers: Imran Hassan, MD, Assistant Professor, Department of Surgery, SIU School of Medicine, 701 North Rutledge, PO Box 19638, Springfield, IL 62794, United States; Jon C Gould, MD, FACS, Associate Professor of Surgery, University of Wisconsin School of Medicine and Public Health, 600 Highland Avenue, H4/726, Madison, WI 53792, United States
S- Editor Tian L L- Editor Cant MR E- Editor Lin YP
| 1. | Painter NS, Burkitt DP. Diverticular disease of the colon, a 20th century problem. Clin Gastroenterol. 1975;4:3-21. |
| 2. | Ferzoco LB, Raptopoulos V, Silen W. Acute diverticulitis. N Engl J Med. 1998;338:1521-1526. |
| 3. | Parks TG. Natural history of diverticular disease of the colon. Clin Gastroenterol. 1975;4:53-69. |
| 4. | Hughes LE. Postmortem survey of diverticular disease of the colon. II. The muscular abnormality of the sigmoid colon. Gut. 1969;10:344-351. |
| 5. | Pohlman T. Diverticulitis. Gastroenterol Clin North Am. 1988;17:357-385. |
| 6. | Floch MH, White JA. Management of diverticular disease is changing. World J Gastroenterol. 2006;12:3225-3228. |
| 7. | Schauer PR, Ramos R, Ghiatas AA, Sirinek KR. Virulent diverticular disease in young obese men. Am J Surg. 1992;164:443-446; discussion 446-448. |
| 8. | Ambrosetti P, Robert JH, Witzig JA, Mirescu D, Mathey P, Borst F, Rohner A. Acute left colonic diverticulitis in young patients. J Am Coll Surg. 1994;179:156-160. |
| 10. | Farrell RJ, Farrell JJ, Morrin MM. Diverticular disease in the elderly. Gastroenterol Clin North Am. 2001;30:475-496. |
| 11. | Wong WD, Wexner SD, Lowry A, Vernava A 3rd, Burnstein M, Denstman F, Fazio V, Kerner B, Moore R, Oliver G. Practice parameters for the treatment of sigmoid diverticulitis--supporting documentation. The Standards Task Force. The American Society of Colon and Rectal Surgeons. Dis Colon Rectum. 2000;43:290-297. |
| 12. | Kaiser AM, Jiang JK, Lake JP, Ault G, Artinyan A, Gonzalez-Ruiz C, Essani R, Beart RW Jr. The management of complicated diverticulitis and the role of computed tomography. Am J Gastroenterol. 2005;100:910-917. |
| 13. | Parks TG. Natural history of diverticular disease of the colon. A review of 521 cases. Br Med J. 1969;4:639-642. |
| 15. | Janes S, Meagher A, Frizelle FA. Elective surgery after acute diverticulitis. Br J Surg. 2005;92:133-142. |
| 16. | Haglund U, Hellberg R, Johnsén C, Hultén L. Complicated diverticular disease of the sigmoid colon. An analysis of short and long term outcome in 392 patients. Ann Chir Gynaecol. 1979;68:41-46. |
| 17. | Guzzo J, Hyman N. Diverticulitis in young patients: is resection after a single attack always warranted? Dis Colon Rectum. 2004;47:1187-1190; discussion 1190-1191. |
| 18. | Farmakis N, Tudor RG, Keighley MR. The 5-year natural history of complicated diverticular disease. Br J Surg. 1994;81:733-735. |
| 19. | Biondo S, Parés D, Martí Ragué J, Kreisler E, Fraccalvieri D, Jaurrieta E. Acute colonic diverticulitis in patients under 50 years of age. Br J Surg. 2002;89:1137-1141. |
| 20. | Alexander J, Karl RC, Skinner DB. Results of changing trends in the surgical management of complications of diverticular disease. Surgery. 1983;94:683-690. |
| 21. | Mäkelä J, Vuolio S, Kiviniemi H, Laitinen S. Natural history of diverticular disease: when to operate? Dis Colon Rectum. 1998;41:1523-1528. |
| 22. | Ouriel K, Schwartz SI. Diverticular disease in the young patient. Surg Gynecol Obstet. 1983;156:1-5. |
| 23. | Rodkey GV, Welch CE. Changing patterns in the surgical treatment of diverticular disease. Ann Surg. 1984;200:466-478. |
| 24. | Freischlag J, Bennion RS, Thompson JE Jr. Complications of diverticular disease of the colon in young people. Dis Colon Rectum. 1986;29:639-643. |
| 25. | Chappuis CW, Cohn I Jr. Acute colonic diverticulitis. Surg Clin North Am. 1988;68:301-313. |
| 26. | Simonowitz D, Paloyan D. Diverticular disease of the colon in patients under 40 years of age. Am J Gastroenterol. 1977;67:69-72. |
| 27. | Schweitzer J, Casillas RA, Collins JC. Acute diverticulitis in the young adult is not "virulent.". Am Surg. 2002;68:1044-1047. |
| 28. | Minardi AJ Jr, Johnson LW, Sehon JK, Zibari GB, McDonald JC. Diverticulitis in the young patient. Am Surg. 2001;67:458-461. |
| 29. | Hinchey EJ, Schaal PG, Richards GK. Treatment of perforated diverticular disease of the colon. Adv Surg. 1978;12:85-109. |
| 30. | Lahat A, Menachem Y, Avidan B, Yanai H, Sakhnini E, Bardan E, Bar-Meir S. Diverticulitis in the young patient--is it different ? World J Gastroenterol. 2006;12:2932-2935. |
| 31. | Ambrosetti P, Grossholz M, Becker C, Terrier F, Morel P. Computed tomography in acute left colonic diverticulitis. Br J Surg. 1997;84:532-534. |
| 32. | Cho KC, Morehouse HT, Alterman DD, Thornhill BA. Sigmoid diverticulitis: diagnostic role of CT--comparison with barium enema studies. Radiology. 1990;176:111-115. |
| 33. | Ambrosetti P, Jenny A, Becker C, Terrier TF, Morel P. Acute left colonic diverticulitis--compared performance of computed tomography and water-soluble contrast enema: prospective evaluation of 420 patients. Dis Colon Rectum. 2000;43:1363-1367. |
| 34. | Stollman NH, Raskin JB. Diverticular disease of the colon. J Clin Gastroenterol. 1999;29:241-252. |
| 35. | Chautems RC, Ambrosetti P, Ludwig A, Mermillod B, Morel P, Soravia C. Long-term follow-up after first acute episode of sigmoid diverticulitis: is surgery mandatory?: a prospective study of 118 patients. Dis Colon Rectum. 2002;45:962-966. |
| 36. | Anderson DN, Driver CP, Davidson AI, Keenan RA. Diverticular disease in patients under 50 years of age. J R Coll Surg Edinb. 1997;42:102-104. |
| 38. | Spivak H, Weinrauch S, Harvey JC, Surick B, Ferstenberg H, Friedman I. Acute colonic diverticulitis in the young. Dis Colon Rectum. 1997;40:570-574. |
| 39. | Vignati PV, Welch JP, Cohen JL. Long-term management of diverticulitis in young patients. Dis Colon Rectum. 1995;38:627-629. |
| 40. | West SD, Robinson EK, Delu AN, Ligon RE, Kao LS, Mercer DW. Diverticulitis in the younger patient. Am J Surg. 2003;186:743-746. |
| 41. | Rafferty J, Shellito P, Hyman NH, Buie WD. Practice parameters for sigmoid diverticulitis. Dis Colon Rectum. 2006;49:939-944. |
| 42. | Jacobs DO. Clinical practice. Diverticulitis. N Engl J Med. 2007;357:2057-2066. |
| 43. | Floch CL. Emergent and elective surgery for diverticulitis. J Clin Gastroenterol. 2008;42:1152-1153. |
| 44. | Issa N, Dreznik Z, Dueck DS, Arish A, Ram E, Kraus M, Gutman M, Neufeld D. Emergency surgery for complicated acute diverticulitis. Colorectal Dis. 2009;11:198-202. |
| 45. | Chapman JR, Dozois EJ, Wolff BG, Gullerud RE, Larson DR. Diverticulitis: a progressive disease? Do multiple recurrences predict less favorable outcomes? Ann Surg. 2006;243:876-830; discussion 880-883. |
| 46. | Nelson RS, Velasco A, Mukesh BN. Management of diverticulitis in younger patients. Dis Colon Rectum. 2006;49:1341-1345. |