Published online Feb 14, 2010. doi: 10.3748/wjg.v16.i6.663
Revised: November 26, 2009
Accepted: December 3, 2009
Published online: February 14, 2010
The carcinogenic potential of iron in colorectal cancer (CRC) is not fully understood. Iron is able to undergo reduction and oxidation, making it important in many physiological processes. This inherent redox property of iron, however, also renders it toxic when it is present in excess. Iron-mediated generation of reactive oxygen species via the Fenton reaction, if uncontrolled, may lead to cell damage as a result of lipid peroxidation and oxidative DNA and protein damage. This may promote carcinogenesis through increased genomic instability, chromosomal rearrangements as well as mutations of proto-oncogenes and tumour suppressor genes. Carcinogenesis is also affected by inflammation which is exacerbated by iron. Population studies indicate an association between high dietary iron intake and CRC risk. In this editorial, we examine the link between iron-induced oxidative stress and inflammation on the pathogenesis of CRC.
- Citation: Chua AC, Klopcic B, Lawrance IC, Olynyk JK, Trinder D. Iron: An emerging factor in colorectal carcinogenesis. World J Gastroenterol 2010; 16(6): 663-672
- URL: https://www.wjgnet.com/1007-9327/full/v16/i6/663.htm
- DOI: https://dx.doi.org/10.3748/wjg.v16.i6.663
Colorectal cancer (CRC) is the second most common cancer in developed countries. Apart from genetic mutations, environmental factors appear to play a role in intestinal carcinogenesis. Results from numerous population studies support the idea that dietary iron and/or elevated iron levels increase the risk of cancers including CRC, hepatocellular carcinoma (HCC) and lung cancer[1-6]. HCC occurs at a higher incidence in hereditary haemochromatosis (HH) patients with hepatic iron overload than in the normal population[7] and recently, an increased risk for CRC and breast cancer development in patients with HH has also been demonstrated[8,9]. Further support for a role of iron in carcinogenesis comes from animal studies. Multiple injections of iron compounds, such as iron dextran complex, ferric nitriloacetate and ferric saccharate in rodents result in the formation of sarcomas, renal cell carcinoma and mesothelioma, respectively[10-12]. Iron has also been implicated in intestinal carcinogenesis in rodent models of CRC[13,14].
Iron, whilst indispensable for life, can cause tissue injury through the formation of reactive oxygen species (ROS) and the high oxidative potential of iron and its participation in oxidative stress-related carcinogenesis have been reviewed in detail elsewhere[15-17]. The excessive generation of oxidative stress can lead to carcinogenic events, and this has been the premise for the hypothesis that high iron levels may potentiate the risk of cancer. In addition, iron is a source of sustenance for cancer cell growth and proliferation. Cancer cell growth is enhanced by iron administration and has been shown to be retarded by both dietary iron deprivation[18] and treatment with iron chelators[19-21]. It is thought that genetic modifications and continual activation of the signalling pathways of cell proliferation by ROS synergistically promote carcinogenesis[16,22]. Chronic inflammation also induces cell oxidative stress, which promotes the onset of dysplasia[23] and is accompanied by a dysregulation in iron metabolism[24]. Nonetheless, the mechanistic link among iron, oxidative stress, inflammation and colorectal carcinogenesis remains to be elucidated.
The incidence of CRC varies among countries and this has been mainly attributed to environmental factors, although genetic factors are also important. Environmental risk factors for colorectal carcinogenesis include many dietary factors such as high red meat and alcohol consumption as well as low fibre and vegetable intake[25,26].
The majority of CRCs originate from pre-existing adenomatous polyps of the colonic mucosa[27]. These are defined as well demarcated masses of epithelial mucosa with increased crypt proliferation. Eventually, neoplastic cells migrate through the muscularis mucosa and it is once the basement membrane surrounding these cells is breached that the lesions are classified as malignant. These morphological and histopathological changes are accompanied by sequential dysregulation of key molecular pathways of cell division and tissue homeostasis[28]. Several syndromes have been described in families with a history of CRC, which involve mutations in components of these pathways[29]. Affected persons with familial adenomatous polyposis (FAP) develop hundreds of adenomatous polyps throughout their lifetime, some of which inevitably progress to malignancy. Genetic studies in subjects with FAP led to the discovery of the adenomatous polyposis coli (APC) gene, a key gene in the regulation of mucosal epithelial maturation via the Wnt signalling pathway[30,31]. In contrast, hereditary non-polyposis colorectal cancer (HNPCC) is characterised by an increased risk for developing CRC in the absence of a germ line mutation in the APC gene and is typically accompanied by the loss of DNA mismatch repair genes which impacts on other signalling pathways[32,33]. Although there is currently no evidence that iron plays a role in the pathology of these syndromes, it is interesting to note that iron can increase Wnt signalling in the absence of APC[34] and that mutations in the haemochromatosis gene (HFE) act as a genetic modifier of HNPCC disease expression[35].
Although there is a strong role for genes in the pathogenesis of CRC in the above-mentioned risk groups, environmental factors seem to play a more significant role. Population based studies have shown that in immigrant groups, the incidence of CRC changes towards that observed in the host country[36,37]. Another indication of the importance of environmental factors is that in Japan, a country with a traditionally low incidence of CRC, the rate has rapidly increased in recent years, a circumstance that has been primarily attributed to changes in life-style in the recent decades[29]. It is also interesting to note, that one of the highest incidences of CRC in the United States can be found in Japanese Hawaiians, highlighting the significance of environment vs genes[38].
Of the environmental risk factors, the diet is of particular interest since it impacts on the composition of the intestinal luminal contents, which are in direct contact with the colonic mucosa. Diets between countries vary significantly in their iron content and iron-rich food components, suggesting that iron intake could be one of the factors influencing CRC incidence in different populations. Dietary iron as an environmental modifier of CRC has been examined in population-based studies and there is evidence that both dietary iron[4,5,39] and/or increased body iron stores[1,2,40] enhance the risk of CRC.
Iron is a vital trace element participating in numerous biological and cellular processes such as electron transfer, oxygen transport and DNA synthesis as well as cell cycle progression and growth[41]. Iron absorption from the diet occurs mainly in the duodenum by a tightly regulated process. Most of the absorbed iron is utilized for erythropoiesis and any excessive iron is stored mainly in the liver[42]. Dietary iron occurs in two forms, haem iron from red meat and non-haem iron from plants and dairy products. Both forms of iron are taken up from the intestinal lumen into the enterocyte by different pathways. Haem iron is taken up as an intact metalloporphyrin by a haem transporter, the identity of which has yet to be confirmed. After entering the enterocyte, haem is broken down by haem oxygenase into free iron, biliverdin that is rapidly converted to bilirubin and carbon monoxide[43,44]. In contrast, non-haem ferric iron is reduced to ferrous iron by a ferrireductase and is then taken up by divalent metal transporter 1 at the apical surface of enterocytes. The iron from both sources enters a common intracellular iron pool and is stored as ferritin or transferred across the basolateral membrane of the enterocyte into the circulation by ferroportin. Upon release, iron is oxidised by hephaestin and binds to plasma transferrin. Transferrin-bound iron is taken up by cells via transferrin receptors. Iron absorption is inversely regulated by body iron levels, increasing during iron deficiency and decreasing in conditions of iron excess. Iron metabolism is regulated by the hepatic hormone, hepcidin, and its expression is controlled by many factors including iron stores, hypoxia, inflammation, anaemia and erythropoiesis[45,46]. The regulation of cellular iron metabolism has been extensively reviewed elsewhere[42,47-49].
The association between dietary iron and CRC risk has been examined in many population-based studies. A meta-analysis of studies investigating dietary iron intake, body iron stores and CRC demonstrated a positive correlation between iron in the diet and CRC risk[50]. Notably, two large prospective cohort studies have found that high iron intake and CRC risk were associated with other factors such as a high fat diet or bile acids[4,5] and at least three other case control studies have corroborated the positive correlation between dietary iron and CRC[39,51,52]. Of the studies analysing body iron stores and CRC, one large cohort study observed an association between transferrin saturation and CRC risk[2] whilst three case control studies found a positive correlation between serum ferritin levels and the formation of colorectal adenomatous polyps[1,40,53]. Other studies, however, reported inverse correlations between transferrin saturation[54] or ferritin levels[5] and CRC risk. The role of body iron stores in CRC appears more complex than that of dietary iron and the influence of genetic factors on body iron stores will be discussed in more detail below.
The effect of high red meat consumption, as a dietary source of iron, on the pathogenesis of CRC has been of considerable interest. Red meat is a major component of the human diet in some societies and contains a high amount of myoglobin and haemoglobin. Both contain haem, a porphyrin structure that contains a central iron atom and it has been suggested that the haem content in red meat promotes colorectal carcinogenesis[55,56]. A meta-analysis of 48 studies specifically addressing red meat consumption showed a significantly increased risk of developing CRC in people with a high intake of red meat as well as processed meat in most of the studies[57]. Of interest is a recent very large prospective cohort study investigating nutrition and disease that described an increased risk of CRC in people who consumed red meat rich in haem, whilst no increased risk was identified for poultry and an inverse correlation was observed for fish, both of which have a lower haem content[58]. Another two prospective cohort studies also reported that haem iron was associated with a higher risk of CRC especially in those who consumed alcohol[59] or those with a low intake of chlorophyll[26]. It is, however, unclear whether the effects of red meat on colorectal carcinogenesis are due to haem, the iron bound to haem, or a combination of both.
HH is a common disorder of iron metabolism that usually results from a homozygous C282Y mutation in the HFE gene. HFE protein is a key regulator of hepcidin, and in HH, the HFE-mediated regulation of hepcidin is impaired resulting in excessive absorption of iron and increased deposition of iron primarily in the liver[47,60]. As mentioned earlier, these individuals have an increased risk of developing CRC. This is exemplified by the recent findings that patients homozygous for the C282Y mutation have a 2.4-fold increased risk of developing CRC[9]. This is of particular significance considering that the C282Y mutation is one of the most abundant autosomal mutations in some Western societies with homozygosity rates ranging from 1/102 in Northern Ireland to less than 1/100 000 in Greece[61]. The homozygosity rate of 1/385 in the United States of America calculates as an estimated number of 718 000 affected individuals with an increased risk of CRC[61]. Another study describing a 7.7-fold increased risk of developing CRC in patients with homozygous mutations in both HFE and TFR1 genes, further implicates a dysregulation in iron metabolism as a possible mechanism contributing to colorectal carcinogenesis[62]. An increased risk for CRC has also been described for compound C282Y/H63D heterozygotes, single C282Y or H63D heterozygotes and H63D homozygotes[35,63-65], but not all studies detected a significant correlation between HFE mutations and CRC[65-73]. Knekt et al[2], however, reported a 3-fold increased risk for CRC that was associated with a transferrin saturation level exceeding 60% in a large cohort from Finland, which may include subjects with mutations in the HFE gene. Furthermore, systemic iron reduction by phlebotomy decreases visceral malignancies and mortality in patients with peripheral arterial disease[74] and in blood donors the number of non-haematological malignancies is significantly reduced[75] indicating that reduction of iron levels might decrease CRC risk. The availability of mouse models of HH enables future studies to investigate the interaction of dietary iron, regulation of body iron stores and colorectal carcinogenesis.
There is evidence suggesting that significant iron absorption may occur in the colon[76,77]. Increased iron intake results in higher levels of iron in colonic epithelial cells in rats[78], and although divalent metal transporter 1, ferroportin and hephaestin mRNA expression is highest in the duodenum and decreases along with the length of the small intestine[79], their expression is still relatively high in the colon, especially hephaestin and ferroportin. These findings suggest that there may be significant colonic iron transport, which impacts on cell proliferation and cancer development. In a recent study, it was shown that there was increased iron staining in human colorectal tumours[80]. The expression of proteins involved in cellular iron uptake such as divalent metal transporter 1 and transferrin receptor 1 was also upregulated. The expression of the iron exporter, ferroportin, was increased but it was located intracellularly whilst hephaestin expression was decreased, suggesting decreased release of iron from the cells. These results suggest that the retention of iron by tumours may facilitate cell proliferation.
Further support for the concept of iron as a risk factor for CRC has been demonstrated in animal studies. Elevated dietary iron levels increased the incidence of tumours in rodent models of CRC induced by inflammatory or carcinogenic agents[13,14]. In these experiments, however, iron was supplemented with inorganic carbonyl iron, a form that does not constitute a major component of natural diets. Interestingly, in the inflammatory model, Seril and colleagues demonstrated that systemic iron supplementation did not increase tumour incidence. This suggests that increased luminal iron but not systemic iron levels increase colorectal carcinogenesis in an inflammatory model of CRC[81]. In a carcinogen-induced CRC model, the number of preneoplastic lesions increased with the amount of haem in the diet[82]. Haem iron is more bioavailable than non-haem iron and has been shown to increase mucosal proliferation and cytotoxicity, indicating that haem may have a greater propensity for inducing malignancy compared with other forms of dietary iron[55,56]. Haem has been shown to stimulate the production of endogenous N-nitroso compounds in the large intestine after red meat ingestion, many of which are pro-carcinogenic[83]. The genotoxic effect of haem has also been demonstrated in human colonic cells, where DNA damage was induced by haemoglobin and haemin[84]. The exact mechanism by which luminal iron (haem and/or non-haem), iron transport, systemic iron levels and their regulation impact the pathogenesis of CRC, however, remains to be elucidated.
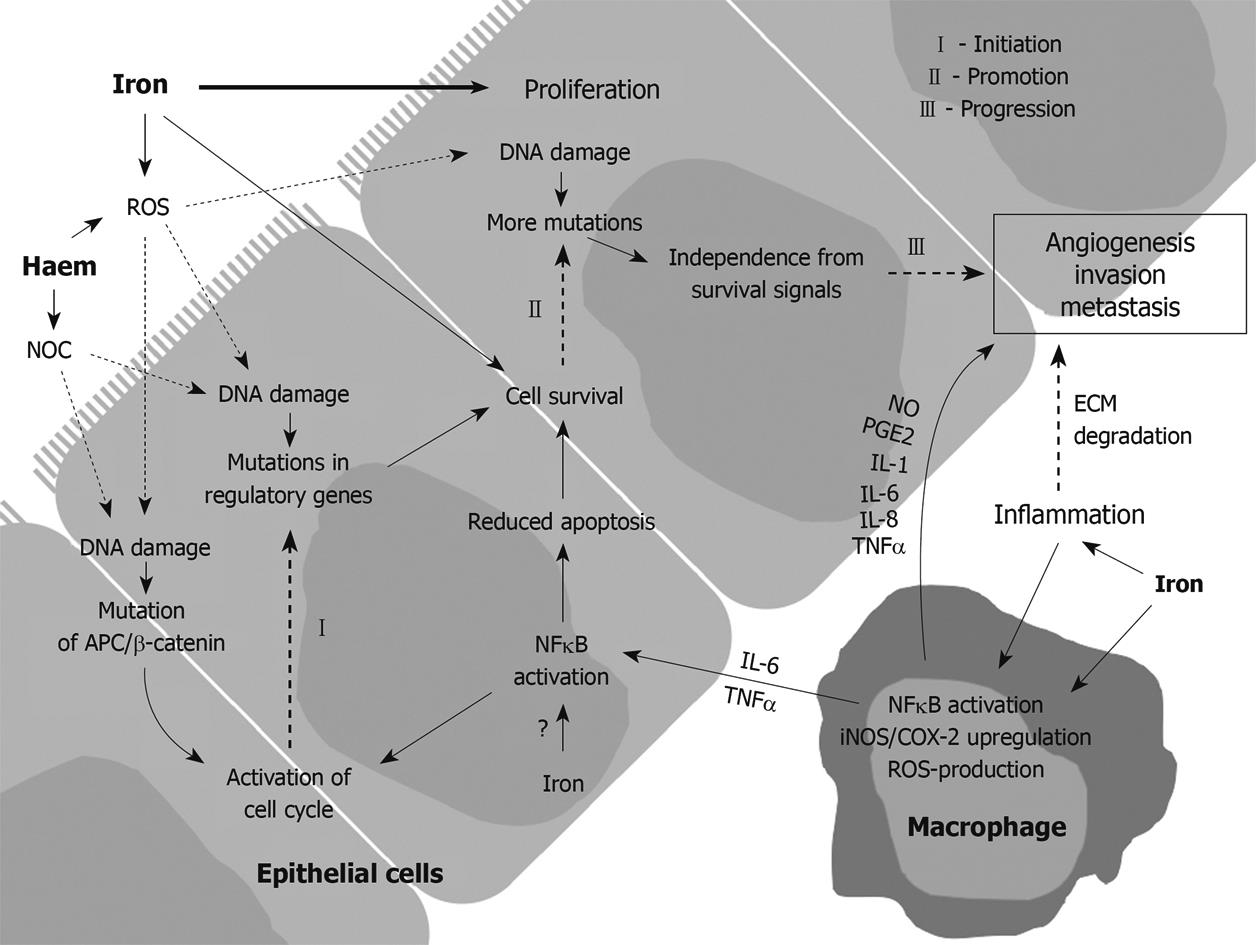
Colorectal carcinogenesis is a multi-step process involving the formation of adenomatous polyps and their subsequent progression to malignancy. At the molecular level, this process is reflected by sequential events of gene mutation and activation of key molecular pathways[85,86]. Some of these pathways may be altered by iron and iron-mediated generation of ROS (Figure 1). The APC gene was initially identified in patients with FAP and subsequently shown to be mutated in > 80% of human colorectal neoplasia[85]. APC mutations involved in carcinogenesis led to nuclear accumulation of β-catenin and constitutive activation of the wnt/β-catenin/T cell factor (TCF) signalling pathway. In rodent models of azoxymethane-induced CRC, the majority of colonic tumours harbour mutations in APC and/or β-catenin genes[87,88]. Activation of the wnt/β-catenin/TCF pathway results in increased expression of cyclin D1 and c-myc, both of which are positive regulators of cell proliferation[89,90]. Iron has been implicated in APC loss[34] and iron chelators decrease the expression of cyclin D1 and c-myc[41].
The molecular pathogenesis of ulcerative colitis-associated colorectal carcinogenesis has been extensively studied[91]. Events such as chromosomal and microsatellite instability and alterations in tumour suppressor genes (p53 and APC mutations) and DNA mismatch repair genes have been documented[92,93]. The inhibitor of NFκB kinase (IKKβ)/nuclear factor kappa B (NFκB) signalling pathway constitutes a key molecular link between inflammation and carcinogenesis. NFκB is activated in colorectal carcinogenesis and is influenced by both inflammation and oxidative stress[94]. NFκB targets the genes that control cell proliferation, apoptosis, angiogenesis and metastasis[94,95]. A direct stimulatory effect of iron on NFκB signalling has also been demonstrated in hepatic macrophages[96,97].
Other pathways involved in colorectal carcinogenesis include cyclo-oxygenase (COX)-2 mediated prostaglandin E2 synthesis and inducible nitric oxide synthase (iNOS)-mediated generation of nitric oxide[98]. Prostaglandin E2 is involved in regulating angiogenesis and inhibiting apoptosis[99] whilst iNOS activity induces DNA damage and promotes microvascularisation[100]. Both COX-2 and iNOS are frequently over-expressed in human CRC[101,102] and inhibition of their activity has been shown to decrease tumorigenesis in rodent models of CRC[103,104]. Furthermore, iNOS is a target of the wnt/β-catenin/TCF pathway and its production of ROS through nitric oxide is catalysed by iron[105].
Oxidative stress occurs when the body or cell is unable to combat the deleterious effects of overproduction of oxidants or free radicals due to decreased anti-oxidant activity to counterbalance or eliminate them. Oxidative stress is related to many pathological conditions such as infection, inflammation, iron and other transition metal overload. It has also been implicated in carcinogenesis[15,106,107]. Although many reactive species and free radicals such as reactive nitrogen species contribute to oxidative stress, the role of ROS in colorectal carcinogenesis will mainly be discussed here. ROS is generated through the partial reduction of oxygen which results in superoxide anion, singlet oxygen, hydrogen peroxide and hydroxyl radical formation. ROS plays a dual role in biological systems. When the balance between oxidant and anti-oxidant activity is maintained, ROS can participate as secondary messengers in intracellular signal transduction cascades, whilst the presence of excessive ROS induces tissue damage.
Iron is a strong oxidant and when present at high levels, it generates ROS via the Haber-Weiss-Fenton reaction. Iron-mediated generation of ROS can cause oxidative damage to lipids, nucleic acids or proteins[86]. Oxidative damage to proteins and lipids can generate reactive intermediates that can couple to DNA bases resulting in DNA lesions[86]. DNA damage as a consequence of prolonged oxidative stress can result in mutation of proto-oncogenes and tumour suppressor genes, microsatellite instability and chromosomal rearrangements as well as a dysregulation in transcription, signal transduction and replication, all of which are associated with carcinogenesis[15,86,108]. Haem is also an oxidant[109], and despite being essential for many biological processes and enzyme systems, excessive free haem catalyses ROS production, resulting in oxidative stress[110]. The degradation of haem by haem oxygenase-1 alleviates oxidative stress[111] and bilirubin, a by-product of haem breakdown, is anti-oxidative and has been shown to scavenge peroxyl radicals in plasma[112,113]. In mice lacking copper-and zinc-containing superoxide dismutase, oxidative damage is pervasive and the rate of liver cancer development is increased later in life[114] whilst, mice with decreased manganese-containing superoxide dismutase activity have an increased risk for lymphoma and adenocarcinoma[115]. These results suggest that reduced anti-oxidant activity can lead to cancer.
Oxidative stress is enhanced in neoplastic tissue from the colonic mucosa of CRC patients[116,117]. In these patients, lipid peroxidation is increased in colonic tumours compared with normal mucosa[116]. In addition, there is a greater extent of DNA strand breakage in colonic mucosal cells isolated from neoplastic tissues compared with normal tissues from cancer patients[117]. Oxidative damage is also more evident in the earlier stages of CRC than in the more advanced stages of cancer. The accumulation of iron in a human colon cancer cell line has been shown to correlate with increased oxidative protein and DNA damage[118]. In rodent studies, mice and rats fed a diet high in iron[119-122] and haem[82] exhibited greater lipid peroxidation activity in the colon and increased colonic aberrant crypt foci, which are pre-neoplastic lesions[82,123,124]. Oxidative damage markers are increased in the colons of Hfe knockout mice[125], indicating an increased presence of ROS in these iron-loaded mice. Oxidative stress due to high iron and/or haem levels may, therefore, be instrumental in mediating colorectal carcinogenesis.
The relationship between inflammation and tumour development has been a major focus in recent cancer research. Persistent inflammation as a result of infection promotes carcinogenesis; for example, infection with hepatitis B and C and human papilloma viruses are associated with HCC and cervical cancer, respectively, whilst Helicobacter pylori infection is linked to gastric cancer[126]. Furthermore, subjects with inflammatory bowel disease such as ulcerative colitis and Crohn’s disease suffer from recurring inflammation in the colonic mucosa and are at an increased risk of developing CRC[127].
Chronic inflammation and metabolites from phagocytic processes result in formation of excessive ROS and nitric oxide[98,128], which as mentioned above, can directly cause damage to DNA, protein or lipids. Inflammatory cells active in chronic inflammation are also present within a tumour and its surrounding tissue. This suggests that the presence of oxidative stress and the network of inflammatory cytokines and chemokines in a tumour microenvironment may perpetuate carcinogenesis by promoting genotoxicity, proliferation and survival as well as angiogenesis, cell invasion and metastasis[129]. Cytokines that are frequently associated with carcinogenesis include TNFα and IL-6, which promote cell proliferation and survival[130]. Angiogenesis, invasion and metastasis are influenced by the cytokines, TNFα, IL-1, -6 and -8[129].
CRC occurs in approximately 4% of patients with ulcerative colitis[131] where the risk for CRC has been reported to be approximately 10-fold higher than in the normal population[127,132]. The risk for cancer increases with longer duration and the extent of colon affected by this disease, and how well the inflammation is controlled, indicating that it is the prolonged inflammatory stimulus that directly affects the pathogenesis of CRC in these patients. In addition to the blood loss and iron deficiency due to the chronic intestinal inflammation, patients with ulcerative colitis and Crohn’s disease may also develop iron deficiency anaemia secondary to inflammation and reduced mobilization of bone marrow iron and are frequently treated with oral iron supplementation. Hence, the effects of iron on colitis-associated colorectal carcinogenesis have also been examined. Chronic inflammation induced in mice treated with dextran sodium sulphate resulted in colorectal tumorigenesis which became worse with dietary iron supplementation, indicating the tumour-promoting role of iron when inflammation was present[13]. This was accompanied by the increased presence of enhanced nitrotyrosine and iNOS expression, which implicates a role for oxidative stress in inflammation-associated carcinogenesis. Further evidence comes from experimental models where colitis is attenuated when anti-oxidant activity is increased[133,134]. In addition, the formation of pro-oxidants due to increased activity of phagocytic leukocytes in the colons of ulcerative colitis patients has been reported[135]. These findings suggest that oxidative stress induced by both inflammation and iron plays a major role in inflammation-associated colorectal carcinogenesis.
Better understanding about the relationship between inflammation and iron metabolism has been achieved since the identification of hepcidin. Inflammation affects iron homeostasis by inducing hepcidin through an IL-6-mediated pathway[45]. Increased hepcidin levels caused decreased iron absorption[136,137] as well as iron retention by reticulo-endothelial macrophages, which may result in hypoferraemia (low serum iron concentration)[138]. Hypoferraemia is associated with the anaemia of chronic disease, also known as anaemia of inflammation. Haem, like iron, is pro-inflammatory and increases the expression of inflammatory adhesion molecules in endothelial cells[109]. Haem oxygenase 1 knockout mice suffer from anaemia, tissue iron loading and severe inflammation, having enlarged spleens and lymph nodes, vasculitis and inflammatory cell infiltrates in the liver[139]. Carbon monoxide, a by-product of haem degradation, ameliorates inflammation in a mouse model of colitis[140]. High dietary iron and/or haem levels are likely to contribute to the pathogenesis of inflammation-associated colorectal carcinogenesis.
Population-based studies as well as animal studies point to a role for dietary and/or systemic body iron levels in colorectal carcinogenesis. The effect of high iron levels on regulatory pathways of iron metabolism through HFE and hepcidin as well as the increased production of ROS in the presence of iron provide potential mechanisms. The interference of high iron levels with ROS and/or inflammation and their effects on pathways involved in colorectal carcinogenesis remains poorly understood. Future studies in mouse models of HH, dietary iron overload and colorectal carcinogenesis will provide valuable insights into this fascinating aspect of iron biology and CRC.
Peer reviewer: William Dickey, MD, PhD, Altnagelvin Hospital, Londonderry, BT47 6SB, Northern Ireland, United Kingdom
S- Editor Tian L L- Editor Ma JY E- Editor Lin YP
| 1. | Nelson RL, Davis FG, Sutter E, Sobin LH, Kikendall JW, Bowen P. Body iron stores and risk of colonic neoplasia. J Natl Cancer Inst. 1994;86:455-460. [Cited in This Article: ] |
| 2. | Knekt P, Reunanen A, Takkunen H, Aromaa A, Heliövaara M, Hakulinen T. Body iron stores and risk of cancer. Int J Cancer. 1994;56:379-382. [Cited in This Article: ] |
| 3. | Hsing AW, McLaughlin JK, Olsen JH, Mellemkjar L, Wacholder S, Fraumeni JF Jr. Cancer risk following primary hemochromatosis: a population-based cohort study in Denmark. Int J Cancer. 1995;60:160-162. [Cited in This Article: ] |
| 4. | Wurzelmann JI, Silver A, Schreinemachers DM, Sandler RS, Everson RB. Iron intake and the risk of colorectal cancer. Cancer Epidemiol Biomarkers Prev. 1996;5:503-507. [Cited in This Article: ] |
| 5. | Kato I, Dnistrian AM, Schwartz M, Toniolo P, Koenig K, Shore RE, Zeleniuch-Jacquotte A, Akhmedkhanov A, Riboli E. Iron intake, body iron stores and colorectal cancer risk in women: a nested case-control study. Int J Cancer. 1999;80:693-698. [Cited in This Article: ] |
| 6. | Zhou W, Park S, Liu G, Miller DP, Wang LI, Pothier L, Wain JC, Lynch TJ, Giovannucci E, Christiani DC. Dietary iron, zinc, and calcium and the risk of lung cancer. Epidemiology. 2005;16:772-779. [Cited in This Article: ] |
| 7. | Niederau C, Fischer R, Sonnenberg A, Stremmel W, Trampisch HJ, Strohmeyer G. Survival and causes of death in cirrhotic and in noncirrhotic patients with primary hemochromatosis. N Engl J Med. 1985;313:1256-1262. [Cited in This Article: ] |
| 8. | Kallianpur AR, Hall LD, Yadav M, Christman BW, Dittus RS, Haines JL, Parl FF, Summar ML. Increased prevalence of the HFE C282Y hemochromatosis allele in women with breast cancer. Cancer Epidemiol Biomarkers Prev. 2004;13:205-212. [Cited in This Article: ] |
| 9. | Osborne NJ, Gurrin LC, Allen KJ, Constantine CC, Delatycki MB, McLaren CE, Gertig DM, Anderson GJ, Southey MC, Olynyk JK. HFE C282Y homozygotes are at increased risk of breast and colorectal cancer. Hepatology. 2010;In press. [Cited in This Article: ] |
| 10. | Richmond HG. Induction of sarcoma in the rat by iron-dextran complex. Br Med J. 1959;1:947-949. [Cited in This Article: ] |
| 11. | Li JL, Okada S, Hamazaki S, Ebina Y, Midorikawa O. Subacute nephrotoxicity and induction of renal cell carcinoma in mice treated with ferric nitrilotriacetate. Cancer Res. 1987;47:1867-1869. [Cited in This Article: ] |
| 12. | Okada S, Hamazaki S, Toyokuni S, Midorikawa O. Induction of mesothelioma by intraperitoneal injections of ferric saccharate in male Wistar rats. Br J Cancer. 1989;60:708-711. [Cited in This Article: ] |
| 13. | Seril DN, Liao J, Ho KL, Warsi A, Yang CS, Yang GY. Dietary iron supplementation enhances DSS-induced colitis and associated colorectal carcinoma development in mice. Dig Dis Sci. 2002;47:1266-1278. [Cited in This Article: ] |
| 14. | Ilsley JN, Belinsky GS, Guda K, Zhang Q, Huang X, Blumberg JB, Milbury PE, Roberts LJ 2nd, Stevens RG, Rosenberg DW. Dietary iron promotes azoxymethane-induced colon tumors in mice. Nutr Cancer. 2004;49:162-169. [Cited in This Article: ] |
| 15. | Klaunig JE, Kamendulis LM. The role of oxidative stress in carcinogenesis. Annu Rev Pharmacol Toxicol. 2004;44:239-267. [Cited in This Article: ] |
| 16. | Galaris D, Skiada V, Barbouti A. Redox signaling and cancer: the role of "labile" iron. Cancer Lett. 2008;266:21-29. [Cited in This Article: ] |
| 17. | Toyokuni S. Role of iron in carcinogenesis: cancer as a ferrotoxic disease. Cancer Sci. 2009;100:9-16. [Cited in This Article: ] |
| 18. | Weinberg ED. Iron withholding: a defense against infection and neoplasia. Physiol Rev. 1984;64:65-102. [Cited in This Article: ] |
| 19. | Kicic A, Chua AC, Baker E. The desferrithiocin (DFT) class of iron chelators: potential as antineoplastic agents. Anticancer Drug Des. 2001;16:195-207. [Cited in This Article: ] |
| 20. | Kicic A, Chua AC, Baker E. Effect of iron chelators on proliferation and iron uptake in hepatoma cells. Cancer. 2001;92:3093-3110. [Cited in This Article: ] |
| 21. | Yuan J, Lovejoy DB, Richardson DR. Novel di-2-pyridyl-derived iron chelators with marked and selective antitumor activity: in vitro and in vivo assessment. Blood. 2004;104:1450-1458. [Cited in This Article: ] |
| 22. | Toyokuni S, Okamoto K, Yodoi J, Hiai H. Persistent oxidative stress in cancer. FEBS Lett. 1995;358:1-3. [Cited in This Article: ] |
| 23. | Rakoff-Nahoum S. Why cancer and inflammation? Yale J Biol Med. 2006;79:123-130. [Cited in This Article: ] |
| 24. | Ganz T, Nemeth E. Iron sequestration and anemia of inflammation. Semin Hematol. 2009;46:387-393. [Cited in This Article: ] |
| 25. | Bingham SA, Day NE, Luben R, Ferrari P, Slimani N, Norat T, Clavel-Chapelon F, Kesse E, Nieters A, Boeing H. Dietary fibre in food and protection against colorectal cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC): an observational study. Lancet. 2003;361:1496-1501. [Cited in This Article: ] |
| 26. | Balder HF, Vogel J, Jansen MC, Weijenberg MP, van den Brandt PA, Westenbrink S, van der Meer R, Goldbohm RA. Heme and chlorophyll intake and risk of colorectal cancer in the Netherlands cohort study. Cancer Epidemiol Biomarkers Prev. 2006;15:717-725. [Cited in This Article: ] |
| 27. | Tanaka T. Colorectal carcinogenesis: Review of human and experimental animal studies. J Carcinog. 2009;8:5. [Cited in This Article: ] |
| 28. | Vogelstein B, Fearon ER, Hamilton SR, Kern SE, Preisinger AC, Leppert M, Nakamura Y, White R, Smits AM, Bos JL. Genetic alterations during colorectal-tumor development. N Engl J Med. 1988;319:525-532. [Cited in This Article: ] |
| 29. | Potter JD. Colorectal cancer: molecules and populations. J Natl Cancer Inst. 1999;91:916-932. [Cited in This Article: ] |
| 30. | Bodmer WF, Bailey CJ, Bodmer J, Bussey HJ, Ellis A, Gorman P, Lucibello FC, Murday VA, Rider SH, Scambler P. Localization of the gene for familial adenomatous polyposis on chromosome 5. Nature. 1987;328:614-616. [Cited in This Article: ] |
| 31. | Leppert M, Dobbs M, Scambler P, O'Connell P, Nakamura Y, Stauffer D, Woodward S, Burt R, Hughes J, Gardner E. The gene for familial polyposis coli maps to the long arm of chromosome 5. Science. 1987;238:1411-1413. [Cited in This Article: ] |
| 32. | Bronner CE, Baker SM, Morrison PT, Warren G, Smith LG, Lescoe MK, Kane M, Earabino C, Lipford J, Lindblom A. Mutation in the DNA mismatch repair gene homologue hMLH1 is associated with hereditary non-polyposis colon cancer. Nature. 1994;368:258-261. [Cited in This Article: ] |
| 33. | Rodriguez-Bigas MA, Boland CR, Hamilton SR, Henson DE, Jass JR, Khan PM, Lynch H, Perucho M, Smyrk T, Sobin L. A National Cancer Institute Workshop on Hereditary Nonpolyposis Colorectal Cancer Syndrome: meeting highlights and Bethesda guidelines. J Natl Cancer Inst. 1997;89:1758-1762. [Cited in This Article: ] |
| 34. | Brookes MJ, Boult J, Roberts K, Cooper BT, Hotchin NA, Matthews G, Iqbal T, Tselepis C. A role for iron in Wnt signalling. Oncogene. 2008;27:966-975. [Cited in This Article: ] |
| 35. | Shi Z, Johnstone D, Talseth-Palmer BA, Evans TJ, Spigelman AD, Groombridge C, Milward EA, Olynyk JK, Suchy J, Kurzawski G. Haemochromatosis HFE gene polymorphisms as potential modifiers of hereditary nonpolyposis colorectal cancer risk and onset age. Int J Cancer. 2009;125:78-83. [Cited in This Article: ] |
| 36. | Haenszel W. Cancer mortality among the foreign-born in the United States. J Natl Cancer Inst. 1961;26:37-132. [Cited in This Article: ] |
| 37. | McMichael AJ, Giles GG. Cancer in migrants to Australia: extending the descriptive epidemiological data. Cancer Res. 1988;48:751-756. [Cited in This Article: ] |
| 38. | Parkin DM, Muir CS. Cancer Incidence in Five Continents. Comparability and quality of data. IARC Sci Publ. 1992;45-173. [Cited in This Article: ] |
| 39. | Senesse P, Meance S, Cottet V, Faivre J, Boutron-Ruault MC. High dietary iron and copper and risk of colorectal cancer: a case-control study in Burgundy, France. Nutr Cancer. 2004;49:66-71. [Cited in This Article: ] |
| 40. | Bird CL, Witte JS, Swendseid ME, Shikany JM, Hunt IF, Frankl HD, Lee ER, Longnecker MP, Haile RW. Plasma ferritin, iron intake, and the risk of colorectal polyps. Am J Epidemiol. 1996;144:34-41. [Cited in This Article: ] |
| 41. | Le NT, Richardson DR. The role of iron in cell cycle progression and the proliferation of neoplastic cells. Biochim Biophys Acta. 2002;1603:31-46. [Cited in This Article: ] |
| 42. | Chua AC, Graham RM, Trinder D, Olynyk JK. The regulation of cellular iron metabolism. Crit Rev Clin Lab Sci. 2007;44:413-459. [Cited in This Article: ] |
| 43. | Raffin SB, Woo CH, Roost KT, Price DC, Schmid R. Intestinal absorption of hemoglobin iron-heme cleavage by mucosal heme oxygenase. J Clin Invest. 1974;54:1344-1352. [Cited in This Article: ] |
| 44. | Montellano PR. The mechanism of heme oxygenase. Curr Opin Chem Biol. 2000;4:221-227. [Cited in This Article: ] |
| 45. | Nicolas G, Chauvet C, Viatte L, Danan JL, Bigard X, Devaux I, Beaumont C, Kahn A, Vaulont S. The gene encoding the iron regulatory peptide hepcidin is regulated by anemia, hypoxia, and inflammation. J Clin Invest. 2002;110:1037-1044. [Cited in This Article: ] |
| 46. | Viatte L, Vaulont S. Hepcidin, the iron watcher. Biochimie. 2009;91:1223-1228. [Cited in This Article: ] |
| 47. | Olynyk JK, Trinder D, Ramm GA, Britton RS, Bacon BR. Hereditary hemochromatosis in the post-HFE era. Hepatology. 2008;48:991-1001. [Cited in This Article: ] |
| 48. | Fleming MD. The regulation of hepcidin and its effects on systemic and cellular iron metabolism. Hematology Am Soc Hematol Educ Program. 2008;151-158. [Cited in This Article: ] |
| 49. | Zhang AS, Enns CA. Iron homeostasis: recently identified proteins provide insight into novel control mechanisms. J Biol Chem. 2009;284:711-715. [Cited in This Article: ] |
| 50. | Nelson RL. Iron and colorectal cancer risk: human studies. Nutr Rev. 2001;59:140-148. [Cited in This Article: ] |
| 51. | Freudenheim JL, Graham S, Marshall JR, Haughey BP, Wilkinson G. A case-control study of diet and rectal cancer in western New York. Am J Epidemiol. 1990;131:612-624. [Cited in This Article: ] |
| 52. | Deneo-Pellegrini H, De Stéfani E, Boffetta P, Ronco A, Mendilaharsu M. Dietary iron and cancer of the rectum: a case-control study in Uruguay. Eur J Cancer Prev. 1999;8:501-508. [Cited in This Article: ] |
| 53. | Tseng M, Greenberg ER, Sandler RS, Baron JA, Haile RW, Blumberg BS, McGlynn KA. Serum ferritin concentration and recurrence of colorectal adenoma. Cancer Epidemiol Biomarkers Prev. 2000;9:625-630. [Cited in This Article: ] |
| 54. | Herrinton LJ, Friedman GD, Baer D, Selby JV. Transferrin saturation and risk of cancer. Am J Epidemiol. 1995;142:692-698. [Cited in This Article: ] |
| 55. | Sesink AL, Termont DS, Kleibeuker JH, Van der Meer R. Red meat and colon cancer: the cytotoxic and hyperproliferative effects of dietary heme. Cancer Res. 1999;59:5704-5709. [Cited in This Article: ] |
| 56. | Sesink AL, Termont DS, Kleibeuker JH, Van Der Meer R. Red meat and colon cancer: dietary haem, but not fat, has cytotoxic and hyperproliferative effects on rat colonic epithelium. Carcinogenesis. 2000;21:1909-1915. [Cited in This Article: ] |
| 57. | Norat T, Lukanova A, Ferrari P, Riboli E. Meat consumption and colorectal cancer risk: dose-response meta-analysis of epidemiological studies. Int J Cancer. 2002;98:241-256. [Cited in This Article: ] |
| 58. | Norat T, Bingham S, Ferrari P, Slimani N, Jenab M, Mazuir M, Overvad K, Olsen A, Tjønneland A, Clavel F. Meat, fish, and colorectal cancer risk: the European Prospective Investigation into cancer and nutrition. J Natl Cancer Inst. 2005;97:906-916. [Cited in This Article: ] |
| 59. | Lee DH, Anderson KE, Harnack LJ, Folsom AR, Jacobs DR Jr. Heme iron, zinc, alcohol consumption, and colon cancer: Iowa Women's Health Study. J Natl Cancer Inst. 2004;96:403-407. [Cited in This Article: ] |
| 60. | Ayonrinde OT, Milward EA, Chua AC, Trinder D, Olynyk JK. Clinical perspectives on hereditary hemochromatosis. Crit Rev Clin Lab Sci. 2008;45:451-484. [Cited in This Article: ] |
| 61. | Heath AL, Fairweather-Tait SJ. Health implications of iron overload: the role of diet and genotype. Nutr Rev. 2003;61:45-62. [Cited in This Article: ] |
| 62. | Beckman LE, Van Landeghem GF, Sikström C, Wahlin A, Markevärn B, Hallmans G, Lenner P, Athlin L, Stenling R, Beckman L. Interaction between haemochromatosis and transferrin receptor genes in different neoplastic disorders. Carcinogenesis. 1999;20:1231-1233. [Cited in This Article: ] |
| 63. | Nelson RL, Davis FG, Persky V, Becker E. Risk of neoplastic and other diseases among people with heterozygosity for hereditary hemochromatosis. Cancer. 1995;76:875-879. [Cited in This Article: ] |
| 64. | Shaheen NJ, Silverman LM, Keku T, Lawrence LB, Rohlfs EM, Martin CF, Galanko J, Sandler RS. Association between hemochromatosis (HFE) gene mutation carrier status and the risk of colon cancer. J Natl Cancer Inst. 2003;95:154-159. [Cited in This Article: ] |
| 65. | Robinson JP, Johnson VL, Rogers PA, Houlston RS, Maher ER, Bishop DT, Evans DG, Thomas HJ, Tomlinson IP, Silver AR. Evidence for an association between compound heterozygosity for germ line mutations in the hemochromatosis (HFE) gene and increased risk of colorectal cancer. Cancer Epidemiol Biomarkers Prev. 2005;14:1460-1463. [Cited in This Article: ] |
| 66. | Altés A, Gimferrer E, Capella G, Barceló MJ, Baiget M. Colorectal cancer and HFE gene mutations. Haematologica. 1999;84:479-480. [Cited in This Article: ] |
| 67. | Macdonald GA, Tarish J, Whitehall VJ, McCann SJ, Mellick GD, Buttenshaw RL, Johnson AG, Young J, Leggett BA. No evidence of increased risk of colorectal cancer in individuals heterozygous for the Cys282Tyr haemochromatosis mutation. J Gastroenterol Hepatol. 1999;14:1188-1191. [Cited in This Article: ] |
| 68. | Nelson RL, Persky V, Davis F, Becker E. Risk of disease in siblings of patients with hereditary hemochromatosis. Digestion. 2001;64:120-124. [Cited in This Article: ] |
| 69. | Elmberg M, Hultcrantz R, Ekbom A, Brandt L, Olsson S, Olsson R, Lindgren S, Lööf L, Stål P, Wallerstedt S. Cancer risk in patients with hereditary hemochromatosis and in their first-degree relatives. Gastroenterology. 2003;125:1733-1741. [Cited in This Article: ] |
| 70. | van der A DL, van der Hel O, Roest M, van der Schouw YT, van Gils CH, Marx JJ, van Noord PA, Peeters PH. Heterozygosity for the Cys282Tyr mutation in the HFE gene and the risk of colorectal cancer (Netherlands). Cancer Causes Control. 2003;14:541-545. [Cited in This Article: ] |
| 71. | McGlynn KA, Sakoda LC, Hu Y, Schoen RE, Bresalier RS, Yeager M, Chanock S, Hayes RB, Buetow KH. Hemochromatosis gene mutations and distal adenomatous colorectal polyps. Cancer Epidemiol Biomarkers Prev. 2005;14:158-163. [Cited in This Article: ] |
| 72. | Chan AT, Ma J, Tranah GJ, Giovannucci EL, Rifai N, Hunter DJ, Fuchs CS. Hemochromatosis gene mutations, body iron stores, dietary iron, and risk of colorectal adenoma in women. J Natl Cancer Inst. 2005;97:917-926. [Cited in This Article: ] |
| 73. | Ellervik C, Birgens H, Tybjaerg-Hansen A, Nordestgaard BG. Hemochromatosis genotypes and risk of 31 disease endpoints: meta-analyses including 66,000 cases and 226,000 controls. Hepatology. 2007;46:1071-1080. [Cited in This Article: ] |
| 74. | Zacharski LR, Chow BK, Howes PS, Shamayeva G, Baron JA, Dalman RL, Malenka DJ, Ozaki CK, Lavori PW. Decreased cancer risk after iron reduction in patients with peripheral arterial disease: results from a randomized trial. J Natl Cancer Inst. 2008;100:996-1002. [Cited in This Article: ] |
| 75. | Merk K, Mattsson B, Mattsson A, Holm G, Gullbring B, Björkholm M. The incidence of cancer among blood donors. Int J Epidemiol. 1990;19:505-509. [Cited in This Article: ] |
| 76. | Wheby MS, Jones LG, Crosby WH. Studies on Iron Absorption. Intestinal Regulatory Mechanisms. J Clin Invest. 1964;43:1433-1442. [Cited in This Article: ] |
| 77. | Chernelch M, Fawwaz R, Sargent T, Winchell HS. Effect of phlebotomy and pH on iron absorption from the colon. J Nucl Med. 1970;11:25-27. [Cited in This Article: ] |
| 78. | Jeffrey GP, Basclain KA, Allen TL. Molecular regulation of transferrin receptor and ferritin expression in the rat gastrointestinal tract. Gastroenterology. 1996;110:790-800. [Cited in This Article: ] |
| 79. | Frazer DM, Vulpe CD, McKie AT, Wilkins SJ, Trinder D, Cleghorn GJ, Anderson GJ. Cloning and gastrointestinal expression of rat hephaestin: relationship to other iron transport proteins. Am J Physiol Gastrointest Liver Physiol. 2001;281:G931-G939. [Cited in This Article: ] |
| 80. | Brookes MJ, Hughes S, Turner FE, Reynolds G, Sharma N, Ismail T, Berx G, McKie AT, Hotchin N, Anderson GJ. Modulation of iron transport proteins in human colorectal carcinogenesis. Gut. 2006;55:1449-1460. [Cited in This Article: ] |
| 81. | Seril DN, Liao J, Yang CS, Yang GY. Systemic iron supplementation replenishes iron stores without enhancing colon carcinogenesis in murine models of ulcerative colitis: comparison with iron-enriched diet. Dig Dis Sci. 2005;50:696-707. [Cited in This Article: ] |
| 82. | Pierre F, Freeman A, Taché S, Van der Meer R, Corpet DE. Beef meat and blood sausage promote the formation of azoxymethane-induced mucin-depleted foci and aberrant crypt foci in rat colons. J Nutr. 2004;134:2711-2716. [Cited in This Article: ] |
| 83. | Cross AJ, Pollock JR, Bingham SA. Haem, not protein or inorganic iron, is responsible for endogenous intestinal N-nitrosation arising from red meat. Cancer Res. 2003;63:2358-2360. [Cited in This Article: ] |
| 84. | Glei M, Klenow S, Sauer J, Wegewitz U, Richter K, Pool-Zobel BL. Hemoglobin and hemin induce DNA damage in human colon tumor cells HT29 clone 19A and in primary human colonocytes. Mutat Res. 2006;594:162-171. [Cited in This Article: ] |
| 85. | Kinzler KW, Vogelstein B. Lessons from hereditary colorectal cancer. Cell. 1996;87:159-170. [Cited in This Article: ] |
| 86. | Valko M, Rhodes CJ, Moncol J, Izakovic M, Mazur M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem Biol Interact. 2006;160:1-40. [Cited in This Article: ] |
| 87. | Takahashi M, Nakatsugi S, Sugimura T, Wakabayashi K. Frequent mutations of the beta-catenin gene in mouse colon tumors induced by azoxymethane. Carcinogenesis. 2000;21:1117-1120. [Cited in This Article: ] |
| 88. | Takahashi M, Wakabayashi K. Gene mutations and altered gene expression in azoxymethane-induced colon carcinogenesis in rodents. Cancer Sci. 2004;95:475-480. [Cited in This Article: ] |
| 89. | He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT, Morin PJ, Vogelstein B, Kinzler KW. Identification of c-MYC as a target of the APC pathway. Science. 1998;281:1509-1512. [Cited in This Article: ] |
| 90. | Tetsu O, McCormick F. Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature. 1999;398:422-426. [Cited in This Article: ] |
| 91. | Seril DN, Liao J, Yang GY, Yang CS. Oxidative stress and ulcerative colitis-associated carcinogenesis: studies in humans and animal models. Carcinogenesis. 2003;24:353-362. [Cited in This Article: ] |
| 92. | Kern SE, Redston M, Seymour AB, Caldas C, Powell SM, Kornacki S, Kinzler KW. Molecular genetic profiles of colitis-associated neoplasms. Gastroenterology. 1994;107:420-428. [Cited in This Article: ] |
| 93. | Fogt F, Zhuang Z, Poremba C, Dockhorn-Dworniczak B, Vortmeyer A. Comparison of p53 immunoexpression with allelic loss of p53 in ulcerative colitis-associated dysplasia and carcinoma. Oncol Rep. 1998;5:477-480. [Cited in This Article: ] |
| 94. | Naugler WE, Karin M. NF-kappaB and cancer-identifying targets and mechanisms. Curr Opin Genet Dev. 2008;18:19-26. [Cited in This Article: ] |
| 95. | Grivennikov S, Karin E, Terzic J, Mucida D, Yu GY, Vallabhapurapu S, Scheller J, Rose-John S, Cheroutre H, Eckmann L. IL-6 and Stat3 are required for survival of intestinal epithelial cells and development of colitis-associated cancer. Cancer Cell. 2009;15:103-113. [Cited in This Article: ] |
| 96. | She H, Xiong S, Lin M, Zandi E, Giulivi C, Tsukamoto H. Iron activates NF-kappaB in Kupffer cells. Am J Physiol Gastrointest Liver Physiol. 2002;283:G719-G726. [Cited in This Article: ] |
| 97. | Xiong S, She H, Takeuchi H, Han B, Engelhardt JF, Barton CH, Zandi E, Giulivi C, Tsukamoto H. Signaling role of intracellular iron in NF-kappaB activation. J Biol Chem. 2003;278:17646-17654. [Cited in This Article: ] |
| 98. | Singer II, Kawka DW, Scott S, Weidner JR, Mumford RA, Riehl TE, Stenson WF. Expression of inducible nitric oxide synthase and nitrotyrosine in colonic epithelium in inflammatory bowel disease. Gastroenterology. 1996;111:871-885. [Cited in This Article: ] |
| 99. | Cianchi F, Cortesini C, Bechi P, Fantappiè O, Messerini L, Vannacci A, Sardi I, Baroni G, Boddi V, Mazzanti R. Up-regulation of cyclooxygenase 2 gene expression correlates with tumor angiogenesis in human colorectal cancer. Gastroenterology. 2001;121:1339-1347. [Cited in This Article: ] |
| 100. | Jenkins DC, Charles IG, Thomsen LL, Moss DW, Holmes LS, Baylis SA, Rhodes P, Westmore K, Emson PC, Moncada S. Roles of nitric oxide in tumor growth. Proc Natl Acad Sci USA. 1995;92:4392-4396. [Cited in This Article: ] |
| 101. | Eberhart CE, Coffey RJ, Radhika A, Giardiello FM, Ferrenbach S, DuBois RN. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology. 1994;107:1183-1188. [Cited in This Article: ] |
| 102. | Ambs S, Merriam WG, Bennett WP, Felley-Bosco E, Ogunfusika MO, Oser SM, Klein S, Shields PG, Billiar TR, Harris CC. Frequent nitric oxide synthase-2 expression in human colon adenomas: implication for tumor angiogenesis and colon cancer progression. Cancer Res. 1998;58:334-341. [Cited in This Article: ] |
| 103. | Ahn B, Ohshima H. Suppression of intestinal polyposis in Apc(Min/+) mice by inhibiting nitric oxide production. Cancer Res. 2001;61:8357-8360. [Cited in This Article: ] |
| 104. | Oshima M, Murai N, Kargman S, Arguello M, Luk P, Kwong E, Taketo MM, Evans JF. Chemoprevention of intestinal polyposis in the Apcdelta716 mouse by rofecoxib, a specific cyclooxygenase-2 inhibitor. Cancer Res. 2001;61:1733-1740. [Cited in This Article: ] |
| 105. | Philippe MA, Ruddell RG, Ramm GA. Role of iron in hepatic fibrosis: one piece in the puzzle. World J Gastroenterol. 2007;13:4746-4754. [Cited in This Article: ] |
| 106. | Halliwell B. Oxidative stress and cancer: have we moved forward? Biochem J. 2007;401:1-11. [Cited in This Article: ] |
| 107. | Toyokuni S. Molecular mechanisms of oxidative stress-induced carcinogenesis: from epidemiology to oxygenomics. IUBMB Life. 2008;60:441-447. [Cited in This Article: ] |
| 108. | Turker MS, Gage BM, Rose JA, Elroy D, Ponomareva ON, Stambrook PJ, Tischfield JA. A novel signature mutation for oxidative damage resembles a mutational pattern found commonly in human cancers. Cancer Res. 1999;59:1837-1839. [Cited in This Article: ] |
| 109. | Wagener FA, Volk HD, Willis D, Abraham NG, Soares MP, Adema GJ, Figdor CG. Different faces of the heme-heme oxygenase system in inflammation. Pharmacol Rev. 2003;55:551-571. [Cited in This Article: ] |
| 110. | Jeney V, Balla J, Yachie A, Varga Z, Vercellotti GM, Eaton JW, Balla G. Pro-oxidant and cytotoxic effects of circulating heme. Blood. 2002;100:879-887. [Cited in This Article: ] |
| 111. | Oates PS, West AR. Heme in intestinal epithelial cell turnover, differentiation, detoxification, inflammation, carcinogenesis, absorption and motility. World J Gastroenterol. 2006;12:4281-4295. [Cited in This Article: ] |
| 112. | Stocker R, Glazer AN, Ames BN. Antioxidant activity of albumin-bound bilirubin. Proc Natl Acad Sci USA. 1987;84:5918-5922. [Cited in This Article: ] |
| 113. | Minetti M, Mallozzi C, Di Stasi AM, Pietraforte D. Bilirubin is an effective antioxidant of peroxynitrite-mediated protein oxidation in human blood plasma. Arch Biochem Biophys. 1998;352:165-174. [Cited in This Article: ] |
| 114. | Elchuri S, Oberley TD, Qi W, Eisenstein RS, Jackson Roberts L, Van Remmen H, Epstein CJ, Huang TT. CuZnSOD deficiency leads to persistent and widespread oxidative damage and hepatocarcinogenesis later in life. Oncogene. 2005;24:367-380. [Cited in This Article: ] |
| 115. | Van Remmen H, Ikeno Y, Hamilton M, Pahlavani M, Wolf N, Thorpe SR, Alderson NL, Baynes JW, Epstein CJ, Huang TT. Life-long reduction in MnSOD activity results in increased DNA damage and higher incidence of cancer but does not accelerate aging. Physiol Genomics. 2003;16:29-37. [Cited in This Article: ] |
| 116. | Rainis T, Maor I, Lanir A, Shnizer S, Lavy A. Enhanced oxidative stress and leucocyte activation in neoplastic tissues of the colon. Dig Dis Sci. 2007;52:526-530. [Cited in This Article: ] |
| 117. | Ribeiro ML, Priolli DG, Miranda DD, Arçari DP, Pedrazzoli J Jr, Martinez CA. Analysis of oxidative DNA damage in patients with colorectal cancer. Clin Colorectal Cancer. 2008;7:267-272. [Cited in This Article: ] |
| 118. | Núñez MT, Tapia V, Toyokuni S, Okada S. Iron-induced oxidative damage in colon carcinoma (Caco-2) cells. Free Radic Res. 2001;34:57-68. [Cited in This Article: ] |
| 119. | Younes M, Trepkau HD, Siegers CP. Enhancement by dietary iron of lipid peroxidation in mouse colon. Res Commun Chem Pathol Pharmacol. 1990;70:349-354. [Cited in This Article: ] |
| 120. | Rimbach G, Markant A, Most E, Pallauf J. Liver and colon oxidant status in growing rats fed increasing levels of dietary iron. J Trace Elem Med Biol. 1997;11:99-104. [Cited in This Article: ] |
| 121. | Carrier J, Aghdassi E, Platt I, Cullen J, Allard JP. Effect of oral iron supplementation on oxidative stress and colonic inflammation in rats with induced colitis. Aliment Pharmacol Ther. 2001;15:1989-1999. [Cited in This Article: ] |
| 122. | Lund EK, Fairweather-Tait SJ, Wharf SG, Johnson IT. Chronic exposure to high levels of dietary iron fortification increases lipid peroxidation in the mucosa of the rat large intestine. J Nutr. 2001;131:2928-2931. [Cited in This Article: ] |
| 123. | Liu Z, Tomotake H, Wan G, Watanabe H, Kato N. Combined effect of dietary calcium and iron on colonic aberrant crypt foci, cell proliferation and apoptosis, and fecal bile acids in 1,2-dimethylhydrazine-treated rats. Oncol Rep. 2001;8:893-897. [Cited in This Article: ] |
| 124. | Pierre F, Taché S, Petit CR, Van der Meer R, Corpet DE. Meat and cancer: haemoglobin and haemin in a low-calcium diet promote colorectal carcinogenesis at the aberrant crypt stage in rats. Carcinogenesis. 2003;24:1683-1690. [Cited in This Article: ] |
| 125. | Stevens RG, Morris JE, Cordis GA, Anderson LE, Rosenberg DW, Sasser LB. Oxidative damage in colon and mammary tissue of the HFE-knockout mouse. Free Radic Biol Med. 2003;34:1212-1216. [Cited in This Article: ] |
| 126. | Parkin DM. The global health burden of infection-associated cancers in the year 2002. Int J Cancer. 2006;118:3030-3044. [Cited in This Article: ] |
| 127. | Ekbom A, Helmick C, Zack M, Adami HO. Ulcerative colitis and colorectal cancer. A population-based study. N Engl J Med. 1990;323:1228-1233. [Cited in This Article: ] |
| 128. | Babbs CF. Free radicals and the etiology of colon cancer. Free Radic Biol Med. 1990;8:191-200. [Cited in This Article: ] |
| 129. | Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. 2001;357:539-545. [Cited in This Article: ] |
| 130. | Lin WW, Karin M. A cytokine-mediated link between innate immunity, inflammation, and cancer. J Clin Invest. 2007;117:1175-1183. [Cited in This Article: ] |
| 131. | Eaden JA, Abrams KR, Mayberry JF. The risk of colorectal cancer in ulcerative colitis: a meta-analysis. Gut. 2001;48:526-535. [Cited in This Article: ] |
| 132. | Prior P, Gyde SN, Macartney JC, Thompson H, Waterhouse JA, Allan RN. Cancer morbidity in ulcerative colitis. Gut. 1982;23:490-497. [Cited in This Article: ] |
| 133. | Keshavarzian A, Morgan G, Sedghi S, Gordon JH, Doria M. Role of reactive oxygen metabolites in experimental colitis. Gut. 1990;31:786-790. [Cited in This Article: ] |
| 134. | Keshavarzian A, Haydek J, Zabihi R, Doria M, D'Astice M, Sorenson JR. Agents capable of eliminating reactive oxygen species. Catalase, WR-2721, or Cu(II)2(3,5-DIPS)4 decrease experimental colitis. Dig Dis Sci. 1992;37:1866-1873. [Cited in This Article: ] |
| 135. | Babbs CF. Oxygen radicals in ulcerative colitis. Free Radic Biol Med. 1992;13:169-181. [Cited in This Article: ] |
| 136. | Nicolas G, Bennoun M, Porteu A, Mativet S, Beaumont C, Grandchamp B, Sirito M, Sawadogo M, Kahn A, Vaulont S. Severe iron deficiency anemia in transgenic mice expressing liver hepcidin. Proc Natl Acad Sci USA. 2002;99:4596-4601. [Cited in This Article: ] |
| 137. | Laftah AH, Ramesh B, Simpson RJ, Solanky N, Bahram S, Schümann K, Debnam ES, Srai SK. Effect of hepcidin on intestinal iron absorption in mice. Blood. 2004;103:3940-3944. [Cited in This Article: ] |
| 138. | Weiss G. Pathogenesis and treatment of anaemia of chronic disease. Blood Rev. 2002;16:87-96. [Cited in This Article: ] |
| 139. | Poss KD, Tonegawa S. Heme oxygenase 1 is required for mammalian iron reutilization. Proc Natl Acad Sci USA. 1997;94:10919-10924. [Cited in This Article: ] |