Published online Sep 7, 2009. doi: 10.3748/wjg.15.4199
Revised: July 18, 2009
Accepted: July 25, 2009
Published online: September 7, 2009
Malignant tumors of papilla are usually adenocarcinomas. We present a 67-year-old female who became icteric as result of a malignant tumor infiltrating the papilla of Vater. Histopathological assessment of surgically excised tumor showed both neuroendocrine and adenocarcinomatous features. To our knowledge, this is the seventh report of this rare neoplastic association in the duodenal periampullary region.
- Citation: Musialik JA, Kohut MJ, Marek T, Wodołażski A, Hartleb M. Composite neuroendocrine and adenomatous carcinoma of the papilla of Vater. World J Gastroenterol 2009; 15(33): 4199-4200
- URL: https://www.wjgnet.com/1007-9327/full/v15/i33/4199.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.4199
In adults the commonest causes of obstructive jaundice are biliary stone disease, pancreatic cancer or chronic pancreatitis. A less frequent cause is a benign or malignant tumor arising from the papilla of Vater. Malignant tumors of papilla are usually adenocarcinomas. We present a patient who became icteric as a consequence of a malignant tumor infiltrating the papilla of Vater which histologically showed both neuroendocrine and adenocarcinomatous features.
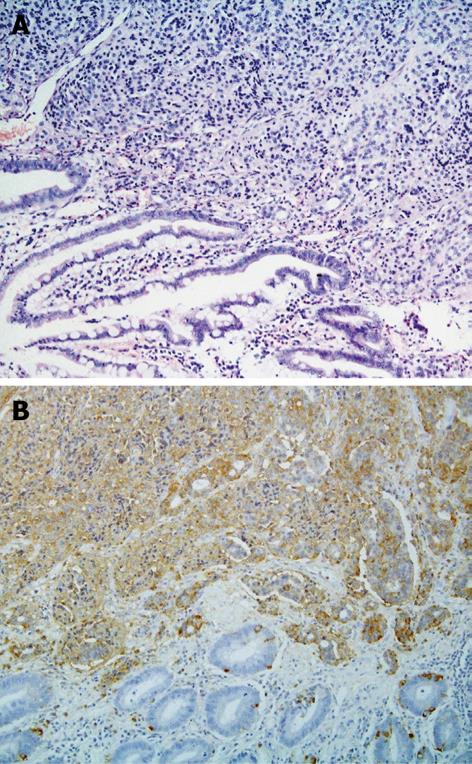
A 67-year-old cholecystectomized woman was admitted to our department with symptoms of obstructive jaundice and cholangitis. Severe epigastric pain, fever and jaundice developed 1 d before presentation. The serum bilirubin level was 5.3 mg/dL, white cell blood count was 20.4 × 103/μL and serum amylase and lipase activities were elevated to about six times the normal upper limit. Abdominal ultrasonography showed moderate dilatation of common bile duct. The patient underwent endoscopic retrograde cholangiopancreatography (ERCP) revealing the papilla of Vater was suggestive of adenoma i.e., was markedly enlarged and covered with smooth surfaced mucosa. Sphincterotomy was performed and biopsies were taken from the inner part of the ampulla. The histopathological diagnosis from biopsy specimens was carcinoid tumor, staining positive for chromogranin A and synaptophysin. The patient underwent surgical transduodenal excision of the ampulla. The postoperative histopathological examination showed a composite tumor i.e. mixed neuroendocrine carcinoma (NE)-adenocarcinoma neoplasm (Figure 1A and B). Immunocytochemical studies were used to delinate the NE component (chromogranin, synaptophisin, EMA, cytokeratin). The NE-cell component represented about 80% of the tumor’s
area. NE tumor cells were well-differentiated, lacked significant atypia and showed low grade malignancy (Ki-67 labelling index was 5%). The glandular component presented well- differentiated tubules (grade I). No necrosis was found in either the NE or glandular areas. The tumor focally involved lymphatic vessels and nerves and infiltrated the muscle of the ampulla. The margins of the excised tissue specimen were free of neoplasm and no neoplastic cells were found in periduodenal lymphatic nodes (T1N0M0). The patient has been put under endoscopic, serological (CA 19-9, CEA, chromogranin A) and ultrasound surveillance. Six months after surgery she is doing well.
Gastroenteropancreatic Neuroendocrine Tumors (GEP/NETs) are rare neoplasms originating from the diffuse neuroendocrine system, involving 15 types of highly differentiated ectodermal cells located in the gastrointestinal tract and pancreas. The most common location of GEP/NETs is in the appendix (62% of cases), but they may also be found in small intestine (27%), lung (15%), undefined primary locations with hepatic metastases (12%) and other organs (3%)[1]. GEP/NETs secrete peptides and neuroamines that cause distinct clinical syndromes, including carcinoid syndrome. Many tumors are, however, clinically silent until late presentation associated with mass effects. The current WHO classification categorizes GEP/NETs into: (1) well-differentiated neuroendocrine tumor; (2) low-grade malignant carcinoma; (3) high-grade malignant carcinoma and (4) mixed tumor: adenocarcinoma/neuroendocrine carcinoma[2]. At present 14 terms are used to define tumors with mixed exocrine-endocrine features. Volante et al[3] proposed classification based on the extension of each component and structural features of the NE component. Three separate patterns can be distinguished i.e. (1) NE tumors with focal non-NE component occupying less than 30% of the tumor, (2) mixed exocrine-endocrine carcinomas (NE or non-NE cells > 30%) and (3) non-NE carcinoma with NE component (< 30%). The type of tumor influences the prognosis, which improves with increasing contribution of NE component[3]. About 50% of GEP/NETs are carcinoids found by chance during laparotomy or as hepatic metastases.
Carcinoids involving the papilla of Vater are rare lesions, accounting for 0.35% of all gastrointestinal carcinoids. So far, only 110 cases have been reported in the literature, mostly as individual case reports[4]. These tumors are predominantly found on ERCP in patients with obstructive jaundice or acute biliary pancreatitis. In the presented case the tumor histologically was a mixed carcinoid-adenocarcinoma neoplasm (type IV). The concurrence of carcinoid with adenocarcinoma is an unusual phenomenon in the gastrointestinal tract, that has been reported in the oesophagus, stomach, small intestine, appendix, colon and rectum[5-7]. To our knowledge, this is the seventh report of this rare neoplastic association in the duodenal periampullary region.
| 1. | Doherty GM. Rare endocrine tumours of the GI tract. Best Pract Res Clin Gastroenterol. 2005;19:807-817. [Cited in This Article: ] |
| 2. | Rindi G, Capella C, Solcia E. Introduction to a revised clinicopathological classification of neuroendocrine tumors of the gastroenteropancreatic tract. Q J Nucl Med. 2000;44:13-21. [Cited in This Article: ] |
| 3. | Volante M, Rindi G, Papotti M. The grey zone between pure (neuro)endocrine and non-(neuro)endocrine tumours: a comment on concepts and classification of mixed exocrine-endocrine neoplasms. Virchows Arch. 2006;449:499-506. [Cited in This Article: ] |
| 4. | Selvakumar E, Rajendran S, Balachandar TG, Kannan DG, Jeswanth S, Ravichandran P, Surendran R. Neuroendocrine carcinoma of the ampulla of Vater: a clinicopathologic evaluation. Hepatobiliary Pancreat Dis Int. 2008;7:422-425. [Cited in This Article: ] |
| 5. | Levendoglu H, Cox CA, Nadimpalli V. Composite (adenocarcinoid) tumors of the gastrointestinal tract. Dig Dis Sci. 1990;35:519-525. [Cited in This Article: ] |
| 6. | Costantini M, Montalti R, Rossi G, Luisa L, Masetti M, Di Benedetto F, Giorgio G. Adenocarcinoid tumor of the extrahepatic biliary tract. Int J Surg Pathol. 2008;16:455-457. [Cited in This Article: ] |
| 7. | Liu SH, Tsay SH. Coexistence of large cell neuroendocrine carcinoma and adenocarcinoma of the ampulla of vater. J Chin Med Assoc. 2008;71:536-540. [Cited in This Article: ] |