Published online Jan 21, 2009. doi: 10.3748/wjg.15.280
Revised: November 19, 2008
Accepted: November 26, 2008
Published online: January 21, 2009
About 30% of patients with cirrhosis have diabetes mellitus (DM). Nowadays, it is a matter for debate whether type 2 DM in the absence of obesity and hypertriglyceridemia may be a risk factor for chronic liver disease. DM, which develops as a complication of cirrhosis, is known as “hepatogenous diabetes”. Insulin resistance in muscular and adipose tissues and hyperinsulinemia seem to be the pathophysiologic bases of diabetes in liver disease. An impaired response of the islet β-cells of the pancreas and hepatic insulin resistance are also contributory factors. Non-alcoholic fatty liver disease, alcoholic cirrhosis, chronic hepatitis C (CHC) and hemochromatosis are more frequently associated with DM. Insulin resistance increases the failure of the response to treatment in patients with CHC and enhances progression of fibrosis. DM in cirrhotic patients may be subclinical. Hepatogenous diabetes is clinically different from that of type 2 DM, since it is less frequently associated with microangiopathy and patients more frequently suffer complications of cirrhosis. DM increases the mortality of cirrhotic patients. Treatment of the diabetes is complex due to liver damage and hepatotoxicity of oral hypoglycemic drugs. This manuscript will review evidence that exists in relation to: type 2 DM alone or as part of the metabolic syndrome in the development of liver disease; factors involved in the genesis of hepatogenous diabetes; the impact of DM on the clinical outcome of liver disease; the management of DM in cirrhotic patients and the role of DM as a risk factor for the occurrence and exacerbation of hepatocellular carcinoma.
- Citation: Garcia-Compean D, Jaquez-Quintana JO, Gonzalez-Gonzalez JA, Maldonado-Garza H. Liver cirrhosis and diabetes: Risk factors, pathophysiology, clinical implications and management. World J Gastroenterol 2009; 15(3): 280-288
- URL: https://www.wjgnet.com/1007-9327/full/v15/i3/280.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.280
Up to 96% of patients with cirrhosis may be glucose intolerant and 30% may be clinically diabetic[1]. Currently, it is a matter for debate whether type 2 diabetes mellitus (DM), in the absence of other risk factors contributing to metabolic syndrome (obesity and hypertriglyceridemia), could be a risk factor for the development and progression of liver disease[2–4]. On the other hand, the diabetes which develops as a complication of cirrhosis is known as “hepatogenous diabetes” and is not recognized by the American Diabetes Association and the World Health Organization as a specific independent entity[5].
The liver has an important role in carbohydrate metabolism since it is responsible for the balance of blood glucose levels by means of glycogenogenesis and glycogenolysis[5–11]. In the presence of hepatic disease, the metabolic homeostasis of glucose is impaired as a result of disorders such as insulin resistance, glucose intolerance and diabetes[681112]. Insulin resistance occurs not only in muscular tissue, but also in adipose tissue[13], and this combined with hyperinsulinemia seem to be important pathophysiologic bases of diabetes in liver disease[135614–17]. Additionally, the etiology of liver disease is important in the incidence of DM, since non-alcoholic fatty liver disease (NAFLD), alcohol, hepatitis C virus (HCV) and hemochromatosis are frequently associated with DM[1–3718].
DM in patients with compensated liver cirrhosis may be subclinical, since fasting serum glucose levels may be normal. In these cases, it is necessary to perform an oral glucose tolerance test (OGTT) to detect an impairment of glucose metabolism[19]. The natural history of hepatogenous diabetes is different from that of hereditary type 2 DM, since it is less frequently associated with microangiopathy. In contrast, the patient with cirrhosis and diabetes suffers more frequently from complications of cirrhosis, which can cause death[2419].
Treatment of diabetes in the cirrhotic patient is complex because of the presence of liver damage and the hepatotoxicity of oral hypoglycemic drugs. Therefore, pharmacological therapy must be closely monitored for the risk of hypoglycemia[3519].
This review will present evidence that exists in the literature in relation to: (1) type 2 DM alone or as part of the metabolic syndrome in the development of liver disease; (2) factors involved in the genesis of hepatogenous diabetes; (3) the impact of DM on the clinical outcome of liver disease; (4) the management of DM in cirrhotic patients. Similarly, we will review the role of type 2 DM and hepatogenous diabetes as risk factors for the occurrence and exacerbation of hepatocellular carcinoma (HCC).
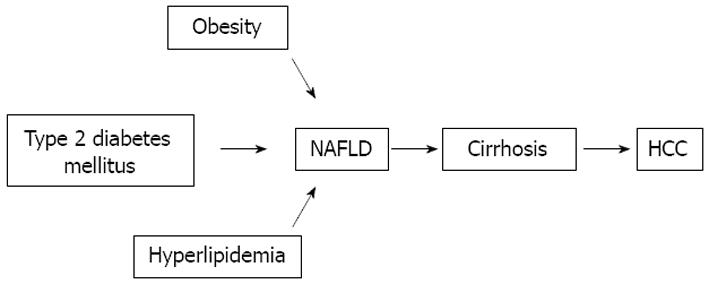
Several studies suggest that type 2 DM may have an etiological role in chronic liver disease and HCC regardless of alcohol and viruses[4] (Figure 1).
A total of 173643 patients with type 2 DM and 650620 patients without type 2 DM, in whom chronic liver disease was excluded at the time of enrollment and a year later, were observed over a 10-year period in a cohort study. The incidence of non-alcoholic chronic liver disease and HCC was significantly higher in diabetic patients compared with non-diabetic patients. This risk was 2-fold greater and was independent of alcoholic liver disease, viral hepatitis and demographic factors[4]. Although this study has the strength of having a very large number of individuals, it has been criticized because it included a population comprised almost entirely of men (98%) from the Department of Veterans Affairs, and the diagnoses of type 2 DM, chronic liver disease and HCC were taken from a database, and consequently they were not verified biochemically and histopathologically. Additionally, other factors that are part of the metabolic syndrome (which already have a proven influence on the occurrence of NAFLD such as obesity and dyslipidemia) were not taken into account[20].
In a study with a large number of patients carried out in Denmark, the standardized incidence of HCC was higher in men (4.0, 95% CI: 3.5-4.6) and women (2.1, 95% CI: 1.6-2.7) with type 2 DM compared with the general population[21]. Other studies with fewer patients have yielded similar results[2223].
In a recent case-control study that included 465 patients, DM prevalence was higher in patients with HCC than in controls (31.2% vs 12.7%, OR 3.12 95% CI: 2.22-4.43). The DM had been diagnosed prior to the occurrence of HCC in 84% of cases with an average duration of 181.4 mo indicating that it was type 2 DM in most cases[24]. The above data suggests that type 2 DM itself might be a risk factor for the occurrence of HCC. Other studies showed that in the presence HCV, liver fibrosis and alcohol, the risk is higher. Recently, it was observed that patients suffering from chronic hepatitis C (CHC), DM and advanced fibrosis had a 3-fold greater risk than non-diabetic patients with mild to moderate fibrosis of developing HCC in 5 years of follow-up (13% vs 5%)[25].
NAFLD comprises a series of liver disorders such as simple steatosis, steatohepatitis, fibrosis and cirrhosis. It is estimated that one third of American adult individuals may suffer from fatty liver[26], which is considered the most benign manifestation of NAFLD. The primary fatty liver results from the accumulation of fat, mainly of triglycerides in liver cells in the presence of insulin resistance, and frequently occurs as part of the metabolic syndrome which is made up of obesity, type 2 DM and dyslipidemia[27]. Non-alcoholic steatohepatitis (NASH) is a severe manifestation of NAFLD, since it causes not only steatosis, but tissue inflammation, cell damage and fibrosis. Nevertheless, the prevalence of NASH is estimated at 2%-3%. NASH is regarded as an entity that can progress to cirrhosis and liver failure, and currently it is estimated to be the most common cause of cryptogenic cirrhosis[2829].
The mechanisms by which type 2 DM might cause NAFLD are complex and have been studied in a fragmented manner mainly in isolated biological systems.
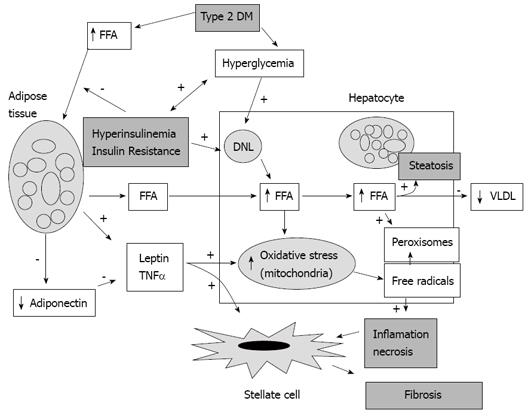
It has been observed that the fatty liver, obesity and insulin resistance act as co-factors to cause liver damage[134]. Fatty liver is the result of an intracellular accumulation of triglycerides because of increased uptake of free fatty acids and de novo liponeogenesis in the hepatocytes. At the same time, there is a reduction in the hepatic secretion of very low density lipoproteins. The liver damage consists of cellular necrosis and inflammation, and these disorders result from an increase in mitochondrial oxidative stress on triglycerides with the consequential generation of free radicals and peroxisomes[3031]. The mitochondrial oxidative stress is increased also by the action of adipokines (cytokines produced by the adipocytes) such as leptin and tumor necrosis factor-α (TNF-α), which are produced in excess[32]. The reduction of adiponectin, which is a regulatory adipokine, favors the activity of inflammatory adipokines[33]. These chemical mediators, derived from inflammation and cell necrosis, as well as the adipokines activate the liver stellate cells and induce them to increase production of collagen, connective tissue growth factor and accumulation of extracellular matrix, in turn favoring fibrosis[34] (Figure 2).
Depending on the etiology, the degree of liver damage and the diagnostic criteria, the reported incidence of glucose intolerance varies from 60 to 80% and that of diabetes between 20 and 60%[351619]. It is known that from the early stages of chronic liver disease, insulin resistance and glucose intolerance may be found in most of these patients[3536]. The diabetes manifests clinically as the liver function deteriorates, thus hepatogenous diabetes can be considered as an indicator of advanced liver disease[37].
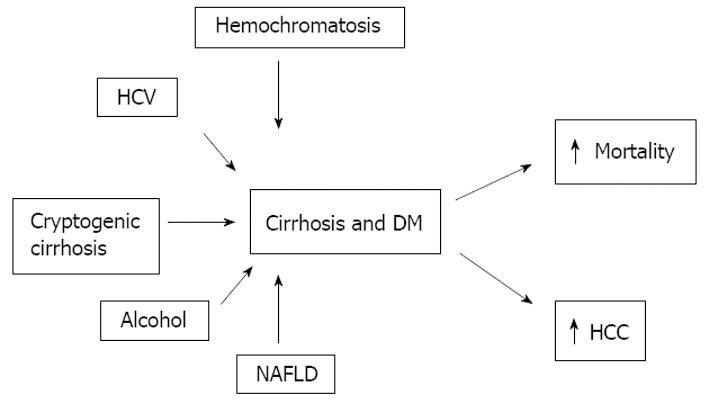
The etiology of chronic liver disease is crucial in the development of hepatogenous diabetes: alcohol, HCV, hemochromatosis and NASH (Figure 3).
NASH: NASH is a severe manifestation of NAFLD. NASH is associated with visceral obesity, hypertriglyceri-demia, and virtually all patients have insulin resistance. Therefore, it is not surprising that type 2 DM is present in 30%-45% of patients with NASH[38].
On the other hand, it has been observed that obesity itself is an independent risk factor for severe liver disease[39]. Obesity is characterized by expanded adipose tissue which is in a state of chronic inflammation resulting in an increase in the secretion of adipokines. These adipose tissue cytokines have a systemic effect particularly on the liver, which leads to an altered metabolic state with insulin resistance, hyperglycemia and hyperinsulinemia; these abnormalities disrupt the liver metabolism of lipids[40]. Cytokines, of which TNF-α is the most studied member, stimulate the liver stellate cells directly inducing hepatic fibrosis[41]. The body weight reduction improves metabolic abnormalities that accompany the metabolic syndrome such as hyperlipidemia and fatty liver[42].
CHC and HCV: In a study conducted by The National Health and Nutrition Examination Survey, a 3-fold higher risk of DM was identified in individuals over 40 years of age with CHC, compared with those patients with non-C chronic hepatitis[43]. Knobler et al observed a prevalence of DM of 33% in non-cirrhotic patients with CHC, compared with 5.6% in a control group[44]. In patients chronically infected with HCV, fatty liver was observed in 30%-70% of cases[45].
In patients with CHC, a high prevalence of glycometabolic abnormalities is reported such as glucose intolerance in more than 40% and DM in more than 17%. Additionally, the insulin resistance observed in these patients is an independent risk factor for steatosis in relation to the severity of fibrosis[746–48].
The mechanisms by which HCV produces insulin resistance and DM are not clearly known. It has been observed that HCV induces insulin resistance regardless of body mass index and fibrosis stage. In a study conducted in a transgenic animal model, the HCV core protein was able to induce insulin resistance, steatosis and DM. TNF-α overproduction seems to have been the primary mechanism. This cytokine phosphorylates the serine residues of the insulin receptor (IRS-1 and IRS-2), and stimulates the overproduction of suppressor of cytokines (SOC-3). SOC-3 inhibits phosphorylation of Akt and phosphatidylinositol 3-kinase. All these disorders, related to intracellular signaling of insulin, could block the transactivation of GLUT-4, which would result in block of glucose uptake at the cellular level. Indeed, in the transgenic mouse, TNF-α correlates with the hyperinsulinism and TNF-α block occasioned by the administration of anti-TNF-α drugs such as infliximab avoids the appearance of insulin resistance. Therefore, the mechanisms by which HCV induces insulin resistance include: production of TNF-α, serine phosphorylation of IRS and overexpression of SOCs. Furthermore, the overproduction of TNF-α in patients with CHC correlates with a faster progression of fibrosis and a lower response to interferon[40].
On the other hand, HCV genotype may be of importance in the occurrence of glucose metabolic disorders, as genotypes 1 and 4 are significantly associated with insulin resistance more frequently than genotypes 2 and 3 (37% vs 17%)[47]. It is well demonstrated that genotypes 1 and 4 are associated with a lower viral sustained response to antiviral therapy than genotypes 2 and 3. Insulin resistance may be a cofactor that increases the failure of the response to antiviral treatment observed in these patients. In accordance with this, in a recent study in patients with HCV genotype 1, those with HOMA > 2 (insulin resistance) had a 2-fold lower sustained response to treatment than patients with HOMA < 2 (32.8% vs 60.5%, respectively)[49]. For sustaining this idea, it is important to note that in experiments carried out with Huh-7 cells infected with HCV RNA, viral replication was blocked by adding interferon to the system. However, the ability of interferon to block viral replication was abolished when insulin was added to interferon at a dose of 128 mcU/mL (similar to that seen in the hyperinsulinemic states)[50]. Finally, it has been reported that patients with CHC and insulin resistance have a less sustained response to peginterferon plus ribavirin treatment compared with patients without insulin resistance[4049].
It seems that once the insulin resistance and DM-inducing mechanisms in CHC are fired, their courses are not affected by the presence or absence of viral activity. Indeed, in a recent study it was observed that HCV clearance by pegylated interferon and ribavirin treatment did not reduce the risk of DM in patients with chronic hepatitis and normal fasting blood glucose during a period of 8 years of follow-up after treatment. Patients with a sustained response had a similar incidence of DM compared with those who did not respond to treatment (14.8% vs 18.5%, respectively)[51].
Alcohol: Patients with alcoholic liver disease have a high relative risk of suffering diabetes[52]. This risk is directly related to the amount of ingested alcohol, as it rises 2-fold in patients ingesting more than 270 g of alcohol per week compared with those ingesting less than 120 g/wk[53]. Acute alcohol ingestion produces a significant reduction in insulin-mediated glucose uptake. On the other hand, patients with chronic alcoholism frequently have chronic pancreatic damage and injury of pancreatic islet β-cells resulting in DM[1].
Hemochromatosis: Hereditary hemochromatosis is a disease characterized by iron accumulation in several organs-particularly in the liver - as a result of a disorder of the metabolism of this metal. This abnormality is produced by a mutation of the HFE gene. In addition, the iron can infiltrate the pancreas and myocardium. In the pancreas, the concentration of iron is predominantly in the acinus of exocrine secretion. However, infiltration of Langerhans islets with damage to the insulin-producing β-cells can also be observed. This is the reason why DM can be observed in 50%-85% of patients with hereditary hemochromatosis in advanced stages[54]. Additionally, glucose metabolic disorders resulting from the liver damage probably contribute to the high frequency of DM[15].
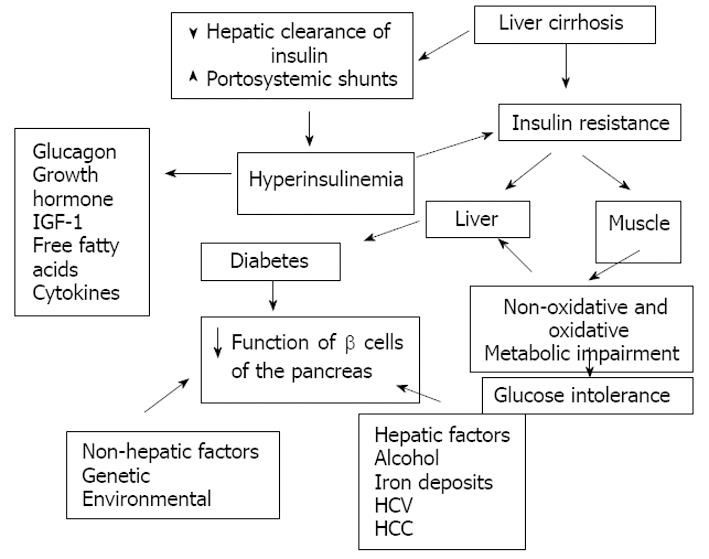
The pathophysiology of hepatogenous diabetes is complex and not precisely known. Insulin resistance in peripheral tissues (adipose and muscular tissue) plays a central role in the glucose metabolism disturbance[1291114–17]. It has also been proposed that reduced insulin extraction by the damaged liver and portosystemic shunts result in hyperinsulinemia which is potentiated by raised levels of contra-insulin hormones (glucagon, growth hormone, insulin-like growth factor, free fatty acids and cytokines)[2111517]. However, a recent study reports that in patients with Child B grade liver cirrhosis the hyperinsulinism may be produced by an increase of the pancreatic β-cell sensitivity to glucose, whereas disturbance of hepatic insulin extraction does not seem to have a significant role[55]. It has also been speculated that genetic and environmental factors and some etiologic agents in liver disease such as HCV, alcohol, and iron infiltration impair the insulin secretion activity of the β-cells of the pancreas[9]. In conclusion it seems that glucose intolerance may result from two abnormalities that occur simultaneously: (1) insulin resistance of muscle and (2) an inadequate response of the β-cells to appropriately secrete insulin to overcome the defect in insulin action. On the other hand DM develops as the result of progressive impairment in insulin secretion together with the development of hepatic insulin resistance leading to fasting hyperglycemia and a diabetic glucose tolerance profile[1415] (Figure 4).
Discrimination between hepatogenous diabetes and type 2 DM may be difficult. In a recent study comparing patients with hepatogenous diabetes vs patients with type 2 DM, the ratios of postprandial plasma glucose (PP2h) to fasting plasma glucose (FPG) (2.27 vs 1.69), fasting insulin (23.2 vs 11.6 microIU/mL) and HOMA-Insulin Resistance index (8.38 vs 3.52) were significantly higher in patients with hepatogenous diabetes. Therefore, insulin resistance in liver cirrhosis is higher than in type 2 DM, and impairment of hepatic insulin degradation may be an important mechanism of hyperinsulinemia in liver cirrhosis[56].
The effect of type 2 DM and hepatogenous diabetes on the clinical outcome of cirrhosis and HCC has been evaluated in only few studies. In cross-sectional retrospective studies in patients with cirrhosis of any etiology it has been observed that the DM is associated with an increased risk of complications[5385758]. According to the Verona study, which is based on a population of more than 7000 individuals suffering from type 2 DM, the risk of death at 5 years was 2.52-fold greater (CI 1.96-3.2) than in the general population[59]. Other studies report that DM, obesity and steatosis are associated with liver disease and more severe fibrosis in CHC[6061].
It is important to note that neither the Child-Pugh nor Model for End-Stage Liver Disease (MELD) Scores (which are widely used as prognostic instruments of morbidity and mortality in the short and long term for cirrhotic patients) include in their parameters DM or glucose intolerance[6263]. Nevertheless, interesting data have been observed in some prospective longitudinal studies involving cirrhotic patients where DM has been studied as an independent prognostic factor. In a retrospective and prospective study 354 (98 with diabetes) of 382 eligible patients were followed for 6 years after inclusion into the study: 110 were alive at the end of follow-up. Prognostic factors identified by Kaplan-Meier analysis, followed by Cox’s stepwise regression demonstrated in sequence, albumin, ascites, age, encephalopathy, bilirubin, diabetes, and platelets as prognostic factors of mortality. The larger mortality rate in patients with diabetes, was not due to complications of diabetes but to an increased risk of hepatocellular failure[58]. Diabetes was no longer a risk factor as a covariate in a subgroup of 271 patients when varices were added but was again significant when patients who died of gastrointestinal bleeding were excluded.
In another study carried out in patients suffering from cirrhosis and refractory ascites on the waiting list for liver transplantation it was observed that the HCC and DM, but not the Child-Pugh score, were independent predictive factors of mortality. The patients suffering from refractory ascites and DM showed a 1- and 2-year probability of survival of 32% and 18%, respectively. By contrast survival rates of patients with refractory ascites without DM were 62% and 58%, respectively[64].
Nishida et al performed the OGTT on a group of 56 patients with cirrhosis and normal fasting blood glucose. A total of 38% of patients were diagnosed with DM, 23% with glucose intolerance, and 39% were normal. After 5 years of follow-up, patients with diabetes and glucose intolerance had significantly higher mortality than normal patients (44% and 32% vs 5%, respectively). From a multiple regression analysis only serum albumin and DM were independent negative predictive factors of survival[19].
Hepatogenous diabetes has a clinical behavior different from that of hereditary type 2 DM, since it is less frequently associated with retinopathy and cardiovascular and renal complications[558]. In cirrhotic patients with diabetes, the most recurrent cause of death is liver failure[41958].
At present, type 2 DM is considered a risk factor for the occurrence of HCC. Hepatogenous diabetes together with hepatitis B and C virus infection and alcoholic liver cirrhosis increases the risk of HCC by 10-fold[12].
Patients with HCC and DM have a mortality risk higher than patients with HCC without DM. In another study involving 160 patients suffering from HCC, those who had DM had a 1-year mortality rate higher than those patients without DM. Additionally, they had more extensive disease[65].
The mechanisms by which diabetes worsens the clinical course of liver cirrhosis have not been clearly established. Firstly, DM accelerates liver fibrosis and inflammation giving rise to more severe liver failure. Secondly, DM may potentiate the incidence of bacterial infections in cirrhotic patients which are associated with increased mortality[6667].
In relation to the first mechanism, insulin resistance increases adipokine production (cytokines secreted by adipose tissue), such as leptin and TNF-α, which activates the inflammatory pathways that exacerbate liver damage[68]. In contrast, another cytokine produced by adipose tissue, adiponectin, is a regulator of insulin sensitivity and tissue inflammation[69]. A reduction in the adiponectin levels reflects peripheral and hepatic insulin resistance[70]. There has been speculation that hypoadiponectinemia may play a role in liver disease progression[7071].
Regarding the second mechanism, DM may worsen immunodepression in cirrhotic patients thus increasing the incidence of severe infections which may have deleterious effect on liver function. It should be noted that cirrhotic patients with spontaneous bacterial peritonitis have a high hospital mortality rate due to sepsis, liver failure and hepatorenal syndrome. On the other hand, patients with esophageal variceal bleeding have a high incidence of infections that increase their in-hospital mortality rate[72]. Notwithstanding, it has not been established if DM increases the mortality rate in patients with other complications of cirrhosis.
In future, the precise mechanisms by which DM may worsen liver function should be clarified, since manipulation of these may be useful for reduction of complications.
Clinical manifestations of DM in the early stages of cirrhosis are virtually absent. In a recently published study involving compensated cirrhotic patients with normal fasting serum glucose and without a family history of type 2 DM, up to 77% had DM or glucose intolerance diagnosed by means of OGTT. In 38% of cases, DM was subclinical[19]. As liver function deteriorates, the incidence of diabetes increases so that clinical diabetes may be seen as a marker of liver failure.
Hepatogenous diabetes has particular clinical characteristics: (1) unlike the hereditary type 2 DM, it is less frequently associated with risk factors such as age, body mass index, and family history of diabetes; (2) it is less frequently associated with retinopathy and cardiovascular and renal complications; (3) it is more frequently associated with hypoglycemic episodes as a result of impaired liver function[219].
Although the incidence of obesity, DM and metabolic syndrome have increased in the world, reaching epidemic proportions, the role of DM as a prognostic factor of morbidity and mortality in cirrhotic patients has been scarcely studied. In addition, the impact of early diagnosis and treatment of DM on the clinical course of cirrhosis is unknown.
Few studies have evaluated what is the most efficacious therapy for DM in cirrhotic patients and what is the impact of treatment of DM on the clinical course of liver disease.
The treatment of DM of cirrhotic patients has particular characteristics that make it different from type 2 DM without liver disease: (1) about half the patients have malnutrition; (2) when clinical DM is diagnosed, the patient has advanced liver disease; (3) most of the oral hypoglycemic agents are metabolized in the liver; (4) patients often have episodes of hypoglycemia[3].
The initial treatment of patients suffering from mild to moderate hyperglycemia and compensated liver disease may be a lifestyle change, since at this stage the insulin resistance is a dominant factor. However, these therapeutic measures may be compromised by very restrictive diets, since they might aggravate malnutrition in some patients. On the other hand, physical exercise which improves insulin resistance may not be an appropriate measure in patients with active liver disease[15].
When DM manifests in advanced stages of liver disease, the use of oral hypoglycemic drugs may be required. However, most of these drugs are metabolized in the liver, therefore, the blood glucose levels during treatment shall be closely monitored in order to avoid hypoglycemia[73]. In these cases, biguanides, which reduce resistance to insulin, may be useful. Metformin is a biguanide that is relatively contraindicated in patients with advanced liver failure and patients who continue to ingest alcohol, because of the risk of lactic acidosis[74].
On the other hand, insulin secretagogues, despite the fact that they are safe drugs in patients with liver disease, probably are not useful, since they do not modify insulin resistance and patients with alcoholic cirrhosis often have pancreatic islet β-cell damage[75]. These patients have chronic compensatory hyperinsulinemia until the islet β-cells are exhausted.
Alpha-glucosidase inhibitors can be useful in patients suffering from liver cirrhosis, since their mechanism of action is to reduce carbohydrate absorption in the bowel, thus reducing the risk of postprandial hyperglycemia that is common in these patients. In a randomized, double-blind study involving 100 patients with compensated liver cirrhosis and insulin-treated DM, the control of postprandial and fasting blood glucose levels improved significantly with the use of acarbose, an alpha-glucosidase[76]. In another crossover placebo-controlled study involving patients with hepatic encephalopathy, acarbose produced a significant improvement in postprandial blood glucose level. Additionally, the patients had a reduction in plasma ammonia levels and an increase in the frequency of bowel movements[77]. The reduction in ammonia levels was probably the result of a decrease in the proliferation of intestinal proteolytic bacteria caused by bowel movement[77].
Thiazolidines may be particularly useful in cirrhotic patients with DM, since they increase the insulin sensitivity. However, troglitazone has been withdrawn from the market because of its potential hepatotoxic effects. Nevertheless, rosiglitazone and pioglitazone appear to be safer drugs[78], but it is recommended that these drugs should not be initiated if there is evidence of active liver disease or if alanine transaminase levels are above 2.5 times the upper limit of normal. The use of these drugs should be monitored closely whenever necessary.
Insulin requirements in the cirrhotic patient with diabetes may vary. In patients with compensated cirrhosis, requirements may be greater compared to patients with decompensated cirrhosis, since insulin resistance predominates in the former while in the latter liver metabolism of insulin is greatly reduced. Therefore, therapy with insulin must be preferably performed in hospitalized patients with close monitoring of blood glucose levels for development of hypoglycemia[79].
Finally, liver transplantation rapidly normalizes glucose tolerance and insulin sensitivity. It is thought that this effect is due to an improvement in the hepatic clearance and peripheral glucose disposal. The latter effect could be secondary to a correction of chronic hyperinsulinemia[1680]. It has been observed that liver transplantation, in reducing insulin resistance, cures hepatogenous diabetes in 67% of cirrhotic-diabetic patients. In 33% of patients diabetes was not corrected because of persistence of a reduced β-cell function measured by means of an OGTT. This abnormality would make these patients eventually eligible for combined islet transplantation[81].
Future research in the field of DM in cirrhotic patients should clarify the following issues: (1) the role of isolated type 2 DM in the genesis of chronic liver disease and the factors involved in this complication; (2) the impact of hepatogenous diabetes in the natural history of cirrhotic patients; (3) the impact of early diagnosis and treatment of hepatogenous diabetes (through OGTT) in reducing mortality; (4) the benefits of controlling the DM in the management of complications of liver cirrhosis; (5) the mechanisms by which hepatogenous diabetes increases morbidity and mortality of cirrhotic patients, as well as the impact of the manipulation of these mechanisms on the patients; (6) the establishment of clearer guidelines for management of diabetes in the cirrhotic patient.
Perhaps the combination of DM with the currently used scores (Child-Pugh and MELD scores) may enhance the sensitivity and the specificity for prediction of morbidity and mortality rates in cirrhotic patients.
| 1. | Hickman IJ, Macdonald GA. Impact of diabetes on the severity of liver disease. Am J Med. 2007;120:829-834. [Cited in This Article: ] |
| 2. | El-Serag HB, Tran T, Everhart JE. Diabetes increases the risk of chronic liver disease and hepatocellular carcinoma. Gastroenterology. 2004;126:460-468. [Cited in This Article: ] |
| 3. | Tolman KG, Fonseca V, Dalpiaz A, Tan MH. Spectrum of liver disease in type 2 diabetes and management of patients with diabetes and liver disease. Diabetes Care. 2007;30:734-743. [Cited in This Article: ] |
| 4. | El-Serag HB, Everhart JE. Diabetes increases the risk of acute hepatic failure. Gastroenterology. 2002;122:1822-1828. [Cited in This Article: ] |
| 5. | Holstein A, Hinze S, Thiessen E, Plaschke A, Egberts EH. Clinical implications of hepatogenous diabetes in liver cirrhosis. J Gastroenterol Hepatol. 2002;17:677-681. [Cited in This Article: ] |
| 6. | Picardi A, D'Avola D, Gentilucci UV, Galati G, Fiori E, Spataro S, Afeltra A. Diabetes in chronic liver disease: from old concepts to new evidence. Diabetes Metab Res Rev. 2006;22:274-283. [Cited in This Article: ] |
| 7. | Custro N, Carroccio A, Ganci A, Scafidi V, Campagna P, Di Prima L, Montalto G. Glycemic homeostasis in chronic viral hepatitis and liver cirrhosis. Diabetes Metab. 2001;27:476-481. [Cited in This Article: ] |
| 8. | Postic C, Dentin R, Girard J. Role of the liver in the control of carbohydrate and lipid homeostasis. Diabetes Metab. 2004;398-408. [Cited in This Article: ] |
| 9. | Barthel A, Schmoll D. Novel concepts in insulin regulation of hepatic gluconeogenesis. Am J Physiol Endocrinol Metab. 2003;285:E685-E692. [Cited in This Article: ] |
| 10. | Cotrozzi G, Casini Raggi V, Relli P, Buzzelli G. [Role of the liver in the regulation of glucose metabolism in diabetes and chronic liver disease]. Ann Ital Med Int. 1997;12:84-91. [Cited in This Article: ] |
| 11. | Tappy L, Minehira K. New data and new concepts on the role of the liver in glucose homeostasis. Curr Opin Clin Nutr Metab Care. 2001;4:273-277. [Cited in This Article: ] |
| 12. | Nielsen MF, Caumo A, Aagaard NK, Chandramouli V, Schumann WC, Landau BR, Schmitz O, Vilstrup H. Contribution of defects in glucose uptake to carbohydrate intolerance in liver cirrhosis: assessment during physiological glucose and insulin concentrations. Am J Physiol Gastrointest Liver Physiol. 2005;288:G1135-G1143. [Cited in This Article: ] |
| 13. | Steppan CM, Bailey ST, Bhat S, Brown EJ, Banerjee RR, Wright CM, Patel HR, Ahima RS, Lazar MA. The hormone resistin links obesity to diabetes. Nature. 2001;409:307-312. [Cited in This Article: ] |
| 14. | Petrides AS, Vogt C, Schulze-Berge D, Matthews D, Strohmeyer G. Pathogenesis of glucose intolerance and diabetes mellitus in cirrhosis. Hepatology. 1994;19:616-627. [Cited in This Article: ] |
| 15. | Petrides AS, Stanley T, Matthews DE, Vogt C, Bush AJ, Lambeth H. Insulin resistance in cirrhosis: prolonged reduction of hyperinsulinemia normalizes insulin sensitivity. Hepatology. 1998;28:141-149. [Cited in This Article: ] |
| 16. | Merli M, Leonetti F, Riggio O, Valeriano V, Ribaudo MC, Strati F, Tisone G, Casciani CU, Capocaccia L. Glucose intolerance and insulin resistance in cirrhosis are normalized after liver transplantation. Hepatology. 1999;30:649-654. [Cited in This Article: ] |
| 17. | Petrides AS, Groop LC, Riely CA, DeFronzo RA. Effect of physiologic hyperinsulinemia on glucose and lipid metabolism in cirrhosis. J Clin Invest. 1991;88:561-570. [Cited in This Article: ] |
| 18. | Lecube A, Hernandez C, Genesca J, Esteban JI, Jardi R, Simo R. High prevalence of glucose abnormalities in patients with hepatitis C virus infection: a multivariate analysis considering the liver injury. Diabetes Care. 2004;27:1171-1175. [Cited in This Article: ] |
| 19. | Nishida T, Tsuji S, Tsujii M, Arimitsu S, Haruna Y, Imano E, Suzuki M, Kanda T, Kawano S, Hiramatsu N. Oral glucose tolerance test predicts prognosis of patients with liver cirrhosis. Am J Gastroenterol. 2006;101:70-75. [Cited in This Article: ] |
| 20. | Di Bisceglie AM. What every hepatologist should know about endocrinology: obesity, diabetes, and liver disease. Gastroenterology. 2004;126:604-606. [Cited in This Article: ] |
| 21. | Wideroff L, Gridley G, Mellemkjaer L, Chow WH, Linet M, Keehn S, Borch-Johnsen K, Olsen JH. Cancer incidence in a population-based cohort of patients hospitalized with diabetes mellitus in Denmark. J Natl Cancer Inst. 1997;89:1360-1365. [Cited in This Article: ] |
| 22. | Fujino Y, Mizoue T, Tokui N, Yoshimura T. Prospective study of diabetes mellitus and liver cancer in Japan. Diabetes Metab Res Rev. 2001;17:374-379. [Cited in This Article: ] |
| 23. | Tazawa J, Maeda M, Nakagawa M, Ohbayashi H, Kusano F, Yamane M, Sakai Y, Suzuki K. Diabetes mellitus may be associated with hepatocarcinogenesis in patients with chronic hepatitis C. Dig Dis Sci. 2002;47:710-715. [Cited in This Article: ] |
| 24. | Donadon V, Balbi M, Casarin P, Vario A, Alberti A. Association between hepatocellular carcinoma and type 2 diabetes mellitus in Italy: Potential role of insulin. World J Gastroenterol. 2008;14:5695-5700. [Cited in This Article: ] |
| 25. | Veldt BJ, Chen W, Heathcote EJ, Wedemeyer H, Reichen J, Hofmann WP, de Knegt RJ, Zeuzem S, Manns MP, Hansen BE. Increased risk of hepatocellular carcinoma among patients with hepatitis C cirrhosis and diabetes mellitus. Hepatology. 2008;47:1856-1862. [Cited in This Article: ] |
| 26. | Browning JD, Szczepaniak LS, Dobbins R, Nuremberg P, Horton JD, Cohen JC, Grundy SM, Hobbs HH. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology. 2004;40:1387-1395. [Cited in This Article: ] |
| 27. | Angulo P. GI epidemiology: nonalcoholic fatty liver disease. Aliment Pharmacol Ther. 2007;25:883-889. [Cited in This Article: ] |
| 28. | Caldwell SH, Oelsner DH, Iezzoni JC, Hespenheide EE, Battle EH, Driscoll CJ. Cryptogenic cirrhosis: clinical characterization and risk factors for underlying disease. Hepatology. 1999;29:664-669. [Cited in This Article: ] |
| 29. | Tellez-Avila FI, Sanchez-Avila F, Garcia-Saenz-de-Sicilia M, Chavez-Tapia NC, Franco-Guzman AM, Lopez-Arce G, Cerda-Contreras E, Uribe M. Prevalence of metabolic syndrome, obesity and diabetes type 2 in cryptogenic cirrhosis. World J Gastroenterol. 2008;14:4771-4775. [Cited in This Article: ] |
| 30. | Chalasani N, Gorski JC, Asghar MS, Asghar A, Foresman B, Hall SD, Crabb DW. Hepatic cytochrome P450 2E1 activity in nondiabetic patients with nonalcoholic steatohepatitis. Hepatology. 2003;37:544-550. [Cited in This Article: ] |
| 31. | Pessayre D, Fromenty B, Mansouri A. Mitochondrial injury in steatohepatitis. Eur J Gastroenterol Hepatol. 2004;16:1095-1105. [Cited in This Article: ] |
| 32. | Crespo J, Cayon A, Fernandez-Gil P, Hernandez-Guerra M, Mayorga M, Dominguez-Diez A, Fernandez-Escalante JC, Pons-Romero F. Gene expression of tumor necrosis factor alpha and TNF-receptors, p55 and p75, in nonalcoholic steatohepatitis patients. Hepatology. 2001;34:1158-1163. [Cited in This Article: ] |
| 33. | Sanyal AJ. AGA technical review on nonalcoholic fatty liver disease. Gastroenterology. 2002;123:1705-1725. [Cited in This Article: ] |
| 34. | Bertolani C, Marra F. The role of adipokines in liver fibrosis. Pathophysiology. 2008;15:91-101. [Cited in This Article: ] |
| 35. | Buzzelli G, Chiarantini E, Cotrozzi G, Relli P, Matassi L, Romanelli RG, Gentilini P. Estimate of prevalence of glucose intolerance in chronic liver disease. Degree of agreement among some diagnostic criteria. Liver. 1988;8:354-359. [Cited in This Article: ] |
| 36. | Niederau C, Fischer R, Purschel A, Stremmel W, Haussinger D, Strohmeyer G. Long-term survival in patients with hereditary hemochromatosis. Gastroenterology. 1996;110:1107-1119. [Cited in This Article: ] |
| 37. | Del Vecchio Blanco C, Gentile S, Marmo R, Carbone L, Coltorti M. Alterations of glucose metabolism in chronic liver disease. Diabetes Res Clin Pract. 1990;8:29-36. [Cited in This Article: ] |
| 38. | Harrison SA. Liver disease in patients with diabetes mellitus. J Clin Gastroenterol. 2006;40:68-76. [Cited in This Article: ] |
| 39. | Angulo P, Keach JC, Batts KP, Lindor KD. Independent predictors of liver fibrosis in patients with nonalcoholic steatohepatitis. Hepatology. 1999;30:1356-1362. [Cited in This Article: ] |
| 40. | Romero-Gomez M. Insulin resistance and hepatitis C. World J Gastroenterol. 2006;12:7075-7080. [Cited in This Article: ] |
| 41. | Qureshi K, Abrams GA. Metabolic liver disease of obesity and role of adipose tissue in the pathogenesis of nonalcoholic fatty liver disease. World J Gastroenterol. 2007;13:3540-3553. [Cited in This Article: ] |
| 42. | Hatzitolios A, Savopoulos C, Lazaraki G, Sidiropoulos I, Haritanti P, Lefkopoulos A, Karagiannopoulou G, Tzioufa V, Dimitrios K. Efficacy of omega-3 fatty acids, atorvastatin and orlistat in non-alcoholic fatty liver disease with dyslipidemia. Indian J Gastroenterol. 2004;23:131-134. [Cited in This Article: ] |
| 43. | Mehta SH, Brancati FL, Sulkowski MS, Strathdee SA, Szklo M, Thomas DL. Prevalence of type 2 diabetes mellitus among persons with hepatitis C virus infection in the United States. Ann Intern Med. 2000;133:592-599. [Cited in This Article: ] |
| 44. | Knobler H, Schihmanter R, Zifroni A, Fenakel G, Schattner A. Increased risk of type 2 diabetes in noncirrhotic patients with chronic hepatitis C virus infection. Mayo Clin Proc. 2000;75:355-359. [Cited in This Article: ] |
| 45. | Anty R, Gelsi E, Giudicelli J, Marine-Barjoan E, Gual P, Benzaken S, Saint-Paul MC, Sadoul JL, Huet PM, Tran A. Glucose intolerance and hypoadiponectinemia are already present in lean patients with chronic hepatitis C infected with genotype non-3 viruses. Eur J Gastroenterol Hepatol. 2007;19:671-677. [Cited in This Article: ] |
| 46. | Lecube A, Hernandez C, Genesca J, Simo R. Proinflammatory cytokines, insulin resistance, and insulin secretion in chronic hepatitis C patients: A case-control study. Diabetes Care. 2006;29:1096-1101. [Cited in This Article: ] |
| 47. | Moucari R, Asselah T, Cazals-Hatem D, Voitot H, Boyer N, Ripault MP, Sobesky R, Martinot-Peignoux M, Maylin S, Nicolas-Chanoine MH. Insulin resistance in chronic hepatitis C: association with genotypes 1 and 4, serum HCV RNA level, and liver fibrosis. Gastroenterology. 2008;134:416-423. [Cited in This Article: ] |
| 48. | Hui JM, Sud A, Farrell GC, Bandara P, Byth K, Kench JG, McCaughan GW, George J. Insulin resistance is associated with chronic hepatitis C virus infection and fibrosis progression [corrected]. Gastroenterology. 2003;125:1695-1704. [Cited in This Article: ] |
| 49. | Romero-Gomez M, Del Mar Viloria M, Andrade RJ, Salmeron J, Diago M, Fernandez-Rodriguez CM, Corpas R, Cruz M, Grande L, Vazquez L. Insulin resistance impairs sustained response rate to peginterferon plus ribavirin in chronic hepatitis C patients. Gastroenterology. 2005;128:636-641. [Cited in This Article: ] |
| 50. | Sanyal AJ, Chand N, Comar K, Mirshahi F. Hyperinsulinemia blocks the inhibition of hepatitis C virus (HCV) replication by interferon: a potential mechanism for failure of interferon therapy in subjects with HCV and nonalcoholic liver disease. Hepatology. 2004;40:179A. [Cited in This Article: ] |
| 51. | Giordanino C, Bugianesi E, Smedile A, Ciancio A, Abate ML, Olivero A, Pellicano R, Cassader M, Gambino R, Bo S. Incidence of type 2 diabetes mellitus and glucose abnormalities in patients with chronic hepatitis C infection by response to treatment: results of a cohort study. Am J Gastroenterol. 2008;103:2481-2487. [Cited in This Article: ] |
| 52. | Zein NN, Abdulkarim AS, Wiesner RH, Egan KS, Persing DH. Prevalence of diabetes mellitus in patients with end-stage liver cirrhosis due to hepatitis C, alcohol, or cholestatic disease. J Hepatol. 2000;32:209-217. [Cited in This Article: ] |
| 53. | Wei M, Gibbons LW, Mitchell TL, Kampert JB, Blair SN. Alcohol intake and incidence of type 2 diabetes in men. Diabetes Care. 2000;23:18-22. [Cited in This Article: ] |
| 54. | Adams PC, Kertesz AE, Valberg LS. Clinical presentation of hemochromatosis: a changing scene. Am J Med. 1991;90:445-449. [Cited in This Article: ] |
| 55. | Greco AV, Mingrone G, Mari A, Capristo E, Manco M, Gasbarrini G. Mechanisms of hyperinsulinaemia in Child’s disease grade B liver cirrhosis investigated in free living conditions. Gut. 2002;51:870-875. [Cited in This Article: ] |
| 56. | Kim MG, Choi WC. [Differential diagnosis of diabetes mellitus caused by liver cirrhosis and other type 2 diabetes mellitus]. Korean J Hepatol. 2006;12:524-529. [Cited in This Article: ] |
| 57. | de Marco R, Locatelli F, Zoppini G, Verlato G, Bonora E, Muggeo M. Cause-specific mortality in type 2 diabetes. The Verona Diabetes Study. Diabetes Care. 1999;22:756-761. [Cited in This Article: ] |
| 58. | Bianchi G, Marchesini G, Zoli M, Bugianesi E, Fabbri A, Pisi E. Prognostic significance of diabetes in patients with cirrhosis. Hepatology. 1994;20:119-125. [Cited in This Article: ] |
| 59. | Trombetta M, Spiazzi G, Zoppini G, Muggeo M. Review article: type 2 diabetes and chronic liver disease in the Verona diabetes study. Aliment Pharmacol Ther. 2005;22 Suppl 2:24-27. [Cited in This Article: ] |
| 60. | Hourigan LF, Macdonald GA, Purdie D, Whitehall VH, Shorthouse C, Clouston A, Powell EE. Fibrosis in chronic hepatitis C correlates significantly with body mass index and steatosis. Hepatology. 1999;29:1215-1219. [Cited in This Article: ] |
| 61. | Taura N, Ichikawa T, Hamasaki K, Nakao K, Nishimura D, Goto T, Fukuta M, Kawashimo H, Fujimoto M, Kusumoto K. Association between liver fibrosis and insulin sensitivity in chronic hepatitis C patients. Am J Gastroenterol. 2006;101:2752-2759. [Cited in This Article: ] |
| 62. | Flores-Rendon AR, Gonzalez-Gonzalez JA, Garcia-Compean D, Maldonado-Garza HJ, Garza-Galindo AA. Model for end stage of liver disease (MELD) is better than the Child-Pugh score for predicting in-hospital mortality related to esophageal variceal bleeding. Ann Hepatol. 2008;7:230-234. [Cited in This Article: ] |
| 63. | Durand F, Valla D. Assessment of the prognosis of cirrhosis: Child-Pugh versus MELD. J Hepatol. 2005;42 Suppl:S100-S107. [Cited in This Article: ] |
| 64. | Moreau R, Delegue P, Pessione F, Hillaire S, Durand F, Lebrec D, Valla DC. Clinical characteristics and outcome of patients with cirrhosis and refractory ascites. Liver Int. 2004;24:457-464. [Cited in This Article: ] |
| 65. | Amarapurkar DN, Patel ND, Kamani PM. Impact of diabetes mellitus on outcome of HCC. Ann Hepatol. 2008;7:148-151. [Cited in This Article: ] |
| 66. | Garcia-Tsao G. Bacterial infections in cirrhosis: treatment and prophylaxis. J Hepatol. 2005;42 Suppl:S85-S92. [Cited in This Article: ] |
| 67. | Cheruvattath R, Balan V. Infections in Patients With End-stage Liver Disease. J Clin Gastroenterol. 2007;41:403-411. [Cited in This Article: ] |
| 68. | Roden M. Mechanisms of Disease: hepatic steatosis in type 2 diabetes--pathogenesis and clinical relevance. Nat Clin Pract Endocrinol Metab. 2006;2:335-348. [Cited in This Article: ] |
| 69. | Whitehead JP, Richards AA, Hickman IJ, Macdonald GA, Prins JB. Adiponectin--a key adipokine in the metabolic syndrome. Diabetes Obes Metab. 2006;8:264-280. [Cited in This Article: ] |
| 70. | Jonsson JR, Moschen AR, Hickman IJ, Richardson MM, Kaser S, Clouston AD, Powell EE, Tilg H. Adiponectin and its receptors in patients with chronic hepatitis C. J Hepatol. 2005;43:929-936. [Cited in This Article: ] |
| 71. | Svegliati-Baroni G, Ridolfi F, Di Sario A, Casini A, Marucci L, Gaggiotti G, Orlandoni P, Macarri G, Perego L, Benedetti A. Insulin and insulin-like growth factor-1 stimulate proliferation and type I collagen accumulation by human hepatic stellate cells: differential effects on signal transduction pathways. Hepatology. 1999;29:1743-1751. [Cited in This Article: ] |
| 72. | Hou MC, Lin HC, Liu TT, Kuo BI, Lee FY, Chang FY, Lee SD. Antibiotic prophylaxis after endoscopic therapy prevents rebleeding in acute variceal hemorrhage: a randomized trial. Hepatology. 2004;39:746-753. [Cited in This Article: ] |
| 73. | Marks V, Teale JD. Drug-induced hypoglycemia. Endocrinol Metab Clin North Am. 1999;28:555-577. [Cited in This Article: ] |
| 74. | Nair S, Diehl AM, Wiseman M, Farr GH Jr, Perrillo RP. Metformin in the treatment of non-alcoholic steatohepatitis: a pilot open label trial. Aliment Pharmacol Ther. 2004;20:23-28. [Cited in This Article: ] |
| 75. | Choudhury S, Hirschberg Y, Filipek R, Lasseter K, McLeod JF. Single-dose pharmacokinetics of nateglinide in subjects with hepatic cirrhosis. J Clin Pharmacol. 2000;40:634-640. [Cited in This Article: ] |
| 76. | Gentile S, Turco S, Guarino G, Oliviero B, Annunziata S, Cozzolino D, Sasso FC, Turco A, Salvatore T, Torella R. Effect of treatment with acarbose and insulin in patients with non-insulin-dependent diabetes mellitus associated with non-alcoholic liver cirrhosis. Diabetes Obes Metab. 2001;3:33-40. [Cited in This Article: ] |
| 77. | Gentile S, Guarino G, Romano M, Alagia IA, Fierro M, Annunziata S, Magliano PL, Gravina AG, Torella R. A randomized controlled trial of acarbose in hepatic encephalopathy. Clin Gastroenterol Hepatol. 2005;3:184-191. [Cited in This Article: ] |
| 78. | Lebovitz HE, Kreider M, Freed MI. Evaluation of liver function in type 2 diabetic patients during clinical trials: evidence that rosiglitazone does not cause hepatic dysfunction. Diabetes Care. 2002;25:815-821. [Cited in This Article: ] |
| 79. | Petrides AS. Hepatogenic diabetes: pathophysiology, therapeutic options and prognosis. Z Gastroenterology. 1999;16:15-21. [Cited in This Article: ] |
| 80. | Blanco JJ, Herrero JI, Quiroga J, Sangro B, Gomez-Manero N, Pardo F, Cienfuegos JA, Prieto J. Liver transplantation in cirrhotic patients with diabetes mellitus: midterm results, survival, and adverse events. Liver Transpl. 2001;7:226-233. [Cited in This Article: ] |
| 81. | Perseghin G, Mazzaferro V, Sereni LP, Regalia E, Benedini S, Bazzigaluppi E, Pulvirenti A, Leao AA, Calori G, Romito R. Contribution of reduced insulin sensitivity and secretion to the pathogenesis of hepatogenous diabetes: effect of liver transplantation. Hepatology. 2000;31:694-703. [Cited in This Article: ] |