Published online Apr 14, 2009. doi: 10.3748/wjg.15.1730
Revised: March 5, 2009
Accepted: March 12, 2009
Published online: April 14, 2009
AIM: To investigate the effect of Bu-Zhong-Yi-Qi-Tang (Decoction for Reinforcing Middle Jiao and Replenishing Qi) on deficiency of N-glycan/nitric oxide (NO) and islet damage induced by injecting two medium doses of streptozotocin (STZ).
METHODS: Diabetes was induced by intraperitoneal injection of STZ at 55 mg/kg on day 1 and day 8. Islet damage was evaluated using a scoring system. Nitrite, nitrate, α-mannosidase and amylase activities were measured by colorimetry. N-glycan patterns of amylase were determined with lectin [ConA, pisum sativum agglutinin (PSA), peanut agglutinin (PNA), and lens culinaris agglutinin (LCA)] affinity precipitation method.
RESULTS: Severe islet necrosis and mild islet atrophy were observed in diabetic rats. The number and size of islets, the activities of α-mannosidase, amylase and nitrite were decreased, while the binding of PNA and LCA to amylase was increased. All of which were improved after treatment with Bu-Zhong-Yi-Qi-Tang. Islet damage was significantly correlated with nitrite, nitrate, α-mannosidase, amylase and the binding of LCA, PNA, and PSA to amylase.
CONCLUSION: STZ-induced islet damage is related to N-glycan deficiency in proteins by blocking α-mannosidase activity and no deficiency, accumulation of unfolded proteins, and endoplasmic reticulum stress and activation of cellular signals, all of which are improved after treatment with Bu-Zhong-Yi-Qi-Tang.
- Citation: Liu XQ, Wu L, Guo XJ. Effect of Bu-Zhong-Yi-Qi-Tang on deficiency of N-glycan/nitric oxide and islet damage induced by streptozotocin in diabetic rats. World J Gastroenterol 2009; 15(14): 1730-1737
- URL: https://www.wjgnet.com/1007-9327/full/v15/i14/1730.htm
- DOI: https://dx.doi.org/10.3748/wjg.15.1730
N-glycan plays an important role in the quality control (QC) of glycoprotein folding both in endoplasmic reticulum (ER) lumen and in ER-associated degradation (ERAD) of proteins by cytosolic proteasomes[1]. Alpha-mannosidase, a key enzyme converting precursor high-mannose-type N-glycans to matured complex-type structures, contributes to the establishment of an equitable glycoprotein quality control standard, by which the efficiency of Asn-linked glycoprotein conformational maturation results in dislocation of misfolded glycoproteins into the cytosol, where proteins are degraded in proteasomes and maintain the homeostasis of ER[2].
The unfolded protein response (UPR) is a conserved cellular response designed to alleviate damage and promote survival of cells experiencing stress by either shutting down translation to reduce the protein flow into ER, or by up-regulating molecules that protect cells or clear misfolded proteins from the ER, thus alleviating ER stress. Nitric oxide (NO) is a known activator of the UPR[3–5], and NO-induced UPR activation may function to assist in the recovery of β-cells from NO-mediated damage, which is associated with enhanced expression of heat shock proteins and chaperones that assist in protein folding.
Islet β cells have a well-developed ER and secrete a large amount of insulin and glycoproteins. Islet β cells may be at risk of developing ER stress and increase misfolded glycoproteins. Inability to clear misfolded proteins causes early accumulation of unfolded proteins, and ER stress and activation of cellular signals lead to cell death, thus resulting in loss of β cell secretion potential and diabetes.
Xiong et al[6] showed that the pathogenesis of diabetes is closely correlated to Pixu (insufficiency of the spleen), a major pathogenic factor for diabetes syndrome. Pixu is therefore considered in the treatment of diabetes. Diabetes mellitus is caused by Pixu, when diabetes progresses, deficiency of Yin and dryness-heat exist simultaneously, and when diabetes prolongs, deficiency of Yin becomes the key pathogenesis, finally resulting in impaired Yin and Yang[7].
The levels of nitrite and nitrate are significantly decreased in stimulated crude whole saliva of diabetic patients with Pixu[8]. Bu-Zhong-Yi-Qi-Tang, a traditional Chinese medicine, tonifies qi, strengthens the stomach and spleen, and increases sunken Yang qi. It is prescribed mainly for Pixu by improving the deficiency of spleen and stomach qi. It was reported that the lower levels of nitrite, nitrate, and peroxynitrite in gastric mucosa of rats with Pixu can return to normal after treatment with Bu-Zhong-Yi-Qi-Tang[9].
This study was to examine the effect of Bu-Zhong-Yi-Qi-Tang on deficiency of N-glycan/NO and islets damage in rats with diabetes mellitus induced by streptozotocin (STZ).
This study was approved by Guangdong Science and Technology Committee and Guangdong Management Committee for Medical Experimental Animals.
Sprague-Dawley rats were obtained from the Laboratory Animal Research Center of Guangzhou University of Traditional Chinese Medicine. The animals were housed in a plastic cage in an air-conditioned room at 20 ± 2°C with a humidity of 58% ± 5% in a 12-h light and dark cycle, with free access to standard rat food and tap water. The rats were divided into normal control, and diabetic model groups after 1 wk.
A diabetic model of rats was established by intraperitoneal injection of STZ at 55 mg/kg on day 1 and day 8. The rats were bred for 4 wk. Blood was sampled from the tail vein and blood glucose was determined with the one touch ultra blood glucose monitoring system (LifeScan, Inc. Milpitas, CA, USA). Animals with their non-fasting blood glucose less than 16.65 mmol/L were excluded from the study. The rats were divided into diabetics groups and Bu-Zhong-Yi-Qi-Tang treatment group.
Bu-Zhong-Yi-Qi-Tang was made of Radix Astragali, Codonopsis pilosula, Glycyrrhiza uralensisf isch, Rhizoma Atractylodis Macrocephalae, Radix Angelicae Sinensis, Rhizoma Cimicifugae Foetidae, Radix Bupleuri Chinensis, and Pericarpium Citri Reticulatae, which were put into a 20-fold volume of distilled water, decocted from 80°C to 100°C, filtered and concentrated at 40°C-80°C, and stored in a refrigerator until use. Bu-Zhong-Yi-Qi-Tang was administered by gavage, 8 g/kg per day for 4 wk.
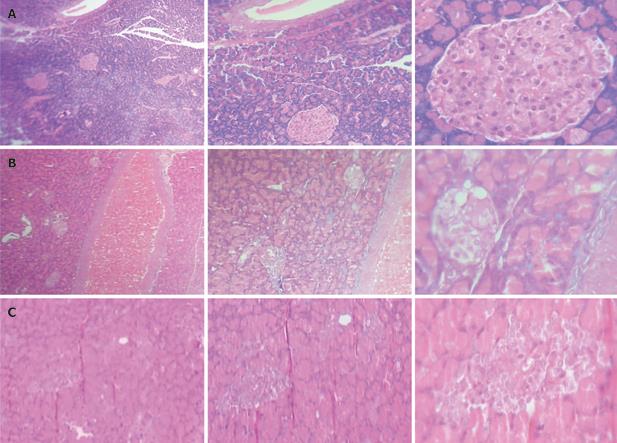
At the end of treatment, the animals were killed by exsanguination from the carotid artery. Blood samples were taken immediately and centrifuged at 3000 r/min for 5 min. Serum was separated for the measurement of nitrite, nitrate, α-mannosidase and amylase activities, and determination of the N-glycan patterns of amylase. The pancreas was removed from each rat and dissected longitudinally. One half was dried and homogenized. Supernatant was removed for the measurement of nitrite, nitrate, α-mannosidase and amylase activities, and determination of the N-glycan patterns of amylase. The other half was fixed in Bouin’s solution for 24 h. The pancreatic tissue, embedded in paraffin wax, was cut into 3-4-&mgr;m-thick sections which were stained with hematoxylin and eosin (HE) for histopathological examination.
Bouin’s-solution-fixed pancreatic tissue was cut into eight sections (approximately 1 mm × 1 mm × 1 mm). Four sections were randomly selected, stained with HE, and observed under a microscope with a digital camera (C170 D425, Olympus Imaging Corp). Digital micrographs were analyzed for estimating the number, size, necrosis and atrophy of islets and insulitis by two observers unaware of the status and/or treatment modalities.
The number of islets was calculated as previously described[10] with some minor modifications and graded as 0, no visible islets; 1, < 5 islets; 2, 6-10 islets; 3, 11-20 islets; 4, > 20 islets.
The size of islets was measured as previously described[11] with some minor modifications and graded as 1, 1-40 cells per section; 2, 41-100 cells per section; 3, 101-200 cells per section; 4, 201-400 cells per section; 5, > 400 cells per section.
Insulitis was evaluated by examining at least 10 islets from each treatment group and graded as 0, no mononuclear cell infiltration; 1, very mild mononuclear cell infiltration in and around the islets; 2, mild cell infiltration in and around the islets; 3, moderate cell infiltration in and around the islets; 4, marked cell infiltration observed as the structure of islets was destroyed. Pancreas was graded for insulitis according to the most severely involved islets in each pancreas.
Necrosis of islets was graded as 0, no islet necrosis; 1, very mild islet necrosis; 2, mild islet necrosis; 3, moderate islet necrosis; 4, marked islets necrosis.
Atrophy of islets was graded as 0, no islet atrophy; 1, very mild islet atrophy; 2, mild islet atrophy; 3, moderate islet atrophy; 4, marked islet atrophy.
Pancreatic tissue was homogenized, supernatant and serum were initially deproteinized with Somogyi reagent[12] (using 2.0 mL 55 mmol/L NaOH followed by 2.0 mL 75 mmol/L ZnSO4). After centrifugation, aliquots of supernatant were mixed with an equal volume of Griess reagent[13]. The absorbance was measured at 546 nm. Nitrite level was measured using 120 &mgr;mol/L sodium nitrite solution diluted to 10 &mgr;mol/L intermediate dissolution. Results were expressed as &mgr;mol/L per gram wet tissue, and serum nitrite was expressed as &mgr;mol/L.
Pancreatic tissue was homogenized, supernatant and serum were initially deproteinized with Somogyi reagent[12] (using 2.0 mL 55 mmol/L NaOH followed by 2.0 mL 75 mmol/L ZnSO4). After centrifugation, excess nitrite was eliminated with 10 mg/mL ammonium sulfamate and acidified with 1 mol/L hydrochloric acid which prevented interference with hydroxide or carbonate. Aliquots of supernatant nitrate were determined by dual-wavelength ultraviolet spectrop-otometry[1415]. Nitrate absorbance was calculated by subtracting the absorbance at 275 nm for background correction from the absorbance at 220 nm. Nitrate level was meausred using 120 &mgr;mol/L standard sodium nitrate solution diluted to 10 &mgr;mol/L intermediate dissolution. Results were expressed as &mgr;mol/L per gram wet tissue, and serum nitrate was expressed as &mgr;mol/L.
The animals were killed by exsanguination from the carotid artery. Blood samples were taken immediately. The serum was stored at -18°C. Pancreas was removed, fragmented and stored at -20°C until required. Extracts from pancreatic tissue were prepared by homogenization in 0.05 mol/L sodium phosphate (pH 6.5) containing 5 mmol/L MgCl2 and 0.1% Triton X-100 (a DY89-1 tissue homogenizer) at 4°C. Tissue homogenate was centrifuged at 16 000 r/min for 30 min. Clear supernatant and serum were used to determine the enzyme activity. The level of α-mannosidase was measured by spectrophotometry as previously described[16] with some minor modifications. Fifty microliters of supernatant (or serum) was added to 50 &mgr;L 4 mmol/L p-nitrophenyl-α-D-mannopyranoside substrate (Fluka, Bucks, Switzerland) in 0.1 mol/L sodium acetate-acetic acid buffer (pH 5.8). Reaction mixtures were incubated at 37°C for 60 min. The reaction was terminated by adding 3 mL glycine-sodium hydroxide (0.1 mol/L, pH 10.14). The amount of liberated p-nitrophenol was determined by spectrophotometry at 405 nm. One international unit of enzyme was defined as the amount required for catalyzing the release of 1 &mgr;mol p-nitrophenol from p-nitrophenyl-α-D-mannopyranoside substrate per hour at 37°C under the conditions described above. Tissue enzyme activities were expressed as U/g wet tissue, and serum enzyme activities were expressed as U/mL serum.
Extracts from pancreatic tissue were prepared by homogenization in 9 g/L NaCl (a DY89-1 tissue homogenizer) at 4°C. Tissue homogenate was centrifuged at 16 000 r/min for 30 min. Clear supernatant and serum were used to determine the amylase activity and binding of lectin to amylase. Amylase N-glycan patterns were analyzed with lectin [ConA, pisum sativum agglutinin (PSA), peanut agglutinin (PNA), lens culinaris agglutinin (LCA)] affinity precipitation method. The precipitation procedure was performed as previously described[1718]. The clear supernatant and serum were mixed with 400 &mgr;L of an aqueous solution of ConA, PSA, PNA, LCA (5 g/L in distilled water). The mixture was incubated for 30 min at 37°C and centrifuged at 2000 r/min for 15 min. Without disturbing the precipitate, we removed the supernatant and measured its amylase activity. Binding of lectin to amylase was calculated by subtracting the corrected value from the total amylase activity. ConA, PSA, PNA, LCA were purchased from Sigma (St. Louis, MO, USA).
All data were presented as mean ± SD. Statistical significance was calculated using unpaired Student’s t test. The correlation coefficient between islet histomorphometry and nitrite, nitrate, binding of lectin to amylase, activity of α-mannosidase and amylase was calculated. P < 0.05 was considered statistically significant. All analyses were performed using Excel 2003.
The body weight of normal rats increased over the 12 wk experimental period. However, diabetic rats lost 22.1% of their body weight within 12 wk, the non-fasting blood glucose level increased from 16.62 mmol/L to 31.63 mmol/L 4 wk after injection of STZ, and remained significantly elevated for 8 wk thereafter.
Relatively well documented pancreatic islets and tightly arranged islet cells were observed in the normal control group. Severe islet necrosis and mild islet atrophy were detected in the diabetic group, and improved after treatment with Bu-Zhong-Yi-Qi-Tang compared to the diabetic group (Figure 1). Histomorphometrical analysis showed that the number and size of islets were significantly decreased, and the necrosis and atrophy of islets were significantly exacerbated in the diabetic group compared to the normal control group. However, the size of islets was significantly increased, and the necrosis and atrophy of islets were significantly improved in the Bu-Zhong-Yi-Qi-Tang treatment group compared to the diabetic group (Table 1).
The levels of nitrite and nitrate in serum and pancreas tissue of the diabetic group were significantly lower than those in the normal control group, and significantly increased in the Bu-Zhong-Yi-Qi-Tang treatment group compared to the diabetic group (Table 2).
| Treatment | Nitrite &mgr;mol/L serum | Nitrate &mgr;mol/L serum | Nitrite &mgr;mol/L per gram wet tissue | Nitrate &mgr;mol/L per gram wet tissue |
| Normal rats | 21.33 ± 0.25 | 98.78 ± 49.20 | 146.70 ± 24.77 | 13.29 ± 10.73 |
| Diabetic rats | 14.00 ± 0.19b | 85.76 ± 30.08 | 95.60 ± 28.89b | 11.59 ± 9.54 |
| Bu-Zhong-Yi-Qi-Tang-treated rats | 25.98 ± 0.74bd | 114.78 ± 30.09d | 132.82 ± 54.03d | 21.30 ± 18.73 |
The activity of α-mannosidase and amylase in pancreas tissue in the diabetic group was significantly lower than that in the normal control group. However, the serum α-mannosidase activity was not significantly different between the two groups. The activity of α-mannosidase and amylase in pancreas tissue was significantly increased in the Bu-Zhong-Yi-Qi-Tang treatment group compared to the diabetic group (Table 3).
| Treatment | Serum enzyme activities (U/mL serum) | Tissue enzyme activities (U/g wet tissue) | ||
| α-mannosidase | Amylase | α-mannosidase | Amylase | |
| Normal rats | 3.60 ± 2.43 | 28.71 ± 11.07 | 81.78 ± 30.34 | 19.53 ± 3.84 |
| Diabetic rats | 3.17 ± 7.07 | 17.35 ± 13.76a | 20.26 ± 20.42b | 13.15 ± 0.98a |
| Bu-Zhong-Yi-Qi-Tang-treated rats | 1.53 ± 1.72 | 25.14 ± 8.76c | 44.79 ± 26.93bd | 17.94 ± 2.20d |
Binding of LCA, and PNA to serum amylase and binding of LCA to pancreas amylase in the diabetic group were significantly higher than those in the normal control group, while the binding of ConA and PSA to amylase was not significantly different between the two groups. After treatment with Bu-Zhong-Yi-Qi-Tang, the binding of ConA, PNA and LCA to amylase was significantly lower than that in the diabetic group (Tables 4 and 5).
| Treatment | ConA-binding% (0-binding%) | LCA-binding% (0-binding%) | PNA-binding% (0-binding%) | PSA-binding% (0-binding%) |
| Normal rats | 22.18 ± 42.27 (71.43) | 2.61 ± 6.78 (85.71) | 3.89 ± 9.92 (85.71) | 6.52 ± 14.22 (78.57) |
| Diabetic rats | 31.18 ± 43.91 (58.33) | 27.84 ± 44.15a (58.33) | 32.86 ± 41.01a (50.0) | 25.0 ± 45.23 (75.0) |
| Bu-Zhong-Yi-Qi-Tang-treated rats | 1.20 ± 3.62ac (87.50) | 16.41 ± 34.06 (75.0) | 1.78 ± 5.0c (87.50) | 9.09 ± 16.44 (62.50) |
| Treatment | ConA-binding% (0-binding%) | LCA-binding% (0-binding%) | PSA-binding% (0-binding%) |
| Normal rats | 23.53 ± 27.83 (40.0) | 7.06 ± 15.78 (80.0) | 20.0 ± 44.72 (80.0) |
| Diabetic rats | 20.0 ± 35.46 (62.5) | 47.30 ± 40.53a (12.5) | 14.87 ± 27.68 (75.0) |
| Bu-Zhong-Yi-Qi-Tang-treated rats | 16.82 ± 31.18 (50.0) | 35.91 ± 37.08a (30.0) | 22.0 ± 41.46 (50.0) |
The number, size, necrosis and atrophy of islets were significantly correlated with the activity of nitrite, nitrate, α-mannosidase, amylase and binding of LCA, PNA, and PSA to amylase (Table 6).
| Biochemistry index | n | Islet number | Islet size | Necrosis | Atrophy |
| Serum nitrite | 50 | 0.404b | 0.736b | -0.319b | -0.866b |
| Serum nitrate | 50 | 0.181 | 0.194 | -0.126 | -0.245a |
| Pancreas tissue nitrite | 50 | 0. 30a | 0.456b | -0.222 | -0.480b |
| Pancreas tissue nitrate | 50 | 0.101 | 0.157 | -0.041 | -0.317b |
| LCA-binding of amylase in serum | 61 | -0.254a | -0.290a | 0.077 | 0.228a |
| ConA-binding of amylase in serum | 61 | -0.048 | -0.10 | -0.011 | -0.016 |
| PNA-binding of amylase in serum | 61 | -0.114 | -0.261a | 0.283a | 0.041 |
| PSA-binding of amylase in serum | 61 | -0.143 | -0.26a | 0.165 | 0.153 |
| LCA-binding of amylase in pancreas tissue | 43 | -0.442b | -0.253a | 0.174 | 0.183 |
| ConA-binding of amylase in pancreas tissue | 43 | -0.117 | -0.095 | -0.192 | 0.232 |
| PSA-binding of amylase in pancreas tissue | 43 | -0.095 | -0.037 | -0.288a | 0.106 |
| The activity of α-mannosidase in serum | 61 | 0.067 | 0.050 | 0.026 | -0.108 |
| The activity of α-mannosidase in pancreas tissue | 43 | 0.638b | 0.747b | -0.508b | -0.714b |
| The activity of amylase in serum | 61 | 0.158 | 0.365b | -0.215a | -0.310b |
| The activity of amylase in pancreas tissue | 43 | 0.428b | 0.639b | -0.314a | -0.631b |
In the present study, severe necrosis and mild atrophy of islets were observed in diabetic rats. The number and size of islets. The activity of α-mannosidase and amylase were decreased, and the binding of PNA and LCA to amylase were increased. All of which were improved after treatment with Bu-Zhong-Yi-Qi-Tang.
Islet damage was found to be related to the levels of nitrite, nitrate, α-mannosidase and amylase, and the binding of LCA, PNA and PSA to amylase.
Trimannosyl oligosaccharide is a unique moiety recognized by concanavalin A lectin (ConA) for high affinity and extended site binding. ConA primarily binds to the outer trimannosyl region of high mannose and bisected hybrid-type glycopeptides rather than to the central trimannosyl region of complex glycopeptides[19]. Fucosyl residues in the outer chain moieties have a shielding effect on the neighboring α-mannosyl residues and can eliminate this type of interaction[20]. PNA is a lectin with a high binding affinity for galactose-galactosamine disaccharide[21]. NeuAc caps the galactose-terminated chains[22]. Like ConA, LCA is able to bind (or not bind) to the same glycopeptides. LCA additionally requires the fucosyl- N-glycan-asparaginyl core for high-affinity binding. The presence of a core fucose residue greatly enhances recognition of N-linked sugar chains by LCA. Exposure of terminal N-acetylglucosamine (GlcNAc) residues to glycopeptides can enhance the binding of glycopeptide to LCA[23]. Like ConA, PSA is able to bind (or not bind) to the same glycopeptides. PSA additionally requires the fucosyl-N-glycan-asparaginyl core for high affinity binding. The presence of a core fucose residue greatly enhances recognition of N-linked sugar chains by PSA. Exposure of terminal mannose residues to glycopeptides can enhance the binding of glycopeptide to PSA[23].
These findings indicate that N-glycan processing is deficient in diabetic rats. Amylase core-fucosylate, high-mannose-type, hybrid-type sugar chains are increased while terminal sialic acid and fucose on the sugar chain are decreased, thus resulting in exposure of terminal galactose and GlcNAc residues to PNA and LCA.
N-glycan plays an important role in the quality control of glycoprotein folding in lumen and ERAD of proteins by cytosolic proteasomes[1]. Alpha-mannosidase, a key enzyme converting precursor high-mannose-type N-glycan to matured complex-type structure, contributes to the establishment of an equitable glycoprotein quality control standard, by which the efficiency of Asn-linked glycoprotein conformational maturation results in dislocation of misfolded glycoproteins into the cytosol where proteins are degraded in the proteasome and maintain the homeostasis of ER[2].
Lin et al[24] cultured neurons derived from embryonic chicken brains with tunicamycin (TM), an inhibitor of N-linked glycosylation, and found that the light neurons resembled necrotic cells, but the dense neurons exhibited distinct morphological features of necrosis and apoptosis, indicating that TM has an irreversible toxicity to the neurons and a different mechanism underlying neuron death.
Finnie et al[25] showed that cultured rat hepatocytes exposed to TM have degenerative changes characterized by marked cytoplasmic lipid accumulation and dilatation of cisternae of rough ER or necrosis.
Shi et al[2627] reported that inhibition of 6A8 alpha-mannosidase causes oncosis-like death of BJAB cells with no apoptotic bodies, annexin-V staining DNA fragmentation assay cannot show any evidence of apoptosis in these cells. However, binding of ConA to the cells transduced with the antisense 6A8 DNA is increased, but ConA-binding to the cells transduced with the sense 6A8 DNA is decreased.
The UPR is a conserved cellular response designed to alleviate damage and promote survival of cells experiencing stress by either shutting down translation to reduce the protein flow into the ER or by up-regulating molecules that protect cells or clear misfolded proteins from the ER and alleviate stress. NO, a known activator of UPR[3–5], may function to assist in the recovery of β-cells from NO-mediated damage, which is associated with enhanced expression of heat shock proteins and chaperones that assist in protein folding.
Islet β cells have a well-developed ER, reflecting their role in secreting a large amount of insulin and glycoproteins. Islet β cells may be at risk for ER stress. Inability to clear misfolded proteins causes accumulation of unfolded proteins, ER stress and activation of cellular signals, leading to cell death, loss of β cell secretion potential, and diabetes.
These findings indicate that STZ induces islet damage, which might be related to the N-glycan processing deficiency of proteins. Blocking of α-mannosidase activity and NO deficiency can easily cause accumulation of unfolded proteins, ER stress and activation of cellular signals leading to cell death.
Diabetes (Xiaoke) was firstly recorded in the Yellow Emperor’s Canon of Medicine (722-221 B.C.), a classic medical book, in which Xiaozhong (polyorexia) and Xiaodan (diabetes) are used to describe diabetes. It points out that the etiologic factors excess fat and sugar, and imbalance of emotion. The General Treatise on the Etiology and Symptomology (618-907 AD) say that diabetes patients often suffer from carbuncle and deep-root carbuncle. The Medical Secrets of an Official (670-755 AD) describes that urine of patients with diabetes is sweet, indicating that glucose can be found in the urine of diabetes patients early.
It has been shown that the pathogenesis of diabetes is closely related to Pixu[6], a major pathogenic factor for diabetes syndromes. Improving Pixu is therefore appropriate in the treatment of diabetes. In this study, the nitrite and nitrate levels were signifcantly decreased in diabetic rats, which is consistent with the reported fndings in patients with Pixu[8].
Bu-Zhong-Yi-Qi-Tang, a traditional Chinese medicine, tonifies qi, strengthens the stomach and spleen, and increases sunken Yang qi, and is prescribed mainly for the deficiency of spleen and stomach qi in the treatment of Pixu. It was reported that the levels of nitrite, nitrate, and peroxynitrite in gastric mucosa are lower in Pixu rats, and return to normal after treatment with Bu-Zhong-Yi-Qi-Tang[9].
In conclusion, Bu-Zhong-Yi-Qi-Tang can improve these abnormal conditions of diabetic rats by increasing the levels of nitric oxide and mannosidase activation, promoting N-glycosylation of proteins, assisting in the quality control of glycoprotein folding in ER lumen, and preventing accumulation of unfolded proteins, ER stress, activation of cellular signals.
N-glycans, α-mannosidase and nitric oxide (NO) play an important role in the quality control of glycoprotein folding in endoplasmic reticulum (ER) lumen, promote ER homeostasis and prevent ER-stress-related cell damage. There is evidence that ER stress plays a role in the pathogenesis of diabetes, contributing to loss of pancreatic beta-cells and insulin resistance. However, little attention has been paid to islet damage associated with the deficiency of N-glycan/nitric oxide in diabetes mellitus patients. It has been shown that the pathogenesis of diabetes is closely related to Pixu, a major pathogenic factor for diabetes syndromes. Improving Pixu is therefore appropriate in the treatment of diabetes. Bu-Zhong-Yi-Qi-Tang, a traditional Chinese decoction, is prescribed mainly for Pixu. However, little attention has been paid to its activity in islet damage and related mode of action in patients with diabetes.
N-glycans, α-mannosidase and NO play an important role in the quality control of glycoprotein folding in ER lumen, promote ER homeostasis and prevent ER-stress-related cell damage. There is evidence that ER stress plays a role in the pathogenesis of diabetes, contributing to loss of pancreatic beta-cells and insulin resistance. The results of this study indicate that streptozotocin induces islet damage, which might be related to the N-glycan processing deficiency of proteins. Blocking of α-mannosidase activity and nitric NO easily causes accumulation of unfolded proteins, ER stress and activation of cellular signals leading to cell death. Bu-Zhong-Yi-Qi-Tang could improve these abnormal conditions of diabetic rats by increasing the levels of NO and mannosidase activity, promoting N-glycosylation of proteins, assisting in quality control of glycoprotein folding in ER lumen, and preventing accumulation of unfolded proteins, ER stress, activation of cellular signals leading to cell death.
Bu-Zhong-Yi-Qi-Tang: a traditional Chinese medicine, consisting of Radix Astragali, Codonopsis pilosula, Glycyrrhiza uralensisf isch, Rhizoma Atractylodis Macrocephalae, Radix Angelicae Sinensis, Rhizoma Cimicifugae Foetidae, Radix Bupleuri Chinensis, Pericarpium Citri Reticulatae, is prescribed mainly for deficiency of spleen and stomach qi in treatment of Pixu. Streptozotocin (STZ): 2-deoxy-2-(3-methyl-3-nitrosourea)-1-D-glucopyranose, is actively transported into pancreatic β cells via the Glut-2 glucose transporter. Alpha-mannosidase: a key enzyme converting precursor high-mannose-type N-glycans to matured complex-type structure, contributes to the establishment of an equitable glycoprotein quality control standard, by which the efficiency of Asn-linked glycoprotein conformational maturation results in dislocation of misfolded glycoproteins into the cytosol, where proteins are degraded in proteasome and maintain the homeostasis of ER.
This is a good descriptive study. Experiments were well designed and the data contained novelty. The authors investigate the deficiency of N-glycan/nitric oxide in diabetic rats, and evaluated the effects of Bu-Zhong-Yi-Qi-Tang on diabetics, showing that STZ can induce islet damage, which might be related to the N-glycan processing deficiency of proteins. Blocking of α-mannosidase activity and nitric oxide deficiency could easily cause accumulation of unfolded proteins, ER stress and activation of cellular signals leading to cell death. Bu-Zhong-Yi-Qi-Tang was found to be effective against these abnormal conditions.
| 1. | Banerjee S, Vishwanath P, Cui J, Kelleher DJ, Gilmore R, Robbins PW, Samuelson J. The evolution of N-glycan-dependent endoplasmic reticulum quality control factors for glycoprotein folding and degradation. Proc Natl Acad Sci USA. 2007;104:11676-11681. [Cited in This Article: ] |
| 2. | Vallée F, Lipari F, Yip P, Sleno B, Herscovics A, Howell PL. Crystal structure of a class I alpha1,2-mannosidase involved in N-glycan processing and endoplasmic reticulum quality control. EMBO J. 2000;19:581-588. [Cited in This Article: ] |
| 3. | Cardozo AK, Ortis F, Storling J, Feng YM, Rasschaert J, Tonnesen M, Van Eylen F, Mandrup-Poulsen T, Herchuelz A, Eizirik DL. Cytokines downregulate the sarcoendoplasmic reticulum pump Ca2+ ATPase 2b and deplete endoplasmic reticulum Ca2+, leading to induction of endoplasmic reticulum stress in pancreatic beta-cells. Diabetes. 2005;54:452-461. [Cited in This Article: ] |
| 4. | Oyadomari S, Takeda K, Takiguchi M, Gotoh T, Matsumoto M, Wada I, Akira S, Araki E, Mori M. Nitric oxide-induced apoptosis in pancreatic beta cells is mediated by the endoplasmic reticulum stress pathway. Proc Natl Acad Sci USA. 2001;98:10845-10850. [Cited in This Article: ] |
| 5. | Chambers KT, Unverferth JA, Weber SM, Wek RC, Urano F, Corbett JA. The role of nitric oxide and the unfolded protein response in cytokine-induced beta-cell death. Diabetes. 2008;57:124-132. [Cited in This Article: ] |
| 6. | Xiong MQ, Li HL. Relation Between Diabetes and Spleen-deficiency. Guangzhou Zhongyiyao Daxue Xuebao. 1991;8:1-4. [Cited in This Article: ] |
| 7. | Lin L, Wei JP. Minutes of the 5th National Academic Symposia on Diabetes mellitus Medicine of Integrative Chinese and Western Medicine. Zhongguo Zhongxiyi Jiehe Zazhi. 2000;20:875. [Cited in This Article: ] |
| 8. | Liu XQ, Liang YY, Ja XL, Tang HQ, Chai WC, Wang JH. Preliminary study on the correlation of changes in salivary amylase activity and nitric oxide in diabetic patients with Pi-Xu. Zhongyao Yaoli Yu Linchuang. 2002;18:45-47. [Cited in This Article: ] |
| 9. | Xu Q, Liu XQ, Wang JH, Tang HQ, Wang RJ. Effect of Buzhong Yiqi Tang on the nitric oxide in gastric mucosa in rat with deficiency of the Pi. Zhongyao Yaoli Yu Linchuang. 2003;19:7-8. [Cited in This Article: ] |
| 10. | Inuwa IM, El Mardi AS. Correlation between volume fraction and volume-weighted mean volume, and between total number and total mass of islets in post-weaning and young Wistar rats. J Anat. 2005;206:185-192. [Cited in This Article: ] |
| 11. | Pei XH, Bai F, Tsutsui T, Kiyokawa H, Xiong Y. Genetic evidence for functional dependency of p18Ink4c on Cdk4. Mol Cell Biol. 2004;24:6653-6664. [Cited in This Article: ] |
| 12. | Somogyi M. A method for the preparation of blood filtrates for the determination of sugar. J Biol Chem. 1930;86:655-663. [Cited in This Article: ] |
| 13. | Cortas NK, Wakid NW. Determination of inorganic nitrate in serum and urine by a kinetic cadmium-reduction method. Clin Chem. 1990;36:1440-1443. [Cited in This Article: ] |
| 14. | Norman RJ, Edberg JC, Stucki JW. Determination of nitrate in soil extracts by dual-wavelength ultraviolet spectrophotometry. Soil Sci Soc Am J. 1985;49:1182-1185. [Cited in This Article: ] |
| 15. | Gheisari MM, Messripour M, Hoodaji M, Noroozi M, Abdollahi1 A. Nitrate Intake from Drinking Water in Isfahan in 2004. J Sci I R Iran. 2005;16:113-116. [Cited in This Article: ] |
| 16. | Kilian M, Bülow P. Rapid diagnosis of Enterobacteriaceae. I. Detection of bacterial glycosidases. Acta Pathol Microbiol Scand [B]. 1976;84B:245-251. [Cited in This Article: ] |
| 17. | Behr W, Barnert J. Quantification of bone alkaline phosphatase in serum by precipitation with wheat-germ lectin: a simplified method and its clinical plausibility. Clin Chem. 1986;32:1960-1966. [Cited in This Article: ] |
| 18. | Liu XQ, Zhu HL, Ye XW, Tang HQ. The aberrant sugar chains of amylase and different TCM syndrome patterns in primary hepatic cancer as well as the related mechanism. Zhong Liu. 2008;28:322-325. [Cited in This Article: ] |
| 19. | Brewer CF, Bhattacharyya L. Specificity of concanavalin A binding to asparagine-linked glycopeptides. A nuclear magnetic relaxation dispersion study. J Biol Chem. 1986;261:7306-7310. [Cited in This Article: ] |
| 20. | Yamashita K, Tachibana Y, Nakayama T, Kitamura M, Endo Y, Kobata A. Structural studies of the sugar chains of human parotid alpha-amylase. J Biol Chem. 1980;255:5635-5642. [Cited in This Article: ] |
| 21. | Chacko BK, Appukuttan PS. Peanut (Arachis hypogaea) lectin recognizes alpha-linked galactose, but not N-acetyl lactosamine in N-linked oligosaccharide terminals. Int J Biol Macromol. 2001;28:365-371. [Cited in This Article: ] |
| 22. | Chavan MM, Kawle PD, Mehta NG. Increased sialylation and defucosylation of plasma proteins are early events in the acute phase response. Glycobiology. 2005;15:838-848. [Cited in This Article: ] |
| 23. | Kornfeld K, Reitman ML, Kornfeld R. The carbohydrate-binding specificity of pea and lentil lectins. Fucose is an important determinant. J Biol Chem. 1981;256:6633-6640. [Cited in This Article: ] |
| 24. | Lin TY, Wang SM, Fu WM, Chen YH, Yin HS. Toxicity of tunicamycin to cultured brain neurons: ultrastructure of the degenerating neurons. J Cell Biochem. 1999;74:638-647. [Cited in This Article: ] |
| 25. | Finnie JW. Effect of tunicamycin on hepatocytes in vitro. J Comp Pathol. 2001;125:318-321. [Cited in This Article: ] |
| 26. | Shi GX, Liu Y, Zhao FT, Zhu LP. BJAB cells undergo an oncosis-like cell death after transduction with an antisense DNA to human 6A8α-mannosidase gene. Zhonghua Weishengwu He Mianyixue Zazhi. 2001;21:480-485. [Cited in This Article: ] |
| 27. | Shi GX, Liu Y, Li L, Zhu LP. Inhibition of 6A8 alpha-mannosidase causes oncosis-like death of BJAB cells. Cell Mol Biol (Noisy-le-grand). 2002;48 Online Pub:OL369-OL377. [Cited in This Article: ] |