Published online Aug 28, 2021. doi: 10.37126/aige.v2.i4.168
Peer-review started: May 25, 2021
First decision: June 18, 2021
Revised: June 20, 2021
Accepted: July 6, 2021
Article in press: July 6, 2021
Published online: August 28, 2021
Processing time: 104 Days and 6.3 Hours
Pancreatic malignancy still becomes a major global problem and is considered as one of the most lethal cancers in the field of gastroenterology. Most patients come in the late stage of the disease due to organ’s location, and until now the treatment result is still far away from satisfaction. Early detection is still the main key for good, prolonged survival. However, discerning from other types of tumor sometimes is not easy. Endoscopic ultrasound (EUS) is still the best tool for pancreatic assessment, whereas fine-needle aspiration biopsy (FNAB) is considered as the cornerstone for further management of pancreatic malignancy. Several conditions have become a concern for EUS-FNAB procedure, such as risk of bleeding, pancreatitis, and even needle track-seeding. Recently, an artificial intelligence innovation, such as EUS elastography has been developed to improve diagnostic accuracy in pancreatic lesions evaluation. Studies have shown the promising results of EUS elastography in improving diagnostic accuracy, as well as discerning from other tumor types. However, more studies are still needed with further considerations, such as adequate operator training, expertise, availability, and its cost-effectiveness in comparison to other imaging options.
Core Tip: The application of endoscopic ultrasound (EUS) elastography is one of the most potential roles of artificial intelligence in pancreaticobiliary disorders. EUS elastography becomes a promising method to evaluate pancreatic lesions by providing information of tissue elasticity, which may correlate with malignant characteristics. Incomplete elastographic delineation, especially in large tumor size, as well as compelling intra-/inter-observer variability also still become limitations in performing adequate EUS elastography examination on pancreatic lesions.
- Citation: Lesmana CRA, Paramitha MS. Impact of endoscopic ultrasound elastography in pancreatic lesion evaluation. Artif Intell Gastrointest Endosc 2021; 2(4): 168-178
- URL: https://www.wjgnet.com/2689-7164/full/v2/i4/168.htm
- DOI: https://dx.doi.org/10.37126/aige.v2.i4.168
Pancreatic malignancy is still considered as the most lethal cancer in the field of gastroenterology. Based on Global Cancer Observatory database 2020, it is still holding the 12th rank of the most common malignancies all over the world. The mortality rate related to pancreatic cancer has increased more than double within 27 years. The survival rate has also been considered far from satisfaction with regards to the standard treatment development. In Asian population, the incidence and mortality related to pancreatic cancer are also quite high (47.1% and 48.1%, respectively)[1]. Most of the patients are diagnosed at the late stage due to organ’s location, non-specific clinical manifestation in early stages, and the absence of simple screening test with high accuracy for early stages of the disease.
In the evaluation of pancreatic cancer, imaging has been proven to play a central and critical role. Imaging modalities are expected to be able to detect and characterize the tumor mass, evaluate local and vascular involvement, evaluate lymphatic and perineural invasion, and find any metastases. Evolution of diagnostic imaging examination such as abdominal computed tomography (CT) scan and magnetic resonance imaging (MRI) have shown good accuracy for detecting pancreatic lesion. A single-center retrospective study in 140 subjects showed higher sensitivity (89.5% vs 81.4%) and specificity (63.4% vs 43%) in MRI compared to CT-scan for evaluating pancreatic adenocarcinoma. This study also showed that only 14% of the patients were diagnosed in the early stage at the time of diagnosis. Nevertheless, in the setting of small size of tumor mass, uncooperative patients for MRI evaluation, availability of MRI, lack of clinicians’ familiarity with the device, and high cost of performing MRI still become the limitations in clinical practice. Additionally, from the same study, the highest diagnostic accuracy was shown by endoscopic ultrasound (EUS) (sensitivity 97.5%, specificity 90.3%). In the new era of the old instrument development, EUS has become the cornerstone in pancreatic malignancy, as it has a high sensitivity for small tumor size (< 2 cm), evaluation of staging (including the presence of lymph nodes, ascites, liver metastasis, and vascular involvement), and to perform direct tissue sampling[2,3]. However, in the conditions of uncertain malignant condition, normal tumor markers level, and possibility of needle tract seeding, a dilemmatic condition on whether the lesion should be punctured or not may arise[3-5]. Learning from the non-invasive tool development, such as elasticity evaluation, has opened a better insight for utilizing EUS, not only for diagnostic purpose, but also for therapeutic purpose.
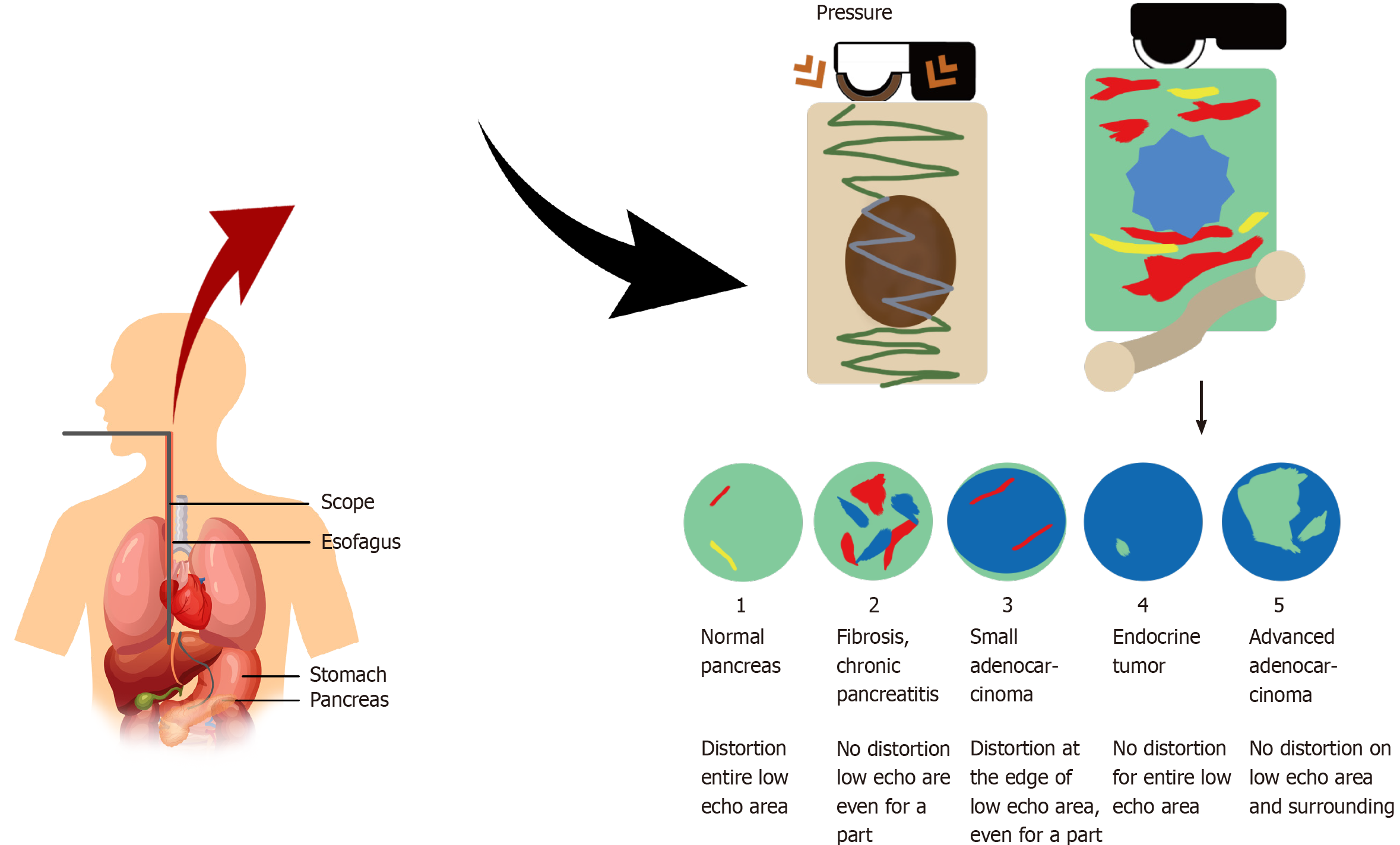
The concept of utilizing combination of elastography (EG) and ultrasonography in diagnosing pancreatic disorders has been proposed as a way to overcome the diagnostic problem of solid pancreatic lesions (Figure 1). A prospective study conducted by Uchida et al[6] showed that real-time tissue EG and transcutaneous ultrasonography can provide real-time visualization and information of pancreatic tissue elasticity. Combination of sonic and ultrasound waves will cause less compression in fibrotic and stiff tissue, in comparison to softer and healthy tissue. This characteristic may overcome the limitation of conventional EUS, especially in patients with coexistent chronic pancreatitis or “pseudotumoral” pancreatitis[7]. As one of the most recent approaches in gastrointestinal endoscopy, EUS real-time tissue EG has more diagnostic potentials compared to EUS with only a B-mode imaging ability. In general, EUS EG provides information of tissue elasticity through differences in deformation and displacement among tissue areas, as well as different amount of tissue distortion attained from spatial differentiation. Tissue consistency may correlate with malignancy characteristics, in which malignant tissues have harder consistency than benign tissues[8].
Reported for the first time in 2006 for evaluating pancreatic tissues, EUS EG has been continuously developed for tissue elasticity assessment. Two methods have been differently proposed and compared for each diagnostic performance, i.e., strain and shear-wave EG. Generally, strain elastograms are produced by internal physiological pulsations from respiratory contractions. Estimation of the target tissue’s stiffness is conducted with semiquantitative real-time elastography (RTE) using strain histogram (SH), and quantitative strain ration (SR) histogram EG. In particular, SR is a semi-quantitative method to calculate relative tissue stiffness by dividing mean strain of reference area and mean strain in lesion of interest. Meanwhile, the global hardness of a lesion is expressed by the mean histogram value (numerical values from SH)[3,9]. There are three major important principles when RTE is applied for tissue elasticity evaluation, i.e., the stress compression, the region of interest (ROI), and the tissue displacement. Semi-quantitative SH EUS EG uses the manual method through tissue compression effect or pressure application, which will create color-based results. Quantitative strain elastograms or SH needs to calculate the ratio; however, this can be a combined assessment. This software methods usually will be incorporated to the echoendoscope for pancreatic tissue assessment[3,8]. In a healthy pancreatic tissue, the internal structure is isoechoic with soft elastogram. In elderly, the consistency of pancreatic tissue is remarkably harder, but not as hardened as the histogram result of chronic pancreatitis. In acute pancreatitis, softer consistency can be observed in the necrotic zones. Significantly higher stiffness (often unequivocal) can be found in ductal adenocarcinoma. The hue color-based parameter, where it is used for tissue elasticity evaluation, consists of red, green, and blue color. Soft tissue appears as red color, whereas intermediate tissue appears as green color, and blue color will represent hard tissue. However, perception errors or variability of interpretation between endosonographers may occur in the characterization of hue color-based parameter[8,9].
On the other hand, shear-wave EG is a quantitative tissue elasticity assessment, where it has been mostly used for liver, breast, prostate, rectum, and lymph node. In shear wave EG, focused ultrasound from the probe to target tissue is emitted and evaluation of target tissue’s stiffness is performed afterwards by measuring the shear wave’s propagation speed. An exploratory study of EUS shear-wave measurement (EUS-SWM) in the assessment and treatment of autoimmune pancreatitis showed approximately 97.6% success rate with no significant difference of success rate in the head, body, and tail of the pancreas (P = 0.4997)[10]. Another preliminary study also demonstrated similarly high success rate (96.8%) without any adverse events. In addition, the elastic value with unique reliability index of the velocity of shear wave measurement also allows more objective and repeated measurement with EUS-SWM[11]. However, compared to strain EG, varying results with EUS-SWM are still found from previous study by Carlsen et al[12] This study also showed that target diameter had the most significant effect for all methods of shear-wave EG measurement, while target depth only affected shear-wave velocity measurement in targets with hard consistency.
Throughout the years, evidences related to the use of EUS EG in pancreatic lesion evaluation keep emerging (Table 1). A pioneer study by Giovannini et al[13], 2006 showed the impact of endosonoelastography examination for pancreatic masses evaluation in 49 patients, where the sensitivity and specificity in diagnosing malignant lesion were 100% and 67%, respectively. In this study, there were two misdiagnosed cases (neuroendocrine tumor and benign fibromyoblastic tumor of surgically resected pancreas). The sensitivity and specificity of endosonoelastography in assessing malignant lymph node invasion in this study were 100% and 50%, respectively. As mentioned in the previous section, the first experimental study for real-time tissue EG for pancreatic tissue assessment was investigated by Uchida et al[6], 2009, in which a linear probe, with B-mode and EG mode, was used to visualize the object. The color-based (blue for hard and red for soft) was used in the ROI. In pancreatic cancer, the lesion was identified with blue color, which was subsequently confirmed through histopathologic examination result. Combination of B-mode and EG mode increased the diagnosis accuracy of pancreatic cancer from 73.3% to 100%, corrected by operator. The sensitivity and specificity between operator and another reviewer showed the same results for EG mode evaluation (64.3% vs 60.7% and 88% vs 88%). In the case of pancreatic endocrine tumor, the diagnosis accuracy also increased from 66.7% to 100%[6]. In 2009, a prospective study by Iglesias-Garcia et al[14], where the EG pattern was compared to histological specimen, showed the blue color pattern supported the malignant pancreatic lesions, whereas the green color pattern excluded malignant lesions. The sensitivity and specificity of EG diagnosis in malignant pancreatic lesions were 100% and 85.5%, respectively. This study concluded that the overall diagnostic accuracy of EUS EG for malignancy was 94%. Further concordance analysis by two endosonographers yielded agreement of elastographic pattern by both of them in 93.1% of the cases. This study also addressed the possibility of EUS EG in tackling the limitation of EUS-guided fine needle aspiration (EUS-FNA). One of the major drawbacks of EUS-FNA was interposition of malignant tissue and vascular structures, which may contribute to false negative results. EUS EG can overcome this limitation by assessing tissue elasticity and discerning hardness between normal and malignant tissues[14].
| Ref. | Population of the study | Key findings |
| Giovannini et al[13], 2006 | 24 patients with pancreatic masses. | Sensitivity 100% and specificity 67% in diagnosing malignant lesions. |
| Sensitivity 100% and specificity 50% in diagnosing malignant invasion of lymph nodes. | ||
| Uchida et al[6], 2009 | Phase 1: pancreatic cancer (5 subjects), endocrine tumor (2 subjects), chronic pancreatitis (5 subjects), intraductal papillary mucinous neoplasm. | Diagnostic performance of real-time tissue elastography mode for diagnosing malignancy: Operator vs another reviewer |
| Sensitivity: 64.3% vs 60.7%. | ||
| Specificity: 88% vs 88%. | ||
| Positive predictive value: 85.7% vs 85%. | ||
| Phase 2: 53 consecutive subjects with pancreatic lesions visible on B-mode images. | Negative predictive value: 68.8% vs 66.7%. | |
| Iglesias-Garcia et al[14], 2009 | 130 consecutive patients with solid pancreatic masses vs 20 subjects with normal pancreases. | Diagnostic performance of EUS elastography in diagnosing malignancy |
| Sensitivity: 100%. | ||
| Specificity: 85.5%. | ||
| Positive predictive value: 90.7%. | ||
| Negative predictive value: 100%. | ||
| Overall accuracy: 94%. | ||
| Hirche et al[4], 2008 | 70 patients with unclassified solid pancreatic lesions vs 10 subjects with healthy pancreas. | Diagnostic performance of EUS elastography in predicting the nature of pancreatic lesions |
| Sensitivity: 41%. | ||
| Specificity: 53%. | ||
| Accuracy: 45%. | ||
| Janssen et al[15], 2007 | 20 patients with chronic pancreatitis vs 33 patients with focal pancreatic lesions vs 20 subjects with normal pancreas. | Diagnostic performance of EUS elastography in diagnosing chronic pancreatitis |
| Sensitivity: 65.9%. | ||
| Specificity: 56.9%. | ||
| Accuracy: 60.2%. | ||
| Diagnostic performance of EUS elastography in diagnosing focal pancreatic lesions | ||
| Sensitivity: 93.8%. | ||
| Specificity: 65.4%. | ||
| Accuracy: 73.5%. | ||
| Diagnostic performance of EUS elastography in differentiating pancreatic adenocarcinoma and inflammatory pancreatic masses | ||
| Li et al[16], 2013 | Meta-analysis of 10 studies with 781 patients. | Diagnostic performance of EUS elastography in differentiating pancreatic adenocarcinoma and inflammatory pancreatic masses |
| AUC: 0.8227. | ||
| In studies with color pattern as the diagnostic standard | ||
| Sensitivity: 99%. | ||
| Specificity: 76%. | ||
| Positive likelihood ratio: 3.36. | ||
| Negative likelihood ratio: 0.03. | ||
| Diagnostic odds ratio: 129.96. | ||
| In studies with hue histogram as the diagnostic standard | ||
| Sensitivity: 92%. | ||
| Specificity: 68%. | ||
| Positive likelihood ratio: 2.84. | ||
| Negative likelihood ratio: 0.12. | ||
| Diagnostic odds ratio: 24.69. | ||
| Xu et al[17], 2013 | Meta-analysis of 9 studies. | Diagnostic performance of EUS elastography in differentiating benign and malignant pancreatic masses |
| In studies with qualitative color pattern as the diagnostic standard | ||
| Sensitivity: 99%. | ||
| Specificity: 74%. | ||
| AUROC: 0.9624. | ||
| In studies with quantitative hue histogram value as the diagnostic standard | ||
| Sensitivity: 85%-93%. | ||
| Specificity: 64%-76%. | ||
| Mei et al[18], 2013 | Meta-analysis of 13 studies with 1044 patients. | Diagnostic performance of EUS elastography in differentiating benign and malignant solid pancreatic masses |
| Sensitivity: 95%. | ||
| Specificity: 67%. | ||
| Diagnostic odds ratio: 42.28. | ||
| Altonbary et al[19], 2019 | 97 patients with malignant lesions vs 19 patients with benign lesions | Diagnostic performance of combined elasticity score and strain ratio in differentiating benign and malignant pancreatic lesions (cut-off point: 7.75) |
| Sensitivity: 99%. | ||
| Specificity: 94.6%. | ||
| Positive predictive value: 98%. | ||
| Negative predictive value: 98.5%. | ||
| Accuracy: 97%. | ||
| Ignee et al[20], 2018 | 218 patients with solid pancreatic lesions sized ≤ 15 mm and a definite diagnosis. | Diagnostic performance of EUS elastography with high stiffness of the lesion in diagnosing malignancy |
| Sensitivity: 84%. | ||
| Specificity: 67%. | ||
| Positive predictive value: 56%. | ||
| Negative predictive value: 89%. | ||
| Diagnostic performance of EUS elastography in diagnosing pancreatic ductal adenocarcinoma | ||
| Sensitivity: 96%. | ||
| Specificity: 64%. | ||
| Positive predictive value: 45%. | ||
| Negative predictive value: 98%. |
In contrast to previous evidences, a prospective study by Hirche et al[4] showed that EUS-EG had low sensitivity (41%), specificity (53%), and accuracy (45%) in predicting malignant pancreatic lesion. A subgroup analysis in ductal adenocarcinoma also demonstrated poor sensitivity (50%). Moderate intraobserver and interobserver reproducibilities were also demonstrated from the findings. However, in this study, the sample size was considered small. Additionally, some patients were diagnosed with cystic lesion tumor, suggesting that presence of fluid might interfere the elastographic pattern. On the other hand, larger tumor size was causing the inaccurate distance between the EUS probe and the mucosal wall. Incomplete border delineation by EUS- EG was also shown in lesions with a larger diameter, leading to insufficient display of surrounding pancreatic parenchyma[4]. In another small prospective single-center study by Janssen et al[15], three groups were classified as normal pancreas, chronic pancreatitis, and focal pancreatic lesions. The elastographic pattern classification (homogenous, different colors, and honeycomb pattern) and elastographic colors classification (blue, green/yellow, and red) were combinedly used. In normal pancreas group, all showed homogenous green color interfered with blue clouds’ color. Whereas, in chronic pancreatitis group showed hard (blue) with honeycomb pattern. In pancreatic focal lesions’ group, examination showed that almost all patients had blue/green honeycomb pattern. Only one patient which has tumorlike due to chronic pancreatitis showed blue/green honeycomb pattern. The sensitivity and specificity for group with chronic pancreatitis were 65.9% and 56.9%, respectively, with diagnostic accuracy of 60.2%; while the sensitivity and specificity in group with focal pancreatic lesions were 93.8% and 65.4%, respectively, with slightly higher diagnostic accuracy (73.5%). The findings from this study also addressed the limitation of EUS EG in distinguishing the elastographic patterns of chronic pancreatitis and malignant tumors due to the corresponding amount of fibrous pattern of chronic pancreatitis, which can also be found in desmoplastic pancreatic carcinomas and microcystic adenomas[15]. Another meta-analysis, which evaluated the use of EUS EG in discernment of pancreatic adenocarcinoma and inflammatory masses, indicated slightly better diagnostic performance in studies with color pattern as the diagnostic standard (sensitivity 99%, specificity 76%) compared to studies with hue histogram as the diagnostic standard (sensitivity 92%, specificity 68%)[16]. In differentiating benign and malignant pancreatic masses, better diagnostic performance was also demonstrated by studies using qualitative color pattern as the diagnostic standard (sensitivity 99%, specificity 74%) in comparison to studies using hue histogram as the diagnostic standard (sensitivity 85%-93%, specificity 64%-76%). This meta-analysis also acknowledged the difficulties in distinguishing neuroendocrine tumors and adenocarcinomas due to their similar hardness[17]. Regardless of the low specificity, EUS EG can still be considered as a complementary diagnostic method. A meta-analysis by Mei et al[18] showed high pooled sensitivity (95%) with acceptable pooled specificity (67%) and moderate accuracy (summary Receiver Operating Characteristic: 90.46%) of EUS EG in diagnosing solid pancreatic masses. Improvement of diagnostic accuracy may be achieved with application of more meticulous computer-aided diagnosis method for EUS-EG[18]. Recent findings from a single center retrospective study by Altonbary et al[19] also demonstrated promising results of EUS EG with combination of elasticity score and strain ratio in discerning solid pancreatic lesions (sensitivity 99%, specificity 94.6%, and accuracy 97%). Moderately well diagnostic performance in ruling out malignancy was also demonstrated by a multicenter study conducted in 218 patients with small (< 15 mm) solid pancreatic lesions (sensitivity 84%). Higher sensitivity (96%) was shown when EUS EG was used in diagnosing Pancreatic Ductal Adenocarcinoma (PDAC)[20].
Several conditions have been considered as clinical dilemma, such as small pancreatic lesion which also can be found incidentally, pseudo-tumoral in chronic pancreatitis, negative FNA biopsy (FNAB) results, and possibility of needle tract tumor seeding[3-5]. It has been known that pancreatic cancer is mostly dominated by PDAC, a highly aggressive tumor with very poor prognosis and high mortality rate. It has been reported that Negative Predictive Value (NPV) of FNAB result can vary, ranging from 16% to 85%. In the case of negative biopsy, patients with suspicion of PDAC should be referred immediately for surgical approach consideration. Spier et al[21] published a small retrospective EUS-FNA study in patients who had suspected pancreatic lesions with negative biopsy results. The study found that 30.9% of patients with negative/ non-diagnostic FNA results were later diagnosed with pancreatic cancer (mean time 66 d to 360 d after FNA procedure)[21]. RTE has been proposed as a supplementary method to improve diagnostic performance of EUS-FNA, especially in terms of available rapid on-site tissue evaluation by a cytopathologist[22,23]. A retrospective study in 54 subjects with solid pancreatic lesions highlighted the benefit of combining RTE and EUS-FNA (sensitivity 94.4%, specificity 93.4%, and accuracy 100%) compared to the diagnostic performance of RTE alone (sensitivity 86.9%, specificity 75%, and accuracy 85.1%)[22].
Possibility of tumor seeding has become a challenging issue as it will impact on faster disease progression, patient’s clinical-based management, and patient’s survival after surgery or non-surgical biliary drainage procedure in patients with bile duct obstruction. There has been a debate on whether this tract seeding issue should be underestimated or overestimated, since most of the studies use retrospective study design. Small sample size and no clear tumor dissemination finding also become issues on the studies of needle tract seeding related to EUS-FNA[5]. The first reported case of EUS-FNA-related tumor dissemination was delivered in 2003, in which peritoneal dissemination occurred in intraductal papillary mucinous tumor (T1N0M0)[5]. Approximately 80% of all needle tract seeding cases following EUS-FNA happened in pancreatic cancer and pancreatic cystic tumors located in the body or tail of pancreas. In most of the cases, 22-G FNA needle was used, even though the relationship between needle size or number of needle passes and the risk of tumor seeding is still unclear. The range of interval from EUS-FNA procedure to diagnosis of needle tract seeding is 3-48 mo[24].
EG EUS multicenter study by Ignee et al[20] in small solid pancreatic lesions showed that sensitivity and specificity were 84% and 67%, respectively, with 56% of positive predictive value and 89% of NPV. In PDAC cases, sensitivity and specificity were 96% and 64%, respectively. Based on this study, it is clear that early detection in less than 15 mm pancreatic lesion might prevent the delay for surgery management even though PDAC tends to be found in larger lesions (> 15 mm)[20]. Another prospective study was conducted by Dawwas et al[25] in patients underwent quantitative EUS EG procedure for differentiating pancreatic malignant lesion with pancreatic inflammatory lesion. The examination results were compared to histology or cytology results with follow-up imaging study. The sensitivity and specificity with quantitative EUS EG were 100% and 95.7%, respectively. This study has shown the important value of EUS EG in reducing the need of biopsy as the EUS-FNAB procedure still carries potentially harmful risks, such as pancreatitis and bleeding[25]. In 2018, Dong et al[26] reported the role of combination strategy using B-mode ultrasound, contrast-enhanced ultrasound (CEUS), and EUS EG in small case series of isolated pancreatic tuberculosis (PTB) cases. These findings were then compared with the clinical findings of PDAC cases. In PTB cases, common bile duct and pancreatic duct dilatation are considered to be rare findings, however, it is common to find multiple peripancreatic lymph nodes enlargement. The PTB lesion was showing less demarcation, whereas clear demarcation was found in PDAC cases. It might be difficult to differentiate PTB from PDAC cases by using the tissue stiffness result from EUS elastrography alone, however, with CEUS combination, PTB lesion showed hyperenhancement, whereas in PDAC cases showed hypoenhancement. In addition, peripancreatic pseudocysts were more commonly observed in PTB cases. This non-invasive strategy can be an accurate diagnosis tool with or without biopsy as a clinical-based approach in patients with PTB. Consequently, it can also avoid unnecessary surgical management[26].
A former retrospective analysis study by Iordache et al[27] in 50 consecutive patients with negative results of EUS-FNA who sequentially underwent EUS EG and CE-EUS, found that EUS EG has similar results with CE-EUS in diagnosing possibility of pancreatic malignancy. However, combination of both methods showed excellent specificity (100%). Another interesting finding from this study is the excellent specificity (100%) exhibited by CEH-EUS in patients with soft/mixed or hard (low strain) appearance from EG. Excellent specificity was shown by CEH-EUS for distinguishing chronic pancreatitis in soft/mixed (high strain) appearance; while in hard appearance, CEH-EUS exhibited outstanding specificity (100%) and sensitivity (88.89%) for distinguishing pancreatic cancer. These results suggested that hard hypovascular masses can indicate the presence of pancreatic adenocarcinoma or other malignant masses, whereas soft hyper-/isovascular masses can indicate the presence of chronic pseudotumoral pancreatitis or other benign masses[27]. Another pro
According to most studies on EUS EG, it has been shown that EUS EG has a big role in managing pancreatic lesions. This method can be a primary choice for diagnosis evaluation in patients who have coagulation disorders or history of anticoagulation drugs consumption, who are not suitable yet for chemotherapy, and who have the possibility for direct surgical approach due to the needle tract seeding risk during FNA procedures. In targeting unclear demarcation and pancreatic lesion image, EUS EG can also be an additional tool. However, it cannot be used for pancreatic cystic mass tumor evaluation. Studies to differentiate between malignant and benign pancreatic mass lesion have not shown any strong evidence yet as some studies were only performed with small sample size, and some only used retrospective study analysis.
The main objectives of performing EG for the pancreas are to ensure that the elastogram is sufficiently meticulous to represent the histological structures and to be reproducible adequately. These objectives, however, are hampered by the small size of the pancreas, the depth of its anatomical location in the center of the body, the technical difficulties in extracting biopsy specimens, and the strong influence of aortic pulsation to pancreas. In addition, EG is an operator-independent modality[30]. Other pitfalls of EUS EG are the difficulty in controlling tissue compression by the EUS transducer, the presence of motion artifacts due to respiratory movement, as well as the careful selection of ROI from its surrounding soft tissues[31].
Overall, the application of EUS EG is one of the most potential roles of artificial intelligence (AI) in pancreaticobiliary disorders. In general, AI refers to the capacity of a computer to imitate the cognitive intelligence or the learning capability of human being in order to perform tasks appropriately. In medicine, AI consists of machine learning and deep learning, which are often utilized reciprocally[32]. A cross-sectional feasibility study in Denmark established the importance of AI in distinguishing pancreatic cancer from chronic pancreatitis through the application of neural network analysis of dynamic sequences of EUS EG. In this study, the sensitivity, specificity, and accuracy were 91.4%, 87.9%, and 89.7%, respectively. In addition, the application of multilayer perceptron neural networks with high training performance was able to reach an accuracy as high as 97%[33]. Another prospective and multicenter study in 258 patients by Săftoiu et al[34] also highlighted the efficacy of AI in EUS EG. The utilization of multilayer perceptron as an artificial neural network demonstrated moderately high diagnostic performance (sensitivity 87.59%, specificity 82.94%, AUROC 0.94, training accuracy 91.14%, and testing accuracy 84.27%) in diagnosing focal pancreatic lesions.
EUS EG is a promising method to improve the diagnostic accuracy as well as helping to decide which type of management is probably more suitable for patients with pancreatic mass lesion. However, it would still need more studies with further considerations, such as adequate operator training, expertise, availability, and its cost-effectiveness in comparison to other imaging options. Integrating clinical data into artificial intelligence techniques concomitantly with real-time imaging results is potentially favorable for faster and more accurate clinical-decision making in pancreatic lesion evaluation.
I would like to thank to Professor Ho Khek Yu, Past President of Asian EUS Group (AEG), and Professor Laurentius A Lesmana, Chair of Digestive Disease & GI Oncology Center, Medistra Hospital, Jakarta who had given big support and contributions in the development of endoscopic ultrasound (EUS) in Indonesia.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Indonesia
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Altonbary AY S-Editor: Liu M L-Editor: A P-Editor: Wang LYT
| 1. | World Health Organization International Agency for Research on Cancer (IARC). GLOBOCAN 2020: Pancreas. [homepage on the internet]; 2020 [cited 2021 May 22]. Available from: https://gco.iarc.fr/today/data/factsheets/cancers/13-Pancreas-fact-sheet.pdf. |
| 2. | Costache MI, Costache CA, Dumitrescu CI, Tica AA, Popescu M, Baluta EA, Anghel AC, Saftoiu A, Dumitrescu D. Which is the Best Imaging Method in Pancreatic Adenocarcinoma Diagnosis and Staging - CT, MRI or EUS? Curr Health Sci J. 2017;43:132-136. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 24] [Reference Citation Analysis (0)] |
| 3. | Kitano M, Yoshida T, Itonaga M, Tamura T, Hatamaru K, Yamashita Y. Impact of endoscopic ultrasonography on diagnosis of pancreatic cancer. J Gastroenterol. 2019;54:19-32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 170] [Cited by in RCA: 217] [Article Influence: 36.2] [Reference Citation Analysis (0)] |
| 4. | Hirche TO, Ignee A, Barreiros AP, Schreiber-Dietrich D, Jungblut S, Ott M, Hirche H, Dietrich CF. Indications and limitations of endoscopic ultrasound elastography for evaluation of focal pancreatic lesions. Endoscopy. 2008;40:910-917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 100] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 5. | Minaga K, Takenaka M, Katanuma A, Kitano M, Yamashita Y, Kamata K, Yamao K, Watanabe T, Maguchi H, Kudo M. Needle Tract Seeding: An Overlooked Rare Complication of Endoscopic Ultrasound-Guided Fine-Needle Aspiration. Oncology. 2017;93 Suppl 1:107-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 6. | Uchida H, Hirooka Y, Itoh A, Kawashima H, Hara K, Nonogaki K, Kasugai T, Ohno E, Ohmiya N, Niwa Y, Katano Y, Ishigami M, Goto H. Feasibility of tissue elastography using transcutaneous ultrasonography for the diagnosis of pancreatic diseases. Pancreas. 2009;38:17-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 44] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 7. | Gill KR, Wallace MB. EUS elastography for pancreatic mass lesions: between image and FNA? Gastrointest Endosc. 2008;68:1095-1097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 8. | Dietrich CF, Bibby E, Jenssen C, Saftoiu A, Iglesias-Garcia J, Havre RF. EUS elastography: How to do it? Endosc Ultrasound. 2018;7:20-28. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 9. | Costache MI, Dumitrescu D, Săftoiu A. Technique of qualitative and semiquantitative EUS elastography in pancreatic examination. Endosc Ultrasound. 2017;6:S111-S114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 10. | Ohno E, Hirooka Y, Kawashima H, Ishikawa T, Tanaka H, Sakai D, Ishizu Y, Kuzuya T, Nakamura M, Honda T. Feasibility and usefulness of endoscopic ultrasonography-guided shear-wave measurement for assessment of autoimmune pancreatitis activity: a prospective exploratory study. J Med Ultrason (2001). 2019;46:425-433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 28] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 11. | Ohno E, Hirooka Y, Kawashima H, Ishikawa T. Feasibility of EUS-guided shear-wave measurement: A preliminary clinical study. Endosc Ultrasound. 2019;8:215-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 12. | Carlsen JF, Pedersen MR, Ewertsen C, Săftoiu A, Lönn L, Rafaelsen SR, Nielsen MB. A comparative study of strain and shear-wave elastography in an elasticity phantom. AJR Am J Roentgenol. 2015;204:W236-W242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 51] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 13. | Giovannini M, Hookey LC, Bories E, Pesenti C, Monges G, Delpero JR. Endoscopic ultrasound elastography: the first step towards virtual biopsy? Endoscopy. 2006;38:344-348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 215] [Cited by in RCA: 214] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 14. | Iglesias-Garcia J, Larino-Noia J, Abdulkader I, Forteza J, Dominguez-Munoz JE. EUS elastography for the characterization of solid pancreatic masses. Gastrointest Endosc. 2009;70:1101-1108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 124] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 15. | Janssen J, Schlörer E, Greiner L. EUS elastography of the pancreas: feasibility and pattern description of the normal pancreas, chronic pancreatitis, and focal pancreatic lesions. Gastrointest Endosc. 2007;65:971-978. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 198] [Cited by in RCA: 178] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 16. | Li X, Xu W, Shi J, Lin Y, Zeng X. Endoscopic ultrasound elastography for differentiating between pancreatic adenocarcinoma and inflammatory masses: a meta-analysis. World J Gastroenterol. 2013;19:6284-6291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 47] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 17. | Xu W, Shi J, Li X, Zeng X, Lin Y. Endoscopic ultrasound elastography for differentiation of benign and malignant pancreatic masses: a systemic review and meta-analysis. Eur J Gastroenterol Hepatol. 2013;25:218-224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 18. | Mei M, Ni J, Liu D, Jin P, Sun L. EUS elastography for diagnosis of solid pancreatic masses: a meta-analysis. Gastrointest Endosc. 2013;77:578-589. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 85] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 19. | Altonbary AY, Hakim H, El-Shamy AM. Diagnostic Efficacy of Endoscopic Ultrasound Elastography in Differentiating Solid Pancreatic Lesions: A Single-Center Experience. Clin Endosc. 2019;52:360-364. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 20. | Ignee A, Jenssen C, Arcidiacono PG, Hocke M, Möller K, Saftoiu A, Will U, Fusaroli P, Iglesias-Garcia J, Ponnudurai R, Petrone MC, Braden B, Burmester E, Dong Y, Atkinson NS, Dietrich CF. Endoscopic ultrasound elastography of small solid pancreatic lesions: a multicenter study. Endoscopy. 2018;50:1071-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 21. | Spier BJ, Johnson EA, Gopal DV, Frick T, Einstein MM, Byrne S, Koscik RL, Liou JI, Broxmeyer T, Selvaggi SM, Pfau PR. Predictors of malignancy and recommended follow-up in patients with negative endoscopic ultrasound-guided fine-needle aspiration of suspected pancreatic lesions. Can J Gastroenterol. 2009;23:279-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 22. | Facciorusso A, Martina M, Buccino RV, Nacchiero MC, Muscatiello N. Diagnostic accuracy of fine-needle aspiration of solid pancreatic lesions guided by endoscopic ultrasound elastography. Ann Gastroenterol. 2018;31:513-518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 23. | Yang F, Liu E, Sun S. Rapid on-site evaluation (ROSE) with EUS-FNA: The ROSE looks beautiful. Endosc Ultrasound. 2019;8:283-287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 24. | Hirooka Y, Goto H, Itoh A, Hashimoto S, Niwa K, Ishikawa H, Okada N, Itoh T, Kawashima H. Case of intraductal papillary mucinous tumor in which endosonography-guided fine-needle aspiration biopsy caused dissemination. J Gastroenterol Hepatol. 2003;18:1323-1324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 176] [Cited by in RCA: 148] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 25. | Dawwas MF, Taha H, Leeds JS, Nayar MK, Oppong KW. Diagnostic accuracy of quantitative EUS elastography for discriminating malignant from benign solid pancreatic masses: a prospective, single-center study. Gastrointest Endosc. 2012;76:953-961. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 80] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 26. | Dong Y, Jürgensen C, Puri R, D'Onofrio M, Hocke M, Wang WP, Atkinson N, Sharma M, Dietrich CF. Ultrasound imaging features of isolated pancreatic tuberculosis. Endosc Ultrasound. 2018;7:119-127. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Iordache S, Costache MI, Popescu CF, Streba CT, Cazacu S, Săftoiu A. Clinical impact of EUS elastography followed by contrast-enhanced EUS in patients with focal pancreatic masses and negative EUS-guided FNA. Med Ultrason. 2016;18:18-24. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 32] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 28. | Costache MI, Cazacu IM, Dietrich CF, Petrone MC, Arcidiacono PG, Giovannini M, Bories E, Garcia JI, Siyu S, Santo E, Popescu CF, Constantin A, Bhutani MS, Saftoiu A. Clinical impact of strain histogram EUS elastography and contrast-enhanced EUS for the differential diagnosis of focal pancreatic masses: A prospective multicentric study. Endosc Ultrasound. 2020;9:116-121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 29. | Jafri M, Sachdev AH, Khanna L, Gress FG. The Role of Real Time Endoscopic Ultrasound Guided Elastography for Targeting EUS-FNA of Suspicious Pancreatic Masses: A Review of the Literature and A Single Center Experience. JOP. 2016;17:516-524. [PubMed] |
| 30. | Kawada N, Tanaka S. Elastography for the pancreas: Current status and future perspective. World J Gastroenterol. 2016;22:3712-3724. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 64] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 31. | Lee TH, Cha SW, Cho YD. EUS elastography: advances in diagnostic EUS of the pancreas. Korean J Radiol. 2012;13 Suppl 1:S12-S16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 32. | Goyal H, Mann R, Gandhi Z, Perisetti A, Zhang Z, Sharma N, Saligram S, Inamdar S, Tharian B. Application of artificial intelligence in pancreaticobiliary diseases. Ther Adv Gastrointest Endosc. 2021;14:2631774521993059. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 33. | Săftoiu A, Vilmann P, Gorunescu F, Gheonea DI, Gorunescu M, Ciurea T, Popescu GL, Iordache A, Hassan H, Iordache S. Neural network analysis of dynamic sequences of EUS elastography used for the differential diagnosis of chronic pancreatitis and pancreatic cancer. Gastrointest Endosc. 2008;68:1086-1094. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 169] [Cited by in RCA: 174] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 34. | Săftoiu A, Vilmann P, Gorunescu F, Janssen J, Hocke M, Larsen M, Iglesias-Garcia J, Arcidiacono P, Will U, Giovannini M, Dietrich CF, Havre R, Gheorghe C, McKay C, Gheonea DI, Ciurea T; European EUS Elastography Multicentric Study Group. Efficacy of an artificial neural network-based approach to endoscopic ultrasound elastography in diagnosis of focal pancreatic masses. Clin Gastroenterol Hepatol. 2012;10:84-90.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 129] [Article Influence: 9.9] [Reference Citation Analysis (0)] |