Published online Jun 28, 2020. doi: 10.35711/aimi.v1.i1.31
Peer-review started: May 7, 2020
First decision: June 4, 2020
Revised: June 12, 2020
Accepted: June 17, 2020
Article in press: June 17, 2020
Published online: June 28, 2020
Processing time: 63 Days and 19.3 Hours
Coronary artery disease (CAD) has become a major illness endangering human health. It mainly manifests as atherosclerotic plaques, especially vulnerable plaques without obvious symptoms in the early stage. Once a rupture occurs, it will lead to severe coronary stenosis, which in turn may trigger a major adverse cardiovascular event. Computed tomography angiography (CTA) has become a standard diagnostic tool for early screening of coronary plaque and stenosis due to its advantages in high resolution, noninvasiveness, and three-dimensional imaging. However, manual examination of CTA images by radiologists has been proven to be tedious and time-consuming, which might also lead to intra- and interobserver errors. Nowadays, many machine learning algorithms have enabled the (semi-)automatic diagnosis of CAD by extracting quantitative features from CTA images. This paper provides a survey of these machine learning algorithms for the diagnosis of CAD in CTA images, including coronary artery extraction, coronary plaque detection, vulnerable plaque identification, and coronary stenosis assessment. Most included articles were published within this decade and are found in the Web of Science. We wish to give readers a glimpse of the current status, challenges, and perspectives of these machine learning-based analysis methods for automatic CAD diagnosis.
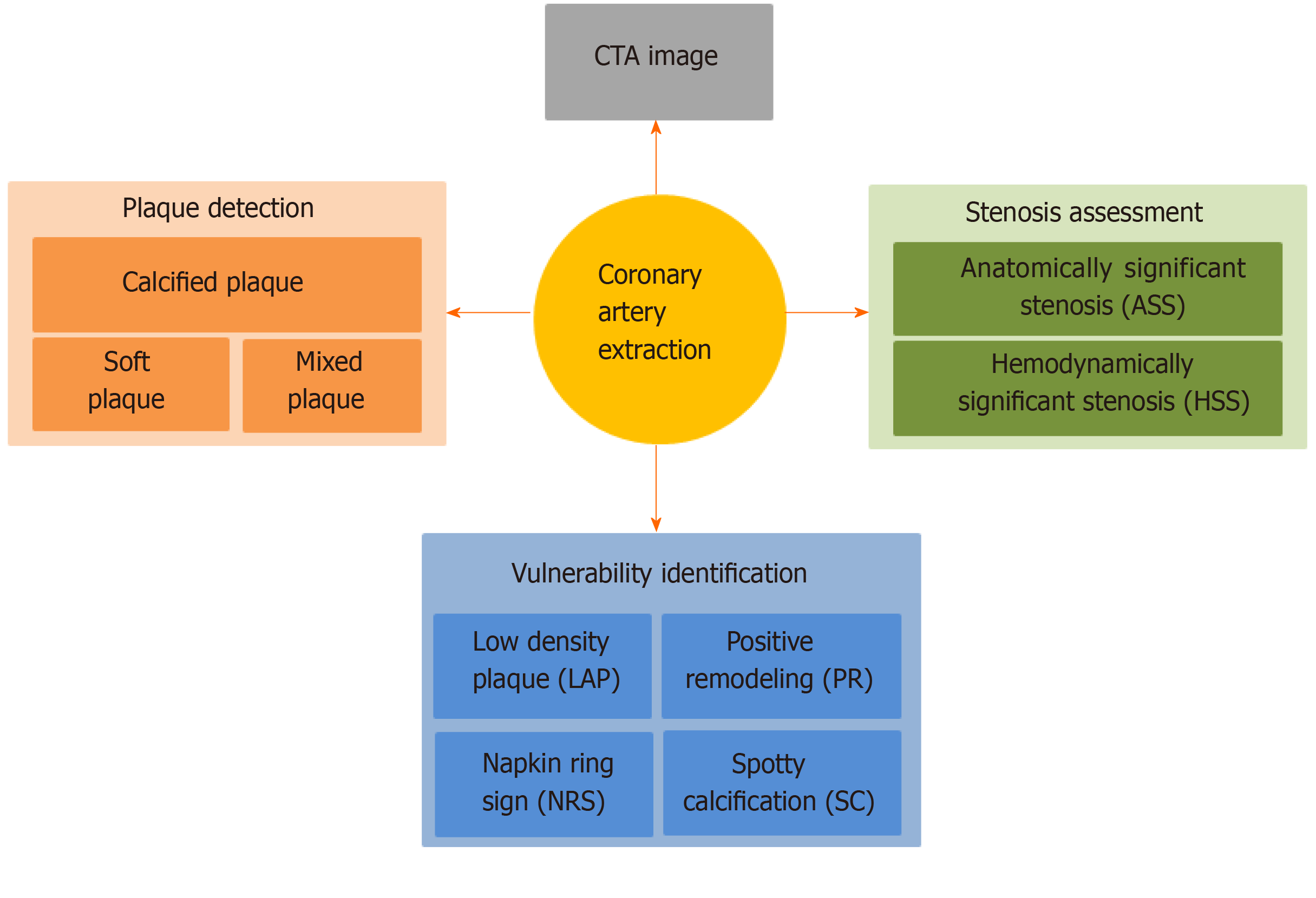
Core tip: There are reviews that contributed to the segmentation of the coronary artery, detection of calcified plaques, and calculation of fractional flow reserve. To the best of our knowledge, this is the first paper to survey the machine learning algorithms for the diagnosis of coronary artery disease in computed tomography angiography images, including extraction of coronary arteries, detection of calcified, soft and mixed plaques, identification of plaque vulnerability features including low density plaque, positive remodeling, spot calcification, and napkin ring sign, assessment of both anatomically and hemodynamically significant stenosis, and the challenges and perspectives of these machine learning-based analysis methods.
- Citation: Zhao FJ, Fan SQ, Ren JF, von Deneen KM, He XW, Chen XL. Machine learning for diagnosis of coronary artery disease in computed tomography angiography: A survey. Artif Intell Med Imaging 2020; 1(1): 31-39
- URL: https://www.wjgnet.com/2644-3260/full/v1/i1/31.htm
- DOI: https://dx.doi.org/10.35711/aimi.v1.i1.31
Coronary artery disease (CAD) has become a major illness endangering human health, which caused more than 17.6 million deaths worldwide in 2016[1]. Atherosclerotic plaque is the pathological basis of CAD, especially vulnerable plaques without obvious symptoms in the early stage. Once a rupture occurs, it will lead to severe coronary stenosis, which in turn may trigger a major adverse cardiovascular event[2]. Therefore in CAD diagnosis, it is urgent to accurately detect coronary plaques, identify their vulnerable features, and assess the resulting stenosis. Computed tomography angiography (CTA) has become a standard diagnostic tool for early screening of CAD due to its advantages in high resolution, noninvasiveness, and three-dimensional (3D) imaging[3]. However, manual examination of CTA images by radiologists has been proven to be tedious and time-consuming, which might also lead to intra- and interobserver errors[4].
To date, many state-of-the-art machine learning (ML) algorithms have enabled the (semi-)automatic diagnosis of CAD by extracting quantitative features from CTA images. These ML algorithms can be grouped into: (1) Conventional ML algorithms that are typically based on the predefined or hand-crafted features, such as linear regression, support vector machine (SVM), and random forests; and (2) Deep learning (DL) algorithms that can directly learn features from original medical images, such as the convolutional neural network (CNN) and recurrent neural network.
There are some reviews that contributed to the segmentation of the coronary artery[5], detection of calcified plaques[6], and calculation of fractional flow reserve (FFR) with both the rule-based (non-ML) and ML-based methods[7]. This paper provides a survey of the above two groups of ML methods in (semi-)automatic diagnosis of CAD, including coronary artery extraction, coronary plaque detection, vulnerable plaque identification, and coronary stenosis assessment (Figure 1 and Table 1). Most included articles were published within this decade and appear in the Web of Science. Instead of exhaustively listing all of the ML methods of coronary plaque diagnosis, we focus on typical ML-based methods with CTA images in recent years and summarize the challenges regarding these methods.
| CAD diagnosis | Method | Task | Category |
| Coronary artery extraction | |||
| Schaap et al[14] | Linear and nonlinear regression | Artery | ML |
| Huang et al[15] | 3D U-net | Artery | DL |
| Kong et al[16] | ConvRNN + ConvGRU | Artery | DL |
| Shen et al[17] | 3D FCN + level set | Artery | DL |
| Wu et al[18] | CNN + nearest neighbor search | Artery | DL |
| Wolterink et al[19] | 3D dilated CNN | Centerline | DL |
| Coronary plaque detection | |||
| Mittal et al[20] | PBT, RF | Calcified | ML |
| Kurkure et al[21] | SVM | Calcified | ML |
| Wei et al[22] | Linear discriminant analysis | Soft | ML |
| Jawaid et al[23] | SVM | Soft | ML |
| Tessmann et al[24] | AdaBoost | Multiple | ML |
| Kelm et al[25] | PBT, RF | Multiple | ML |
| Zhao et al[26] | SVM | Multiple | ML |
| Zreik et al[27] | CNN + RNN | Multiple | DL |
| Huo et al[28] | Attention recognition dual network | Calcified | DL |
| Vulnerable plaque identification | |||
| Kolossváry et al[33] | Radiomics | NRS | ML |
| Kolossváry et al[2] | Radiomics | LAP &NRS | ML |
| Kolossváry et al[34] | Logistic regression, K-nearest neighbors, RF, least angle regression, naive Bayes, Gaussian process classifier, decision trees, DNN | Advanced lesion | ML, DL |
| Coronary stenosis assessment | |||
| Zuluaga et al[36] | SVM | ASS | ML |
| Kang et al[37] | SVM + formula-based analytical method | ASS | ML |
| Zreik et al[27] | CNN + RNN | ASS | DL |
| Itu et al[41] | DNN | HSS | DL |
| Wang et al[42] | DeepVessel-FFR | HSS | DL |
| Dey et al[43] | Boosted ensemble algorithm | HSS | ML |
| Kumamaru et al[44] | 2D conditional generative adversarial network + 3D convolutional ladder network | HSS | DL |
Due to the tortuous structure of the coronary arteries, it is necessary to perform multiplanar reconstruction or curved planar reconstruction visualization of CTA images before CAD diagnosis[8]. The reconstruction of both multiplanar reconstruction and curved planar reconstruction images relies on the extraction of coronary artery trees. In addition, some studies have directly carried out a plaque analysis along the cross-section perpendicular to the coronary artery[9,10]. It can be seen that the accurate extraction of coronary arteries plays an indispensable role in CAD diagnosis. Manual extraction of the coronary arteries is labor intensive and observer dependent. Therefore, automatic/semi-automatic extraction methods have been adopted, such as the Hessian matrix method, mathematical morphology, and minimal cost path[5,11]. These traditional methods discriminate coronary arteries from the background based on intuitively and exquisitely designed models[12].
ML methods transfer the segmentation into the problem of pixel classification by assigning each pixel as the coronary artery or background[13]. Specifically, Schaap et al[14] employed both linear regression and nonlinear regression to learn the arterial geometry and appearance from annotated CTA images, and then made full use of the learned knowledge to segment coronary arteries in unseen CTA images. Huang et al[15] introduced the 3D U-net, a typical fully convolutional network (FCN), to segment the coronary artery, which densely performed the pixel-wise classification via directly extracting features from CTA images. Alternatively, Kong et al[16] employed a convolutional recurrent neural network and a tree-structured convolutional gated recurrent unit to learn the anatomical structure of the coronary artery, and hereby they achieved accurate segmentation of coronary arteries. Recently, the combination of traditional methods (such as level set and nearest neighbor search) and DL methods (such as fully convolutional network and CNN) were also devised for coronary artery segmentation[17,18]. Moreover, Wolterink et al[19] trained a 3D dilated CNN to iteratively track the centerline points in CTA images in which the coronary artery could be reconstructed based on the extracted centerline and the radius of each centerline point.
Depending on the degree of calcification, coronary plaques can be divided into calcified plaques (full calcification), soft plaques (no calcification), and mixed plaques (partial calcification). Mittal et al[20] used probability boosting trees and random forests to detect coronary calcified plaques with the designed rotation invariant features along the coronary centerline. Kurkure et al[21] adopted an SVM-based method to detect the calcification positions in the aorta and coronary arteries, amongst which they selected coronary calcified plaques. Wei et al[22] proposed a topological soft gradient pre-screening method to obtain candidate soft plaques and then detected soft plaques from the candidate set by a linear discriminant analysis. Jawaid et al[23] divided the coronary cross-section into eight concentric circles. Then they constructed an SVM to identify abnormal coronary segments caused by soft plaques based on the difference in strength stability and localized and identified soft plaques. However, due to large morphological differences between different types of plaques, it is challenging to simultaneously detect multiple types of coronary plaques.
Thus, Tessmann et al[24] performed feature extraction on a cylindrical coronary region of interest and introduced the AdaBoost algorithm to identify calcified plaques and soft plaques. Kelm et al[25] regressed the vessel radius based on the pre-acquired centerline to evaluate stenosis and then constructed a classifier (similar to[20]) to determine the type of coronary plaques that caused the stenosis, so as to realize the classification of multiple types of plaques. Zhao et al[26] designed a random radial symmetric feature vector and augmented the training data by rotating the cross-section with random angles. Then they trained an SVM to detect and classify multiclass coronary plaques. With the advantages in representing the complex texture of medical images, DL methods have been brought to the domain of plaque image analysis. Zreik et al[27] constructed a CNN model to extract the image features of coronary artery sections, and then used a recurrent neural network to fuse the features extracted by multiple CNNs. Finally, they realized the detection and classification of different coronary plaques. Huo et al[28] proposed a weak supervised attention recognition dual network to perform the detection of calcified plaques, which required only scan-level labels instead of pixel-level labels.
CTA imaging can evaluate plaque components in coronary arteries with the diameter greater than 1.5 mm[29]. Studies found that the plaque vulnerability in CTA images was closely related to low density plaque, positive remodeling, spotty calcification, and napkin ring sign (NRS)[30,31]. If a coronary plaque contains two or more of the above four vulnerable features, the plaque is more likely to be a vulnerable plaque[32]. Traditionally, visual inspection performed by radiologists is used to determine whether a coronary plaque contains the above vulnerable features. However, different patients have large individual differences in CTA imaging, resulting in the visual inspection relying heavily on experienced radiologists.
ML-based radiomics can extract a large number of quantitative features from the image to describe the complex texture and spatial structure of the lesion area, providing an automated solution for plaque vulnerability analysis. Kolossváry et al[33] applied radiomics to the identification of NRS in coronary CTA images, and the results showed that radiomic features were superior to traditional imaging parameters in distinguishing NRS and non-NRS plaques. Afterwards, they identified the low density plaque, NRS, and Na18F-positive vulnerable features in CTA images[2]. The results demonstrated that noninvasive CTA diagnosis could accurately distinguish high risk plaques that were previously diagnosed by intravascular ultrasound, optical coherence tomography, and positron emission tomography. In addition, they also collaborated with researchers from the Massachusetts General Hospital to identify advanced coronary atherosclerotic lesions through an ML-based radiomics analysis of ex vivo coronary CTA imaging[34]. The identification results on the cross-section were better than the visual inspection and histogram evaluation.
Various types of plaques are the main causes of coronary stenosis, i.e. narrowing of the coronary artery lumen, which will restrain blood flow to the myocardium and potentially lead to myocardial ischemia[35]. Therefore, the assessment of coronary stenosis is also an important aspect in the diagnosis of CAD. Taking physiology into account, coronary stenotic lesions are generally categorized as anatomically significant stenosis and hemodynamically significant stenosis, both of which can be noninvasively assessed by CTA imaging. Anatomically significant stenosis refers to the narrowing of the coronary lumen of at least 50%, which acts as the early assessment for the severity of stenosis in CAD patients. Zuluaga et al[36] employed SVM to detect coronary stenosis and arterial bifurcation based on the features of concentric circles in two-dimensional cross-sectional images. Kang et al[37] developed a structured learning algorithm based on SVM and a formula-based analytical method to detect both obstructive (with over 50% stenosis) and non-obstructive (with stenosis between 25% and 50%) lesions. Furthermore, Zreik et al[27] applied a recurrent CNN on coronary artery multiplanar reconstruction images to detect different grades of anatomically significant stenosis, including no stenosis, nonsignificant stenosis (with less than 50% narrowing), and significant stenosis (with over 50% narrowing). However, the detected anatomically significant stenosis from CTA images has only moderate specificity for predicting hemodynamically significant stenosis (HSS) that causes myocardial ischemia[38].
Currently, FFR is the standard examination for diagnosis of HSS, which invasively measures the ratio of distal blood flow to the proximal blood flow of the stenosis by inserting a special catheter. FFR estimation based on CTA images (FFTCT) provides a noninvasive alternative for evaluating HSS based on computational fluid dynamics[39,40], which is accurate but computationally demanding due to the complex iterative computation. To improve the computation efficiency, Itu et al[41] proposed an artificial neural network to predict the FFR value of each coronary artery segment based on the geometry and global features extracted from the most severe stenosis. Wang et al[42] developed a DL method (DEEPVESSEL-FFR) to calculate the FFR value from CTA images and predicted the ischemic risk of HSS. Both of the above ML-based FFR prediction methods only rely on the geometry of the coronary artery, leading to their susceptibility to the errors of coronary artery segmentation. Therefore, Dey et al[43] performed the HSS identification with a boosted ensemble algorithm, which combined the geometric features of stenosis with the volumes of plaques, the contrast density difference, and the plaque length. Moreover, Kumamaru et al[44] proposed a 3D DL model to identify patients with at least one HSS, where the model could automatically extract the representative features from the CTA dataset without segmentation or other data manipulation.
ML algorithms have been widely used in the analysis of CTA images for CAD diagnosis, including the extraction of coronary arteries, diagnosis of plaques, and assessment of stenotic lesions. In particular, DL methods can directly extract task-specific features from input CTA images, which have partially replaced conventional ML methods that depend on the hand-crafted features (or engineered features). Nevertheless, there are some merits and challenges for both the conventional ML methods and DL-based methods. (1) Conventional ML methods are more often involved in plaque and stenosis diagnosis, where the used hand-crafted features were designed according to the visual and clinical experience of radiologists. For this reason, the diagnostic results of these ML methods are inherently explainable, which means they can explicitly show task-relevant quantitative features. Moreover, these ML models are relatively simple and easy to train with only a small number of CTA images. However, the quantitative features used in the ML methods heavily depend on the careful designing by computer vision experts. How to develop or select task-specific quantitative features requires extensive experience accumulation; and (2) DL-based methods are sometimes applied in both coronary artery extraction, and stenosis and plaque diagnosis. DL methods can integrate the whole ML-based analysis workflow including (hand-crafted) feature extraction, feature selection, and classifier training into only one DL model, whose performance would be continuously improved via end-to-end learning as long as enough training samples are provided[45]. However, DL methods generally require a large number of training samples. As is known, manual labeling of coronary data is time-consuming and laborious, so the number of labeled samples is still very limited, even though there are large amounts of patient data in the clinics. Moreover, difficulty in interpretability may also prevent using the DL methods in clinical diagnosis of CAD.
Nevertheless, the DL method has become an important branch in the family of ML algorithms, especially for coronary artery segmentation and coronary stenosis assessment. It is foreseeable that most tasks in CAD diagnosis may start using DL methods or at least the combination of DL and conventional ML methods. For the latter, the DL method functions like a feature extractor, and the classifier from the conventional ML method carries out the subsequent classification. There are some solutions that may address the shortcomings of DL methods. For example, semi-supervised DL methods in natural image processing can potentially solve the classification with only small labeled data. It is reported the prediction error of semi-supervised methods using only 4000 labeled samples in the CIFAR-10 dataset was approximated to supervised learning with 50000 labeled samples[46,47]. Moreover, some studies tried to explain the decision made by a DL model by double-checking the results with an expert[48], generating a heat-map to highlight the input regions responsible for a specific task[49], or projecting the high-dimensional feature space to a bi-dimensional plane[50].
In conclusion, we have surveyed the ML-based CAD diagnostic methods in CTA images in recent years and highlighted the most typical application of both conventional ML and DL methods. We wish to give the readers a glimpse of the current status, challenges, and perspectives of these ML-based analysis methods for automatic CAD diagnosis.
Manuscript source: Invited manuscript
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Chello M, Dai X, Korosoglou G S-Editor: Wang JL L-Editor: Filipodia E-Editor: Xing YX
| 1. | Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Das SR, Delling FN, Djousse L, Elkind MSV, Ferguson JF, Fornage M, Jordan LC, Khan SS, Kissela BM, Knutson KL, Kwan TW, Lackland DT, Lewis TT, Lichtman JH, Longenecker CT, Loop MS, Lutsey PL, Martin SS, Matsushita K, Moran AE, Mussolino ME, O'Flaherty M, Pandey A, Perak AM, Rosamond WD, Roth GA, Sampson UKA, Satou GM, Schroeder EB, Shah SH, Spartano NL, Stokes A, Tirschwell DL, Tsao CW, Turakhia MP, VanWagner LB, Wilkins JT, Wong SS, Virani SS; American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation. 2019;139:e56-e528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4294] [Cited by in RCA: 5877] [Article Influence: 979.5] [Reference Citation Analysis (5)] |
| 2. | Kolossváry M, Park J, Bang JI, Zhang J, Lee JM, Paeng JC, Merkely B, Narula J, Kubo T, Akasaka T, Koo BK, Maurovich-Horvat P. Identification of invasive and radionuclide imaging markers of coronary plaque vulnerability using radiomic analysis of coronary computed tomography angiography. Eur Heart J Cardiovasc Imaging. 2019;20:1250-1258. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 110] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 3. | Hampe N, Wolterink JM, van Velzen SGM, Leiner T, Išgum I. Machine Learning for Assessment of Coronary Artery Disease in Cardiac CT: A Survey. Front Cardiovasc Med. 2019;6:172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 4. | Litjens G, Kooi T, Bejnordi BE, Setio AAA, Ciompi F, Ghafoorian M, van der Laak JAWM, van Ginneken B, Sánchez CI. A survey on deep learning in medical image analysis. Med Image Anal. 2017;42:60-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5573] [Cited by in RCA: 4920] [Article Influence: 615.0] [Reference Citation Analysis (0)] |
| 5. | Lesage D, Angelini ED, Bloch I, Funka-Lea G. A review of 3D vessel lumen segmentation techniques: models, features and extraction schemes. Med Image Anal. 2009;13:819-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 660] [Cited by in RCA: 461] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 6. | Al'Aref SJ, Anchouche K, Singh G, Slomka PJ, Kolli KK, Kumar A, Pandey M, Maliakal G, van Rosendael AR, Beecy AN, Berman DS, Leipsic J, Nieman K, Andreini D, Pontone G, Schoepf UJ, Shaw LJ, Chang HJ, Narula J, Bax JJ, Guan Y, Min JK. Clinical applications of machine learning in cardiovascular disease and its relevance to cardiac imaging. Eur Heart J. 2019;40:1975-1986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 273] [Article Influence: 54.6] [Reference Citation Analysis (0)] |
| 7. | Cook CM, Petraco R, Shun-Shin MJ, Ahmad Y, Nijjer S, Al-Lamee R, Kikuta Y, Shiono Y, Mayet J, Francis DP, Sen S, Davies JE. Diagnostic Accuracy of Computed Tomography-Derived Fractional Flow Reserve : A Systematic Review. JAMA Cardiol. 2017;2:803-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 169] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 8. | Cademartiri F, La Grutta L, Palumbo A, Malagutti P, Pugliese F, Meijboom WB, Baks T, Mollet NR, Bruining N, Hamers R, de Feyter PJ. Non-invasive visualization of coronary atherosclerosis: state-of-art. J Cardiovasc Med (Hagerstown). 2007;8:129-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 21] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 9. | Sankaran S, Schaap M, Hunley SC, Min JK, Taylor CA, Grady L. In: Ourselin S, Joskowicz L, Sabuncu M, Unal G, Wells W, editors. Medical Image Computing and Computer-Assisted Intervention - MICCAI 2016. MICCAI 2016. Lecture Notes in Computer Science, vol 9902. Cham: Springer 2016; 380-387. [RCA] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Zuluaga MA, Hernandez Hoyos M, Orkisz M. Feature selection based on empirical-risk function to detect lesions in vascular computed tomography. Irbm. 2014;35:244-254. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 11. | Kirbas C, Quek F. A review of vessel extraction techniques and algorithms. ACM Comput Surv. 2004;36:81-121. [DOI] [Full Text] |
| 12. | Zhao F, Chen Y, Hou Y, He X. Segmentation of blood vessels using rule-based and machine-learning-based methods: a review. Multimed Syst. 2019;25:109-118. [DOI] [Full Text] |
| 13. | Tetteh G, Efremov V, Forkert ND, Schneider M, Kirschke J, Weber B, Zimmer C, Piraud M, Menze BH. DeepVesselNet: Vessel Segmentation, Centerline Prediction, and Bifurcation Detection in 3-D Angiographic Volumes. 2018 Preprint. Available from: arXiv:1803.09340. |
| 14. | Schaap M, van Walsum T, Neefjes L, Metz C, Capuano E, de Bruijne M, Niessen W. Robust shape regression for supervised vessel segmentation and its application to coronary segmentation in CTA. IEEE Trans Med Imaging. 2011;30:1974-1986. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 30] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | Huang W, Huang L, Lin Z, Huang S, Chi Y, Zhou J, Zhang J, Tan RS, Zhong L. Coronary Artery Segmentation by Deep Learning Neural Networks on Computed Tomographic Coronary Angiographic Images. Conf Proc IEEE Eng Med Biol Soc. 2018;2018:608-611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 16. | Kong B, Wang X, Bai J, Lu Y, Gao F, Cao K, Xia J, Song Q, Yin Y. Learning tree-structured representation for 3D coronary artery segmentation. Comput Med Imaging Graph. 2020;80:101688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 17. | Shen Y, Fang Z, Gao Y, Xiong N, Zhong C, Tang X. Coronary Arteries Segmentation Based on 3D FCN With Attention Gate and Level Set Function. IEEE Access. 2019;7:42826-42835. [DOI] [Full Text] |
| 18. | Wu A, Xu Z, Gao M, Buty M, Mollura DJ. Deep vessel tracking: A generalized probabilistic approach via deep learning. In: 2016 IEEE 13th International Symposium on Biomedical Imaging (ISBI). IEEE, 2016: 1363-1367. [DOI] [Full Text] |
| 19. | Wolterink JM, van Hamersvelt RW, Viergever MA, Leiner T, Išgum I. Coronary artery centerline extraction in cardiac CT angiography using a CNN-based orientation classifier. Med Image Anal. 2019;51:46-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 102] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 20. | Mittal S, Zheng Y, Georgescu B, Vega-Higuera F, Zhou SK, Meer P, Comaniciu D. Fast Automatic Detection of Calcified Coronary Lesions in 3D Cardiac CT Images. In: Wang F, Yan P, Suzuki K, Shen D, editors. Machine Learning in Medical Imaging. MLMI 2010. Lecture Notes in Computer Science, vol 6357. Berlin, Heidelberg: Springer 2010; 1-9. [RCA] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 21. | Kurkure U, Chittajallu DR, Brunner G, Le YH, Kakadiaris IA. A supervised classification-based method for coronary calcium detection in non-contrast CT. Int J Cardiovasc Imaging. 2010;26:817-828. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 30] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 22. | Wei J, Zhou C, Chan HP, Chughtai A, Agarwal P, Kuriakose J, Hadjiiski L, Patel S, Kazerooni E. Computerized detection of noncalcified plaques in coronary CT angiography: evaluation of topological soft gradient prescreening method and luminal analysis. Med Phys. 2014;41:081901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 23. | Jawaid MM, Riaz A, Rajani R, Reyes-Aldasoro CC, Slabaugh G. Framework for detection and localization of coronary non-calcified plaques in cardiac CTA using mean radial profiles. Comput Biol Med. 2017;89:84-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 24. | Tessmann M, Vega-Higuera F, Fritz D, Scheuering M, Greiner G. Multi-scale feature extraction for learning-based classification of coronary artery stenosis. SPIE. 2009;726002. |
| 25. | Kelm BM, Mittal S, Zheng Y, Tsymbal A, Bernhardt D, Vega-Higuera F, Zhou SK, Meer P, Comaniciu D. Detection, grading and classification of coronary stenoses in computed tomography angiography. Med Image Comput Comput Assist Interv. 2011;14:25-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 26. | Zhao F, Wu B, Chen F, Cao X, Yi H, Hou Y, He X, Liang J. An automatic multi-class coronary atherosclerosis plaque detection and classification framework. Med Biol Eng Comput. 2019;57:245-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 27. | Zreik M, van Hamersvelt RW, Wolterink JM, Leiner T, Viergever MA, Isgum I. A Recurrent CNN for Automatic Detection and Classification of Coronary Artery Plaque and Stenosis in Coronary CT Angiography. IEEE Trans Med Imaging. 2019;38:1588-1598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 140] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 28. | Huo Y, Terry JG, Wang J, Nath V, Bermudez C, Bao S, Parvathaneni P, Carr JJ, Landman BA. Coronary Calcium Detection using 3D Attention Identical Dual Deep Network Based on Weakly Supervised Learning. Proc SPIE Int Soc Opt Eng. 2019;10949:1094917. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 29. | Kolossváry M, Szilveszter B, Merkely B, Maurovich-Horvat P. Plaque imaging with CT-a comprehensive review on coronary CT angiography based risk assessment. Cardiovasc Diagn Ther. 2017;7:489-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 93] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 30. | Rodriguez-Granillo GA, Carrascosa P, Bruining N, Waksman R, Garcia-Garcia HM. Defining the non-vulnerable and vulnerable patients with computed tomography coronary angiography: evaluation of atherosclerotic plaque burden and composition. Eur Heart J Cardiovasc Imaging. 2016;17:481-491. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 31. | Motoyama S, Sarai M, Narula J, Ozaki Y. Coronary CT angiography and high-risk plaque morphology. Cardiovasc Interv Ther. 2013;28:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 43] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 32. | Lee JM, Bang JI, Koo BK, Hwang D, Park J, Zhang J, Yaliang T, Suh M, Paeng JC, Shiono Y, Kubo T, Akasaka T. Clinical Relevance of 18F-Sodium Fluoride Positron-Emission Tomography in Noninvasive Identification of High-Risk Plaque in Patients With Coronary Artery Disease. Circ Cardiovasc Imaging. 2017;10:e006704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 59] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 33. | Kolossváry M, Karády J, Szilveszter B, Kitslaar P, Hoffmann U, Merkely B, Maurovich-Horvat P. Radiomic Features Are Superior to Conventional Quantitative Computed Tomographic Metrics to Identify Coronary Plaques With Napkin-Ring Sign. Circ Cardiovasc Imaging. 2017;10:e006843. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 108] [Cited by in RCA: 154] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 34. | Kolossváry M, Karády J, Kikuchi Y, Ivanov A, Schlett CL, Lu MT, Foldyna B, Merkely B, Aerts HJ, Hoffmann U, Maurovich-Horvat P. Radiomics versus Visual and Histogram-based Assessment to Identify Atheromatous Lesions at Coronary CT Angiography: An ex Vivo Study. Radiology. 2019;293:89-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 98] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 35. | Fuchs TA, Fiechter M, Gebhard C, Stehli J, Ghadri JR, Kazakauskaite E, Herzog BA, Husmann L, Gaemperli O, Kaufmann PA. CT coronary angiography: impact of adapted statistical iterative reconstruction (ASIR) on coronary stenosis and plaque composition analysis. Int J Cardiovasc Imaging. 2013;29:719-724. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 42] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 36. | Zuluaga MA, Magnin IE, Hernández Hoyos M, Delgado Leyton EJ, Lozano F, Orkisz M. Automatic detection of abnormal vascular cross-sections based on density level detection and support vector machines. Int J Comput Assist Radiol Surg. 2011;6:163-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 37. | Kang D, Dey D, Slomka PJ, Arsanjani R, Nakazato R, Ko H, Berman DS, Li D, Kuo CC. Structured learning algorithm for detection of nonobstructive and obstructive coronary plaque lesions from computed tomography angiography. J Med Imaging (Bellingham). 2015;2:014003. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 59] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 38. | Meijboom WB, Van Mieghem CA, van Pelt N, Weustink A, Pugliese F, Mollet NR, Boersma E, Regar E, van Geuns RJ, de Jaegere PJ, Serruys PW, Krestin GP, de Feyter PJ. Comprehensive assessment of coronary artery stenoses: computed tomography coronary angiography versus conventional coronary angiography and correlation with fractional flow reserve in patients with stable angina. J Am Coll Cardiol. 2008;52:636-643. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 495] [Cited by in RCA: 491] [Article Influence: 28.9] [Reference Citation Analysis (0)] |
| 39. | Taylor CA, Fonte TA, Min JK. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: scientific basis. J Am Coll Cardiol. 2013;61:2233-2241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 744] [Cited by in RCA: 833] [Article Influence: 69.4] [Reference Citation Analysis (0)] |
| 40. | Tesche C, Vliegenthart R, Duguay TM, De Cecco CN, Albrecht MH, De Santis D, Langenbach MC, Varga-Szemes A, Jacobs BE, Jochheim D, Baquet M, Bayer RR Nd, Litwin SE, Hoffmann E, Steinberg DH, Schoepf UJ. Coronary Computed Tomographic Angiography-Derived Fractional Flow Reserve for Therapeutic Decision Making. Am J Cardiol. 2017;120:2121-2127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 41. | Itu L, Rapaka S, Passerini T, Georgescu B, Schwemmer C, Schoebinger M, Flohr T, Sharma P, Comaniciu D. A machine-learning approach for computation of fractional flow reserve from coronary computed tomography. J Appl Physiol (1985). 2016;121:42-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 260] [Article Influence: 28.9] [Reference Citation Analysis (0)] |
| 42. | Wang ZQ, Zhou YJ, Zhao YX, Shi DM, Liu YY, Liu W, Liu XL, Li YP. Diagnostic accuracy of a deep learning approach to calculate FFR from coronary CT angiography. J Geriatr Cardiol. 2019;16:42-48. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 38] [Reference Citation Analysis (0)] |
| 43. | Dey D, Gaur S, Ovrehus KA, Slomka PJ, Betancur J, Goeller M, Hell MM, Gransar H, Berman DS, Achenbach S, Botker HE, Jensen JM, Lassen JF, Norgaard BL. Integrated prediction of lesion-specific ischaemia from quantitative coronary CT angiography using machine learning: a multicentre study. Eur Radiol. 2018;28:2655-2664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 119] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 44. | Kumamaru KK, Fujimoto S, Otsuka Y, Kawasaki T, Kawaguchi Y, Kato E, Takamura K, Aoshima C, Kamo Y, Kogure Y, Inage H, Daida H, Aoki S. Diagnostic accuracy of 3D deep-learning-based fully automated estimation of patient-level minimum fractional flow reserve from coronary computed tomography angiography. Eur Heart J Cardiovasc Imaging. 2020;21:437-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 23] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 45. | Afshar P, Mohammadi A, Plataniotis KN, Oikonomou A, Benali H. From Handcrafted to Deep-Learning-Based Cancer Radiomics: Challenges and Opportunities. IEEE Signal Process Mag. 2019;36:132-160. [DOI] [Full Text] |
| 46. | Berthelot D, Carlini N, Goodfellow I, Papernot N, Oliver A, Raffel C. MixMatch: a holistic approach to semi-supervised learning. 2019 Preprint. Available from: arXiv:1905.02249. |
| 47. | Qizhe X, Dai Z, Hovy E, Luong MT, Le QV. Unsupervised Data Augmentation for Consistency Training. 2019 Preprint. Available from: arXiv:1904.12848v12844. |
| 48. | Oakden-Rayner L, Carneiro G, Bessen T, Nascimento JC, Bradley AP, Palmer LJ. Precision Radiology: Predicting longevity using feature engineering and deep learning methods in a radiomics framework. Sci Rep. 2017;7:1648. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 94] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 49. | Jamaludin A, Kadir T, Zisserman A. SpineNet: Automated classification and evidence visualization in spinal MRIs. Med Image Anal. 2017;41:63-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 98] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 50. | Ciompi F, Chung K, van Riel SJ, Setio AAA, Gerke PK, Jacobs C, Scholten ET, Schaefer-Prokop C, Wille MMW, Marchianò A, Pastorino U, Prokop M, van Ginneken B. Towards automatic pulmonary nodule management in lung cancer screening with deep learning. Sci Rep. 2017;7:46479. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 156] [Cited by in RCA: 196] [Article Influence: 24.5] [Reference Citation Analysis (0)] |