Published online Feb 28, 2021. doi: 10.13105/wjma.v9.i1.12
Peer-review started: October 21, 2020
First decision: November 3, 2020
Revised: December 15, 2020
Accepted: February 22, 2021
Article in press: February 22, 2021
Published online: February 28, 2021
Processing time: 131 Days and 17.2 Hours
Exosomes are small plasma membrane-bound multivesicular bodies ranging in size from 20-100 nm. Exosomes are degraded fragments of mRNA, microRNA, and enriched in proteins, lipids, and nucleic acid. They are produced in the endosomes of most eukaryotic cells and once secreted, exosomes are involved in cell to cell communication and remodeling of the matrix in the extracellular compartment. Exosome biogenesis plays a crucial role in cellular development, inflammation, immunity, hemostasis, carcinogenesis, and degeneration. Due to their unique biochemical and biophysical properties, exosomes serve a variety of functions including biomarkers of diagnostic and prognostic significance. Besides, there is an increasing level of evidence to expand our understanding of the exosomes as novel therapeutic agents. Inflammatory bowel disease (IBD) such as Crohn's disease and ulcerative colitis, hepatic fibrosis, and gastrointestinal malignancies such as colorectal cancer are the potential avenues where exosomes can be applied as cell therapy and immunotherapy and have shown promising results in several in-vitro and animal models. The purpose of this review article is to highlight the emerging role of exosomes as the diagnostic and therapeutic tool in various diseases involving the gastrointestinal tract like IBD, hepatocellular carcinoma, and colon cancer. A thorough literature search was performed on databases such as PubMed, Ovid Medline, and EMBASE to achieve the objectives of this review article.
Core Tip: Exosomes are tiny membrane-bound extracellular vesicles that are becoming increasingly popular due to their potential role in regulating various intracellular and extracellular signaling pathways. Exosome biogenesis has been shown to plays a role in cellular development, inflammation, immunity, hemostasis, carcinogenesis, and degeneration. Due to their unique properties, exosomes have the potential to serve as diagnostic, prognostic, and therapeutic vectors. This review provides a concise summary of the role of exosomes as the diagnostic and therapeutic tool in various diseases involving the gastrointestinal tract.
- Citation: Naseer M, Hadi S, Syed A, Safdari A, Tahan V. Exosomes: A new frontier under the spotlight for diagnosis and treatment of gastrointestinal diseases. World J Meta-Anal 2021; 9(1): 12-28
- URL: https://www.wjgnet.com/2308-3840/full/v9/i1/12.htm
- DOI: https://dx.doi.org/10.13105/wjma.v9.i1.12
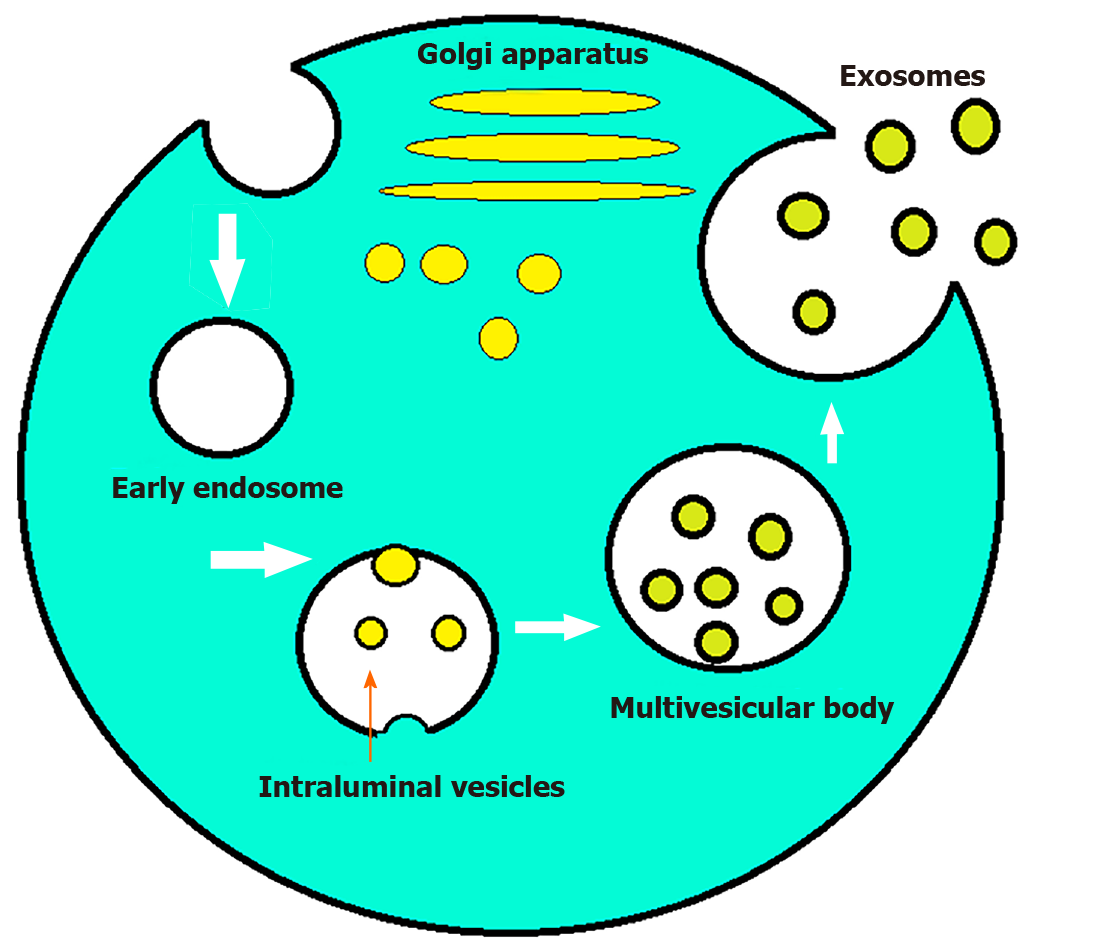
Most eukaryotic cells can internalize portions of their intracellular components to create endosomes. These endosomes then serve as a recycling plant for the cells[1]. However, some endosomes can internalize parts of their membranes to create multivesicular bodies. Vesicles found within the endosomes are called intraluminal endosomal vesicles. If these multivesicular bodies fuse with the plasma membrane releasing their intraluminal content extracellularly, then those vesicles are referred to as exosomes[2,3]. The size of the parent multivesicular bodies ultimately determine the size of the exosomes, which can range between 20-100 nm[4]. Although poorly understood at the time, exosomes first made their appearance in literature as early as the 1940s[5]. In the 1970s these extracellular “membrane fragments” were considered a universal feature of viable cells. In the early 1980s exosomes were considered nothing more than cellular debris that budded directly from the plasma membrane[6]. In 1987 exosomes were given their name by Rose Johnstone who able to isolate endosome-derived vesicles proving their intracellular origin[7]. The next major advancement in the history of exosomes was the discovery of mRNA, microRNA (miR), and proteins found within them[8,9]. Exosome protein composition can be divided into two categories. The first category depends on the endosome from which they originated and contains components from the Golgi apparatus, endoplasmic reticulum, nucleus, and mitochondria. The second category consists of evolutionarily conserved protein molecules that have the potential to serve as a biomarker (Figure 1). Exosomes also contain mRNA or miR which have been shown to alter gene expression in target cells[8]. This has led to increased research in the field and exosomes have now been shown to patriciate in biological processes including signaling, inflammation, waste management, immune response, coagulation, cell death, angiogenesis, neurodegenerative diseases, and cancer[10,11]. Exosomes also play a role in altering the microenvironment of tumor cells to help facilitate angiogenesis and metastasis to a more favorable environment in the setting of hypoxia[12]. Multiple studies have demonstrated the potential for exosomes to be used as novel biomarkers and therapeutic tools in combating cancer. Current research is focused on bringing exosomes to the clinical arena as said biomarkers and therapeutic vectors[13]. The purpose of this review article is to highlight the emerging role in the diagnostic and therapeutic utility of exosomes in various cancerous and non-cancerous diseases involving the gastrointestinal tract.
Intestinal epithelial cells interact daily with numerous dietary-derived antigens and bacteria without eliciting immune reactions. Yet, when enteric pathogens are introduced, the immune system activates to fight the novel insult. Homeostasis depends on the ability of the immune system to distinguish normal intraluminal content from invading pathogens. The failure of this process results in inflammatory bowel diseases[14-16]. Although the exact etiology of the disease remains debated, a combination of factors including genetic, immunoregulatory, and environmental likely play a major role[17]. Inflammatory bowel disease (IBD) is a systemic, chronic, noninfectious, and relapsing inflammatory disorder of the gastrointestinal tract consisting primarily of Crohn’s disease and ulcerative colitis. The symptoms of the disease commonly include abdominal pain, bloody diarrhea, weight loss, and fever[18,19]. Current therapy for the disease focuses primarily on immunosuppression with agents such as anti-tumor necrosis factor (TNF) monoclonal antibodies, immunomodulators, anti-inflammatory drugs, and thiopurine agents[20]. The incidence of IBD is increasing in developed countries and the United States alone one to 1.5 million people estimated to be affected by the disease[21-23]. Patients with the disease experience a reduction in their overall quality of life and increased mortality primarily due to an increased risk for colitis associated colorectal cancer (CRC)[24-26]. Current therapies for IBD have their limitations and many patients are unable to achieve remission. As such, novel therapies ranging from the use of herbal extracts to the introduction of harmless parasites have been proposed[27-29]. One potential avenue worth exploring in the treatment of IBD is the use of exosomes as novel biomarkers and therapeutic vectors.
Currently, the gold standard for diagnosing IBD is ileocolonoscopy. Endoscopic visualization is necessary due to the discrepancy between clinical symptoms and the extent of the disease. The endoscopic assessment provides critical information about the severity of the disease and aids in decision making regarding medical management. However, colonoscopies are associated with increased cost and patient discomfort. As such development of less invasive tools for diagnosing IBD are necessary.
Exosomes show promise as novel biomarkers for diagnosing IBD and assessing its progression (Table 1). One such biomarker is proteasomes subunit alpha type 7 (PSMA7) which is markedly upregulated in the salivary exosomes of a patient with IBD when compared to healthy individuals[30]. The exact relationship between the development of IBD and salivary exosomal PSMA7 has yet to be elucidated, but the ease with which they can be collected may represent a better diagnostic approach towards IBD[30]. Another potential biomarker for diagnosing IBD is annexin-1 containing exosomes. These exosomes arise from the intestinal epithelial cell and are overexpressed during inflammatory responses. Annexin-1 has been shown to play a role in the healing process that occurs in intestinal epithelial cells following mucosal damage. Patients with IBD have shown have increased levels of annexin-1 containing exosomes making them a potential new biomarker for diagnosing IBD[31].
| Exosomal component | Origins | Effects and mechanisms | Significance | Exosome structure effects |
| Baseline proteins in regular controls[16] | Intestinal epithelial cells | TGF-β1-dependent immunosuppression of T cells and dendritic cells | Treatment | Localization in intestinal tract and epithelial cell adhesion |
| PSMA7[30] | Saliva obtained from IBD patients | Higher expression in IBD patients when compared to normal | Diagnosis | Long distance transportation and protection |
| Annexin-1[31] | Serum of dextran sulfate sodium induced colitis in mice | Protection of epithelial cells anti-inflammatory | Diagnosis/treatment | Annexin-1 efficiency enhancement |
| Inflammatory proteins and mRNA[32] | Patients with IBD with a high endoscopic score (≥ 1) | Increased translation of IL-8 protein | Diagnosis/treatment | Membrane carrier |
| Acute phase proteins in IBD[34] | Serum of dextran sulfate sodium induced colitis in mice | Phosphorylation of p38 and ERK and production of tumor necrosis factor α resulting in macrophage activation | Diagnosis/treatment | Membrane carrier |
The pathophysiology of IBD involves a variety of factors including environmental factors, genetic factors, microbial dysbiosis, and immune system dysregulation. Current treatment for IBD centers on maintaining immuno-tolerance to obtain remission. Current research suggests that immune tolerance may be established through the regulation of activated T cells. Exosomes have been shown to have an immunosuppressive activity that can down-regulate T cells resulting in decreased severity of the disease[16]. Although the mechanism is not fully understood, the transfer of intestinal exosomes from healthy mice into those with IBD has been shown to decrease the severity of IBD[16]. On the other hand, the transfer of exosomes from IBD patients into human colonocytes cell line DLD-1 has been shown to increase the expression of inflammatory markers such as interleukin (IL)-8 within the cell line[32]. Another way that immune tolerance may be established is through the suppression of macrophages. The activation of macrophages has been suggested to play a critical role in the pathogenesis of IBD[33]. Exosomes have been shown to play a role in intercellular communication and can thus modulate the macrophage-mediated immune response[34]. Inducing acute colitis in mice results in the exosomal expression of 56 proteins to change when compared to healthy controls[35]. Most of the proteins that are expressed are acute phase immunoglobulins and proteins which participate in the coagulation cascade and the complement pathway[36]. As such exosomes represent excellent targets for blocking the activation of macrophages.
Chronic liver injury and inflammation result in the production of insoluble collagen fibers and extracellular matrix components resulting in fibrosis and eventually cirrhosis. Exosomes have been shown to participate in this process and thus can serve as potential biomarkers and therapeutic vectors[37]. The gold standard for diagnosis of liver fibrosis and cirrhosis is a biopsy. This is an invasive and expensive procedure the carries the risk of infection, internal bleeding, and injury to the biliary tract. Early research in exosomes shows promise in detecting fibrosis and cirrhosis. Exosomes collected from the serum of a patient with chronic hepatitis C (HCV) have been associated with inflammation and severity of fibrosis when enriched with CD81[38]. Human patients with F3-4 fibrosis were also noticed to have decreased levels of miR-34c, miR-151-3p, miR-483-5p, or miR-532-5p in their serum exosomes. These same miRNAs were also decreased in the serum exosomes of mice with carbon tetrachloride-induced liver fibrosis[39]. Although promising, more research is needed before exosomes can be used in a clinical setting. However, the potential of exosomes and the ease in which they can be collected has the promise of reducing invasive procedures in the future for diagnosing fibrosis or cirrhosis.
Emerging research shows that exosomes play a role in pancreatic diseases including acute pancreatitis, chronic pancreatitis, and pancreatic cancer. In acute pancreatitis exosomes have been shown to participate in pulmonary dysfunction by penetrating the alveolar endothelial barrier and activating macrophages by changing their phenotype from M2 to M1 resulting in injury to the lungs[40]. In pancreatic cancer exosomes derived from pancreatic ductal adenocarcinoma are internalized by Kupffer cells and cause secretion of transforming growth factor (TGF)-β. This results in increased fibronectin production by hepatic stellate cells resulting in activation of fibrotic pathways and proinflammatory milieu to facilitate tumor cell metastasis[41]. Exosomes have been shown to participate in complex signal pathways and such have a potential to serve as novel biomarkers when it comes to pancreatic diseases. In the case of pancreatic cancer, the first presentation are the clinical symptoms and the workup to confirm the diagnosis is costly and invasive in the form to computed tomography scan, endoscopic ultrasound, and biopsy. The current biomarker for pancreatic cancer is carbohydrate antigen 19-9 (CA19-9) which has a sensitivity and specificity of 79%-81% and 82%-90% respectively[42]. However, this is only the case in which patients are suspected to have symptoms of pancreatic cancer. When it comes to its function as a screening maker it has a low positive predictive value (0.5%-0.9%)[42]. As a result of this, better biomarker is necessary, and exosomes have the potential to fill this role.
Most of the research concerning the use of exosomes as biomarkers is in its early stages and more rigorous clinical studies are needed before they play a role in the clinical arena. However, two candidates merit further attention. One is the combination of proteoglycan glypican-1 and circulating exosomes. These exosomal markers are found to be significantly elevated in patients with pancreatic ductal adenocarcinoma when compared to healthy control[43]. Furthermore, these markers could discriminate between patients with each stage of pancreatic cancer (carcinoma in situ, stage I as well as stages II-IV) from patients with benign pancreatic disease with a 100% sensitivity and specificity[43]. Second is a recent study which compared exosomal proteoglycan glypican-1 and CA19-9 to high exosomal levels of miR-10b, miR-21, miR-30c, and miR-181a and low miR-let7a. This study found the exosomal miRNAs mentioned could not only differentiate pancreatic ductal adenocarcinoma patients from healthy controls, but also those with chronic pancreatitis. Furthermore, these exosomal levels decreased to normal values 24 h following resection whereas the levels of proteoglycan glypican-1 remained elevated[44]. Exosomes can serve as a frontier for much needed novel biomarkers in the arena of pancreatic diseases and the early research have been promising. With further studies these markers can soon have a role to play in the clinical setting as well.
Chronic gastritis occurs when the stomach lining becomes inflamed. This can be due to various factors including helicobacter pylori, ethanol use, autoimmune diseases, chronic stress, or certain medication. Currently this disease is diagnosed clinically or with the aid of endoscopy. Untreated chronic atrophic gastritis has the potential for evolving into gastric cancer. Exosomes have been suggested as novel biomarkers for the detection of chronic gastritis. Currently more research is needed in this field, but early studies suggest that exosomes play a role in the development of chronic gastritis. In one study, exomes from chronic gastritis patients with Helicobacter pylori (H. pylori) infection were shown to up regulate the expression of a proinflammatory cytokine (IL-1α) via IL-6 trans-signaling[45]. The proinflammatory effect that results due to the influence of IL-1α is a known initiator of inflammation seen in human diseases[46]. In another study, the exosomal miRNA of patients with chronic atrophic gastritis was compared to those with chronic non-atrophic gastritis. The results suggested that hsa-miR-122-5p is significantly upregulated compared to other components and has the potential to serve as a biomarker for chronic atrophic gastritis[47]. Exosomes also have been shown to play a role in the protective effect seen with H. pylori infection in inflammatory bowel disease. Exosomes derived from H. pylori infected patient were shown to promote NLRP12 expression in vitro human intestinal epithelial cells. NLRP12 was then shown to inhibit the Notch signaling pathway resulting in a decrease in chemokine MCP-1 and MIP-1α. These exosomes where then injected into dextran sulfate sodium induced colitis mice which had an improvement in colitis symptoms associated with an increase in NLRP12[48]. Currently the studies looking at the relationship between exosomes and chronic gastritis are ongoing. Early data shows that exosomes do play a role a role in gastritis, but further studies are needed to figure out the nature of this interaction.
Exosomes have been known to participate in complex cellular communication. One way in which they accomplish this is by transfer genetic material between cells. This normal process, however, can also be hijacked by viruses to promote proliferation and avoid immune response. In a study by Lin et al[49] the exosomes found within the serum of patients with chronic hepatitis B (HBV) infection contained HBV proteins and nucleic acids. Furthermore, these components were transferred to hepatocytes in an active manner. Natural killer cells from healthy donors and those with chronic HBV infection were shown to interact with and uptake exosomes containing HBV nucleic acids[49]. These exosomes then impaired natural killer cells resulting in dysfunction[49]. Similarly, patients infected with HCV were also found to have HCV viral RNA and proteins within their exosomes[50]. Exosomes that contained HCV were shown to increase the infectivity of HCV compared to exosomes free of HCV[51]. Also, exosomes helped HCV escape immune response up upregulating Gal-9 which is associated with T-cell inhibition[52]. Due to the role exosomes play in propagating infection by HBV and HCV, they also have the potential to serve as novel therapeutic targets. More research is needed to further understand the nature of exosome use by viruses and development of therapies.
Emerging evidence is beginning to show the potential for exosomes in the disease process of CRC. Due to its resistance to treatment and metastatic properties, CRC is the third leading cause of cancer mortality worldwide[53]. CRC has a high prevalence in both men and women and carries a low 5-year survival, especially when it has progressed to stage IV[54]. Of those newly diagnosed with CRC, approximately 25% have distant metastases at the time of presentation[55,56]. Furthermore, an additional 25% will develop distant metastases two years after the initial diagnosis. The prevalence of metastasis observed in CRC serves as a major challenge in its treatment. CRC is known to preferentially metastasize to the liver and a lesser extent the brain, bones, and lungs[57,58]. Exosomes from cancer cells have been shown to preferentially fuse at predicted sites of their metastasis[59]. The unique integrin expression pattern is thought to be the cause of this observed effect[59]. As exosomes likely play a role in the metastasis of CRC; they have the potential to serve as targets for therapeutic interventions. Exosomes also have the potential to serve as novel biomarkers for detecting and monitoring the progression of the disease.
Cancers of different types are known to be associated with their specific exosomal membrane and cytosolic proteins. These proteins can serve as potential biomarkers for detecting CRC in patients. In the exosomes of a patient with CRC different proteins are upregulated when compared to healthy controls[60]. An interaction between FN1 and matrix metalloproteinase 9, both of which are upregulated in patients with CRC, has been shown to cause changes in tumor microenvironment favoring progression and metastasis by integrin signaling and cytoskeletal reorganization[60]. Another potential biomarker for the diagnosis of CRC is a heat shock protein 60 (Hsp60). This protein is known to play a pivotal role in tumorigenesis and is upregulated in exosomes of patients with CRC. Hsp60 can also be used to monitor response to treatment as its level return to normal after resection of CRC tumor[61].
Exosomes also carry miRNAs which are known to play an important role in the progression of CRC. The expression of these molecules differs in CRC when compared to healthy controls. As such, miRNAs can serve as reliable biomarkers for the diagnosis and staging of CRC. Seven of these molecules (let-7a, miR-21, miR-23a, miR-150, miR-223, miR-1229, and miR-1246) are known to be upregulated even in the early stages of CRC. These markers can also be used to monitor treatment response, as their levels in the serum drop after tumor resection[62]. Another potential biomarker that can help in the staging of CRC is miR-193a. Higher levels of circulating miR-193a are associated with advanced stages of CRC, whereas lower levels are seen in the early stages and healthy controls[63]. In addition, miRNAs can be used to monitor for the recurrence of CRC as higher levels of miR-17-92a after therapy correlates with recurrence. The difference in expression of miRNA can also be used as a prognostic tool, as a higher expression of miRNA-19a is associated with a poorer prognosis when compared to those with low expression[64].
Through their role in altering the tumor microenvironment, exosomes have been shown to impact tumor initiation, progression, chemoresistance, and even metastasis. The precise mechanism by which they accomplish this has not been delineated. As such, more research is needed before exosomes can play a central role as novel biomarkers. Although the use of exosomes as biomarker show promise in research done on cell lines, animal models, and human body fluids; large randomized clinical trials are still needed for validation. Moreover, further studies comparing the existing modalities and exosomes are still needed to show if exosomal biomarkers allow for earlier detection of CRC.
Research focusing on strengthening host immunity to combat CRC via immunotherapy shows promise. Dendritic cells (DCs) are potent antigen-presenting cells that play an important role in regulating both the adaptive and innate immune responses[65]. Dendritic cell-derived exosomes (Dex) are exosomes that are secreted by DCs and have been shown to elicit a strong immune response against cancer cells[50]. This response is due to an abundance of immunostimulatory membrane proteins being present on the surface of Dex (integrins, CD81, CD63, MHC class I and II)[66,67]. In mouse models, Dex loaded with tumor peptides can invoke a stronger immune response in cytotoxic T cells than even tumor peptide loaded DCs themselves[68]. Exosomes produced by cancer cells inhibit the proliferation and cytotoxic effects of natural killer cells[69]. Furthermore, they carry the Fas ligand on their surface to induce T cell apoptosis[70]. In contrast, exosomes produced by DCs activate anti-tumor immunity and cause tumor cell lysis[71,72]. Dex loaded with tumor antigens have already been tested in clinical trials and were found to be safe and effective in activating antitumor immunity in the cases of melanoma and non-small cell lung cancer[73,74]. While the use of Dex in the treatment of CRC remains to be further studied, phase I clinical trials have already begun. In the cases of advanced CRC, the combination of ascites-derived exosomes and granulocyte-macrophage colony-stimulating factor has been shown to induce the activation of antitumor cytotoxic T cells. Treatment with this immunotherapy was not only safe but also prolonged the survival of patients with CRC in the metastatic stage of the disease[75]. While the use of Dex in the treatment of CRC shows great promise, its success has only been observed in a few small trials. Large randomized clinical trials are still needed before Dex can play a role in the clinical arena. Furthermore, research is still needed to modify Dex to elicit a stronger immune response from innate host immunity.
Exosomes play an important role in the progression of CRC and thus serve as excellent targets for therapeutic interventions. They have the potential to serve as delivery vectors for chemotherapeutic agents. Since exosomes have a slower rate of clearance from the bloodstream, they are superior to existing modalities such as liposomes. They also boast the advantage of a greater degree of specificity for CRC cells. Exosomes can also serve to enhance host immunity against cancer cells as shown by dendritic cell-derived exosomes. They could also be used to enhance chemosensitivity in cancer cells through the use of miRNA, increasing the efficacy of chemotherapeutic agents. While large clinical trials and translational studies are still needed to validate the use of exosomes in a clinical setting; they represent promising therapeutic opportunities in the fight against CRC.
The incidence of hepatocellular carcinoma (HCC), one of the most frequently diagnosed cancer, is increasing worldwide[76-78]. Increased technological development and the availability of novel screening methods are likely contributing factors; but despite advancements in screening and prevention mortality rates continue to increase[79,80]. Current screening and diagnostic tools are limited to abdominal ultrasound, computed tomography scans, magnetic resonance imagines, liver biopsies, and surveillance by alpha-fetoprotein (AFP) levels[81]. Although AFP levels > 400 ng/mL are highly correlated with HCC, only 30% of patients with HCC present with elevated AFP levels[81]. As a result, early-stage tumors are often missed causing patients to present with advanced stages of the diseases; less than 30% of these cases are amenable to surgical intervention[82-85]. As such, more sensitive methods are needed to detect these tumors. Exosomes have the potential to not only offer new tools for detection and surveillance but also open new avenues in the treatment of HCC.
The intercellular communication via exosomes has been linked with helping cancer cells create a microenvironment that serves to induce angiogenesis, avoid host cell immune response, interact with the extracellular matrix, create a pre-metastatic niche, and even develop resistance to chemotherapeutic agents[86-91]. As such, the information contained within exosomes reflects the real-time status of cancer cells. Analysis of this data can help in cancer detection, determining the stage of cancer, its recurrence, and response to chemotherapy. This information presents itself in the form of miR or protein biomarkers that have been concentrated within the exosomes which would otherwise go undetected in serum[92].
miRNA expression varies based on conditions and diseases affecting hepatocytes. Patients with HCC have higher exosomal levels of miR-18a, miR-221, miR-222, and miR224 when compared with those who have chronic hepatitis B or cirrhosis without HCC[75]. A strong correlation was also noticed between miR-21 and patients with chronic HBV[92]. A subsequent study confirmed that high levels of miR-21 were correlated with the advanced tumor stage. Patients with HCC had higher levels of miR-21 when compared to those with chronic HBV or healthy volunteers[93]. Certain miRs (939, 595, and 519d) have also been shown to be superior to AFP as a diagnostic biomarker based on receiver operating characteristic curve analysis[94].
In a separate study comparing patients with chronic HCV vs those with HCC secondary to HCV, patients with HCC had significantly lower miR-16 Levels. Moreover, miR-16 could be used to discriminate HCC from chronic HCV with a sensitivity of 57.5% and specificity of 70%. When miR-16 was combined with AFP, the sensitivity of the test improved to 85%, and the diagnostic accuracy improved to 87.5%. Additional associations were also noticed between miR-16 and the number of HCC tumors and their size[95]. In rat models, hepatocytes were converted into HCC cells via diethylnitrosamine. Variation in miR-10b, miR-21, miR-122, and miR-200a expression gradually began to appear with the development of liver disease and achieved the greatest change as cells transitioned to cirrhosis and HCC. In comparison, AFP levels remained unchanged until the early stages of HCC[96].
These studies demonstrate that exosomes directed research continues to progress in the area of primary liver disease. Current trends indicated that exosomes may have a role to play in the surveillance and diagnosis of HCC in the future clinical setting. However, further studies are needed to find reliable exosomal biomarkers and more work is needed in developing technologies to extract and accurately test the samples obtained.
Although the mechanism of resistance is not yet fully understood, HCC is known to be highly resistant to chemotherapy. Mounting evidence is showing that exosomes facilitate intercellular communications between HCC cells allowing them to develop resistance to chemotherapeutic agents. One study demonstrated that treatment with TGF-β and sorafenib caused increased expression of long-noncoding RNA within exosomes. Long intergenic non-protein coding RNA, regulator of reprogramming was then shown to enhance the expression of CD133+ tumor-initiating cells resulting in chemoresistance[97]. Another study demonstrated exosomal lincRNA-VLDLR not only promoted cell cycle progression in cancer cells, but also helped to develop resistance by expression of ABCG2 transporter which actively exported chemotherapeutic agents such as sorafenib, camptothecin, and doxorubicin out of the cell[98]. An in vitro model demonstrated that exosomes derived from patients with HCC activated the HGF/c-Met/Akt signaling pathway inducing resistance to sorafenib via inhibition of sorafenib-induced apoptosis[99].
Exosomes present a novel avenue to explore in the treatment of HCC, as they serve an integral part in cancer cell communication. Vps4A is a protein that weakens cell response to exosomes and thus shows potential as a therapeutic agent for blocking said communication. This molecule is frequently downregulated in HCC and its expression deactivates the PI3K/AKT pathway resulting in decreased growth, migration, and invasion by HCC cells in vitro[100]. Exosomes naturally accumulate in the liver after intravenous delivery. This makes them an excellent vehicle for drug delivery; they are also stable, biocompatible, and resistant to proteases or RNases[101]. MiR-355 is a molecule that has shown to have both anti-fibrotic and anti-neoplastic capabilities. In one study, the hepatic stellate cell was used to generate and deliver exosomes containing miR-335 to HCC cells. When this treatment was administered to HCC cells in vitro, inhibition of cell proliferation, and invasion was noted. When tumor-bearing mice were treated, tumor shrinkage was observed[102].
A potential therapeutic option for HCC might lie in strengthening host immunity. Exosomes are efficiently taken up by dendritic cells and can participate in antigen presentation to tumor-specific cytotoxic T cells. In mice exosomes from HCC cells have been used to confirm cross-protection against various tumors with shared antigens independent of the MHC molecules of the donors[103]. In a separate study, a tumor-suppressive effect was noticed when exosomes derived from HCC cells expressed CD9 and CD63 surface markers. These exosome markers are known to induce the cytotoxic effects of dendritic and NK cells. The activation of this cell-mediated immunity was likely the cause of the observed effect[102]. In a separate study, a significant decrease in tumor growth and prolonged survival rates were observed; when mice with HCC were exposed to exosomes obtained from AFP-expressing dendritic cells. These exosomes stimulated CD8+ T cells, activated interferon gamma, IL-2, IL-10, TGF-β, and inhibited CD25+, Foxp3+, regulatory T cells resulting in a strong antigen-specific immune response. The same response was observed with exosomes obtained from AFP-expressing bone marrow stromal cells[104].
Lastly, exosomes may also have the potential to increase chemosensitivity in cancer cells resulting in increased efficacy of chemotherapeutic agents. MiR-122 has been shown to promote chemosensitivity in HCC cells by down-regulating IGF1-R, metalloprotease 10, and cyclin G1[105]. When HCC cells were treated with miR-122 containing exosomes, they displayed increased sensitivity to sorafenib and 5 fluorouracil[106]. Despite the potential of exosomes, more research is needed before they can play a role in a clinical setting. One significant flaw of exosomes lies in the ability to produce exosomes that contain therapeutic siRNA and active drugs. Preliminary research in the field shows promise, but more work is still needed. Mesenchymal stem cells derived exosomes have been used to pack and deliver therapeutic siRNA and active drugs such as paclitaxel and deserve further investigation[107].
Gastric cancer (GC) is associated with poor prognosis despite all the recent advancements in chemotherapy, surgical techniques, and radiotherapy[108]. This in part is due to GC presenting in later stages of the disease due to its relatively asymptomatic nature early in its course. Exosomes have been shown to participate in the progression of GC and as such have the potential to serve as novel biomarkers resulting in early diagnosis and increased long-term survival of patients. Examples include exosomes derived from GC cells which have been shown to promote proliferation in an autocrine manner by activation of PI3K/Akt and MAPK/ERK pathways[93]. GC cell exosomes have also been shown to increase proliferation and invasion in an exosome-dependent manner by activation of the MAPK signaling pathway via CD 97[109,110].
Potential GC biomarker targets include the BARHL2 gene found within the exosomes in gastric juice. By looking at the methylation level of the gene, GC patients can be discriminated from non-GC control with a sensitivity of 90% and a specificity of 100%. However, this study only contained 20 GC patients and 10 non-GC control and further studies with a larger sample are needed[96]. Other promising biomarkers include exosomal proteins such as GKN1, TGF-β1, and TRIM3. GNK1 Levels are lower in healthy controls when compared to a patient with GC[111]. Increased exosomal TGF-β1 Levels are associated with the advanced stage of GC and increased TRIM3 Levels are associated with GC when compared to healthy controls[112-115]. Other potential biomarkers include exosomal miRNA which has also been shown to not only have diagnostic, but prognostic value as well. Elevated miR-19b-3p and miR-106a-5p levels in serum exosomes are more sensitive and specific for GC than AFP and CA-199[116]. High levels of miR-423-5p in serum exosomes have been correlated with lymph node metastasis and poor prognosis[117]. Post-surgery higher miR-451 Levels predict a significantly poorer prognosis than lower level[118]. Lastly, exosomal lncRNA could also serve as promising new biomarkers. Exosomes found in the plasma of stage I GC patients have significantly upregulated lncUEGC1 when compared to healthy controls. Plasma exosomal lncUEGC1 had a better diagnostic value than CEA in distinguishing stage I GC patients from healthy controls[49]. Overall exosomes show great potential as diagnostic and prognostic biomarkers for GC. However, further research is still needed to test these biomarkers in larger clinical trials to validate preliminary observations.
Globally, pancreatic cancer remained one of the most aggressive malignancy with 5- year relative survival rate as low as 40% in patients with localized disease and 3% in patients with distant metastasis[119]. The poor prognosis is attributed to factors such as lack of early diagnosis, high risk of lymphovascular invasion leading to early distant metastasis, and sub-optimal response to traditional therapies[107]. Exosomes have been identified to play a key role in cellular communication, signaling, and transportation. Due to these important properties, exosomes can serve as potential biomarkers for a better understanding of the tumor behavior and potential for metastasis. A recent study published by Que et al[120] highlights the use of pancreatic adenocarcinoma-relate miRNAs (miR-21, miR-155, miR-17-5p, and miR-196a) as potential biomarkers in 49 patients. Compared to controls, patients with advanced or unresectable pancreatic adenocarcinoma were found to have to higher expression of miR-17-5p and miR-21[108]. However, for localized pancreatic cancer i.e. stages I and IIA plasma microsomal exosome miR-196a is found to be elevated in pancreatic ductal adenocarcinoma, and miR-1246 is significantly elevated in patients with intraductal papillary mucinous neoplasms[121]. Certain exosomal proteins are also found to be elevated in a patient with pancreatic cancer. A study published by Melo et al[122] indicated that exosomes in a patient with pancreatic cancer are enriched in glypical-1 and the levels were significantly elevated in the cancer group than in controls with sensitivity and specificity of 100%. Other exosomes that were found to be expressed by the metastatic pancreatic cell to liver and lung are macrophage migration inhibitory factor and exosomal integrins such as α6β4 and α6β1[41,43]. In addition, exosomes have a role in the management of pancreatic cancer due to their unique biochemical structure and protein arrangement which enable them to act either as potential therapeutic targets or as nanoparticle carriers to transport proteins and drugs across cell membranes[123].
Cholangiocarcinoma (CCA) is a rare but aggressive malignancy involving the biliary tree. Globally, CCA accounts for 10%-25% of the primary liver cancers which constitute second most common cancer after HCC[124]. Though CCA is more prevalent in Asian countries but in recent years the incidence is even rising in Western world. CCA arises from biliary epithelium and classified as intra-hepatic and extra-hepatic (peri-hilar and distal) subtypes based on location[125]. Chronic inflammatory diseases such as primary sclerosing cholangitis, chronic cholestasis and infections are the common risk factors for CCA. However, there are significant number of cases arising in the absence of known risk factors. The cancer carries poor prognosis and so far, surgical resection is the only curative treatment available with chemoradiation therapy for locally advanced and distantly metastatic cancers[126].
There is substantial evidence that exosomes play important role in the progression of the CCA through regulating tumor microenvironment[127]. CCA microenvironment consists of dense stroma enriched with immune cells and extracellular matrix. Exosomes derived from CCA modulates tumor microenvironment and promotes formation of the tumor stroma by regulating the expression of α-smooth muscle actin mRNA, differentiation of fibroblasts particularly cancer-associated fibroblasts (CAFs) and interleukin-6 by interacting with mesenchymal stem cells[128]. CAFs are known to promote tumorigenesis by producing and releasing tumor growth factors like fibroblast growth factors and vascular endothelial growth factor. There is increasing evidence that CCA-derived exosomes are involved in cell-cell communication and transfer of the oncogenic proteins. Dutta et al[129] studied the protein profiles of CCA-derived exosomes and their potential roles in CCA progression. In this study, CCA derived exosomes named KKU-M213 and KKU-100 were incubated with normal human cholangiocyte cells i.e. H69 and proteomic analysis was performed. These isolated exosomes were found to be internalized into H69 cells, leading to migration and invasions of these cells by increasing the expression β-catenin and reducing the expression of E-cadherin. In addition, they are involved in metastasis by inhibiting the function of tumor suppressing genes such as p53 and chemoresistance. Furthermore, by inhibiting cytokine-induced killer (CIK) cells exosomes enables tumor cells for immune escape[130]. Like the proteins in exosomes, non-coding RNA such as miRs and circular RNAs are found to be involved in the progression of CCA. Particularly, miR-205-5p and circ-0000284 Levels were associated with invasion and migration of CCA cells in several studies[131,132].
Therapeutic role of exosomes in the management of CCA has been studied well in human and animal models both as a primary target to inhibit tumor progression and drug delivery. Chen et al[133] explored the impact of tumor derived exosomes on the antitumor activities of CIK cells. The results of the study showed that by down regulating the population of CD3+, CD8+, NK (CD56+), CD3+CD56+ cells and decreasing secretion of TNF-α and perforin, RBA derived exosomes inhibit the antitumor activity of CIK cells. In another study conducted by Wang and coauthors, exosomal circ-0000284 found to be a therapeutic target for CCA[134].
Exosomal composition changes in response to the state of the cells from which they are released. They provide a view of the physio-pathological process occurring within cells in real-time. Current research indicates that exosomes play a role in modulating gene expression and cell function. They are also readily available in blood, urine, and saliva marking them as promising candidates as a novel biomarker in the detection of IBD and cancer. An increasing number of studies are expanding our understanding of the potential of exosomes in diagnostic testing. However, a standard method to isolate, analyze, and track exosomes has yet to be developed limiting their application in a clinical setting. Tumorigenesis is a complex process and while certain molecules show promise, further research is still needed to identify biomarkers unique to cancer-derived exosomes. These new biomarkers then need to be tested against current detection and surveillance techniques in large randomized clinical trials. Exosomes have also been shown to play a role in managing the tumor microenvironment, helping cancer cells grow, metastasize, and avoid host immune response. They have also been shown to play a role in the development of cancers such as HCC and CRC. Given the key role they play in the propagation of cancer, they have the potential to serve as novel therapeutic vectors. The biocompatibility of exosomes makes them excellent biocarriers. Many different cell types have been used to engineer exosomes for delivering therapeutic cargos as a means for targeted therapy. The clinical application of current models, however, is limited by inefficiency. As research in the field expands, more efficient methods for generating exosomes will emerge, allowing their transition into the clinical arena. Exosomes carry miRNA and proteins that have been shown to have both a negative and positive effect on cancer cells. These molecules have been shown to limit cancer growth, increase sensitivity to chemotherapy, and activate the immune response resulting in tumor lysis. On the other hand, miRNA and exosomal proteins have also been linked to cancer growth, propagation, and metastasis. Different tumors have different characteristics and likewise, the effect of exosomal cargo on them is equally varied. Further research is needed to characterize these effects so appropriate therapeutic vectors can be selected. Cancer is an adaptive process and more innovation is needed before exosomes can be used for cancer therapy. However, early studies show considerable promise and with further research, exosomes may yet play an increasing role in a clinical setting.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): 0
Grade D (Fair): D, D
Grade E (Poor): 0
P-Reviewer: Rostami-Nejad M, Zhao CF S-Editor: Wang JL L-Editor: A P-Editor: Li X
| 1. | Huotari J, Helenius A. Endosome maturation. EMBO J. 2011;30:3481-3500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1521] [Cited by in RCA: 1803] [Article Influence: 128.8] [Reference Citation Analysis (0)] |
| 2. | Gruenberg J, van der Goot FG. Mechanisms of pathogen entry through the endosomal compartments. Nat Rev Mol Cell Biol. 2006;7:495-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 280] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 3. | van Niel G, D'Angelo G, Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19:213-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3060] [Cited by in RCA: 5620] [Article Influence: 802.9] [Reference Citation Analysis (0)] |
| 4. | Théry C, Ostrowski M, Segura E. Membrane vesicles as conveyors of immune responses. Nat Rev Immunol. 2009;9:581-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2703] [Cited by in RCA: 3108] [Article Influence: 194.3] [Reference Citation Analysis (0)] |
| 5. | Chargaff E, West R. The biological significance of the thromboplastic protein of blood. J Biol Chem. 1946;166:189-197. [PubMed] |
| 6. | Wolf P. The nature and significance of platelet products in human plasma. Br J Haematol. 1967;13:269-288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1295] [Cited by in RCA: 1220] [Article Influence: 21.0] [Reference Citation Analysis (1)] |
| 7. | Johnstone RM, Adam M, Hammond JR, Orr L, Turbide C. Vesicle formation during reticulocyte maturation. Association of plasma membrane activities with released vesicles (exosomes). J Biol Chem. 1987;262:9412-9420. [PubMed] |
| 8. | Valadi H, Ekström K, Bossios A, Sjöstrand M, Lee JJ, Lötvall JO. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat Cell Biol. 2007;9:654-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8246] [Cited by in RCA: 9798] [Article Influence: 544.3] [Reference Citation Analysis (0)] |
| 9. | Raposo G, Stoorvogel W. Extracellular vesicles: exosomes, microvesicles, and friends. J Cell Biol. 2013;200:373-383. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4900] [Cited by in RCA: 6160] [Article Influence: 513.3] [Reference Citation Analysis (0)] |
| 10. | van der Pol E, Böing AN, Harrison P, Sturk A, Nieuwland R. Classification, functions, and clinical relevance of extracellular vesicles. Pharmacol Rev. 2012;64:676-705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1439] [Cited by in RCA: 1348] [Article Influence: 103.7] [Reference Citation Analysis (0)] |
| 11. | Howitt J, Hill AF. Exosomes in the Pathology of Neurodegenerative Diseases. J Biol Chem. 2016;291:26589-26597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 206] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 12. | Park JE, Tan HS, Datta A, Lai RC, Zhang H, Meng W, Lim SK, Sze SK. Hypoxic tumor cell modulates its microenvironment to enhance angiogenic and metastatic potential by secretion of proteins and exosomes. Mol Cell Proteomics. 2010;9:1085-1099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 381] [Cited by in RCA: 451] [Article Influence: 30.1] [Reference Citation Analysis (0)] |
| 13. | Dhondt B, Van Deun J, Vermaerke S, de Marco A, Lumen N, De Wever O, Hendrix A. Urinary extracellular vesicle biomarkers in urological cancers: From discovery towards clinical implementation. Int J Biochem Cell Biol. 2018;99:236-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 47] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 14. | Dupaul-Chicoine J, Dagenais M, Saleh M. Crosstalk between the intestinal microbiota and the innate immune system in intestinal homeostasis and inflammatory bowel disease. Inflamm Bowel Dis. 2013;19:2227-2237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 15. | Xu AT, Lu JT, Ran ZH, Zheng Q. Exosome in intestinal mucosal immunity. J Gastroenterol Hepatol. 2016;31:1694-1699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 55] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 16. | Jiang L, Shen Y, Guo D, Yang D, Liu J, Fei X, Yang Y, Zhang B, Lin Z, Yang F, Wang X, Wang K, Wang J, Cai Z. EpCAM-dependent extracellular vesicles from intestinal epithelial cells maintain intestinal tract immune balance. Nat Commun. 2016;7:13045. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 107] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 17. | Gracie DJ, Ford AC. IBS-like symptoms in patients with ulcerative colitis. Clin Exp Gastroenterol. 2015;8:101-109. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 31] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 18. | Rönnblom A, Samuelsson SM, Ekbom A. Ulcerative colitis in the county of Uppsala 1945-2007: incidence and clinical characteristics. J Crohns Colitis. 2010;4:532-536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 19. | Boldeanu MV, Siloşi I, Ghiluşi M, Cojocaru M, Biciuşcă V, Avrămescu CS, Cojocaru IM, Ciurea T, Albu DF, Siloşi CA. Investigation of inflammatory activity in ulcerative colitis. Rom J Morphol Embryol. 2014;55:1345-1351. [PubMed] |
| 20. | Uranga JA, López-Miranda V, Lombó F, Abalo R. Food, nutrients and nutraceuticals affecting the course of inflammatory bowel disease. Pharmacol Rep. 2016;68:816-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 94] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 21. | Owczarek D, Rodacki T, Domagała-Rodacka R, Cibor D, Mach T. Diet and nutritional factors in inflammatory bowel diseases. World J Gastroenterol. 2016;22:895-905. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 207] [Cited by in RCA: 177] [Article Influence: 19.7] [Reference Citation Analysis (2)] |
| 22. | Andrews C, McLean MH, Durum SK. Interleukin-27 as a Novel Therapy for Inflammatory Bowel Disease: A Critical Review of the Literature. Inflamm Bowel Dis. 2016;22:2255-2264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 48] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 23. | Kappelman MD, Rifas-Shiman SL, Porter CQ, Ollendorf DA, Sandler RS, Galanko JA, Finkelstein JA. Direct health care costs of Crohn's disease and ulcerative colitis in US children and adults. Gastroenterology. 2008;135:1907-1913. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 524] [Cited by in RCA: 532] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 24. | Dyson JK, Rutter MD. Colorectal cancer in inflammatory bowel disease: what is the real magnitude of the risk? World J Gastroenterol. 2012;18:3839-3848. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 147] [Cited by in RCA: 161] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 25. | Itzkowitz SH, Yio X. Inflammation and cancer IV. Colorectal cancer in inflammatory bowel disease: the role of inflammation. Am J Physiol Gastrointest Liver Physiol. 2004;287:G7-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 870] [Cited by in RCA: 907] [Article Influence: 43.2] [Reference Citation Analysis (0)] |
| 26. | Breynaert C, Vermeire S, Rutgeerts P, Van Assche G. Dysplasia and colorectal cancer in inflammatory bowel disease: a result of inflammation or an intrinsic risk? Acta Gastroenterol Belg. 2008;71:367-372. [PubMed] |
| 27. | Chung HL, Yue GG, To KF, Su YL, Huang Y, Ko WH. Effect of Scutellariae Radix extract on experimental dextran-sulfate sodium-induced colitis in rats. World J Gastroenterol. 2007;13:5605-5611. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 41] [Cited by in RCA: 48] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 28. | Lenoir L, Joubert-Zakeyh J, Texier O, Lamaison JL, Vasson MP, Felgines C. Aloysia triphylla infusion protects rats against dextran sulfate sodium-induced colonic damage. J Sci Food Agric. 2012;92:1570-1572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 29. | Moreels TG, Pelckmans PA. Gastrointestinal parasites: potential therapy for refractory inflammatory bowel diseases. Inflamm Bowel Dis. 2005;11:178-184. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 18] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 30. | Zheng X, Chen F, Zhang Q, Liu Y, You P, Sun S, Lin J, Chen N. Salivary exosomal PSMA7: a promising biomarker of inflammatory bowel disease. Protein Cell. 2017;8:686-695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 102] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 31. | Leoni G, Neumann PA, Kamaly N, Quiros M, Nishio H, Jones HR, Sumagin R, Hilgarth RS, Alam A, Fredman G, Argyris I, Rijcken E, Kusters D, Reutelingsperger C, Perretti M, Parkos CA, Farokhzad OC, Neish AS, Nusrat A. Annexin A1-containing extracellular vesicles and polymeric nanoparticles promote epithelial wound repair. J Clin Invest. 2015;125:1215-1227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 201] [Cited by in RCA: 269] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 32. | Mitsuhashi S, Feldbrügge L, Csizmadia E, Mitsuhashi M, Robson SC, Moss AC. Luminal Extracellular Vesicles (EVs) in Inflammatory Bowel Disease (IBD) Exhibit Proinflammatory Effects on Epithelial Cells and Macrophages. Inflamm Bowel Dis. 2016;22:1587-1595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 92] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 33. | Steinbach EC, Plevy SE. The role of macrophages and dendritic cells in the initiation of inflammation in IBD. Inflamm Bowel Dis. 2014;20:166-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 191] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 34. | Cai Z, Zhang W, Yang F, Yu L, Yu Z, Pan J, Wang L, Cao X, Wang J. Immunosuppressive exosomes from TGF-β1 gene-modified dendritic cells attenuate Th17-mediated inflammatory autoimmune disease by inducing regulatory T cells. Cell Res. 2012;22:607-610. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 122] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 35. | Munagala R, Aqil F, Jeyabalan J, Gupta RC. Bovine milk-derived exosomes for drug delivery. Cancer Lett. 2016;371:48-61. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 686] [Article Influence: 68.6] [Reference Citation Analysis (0)] |
| 36. | Wong WY, Lee MM, Chan BD, Kam RK, Zhang G, Lu AP, Tai WC. Proteomic profiling of dextran sulfate sodium induced acute ulcerative colitis mice serum exosomes and their immunomodulatory impact on macrophages. Proteomics. 2016;16:1131-1145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 70] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 37. | Ban LA, Shackel NA, McLennan SV. Extracellular Vesicles: A New Frontier in Biomarker Discovery for Non-Alcoholic Fatty Liver Disease. Int J Mol Sci. 2016;17:376. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 57] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 38. | Welker MW, Reichert D, Susser S, Sarrazin C, Martinez Y, Herrmann E, Zeuzem S, Piiper A, Kronenberger B. Soluble serum CD81 is elevated in patients with chronic hepatitis C and correlates with alanine aminotransferase serum activity. PLoS One. 2012;7:e30796. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 74] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 39. | Alhomrani M, Correia J, Zavou M, Leaw B, Kuk N, Xu R, Saad MI, Hodge A, Greening DW, Lim R, Sievert W. The Human Amnion Epithelial Cell Secretome Decreases Hepatic Fibrosis in Mice with Chronic Liver Fibrosis. Front Pharmacol. 2017;8:748. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 67] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 40. | Bonjoch L, Casas V, Carrascal M, Closa D. Involvement of exosomes in lung inflammation associated with experimental acute pancreatitis. J Pathol. 2016;240:235-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 62] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 41. | Costa-Silva B, Aiello NM, Ocean AJ, Singh S, Zhang H, Thakur BK, Becker A, Hoshino A, Mark MT, Molina H, Xiang J, Zhang T, Theilen TM, García-Santos G, Williams C, Ararso Y, Huang Y, Rodrigues G, Shen TL, Labori KJ, Lothe IM, Kure EH, Hernandez J, Doussot A, Ebbesen SH, Grandgenett PM, Hollingsworth MA, Jain M, Mallya K, Batra SK, Jarnagin WR, Schwartz RE, Matei I, Peinado H, Stanger BZ, Bromberg J, Lyden D. Pancreatic cancer exosomes initiate pre-metastatic niche formation in the liver. Nat Cell Biol. 2015;17:816-826. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1554] [Cited by in RCA: 2061] [Article Influence: 206.1] [Reference Citation Analysis (0)] |
| 42. | Ballehaninna UK, Chamberlain RS. The clinical utility of serum CA 19-9 in the diagnosis, prognosis and management of pancreatic adenocarcinoma: An evidence based appraisal. J Gastrointest Oncol. 2012;3:105-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 352] [Reference Citation Analysis (3)] |
| 43. | Babic A, Wolpin BM. Circulating Exosomes in Pancreatic Cancer: Will They Succeed on the Long, Littered Road to Early Detection Marker? Clin Chem. 2016;62:307-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 44. | Lai X, Wang M, McElyea SD, Sherman S, House M, Korc M. A microRNA signature in circulating exosomes is superior to exosomal glypican-1 Levels for diagnosing pancreatic cancer. Cancer Lett. 2017;393:86-93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 265] [Cited by in RCA: 261] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 45. | Chen Y, Wang X, Yu Y, Xiao Y, Huang J, Yao Z, Chen X, Zhou T, Li P, Xu C. Serum exosomes of chronic gastritis patients infected with Helicobacter pylori mediate IL-1α expression via IL-6 trans-signalling in gastric epithelial cells. Clin Exp Immunol. 2018;194:339-349. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 24] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 46. | Malik A, Kanneganti TD. Function and regulation of IL-1α in inflammatory diseases and cancer. Immunol Rev. 2018;281:124-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 232] [Article Influence: 33.1] [Reference Citation Analysis (0)] |
| 47. | Liu H, Li PW, Yang WQ, Mi H, Pan JL, Huang YC, Hou ZK, Hou QK, Luo Q, Liu FB. Identification of non-invasive biomarkers for chronic atrophic gastritis from serum exosomal microRNAs. BMC Cancer. 2019;19:129. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 48. | Chen Y, Huang J, Li H, Li P, Xu C. Serum exosomes derived from Hp-positive gastritis patients inhibit MCP-1 and MIP-1α expression via NLRP12-Notch signaling pathway in intestinal epithelial cells and improve DSS-induced colitis in mice. Int Immunopharmacol. 2020;88:107012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 49. | Lin LY, Yang L, Zeng Q, Wang L, Chen ML, Zhao ZH, Ye GD, Luo QC, Lv PY, Guo QW, Li BA, Cai JC, Cai WY. Tumor-originated exosomal lncUEGC1 as a circulating biomarker for early-stage gastric cancer. Mol Cancer. 2018;17:84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 181] [Article Influence: 25.9] [Reference Citation Analysis (0)] |
| 50. | Yang Y, Han Q, Hou Z, Zhang C, Tian Z, Zhang J. Exosomes mediate hepatitis B virus (HBV) transmission and NK-cell dysfunction. Cell Mol Immunol. 2017;14:465-475. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 115] [Cited by in RCA: 173] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 51. | Masciopinto F, Giovani C, Campagnoli S, Galli-Stampino L, Colombatto P, Brunetto M, Yen TS, Houghton M, Pileri P, Abrignani S. Association of hepatitis C virus envelope proteins with exosomes. Eur J Immunol. 2004;34:2834-2842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 157] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 52. | Bukong TN, Momen-Heravi F, Kodys K, Bala S, Szabo G. Exosomes from hepatitis C infected patients transmit HCV infection and contain replication competent viral RNA in complex with Ago2-miR122-HSP90. PLoS Pathog. 2014;10:e1004424. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 277] [Cited by in RCA: 342] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 53. | Ewing I, Hurley JJ, Josephides E, Millar A. The molecular genetics of colorectal cancer. Frontline Gastroenterol. 2014;5:26-30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 73] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 54. | Chiba M, Kimura M, Asari S. Exosomes secreted from human colorectal cancer cell lines contain mRNAs, microRNAs and natural antisense RNAs, that can transfer into the human hepatoma HepG2 and lung cancer A549 cell lines. Oncol Rep. 2012;28:1551-1558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 172] [Cited by in RCA: 200] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 55. | Takayama T, Miyanishi K, Hayashi T, Sato Y, Niitsu Y. Colorectal cancer: genetics of development and metastasis. J Gastroenterol. 2006;41:185-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 170] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 56. | Markowitz SD, Bertagnolli MM. Molecular origins of cancer: Molecular basis of colorectal cancer. N Engl J Med. 2009;361:2449-2460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1274] [Cited by in RCA: 1392] [Article Influence: 87.0] [Reference Citation Analysis (2)] |
| 57. | Fearon ER. Molecular genetics of colorectal cancer. Annu Rev Pathol. 2011;6:479-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1163] [Cited by in RCA: 1271] [Article Influence: 90.8] [Reference Citation Analysis (1)] |
| 58. | Qiu M, Hu J, Yang D, Cosgrove DP, Xu R. Pattern of distant metastases in colorectal cancer: a SEER based study. Oncotarget. 2015;6:38658-38666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 134] [Cited by in RCA: 168] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 59. | Hoshino A, Costa-Silva B, Shen TL, Rodrigues G, Hashimoto A, Tesic Mark M, Molina H, Kohsaka S, Di Giannatale A, Ceder S, Singh S, Williams C, Soplop N, Uryu K, Pharmer L, King T, Bojmar L, Davies AE, Ararso Y, Zhang T, Zhang H, Hernandez J, Weiss JM, Dumont-Cole VD, Kramer K, Wexler LH, Narendran A, Schwartz GK, Healey JH, Sandstrom P, Labori KJ, Kure EH, Grandgenett PM, Hollingsworth MA, de Sousa M, Kaur S, Jain M, Mallya K, Batra SK, Jarnagin WR, Brady MS, Fodstad O, Muller V, Pantel K, Minn AJ, Bissell MJ, Garcia BA, Kang Y, Rajasekhar VK, Ghajar CM, Matei I, Peinado H, Bromberg J, Lyden D. Tumour exosome integrins determine organotropic metastasis. Nature. 2015;527:329-335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2697] [Cited by in RCA: 3761] [Article Influence: 376.1] [Reference Citation Analysis (0)] |
| 60. | Chen Y, Xie Y, Xu L, Zhan S, Xiao Y, Gao Y, Wu B, Ge W. Protein content and functional characteristics of serum-purified exosomes from patients with colorectal cancer revealed by quantitative proteomics. Int J Cancer. 2017;140:900-913. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 98] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 61. | Campanella C, Rappa F, Sciumè C, Marino Gammazza A, Barone R, Bucchieri F, David S, Curcurù G, Caruso Bavisotto C, Pitruzzella A, Geraci G, Modica G, Farina F, Zummo G, Fais S, Conway de Macario E, Macario AJ, Cappello F. Heat shock protein 60 Levels in tissue and circulating exosomes in human large bowel cancer before and after ablative surgery. Cancer. 2015;121:3230-3239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 118] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 62. | Ogata-Kawata H, Izumiya M, Kurioka D, Honma Y, Yamada Y, Furuta K, Gunji T, Ohta H, Okamoto H, Sonoda H, Watanabe M, Nakagama H, Yokota J, Kohno T, Tsuchiya N. Circulating exosomal microRNAs as biomarkers of colon cancer. PLoS One. 2014;9:e92921. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 557] [Cited by in RCA: 647] [Article Influence: 58.8] [Reference Citation Analysis (1)] |
| 63. | Teng Y, Ren Y, Hu X, Mu J, Samykutty A, Zhuang X, Deng Z, Kumar A, Zhang L, Merchant ML, Yan J, Miller DM, Zhang HG. MVP-mediated exosomal sorting of miR-193a promotes colon cancer progression. Nat Commun. 2017;8:14448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 240] [Cited by in RCA: 373] [Article Influence: 46.6] [Reference Citation Analysis (0)] |
| 64. | Matsumura T, Sugimachi K, Iinuma H, Takahashi Y, Kurashige J, Sawada G, Ueda M, Uchi R, Ueo H, Takano Y, Shinden Y, Eguchi H, Yamamoto H, Doki Y, Mori M, Ochiya T, Mimori K. Exosomal microRNA in serum is a novel biomarker of recurrence in human colorectal cancer. Br J Cancer. 2015;113:275-281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 332] [Cited by in RCA: 408] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 65. | O'Neill DW, Adams S, Bhardwaj N. Manipulating dendritic cell biology for the active immunotherapy of cancer. Blood. 2004;104:2235-2246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 248] [Cited by in RCA: 237] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 66. | Zhang B, Yin Y, Lai RC, Lim SK. Immunotherapeutic potential of extracellular vesicles. Front Immunol. 2014;5:518. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 103] [Cited by in RCA: 139] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 67. | Zitvogel L, Regnault A, Lozier A, Wolfers J, Flament C, Tenza D, Ricciardi-Castagnoli P, Raposo G, Amigorena S. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med. 1998;4:594-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1514] [Cited by in RCA: 1699] [Article Influence: 62.9] [Reference Citation Analysis (0)] |
| 68. | Théry C, Regnault A, Garin J, Wolfers J, Zitvogel L, Ricciardi-Castagnoli P, Raposo G, Amigorena S. Molecular characterization of dendritic cell-derived exosomes. Selective accumulation of the heat shock protein hsc73. J Cell Biol. 1999;147:599-610. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 794] [Cited by in RCA: 874] [Article Influence: 33.6] [Reference Citation Analysis (0)] |
| 69. | Clayton A, Mitchell JP, Court J, Linnane S, Mason MD, Tabi Z. Human tumor-derived exosomes down-modulate NKG2D expression. J Immunol. 2008;180:7249-7258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 373] [Cited by in RCA: 445] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 70. | Andreola G, Rivoltini L, Castelli C, Huber V, Perego P, Deho P, Squarcina P, Accornero P, Lozupone F, Lugini L, Stringaro A, Molinari A, Arancia G, Gentile M, Parmiani G, Fais S. Induction of lymphocyte apoptosis by tumor cell secretion of FasL-bearing microvesicles. J Exp Med. 2002;195:1303-1316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 542] [Cited by in RCA: 573] [Article Influence: 24.9] [Reference Citation Analysis (0)] |
| 71. | Hwang I, Shen X, Sprent J. Direct stimulation of naive T cells by membrane vesicles from antigen-presenting cells: distinct roles for CD54 and B7 molecules. Proc Natl Acad Sci USA. 2003;100:6670-6675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 154] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 72. | Skokos D, Botros HG, Demeure C, Morin J, Peronet R, Birkenmeier G, Boudaly S, Mécheri S. Mast cell-derived exosomes induce phenotypic and functional maturation of dendritic cells and elicit specific immune responses in vivo. J Immunol. 2003;170:3037-3045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 296] [Cited by in RCA: 327] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 73. | Morse MA, Garst J, Osada T, Khan S, Hobeika A, Clay TM, Valente N, Shreeniwas R, Sutton MA, Delcayre A, Hsu DH, Le Pecq JB, Lyerly HK. A phase I study of dexosome immunotherapy in patients with advanced non-small cell lung cancer. J Transl Med. 2005;3:9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 678] [Cited by in RCA: 882] [Article Influence: 44.1] [Reference Citation Analysis (0)] |
| 74. | Escudier B, Dorval T, Chaput N, André F, Caby MP, Novault S, Flament C, Leboulaire C, Borg C, Amigorena S, Boccaccio C, Bonnerot C, Dhellin O, Movassagh M, Piperno S, Robert C, Serra V, Valente N, Le Pecq JB, Spatz A, Lantz O, Tursz T, Angevin E, Zitvogel L. Vaccination of metastatic melanoma patients with autologous dendritic cell (DC) derived-exosomes: results of thefirst phase I clinical trial. J Transl Med. 2005;3:10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 763] [Cited by in RCA: 1006] [Article Influence: 50.3] [Reference Citation Analysis (0)] |
| 75. | Dai S, Wei D, Wu Z, Zhou X, Wei X, Huang H, Li G. Phase I clinical trial of autologous ascites-derived exosomes combined with GM-CSF for colorectal cancer. Mol Ther. 2008;16:782-790. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 529] [Cited by in RCA: 639] [Article Influence: 37.6] [Reference Citation Analysis (0)] |
| 76. | Cancer Genome Atlas Research Network. Comprehensive and Integrative Genomic Characterization of Hepatocellular Carcinoma. Cell 2017; 169: 1327-1341. e23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1578] [Cited by in RCA: 1732] [Article Influence: 216.5] [Reference Citation Analysis (1)] |
| 77. | El-Serag HB. Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology 2012; 142: 1264-1273. e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2183] [Cited by in RCA: 2508] [Article Influence: 192.9] [Reference Citation Analysis (2)] |
| 78. | El-Serag HB, Rudolph KL. Hepatocellular carcinoma: epidemiology and molecular carcinogenesis. Gastroenterology. 2007;132:2557-2576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3846] [Cited by in RCA: 4267] [Article Influence: 237.1] [Reference Citation Analysis (2)] |
| 79. | Schütte K, Bornschein J, Malfertheiner P. Hepatocellular carcinoma--epidemiological trends and risk factors. Dig Dis. 2009;27:80-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 239] [Cited by in RCA: 309] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 80. | Bruix J, Han KH, Gores G, Llovet JM, Mazzaferro V. Liver cancer: Approaching a personalized care. J Hepatol. 2015;62:S144-S156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 231] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 81. | Bruix J, Llovet JM. Major achievements in hepatocellular carcinoma. Lancet. 2009;373:614-616. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 233] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 82. | Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet. 2012;379:1245-1255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3249] [Cited by in RCA: 3596] [Article Influence: 276.6] [Reference Citation Analysis (4)] |
| 83. | Moris D, Vernadakis S, Papalampros A, Petrou A, Dimitroulis D, Spartalis E, Felekouras E, Fung JJ. The effect of Guidelines in surgical decision making: The paradigm of hepatocellular carcinoma. J BUON. 2016;21:1332-1336. [PubMed] |
| 84. | Moris D, Felekouras E. Ignore reality but not the consequences of its ignorance: Broaden guidelines in surgery of hepatocellular carcinoma. Hepatology. 2017;65:1772-1773. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 85. | Kosaka N. Decoding the Secret of Cancer by Means of Extracellular Vesicles. J Clin Med. 2016;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 56] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 86. | Grange C, Tapparo M, Collino F, Vitillo L, Damasco C, Deregibus MC, Tetta C, Bussolati B, Camussi G. Microvesicles released from human renal cancer stem cells stimulate angiogenesis and formation of lung premetastatic niche. Cancer Res. 2011;71:5346-5356. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 630] [Cited by in RCA: 696] [Article Influence: 49.7] [Reference Citation Analysis (0)] |
| 87. | Peinado H, Alečković M, Lavotshkin S, Matei I, Costa-Silva B, Moreno-Bueno G, Hergueta-Redondo M, Williams C, García-Santos G, Ghajar C, Nitadori-Hoshino A, Hoffman C, Badal K, Garcia BA, Callahan MK, Yuan J, Martins VR, Skog J, Kaplan RN, Brady MS, Wolchok JD, Chapman PB, Kang Y, Bromberg J, Lyden D. Melanoma exosomes educate bone marrow progenitor cells toward a pro-metastatic phenotype through MET. Nat Med. 2012;18:883-891. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2866] [Cited by in RCA: 2946] [Article Influence: 226.6] [Reference Citation Analysis (0)] |
| 88. | Zhang X, Yuan X, Shi H, Wu L, Qian H, Xu W. Exosomes in cancer: small particle, big player. J Hematol Oncol. 2015;8:83. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 461] [Cited by in RCA: 605] [Article Influence: 60.5] [Reference Citation Analysis (0)] |
| 89. | Roma-Rodrigues C, Fernandes AR, Baptista PV. Exosome in tumour microenvironment: overview of the crosstalk between normal and cancer cells. Biomed Res Int. 2014;2014:179486. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 143] [Cited by in RCA: 188] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 90. | Minciacchi VR, Freeman MR, Di Vizio D. Extracellular vesicles in cancer: exosomes, microvesicles and the emerging role of large oncosomes. Semin Cell Dev Biol. 2015;40:41-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 491] [Cited by in RCA: 709] [Article Influence: 70.9] [Reference Citation Analysis (0)] |
| 91. | Wang H, Hou L, Li A, Duan Y, Gao H, Song X. Expression of serum exosomal microRNA-21 in human hepatocellular carcinoma. Biomed Res Int. 2014;2014:864894. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 181] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 92. | Fornari F, Ferracin M, Trerè D, Milazzo M, Marinelli S, Galassi M, Venerandi L, Pollutri D, Patrizi C, Borghi A, Foschi FG, Stefanini GF, Negrini M, Bolondi L, Gramantieri L. Circulating microRNAs, miR-939, miR-595, miR-519d and miR-494, Identify Cirrhotic Patients with HCC. PLoS One. 2015;10:e0141448. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 91] [Cited by in RCA: 116] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 93. | Sohn W, Kim J, Kang SH, Yang SR, Cho JY, Cho HC, Shim SG, Paik YH. Serum exosomal microRNAs as novel biomarkers for hepatocellular carcinoma. Exp Mol Med. 2015;47:e184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 343] [Article Influence: 34.3] [Reference Citation Analysis (0)] |
| 94. | El-Abd NE, Fawzy NA, El-Sheikh SM, Soliman ME. Circulating miRNA-122, miRNA-199a, and miRNA-16 as Biomarkers for Early Detection of Hepatocellular Carcinoma in Egyptian Patients with Chronic Hepatitis C Virus Infection. Mol Diagn Ther. 2015;19:213-220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 95. | Liu WH, Ren LN, Wang X, Wang T, Zhang N, Gao Y, Luo H, Navarro-Alvarez N, Tang LJ. Combination of exosomes and circulating microRNAs may serve as a promising tumor marker complementary to alpha-fetoprotein for early-stage hepatocellular carcinoma diagnosis in rats. J Cancer Res Clin Oncol. 2015;141:1767-1778. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 56] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 96. | Takahashi K, Yan IK, Kogure T, Haga H, Patel T. Extracellular vesicle-mediated transfer of long non-coding RNA ROR modulates chemosensitivity in human hepatocellular cancer. FEBS Open Bio. 2014;4:458-467. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 310] [Cited by in RCA: 371] [Article Influence: 33.7] [Reference Citation Analysis (0)] |
| 97. | Takahashi K, Yan IK, Wood J, Haga H, Patel T. Involvement of extracellular vesicle long noncoding RNA (linc-VLDLR) in tumor cell responses to chemotherapy. Mol Cancer Res. 2014;12:1377-1387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 197] [Cited by in RCA: 251] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 98. | Qu Z, Wu J, Wu J, Luo D, Jiang C, Ding Y. Exosomes derived from HCC cells induce sorafenib resistance in hepatocellular carcinoma both in vivo and in vitro. J Exp Clin Cancer Res. 2016;35:159. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 126] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 99. | Wei JX, Lv LH, Wan YL, Cao Y, Li GL, Lin HM, Zhou R, Shang CZ, Cao J, He H, Han QF, Liu PQ, Zhou G, Min J. Vps4A functions as a tumor suppressor by regulating the secretion and uptake of exosomal microRNAs in human hepatoma cells. Hepatology. 2015;61:1284-1294. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 146] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 100. | Wiklander OP, Nordin JZ, O'Loughlin A, Gustafsson Y, Corso G, Mäger I, Vader P, Lee Y, Sork H, Seow Y, Heldring N, Alvarez-Erviti L, Smith CI, Le Blanc K, Macchiarini P, Jungebluth P, Wood MJ, Andaloussi SE. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles. 2015;4:26316. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 723] [Cited by in RCA: 1198] [Article Influence: 119.8] [Reference Citation Analysis (0)] |
| 101. | Wang F, Li L, Piontek K, Sakaguchi M, Selaru FM. Exosome miR-335 as a novel therapeutic strategy in hepatocellular carcinoma. Hepatology. 2018;67:940-954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 214] [Article Influence: 30.6] [Reference Citation Analysis (0)] |
| 102. | Rao Q, Zuo B, Lu Z, Gao X, You A, Wu C, Du Z, Yin H. Tumor-derived exosomes elicit tumor suppression in murine hepatocellular carcinoma models and humans in vitro. Hepatology. 2016;64:456-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 216] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 103. | Lu Z, Zuo B, Jing R, Gao X, Rao Q, Liu Z, Qi H, Guo H, Yin H. Dendritic cell-derived exosomes elicit tumor regression in autochthonous hepatocellular carcinoma mouse models. J Hepatol. 2017;67:739-748. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 308] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 104. | Yang N, Li S, Li G, Zhang S, Tang X, Ni S, Jian X, Xu C, Zhu J, Lu M. The role of extracellular vesicles in mediating progression, metastasis and potential treatment of hepatocellular carcinoma. Oncotarget. 2017;8:3683-3695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 75] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 105. | Lou G, Song X, Yang F, Wu S, Wang J, Chen Z, Liu Y. Exosomes derived from miR-122-modified adipose tissue-derived MSCs increase chemosensitivity of hepatocellular carcinoma. J Hematol Oncol. 2015;8:122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 387] [Cited by in RCA: 555] [Article Influence: 55.5] [Reference Citation Analysis (0)] |
| 106. | Zhang Y, Li L, Yu J, Zhu D, Zhang Y, Li X, Gu H, Zhang CY, Zen K. Microvesicle-mediated delivery of transforming growth factor β1 siRNA for the suppression of tumor growth in mice. Biomaterials. 2014;35:4390-4400. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 84] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 107. | Pascucci L, Coccè V, Bonomi A, Ami D, Ceccarelli P, Ciusani E, Viganò L, Locatelli A, Sisto F, Doglia SM, Parati E, Bernardo ME, Muraca M, Alessandri G, Bondiolotti G, Pessina A. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: a new approach for drug delivery. J Control Release. 2014;192:262-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 487] [Cited by in RCA: 680] [Article Influence: 61.8] [Reference Citation Analysis (0)] |
| 108. | Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68:394-424. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53206] [Cited by in RCA: 55833] [Article Influence: 7976.1] [Reference Citation Analysis (132)] |
| 109. | Qu JL, Qu XJ, Zhao MF, Teng YE, Zhang Y, Hou KZ, Jiang YH, Yang XH, Liu YP. Gastric cancer exosomes promote tumour cell proliferation through PI3K/Akt and MAPK/ERK activation. Dig Liver Dis. 2009;41:875-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 251] [Article Influence: 15.7] [Reference Citation Analysis (0)] |
| 110. | Li C, Liu DR, Li GG, Wang HH, Li XW, Zhang W, Wu YL, Chen L. CD97 promotes gastric cancer cell proliferation and invasion through exosome-mediated MAPK signaling pathway. World J Gastroenterol. 2015;21:6215-6228. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 82] [Cited by in RCA: 97] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 111. | Liu D, Li C, Trojanowicz B, Li X, Shi D, Zhan C, Wang Z, Chen L. CD97 promotion of gastric carcinoma lymphatic metastasis is exosome dependent. Gastric Cancer. 2016;19:754-766. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 60] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 112. | Caspi E, Ron-El R, Golan A, Herman A, Nachum H. Early, late, and sequential embryo transfer in in vitro fertilization program: a preliminary report. Fertil Steril. 1989;52:146-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 113. | Yoon JH, Ham IH, Kim O, Ashktorab H, Smoot DT, Nam SW, Lee JY, Hur H, Park WS. Gastrokine 1 protein is a potential theragnostic target for gastric cancer. Gastric Cancer. 2018;21:956-967. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 114. | Yen EY, Miaw SC, Yu JS, Lai IR. Exosomal TGF-β1 is correlated with lymphatic metastasis of gastric cancers. Am J Cancer Res. 2017;7:2199-2208. [PubMed] |
| 115. | Fu H, Yang H, Zhang X, Wang B, Mao J, Li X, Wang M, Zhang B, Sun Z, Qian H, Xu W. Exosomal TRIM3 is a novel marker and therapy target for gastric cancer. J Exp Clin Cancer Res. 2018;37:162. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 101] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 116. | Wang N, Wang L, Yang Y, Gong L, Xiao B, Liu X. A serum exosomal microRNA panel as a potential biomarker test for gastric cancer. Biochem Biophys Res Commun. 2017;493:1322-1328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 101] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 117. | Yang H, Fu H, Wang B, Zhang X, Mao J, Li X, Wang M, Sun Z, Qian H, Xu W. Exosomal miR-423-5p targets SUFU to promote cancer growth and metastasis and serves as a novel marker for gastric cancer. Mol Carcinog. 2018;57:1223-1236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 121] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 118. | Liu F, Bu Z, Zhao F, Xiao D. Increased T-helper 17 cell differentiation mediated by exosome-mediated microRNA-451 redistribution in gastric cancer infiltrated T cells. Cancer Sci. 2018;109:65-73. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 71] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 119. | Siegel RL, Miller KD, Jemal A. Cancer statistics, 2016. CA Cancer J Clin. 2016;66:7-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12135] [Cited by in RCA: 12991] [Article Influence: 1443.4] [Reference Citation Analysis (2)] |
| 120. | Que R, Ding G, Chen J, Cao L. Analysis of serum exosomal microRNAs and clinicopathologic features of patients with pancreatic adenocarcinoma. World J Surg Oncol. 2013;11:219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 200] [Cited by in RCA: 220] [Article Influence: 18.3] [Reference Citation Analysis (1)] |
| 121. | Xu YF, Hannafon BN, Zhao YD, Postier RG, Ding WQ. Plasma exosome miR-196a and miR-1246 are potential indicators of localized pancreatic cancer. Oncotarget. 2017;8:77028-77040. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 145] [Article Influence: 18.1] [Reference Citation Analysis (0)] |
| 122. | Melo SA, Luecke LB, Kahlert C, Fernandez AF, Gammon ST, Kaye J, LeBleu VS, Mittendorf EA, Weitz J, Rahbari N, Reissfelder C, Pilarsky C, Fraga MF, Piwnica-Worms D, Kalluri R. Glypican-1 identifies cancer exosomes and detects early pancreatic cancer. Nature. 2015;523:177-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2108] [Cited by in RCA: 2203] [Article Influence: 220.3] [Reference Citation Analysis (0)] |
| 123. | Armstrong EA, Beal EW, Chakedis J, Paredes AZ, Moris D, Pawlik TM, Schmidt CR, Dillhoff ME. Exosomes in Pancreatic Cancer: from Early Detection to Treatment. J Gastrointest Surg. 2018;22:737-750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 40] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 124. | Esposito I, Schirmacher P. Pathological aspects of cholangiocarcinoma. HPB (Oxford). 2008;10:83-86. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 125. | Sirica AE. Cholangiocarcinoma: molecular targeting strategies for chemoprevention and therapy. Hepatology. 2005;41:5-15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 260] [Article Influence: 13.0] [Reference Citation Analysis (0)] |
| 126. | Doherty B, Nambudiri VE, Palmer WC. Update on the Diagnosis and Treatment of Cholangiocarcinoma. Curr Gastroenterol Rep. 2017;19:2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 155] [Article Influence: 19.4] [Reference Citation Analysis (1)] |
| 127. | Sirica AE, Gores GJ. Desmoplastic stroma and cholangiocarcinoma: clinical implications and therapeutic targeting. Hepatology. 2014;59:2397-2402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 151] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 128. | Blomme A, Fahmy K, Peulen O, Costanza B, Fontaine M, Struman I, Baiwir D, de Pauw E, Thiry M, Bellahcène A, Castronovo V, Turtoi A. Myoferlin is a novel exosomal protein and functional regulator of cancer-derived exosomes. Oncotarget. 2016;7:83669-83683. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 60] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 129. | Dutta S, Reamtong O, Panvongsa W, Kitdumrongthum S, Janpipatkul K, Sangvanich P, Piyachaturawat P, Chairoungdua A. Proteomics profiling of cholangiocarcinoma exosomes: A potential role of oncogenic protein transferring in cancer progression. Biochim Biophys Acta. 2015;1852:1989-1999. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 130. | Farshidfar F, Zheng S, Gingras MC, Newton Y, Shih J, Robertson AG, Hinoue T, Hoadley KA, Gibb EA, Roszik J, Covington KR, Wu CC, Shinbrot E, Stransky N, Hegde A, Yang JD, Reznik E, Sadeghi S, Pedamallu CS, Ojesina AI, Hess JM, Auman JT, Rhie SK, Bowlby R, Borad MJ; Cancer Genome Atlas Network, Zhu AX, Stuart JM, Sander C, Akbani R, Cherniack AD, Deshpande V, Mounajjed T, Foo WC, Torbenson MS, Kleiner DE, Laird PW, Wheeler DA, McRee AJ, Bathe OF, Andersen JB, Bardeesy N, Roberts LR, Kwong LN. Integrative Genomic Analysis of Cholangiocarcinoma Identifies Distinct IDH-Mutant Molecular Profiles. Cell Rep. 2017;18:2780-2794. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 333] [Cited by in RCA: 381] [Article Influence: 47.6] [Reference Citation Analysis (1)] |
| 131. | Xiao Y, Humphries B, Yang C, Wang Z. MiR-205 Dysregulations in Breast Cancer: The Complexity and Opportunities. Noncoding RNA. 2019;5. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 132. | Louis C, Desoteux M, Coulouarn C. Exosomal circRNAs: new players in the field of cholangiocarcinoma. Clin Sci (Lond). 2019;133:2239-2244. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 133. | Chen JH, Xiang JY, Ding GP, Cao LP. Cholangiocarcinoma-derived exosomes inhibit the antitumor activity of cytokine-induced killer cells by down-regulating the secretion of tumor necrosis factor-α and perforin. J Zhejiang Univ Sci B. 2016;17:537-544. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 134. | Wang S, Hu Y, Lv X, Li B, Gu D, Li Y, Sun Y, Su Y. Circ-0000284 arouses malignant phenotype of cholangiocarcinoma cells and regulates the biological functions of peripheral cells through cellular communication. Clin Sci (Lond). 2019;133:1935-1953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 70] [Article Influence: 11.7] [Reference Citation Analysis (0)] |