Published online Apr 30, 2019. doi: 10.13105/wjma.v7.i4.120
Peer-review started: February 22, 2019
First decision: April 13, 2019
Revised: April 22, 2019
Accepted: April 23, 2019
Article in press: April 23, 2019
Published online: April 30, 2019
Processing time: 67 Days and 7.3 Hours
Biliary complications play a significant role in morbidity of liver transplant recipients. Biliary strictures occur between 10%-25% of patients with a higher incidence in living donor recipients compared to deceased donors. Strictures can be classified as either anastomotic or non-anastomotic and may be related to ischemic events. Endoscopic management of biliary strictures in the post-transplant setting has become the preferred initial approach due to adequate rates of resolution of anastomotic and non-anastomotic strictures (NAS). However, several factors may increase complexity of the endoscopic approach including surgical anatomy, location, number, and severity of bile duct strictures. Many endoscopic tools are available, however, the approach to management of anastomotic and NAS has not been standardized. Multi-disciplinary techniques may be necessary to achieve optimal outcomes in select patients. We will review the risk factors associated with the development of bile duct strictures in the post-transplant setting along with the efficacy and complications of current endoscopic approaches available for the management of bile duct strictures.
Core tip: Biliary strictures occur between 10%-25% of patients with a higher incidence in living donor recipients compared to deceased donors. Strictures can be classified as either anastomotic or non-anastomotic and may be related to ischemic events. Many endoscopic tools are available, however, the approach to management of anastomotic and non-anastomotic strictures has not been standardized. We will review the risk factors associated with the development of bile duct strictures in the post-transplant setting along with the efficacy and complications of current endoscopic approaches available for the management of bile duct strictures.
- Citation: Akhter A, Pfau P, Benson M, Soni A, Gopal D. Endoscopic management of biliary strictures post-liver transplantation. World J Meta-Anal 2019; 7(4): 120-128
- URL: https://www.wjgnet.com/2308-3840/full/v7/i4/120.htm
- DOI: https://dx.doi.org/10.13105/wjma.v7.i4.120
Biliary complications after liver transplantation (LT) is a known and significant cause of morbidity in LT recipients. The incidence of post-LT biliary complications is increasing due to increased volume of transplants and longer survival of LT recipients[1]. It is estimated between 5%-35% of LT recipients have biliary com-plications[2,3]. The incidence of complications can be attributed to various techniques of LT including the use of living and deceased cardiac donors, number of donor bile ducts used, and type of surgical anastomosis[4]. Most often the donor liver and residual native bile duct are established in continuity with the creation of a choledochocholedochostomy[5,6]. However, the presence of primary sclerosing cholangitis (PSC) results in the creation of a hepaticojejunostomy. A roux limb is created and adds to the complexity of endoscopic management of biliary complications and may require the aid of a balloon assisted enteroscope for technical success[7].
There are a variety of biliary complications that can arise which include the development of anastomotic and non-anastomotic strictures (NAS), bile duct leaks, papillary stenosis, and presence of bile duct stones/casts. Diagnosis is usually made with a combination of non-invasive tests including liver chemistries, abdominal ultrasound, and cross-sectional imaging (computed tomography and/or magnetic resonance cholangiopancreatography). It is important to consider non-obstructive causes of cholestasis including cellular rejection, drug induced cholestasis, or recurrence of primary disease as this may prevent a delay in therapeutic intervention. Advancements in endoscopic techniques and tools have allowed endoscopic management to be the preferred method to manage most biliary complications[8,9]. Our review will focus on endoscopic management of biliary strictures that can arise after LT.
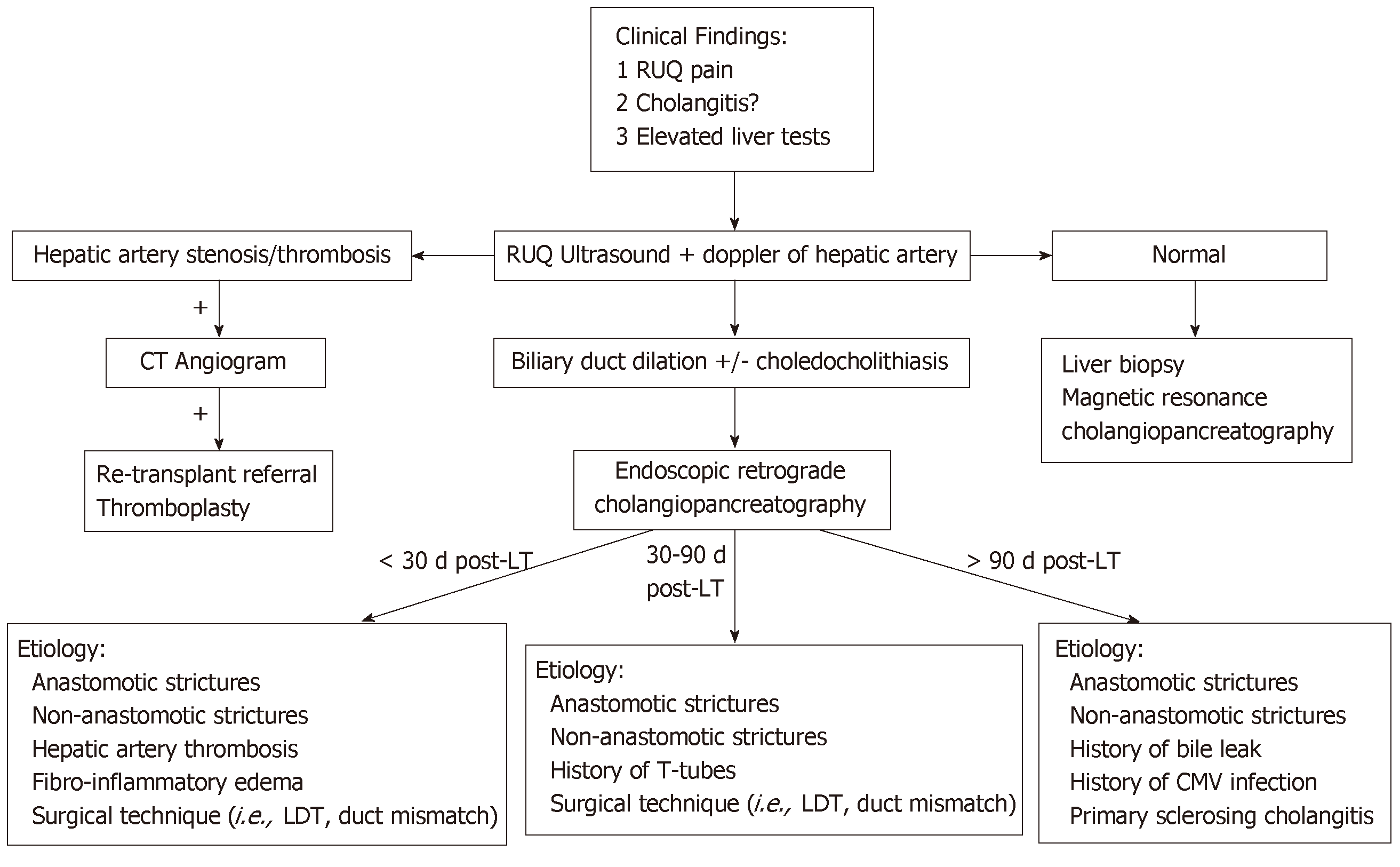
There are several risk factors that predispose to the development of bile duct strictures including hepatic artery thrombosis, donor after cardiac death, ABO incompatibility, preservation injury (cold and warm ischemia time), cytomegalovirus infection, duct mismatch between donor and recipient, presence of PSC, bile duct leaks, placement of T-tubes, and living donor transplantation (LDT)[10-15]. Bile duct strictures can be noted early (< 30 d), delayed (30-90 d), or late (> 90 d) after LT[11,16] (Figure 1). Early complications include hepatic artery thrombosis which can result in ductal stenosis and strictures as well as hepatic ischemia[5]. Post-operative edema can also result in early ductal stenosis. Delayed and late complications can involve biliary obstruction at the anastomotic site or intrahepatic ducts due to ischemia[17]. Bile leaks and recurrence of PSC are risk factors for the development of delayed/late bile duct strictures. T-tubes were previously used more frequently after LT to help maintain the reconstruction of the bile duct anastomosis. However, recent studies have found they may increase the risk of biliary complications including biliary strictures and may be more beneficial for select patients such as those who have a donor-recipient duct mismatch or a bile duct diameter < 7 mm[18,19].
LDT was first performed successfully in 1994 and has been steadily increasing due to limited supply of deceased donors[20]. LDT has advantages over deceased donor transplantation (DDT) including reduction of cold ischemia time and improved graft viability[21,22]. Nonetheless, there is a higher risk of biliary complications and specifically biliary strictures in LDT vs DDT (13%-32% vs 5%-15%)[23-25]. Incidence of biliary strictures in living donors’ range between 0.5%-4%[26,27]. LDT is presumed to carry a higher risk of biliary strictures due to the anastomosis of low-caliber and small ducts as well as increased number of donor ducts needed to establish biliary continuity[23]. Bile duct strictures can be categorized as anastomotic or non-anastomotic with differences in endoscopic management and outcomes.
Anastomotic bile duct strictures (AS) occur in 5%-10% of patients within the first 12 mo of transplantation[28,29]. However, they should always be considered in the setting of a cholestatic pattern of liver injury in LT recipients. As opposed to NAS, AS are segmental, shorter, and localized to the site of anastomosis[23,30]. Bile leaks may be an independent risk factor for the development of an AS. An AS may form within 60 d after LT due to post-operative edema and fibro-inflammatory response along with transient ischemia[1,31,32]. Strictures that form within the first 60 d respond well to 1-2 sessions of endoscopic dilation and plastic stent placement[1].
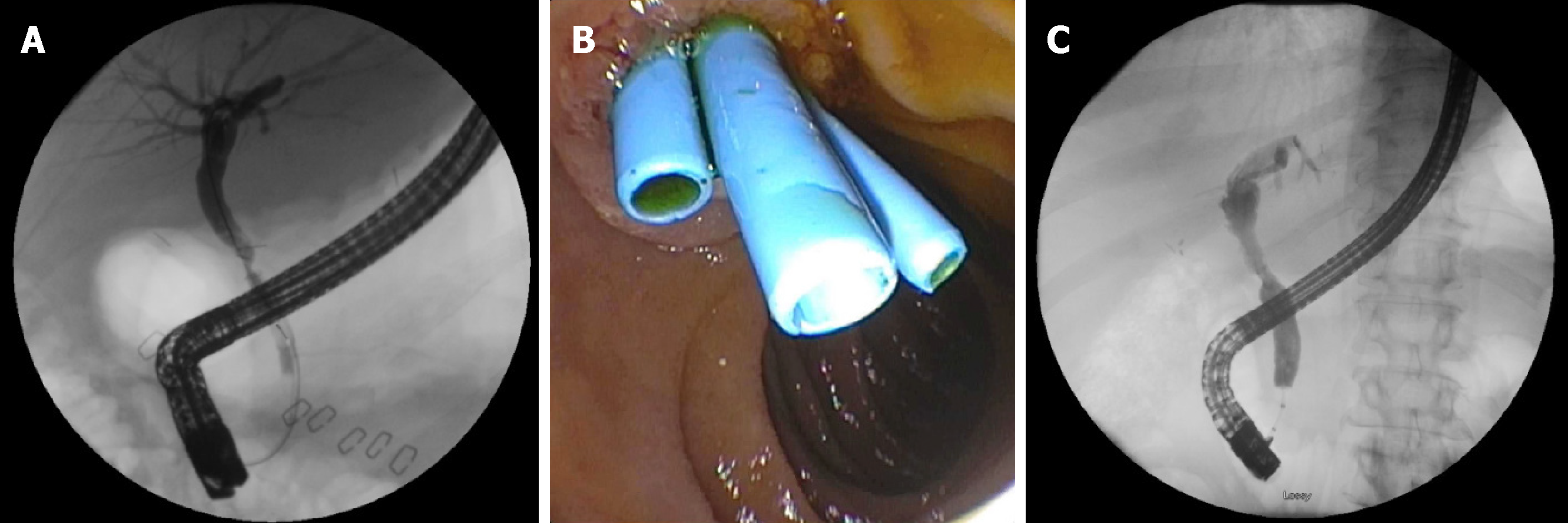
However, biliary strictures that form after 3 mo have a protracted course and require prolonged endoscopic sessions for adequate response. Endoscopic approaches for anastomotic strictures include balloon dilation, passage dilation with a Soehendra biliary dilation catheter, plastic biliary stents, and self-expandable metal stents (SEMS). A guidewire is used to cross the stricture and balloon dilators from 4-10 mm are used to dilate the anastomosis along with placement of 7 Fr to 11.5 Fr plastic stents bridging the anastomosis. The balloon size used to dilate is predicated upon the diameter of the donor bile duct. Soehendra dilators are useful in patients whom the anastomosis is severely stenosed and can be dilated from 4-10 Fr. In addition, balloon dilation is generally avoided in early strictures (< 3 mo) to avoid perforation or leaks of a recently constructed anastomosis. Most patients with an AS and those who present after 3 mo of LT, require several endoscopic sessions (3-5) for long-term success[28,33]. The patency of most plastic biliary stents is 3 mo and thus, endoscopic sessions are performed at 8-12-wk intervals to prevent biliary obstruction[16]. The pre-existing stent is removed using a snare or forceps and a cholangiogram is performed to evaluate the patency of the anastomosis. There is no standardized bile duct diameter that corresponds to a clinically significant bile duct stenosis. However, cholangiogram features of a thin focal narrowing with proximal bile duct dilation along with evaluating the resistance encountered with anterograde and/or retrograde biliary balloon sweeps with an 8.5 mm or 11.5 mm biliary balloon across the anastomosis can help determine the patency of the anastomosis. In general, the goal is to dilate the anastomosis with larger sized dilators and in combination with increasing size or number of plastic biliary stents until patency is achieved and a waist is no longer seen (Figure 2). Combination of balloon dilation and biliary stenting have shown to be more effective than balloon dilation alone[34,35]. Balloon dilation alone has a high recurrence rate of stricture formation when compared to balloon dilation and biliary stenting (62% vs 31%)[35]. LDT has lower success rates of stricture resolution compared to DDT despite similar techniques of balloon dilation plus plastic biliary stents (37%-71% vs 75%-91%)[36-39]. This may in part be explained due to the use of peripheral ducts and presence of smaller multiple anastomotic strictures[1]. Resolution of anastomotic strictures are improved with multiple and maximum number of plastic biliary stents. Several studies evaluating anastomotic stricture resolution in LT recipients found resolution rates to range between 87%-100% with recurrence in 0%-18% of patients[32,40-43]. Number of endoscopic sessions to achieve stricture resolution ranged between 3-4 with a complication rate of 1.5%-5%. Complications were primarily related to pancreatitis and cholangitis.
An alternative strategy is to place a SEMS to prevent or reduce the need for frequent ERCPs that is necessary in the setting of plastic biliary stenting. Covered metallic stents have been used as uncovered SEMS may not be able to be removed and may preclude surgical bile duct intervention. In addition, a metallic stent may lead to hyperplasia leading to the formation of sludge/stone formation proximal to the stent[1]. The role of covered SEMS has yet to be precisely defined but can be useful because of their larger diameter (10 mm), longer patency, and ability to be removed. However, they are limited because of rates of stent migration (4%-38%). Several studies examining the utility of covered SEMS after LT found resolution rates of anastomotic strictures between 61%-83%[44-48]. Recurrence rates were higher in those who received SEMS ranging between 7%-32%[44-49]. A randomized trial evaluating covered SEMS and plastic biliary stents found in sub-group analysis of post-transplant patients resolution rates of 89% vs 86% with 158 to 194 d till resolution respectively. Stricture recurrence was higher in the covered SEMS group and stent migration occurred more frequently in post-transplant AS compared to all other cases[50]. To mitigate the risks of stent migration an alternative is to use partially covered SEMS or stents with special anchoring flanges and anti-migration waists[51]. A systematic review of case series including 446 patients by Kao et al[52] did not find SEMS to have a clear advantage over multiple plastic biliary stents in LT recipients but found stricture resolution was improved in those patients whom the stent duration was longer than 3 mo. A recent meta-analysis of four randomized controlled trials comparing plastic stents to fully covered SEMS found no difference between stricture resolution, stricture recurrence, and adverse events. However, those who received a metal stent did have fewer ERCPs performed as compared to those who had plastic stents[53]. Currently, there is no standardized approach for endoscopic management of AS. The use of multiple plastic biliary stents with balloon dilation and fully covered SEMS can provide similar resolution rates of AS after LT with overall low risk of adverse events.
NAS of the bile ducts have an incidence of 5%-10% after LT[33,54,55]. The definition of a NAS is the presence of stenosis > 5 mm away from the anastomosis and may be located within the intrahepatics, hilum, or anywhere else along the bile duct (including the recipient duct). In contrast to AS, NAS may be multiple and longer in length. Recurrent PSC in the allograft or vascular insufficiency may result in the development of NAS. Vascular ischemia secondary to hepatic artery thrombosis results in biliary destruction and warm and cold ischemia, donation after cardiac death, ABO incompatibility, and chronic rejection are also risk factors for the development of NAS[1,30]. NAS tend to occur 3-6 mo after LT though as many as 50% of patients may develop NAS after the first-year post-transplant[54,56,57].
The principles regarding the management of NAS are similar to anastomotic strictures, however, the optimal protocol has not been established. Balloon dilation with placement of plastic biliary stents have shown to be helpful though with less success and longer time to resolution as compared to anastomotic strictures[33,58]. Balloon dilation is often not as aggressive as in AS with 4-6 mm biliary balloons commonly used. Overall, resolution rates of NAS range between 50%-75% and are associated with worse graft survival[30,59]. However, a study of 48 patients comparing balloon dilation alone vs balloon dilation and plastic biliary stents found a significant difference and improvement in stricture resolution in those who only underwent balloon dilation (91% vs 31%)[60]. This may in part be explained by most of these strictures being located extra-hepatic.
Bile duct strictures involving the hilum and intrahepatics may be more challenging due to difficulty with traversing the stricture secondary to the small caliber of these ducts as well as tortuosity that may be encountered. Longer, fenestrated stents (Johlin), are flexible and can be used for intrahepatic strictures and allow for adequate drainage via multiple side holes and interstent space[61]. Covered metal stents have not been readily used as they may impede flow from surrounding bile ducts and potentially increase risk of cholangitis.
NAS may progress despite improvement in liver enzymes in up to two-third of patients[40,54]. Progression of NAS is more common in patients who develop NAS within the first year after transplantation or who have recurrent cholangitis[57]. Like AS endoscopic resolution rates for NAS in LDT is lower than in DDT 25%-33% vs 50%-60% respectively[39,62]. Currently there is no standard protocol for management of NAS. NAS are of varying complexity with intrahepatic and hilar strictures providing an especially unique challenge to the endoscopist which may require alternative approaches for access and therapeutic interventions to the bile duct.
Endoscopic methods may not be feasible due to surgical anatomy (bilio-enteric anastomosis), tortuosity and angulation of the bile duct, or severity and location of the stricture which prevents a guidewire or dilation devices to traverse the stricture. Roux-en-Y hepaticojejunosotomy or roux-en-Y gastric bypass require deep ERCP methods such as balloon-assisted enteroscopy, endoscopic ultrasonography-directed transgastric ERCP, or percutaneous transhepatic cholangiography (PTC). A multi-center trial showed balloon assisted enteroscopy to be successful in two thirds of cases and in 88% of patients in whom the papilla is reached. Single or double balloon assisted enteroscopy may be an alternative before pursuing PTC or surgical al-ternatives[63].
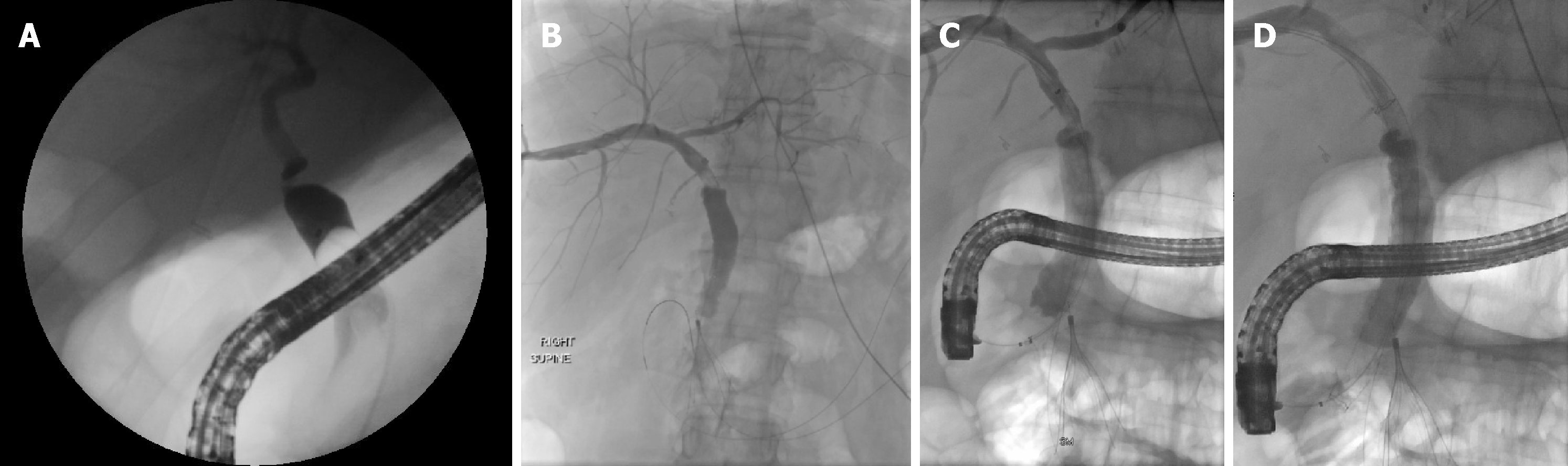
A rendezvous technique may also be used which combines PTC and an endoscopic transpapillary approach to access the bile duct and traverse the stricture that otherwise may have failed with conventional endoscopy (Figure 3). PTC in cases of benign bilio-enteric anastomotic strictures are reported to have an overall success rate of 80%[64]. It is also especially helpful in those with intractable or multiple intrahepatic strictures as internal-external stents can be placed and relieve the obstruction. In addition, the potential of swing-tip cannulas in accessing tight intrahepatic strictures have been reported and may also help achieve faster cannulation of the bile duct[65,66].
Single-operator peroral cholangioscopy can also be used in the treatment of bile duct strictures by providing direct visualization of the lumen of the bile ducts. Direct visualization of the inside of the bile duct may help predict outcomes of endoscopic therapy based upon the pattern and severity of edema and inflammation seen[67]. In addition, direct visualization can also be used in conjunction with the rendezvous technique to puncture the bile duct and safely traverse a completely obstructed duct[68,69].
Magnetic compression anastomosis (MCA) is a rescue technique used in the setting of complete biliary obstruction. A magnet is advanced to the site of the stricture via ERCP and another magnet is advanced percutaneously via PTC. Fluoroscopy is used to properly align the magnets and a hole in the center of the magnets allow a guidewire to be advanced. Recanalization can be achieved via PTC and serial biliary stenting can be performed. Magnet approximation and recanalization have been reported to be successful in 84% and 77% of patients respectively. MCA has been shown to be effective for short strictures (< 1 cm) with a low stricture recurrence rate[70,71].
Biliary strictures play a significant role in morbidity of LT recipients. There exists a variety of techniques to approach anastomotic and NAS. However, despite the use of balloon and passage dilators along with plastic and metal stents, there is no standardized method to approach intrahepatic, hilar, or extra-hepatic bile duct strictures. In addition, patients with altered surgical anatomy and increasing use of LDT add to the complexity of providing successful outcomes. Nonetheless, en-doscopic management of anastomotic and NAS are predominantly successful with relatively low complication rates. Further larger and comparative trials along with the advent of more endoscopic tools may allow for increasing rates of success and improved times till resolution of biliary strictures.
John McDermott, Department of Radiology, University of Wisconsin Hospitals and Clinics, Madison, WI 53719, United States.
Manuscript source: Invited manuscript
Specialty type: Medicine, research and experimental
Country of origin: United States
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: de Moura DTH, Hu B, Tsou YK S-Editor: Ji FF L-Editor: A E-Editor: Wu YXJ
| 1. | Girotra M, Soota K, Klair JS, Dang SM, Aduli F. Endoscopic management of post-liver transplant biliary complications. World J Gastrointest Endosc. 2015;7:446-459. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 18] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 2. | Ayoub WS, Esquivel CO, Martin P. Biliary complications following liver transplantation. Dig Dis Sci. 2010;55:1540-1546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 82] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 3. | Balderramo D, Navasa M, Cardenas A. Current management of biliary complications after liver transplantation: emphasis on endoscopic therapy. Gastroenterol Hepatol. 2011;34:107-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 4. | Lee HW, Shah NH, Lee SK. An Update on Endoscopic Management of Post-Liver Transplant Biliary Complications. Clin Endosc. 2017;50:451-463. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (1)] |
| 5. | Ostroff JW. Post-transplant biliary problems. Gastrointest Endosc Clin N Am. 2001;11:163-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 32] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 6. | Stratta RJ, Wood RP, Langnas AN, Hollins RR, Bruder KJ, Donovan JP, Burnett DA, Lieberman RP, Lund GB, Pillen TJ. Diagnosis and treatment of biliary tract complications after orthotopic liver transplantation. Surgery. 1989;106:675-683; discussion 683-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 7. | Scatton O, Meunier B, Cherqui D, Boillot O, Sauvanet A, Boudjema K, Launois B, Fagniez PL, Belghiti J, Wolff P, Houssin D, Soubrane O. Randomized trial of choledochocholedochostomy with or without a T tube in orthotopic liver transplantation. Ann Surg. 2001;233:432-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 182] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 8. | Scanga AE, Kowdley KV. Management of biliary complications following orthotopic liver transplantation. Curr Gastroenterol Rep. 2007;9:31-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 28] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 9. | Shah SA, Grant DR, McGilvray ID, Greig PD, Selzner M, Lilly LB, Girgrah N, Levy GA, Cattral MS. Biliary strictures in 130 consecutive right lobe living donor liver transplant recipients: results of a Western center. Am J Transplant. 2007;7:161-167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 156] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 10. | Greif F, Bronsther OL, Van Thiel DH, Casavilla A, Iwatsuki S, Tzakis A, Todo S, Fung JJ, Starzl TE. The incidence, timing, and management of biliary tract complications after orthotopic liver transplantation. Ann Surg. 1994;219:40-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 373] [Cited by in RCA: 357] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 11. | Thuluvath PJ, Pfau PR, Kimmey MB, Ginsberg GG. Biliary complications after liver transplantation: the role of endoscopy. Endoscopy. 2005;37:857-863. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 157] [Cited by in RCA: 153] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 12. | Pascher A, Neuhaus P. Biliary complications after deceased-donor orthotopic liver transplantation. J Hepatobiliary Pancreat Surg. 2006;13:487-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 103] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 13. | Dacha S, Barad A, Martin J, Levitsky J. Association of hepatic artery stenosis and biliary strictures in liver transplant recipients. Liver Transpl. 2011;17:849-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 14. | Brunner SM, Junger H, Ruemmele P, Schnitzbauer AA, Doenecke A, Kirchner GI, Farkas SA, Loss M, Scherer MN, Schlitt HJ, Fichtner-Feigl S. Bile duct damage after cold storage of deceased donor livers predicts biliary complications after liver transplantation. J Hepatol. 2013;58:1133-1139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 87] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 15. | Sundaram V, Jones DT, Shah NH, de Vera ME, Fontes P, Marsh JW, Humar A, Ahmad J. Posttransplant biliary complications in the pre- and post-model for end-stage liver disease era. Liver Transpl. 2011;17:428-435. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 16. | Gopal DV, Pfau PR, Lucey MR. Endoscopic Management of Biliary Complications After Orthotopic Liver Transplantation. Curr Treat Options Gastroenterol. 2003;6:509-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 16] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 17. | Mosca S, Militerno G, Guardascione MA, Amitrano L, Picciotto FP, Cuomo O. Late biliary tract complications after orthotopic liver transplantation: diagnostic and therapeutic role of endoscopic retrograde cholangiopancreatography. J Gastroenterol Hepatol. 2000;15:654-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 18. | Sotiropoulos GC, Sgourakis G, Radtke A, Molmenti EP, Goumas K, Mylona S, Fouzas I, Karaliotas C, Lang H. Orthotopic liver transplantation: T-tube or not T-tube? Systematic review and meta-analysis of results. Transplantation. 2009;87:1672-1680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 64] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 19. | López-Andújar R, Orón EM, Carregnato AF, Suárez FV, Herraiz AM, Rodríguez FS, Carbó JJ, Ibars EP, Sos JE, Suárez AR, Castillo MP, Pallardó JM, De Juan Burgueño M. T-tube or no T-tube in cadaveric orthotopic liver transplantation: the eternal dilemma: results of a prospective and randomized clinical trial. Ann Surg. 2013;258:21-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 50] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 20. | Yamaoka Y, Washida M, Honda K, Tanaka K, Mori K, Shimahara Y, Okamoto S, Ueda M, Hayashi M, Tanaka A. Liver transplantation using a right lobe graft from a living related donor. Transplantation. 1994;57:1127-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.0] [Reference Citation Analysis (0)] |
| 21. | Simoes P, Kesar V, Ahmad J. Spectrum of biliary complications following live donor liver transplantation. World J Hepatol. 2015;7:1856-1865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 47] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 22. | Maluf DG, Stravitz RT, Cotterell AH, Posner MP, Nakatsuka M, Sterling RK, Luketic VA, Shiffman ML, Ham JM, Marcos A, Behnke MK, Fisher RA. Adult living donor versus deceased donor liver transplantation: a 6-year single center experience. Am J Transplant. 2005;5:149-156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 125] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 23. | Akamatsu N, Sugawara Y, Hashimoto D. Biliary reconstruction, its complications and management of biliary complications after adult liver transplantation: a systematic review of the incidence, risk factors and outcome. Transpl Int. 2011;24:379-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 247] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 24. | Freise CE, Gillespie BW, Koffron AJ, Lok AS, Pruett TL, Emond JC, Fair JH, Fisher RA, Olthoff KM, Trotter JF, Ghobrial RM, Everhart JE; A2ALL Study Group. Recipient morbidity after living and deceased donor liver transplantation: findings from the A2ALL Retrospective Cohort Study. Am J Transplant. 2008;8:2569-2579. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 236] [Cited by in RCA: 213] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 25. | Wadhawan M, Kumar A, Gupta S, Goyal N, Shandil R, Taneja S, Sibal A. Post-transplant biliary complications: an analysis from a predominantly living donor liver transplant center. J Gastroenterol Hepatol. 2013;28:1056-1060. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 26. | Woo HY, Lee IS, Chang JH, Youn SB, Bae SH, Choi JY, Chun HJ, You YK, Kim DG, Yoon SK. Outcome of donor biliary complications following living donor liver transplantation. Korean J Intern Med. 2018;33:705-715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 27. | Gruttadauria S, Marsh JW, Vizzini GB, di Francesco F, Luca A, Volpes R, Marcos A, Gridelli B. Analysis of surgical and perioperative complications in seventy-five right hepatectomies for living donor liver transplantation. World J Gastroenterol. 2008;14:3159-3164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 48] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 28. | Verdonk RC, Buis CI, Porte RJ, van der Jagt EJ, Limburg AJ, van den Berg AP, Slooff MJ, Peeters PM, de Jong KP, Kleibeuker JH, Haagsma EB. Anastomotic biliary strictures after liver transplantation: causes and consequences. Liver Transpl. 2006;12:726-735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 236] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 29. | Albert JG, Filmann N, Elsner J, Moench C, Trojan J, Bojunga J, Sarrazin C, Friedrich-Rust M, Herrmann E, Bechstein WO, Zeuzem S, Hofmann WP. Long-term follow-up of endoscopic therapy in stenosis of the bilio-biliary anastomosis associated with orthotopic liver transplantation. Liver Transpl. 2013;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 30. | Sharma S, Gurakar A, Jabbour N. Biliary strictures following liver transplantation: past, present and preventive strategies. Liver Transpl. 2008;14:759-769. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 263] [Cited by in RCA: 275] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 31. | Verdonk RC, Buis CI, Porte RJ, Haagsma EB. Biliary complications after liver transplantation: a review. Scand J Gastroenterol Suppl. 2006;89-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 133] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 32. | Pasha SF, Harrison ME, Das A, Nguyen CC, Vargas HE, Balan V, Byrne TJ, Douglas DD, Mulligan DC. Endoscopic treatment of anastomotic biliary strictures after deceased donor liver transplantation: outcomes after maximal stent therapy. Gastrointest Endosc. 2007;66:44-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 117] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 33. | Graziadei IW, Schwaighofer H, Koch R, Nachbaur K, Koenigsrainer A, Margreiter R, Vogel W. Long-term outcome of endoscopic treatment of biliary strictures after liver transplantation. Liver Transpl. 2006;12:718-725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 147] [Cited by in RCA: 157] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 34. | Lee DW, Jo HH, Abdullah J, Kahaleh M. Endoscopic Management of Anastomotic Strictures after Liver Transplantation. Clin Endosc. 2016;49:457-461. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 35. | Zoepf T, Maldonado-Lopez EJ, Hilgard P, Malago M, Broelsch CE, Treichel U, Gerken G. Balloon dilatation vs. balloon dilatation plus bile duct endoprostheses for treatment of anastomotic biliary strictures after liver transplantation. Liver Transpl. 2006;12:88-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 128] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 36. | Morelli J, Mulcahy HE, Willner IR, Cunningham JT, Draganov P. Long-term outcomes for patients with post-liver transplant anastomotic biliary strictures treated by endoscopic stent placement. Gastrointest Endosc. 2003;58:374-379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 137] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 37. | Elmi F, Silverman WB. Outcome of ERCP in the management of duct-to-duct anastomotic strictures in orthotopic liver transplant. Dig Dis Sci. 2007;52:2346-2350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 38. | Kim TH, Lee SK, Han JH, Park DH, Lee SS, Seo DW, Kim MH, Song GW, Ha TY, Kim KH, Hwang S, Lee SG. The role of endoscopic retrograde cholangiography for biliary stricture after adult living donor liver transplantation: technical aspect and outcome. Scand J Gastroenterol. 2011;46:188-196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 46] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 39. | Tsujino T, Isayama H, Sugawara Y, Sasaki T, Kogure H, Nakai Y, Yamamoto N, Sasahira N, Yamashiki N, Tada M, Yoshida H, Kokudo N, Kawabe T, Makuuchi M, Omata M. Endoscopic management of biliary complications after adult living donor liver transplantation. Am J Gastroenterol. 2006;101:2230-2236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 114] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 40. | Rerknimitr R, Sherman S, Fogel EL, Kalayci C, Lumeng L, Chalasani N, Kwo P, Lehman GA. Biliary tract complications after orthotopic liver transplantation with choledochocholedochostomy anastomosis: endoscopic findings and results of therapy. Gastrointest Endosc. 2002;55:224-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 226] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 41. | Alazmi WM, Fogel EL, Watkins JL, McHenry L, Tector JA, Fridell J, Mosler P, Sherman S, Lehman GA. Recurrence rate of anastomotic biliary strictures in patients who have had previous successful endoscopic therapy for anastomotic narrowing after orthotopic liver transplantation. Endoscopy. 2006;38:571-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 77] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 42. | Morelli G, Fazel A, Judah J, Pan JJ, Forsmark C, Draganov P. Rapid-sequence endoscopic management of posttransplant anastomotic biliary strictures. Gastrointest Endosc. 2008;67:879-885. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 78] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 43. | Tabibian JH, Asham EH, Han S, Saab S, Tong MJ, Goldstein L, Busuttil RW, Durazo FA. Endoscopic treatment of postorthotopic liver transplantation anastomotic biliary strictures with maximal stent therapy (with video). Gastrointest Endosc. 2010;71:505-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 104] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 44. | Traina M, Tarantino I, Barresi L, Volpes R, Gruttadauria S, Petridis I, Gridelli B. Efficacy and safety of fully covered self-expandable metallic stents in biliary complications after liver transplantation: a preliminary study. Liver Transpl. 2009;15:1493-1498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 102] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 45. | Poley JW, Cahen DL, Metselaar HJ, van Buuren HR, Kazemier G, van Eijck CH, Haringsma J, Kuipers EJ, Bruno MJ. A prospective group sequential study evaluating a new type of fully covered self-expandable metal stent for the treatment of benign biliary strictures (with video). Gastrointest Endosc. 2012;75:783-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 60] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 46. | Tarantino I, Traina M, Mocciaro F, Barresi L, Curcio G, Di Pisa M, Granata A, Volpes R, Gridelli B. Fully covered metallic stents in biliary stenosis after orthotopic liver transplantation. Endoscopy. 2012;44:246-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 47. | Sauer P, Chahoud F, Gotthardt D, Stremmel W, Weiss KH, Büchler M, Schemmer P, Weitz J, Schaible A. Temporary placement of fully covered self-expandable metal stents in biliary complications after liver transplantation. Endoscopy. 2012;44:536-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 48. | Kahaleh M, Brijbassie A, Sethi A, Degaetani M, Poneros JM, Loren DE, Kowalski TE, Sejpal DV, Patel S, Rosenkranz L, McNamara KN, Raijman I, Talreja JP, Gaidhane M, Sauer BG, Stevens PD. Multicenter trial evaluating the use of covered self-expanding metal stents in benign biliary strictures: time to revisit our therapeutic options? J Clin Gastroenterol. 2013;47:695-699. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 67] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 49. | Martins FP, De Paulo GA, Contini MLC, Ferrari AP. Metal versus plastic stents for anastomotic biliary strictures after liver transplantation: a randomized controlled trial. Gastrointest Endosc. 2018;87:131.e1-131.e13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 85] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 50. | Coté GA, Slivka A, Tarnasky P, Mullady DK, Elmunzer BJ, Elta G, Fogel E, Lehman G, McHenry L, Romagnuolo J, Menon S, Siddiqui UD, Watkins J, Lynch S, Denski C, Xu H, Sherman S. Effect of Covered Metallic Stents Compared With Plastic Stents on Benign Biliary Stricture Resolution: A Randomized Clinical Trial. JAMA. 2016;315:1250-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 168] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 51. | Park DH, Lee SS, Lee TH, Ryu CH, Kim HJ, Seo DW, Park SH, Lee SK, Kim MH, Kim SJ. Anchoring flap versus flared end, fully covered self-expandable metal stents to prevent migration in patients with benign biliary strictures: a multicenter, prospective, comparative pilot study (with videos). Gastrointest Endosc. 2011;73:64-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 120] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 52. | Kao D, Zepeda-Gomez S, Tandon P, Bain VG. Managing the post-liver transplantation anastomotic biliary stricture: multiple plastic versus metal stents: a systematic review. Gastrointest Endosc. 2013;77:679-691. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 110] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 53. | Visconti TAC, Bernardo WM, Moura DTH, Moura ETH, Gonçalves CVT, Farias GF, Guedes HG, Ribeiro IB, Franzini TP, Luz GO, Dos Santos MEDL, de Moura EGH. Metallic vs plastic stents to treat biliary stricture after liver transplantation: a systematic review and meta-analysis based on randomized trials. Endosc Int Open. 2018;6:E914-E923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 54. | Guichelaar MM, Benson JT, Malinchoc M, Krom RA, Wiesner RH, Charlton MR. Risk factors for and clinical course of non-anastomotic biliary strictures after liver transplantation. Am J Transplant. 2003;3:885-890. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 217] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 55. | Koneru B, Sterling MJ, Bahramipour PF. Bile duct strictures after liver transplantation: a changing landscape of the Achilles' heel. Liver Transpl. 2006;12:702-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 79] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 56. | Roos FJM, Poley JW, Polak WG, Metselaar HJ. Biliary complications after liver transplantation; recent developments in etiology, diagnosis and endoscopic treatment. Best Pract Res Clin Gastroenterol. 2017;31:227-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 40] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 57. | Verdonk RC, Buis CI, van der Jagt EJ, Gouw AS, Limburg AJ, Slooff MJ, Kleibeuker JH, Porte RJ, Haagsma EB. Nonanastomotic biliary strictures after liver transplantation, part 2: Management, outcome, and risk factors for disease progression. Liver Transpl. 2007;13:725-732. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 117] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 58. | Rizk RS, McVicar JP, Emond MJ, Rohrmann CA, Kowdley KV, Perkins J, Carithers RL, Kimmey MB. Endoscopic management of biliary strictures in liver transplant recipients: effect on patient and graft survival. Gastrointest Endosc. 1998;47:128-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 106] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 59. | Thuluvath PJ, Atassi T, Lee J. An endoscopic approach to biliary complications following orthotopic liver transplantation. Liver Int. 2003;23:156-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 133] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 60. | Zoepf T, Maldonado de Dechêne EJ, Dechêne A, Malágo M, Beckebaum S, Paul A, Gerken G, Hilgard P. Optimized endoscopic treatment of ischemic-type biliary lesions after liver transplantation. Gastrointest Endosc. 2012;76:556-563. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 61. | Arain MA, Attam R, Freeman ML. Advances in endoscopic management of biliary tract complications after liver transplantation. Liver Transpl. 2013;19:482-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 59] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 62. | Yazumi S, Yoshimoto T, Hisatsune H, Hasegawa K, Kida M, Tada S, Uenoyama Y, Yamauchi J, Shio S, Kasahara M, Ogawa K, Egawa H, Tanaka K, Chiba T. Endoscopic treatment of biliary complications after right-lobe living-donor liver transplantation with duct-to-duct biliary anastomosis. J Hepatobiliary Pancreat Surg. 2006;13:502-510. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 103] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 63. | Shah RJ, Smolkin M, Yen R, Ross A, Kozarek RA, Howell DA, Bakis G, Jonnalagadda SS, Al-Lehibi AA, Hardy A, Morgan DR, Sethi A, Stevens PD, Akerman PA, Thakkar SJ, Brauer BC. A multicenter, U.S. experience of single-balloon, double-balloon, and rotational overtube-assisted enteroscopy ERCP in patients with surgically altered pancreaticobiliary anatomy (with video). Gastrointest Endosc. 2013;77:593-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 228] [Cited by in RCA: 200] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 64. | Kim JH, Lee SK, Kim MH, Song MH, Park DH, Kim SY, Lee SS, Seo DW, Bae JS, Kim HJ, Han J, Sung KB, Min YI. Percutaneous transhepatic cholangioscopic treatment of patients with benign bilio-enteric anastomotic strictures. Gastrointest Endosc. 2003;58:733-738. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 65. | Uchida D, Kato H, Saragai Y, Takada S, Muro S, Tomoda T, Matsumoto K, Horiguchi S, Okada H. Usefulness of a Cannula with a Flexible Tip (Swing Tip) for Managing Severe Biliary Stricture. Can J Gastroenterol Hepatol. 2018;2018:7125714. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 66. | Laasch HU, Tringali A, Wilbraham L, Marriott A, England RE, Mutignani M, Perri V, Costamagna G, Martin DF. Comparison of standard and steerable catheters for bile duct cannulation in ERCP. Endoscopy. 2003;35:669-674. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 31] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 67. | Balderramo D, Sendino O, Miquel R, de Miguel CR, Bordas JM, Martinez-Palli G, Leoz ML, Rimola A, Navasa M, Llach J, Cardenas A. Prospective evaluation of single-operator peroral cholangioscopy in liver transplant recipients requiring an evaluation of the biliary tract. Liver Transpl. 2013;19:199-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 68. | Bukhari MA, Haito-Chavez Y, Ngamruengphong S, Brewer Gutierrez O, Chen YI, Khashab MA. Rendezvous Biliary Recanalization of Complete Biliary Obstruction With Direct Peroral and Percutaneous Transhepatic Cholangioscopy. Gastroenterology. 2018;154:23-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 69. | Gürakar A, Wright H, Camci C, Jaboour N. The application of SpyScope® technology in evaluation of pre and post liver transplant biliary problems. Turk J Gastroenterol. 2010;21:428-432. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 70. | Jang SI, Kim JH, Won JY, Lee KH, Kim HW, You JW, Itoi T, Lee D. Magnetic compression anastomosis is useful in biliary anastomotic strictures after living donor liver transplantation. Gastrointest Endosc. 2011;74:1040-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 71. | Parlak E, Koksal AS, Kucukay F, Eminler AT, Toka B, Uslan MI. A novel technique for the endoscopic treatment of complete biliary anastomosis obstructions after liver transplantation: through-the-scope magnetic compression anastomosis. Gastrointest Endosc. 2017;85:841-847. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |