Peer-review started: April 9, 2020
First decision: April 18, 2020
Revised: April 19, 2020
Accepted: May 12, 2020
Article in press: May 12, 2020
Published online: May 25, 2020
Processing time: 42 Days and 21.5 Hours
Patients with coronavirus disease 2019 (COVID-19) predominantly present with the pulmonary symptoms such as fever, cough, and shortness of breath. We present a case of an 83 years old patient with COVID-19 who presented with only gastrointestinal symptoms without respiratory complaints. Our case raises the concern regarding our current lack of understanding of extrapulmonary manifestations of COVID-19. Given genetic homology between 2019 severe acute respiratory syndrome coronavirus (SARS-CoV) 2 and SARS-CoV, our case underscores the urgent need for further studies to understand the role of the gastrointestinal system in 2019 SARS-CoV-2 transmission and COVID-19 pathogenesis.
Core tip: We present a case of an 83 years old patient with coronavirus disease 2019 (COVID-19) who presented with only gastrointestinal symptoms without respiratory complaints. Our case raises the concern regarding our current lack of understanding of extrapulmonary manifestations of COVID-19 and underscores the urgent need for further studies to understand the role of the gastrointestinal system in 2019 severe acute respiratory syndrome coronavirus 2 transmission and COVID-19 pathogenesis.
- Citation: Kant R, Chandra L, Antony MA, Verma V. Case of COVID-19 presenting with gastrointestinal symptoms. World J Virol 2020; 9(1): 1-4
- URL: https://www.wjgnet.com/2220-3249/full/v9/i1/1.htm
- DOI: https://dx.doi.org/10.5501/wjv.v9.i1.1
Coronavirus disease 2019 (COVID-19) pandemic has resulted in a public health emergency unlike we have seen in generations. Patients with COVID-19 predominantly present with pulmonary symptoms such as fever, cough, and shortness of breath[1]. We present a case of COVID-19 who presented with only gastrointestinal (GI) symptoms without any respiratory complaints.
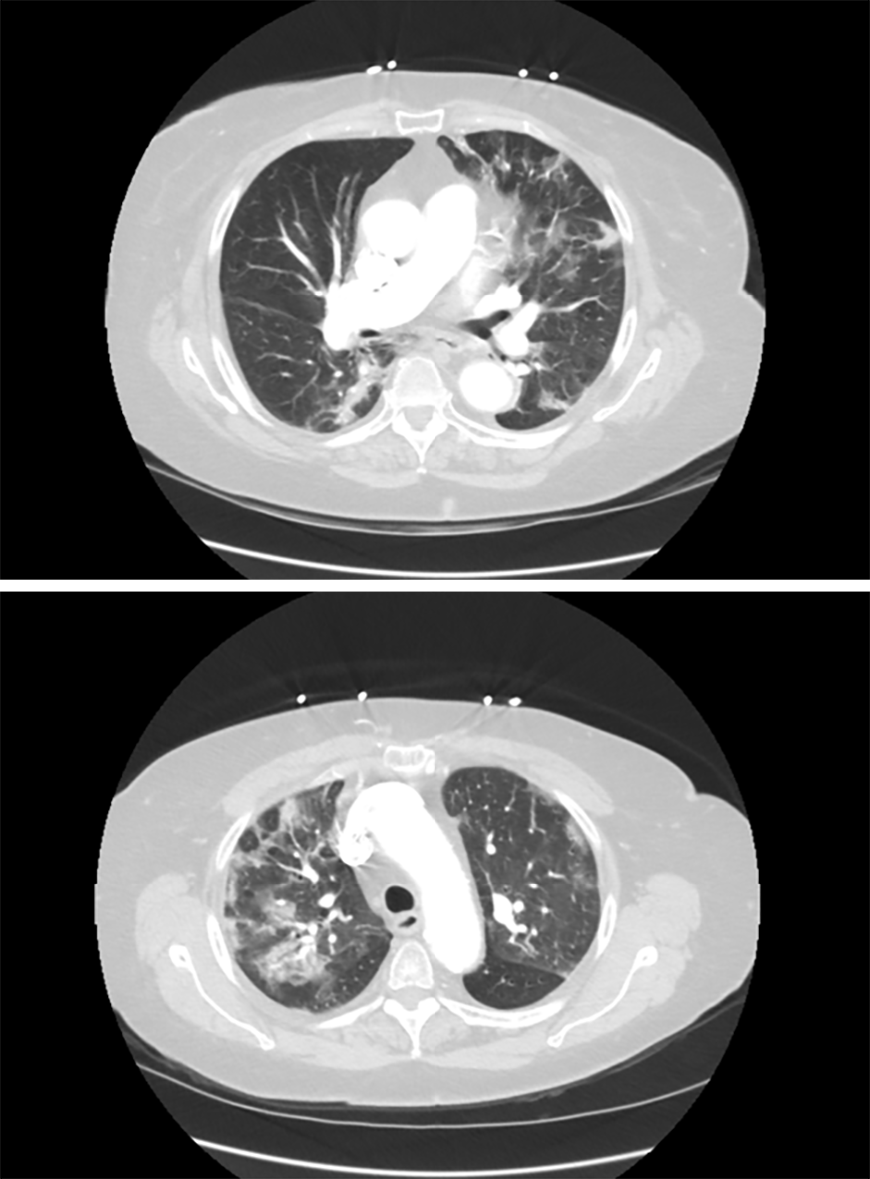
An 83 years old African American woman with a prior history of hypertension, type-2 diabetes mellitus and chronic kidney disease presented to the emergency department with diarrhea and nausea for 3 d. The patient reported 3-5 episodes/d of watery, non-mucoid, and non-bloody diarrhea. Her GI symptoms progressively worsened warranting a visit to the emergency department. The patient denied fever, cough, chills, rigors, night sweats, headache, sore throat, rhinorrhea, dyspnea, orthopnea, recent travel or sick contact. Patient was hemodynamically stable [blood pressure 116/56 mmHg, heart rate 63 beats per minute, respiratory rate 19 per minute, temperature 100.3 ℉, and oxygen saturation (O2 sat) 94%], but noted to have O2 sat of 90%-91% on room air (RA) with ambulation. Labs showed normal white blood cells of 5.6 K/μL (Ref: 4.3 -11.1 K/μL) with differential showing normal lymphocyte of 25% (Ref: 13%-44%) and procalcitonin level of 0.10 ng/mL (Ref: ≤ 0.50 ng/mL). On further evaluation, chest X-ray revealed evidence of multifocal opacities suspicious for pneumonia. Computed tomography chest, abdomen and pelvis showed bilateral ground glass opacities (Figure 1). No acute pathologic findings were identified in the abdomen or pelvis including colitis, diverticulitis, appendicitis or small bowel obstruction.
Patient was admitted under droplet, contact and airborne isolation. Given high suspicion of COVID-19 with superimposed community bacterial pneumonia, she was treated with intravenous ceftriaxone (1 mg daily) and azithromycin (500 mg daily) for 7 d. Meanwhile, emergent disease panel (reverse transcription polymerase chain reaction on nasopharyngeal and oropharyngeal swab) performed by the Department of Health and Environmental Control tested positive for COVID-19. During the hospital stay, patient stayed afebrile without any respiratory distress. She had a benign hospital course and was discharged after 7 d of intravenous antibiotics. Her O2 sat was 98% on RA at rest and 94%-96% on RA with ambulation, and the GI symptoms resolved by discharge.
Our case had a unique presentation with digestive symptoms and raises the concern regarding our current lack of understanding of extrapulmonary manifestations of COVID-19. Diagnosis of COVID-19 cases with predominant extrapulmonary symptoms may be delayed as these symptoms currently may not be on the clinician’s radar. A recent report on 204 patients positive for COVID-19 in Hubei, China showed that approximately 18% of patients had digestive symptoms such as abdominal pain, diarrhea or vomiting[2]. In fact, 6 patients presented just with GI symptoms without respiratory complaints, similar to our patient. In addition, patients with GI symptoms had a longer time from illness onset to admission compared to patients without GI symptoms (8.95 ± 5.40 d vs 7.26 ± 4.20 d)[2].
Genome sequences of 2019 severe acute respiratory syndrome coronavirus 2 (2019 SARS-CoV-2) have demonstrated 79% sequence identity with the severe acute respiratory syndrome-like coronaviruses (SARS-CoV) and 50% sequence identity with the Middle East respiratory syndrome-like coronaviruses[3]. Patients infected with SARS-CoV and Middle East respiratory syndrome-like coronaviruses are known to have digestive symptoms[4-6]. Similar to SARS-CoV, the 2019 SARS-CoV-2 uses the angiotensin-converting enzyme 2 receptors[7]. Presence of angiotensin-converting enzyme 2 protein on small intestine erythrocytes along with epithelia of lung also supports the hypothesis that SARS-CoV-2 may shed through GI tract[8]. Moreover, SARS-CoV-2 was present on fecal swabs and blood samples from patients positive for COVID-19[9]. Intestinal biopsy specimens of patients with SARS have also shown active viral replication suggesting the role of fecal-oral route in SARS-CoV transmission[10].
Several antiviral and antimicrobial drugs such as hydroxychloroquine, remdesivir, lopinavir-ritonavir, and favipiravir are currently being studied for efficacy in patients with COVID-19 but at present, no drug is approved by United States Food and Drug Administration for COVID-19[11,12]. Supportive treatment is the mainstay of COVID-19 management. Azithromycin has been used in some protocols as adjunct therapy, and may benefit from its immunomodulatory properties and/or by preventing bacterial superinfection[11].
Given genetic homology and other similarities between 2019 SARS-CoV2 and SARS-CoV, our case underscores the urgent need for further studies to understand the role of the GI system in 2019 SARS-CoV-2 transmission and COVID-19 pathogenesis. During these unprecedented times, where the primary focus is to screen patients with respiratory symptoms for COVID-19, our case also underscores the importance of maintaining a low threshold of suspicion for COVID-19 in patients presenting with GI symptoms which will not only help with early diagnosis and intervention but may also reduce its associated morbidity and mortality.
Manuscript source: Unsolicited manuscript
Specialty type: Virology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): E
P-Reviewer: Chen YD, Rajcani J, Toyoda T S-Editor: Tang JZ L-Editor: A E-Editor: Li X
| 1. | Arentz M, Yim E, Klaff L, Lokhandwala S, Riedo FX, Chong M, Lee M. Characteristics and Outcomes of 21 Critically Ill Patients With COVID-19 in Washington State. JAMA. 2020;. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1511] [Cited by in F6Publishing: 1601] [Article Influence: 400.3] [Reference Citation Analysis (0)] |
| 2. | Pan L, Mu M, Yang P, Sun Y, Wang R, Yan J, Li P, Hu B, Wang J, Hu C, Jin Y, Niu X, Ping R, Du Y, Li T, Xu G, Hu Q, Tu L. Clinical Characteristics of COVID-19 Patients With Digestive Symptoms in Hubei, China: A Descriptive, Cross-Sectional, Multicenter Study. Am J Gastroenterol. 2020;115:766-773. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1160] [Cited by in F6Publishing: 1160] [Article Influence: 290.0] [Reference Citation Analysis (0)] |
| 3. | Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, Wang W, Song H, Huang B, Zhu N, Bi Y, Ma X, Zhan F, Wang L, Hu T, Zhou H, Hu Z, Zhou W, Zhao L, Chen J, Meng Y, Wang J, Lin Y, Yuan J, Xie Z, Ma J, Liu WJ, Wang D, Xu W, Holmes EC, Gao GF, Wu G, Chen W, Shi W, Tan W. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565-574. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 7915] [Cited by in F6Publishing: 7344] [Article Influence: 1836.0] [Reference Citation Analysis (0)] |
| 4. | Chiu YC, Wu KL, Chou YP, Fong TV, Tsai TL, Kuo CM, Kuo CH, Chiu KW, Liu JW, Eng HL, Jawan B, Cheng YF, Chen CL. Diarrhea in medical care workers with severe acute respiratory syndrome. J Clin Gastroenterol. 2004;38:880-882. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 8] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 5. | Assiri A, Al-Tawfiq JA, Al-Rabeeah AA, Al-Rabiah FA, Al-Hajjar S, Al-Barrak A, Flemban H, Al-Nassir WN, Balkhy HH, Al-Hakeem RF, Makhdoom HQ, Zumla AI, Memish ZA. Epidemiological, demographic, and clinical characteristics of 47 cases of Middle East respiratory syndrome coronavirus disease from Saudi Arabia: a descriptive study. Lancet Infect Dis. 2013;13:752-761. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 969] [Cited by in F6Publishing: 1010] [Article Influence: 91.8] [Reference Citation Analysis (0)] |
| 6. | Dawson P, Malik MR, Parvez F, Morse SS. What Have We Learned About Middle East Respiratory Syndrome Coronavirus Emergence in Humans? A Systematic Literature Review. Vector Borne Zoonotic Dis. 2019;19:174-192. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 35] [Cited by in F6Publishing: 39] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 7. | Guo YR, Cao QD, Hong ZS, Tan YY, Chen SD, Jin HJ, Tan KS, Wang DY, Yan Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak - an update on the status. Mil Med Res. 2020;7:11. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1854] [Cited by in F6Publishing: 1967] [Article Influence: 491.8] [Reference Citation Analysis (0)] |
| 8. | Hamming I, Timens W, Bulthuis ML, Lely AT, Navis G, van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J Pathol. 2004;203:631-637. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 3643] [Cited by in F6Publishing: 4021] [Article Influence: 201.1] [Reference Citation Analysis (0)] |
| 9. | Zhang W, Du RH, Li B, Zheng XS, Yang XL, Hu B, Wang YY, Xiao GF, Yan B, Shi ZL, Zhou P. Molecular and serological investigation of 2019-nCoV infected patients: implication of multiple shedding routes. Emerg Microbes Infect. 2020;9:386-389. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 1192] [Cited by in F6Publishing: 1202] [Article Influence: 300.5] [Reference Citation Analysis (0)] |
| 10. | Leung WK, To KF, Chan PK, Chan HL, Wu AK, Lee N, Yuen KY, Sung JJ. Enteric involvement of severe acute respiratory syndrome-associated coronavirus infection. Gastroenterology. 2003;125:1011-1017. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 127] [Cited by in F6Publishing: 338] [Article Influence: 16.1] [Reference Citation Analysis (1)] |
| 11. | Smith T, Bushek J, LeClaire A, Prosser T. COVID-19 Drug Therapy – Potential Options. Elsevier ©, 2020. Available from: http://www.aesculapseguridaddelpaciente.org.mx/alianzapsqx/docs/informacioncientifica/clinicadiagnostico/COVID-19-Drug-Therapy_Mar-2020.pdf. [Cited in This Article: ] |
| 12. | Singhal T. A Review of Coronavirus Disease-2019 (COVID-19). Indian J Pediatr. 2020;87:281-286. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 2006] [Cited by in F6Publishing: 1506] [Article Influence: 376.5] [Reference Citation Analysis (0)] |