Revised: November 30, 2012
Accepted: January 5, 2013
Published online: March 9, 2013
Diabetic retinopathy (DR) and age-related macular degeneration (AMD) are the leading causes of blindness in adults. The impact of these conditions on the quality of life is increasing in significance with a rise in life expectancy. The role of hyperglycemia, oxidative stress and inflammatory responses in the development and/or progression of DR and AMD, and several other sight threatening ocular diseases, is well established. In proliferative retinopathy, signals sent by the retina for nourishment, triggers the growth of fragile and abnormal blood vessels. Changes in ocular pressure may lead to rupture of these blood vessels causing severe vision problems. Recent in vitro and preclinical studies demonstrate that certain phytochemicals possessing potent antioxidant and anti-inflammatory activity and ocular blood flow enhancing properties may be very useful in the treatment of, or as a prophylactic measure for, DR and AMD. Apart from these properties they have also been investigated for their anti-bacterial, hormonal, enzyme stimulation, and anti-angiogenic activities. The attractive aspect of these potential therapeutic candidates is that they can act on multiple pathways identified in the etiology of DR, AMD, cataract and other ocular diseases. However, results from clinical trials have been somewhat ambiguous, raising questions about the concentrations of these bioflavonoids achieved in the neural retina following oral administration. Unfortunately, as of date, an efficient noninvasive means to deliver therapeutic agents/candidates to the back-of-the eye is still not available. This review examines some of these promising natural agents and discusses the challenges encountered in delivering them to the posterior segment ocular tissues through the oral route.
- Citation: Adelli GR, Srirangam R, Majumdar S. Phytochemicals in ocular health: Therapeutic potential and delivery challenges. World J Pharmacol 2013; 2(1): 18-34
- URL: https://www.wjgnet.com/2220-3192/full/v2/i1/18.htm
- DOI: https://dx.doi.org/10.5497/wjp.v2.i1.18
A report from the World Health Organization (WHO) in 2011 estimated that approximately 285 million people were suffering from visual impairment worldwide out of which 39 million face blindness while 246 million suffer from moderate to severe vision impairment. With an increase in the average survival age and the percentage of diabetics, diabetes related retinopathies are rapidly gaining in significance. It has been predicted that, without additional steps, these numbers will increase to 75 million blind and 200 million visually impaired by the year 2020 (WHO, 2010). Age seems to be a causative factor in blindness as 90% and 58% of the population with blindness are aged above 45 years and 60 years respectively.
Oxidative stress, inflammatory mechanisms and decreased antioxidant capacity in the ocular tissues are all thought to play an active role in the development and progression of these ocular diseases. The following sections briefly highlight the etiology of these ocular disorders, followed by discussions on the therapeutic potential of various phytochemicals and challenges encountered in their delivery to the ocular tissues.
Diabetic retinopathy (DR) is the most common diabetes associated eye disease and is the leading cause of blindness in American adults[1,2]. The WHO estimates that over 360 million worldwide will suffer from diabetes by the year 2030. Currently, 10% of the diabetics suffer from type 1 diabetes whereas 90% suffer from type 2 diabetes. DR in type 1 diabetics approaches 80% and 90 % prevalence rate after 10 years and 20 years of diabetes, respectively. Up to 21% of patients with type 2 diabetes have recently been found to have retinopathy at the time of first diagnosis of diabetes, and most develop some degree of retinopathy over subsequent decades[3].
DR can be broadly categorized into three stages; background DR, pre-proliferative DR and proliferative DR (PDR)[4-6]. In background DR, hyperglycemia is considered to induce thickening of capillary basement membranes and death of pericytes, which support the vessel wall, and endothelial cells of retinal blood vessels. Microaneurysms and vascular leakage follow, and blockage of retinal capillaries take place. In pre-proliferative DR, loss of vascular patency leads to areas of increasing retinal hypoxia and multiple hemorrhages. Increased areas of tissue non-perfusion stimulate the production of angiogenic factors leading to the proliferation of vessels, which is a typical feature of PDR. The newly formed blood vessels by themselves do not lead to vision loss, but leakage of blood through their weak walls can result in severe vision loss and can ultimately lead to complete loss of sight.
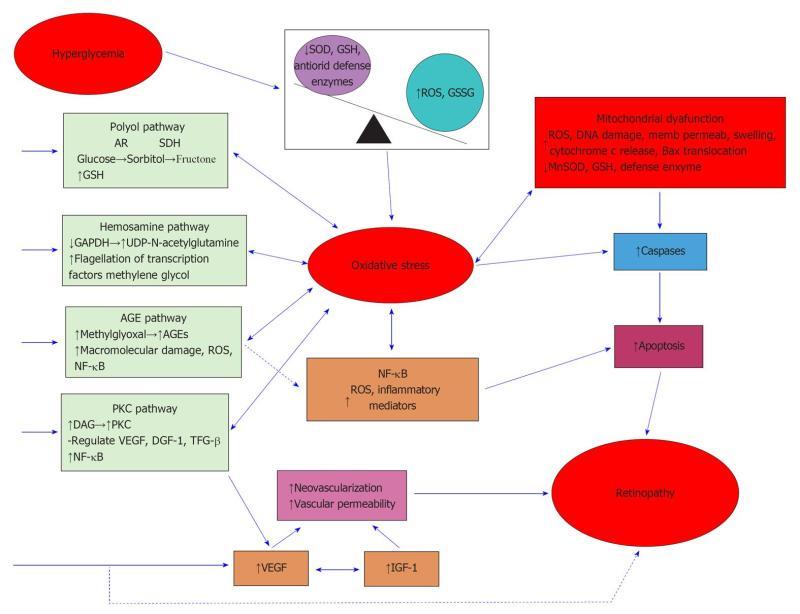
Hyperglycemia and tissue hypoxia, are considered to be principal factors in the DR pathology described above. Multiple studies demonstrate the relationship between high blood glucose, oxidative stress and initiation of DR[6-11] as shown in Figure 1. The retina is highly susceptible to oxidative damage since: (1) it has an abundance of the polyunsaturated fatty acids in its membrane bilayers, whose double bonds are prime targets for peroxidation reactions; (2) it is periodically subjected to continuous light which, by photoexcitation, can initiate free radical formation and peroxidation reactions; and (3) the retina is a highly metabolic tissue that requires a high rate of blood flow in order to receive an adequate oxygen supply. Oxidative damage in biological systems occurs when endogenous antioxidant mechanisms are overwhelmed by free oxygen radicals or reactive oxygen species (ROS). These radicals are extremely unstable and can cause cytotoxicity and cellular damage by reacting with plasma membrane lipids, DNA, RNA and metal-containing compounds[12].
Multiple pathways have been suggested to be activated during hyperglycemia associated oxidative stress, which subsequently leads to damage of retinal blood vessels. These include nitric oxide (NO) synthesis[13,14], nuclear factor-kappa B (NF-κB) expression, secretion of cytokines such as interleukin (IL)-1β, lipid peroxide generation, activation of retinal caspase-3, protein kinase C (PKC) stimulation and alterations of retinal glutamate levels[12,15-20].
In advanced stages, i.e., PDR, reduced retinal blood flow induces retinal ischemia which leads to hypoxic conditions in the retina. Hypoxia stimulates production of a variety of local agents including vascular endothelial growth factor (VEGF)[6,21-25], prostaglandins (PGs), cyclooxygenase-2 (COX-2) and NO, all of which participates in increasing vascular permeability and angiogenesis[6]. The VEGF family plays a key role in the regulation of vascular patency and is involved in both physiological and pathological angiogenesis, stimulating endothelial cells to migrate, proliferate and form tubes[6,26-29]. VEGF is also a potent vascular bed permeability enhancer[30,31]. Hypoxia is a stimulant for COX-2 transcription also in various tissues[6,32-34], including human vascular endothelium[6,32] and neural cells[35]. NF-κB, an oxygen sensitive transcription factor[6,36,37] mediates the induction of COX-2 in hypoxic conditions. The angiogenic properties of COX-2 are likely to directly involve VEGF, as COX-2 has been shown to up-regulate VEGF synthesis which can be inhibited with selective or non-selective COX inhibitors[6,38,39].
In the context of DR, the actions of PGs E2 and I2 are also considered to be important in the development of angiogenesis, breakdown of the blood-retinal barrier and alterations in retinal blood flow[6,40-45]. These PGs are produced via the COX-2 pathway[6,46-48]. Prostaglandins, and in particular PGE2, are also strong inducers of VEGF in cell types such as synovial fibroblasts monocytes and lung and retinal Müller cells. There is evidence that the vascular events prior to angiogenesis may involve the induction of COX-2 followed by VEGF. It has thus been suggested that angiogenesis may be mediated by dual interdependent gene expression pathways that involve COX-2 and VEGF[6,49].
NO is known to participate in vascular permeability and angiogenesis via interactions with VEGF and COX-2. NO reacts with superoxide anions to form peroxynitrite, a highly reactive oxidant. Excess production of peroxynitrite in diabetes may exhibit cytotoxic effects by increasing DNA damage, stimulating lipid peroxidation and depleting glutathione levels[50-52]. Peroxynitrite alters tyrosine in proteins to form nitrotyrosine and nitration of proteins can inactivate mitochondrial and cytosolic proteins and damage cellular elements leading to nitrative stress[53]. PDR is the most damaging stage of DR as it leads to the generation of abnormally located retinal blood vessels with weak capillary walls. Microvascular leakage from these newly formed blood vessels can lead to total blindness through a variety of mechanisms.
In addition to hyperglycemia associated increased ROS generation, it has recently been demonstrated that the total antioxidant capacity (TAC) of the vitreous humor and aqueous humor is lowered in DR. Mancino et al[54] determined the antioxidant capacity of blood, aqueous humor and vitreous of controls (non-diabetic) and of patients with non-proliferative DR (NPDR) and with PDR. The authors observed that the control group displayed significantly higher TAC levels than the diabetic sub-groups in both the vitreous and aqueous humor. PDR patients had decreased TACs in the vitreous and aqueous humor as compared with control subjects and with the NPDR patient subgroup, pointing to the role of oxidative stress in the progression of NPDR to PDR[54]. The results strongly support the need for increased antioxidant levels in the retina, aqueous humor and vitreous humor.
Age-related macular degeneration (AMD), which can be categorized into dry and wet AMD, is the leading cause of irreversible vision loss in the developed world. As per the statistical data published by the National Eye Institute, AMD affected 1.75 million persons in the United States, in 2004, a number which is expected to rise to nearly 3 million by 2020 due to the aging of the population[55]. Like DR, progression of AMD is linked to the activation of inflammatory and immunological pathways[56]. Presence of excess ROS and decreased antioxidant capacity in the ocular tissues is also considered to play a significant role in the initiation and progression of AMD[57,58]. Ding et al[59] provides a summary of the molecular pathways involving inflammation, angiogenesis and oxidative stress, considered to play a role in the development of AMD.
Oxidative stress induced damage to the lens fibers has also been well documented. It is thought that these free radicals accelerate and aggravate cataract development. Additionally, diabetic lenses show an impaired antioxidant capacity that increases their susceptibility to oxidative stress. The loss of antioxidants is exacerbated by glycation and inactivation of lens antioxidant enzymes like superoxide dismutase[60].
During the first two stages of DR blood sugar, pressure, and cholesterol control is recommended. Additionally, antioxidant therapy has shown reasonable promise. Free radical scavenging activity of the antioxidants protects the retinal blood vessel endothelial cells and pericytes from apoptosis in a high glucose and oxygen rich environment, as in the retina[61-67]. PDR is treated with laser surgery. This procedure is called scatter laser treatment. Although some loss of peripheral vision may be noticed, scatter laser treatment can save the remaining sight. Scatter laser treatment works better before the fragile, new blood vessels have started to bleed. Even if bleeding has started, scatter laser treatment may still be possible, depending on the amount of bleeding. If the bleeding is severe, a surgical procedure called a vitrectomy may be needed. Recent studies have demonstrated that VEGF, PKC and COX-2 inhibitors, antibodies and proteins may be effective in controlling PDR[61-66,68-77]. Anti-VEGF injection therapy, photodynamic therapy using intravenous verteporfin and sometimes laser surgery are used to treat and control the progression of AMD. High doses of antioxidants and zinc have also been shown to be useful in AMD[78].
A major shortcoming of the current therapeutic options is that no one candidate appears to be capable of acting on the multiple pathways involved in the initiation and progression of these ocular diseases. Another major challenge is achieving therapeutic concentrations of the active ingredients in the neural retina, where significant damage to the retinal blood vessels and neuronal cells occur. Identification and targeted delivery of compounds that can act on multiple pathways would be a significant advancement in the prevention and treatment of these sight-threatening ocular diseases.
Over the century, flavonoids or bioflavonoids have been identified as the most common group of plant polyphenols that give color and flavor to fruits and vegetables. Till now more than 8000 polyphenolic compounds have been identified and these flavonoids can be classified into different subclasses which include flavones, flavonols, flavanones, flavanols, anthocyanins and isoflavones[79]. Flavonoids have gained prominence in the pharmaceutical arena by virtue of their therapeutically beneficial properties. Bioflavonoids possess antioxidant, anti-angiogenic, and/or anti-inflammatory activities and are also capable of reducing fluid retention and strengthening capillary walls. Interestingly, the etiology of most ocular diseases involve free radical mediated oxidative damage, hypoxia, decreased blood supply to ocular tissues and, in certain conditions, angiogenesis, increased vascular permeability and leakage of vascular contents[80,81]. Thus, select bioflavonoids may be effective in the prevention or treatment of ocular diseases (e.g., DR and macular degeneration) that lead to vision loss if left untreated.
The following section briefly describes various pharmacological activities of a few bioflavonoids that may be useful in the prevention or treatment of DR and AMD and other ocular diseases.
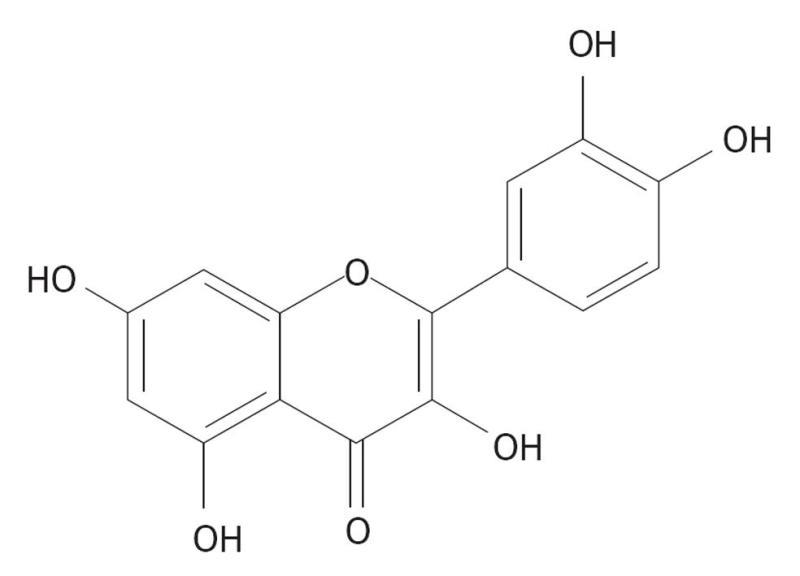
Hesperidin is a flavanone glycoside consisting of the flavanone hesperitin and the disaccharide rutinose. Hesperidin is classified as a citrus flavonoid and is the predominant flavonoid in lemons and oranges. The peel and membranous parts of these fruits have the highest hesperidin concentrations. Therefore, orange juice containing pulp is richer in the flavonoid than that without pulp. Sweet oranges (Citrus sinensis) and tangelos are the richest dietary sources of hesperidin. Hesperidin is metabolized to its aglycone, hesperetin. Figure 2 depicts the structures of hesperidin and hesperetin.
Like some of the other flavonoids, hesperidin and its aglycone hesperetin, has been reported to possess significant radical scavenging, antioxidant and neuroprotective properties[82]. These compounds have been demonstrated to prevent lipopolysaccharide, peroxynitrite, and various other free radical, e.g., azobisisobutyronitrile and benzoylperoxide, mediated cytotoxic effect[83-90]. Additionally, both hesperidin and hesperetin have been observed to exhibit anti-inflammatory activity mediated through the inhibition of the COX-2 pathway, synthesis of PGE2 and nitrogen dioxide, metabolism of arachidonic acid as well as histamine release[84,87,91-94].
What makes hesperidin and hesperetin particularly attractive is their effect on ocular blood flow and vascular permeability. Chiou et al[95] demonstrated that hesperidin, and especially hesperetin, produces marked increase in ocular blood flow and retinal function recovery following retinal ischemia. Additionally, these compounds have been demonstrated to be effective in the treatment of chronic venous insufficiency[96,97]. Reports also suggest that hesperidin can prevent microvascular leakage through their capillary wall strengthening action: hesperidin methyl chalcone, given intravenously significantly inhibited the macromolecular permeability-increasing effect of bradykinin, LTB4, and histamine[98]. Furthermore, hesperidin and hesperitin can reduce platelet aggregation, a factor involved in the blockage of retinal blood vessels[99,100].
Thus, hesperidin and its aglycone hesperetin appear to be capable of modulating multiple pathways involved in the generation and progression of DR and AMD. These compounds can protect against free-radical induced damage to the retinal neuronal cells, protect the health of retinal vascular cells, inhibit inflammatory mechanisms that can lead to the induction of angiogenesis and also prevent microvascular leakage of the retinal blood vessels. Taken together, hesperidin may prove to be a very important therapeutic candidate in the treatment of DR as well as cataract, AMD and ocular tumors through similar mechanisms.
Quercetin, abundantly found in red wine, grapes and other fruits, is one of the most studied flavonoid for its beneficial effects. Quercetin is the aglycone (Figure 3) form of a number of other flavonoid glycosides, such as rutin and quercitrin, found in citrus fruit, buckwheat and onions. Quercetin forms the glycosides quercitrin and rutin with rhamnose and rutinose, respectively.
An extensive amount of in vitro and in vivo animal research has focused on the antioxidant potential of quercetin[101-106]. Recently, its antioxidant activity was studied on retinal cell lines; Hanneken et al[107] investigated quercetin’s ability to protect ARPE-19 and human retinal pigment epithelium (RPE) cells from oxidative stress induced death in vitro. It was found that quercetin exhibited good efficacy, high potency and low toxicity in RPE cells and importantly, it was observed to be effective even after the RPE cells were exposed to oxidative stress, but before cell death occurred. There are a few other investigations that demonstrate the protective effect of quercetin on retinal cell lines[108-110].
Quercetin is also capable of increasing the choroidal blood flow and possesses anti-angiogenic activity. Zhuang et al[111] reported that quercetin inhibited the formation of choroidal neovascularization both in vivo and in vitro and increased choroidal blood flow. Quercetin was studied for its anti-angiogenic activity and was found to inhibit retinal and choroidal angiogenesis in the rhesus choroid-retina endothelial cell line, RF/6A. Quercetin prevented endothelial cell proliferation, migration, and tube formation in a dose dependent manner[112]. Quercetin’s anti-angiogenic activity was thought to be mediated through the inhibition of matrix metalloproteinases (MMP)-2 activation[113]. Other reports also substantiate the anti-angiogenic activity of quercetin[114,115]. However, it has also been reported that one of the metabolites of quercetin has an opposite effect. Quercetin and quercetin-3’-glucuronide were found to inhibit the VEGF receptor-2 but quercetin-3’-sulphate stimulated the VEGF receptor-2[116].
A number of reports also indicate anti-inflammatory activity of quercetin in vivo in animal models[106,117-119]. It’s anti-inflammatory activity was reported to be through the inhibition of COX-2[120,121], iNOS expression[122,123], tumor necrosis factor (TNF)-α, IL-1β, IL-6 and IL-8[124]. Quercetin has also been studied for its positive effect on cataract[125-128].
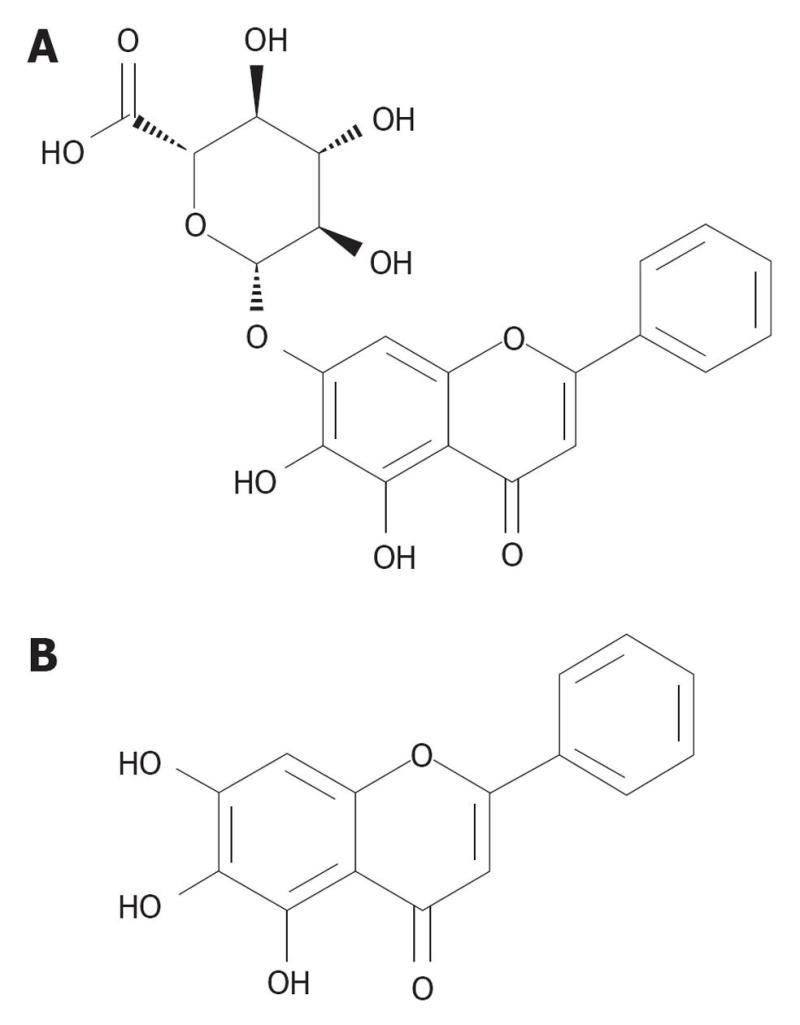
Both baicalin (7-glucuronic acid 5,6-dihydroxyflavone) and its aglycone, baicalein, are known for its strong anti-oxidant properties, anti-inflammatory properties, anti-viral properties, anti-cancer properties, and scavenging potential (Figure 4)[129].
Some recently published work suggests the usefulness of baicalin in countering both diabetes-related complications and metabolic disorders[130,131]. It possesses antioxidant, anti-angiogenic and anti-inflammatory activities. Ling et al[132] reported marked inhibition of angiogenesis, decreased migration of human umbilical vein endothelial cells and reduced VEGF induced new blood vessel growth by baicalein. In another study involving retinal vessels; baicalein treatment ameliorated inflammatory mediators in the retina like IL-18, TNF-α, and IL-1β in the rats with DR, and reduced the glial fibrillary acidic protein and VEGF expression by Muller cells, and significantly reduced vascular abnormality and ganglion cell loss within the retina[133]. Reports also suggest that baicalein is capable of suppressing IL-6 and IL-8 expression in ARPE-19 cell lines[134] and to protect the retinal ganglion cells (RGC) from oxidative stress[109] and ischemia induced[110] cell death. Similar results were observed with ARPE-19 and human RPE cells[107]. Protection of retinal cells against oxidative stress and ischemia/reperfusion (I/R) in vivo were reported for baicalein[135]. Liu et al[105] reported the antioxidant activity of baicalein on human retinal pigment epithelium cells, it was found to down regulate the levels of VEGF and MMP-9.
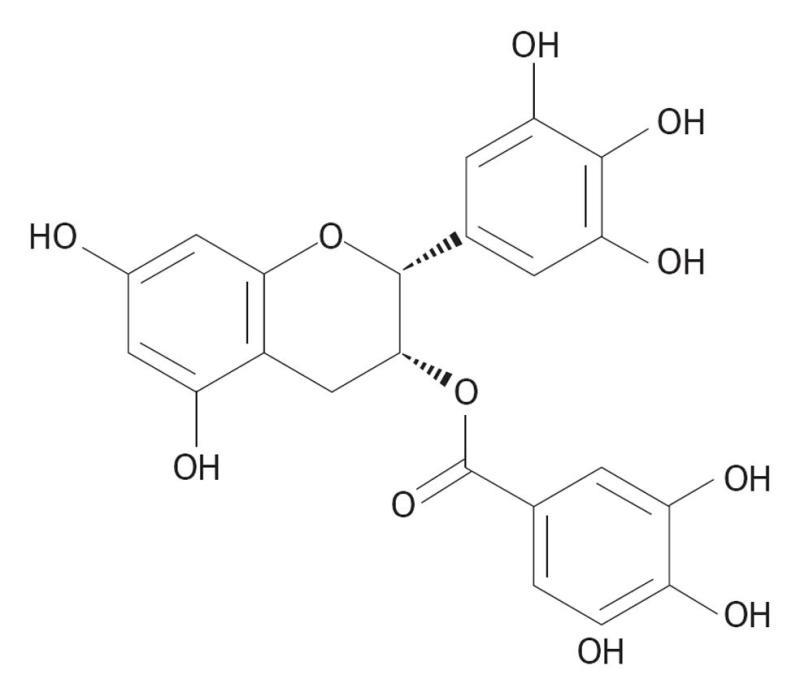
Epigallocatechin gallate (EGCG), also known as epigallocatechin 3-gallate, is the ester of epigallocatechin and gallic acid (Figure 5), and is a type of catechin. EGCG is the most abundant catechin in tea; it is found in green- but not black-tea and is a potent antioxidant that may have therapeutic applications in the treatment of many disorders.
Hanneken et al[107] evaluated the ability of specific dietary and synthetic flavonoids to protect ARPE-19 and human RPE cells from oxidative stress induced death in vitro. Oxidative stress was induced by treatment with t-BOOH or H2O2. It was found that EGCG exhibited good activity and low toxicity in RPE cells. The authors suggested that the flavonoids were probably acting through the inhibition of ROS accumulation and through induction of transcription factor, nuclear erythroid 2 p45-related factor 2, and its downstream phase-2 gene, heme-oxygenase 1, in human RPE cells[107].
EGCG is also an inhibitor of angiogenesis. EGCG was reported to inhibit angiogenesis by inhibiting hypoxia-inducible factor-1α protein expression[136] and in turn VEGF expression[136,137]. Jung et al[138] observed that treatment with EGCG (intraperitoneal administration) in nude mice decreased tumor growth, microvessel density and tumor cell proliferation. However, the authors reported that other tea catechins such as (-)-epigallocatechin, (-)-epicatechin gallate, and (-)-epicatechin were ineffective in vitro against Erk1/2 (extracellularly-regulated kinase-1 and -2; important mediators in the up-regulation of VEGF expression) activation, whereas EGCG inhibited Erk1/2 activation in a dose dependent manner[138].
EGCG exhibited a protective effect against cell death by H2O2 in HLEB-3 cells[139] and ultraviolet irradiation in ARPE-19 cells[140]. In another study by Zhang et al[141] EGCG provided protection to retinal neurons from oxidative stress in RGC-5 cell line and to the retina against I/R in rats. Interestingly, intravenous administration of epigallocatechin and EGCG was found to reduce the intraocular pressure in normotensive rabbits suggesting a possible role in glaucoma therapy[142].
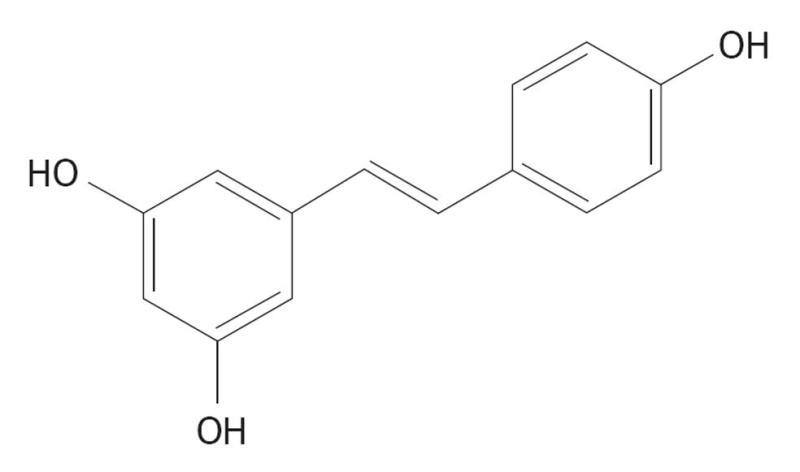
Resveratrol (RES) is a stilbene derivative (Figure 6) found in the skin of grapes and some other fruits. It is a phytoalexin, produced by plants during pathogenic infections. The pharmacological activity of RES was not noticed until 1992 when Baur et al[143] proved its cardio protective action in rodent models.
Most of the ophthalmic diseases such as AMD, PDR, proliferative vitreoretinopathy and cataract are the direct consequences of oxidative stress at a molecular level on various ocular tissues[144,145]. King et al[146] reported that RES prevented oxidative stress in cultured human RPE cells. They observed a 20% decrease in oxidation, compared to the control group, when treated with 100 μmol/Lol per liter RES. Concentration dependent anti-proliferative activity of RES was observed with 25%, 49% and 80% reduction of cell proliferation at 100, 200 and 400 μmol/L RES, respectively. Pintea et al[147] performed similar experiments with cultured human RPE cells and observed reduced levels of ROS following exposure to 500 μmol/L hydrogen peroxide in the presence of 25, 50 and 100 μmol/L RES. They also reported that RES at concentrations of 25-100 μmol/L showed protective effects without any cytotoxicity and increased superoxide dismutase, glutathione peroxidase, and catalase activity in a dose dependent manner. Zheng et al[148] also demonstrated the anti-oxidative activity of RES against oxidative stress induced by hydrogen peroxide in human lens epithelial cells (LEC). The authors observed that pretreatment with 20 μmol/L RES for 12 h is optimum for anti-oxidant activity.
RES has been reported to have both pro-apoptotic and anti-apoptotic activities[149]. Gurong et al reported that these activities of RES depend on the forkhead box O genes in porcine LEC. They observed that when this gene is silenced by specific small interference RNAs the protective activity of RES was lost. The authors also reported a reduction of cell senescence markers in RES treated LEC.
Alex et al[150] performed experiments on anti-proliferative activity of RES, EPCG and curcumin using ARPE-19 and human RPE culture cells at various concentrations. After 72 h RES was found to be the most effective in inhibiting cell growth and cell division. Oxidative stress and over expression of factors such as VEGF, angiotensin-converting enzyme, MMP-9, and endothelial nitric oxide synthase are responsible for ocular neovascularization. Yar et al[151] investigated the effect of RES on ocular neovascularization and found decreased mRNA levels of these factors.
Increased glucose levels in diabetes causes accumulation of inflammatory mediators in RPE[152-157]. As discussed earlier, inflammatory molecules such as cytokines, IL-6, TNF-α, arachidonic acid and COX, PG, intercellular adhesion molecules (ICAM), monocyte chemotactic protein (MCP) are responsible for ocular inflammation and pathological conditions such as DR[158] and AM[159]. Kubota et al[160] used oral doses of 5, 50, 100, 200 mg/kg of bodyweight in endotoxin induced uveitis (EIU) mice model with RES. They found a significant dose dependent reduction in leukocyte adhesion molecules, ICAM. The levels of ICAM and MCP were also reduced in the retina and RPE-choroid on administration of 50 mg/kg of RES in the EIU mice model. All other inflammatory mediator levels were also diminished.
Zhou et al[161] observed increased levels of the glaucoma biomarker, endothelial leukocyte adhesion molecule-1, when acutely treated with H2O2. Chronic treatment resulted in sustained stress response activation[162]. Luna et al163] observed a significant inhibition of these markers on administration of RES. They also noted that at high concentrations (200 μmol/L and 400 μmol/L) RES is cytotoxic and leads to cell death in less than 48 h. At concentrations of 50-100 μmol/L RES exerts its anti-inflammatory activity.
Curcumin is a natural phenol obtained from Curcuma longa of the Zingiberaceae family. It is the major of the three curcuminoids. Curcumin is used as an anti-inflammatory and anti-oxidant in various pathological conditions.
Oxidative stress is one of the major reasons for age related atherogenesis[164-169]. Awasthi et al[170] used an in vitro rat model to establish the activity of curcumin as an antioxidant in treating cataract. When treated with curcumin, 75 mg/kg, there was a significant increase in the glutathione S-transferase isozyme, rGST8-8, which uses 4-hydroxy-2-nonenal/L, a highly electrophilic product of lipid peroxidation, as a substrate and reduces it. It was observed that naphthalene, through an oxidative stress mediated pathway, causes cataract in rat and rabbit models. When treated with 0.005% w/w curcumin, there was significantly less opacification and apoptosis of LEC[171]. Suryanarayana et al[172] also studied the effect of curcumin in galactose induced cataract in rats. They observed that at 0.002% curcumin inhibited the onset and maturity of cataract by inhibiting oxidative species, and at 0.01% delayed the onset but accelerated the maturity of cataract formation indicating the increased oxidative stress conditions in hyperglycemic conditions. The results were supported by the report from Renu and Mamta who used 0.05% of curcumin and observed an increase in oxidative stress factors VGEF, IL-1β and nitrotyrosine and decreased GSH[173]. Manikandan et al[174] reported the protective action of curcumin in selenium induced cataract in Wister rats by inhibiting inducible nitric oxide synthase (iNOS) expression. Kumar et al[175] reported the reduced expression of α A and α B crystallins, produced during oxidative stress conditions in ocular tissues, in the presence of curcumin. Kimura et al[176] reported the inhibition of NF-κB and TNF-α by curcumin in human corneal epithelial cells.
Gupta et al[177] reported the anti-inflammatory effect of curcumin in diabetic rats, investigating the inflammatory mediators VGEF and TNF-α. They observed a significant reduction of these mediators in DR. Chen et al[178] studied the anti-inflammatory activity of curcumin in human corneal cells using sodium chloride in the medium which served as an in vitro dry eye syndrome model. Curcumin, at 5 μmol/L, prevented an increase in IL-1β, p38 mitogen-activated protein (MAP) kinase, JNK MAP kinase and NF-κB. Mandal et al confirmed the anti-inflammatory effect of curcumin in retinal cells. They suggested that curcumin modifies the activity of the inflammatory mediators such as NF-κB, phosphatidylinositol-3-kinase, nuclear factor erythroid 2-related factor 2 and prevents AMD.
An et al[179] evaluated anti-proliferative activity of curcumin in cultured rabbit RPE and reported a dose dependent and time dependent inhibition of RPE cells by increased Ca2+, which causes a decrease in DNA content. Lu et al[180] used N18 mouse-rat hybrid retina ganglion cells for the determination of the anti-proliferative effect of curcumin. In this study, arrest of N18 cells in the G2/M phase induces endoplasmic reticulum stress leading to apoptosis of retinal ganglion based on caspase-3-dependent and independent pathways. Huang et al[181] reported that curcumin induced apoptosis in bovine LEC by lowering the cell DNA levels. Curcumin was also found to inhibit human fetal retinal pigment epithelium cell proliferation by arresting the cell in G2/M phase of cell cycle[182].
Anti-angiogenic activity of curcumin was investigated in corneal endothelial cells of transgenic mouse model[183]. Curcumin was found to have angiostatic activity, inhibiting endogenous gelatinase B expression induced by fibroblast growth factor-2 which is responsible for angiogenesis. Mrudula et al[184] also investigated for anti angiogenic activity of Curcumin in streptozotocin-induced diabetic rat retina at 0.002% or 0.01% curcumin or 0.5% turmeric for a period of 8 wk. They observed inhibition of VGEF expression in treated rats. I/R injury leads to the neuron loss, glial activation and vascular degeneration. When this condition was treated with 0.01%-0.25% Curcumin, there was inhibition of injury induced NF-κB and STAT-3 which leads to inhibition of I/R injury and 0.05% Curcumin lead to vaso-protective effect[185]. Vasireddy et al[186] reported anti-protein aggregation of Curcumin in case of P23H mutation where rhodopsin molecules forms misfolding and forms aggregates leading to loss of activity.
Thus, curcumin seems to be a promising candidate for as antioxidant, anti-inflammatory, anti-proliferative, and anti-angiogenic activity.
Although the potential of the phytochemicals as therapeutic agents for oxidative stress associated ocular diseases has been demonstrated, their delivery to the ocular tissues and physiological diffusion barriers encountered has not been investigated. In order to limit/prevent oxidative damage it is imperative that the bioflavonoids reach the deeper ocular tissues such as the neural retina and lens, the sites of free radical induced damage, in effective concentrations. Unfortunately, drug delivery to the ocular tissues, especially the posterior segment ocular tissues, is a very challenging task.
The eye is a secluded organ, protected by various physiological barriers that restrict entry of xenobiotics into the ocular tissues from the external environment (topical or periocular) or systemic circulation (Table 1). The choroidal blood vessels possess many large fenestrations and pinocytic vesicles that allow free exchange of endogenous/exogenous substances between the choroidal stroma and choroidal vasculature.
| Physiological barriers | Route of administration | |||
| Topical | Peri-ocular | Systemic | Oral | |
| Precorneal loss | √ | |||
| Corneal ultrastructure | √ | |||
| Corneal efflux proteins | √ | |||
| Corneal tight junctions | √ | |||
| Conjunctival efflux proteins | √ | |||
| Conjunctival tight-junctions | √ | |||
| Conjunctival vasculature | √ | |||
| Aqueous humor outflow | √ | |||
| Anterior chamber metabolism | √ | |||
| Scleral vasculature | √ | √ | √ | √ |
| Choroidal vasculature | √ | √ | √ | √ |
| Bruch’s membrane | √ | √ | √ | √ |
| Outer BRB | ||||
| RPE tight junctions | √ | √ | √ | √ |
| RPE efflux pumps | √ | √ | √ | √ |
| Inner BRB | ||||
| Tight-junctions | √ | √ | ||
| Efflux proteins | √ | √ | ||
| Low concentration gradient | √ | √ | ||
| First pass effect | √ | |||
| Absorption limitations | √ | |||
| Metabolism in the GIT | √ | |||
| Systemic metabolism | √ | |||
Thus, systemically administered therapeutic agents can easily diffuse out into the choroidal stroma from the systemic circulation[187]. Bruch’s membrane, separating the choroid from the RPE, acts as a barrier to the diffusion of only macromolecules, like proteins and genes, and thus does not pose much of a diffusional barrier to small molecules. The RPE, on the other hand, presents a formidable permeation barrier to small drug molecules in their diffusion into the neural retina and vitreous humor from the choroidal stroma[187-189]. Epithelial cells of the RPE are joined together by tight junctions[190-194] similar to those observed in the blood-brain barrier, severely restricting paracellular diffusion of hydrophilic molecules. The RPE cells also express the efflux proteins P-gp and multidrug resistance-associated protein (MRP) on the basolateral membrane[195-198]. As a result, trans-retinal permeation of compounds that are substrates of these efflux proteins, from the systemic circulation (choroidal side) into the neural retina/vitreous humor, is strongly modulated by RPE P-gp/MRP mediated efflux. The RPE, thus, presents a major barrier to the exchange of therapeutic agents between the choroidal stroma and the neural retina/vitreous humor, and is referred to as the outer BRB.
Similar to the RPE, the endothelial cells of the blood vessels perfusing the neural retina express efflux proteins and tight-junction proteins[189,199]. The efflux proteins are polarized on the apical membrane of the endothelial cells (facing the lumen of blood vessels) and prevent entry of xenobiotics from the systemic circulation into the neural retina. Like in the outer BRB, tight junction proteins expressed on the inner BRB also severely limits paracellular diffusion of hydrophilic compounds. The barrier properties of the retinal blood vessel endothelial cells are commonly referred to as the inner BRB. Additionally, because of extensive intestinal and hepatic metabolism, the hydrophilic metabolites of the phytochemicals are seen in the systemic circulation and are available for penetration into the ocular tissues. The ambiguity in the results obtained from the limited number of clinical studies that have been carried out with the phytochemicals could be because of the inadequate delivery of the compounds into the tissues of the eye[200].
Periocularly administered agents also encounter the RPE barrier. Thus, the inner and outer BRB acts as a considerable physiological barrier to the ocular delivery of therapeutic agents through the systemic and transcleral routes of administration. Additionally, transcleral permeation is also challenged by the scleral and choroidal blood flow and lymphatics.
Topical administration is the most favored route of administration for ocular disorders. Although significant advances have been made with respect to drug delivery into the front of the eye through the topical route, back-of the eye delivery remains a significant challenge. It is thought that following topical application diffusion into the cornea, followed by lateral migration into the sclera and then diffusion across the choroid and RPE into the vitreous, is the major pathway for topically administered agents[201]. Thus, scleral and choroidal vascular and lymphatic systems as well as the RPE present significant barriers in the diffusional path. Additionally, precorneal loss, corneal ultrastructure and efflux proteins expressed on the corneal membrane present additional physiological barriers to topically administered agents.
The important bioflavonoids are well-established substrates of P-gp/MRP[202-210], and their glucuronides are extremely hydrophilic in nature as well as probable substrates of efflux proteins[211-213]. In view of the ocular barriers, very little, if any, of the parent compound and their metabolites can permeate into the neural retina from the systemic circulation. Considering that the majority of the free radical induced cellular damage is in the neural retina, achieving therapeutic drug concentrations in this tissue is absolutely necessary. It is noted, however, that oral administration may be beneficial in some aspects of the overall disease pathogenesis, e.g., in increasing retinal circulation and in decreasing microvascular leakage. There is some evidence that the hydrophilic metabolites are converted into the parent forms at the site of action[214]. However penetration of these metabolites across the retina is doubtful. Approaches based on direct intravitreal injections and transscleral applications have proven to be effective in delivering therapeutic levels of drug candidates to the back-of-the eye. Although, the intravitreal route is invasive in nature and is associated with various risk factors such as endophthalmitis and retinal detachment, they remain the most effective approach to date since this route avoids all the blood-ocular barriers. Intravitreal nanoparticulate systems can be tailored through judicial formulation approaches to achieve sustained release of the therapeutic agents for prolonged periods of time[215,216] thus minimizing the frequency of injection. Bourges et al[217] injected nanoparticles of different particle sizes and charge into vitreous humor of rabbit eye. They observed the migration of these particles from vitreous humor to retinal layers. They also observed the presence of the nanoparticles even after 4 mo of the single injection. Transscleral approaches, such as subconjunctival delivery, helps in achieving high local concentration gradient of therapeutic agents. However, the challenges posed by the scleral and choroidal lymphatics and vasculature have to be taken into account. Misra et al[218] investigated subconjunctivally biodegradable hydrogel implants for sustained release of insulin to treat DR. Based on hematoxylin and eosin stain, these implants proved to deliver drug to retina for very long time without causing any harmful effects on eye.
Several phytochemicals are potential candidates that can be used as a prophylactic agent in DR and AMD. However, systemically they exist mainly as the hydrophilic metabolites whose permeation into the ocular tissues are questionable and needs investigation. Moreover, activity of these metabolites is also not well established. In view of the challenges faced in the ocular delivery of the phytochemicals through the oral or systemic routes, topical instillation appears to be the most promising mode of administration. Although the retinal barriers, specifically the outer BRB, is still encountered by this route, the high local concentrations achieved and the absence of significant metabolism of the aglycones in the ocular tissues presents a greater likelihood of achieving therapeutic concentrations in the back-of-the eye. Studies carried out in our laboratory suggests that while significant hesperetin levels can be achieved in the RPE-choroid tissue through the topical route, getting a compound into the vitreous humor is still a challenge. Vitreal concentrations reflect free drug concentrations in the neural retina since the diffusional barrier between these two ocular components is thought to be negligible. Concentrations in the RPE-choroid by itself cannot guarantee levels in the neural retina since the compound may be in the choroid or bound to the RPE or Bruch membrane proteins. Thus, penetration into the back-of-the eye is governed by the physicochemical characteristics of the molecules and the susceptibility to being effluxed. Moreover, the compounds are rapidly cleared from the ocular tissues. Thus, further investigations into the development of novel approaches for sustained delivery of the phytochemicals to the posterior segment ocular tissues, through direct intravitreal injections or trans-scleral delivery, are needed. For anterior segment diseases, the topical formulations prepared using currently available technologies, should be an effective means of testing the therapeutic effectiveness of these promising compounds.
P- Reviewer Cherng JY S- Editor Song XX L- Editor A E- Editor Xiong L
| 1. | Arden GB. The absence of diabetic retinopathy in patients with retinitis pigmentosa: implications for pathophysiology and possible treatment. Br J Ophthalmol. 2001;85:366-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 106] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 2. | Kowluru RA, Odenbach S. Role of interleukin-1beta in the development of retinopathy in rats: effect of antioxidants. Invest Ophthalmol Vis Sci. 2004;45:4161-4166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 128] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 3. | Fong DS, Aiello L, Gardner TW, King GL, Blankenship G, Cavallerano JD, Ferris FL, Klein R. Diabetic retinopathy. Diabetes Care. 2003;26 Suppl 1:S99-S102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 148] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 4. | Lieth E, Gardner TW, Barber AJ, Antonetti DA. Retinal neurodegeneration: early pathology in diabetes. Clin Experiment Ophthalmol. 2000;28:3-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 219] [Cited by in RCA: 236] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 5. | Gardner TW, Antonetti DA, Barber AJ, LaNoue KF, Nakamura M. New insights into the pathophysiology of diabetic retinopathy: potential cell-specific therapeutic targets. Diabetes Technol Ther. 2000;2:601-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Wilkinson-Berka JL. Vasoactive factors and diabetic retinopathy: vascular endothelial growth factor, cycoloxygenase-2 and nitric oxide. Curr Pharm Des. 2004;10:3331-3348. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 92] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 7. | Watkins PJ. Retinopathy. BMJ. 2003;326:924-926. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 69] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 8. | Watkins PJ. Diabetic complications: retinopathy. Br Med J (Clin Res Ed). 1982;285:425-427. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 9. | Fong DS, Aiello L, Gardner TW, King GL, Blankenship G, Cavallerano JD, Ferris FL, Klein R. Diabetic retinopathy. Diabetes Care. 2003;26:226-229. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 178] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 10. | Tout S, Chan-Ling T, Holländer H, Stone J. The role of Müller cells in the formation of the blood-retinal barrier. Neuroscience. 1993;55:291-301. [PubMed] |
| 11. | Aizu Y, Oyanagi K, Hu J, Nakagawa H. Degeneration of retinal neuronal processes and pigment epithelium in the early stage of the streptozotocin-diabetic rats. Neuropathology. 2002;22:161-170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 86] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 12. | Niesman MR, Johnson KA, Penn JS. Therapeutic effect of liposomal superoxide dismutase in an animal model of retinopathy of prematurity. Neurochem Res. 1997;22:597-605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 13. | Flynn JT, Bancalari E, Bawol R, Goldberg R, Cassady J, Schiffman J, Feuer W, Roberts J, Gillings D, Sim E. Retinopathy of prematurity. A randomized, prospective trial of transcutaneous oxygen monitoring. Ophthalmology. 1987;94:630-638. [PubMed] |
| 14. | Penn JS. Oxygen-induced retinopathy in the rat: possible contribution of peroxidation reactions. Doc Ophthalmol. 1990;74:179-186. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 15. | Chan PH. Antioxidant-dependent amelioration of brain injury: role of CuZn-superoxide dismutase. J Neurotrauma. 1992;9 Suppl 2:S417-S423. [PubMed] |
| 16. | Stanimirovic DB, Markovic M, Micic DV, Spatz M, Mrsulja BB. Liposome-entrapped superoxide dismutase reduces ischemia/reperfusion ‘oxidative stress’ in gerbil brain. Neurochem Res. 1994;19:1473-1478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 44] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 17. | Nielsen JC, Naash MI, Anderson RE. The regional distribution of vitamins E and C in mature and premature human retinas. Invest Ophthalmol Vis Sci. 1988;29:22-26. [PubMed] |
| 18. | Kowluru RA, Koppolu P, Chakrabarti S, Chen S. Diabetes-induced activation of nuclear transcriptional factor in the retina, and its inhibition by antioxidants. Free Radic Res. 2003;37:1169-1180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 208] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 19. | Kowluru RA. Diabetes-induced elevations in retinal oxidative stress, protein kinase C and nitric oxide are interrelated. Acta Diabetol. 2001;38:179-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 73] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 20. | Serou MJ, DeCoster MA, Bazan NG. Interleukin-1 beta activates expression of cyclooxygenase-2 and inducible nitric oxide synthase in primary hippocampal neuronal culture: platelet-activating factor as a preferential mediator of cyclooxygenase-2 expression. J Neurosci Res. 1999;58:593-598. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 21. | Tilton RG, Kawamura T, Chang KC, Ido Y, Bjercke RJ, Stephan CC, Brock TA, Williamson JR. Vascular dysfunction induced by elevated glucose levels in rats is mediated by vascular endothelial growth factor. J Clin Invest. 1997;99:2192-2202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 143] [Cited by in RCA: 146] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 22. | Sone H, Kawakami Y, Okuda Y, Kondo S, Hanatani M, Suzuki H, Yamashita K. Vascular endothelial growth factor is induced by long-term high glucose concentration and up-regulated by acute glucose deprivation in cultured bovine retinal pigmented epithelial cells. Biochem Biophys Res Commun. 1996;221:193-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 84] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 23. | Brooks SE, Gu X, Kaufmann PM, Marcus DM, Caldwell RB. Modulation of VEGF production by pH and glucose in retinal Müller cells. Curr Eye Res. 1998;17:875-882. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 48] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 24. | Natarajan R, Bai W, Lanting L, Gonzales N, Nadler J. Effects of high glucose on vascular endothelial growth factor expression in vascular smooth muscle cells. Am J Physiol. 1997;273:H2224-H2231. [PubMed] |
| 25. | Kuroki M, Voest EE, Amano S, Beerepoot LV, Takashima S, Tolentino M, Kim RY, Rohan RM, Colby KA, Yeo KT. Reactive oxygen intermediates increase vascular endothelial growth factor expression in vitro and in vivo. J Clin Invest. 1996;98:1667-1675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 307] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 26. | Plouët J, Schilling J, Gospodarowicz D. Isolation and characterization of a newly identified endothelial cell mitogen produced by AtT-20 cells. EMBO J. 1989;8:3801-3806. [PubMed] |
| 27. | Leung DW, Cachianes G, Kuang WJ, Goeddel DV, Ferrara N. Vascular endothelial growth factor is a secreted angiogenic mitogen. Science. 1989;246:1306-1309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3536] [Cited by in RCA: 3553] [Article Influence: 98.7] [Reference Citation Analysis (0)] |
| 28. | Williams B. Vascular permeability/vascular endothelial growth factors: a potential role in the pathogenesis and treatment of vascular diseases. Vasc Med. 1996;1:251-258. [PubMed] |
| 29. | Connolly DT, Heuvelman DM, Nelson R, Olander JV, Eppley BL, Delfino JJ, Siegel NR, Leimgruber RM, Feder J. Tumor vascular permeability factor stimulates endothelial cell growth and angiogenesis. J Clin Invest. 1989;84:1470-1478. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 879] [Cited by in RCA: 916] [Article Influence: 25.4] [Reference Citation Analysis (0)] |
| 30. | Senger DR, Galli SJ, Dvorak AM, Perruzzi CA, Harvey VS, Dvorak HF. Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science. 1983;219:983-985. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2734] [Cited by in RCA: 2677] [Article Influence: 63.7] [Reference Citation Analysis (0)] |
| 31. | Dvorak HF, Brown LF, Detmar M, Dvorak AM. Vascular permeability factor/vascular endothelial growth factor, microvascular hyperpermeability, and angiogenesis. Am J Pathol. 1995;146:1029-1039. [PubMed] |
| 32. | Schmedtje JF, Ji YS, Liu WL, DuBois RN, Runge MS. Hypoxia induces cyclooxygenase-2 via the NF-kappaB p65 transcription factor in human vascular endothelial cells. J Biol Chem. 1997;272:601-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 527] [Cited by in RCA: 541] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 33. | Michiels C, Arnould T, Knott I, Dieu M, Remacle J. Stimulation of prostaglandin synthesis by human endothelial cells exposed to hypoxia. Am J Physiol. 1993;264:C866-C874. [PubMed] |
| 34. | Ji YS, Xu Q, Schmedtje JF. Hypoxia induces high-mobility-group protein I(Y) and transcription of the cyclooxygenase-2 gene in human vascular endothelium. Circ Res. 1998;83:295-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 66] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 35. | Bazan NG, Lukiw WJ. Cyclooxygenase-2 and presenilin-1 gene expression induced by interleukin-1beta and amyloid beta 42 peptide is potentiated by hypoxia in primary human neural cells. J Biol Chem. 2002;277:30359-30367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 78] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 36. | Lukiw WJ, Bazan NG. Strong nuclear factor-kappaB-DNA binding parallels cyclooxygenase-2 gene transcription in aging and in sporadic Alzheimer’s disease superior temporal lobe neocortex. J Neurosci Res. 1998;53:583-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 37. | Baeuerle PA, Baltimore D. NF-kappa B: ten years after. Cell. 1996;87:13-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2488] [Cited by in RCA: 2492] [Article Influence: 85.9] [Reference Citation Analysis (0)] |
| 38. | Takahashi K, Saishin Y, Saishin Y, Mori K, Ando A, Yamamoto S, Oshima Y, Nambu H, Melia MB, Bingaman DP. Topical nepafenac inhibits ocular neovascularization. Invest Ophthalmol Vis Sci. 2003;44:409-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 79] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 39. | Ayalasomayajula SP, Kompella UB. Celecoxib, a selective cyclooxygenase-2 inhibitor, inhibits retinal vascular endothelial growth factor expression and vascular leakage in a streptozotocin-induced diabetic rat model. Eur J Pharmacol. 2003;458:283-289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 115] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 40. | Dodge AB, Hechtman HB, Shepro D. Microvascular endothelial-derived autacoids regulate pericyte contractility. Cell Motil Cytoskeleton. 1991;18:180-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 37] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 41. | Murohara T, Horowitz JR, Silver M, Tsurumi Y, Chen D, Sullivan A, Isner JM. Vascular endothelial growth factor/vascular permeability factor enhances vascular permeability via nitric oxide and prostacyclin. Circulation. 1998;97:99-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 354] [Cited by in RCA: 356] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 42. | Eakins KE. Prostaglandin and non-prostaglandin mediated breeakdown of the blood-aqueous barrier. Exp Eye Res. 1977;25 Suppl:483-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 149] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 43. | Hardy P, Dumont I, Bhattacharya M, Hou X, Lachapelle P, Varma DR, Chemtob S. Oxidants, nitric oxide and prostanoids in the developing ocular vasculature: a basis for ischemic retinopathy. Cardiovasc Res. 2000;47:489-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 86] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 44. | Ziche M, Jones J, Gullino PM. Role of prostaglandin E1 and copper in angiogenesis. J Natl Cancer Inst. 1982;69:475-482. [PubMed] |
| 45. | Form DM, Auerbach R. PGE2 and angiogenesis. Proc Soc Exp Biol Med. 1983;172:214-218. [PubMed] |
| 46. | Gately S. The contributions of cyclooxygenase-2 to tumor angiogenesis. Cancer Metastasis Rev. 2000;19:19-27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 47. | Dannenberg AJ, Altorki NK, Boyle JO, Dang C, Howe LR, Weksler BB, Subbaramaiah K. Cyclo-oxygenase 2: a pharmacological target for the prevention of cancer. Lancet Oncol. 2001;2:544-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 374] [Cited by in RCA: 366] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 48. | Gasparini G, Longo R, Sarmiento R, Morabito A. Inhibitors of cyclo-oxygenase 2: a new class of anticancer agents. Lancet Oncol. 2003;4:605-615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 258] [Cited by in RCA: 266] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 49. | Lukiw WJ, Ottlecz A, Lambrou G, Grueninger M, Finley J, Thompson HW, Bazan NG. Coordinate activation of HIF-1 and NF-kappaB DNA binding and COX-2 and VEGF expression in retinal cells by hypoxia. Invest Ophthalmol Vis Sci. 2003;44:4163-4170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 91] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 50. | Salgo MG, Bermúdez E, Squadrito GL, Pryor WA. Peroxynitrite causes DNA damage and oxidation of thiols in rat thymocytes [corrected]. Arch Biochem Biophys. 1995;322:500-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 220] [Cited by in RCA: 208] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 51. | Salgo MG, Squadrito GL, Pryor WA. Peroxynitrite causes apoptosis in rat thymocytes. Biochem Biophys Res Commun. 1995;215:1111-1118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 115] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 52. | Zhuang S, Simon G. Peroxynitrite-induced apoptosis involves activation of multiple caspases in HL-60 cells. Am J Physiol Cell Physiol. 2000;279:C341-C351. [PubMed] |
| 53. | Halliwell B. What nitrates tyrosine Is nitrotyrosine specific as a biomarker of peroxynitrite formation in vivo. FEBS Lett. 1997;411:157-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 366] [Cited by in RCA: 347] [Article Influence: 12.4] [Reference Citation Analysis (1)] |
| 54. | Mancino R, Di Pierro D, Varesi C, Cerulli A, Feraco A, Cedrone C, Pinazo-Duran MD, Coletta M, Nucci C. Lipid peroxidation and total antioxidant capacity in vitreous, aqueous humor, and blood samples from patients with diabetic retinopathy. Mol Vis. 2011;17:1298-1304. [PubMed] |
| 55. | Kansagara D, Gleitsmann K, Gillingham M, Freeman M, Quiñones A. Nutritional Supplements for Age-Related Macular Degeneration: A Systematic Review [Internet]. Washington DC: Department of Veterans Affairs; 2012; . [PubMed] |
| 56. | Buschini E, Piras A, Nuzzi R, Vercelli A. Age related macular degeneration and drusen: neuroinflammation in the retina. Prog Neurobiol. 2011;95:14-25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 165] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 57. | Khandhadia S, Lotery A. Oxidation and age-related macular degeneration: insights from molecular biology. Expert Rev Mol Med. 2010;12:e34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 124] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 58. | Cano M, Thimmalappula R, Fujihara M, Nagai N, Sporn M, Wang AL, Neufeld AH, Biswal S, Handa JT. Cigarette smoking, oxidative stress, the anti-oxidant response through Nrf2 signaling, and Age-related Macular Degeneration. Vision Res. 2010;50:652-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 144] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 59. | Ding X, Patel M, Chan CC. Molecular pathology of age-related macular degeneration. Prog Retin Eye Res. 2009;28:1-18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 397] [Cited by in RCA: 429] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 60. | Olofsson EM, Marklund SL, Behndig A. Enhanced diabetes-induced cataract in copper-zinc superoxide dismutase-null mice. Invest Ophthalmol Vis Sci. 2009;50:2913-2918. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 61. | Roberts R, Luan H, Berkowitz BA. α-lipoic acid corrects late-phase supernormal retinal oxygenation response in experimental diabetic retinopathy. Invest Ophthalmol Vis Sci. 2006;47:4077-4082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 62. | Lin J, Bierhaus A, Bugert P, Dietrich N, Feng Y, Vom Hagen F, Nawroth P, Brownlee M, Hammes HP. Effect of R-(+)-α-lipoic acid on experimental diabetic retinopathy. Diabetologia. 2006;49:1089-1096. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 66] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 63. | Miranda M, Muriach M, Roma J, Bosch-Morell F, Genovés JM, Barcia J, Araiz J, Díaz-Llospis M, Romero FJ. [Oxidative stress in a model of experimental diabetic retinopathy: the utility of peroxinytrite scavengers]. Arch Soc Esp Oftalmol. 2006;81:27-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 64. | Comer GM, Ciulla TA. Current and future pharmacological intervention for diabetic retinopathy. Expert Opin Emerg Drugs. 2005;10:441-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 65. | Yatoh S, Mizutani M, Yokoo T, Kozawa T, Sone H, Toyoshima H, Suzuki S, Shimano H, Kawakami Y, Okuda Y. Antioxidants and an inhibitor of advanced glycation ameliorate death of retinal microvascular cells in diabetic retinopathy. Diabetes Metab Res Rev. 2006;22:38-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 38] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 67. | Kowluru RA, Engerman RL, Case GL, Kern TS. Retinal glutamate in diabetes and effect of antioxidants. Neurochem Int. 2001;38:385-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 130] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 68. | Zhang SX, Wang JJ, Gao G, Parke K, Ma JX. Pigment epithelium-derived factor downregulates vascular endothelial growth factor (VEGF) expression and inhibits VEGF-VEGF receptor 2 binding in diabetic retinopathy. J Mol Endocrinol. 2006;37:1-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 178] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 69. | Yamagishi S, Nakamura K, Matsui T, Inagaki Y, Takenaka K, Jinnouchi Y, Yoshida Y, Matsuura T, Narama I, Motomiya Y. Pigment epithelium-derived factor inhibits advanced glycation end product-induced retinal vascular hyperpermeability by blocking reactive oxygen species-mediated vascular endothelial growth factor expression. J Biol Chem. 2006;281:20213-20220. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 179] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 70. | Kinose F, Roscilli G, Lamartina S, Anderson KD, Bonelli F, Spence SG, Ciliberto G, Vogt TF, Holder DJ, Toniatti C. Inhibition of retinal and choroidal neovascularization by a novel KDR kinase inhibitor. Mol Vis. 2005;11:366-373. [PubMed] |
| 71. | Kuo GH, Wang A, Emanuel S, Deangelis A, Zhang R, Connolly PJ, Murray WV, Gruninger RH, Sechler J, Fuentes-Pesquera A. Synthesis and discovery of pyrazine-pyridine biheteroaryl as a novel series of potent vascular endothelial growth factor receptor-2 inhibitors. J Med Chem. 2005;48:1886-1900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 72. | Ayalasomayajula SP, Amrite AC, Kompella UB. Inhibition of cyclooxygenase-2, but not cyclooxygenase-1, reduces prostaglandin E2 secretion from diabetic rat retinas. Eur J Pharmacol. 2004;498:275-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 40] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 73. | Unsoeld AS, Junker B, Mazitschek R, Martin G, Hansen LL, Giannis A, Agostini HT. Local injection of receptor tyrosine kinase inhibitor MAE 87 reduces retinal neovascularization in mice. Mol Vis. 2004;10:468-475. [PubMed] |
| 74. | Patel N, Sun L, Moshinsky D, Chen H, Leahy KM, Le P, Moss KG, Wang X, Rice A, Tam D. A selective and oral small molecule inhibitor of vascular epithelial growth factor receptor (VEGFR)-2 and VEGFR-1 inhibits neovascularization and vascular permeability. J Pharmacol Exp Ther. 2003;306:838-845. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 75. | Saishin Y, Saishin Y, Takahashi K, Lima e Silva R, Hylton D, Rudge JS, Wiegand SJ, Campochiaro PA. VEGF-TRAP(R1R2) suppresses choroidal neovascularization and VEGF-induced breakdown of the blood-retinal barrier. J Cell Physiol. 2003;195:241-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 183] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 76. | Joussen AM, Poulaki V, Mitsiades N, Kirchhof B, Koizumi K, Döhmen S, Adamis AP. Nonsteroidal anti-inflammatory drugs prevent early diabetic retinopathy via TNF-α suppression. FASEB J. 2002;16:438-440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 396] [Cited by in RCA: 406] [Article Influence: 17.7] [Reference Citation Analysis (0)] |
| 77. | Berkowitz BA, Roberts R, Luan H, Peysakhov J, Knoerzer DL, Connor JR, Hohman TC. Drug intervention can correct subnormal retinal oxygenation response in experimental diabetic retinopathy. Invest Ophthalmol Vis Sci. 2005;46:2954-2960. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 78. | Available from: http: //www.nei.nih.gov/amd/summary.asp. |
| 79. | Crozier A, Jaganath IB, Clifford MN. Dietary phenolics: chemistry, bioavailability and effects on health. Nat Prod Rep. 2009;26:1001-1043. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1316] [Cited by in RCA: 1215] [Article Influence: 75.9] [Reference Citation Analysis (0)] |
| 80. | Ohia SE, Opere CA, Leday AM. Pharmacological consequences of oxidative stress in ocular tissues. Mutat Res. 2005;579:22-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 97] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 81. | Erickson KK, Sundstrom JM, Antonetti DA. Vascular permeability in ocular disease and the role of tight junctions. Angiogenesis. 2007;10:103-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 140] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 82. | Kaur G, Tirkey N, Chopra K. Beneficial effect of hesperidin on lipopolysaccharide-induced hepatotoxicity. Toxicology. 2006;226:152-160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 98] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 83. | Pollard SE, Whiteman M, Spencer JP. Modulation of peroxynitrite-induced fibroblast injury by hesperetin: a role for intracellular scavenging and modulation of ERK signalling. Biochem Biophys Res Commun. 2006;347:916-923. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 84. | Olszanecki R, Gebska A, Kozlovski VI, Gryglewski RJ. Flavonoids and nitric oxide synthase. J Physiol Pharmacol. 2002;53:571-584. [PubMed] |
| 85. | Jeong YJ, Choi YJ, Kwon HM, Kang SW, Park HS, Lee M, Kang YH. Differential inhibition of oxidized LDL-induced apoptosis in human endothelial cells treated with different flavonoids. Br J Nutr. 2005;93:581-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 63] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 86. | Hirata A, Murakami Y, Shoji M, Kadoma Y, Fujisawa S. Kinetics of radical-scavenging activity of hesperetin and hesperidin and their inhibitory activity on COX-2 expression. Anticancer Res. 2005;25:3367-3374. [PubMed] |
| 87. | Sakata K, Hirose Y, Qiao Z, Tanaka T, Mori H. Inhibition of inducible isoforms of cyclooxygenase and nitric oxide synthase by flavonoid hesperidin in mouse macrophage cell line. Cancer Lett. 2003;199:139-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 76] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 88. | Saija A, Scalese M, Lanza M, Marzullo D, Bonina F, Castelli F. Flavonoids as antioxidant agents: importance of their interaction with biomembranes. Free Radic Biol Med. 1995;19:481-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 499] [Cited by in RCA: 461] [Article Influence: 15.4] [Reference Citation Analysis (0)] |
| 89. | Cirico TL, Omaye ST. Additive or synergetic effects of phenolic compounds on human low density lipoprotein oxidation. Food Chem Toxicol. 2006;44:510-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 64] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 90. | Orallo F, Alvarez E, Basaran H, Lugnier C. Comparative study of the vasorelaxant activity, superoxide-scavenging ability and cyclic nucleotide phosphodiesterase-inhibitory effects of hesperetin and hesperidin. Naunyn Schmiedebergs Arch Pharmacol. 2004;370:452-463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 51] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 91. | Balestrieri ML, Castaldo D, Balestrieri C, Quagliuolo L, Giovane A, Servillo L. Modulation by flavonoids of PAF and related phospholipids in endothelial cells during oxidative stress. J Lipid Res. 2003;44:380-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 33] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 92. | Galati EM, Monforte MT, Kirjavainen S, Forestieri AM, Trovato A, Tripodo MM. Biological effects of hesperidin, a citrus flavonoid. (Note I): antiinflammatory and analgesic activity. Farmaco. 1994;40:709-712. [PubMed] |
| 93. | Ansorge S, Reinhold D, Lendeckel U. Propolis and some of its constituents down-regulate DNA synthesis and inflammatory cytokine production but induce TGF-beta1 production of human immune cells. Z Naturforsch C. 2003;58:580-589. [PubMed] |
| 94. | Guardia T, Rotelli AE, Juarez AO, Pelzer LE. Anti-inflammatory properties of plant flavonoids. Effects of rutin, quercetin and hesperidin on adjuvant arthritis in rat. Farmaco. 2001;56:683-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 442] [Cited by in RCA: 418] [Article Influence: 17.4] [Reference Citation Analysis (0)] |
| 95. | Chiou GC, Xu XR. Effects of some natural flavonoids on retinal function recovery after ischemic insult in the rat. J Ocul Pharmacol Ther. 2004;20:107-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 96. | Calderone V, Chericoni S, Martinelli C, Testai L, Nardi A, Morelli I, Breschi MC, Martinotti E. Vasorelaxing effects of flavonoids: investigation on the possible involvement of potassium channels. Naunyn Schmiedebergs Arch Pharmacol. 2004;370:290-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 112] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 97. | Manthey JA. Biological properties of flavonoids pertaining to inflammation. Microcirculation. 2000;7:S29-S34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 40] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 98. | Bouskela E, Cyrino FZ, Marcelon G. Inhibitory effect of the Ruscus extract and of the flavonoid hesperidine methylchalcone on increased microvascular permeability induced by various agents in the hamster cheek pouch. J Cardiovasc Pharmacol. 1993;22:225-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 99. | Korthuis RJ, Gute DC. Adhesion molecule expression in postischemic microvascular dysfunction: activity of a micronized purified flavonoid fraction. J Vasc Res. 1999;36 Suppl 1:15-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 100. | McGregor L, Bellangeon M, Chignier E, Lerond L, Rousselle C, McGregor JL. Effect of a micronized purified flavonoid fraction on in vivo platelet functions in the rat. Thromb Res. 1999;94:235-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 18] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 101. | de Whalley CV, Rankin SM, Hoult JR, Jessup W, Leake DS. Flavonoids inhibit the oxidative modification of low density lipoproteins by macrophages. Biochem Pharmacol. 1990;39:1743-1750. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 419] [Cited by in RCA: 379] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 102. | Huk I, Brovkovych V, Nanobash Vili J, Weigel G, Neumayer C, Partyka L, Patton S, Malinski T. Bioflavonoid quercetin scavenges superoxide and increases nitric oxide concentration in ischaemia-reperfusion injury: an experimental study. Br J Surg. 1998;85:1080-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 94] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 103. | Mojzis J, Hviscová K, Germanova D, Bukovicová D, Mirossay L. Protective effect of quercetin on ischemia/reperfusion-induced gastric mucosal injury in rats. Physiol Res. 2001;50:501-506. [PubMed] |
| 104. | Seufi AM, Ibrahim SS, Elmaghraby TK, Hafez EE. Preventive effect of the flavonoid, quercetin, on hepatic cancer in rats via oxidant/antioxidant activity: molecular and histological evidences. J Exp Clin Cancer Res. 2009;28:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 86] [Cited by in RCA: 79] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 105. | Liu JH, Wann H, Chen MM, Pan WH, Chen YC, Liu CM, Yeh MY, Tsai SK, Young MS, Chuang HY. Baicalein significantly protects human retinal pigment epithelium cells against H₂O₂-induced oxidative stress by scavenging reactive oxygen species and downregulating the expression of matrix metalloproteinase-9 and vascular endothelial growth factor. J Ocul Pharmacol Ther. 2010;26:421-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 106. | Kelly GS. Quercetin. Monograph. Altern Med Rev. 2011;16:172-194. [PubMed] |
| 107. | Hanneken A, Lin FF, Johnson J, Maher P. Flavonoids protect human retinal pigment epithelial cells from oxidative-stress-induced death. Invest Ophthalmol Vis Sci. 2006;47:3164-3177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 226] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 108. | Kook D, Wolf AH, Yu AL, Neubauer AS, Priglinger SG, Kampik A, Welge-Lüssen UC. The protective effect of quercetin against oxidative stress in the human RPE in vitro. Invest Ophthalmol Vis Sci. 2008;49:1712-1720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 118] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 109. | Maher P, Hanneken A. Flavonoids protect retinal ganglion cells from oxidative stress-induced death. Invest Ophthalmol Vis Sci. 2005;46:4796-4803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 110] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 110. | Maher P, Hanneken A. Flavonoids protect retinal ganglion cells from ischemia in vitro. Exp Eye Res. 2008;86:366-374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 111. | Zhuang P, Shen Y, Lin BQ, Zhang WY, Chiou GC. Effect of Quercetin on Formation of Choroidal Neovascularization (CNV) in Age-related Macular Degeneration(AMD). Yanke Xuebao. 2011;26:23-29. [PubMed] |
| 112. | Chen Y, Li XX, Xing NZ, Cao XG. Quercetin inhibits choroidal and retinal angiogenesis in vitro. Graefes Arch Clin Exp Ophthalmol. 2008;246:373-378. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 46] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 113. | Tan WF, Lin LP, Li MH, Zhang YX, Tong YG, Xiao D, Ding J. Quercetin, a dietary-derived flavonoid, possesses antiangiogenic potential. Eur J Pharmacol. 2003;459:255-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 136] [Cited by in RCA: 135] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 114. | Jackson SJ, Venema RC. Quercetin inhibits eNOS, microtubule polymerization, and mitotic progression in bovine aortic endothelial cells. J Nutr. 2006;136:1178-1184. [PubMed] |
| 115. | Igura K, Ohta T, Kuroda Y, Kaji K. Resveratrol and quercetin inhibit angiogenesis in vitro. Cancer Lett. 2001;171:11-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 163] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 116. | Donnini S, Finetti F, Lusini L, Morbidelli L, Cheynier V, Barron D, Williamson G, Waltenberger J, Ziche M. Divergent effects of quercetin conjugates on angiogenesis. Br J Nutr. 2006;95:1016-1023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 117. | Rogerio AP, Kanashiro A, Fontanari C, da Silva EV, Lucisano-Valim YM, Soares EG, Faccioli LH. Anti-inflammatory activity of quercetin and isoquercitrin in experimental murine allergic asthma. Inflamm Res. 2007;56:402-408. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 216] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 118. | Rogerio AP, Dora CL, Andrade EL, Chaves JS, Silva LF, Lemos-Senna E, Calixto JB. Anti-inflammatory effect of quercetin-loaded microemulsion in the airways allergic inflammatory model in mice. Pharmacol Res. 2010;61:288-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 131] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 119. | Chirumbolo S. The role of quercetin, flavonols and flavones in modulating inflammatory cell function. Inflamm Allergy Drug Targets. 2010;9:263-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 190] [Cited by in RCA: 204] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 120. | Raso GM, Meli R, Di Carlo G, Pacilio M, Di Carlo R. Inhibition of inducible nitric oxide synthase and cyclooxygenase-2 expression by flavonoids in macrophage J774A.1. Life Sci. 2001;68:921-931. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 288] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 121. | Crespo I, García-Mediavilla MV, Gutiérrez B, Sánchez-Campos S, Tuñón MJ, González-Gallego J. A comparison of the effects of kaempferol and quercetin on cytokine-induced pro-inflammatory status of cultured human endothelial cells. Br J Nutr. 2008;100:968-976. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 117] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 122. | Martínez-Flórez S, Gutiérrez-Fernández B, Sánchez-Campos S, González-Gallego J, Tuñón MJ. Quercetin attenuates nuclear factor-kappaB activation and nitric oxide production in interleukin-1beta-activated rat hepatocytes. J Nutr. 2005;135:1359-1365. [PubMed] |
| 123. | Kim BH, Cho SM, Reddy AM, Kim YS, Min KR, Kim Y. Down-regulatory effect of quercitrin gallate on nuclear factor-kappa B-dependent inducible nitric oxide synthase expression in lipopolysaccharide-stimulated macrophages RAW 264.7. Biochem Pharmacol. 2005;69:1577-1583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 50] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 124. | Park HH, Lee S, Son HY, Park SB, Kim MS, Choi EJ, Singh TS, Ha JH, Lee MG, Kim JE. Flavonoids inhibit histamine release and expression of proinflammatory cytokines in mast cells. Arch Pharm Res. 2008;31:1303-1311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 211] [Cited by in RCA: 232] [Article Influence: 13.6] [Reference Citation Analysis (0)] |
| 125. | Cao XG, Li XX, Bao YZ, Xing NZ, Chen Y. Responses of human lens epithelial cells to quercetin and DMSO. Invest Ophthalmol Vis Sci. 2007;48:3714-3718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 126. | Cornish KM, Williamson G, Sanderson J. Quercetin metabolism in the lens: role in inhibition of hydrogen peroxide induced cataract. Free Radic Biol Med. 2002;33:63-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 67] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 127. | Jiang Q, Cao C, Zhou C, Song X, Healey S, Kouttab N, Chu W, Xu A, Bi Z, Wan Y. Quercetin attenuates UV- and H(2)O(2)-induced decrease of collagen type I in cultured human lens epithelial cells. J Ocul Pharmacol Ther. 2008;24:164-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 128. | Sanderson J, McLauchlan WR, Williamson G. Quercetin inhibits hydrogen peroxide-induced oxidation of the rat lens. Free Radic Biol Med. 1999;26:639-645. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 52] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 129. | Srinivas NR. Baicalin, an emerging multi-therapeutic agent: pharmacodynamics, pharmacokinetics, and considerations from drug development perspectives. Xenobiotica. 2010;40:357-367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 99] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 130. | Guo HX, Liu DH, Ma Y, Liu JF, Wang Y, Du ZY, Wang X, Shen JK, Peng HL. Long-term baicalin administration ameliorates metabolic disorders and hepatic steatosis in rats given a high-fat diet. Acta Pharmacol Sin. 2009;30:1505-1512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 120] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 131. | Waisundara VY, Hsu A, Tan BK, Huang D. Baicalin reduces mitochondrial damage in streptozotocin-induced diabetic Wistar rats. Diabetes Metab Res Rev. 2009;25:671-677. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 52] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 132. | Ling Y, Chen Y, Chen P, Hui H, Song X, Lu Z, Li C, Lu N, Guo Q. Baicalein potently suppresses angiogenesis induced by vascular endothelial growth factor through the p53/Rb signaling pathway leading to G1/S cell cycle arrest. Exp Biol Med (Maywood). 2011;236:851-858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 33] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 133. | Yang LP, Sun HL, Wu LM, Guo XJ, Dou HL, Tso MO, Zhao L, Li SM. Baicalein reduces inflammatory process in a rodent model of diabetic retinopathy. Invest Ophthalmol Vis Sci. 2009;50:2319-2327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 116] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 134. | Nakamura N, Hayasaka S, Zhang XY, Nagaki Y, Matsumoto M, Hayasaka Y, Terasawa K. Effects of baicalin, baicalein, and wogonin on interleukin-6 and interleukin-8 expression, and nuclear factor-kappab binding activities induced by interleukin-1beta in human retinal pigment epithelial cell line. Exp Eye Res. 2003;77:195-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 64] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 135. | Jung SH, Kang KD, Ji D, Fawcett RJ, Safa R, Kamalden TA, Osborne NN. The flavonoid baicalin counteracts ischemic and oxidative insults to retinal cells and lipid peroxidation to brain membranes. Neurochem Int. 2008;53:325-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 81] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 136. | Zhang Q, Tang X, Lu Q, Zhang Z, Rao J, Le AD. Green tea extract and (-)-epigallocatechin-3-gallate inhibit hypoxia- and serum-induced HIF-1α protein accumulation and VEGF expression in human cervical carcinoma and hepatoma cells. Mol Cancer Ther. 2006;5:1227-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 132] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 137. | Shirakami Y, Shimizu M, Adachi S, Sakai H, Nakagawa T, Yasuda Y, Tsurumi H, Hara Y, Moriwaki H. (-)-Epigallocatechin gallate suppresses the growth of human hepatocellular carcinoma cells by inhibiting activation of the vascular endothelial growth factor-vascular endothelial growth factor receptor axis. Cancer Sci. 2009;100:1957-1962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 81] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 138. | Jung YD, Kim MS, Shin BA, Chay KO, Ahn BW, Liu W, Bucana CD, Gallick GE, Ellis LM. EGCG, a major component of green tea, inhibits tumour growth by inhibiting VEGF induction in human colon carcinoma cells. Br J Cancer. 2001;84:844-850. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 243] [Cited by in RCA: 230] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 139. | Yao K, Ye P, Zhang L, Tan J, Tang X, Zhang Y. Epigallocatechin gallate protects against oxidative stress-induced mitochondria-dependent apoptosis in human lens epithelial cells. Mol Vis. 2008;14:217-223. [PubMed] |
| 140. | Yang SW, Lee BR, Koh JW. Protective effects of epigallocatechin gallate after UV irradiation in cultured human retinal pigment epithelial cells. Korean J Ophthalmol. 2007;21:232-237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 141. | Zhang B, Safa R, Rusciano D, Osborne NN. Epigallocatechin gallate, an active ingredient from green tea, attenuates damaging influences to the retina caused by ischemia/reperfusion. Brain Res. 2007;1159:40-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 75] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 142. | Hodges LC, Kearse CE, Green K. Intraocular pressure-lowering activity of phenolic antioxidants in normotensive rabbits. Curr Eye Res. 1999;19:234-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 143. | Baur JA, Sinclair DA. Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov. 2006;5:493-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2705] [Cited by in RCA: 2778] [Article Influence: 146.2] [Reference Citation Analysis (0)] |
| 144. | Chiu CJ, Taylor A. Nutritional antioxidants and age-related cataract and maculopathy. Exp Eye Res. 2007;84:229-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 125] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 145. | Liang FQ, Godley BF. Oxidative stress-induced mitochondrial DNA damage in human retinal pigment epithelial cells: a possible mechanism for RPE aging and age-related macular degeneration. Exp Eye Res. 2003;76:397-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 394] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 146. | King RE, Kent KD, Bomser JA. Resveratrol reduces oxidation and proliferation of human retinal pigment epithelial cells via extracellular signal-regulated kinase inhibition. Chem Biol Interact. 2005;151:143-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 127] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 147. | Pintea A, Rugină D, Pop R, Bunea A, Socaciu C, Diehl HA. Antioxidant effect of trans-resveratrol in cultured human retinal pigment epithelial cells. J Ocul Pharmacol Ther. 2011;27:315-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 51] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 148. | Zheng Y, Liu Y, Ge J, Wang X, Liu L, Bu Z, Liu P. Resveratrol protects human lens epithelial cells against H2O2-induced oxidative stress by increasing catalase, SOD-1, and HO-1 expression. Mol Vis. 2010;16:1467-1474. [PubMed] |
| 149. | Steigerwalt RD, Gianni B, Paolo M, Bombardelli E, Burki C, Schönlau F. Effects of Mirtogenol on ocular blood flow and intraocular hypertension in asymptomatic subjects. Mol Vis. 2008;14:1288-1292. [PubMed] |
| 150. | Alex AF, Spitznas M, Tittel AP, Kurts C, Eter N. Inhibitory effect of epigallocatechin gallate (EGCG), resveratrol, and curcumin on proliferation of human retinal pigment epithelial cells in vitro. Curr Eye Res. 2010;35:1021-1033. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 151. | Yar AS, Menevse S, Dogan I, Alp E, Ergin V, Cumaoglu A, Aricioglu A, Ekmekci A, Menevse A. Investigation of ocular neovascularization-related genes and oxidative stress in diabetic rat eye tissues after resveratrol treatment. J Med Food. 2012;15:391-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 152. | Kim YS, Jung DH, Kim NH, Lee YM, Kim JS. Effect of magnolol on TGF-beta1 and fibronectin expression in human retinal pigment epithelial cells under diabetic conditions. Eur J Pharmacol. 2007;562:12-19. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 153. | Malfait M, Gomez P, van Veen TA, Parys JB, De Smedt H, Vereecke J, Himpens B. Effects of hyperglycemia and protein kinase C on connexin43 expression in cultured rat retinal pigment epithelial cells. J Membr Biol. 2001;181:31-40. [PubMed] |
| 154. | Sato T, Haimovici R, Kao R, Li AF, Roy S. Downregulation of connexin 43 expression by high glucose reduces gap junction activity in microvascular endothelial cells. Diabetes. 2002;51:1565-1571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 116] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 155. | Fernandes AF, Zhou J, Zhang X, Bian Q, Sparrow J, Taylor A, Pereira P, Shang F. Oxidative inactivation of the proteasome in retinal pigment epithelial cells. A potential link between oxidative stress and up-regulation of interleukin-8. J Biol Chem. 2008;283:20745-20753. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 56] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 156. | Bailey TA, Kanuga N, Romero IA, Greenwood J, Luthert PJ, Cheetham ME. Oxidative stress affects the junctional integrity of retinal pigment epithelial cells. Invest Ophthalmol Vis Sci. 2004;45:675-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 185] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 157. | Cosentino F, Eto M, De Paolis P, van der Loo B, Bachschmid M, Ullrich V, Kouroedov A, Delli Gatti C, Joch H, Volpe M. High glucose causes upregulation of cyclooxygenase-2 and alters prostanoid profile in human endothelial cells: role of protein kinase C and reactive oxygen species. Circulation. 2003;107:1017-1023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 303] [Cited by in RCA: 305] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 158. | Joussen AM, Poulaki V, Le ML, Koizumi K, Esser C, Janicki H, Schraermeyer U, Kociok N, Fauser S, Kirchhof B. A central role for inflammation in the pathogenesis of diabetic retinopathy. FASEB J. 2004;18:1450-1452. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 713] [Cited by in RCA: 827] [Article Influence: 39.4] [Reference Citation Analysis (0)] |
| 159. | Ambati J, Anand A, Fernandez S, Sakurai E, Lynn BC, Kuziel WA, Rollins BJ, Ambati BK. An animal model of age-related macular degeneration in senescent Ccl-2- or Ccr-2-deficient mice. Nat Med. 2003;9:1390-1397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 407] [Cited by in RCA: 449] [Article Influence: 20.4] [Reference Citation Analysis (0)] |
| 160. | Kubota S, Kurihara T, Mochimaru H, Satofuka S, Noda K, Ozawa Y, Oike Y, Ishida S, Tsubota K. Prevention of ocular inflammation in endotoxin-induced uveitis with resveratrol by inhibiting oxidative damage and nuclear factor-kappaB activation. Invest Ophthalmol Vis Sci. 2009;50:3512-3519. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 114] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 161. | Zhou Q, Liu YQ, Zhao JL, Zhang H. [Effects of oxidative stress on the expression of endothelial-leukocyte adhesion molecule-1 in porcine trabecular meshwork cells]. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2007;29:394-397. [PubMed] |
| 162. | Li G, Luna C, Liton PB, Navarro I, Epstein DL, Gonzalez P. Sustained stress response after oxidative stress in trabecular meshwork cells. Mol Vis. 2007;13:2282-2288. [PubMed] |
| 163. | Luna C, Li G, Liton PB, Qiu J, Epstein DL, Challa P, Gonzalez P. Resveratrol prevents the expression of glaucoma markers induced by chronic oxidative stress in trabecular meshwork cells. Food Chem Toxicol. 2009;47:198-204. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 91] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 164. | Spector A, Wang GM, Wang RR. The prevention of cataract caused by oxidative stress in cultured rat lenses. II. Early effects of photochemical stress and recovery. Exp Eye Res. 1993;57:659-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 23] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 165. | Spector A, Wang GM, Wang RR, Garner WH, Moll H. The prevention of cataract caused by oxidative stress in cultured rat lenses. I. H2O2 and photochemically induced cataract. Curr Eye Res. 1993;12:163-179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 78] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 166. | Bhatnagar A, Ansari NH, Zacarias A, Srivastava SK. Digital image analysis of cultured rat lens during oxidative stress-induced cataractogenesis. Exp Eye Res. 1993;57:385-391. [PubMed] |
| 167. | Spector A, Wang GM, Wang RR, Li WC, Kleiman NJ. A brief photochemically induced oxidative insult causes irreversible lens damage and cataract. II. Mechanism of action. Exp Eye Res. 1995;60:483-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 63] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 168. | Kehrer JP. Free radicals as mediators of tissue injury and disease. Crit Rev Toxicol. 1993;23:21-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 985] [Cited by in RCA: 901] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 169. | Bhuyan KC, Bhuyan DK. Molecular mechanism of cataractogenesis: III. Toxic metabolites of oxygen as initiators of lipid peroxidation and cataract. Curr Eye Res. 1984;3:67-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 136] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 170. | Awasthi S, Srivatava SK, Piper JT, Singhal SS, Chaubey M, Awasthi YC. Curcumin protects against 4-hydroxy-2-trans-nonenal-induced cataract formation in rat lenses. Am J Clin Nutr. 1996;64:761-766. [PubMed] |
| 171. | Pandya U, Saini MK, Jin GF, Awasthi S, Godley BF, Awasthi YC. Dietary curcumin prevents ocular toxicity of naphthalene in rats. Toxicol Lett. 2000;115:195-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 45] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 172. | Suryanarayana P, Krishnaswamy K, Reddy GB. Effect of curcumin on galactose-induced cataractogenesis in rats. Mol Vis. 2003;9:223-230. [PubMed] |
| 173. | Kowluru RA, Kanwar M. Effects of curcumin on retinal oxidative stress and inflammation in diabetes. Nutr Metab (Lond). 2007;4:8. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 213] [Cited by in RCA: 235] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 174. | Manikandan R, Thiagarajan R, Beulaja S, Chindhu S, Mariammal K, Sudhandiran G, Arumugam M. Anti-cataractogenic effect of curcumin and aminoguanidine against selenium-induced oxidative stress in the eye lens of Wistar rat pups: An in vitro study using isolated lens. Chem Biol Interact. 2009;181:202-209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 175. | Kumar PA, Haseeb A, Suryanarayana P, Ehtesham NZ, Reddy GB. Elevated expression of αA- and αB-crystallins in streptozotocin-induced diabetic rat. Arch Biochem Biophys. 2005;444:77-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 73] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 176. | Kimura K, Teranishi S, Fukuda K, Kawamoto K, Nishida T. Delayed disruption of barrier function in cultured human corneal epithelial cells induced by tumor necrosis factor-α in a manner dependent on NF-kappaB. Invest Ophthalmol Vis Sci. 2008;49:565-571. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 67] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 177. | Gupta SK, Kumar B, Nag TC, Agrawal SS, Agrawal R, Agrawal P, Saxena R, Srivastava S. Curcumin prevents experimental diabetic retinopathy in rats through its hypoglycemic, antioxidant, and anti-inflammatory mechanisms. J Ocul Pharmacol Ther. 2011;27:123-130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 133] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 178. | Chen M, Hu DN, Pan Z, Lu CW, Xue CY, Aass I. Curcumin protects against hyperosmoticity-induced IL-1beta elevation in human corneal epithelial cell via MAPK pathways. Exp Eye Res. 2010;90:437-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 66] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 179. | An JB, Ma JX, Liu DY, Gao YJ, Sheng MY, Wang HX, Liu LY. [The effect of curcumin on DNA content, mitochondrial transmembrane potential and calcium of rabbit cultured retinal pigment epithelial cells]. Zhonghua Yan Ke Za Zhi. 2009;45:210-215. [PubMed] |
| 180. | Lu HF, Lai KC, Hsu SC, Lin HJ, Yang MD, Chen YL, Fan MJ, Yang JS, Cheng PY, Kuo CL. Curcumin induces apoptosis through FAS and FADD, in caspase-3-dependent and -independent pathways in the N18 mouse-rat hybrid retina ganglion cells. Oncol Rep. 2009;22:97-104. [PubMed] |
| 181. | Huang XR, Qi MX, Kang KR. [Apoptosis of lens epithelial cell induced by curcumin and its mechanism]. Zhonghua Yanke Zazhi. 2006;42:649-653. [PubMed] |
| 182. | Gong L, Jiang D, Zhu X, Guo L. [Curcumin inhibits the proliferation of cultured human fetal retinal pigment epithelium cells]. Yanke Xuebao. 2004;20:246-248, 258. [PubMed] |
| 183. | Mohan R, Sivak J, Ashton P, Russo LA, Pham BQ, Kasahara N, Raizman MB, Fini ME. Curcuminoids inhibit the angiogenic response stimulated by fibroblast growth factor-2, including expression of matrix metalloproteinase gelatinase B. J Biol Chem. 2000;275:10405-10412. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 179] [Cited by in RCA: 184] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 184. | Mrudula T, Suryanarayana P, Srinivas PN, Reddy GB. Effect of curcumin on hyperglycemia-induced vascular endothelial growth factor expression in streptozotocin-induced diabetic rat retina. Biochem Biophys Res Commun. 2007;361:528-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 88] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 185. | Wang L, Li C, Guo H, Kern TS, Huang K, Zheng L. Curcumin inhibits neuronal and vascular degeneration in retina after ischemia and reperfusion injury. PLoS One. 2011;6:e23194. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 74] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 186. | Vasireddy V, Chavali VR, Joseph VT, Kadam R, Lin JH, Jamison JA, Kompella UB, Reddy GB, Ayyagari R. Rescue of photoreceptor degeneration by curcumin in transgenic rats with P23H rhodopsin mutation. PLoS One. 2011;6:e21193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 73] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 187. | Törnquist P, Alm A, Bill A. Permeability of ocular vessels and transport across the blood-retinal-barrier. Eye (Lond). 1990;4:303-309. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 74] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 188. | Cunha-Vaz JG. The blood-retinal barriers. Doc Ophthalmol. 1976;41:287-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 160] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 189. | Cunha-Vaz J. The blood-ocular barriers. Surv Ophthalmol. 1979;23:279-296. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 249] [Cited by in RCA: 246] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 190. | Peng S, Rahner C, Rizzolo LJ. Apical and basal regulation of the permeability of the retinal pigment epithelium. Invest Ophthalmol Vis Sci. 2003;44:808-817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 35] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 191. | Altunay H. Fine structure of the retinal pigment epithelium, Bruch’s membrane and choriocapillaris in the horse. Anat Histol Embryol. 2000;29:135-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 192. | Rizzolo LJ. Polarity and the development of the outer blood-retinal barrier. Histol Histopathol. 1997;12:1057-1067. [PubMed] |
| 193. | Konari K, Sawada N, Zhong Y, Isomura H, Nakagawa T, Mori M. Development of the blood-retinal barrier in vitro: formation of tight junctions as revealed by occludin and ZO-1 correlates with the barrier function of chick retinal pigment epithelial cells. Exp Eye Res. 1995;61:99-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 47] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 194. | Cunha-Vaz JG. The blood-ocular barriers: past, present, and future. Doc Ophthalmol. 1997;93:149-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 109] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 195. | Duvvuri S, Gandhi MD, Mitra AK. Effect of P-glycoprotein on the ocular disposition of a model substrate, quinidine. Curr Eye Res. 2003;27:345-353. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 31] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 196. | Constable PA, Lawrenson JG, Dolman DE, Arden GB, Abbott NJ. P-Glycoprotein expression in human retinal pigment epithelium cell lines. Exp Eye Res. 2006;83:24-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 47] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 197. | Steuer H, Jaworski A, Elger B, Kaussmann M, Keldenich J, Schneider H, Stoll D, Schlosshauer B. Functional characterization and comparison of the outer blood-retina barrier and the blood-brain barrier. Invest Ophthalmol Vis Sci. 2005;46:1047-1053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 104] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 198. | Aukunuru JV, Sunkara G, Bandi N, Thoreson WB, Kompella UB. Expression of multidrug resistance-associated protein (MRP) in human retinal pigment epithelial cells and its interaction with BAPSG, a novel aldose reductase inhibitor. Pharm Res. 2001;18:565-572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 199. | Duvvuri S, Majumdar S, Mitra AK. Drug delivery to the retina: challenges and opportunities. Expert Opin Biol Ther. 2003;3:45-56. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 144] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 200. | Kowluru RA, Chan PS. Oxidative stress and diabetic retinopathy. Exp Diabetes Res. 2007;2007:43603. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 298] [Cited by in RCA: 415] [Article Influence: 24.4] [Reference Citation Analysis (0)] |
| 201. | Limtrakul P, Khantamat O, Pintha K. Inhibition of P-glycoprotein function and expression by kaempferol and quercetin. J Chemother. 2005;17:86-95. [PubMed] |
| 202. | Takumi H, Nakamura H, Simizu T, Harada R, Kometani T, Nadamoto T, Mukai R, Murota K, Kawai Y, Terao J. Bioavailability of orally administered water-dispersible hesperetin and its effect on peripheral vasodilatation in human subjects: implication of endothelial functions of plasma conjugated metabolites. Food Funct. 2012;3:389-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 63] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 203. | Cermak R, Landgraf S, Wolffram S. The bioavailability of quercetin in pigs depends on the glycoside moiety and on dietary factors. J Nutr. 2003;133:2802-2807. [PubMed] |
| 204. | Erlund I, Kosonen T, Alfthan G, Mäenpää J, Perttunen K, Kenraali J, Parantainen J, Aro A. Pharmacokinetics of quercetin from quercetin aglycone and rutin in healthy volunteers. Eur J Clin Pharmacol. 2000;56:545-553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 361] [Cited by in RCA: 349] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 205. | Li X, Choi JS. Effects of quercetin on the pharmacokinetics of Etoposide after oral or intravenous administration of etoposide in rats. Anticancer Res. 2009;29:1411-1415. [PubMed] |
| 206. | Quintieri L, Bortolozzo S, Stragliotto S, Moro S, Pavanetto M, Nassi A, Palatini P, Floreani M. Flavonoids diosmetin and hesperetin are potent inhibitors of cytochrome P450 2C9-mediated drug metabolism in vitro. Drug Metab Pharmacokinet. 2010;25:466-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 28] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 207. | Breinholt VM, Offord EA, Brouwer C, Nielsen SE, Brøsen K, Friedberg T. In vitro investigation of cytochrome P450-mediated metabolism of dietary flavonoids. Food Chem Toxicol. 2002;40:609-616. [PubMed] |
| 208. | Ueng YF, Shyu CC, Lin YL, Park SS, Liao JF, Chen CF. Effects of baicalein and wogonin on drug-metabolizing enzymes in C57BL/6J mice. Life Sci. 2000;67:2189-2200. [PubMed] |
| 209. | Rao VR, Prescott E, Shelke NB, Trivedi R, Thomas P, Struble C, Gadek T, O’Neill CA, Kompella UB. Delivery of SAR 1118 to the retina via ophthalmic drops and its effectiveness in a rat streptozotocin (STZ) model of diabetic retinopathy (DR). Invest Ophthalmol Vis Sci. 2010;51:5198-5204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 210. | Sigurdsson HH, Konráethsdóttir F, Loftsson T, Stefánsson E. Topical and systemic absorption in delivery of dexamethasone to the anterior and posterior segments of the eye. Acta Ophthalmol Scand. 2007;85:598-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 81] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 211. | Holló G, Whitson JT, Faulkner R, McCue B, Curtis M, Wieland H, Chastain J, Sanders M, DeSantis L, Przydryga J. Concentrations of betaxolol in ocular tissues of patients with glaucoma and normal monkeys after 1 month of topical ocular administration. Invest Ophthalmol Vis Sci. 2006;47:235-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 30] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 212. | Earla R, Boddu SH, Cholkar K, Hariharan S, Jwala J, Mitra AK. Development and validation of a fast and sensitive bioanalytical method for the quantitative determination of glucocorticoids--quantitative measurement of dexamethasone in rabbit ocular matrices by liquid chromatography tandem mass spectrometry. J Pharm Biomed Anal. 2010;52:525-533. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 213. | Loftsson T, Hreinsdóttir D, Stefánsson E. Cyclodextrin microparticles for drug delivery to the posterior segment of the eye: aqueous dexamethasone eye drops. J Pharm Pharmacol. 2007;59:629-635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 84] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 214. | Amado NG, Fonseca BF, Cerqueira DM, Neto VM, Abreu JG. Flavonoids: potential Wnt/beta-catenin signaling modulators in cancer. Life Sci. 2011;89:545-554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 80] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 215. | Sultana Y, Jain R, Aqil M, Ali A. Review of ocular drug delivery. Curr Drug Deliv. 2006;3:207-217. [PubMed] |
| 216. | Vandervoort J, Ludwig A. Ocular drug delivery: nanomedicine applications. Nanomedicine (Lond). 2007;2:11-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 50] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 217. | Bourges JL, Gautier SE, Delie F, Bejjani RA, Jeanny JC, Gurny R, BenEzra D, Behar-Cohen FF. Ocular drug delivery targeting the retina and retinal pigment epithelium using polylactide nanoparticles. Invest Ophthalmol Vis Sci. 2003;44:3562-3569. [PubMed] |
| 218. | Misra GP, Singh RS, Aleman TS, Jacobson SG, Gardner TW, Lowe TL. Subconjunctivally implantable hydrogels with degradable and thermoresponsive properties for sustained release of insulin to the retina. Biomaterials. 2009;30:6541-6547. [PubMed] |