Published online Nov 12, 2013. doi: 10.5318/wjo.v3.i4.32
Revised: November 5, 2013
Accepted: November 10, 2013
Published online: November 12, 2013
Neurohormones melatonin and its analogues are present with an important physiological and pharmacological ability to reduce intraocular pressure (IOP); thus, they are suitable for the treatment of ocular hypertension often associated with glaucoma. It is demonstrated that two of its analogues, 5-MCA-NAT and IIK7, are more effective than melatonin to reduce IOP for a longer period of time. The research for the discovery of better compounds resulted in the development of newer and improved analogues compared to 5-MCA-NAT and IIK7. Furthermore, already commercially available drugs currently used as treatment for other pathologies, presenting a resemblance to the melatonin structure, are being tested as potential glaucoma drugs. In this sense, agomelatine, which is already used as an anti-depressant medicine, is recognized as a worthy candidate since it reduces IOP, even under hypertensive conditions. To sum up, the use of melatonin and its analogues as promising anti-glaucomatous substances is of great importance and should be given serious consideration.
Core tip: This mini review depicts the main features of melatonin and derivatives as interesting agents for the treatment of the ocular hypertension associated with glaucoma.
- Citation: Alkozi HA, Pintor J. Melatonin and derivatives as promising tools for glaucoma treatment. World J Ophthalmol 2013; 3(4): 32-37
- URL: https://www.wjgnet.com/2218-6239/full/v3/i4/32.htm
- DOI: https://dx.doi.org/10.5318/wjo.v3.i4.32
There is a general interest in searching for novel compounds capable of reducing intraocular pressure (IOP) as an improved alternative to the existing drugs. IOP can be lowered through the reduction of aqueous humor production or by increasing its outflow through the trabecular meshwork or uveoscleral pathways. The interest for searching for new compounds relies on the fact that most of the existing drugs produce important side effects, hampering the treatment of certain patients. Side effects are a common issue in glaucoma medications. β-blockers such as timolol can cause bradycardia and hypotension and they are unsuitable for patients suffering from cardio vascular problems[1], asthma, obstructive pulmonary disease or corneal dystrophy[2]. Cholinergic agonists such as pilocarpine produce fixed pupils and induce myopia and cataracts[1], whereas prostaglandins (e.g., latanoprost) cause eyelash growth, iris pigmentation[3], muscle and joint pain[2]. Frequently, ocular redness and ocular surface discomfort obligates patients to abandon the treatment.
Several new compounds and approaches are under development in companies’ pipelines or in academic institutions. Among the plethora of substances, the naturally occurring are more attractive as its administration is expected to result in fewer side effects[4]. Among these, the neurohormone melatonin emerges as a promising substance with interesting hypotensive properties[5]. The use of 5-MCA-NAT (a melatonin analogue, see below) when applied to the eye does not produce severe side effects. It does not affect corneal and lens transparency nor cause redness or corneal edema. No negative effects were noticed in general ocular examinations[5]. It is important to bear in mind that most of melatonin intake is not by prescription as it is considered a dietetic supplement. In this case, high dosages and an elevated number of intakes could produce some minor side effects. The Mayo Clinic indicates that the most common side effects are drowsiness, headache and dizziness. Moreover, large doses of melatonin can interfere with some medications, such as anticoagulants, immunosuppressants, diabetes medications and birth control pills.
There are two interesting works describing the melatonin effect and its analogues on reducing IOP. Serle et al[6] demonstrated that a melatonin analogue was able to reduce IOP in glaucomatous monkeys, suggesting these molecules as a possible treatment of ocular hypertension related to glaucoma. Additionally, a group of ophthalmologists started to use melatonin during cataract surgery because it reduces IOP substantially, which is recommendable during phacoemulsification[7].
From these two relevant works, the question arises as why these groups decided to use melatonin and its analogues for clinical purposes and mainly for reducing IOP. The present mini review introduces the reader to the basis of why melatonin is an attractive molecule to reduce IOP and why it should be considered in the future as a respectable alternative to the current ocular hypertension and glaucoma therapies.
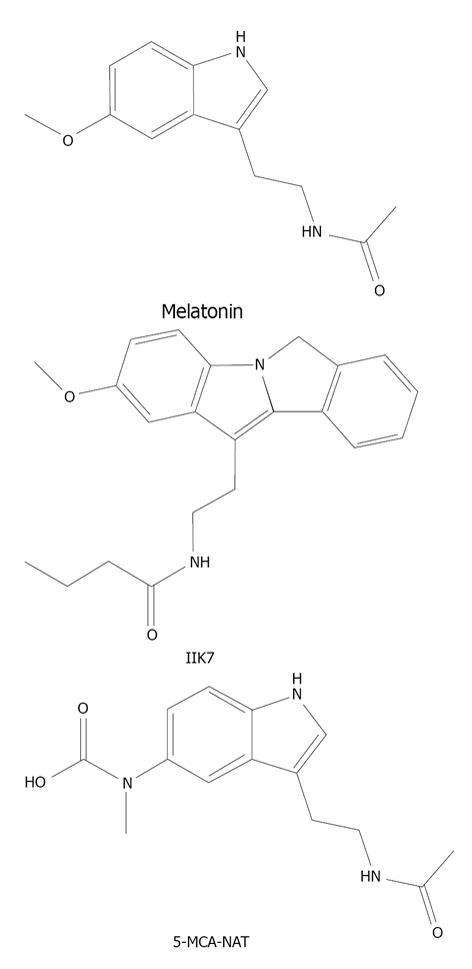
Melatonin is a molecule known by its chemical name N-acetyl-5-methoxytryptamine (Figure 1). It has been traditionally related to a particular area of brain, termed the pineal gland, where it is synthesized in low illumination conditions like during the night[8] and it regulates many day-night processes, called circadian rhythms[9]. It is necessary to emphasize that this substance is also synthesized in other tissues and ocular structures such as the retina, the ciliary body or the lens. This clearly suggests that melatonin can exert some local actions on the tissues where it is synthesized or in surrounding areas. Keeping in mind that melatonin is released by the lens or the ciliary body, its presence in the aqueous humor, modifying the physiology of these structures being bathed in the fluid, can be speculated about. Interestingly, one of the possible physiological processes to be modified is IOP. It is documented that in many animal models there are changes in IOP during the day (high IOP) and night (low IOP). It is possible that both processes are associated considering the circadian pattern of melatonin production. Consequently, we should study what happens if we topically apply melatonin during the day when IOP is high.
When melatonin is topically applied at a single dose of 100 μmol/L in a volume of 10 μL, there is a transient reduction in IOP and values return quickly to initial figures in about 2 h[10]. This effect is similar to that of endogenous melatonin at night which reduces IOP. Despite the acquired hypotensive effect, the rapid return to normal pressure values suggests that either it is necessary to regulate the doses or to look for an alternative compound to produce a more sustained effect[11].
There are several commercially available melatonin analogues depicting similar behavior to melatonin. Two compounds present sharper and long lasting effects on reducing the IOP compared to melatonin. In particular, the compound N-butanoyl-2-2-methoxy-6H-isoindolo ([2,1-a]indol-11-yl) ethanamine (abbreviated as IIK7) has a hypotensive effect that lasts up to 7 hours and the compound 5-methylcarboxyamino-N-acetyltryptamine (also known as 5-MCA-NAT), which can reduce IOP for up to 9 h (Figure 1)[12]. Consequently, 5-MCA-NAT is more interesting since it presents a longer term effect with a significant reduction of IOP for up to 96 h. This remarkable effect has been taken into consideration as we indicate below[11] (Table 1).
| Compound species | IOP reduction | Receptor involved | Ref. |
| Melatonin | |||
| Human | 32.0%± 3.2% | Unknown | [7] |
| Rabbit | 22.0% ± 1.6% | MT2, MT3 | [10,11] |
| Mouse (glaucomatous) | 33.4% ± 2.5% | MT2 | UD |
| 5-MCA-NAT | |||
| Monkey (hypertensive) | 19.2% ± 2.1% | MT3 | [6] |
| Rabbit | 42.5% ± 1.6% | MT3 | [10,11] |
| IIK7 | |||
| Rabbit | 38.5% ± 3.2% | MT2 | [12] |
| INS48848 | |||
| Rabbit | 36.0% ± 2.0% | MT3 | [22] |
| INS48852 | |||
| Rabbit | 33.1% ± 1.4% | MT2 | [22] |
| INS48862 | |||
| Rabbit | 26.0 V ± 1.3 V | MT2 | [22] |
| Agomelatine | |||
| Rabbit | |||
| Normotensive | 20.8% ± 1.4% | MT2 | [25] |
| Hypertensive | 68.8% ± 5.7% | MT2 | [25] |
5-MCA-NAT was tested in normotensive models as well as under hypertensive conditions, including glaucomatous monkeys (Table 1). Interestingly, the effects on the monkeys, a model closer to the human glaucomatous pathology, were extremely interesting. Compared to vehicle treatment, twice daily administration of 5-MCA-NAT for 5 d reduced IOP from 1 to 5 h after the first dose and the IOP-lowering effects were shown to last at least 18 h following administration, based on IOP measurements made after the fourth and eighth doses[6].
One interesting characteristic to take into account was that the ocular hypotensive effect of 5-MCA-NAT was enhanced by repeated dosing. The maximum reduction of IOP was acquired 3 h after each morning dose and was 10% on day 1, 15% on day 3, and 19% on day 5 (control = 100%). No adverse ocular or systemic side effects were observed during the 5 treatment days, suggesting that this compound could be used perfectly as ocular hypertension treatment [6] (Table 1).
IIK7 reduced intraocular pressure by acting through MT2 melatonin receptors, presumably decreasing aqueous humor formation. Its effect is concentration dependent and it can reduce IOP 38.5% ± 3.2% when compared to controls (Table 1). It is important to notice that these experiments have not been performed in glaucomatous monkeys yet but only in rabbits[12].
In summary, it seems that some compounds, such as melatonin, 5-MCA-NAT and IIK7, clearly reduce IOP. But what is the mechanism for this IOP reduction? What receptors activate these substances in order to produce the observed effects?
Melatonin exerts its effect via membrane and nuclear receptors. The protein membrane receptors are better understood and until recently three proteins have been cloned. Two of these membrane receptors, termed MT1 and MT2, are melatonin receptors belonging to the 7-transmembrane G protein-coupled receptor family (GPCR). There have been claims that a third receptor exists, the MT3 melatonin receptor, although it has not been cloned yet. Some authors have identified it as quinone reductase 2 (QR2), demonstrating features of a melatonin receptor in some animal models (for a review see[9]).
MT1, MT2 and the probable MT3 melatonin receptors are present in several ocular structures, according to pharmacological, biochemical and immunological studies[13,14]. This evidence suggests that melatonin plays a role in physiological processes in ocular tissues, such as the modulation of IOP, and it has been documented that MT2 and MT3 are responsible for IOP reduction.
When melatonin, 5-MCA-NAT and IIK7 are applied to normotensive or hypertensive eyes, they produce a dissimilar IOP reduction, depending on the compound under study. The use of selective antagonists for melatonin receptors has allowed identification of the presence of MT2 melatonin receptors in the ciliary body of experimental animals, such as New Zealand white rabbits. This has been confirmed through immunohistochemical studies. In these studies it has been possible to verify the presence of MT2 melatonin receptors on pigmented and non-pigmented ciliary epithelia. Accordingly, the application of melatonin or IIK7, which is a selective MT2 agonist, results in a reduction in the production of the aqueous humor[12] (Figure 2).
5-MCA-NAT has been suggested as an MT3 melatonin receptor agonist that reduces IOP. To date, the location of the receptor is unknown. As there is a controversy with the possible identification of the MT3 receptor which is tentatively identified in some animal models as QR2, some sophisticated experiments were performed to clarify the issue[15-17]. In New Zealand rabbits, the use of a siRNA silencing QR2 (therefore avoiding the expression of this enzyme) did not abolish the hypotensive effect of 5-MCA-NAT, clearly indicating that, in this animal model, MT3≠ QR2, opening the possibility of speculating about the existence of a receptor that needs to be cloned to fully understand its functioning and location[18].
Apart from melatonin and its derivatives, some other compounds, like 5-MCA-NAT, can keep IOP below normal values for up to 5 d. This long-term effect is mediated by the action of melatonin receptors on the expression of genes expressing proteins important for the homeostasis of the aqueous humor.
To date, it has been possible to demonstrate that the 5-MCA-NAT long-term effect is in part the result of the expression inhibition of carbonic anhydrases. This down-regulation means that 24 h after 5-MCA-NAT application there is a reduction in IOP because the amounts of carbonic anhydrases are severely reduced. In particular, when 5-MCA-NAT is applied, carbonic anhydrase 2 is reduced 32% (protein levels), while carbonic anhydrase 12 is reduced 39% (protein levels). This reduction in protein expression mimics the carbonic anhydrase inhibitor action, such as dorzolamide or acetazolamide[19].
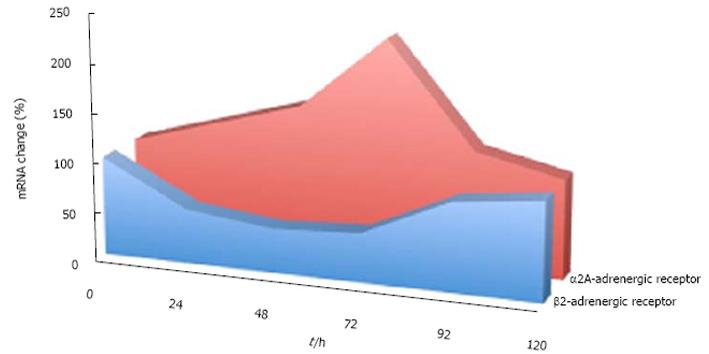
Likewise, the expression of adrenergic receptors is modified by the application of 5-MCA-NAT. Interestingly, this melatonin analogue is able to produce a sequential process consisting of an initial reduction in the β2-adrenoreceptors expression, followed by an increase in α2A-adrenoreceptors[20]. Altogether, these consecutive effects produce a sustained reduction in IOP lasting for at least 96 h[21].
In summary, 5-MCA-NAT, apart from a sharp hypotensive effect, exerts a long term effect, maintaining low IOP for 4 d.
Several aspects need to be studied, taking into account that melatonin and analogues can significantly reduce IOP.
It is clear that it is necessary to research and design new melatonin analogues with more profound and long lasting effects[5]. Inspire Pharmaceuticals Inc. (now absorbed by Merck) has designed several melatonin analogues with interesting hypotensive properties to reduce IOP. In recent studies, melatonin analogues, termed INS48848, INS48852 and INS48862, demonstrated similar behavior to melatonin, 5-MCA-NAT and IIK7[22]. Indeed, these three compounds decreased IOP in a dose-dependent manner similar to melatonin, 5-MCA-NAT and IIK7, confirming their efficiency in decreasing IOP (Table 1). Concerning their selectivity on melatonin receptors, the effects of INS48848 were completely blocked by prazosin, an antagonist of MT3 melatonin receptors, and were potently inhibited by luzindole, a non-selective antagonist of melatonin receptors. However, DH97, a selective MT2 receptor antagonist, had a limited effect against INS48848 and the results obtained from INS48862 and INS48852 were contradictory. Luzindole and prazosin had no significant effects against those two compounds, whereas DH97 blocked them completely. These results strongly suggest that INS48848 could be acting through the MT3 melatonin receptors and that INS48862 and INS48852 could be acting preferentially through MT2 melatonin receptors. In any case, all these compounds are worthy candidates to reduce IOP, especially when it is abnormally elevated[22].
Another alternative to the development of newly synthesized compounds is to search for melatoninergic compounds already used for other medical purposes. Compounds such as ramelteon ((S)-N-[2-(1,6,7,8-tetrahydro-2H-indeno-[5,4-b]furan-8-yl)ethyl]propionamide), also known as Rozerem, used for sleep disorders[23] or agomelatine (N-[2-(7-methoxynaphthalen-1-yl)ethyl]acetamide), known also by the names Valdoxan, Melitor, Thymanax[24], used for the treatment of depression, could be candidates to reduce IOP since their structure is similar to melatonin.
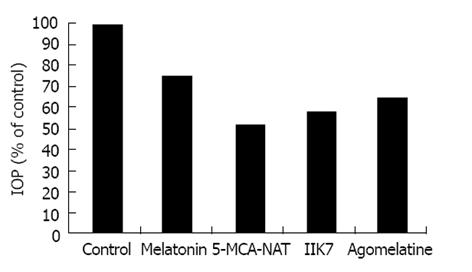
There is a lack of information regarding the use of ramelteon in IOP studies. Agomelatine significantly reduces IOP when topically applied on rabbit eyes. Agomelatine (10 μL, 100 μmol/L) reduced IOP by 20.8% ± 1.4% and its maximal IOP reduction was 180 minutes after the compound application. Interestingly, this compound exhibited an ability to reduce IOP in hypertensive conditions. It is noteworthy to stress that under high IOP the ability of this melatonin analogue to reduce IOP was 68.8% ± 5.7% (Figure 3, Table 1)[25].
There is a clear advantage in using compounds already commercialised for other conditions as the timeline for testing and clinical trials is significantly reduced.
It is necessary to perform an exhaustive study on the role of melatonin and its analogues in the different ocular structures since it is very probable that this knowledge will contribute to the discovery of more effective treatments for pathologies like glaucoma, corneal wound healing, cataracts or retinal pathology[26].
Taking into account the importance of the role of melatonin and its analogues in hypertension, often associated with glaucoma, it is quite evident that these compounds should be used as treatment to reduce IOP. Melatonin or agomelatine can simply and rapidly reduce IOP, although further research is required to prove that they can be safely used as treatment for ocular hypertension.
Most of the presented data resulted from experiments assaying melatonin or its analogues on animal models. We still have a long way to go to test these compounds on human beings. Nevertheless, there are a lot of positive points regarding the efficacy of certain melatoninergic compounds. For instance, melatonin itself is able to reduce IOP in normotensive humans, as previously described[7]. These authors reported an approximate 30% reduction in IOP during cataract surgery compared to the initial patient’s pressures. This is quite interesting because the IOP reduction has been obtained in normotensive patients and it could be even more substantial in hypertensive (glaucomatous) patients. Several experiments in animal models demonstrated that melatonin and analogues are able to reduce IOP equally in normotensive and hypertensive animals, being more effective in hypertensive than in normotensive animals (Table 1). Also, experiments performed with 5-MCA-NAT on hypertensive monkeys, a step before human clinical trials, have proved that this melatonin analogue reduced IOP.
In conclusion, agomelatine is the compound that we strongly believe should be tested in glaucomatous patients for its ability to reduce IOP. Agomelatine is already used as a depression treatment drug under the commercial name Valdoxan[24]. Since many of the pre-clinical tests have already been completed, we should not be surprised if agomelatine clinical trials start and it becomes the first melatoninergic compound to join the group of glaucoma treatment substances.
The Harvard Alumni Association is a fellowship holder of Saudi Arabia Government.
P- Reviewers: Clement CI, Hong YJ, Nowak MS, Zanon-Moreno V S- Editor: Song XX L- Editor: Roemmele A
E- Editor: Lu YJ
| 1. | Hoyng PF, van Beek LM. Pharmacological therapy for glaucoma: a review. Drugs. 2000;59:411-434. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 114] [Cited by in F6Publishing: 124] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 2. | Higginbotham EJ, Schuman JS, Goldberg I, Gross RL, VanDenburgh AM, Chen K, Whitcup SM. One-year, randomized study comparing bimatoprost and timolol in glaucoma and ocular hypertension. Arch Ophthalmol. 2002;120:1286-1293. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 139] [Cited by in F6Publishing: 150] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 3. | Johnstone MA. Hypertrichosis and increased pigmentation of eyelashes and adjacent hair in the region of the ipsilateral eyelids of patients treated with unilateral topical latanoprost. Am J Ophthalmol. 1997;124:544-547. [PubMed] [Cited in This Article: ] |
| 4. | Mediero A, Alarma-Estrany P, Pintor J. New treatments for ocular hypertension. Auton Neurosci. 2009;147:14-19. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 19] [Cited by in F6Publishing: 19] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 5. | Crooke A, Colligris B, Pintor J. Update in glaucoma medicinal chemistry: emerging evidence for the importance of melatonin analogues. Curr Med Chem. 2012;19:3508-3522. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 6. | Serle JB, Wang RF, Peterson WM, Plourde R, Yerxa BR. Effect of 5-MCA-NAT, a putative melatonin MT3 receptor agonist, on intraocular pressure in glaucomatous monkey eyes. J Glaucoma. 2004;13:385-388. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 72] [Cited by in F6Publishing: 72] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 7. | Ismail SA, Mowafi HA. Melatonin provides anxiolysis, enhances analgesia, decreases intraocular pressure, and promotes better operating conditions during cataract surgery under topical anesthesia. Anesth Analg. 2009;108:1146-1151. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 107] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 8. | Ackermann K, Stehle JH. Melatonin synthesis in the human pineal gland: advantages, implications, and difficulties. Chronobiol Int. 2006;23:369-379. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 42] [Cited by in F6Publishing: 41] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 9. | Alarma-Estrany P, Pintor J. Melatonin receptors in the eye: location, second messengers and role in ocular physiology. Pharmacol Ther. 2007;113:507-522. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 74] [Cited by in F6Publishing: 79] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 10. | Pintor J, Martin L, Pelaez T, Hoyle CH, Peral A. Involvement of melatonin MT(3) receptors in the regulation of intraocular pressure in rabbits. Eur J Pharmacol. 2001;416:251-254. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 94] [Cited by in F6Publishing: 97] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 11. | Pintor J, Peláez T, Hoyle CH, Peral A. Ocular hypotensive effects of melatonin receptor agonists in the rabbit: further evidence for an MT3 receptor. Br J Pharmacol. 2003;138:831-836. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 86] [Cited by in F6Publishing: 85] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 12. | Alarma-Estrany P, Crooke A, Mediero A, Peláez T, Pintor J. Sympathetic nervous system modulates the ocular hypotensive action of MT2-melatonin receptors in normotensive rabbits. J Pineal Res. 2008;45:468-475. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 29] [Cited by in F6Publishing: 29] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 13. | Dubocovich ML. Pharmacology and function of melatonin receptors. FASEB J. 1988;2:2765-2773. [PubMed] [Cited in This Article: ] |
| 14. | Dubocovich ML, Markowska M. Functional MT1 and MT2 melatonin receptors in mammals. Endocrine. 2005;27:101-110. [PubMed] [DOI] [Cited in This Article: ] [Cited by in F6Publishing: 2] [Reference Citation Analysis (0)] |
| 15. | Boutin JA, Saunier C, Guenin SP, Berger S, Moulharat N, Gohier A, Delagrange P, Cogé F, Ferry G. Studies of the melatonin binding site location onto quinone reductase 2 by directed mutagenesis. Arch Biochem Biophys. 2008;477:12-19. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 13] [Cited by in F6Publishing: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 16. | Mailliet F, Ferry G, Vella F, Berger S, Cogé F, Chomarat P, Mallet C, Guénin SP, Guillaumet G, Viaud-Massuard MC. Characterization of the melatoninergic MT3 binding site on the NRH: quinone oxidoreductase 2 enzyme. Biochem Pharmacol. 2005;71:74-88. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 92] [Cited by in F6Publishing: 95] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Tan DX, Manchester LC, Terron MP, Flores LJ, Tamura H, Reiter RJ. Melatonin as a naturally occurring co-substrate of quinone reductase-2, the putative MT3 melatonin membrane receptor: hypothesis and significance. J Pineal Res. 2007;43:317-320. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 101] [Cited by in F6Publishing: 100] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 18. | Alarma-Estrany P, Crooke A, Pintor J. 5-MCA-NAT does not act through NQO2 to reduce intraocular pressure in New-Zealand white rabbit. J Pineal Res. 2009;47:201-209. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 24] [Cited by in F6Publishing: 24] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 19. | Crooke A, Huete-Toral F, Martínez-Águila A, Martín-Gil A, Pintor J. Involvement of carbonic anhydrases in the ocular hypotensive effect of melatonin analogue 5-MCA-NAT. J Pineal Res. 2012;52:265-270. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 14] [Cited by in F6Publishing: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 20. | Crooke A, Huete-Toral F, Martínez-Águila A, Alarma-Estrany P, Pintor J. Regulation of ocular adrenoceptor genes expression by 5-MCA-NAT: implications for glaucoma treatment. Pharmacogenet Genomics. 2011;21:587-589. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 8] [Cited by in F6Publishing: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 21. | Crooke A, Huete-Toral F, Martínez-Águila A, Martín-Gil A, Pintor J. Melatonin and its analog 5-methoxycarbonylamino-N-acetyltryptamine potentiate adrenergic receptor-mediated ocular hypotensive effects in rabbits: significance for combination therapy in glaucoma. J Pharmacol Exp Ther. 2013;346:138-145. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 23] [Cited by in F6Publishing: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 22. | Alarma-Estrany P, Guzman-Aranguez A, Huete F, Peral A, Plourde R, Pelaez T, Yerxa B, Pintor J. Design of novel melatonin analogs for the reduction of intraocular pressure in normotensive rabbits. J Pharmacol Exp Ther. 2011;337:703-709. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 25] [Cited by in F6Publishing: 25] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 23. | Laustsen G, Andersen M. Ramelteon (rozerem) a novel approach for insomnia treatment. Nurse Pract. 2006;31:52-55. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 6] [Cited by in F6Publishing: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 24. | Millan MJ, Brocco M, Gobert A, Dekeyne A. Anxiolytic properties of agomelatine, an antidepressant with melatoninergic and serotonergic properties: role of 5-HT2C receptor blockade. Psychopharmacology (Berl). 2005;177:448-458. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 190] [Cited by in F6Publishing: 194] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 25. | Martínez-Águila A, Fonseca B, Bergua A, Pintor J. Melatonin analogue agomelatine reduces rabbit’s intraocular pressure in normotensive and hypertensive conditions. Eur J Pharmacol. 2013;701:213-217. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 33] [Cited by in F6Publishing: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 26. | Crooke A, Huete-Toral F, Martínez-Águila A, Colligris B, Pintor J. Ocular disorders and the utility of animal models in the discovery of melatoninergic drugs with therapeutic potential. Expert Opin Drug Discov. 2012;7:989-1001. [PubMed] [DOI] [Cited in This Article: ] [Cited by in Crossref: 11] [Cited by in F6Publishing: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |